Exhibit 99.1

Enabling oral drug delivery to improve patient compliance March 12, 2018 Corporate Presentation Exhibit 99.1

Forward - Looking Statements This presentation contains forward - looking statements about Lipocine Inc. (the “Company”). These forward - looking statements are made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. These forward - looking statement s relate to the Company’s product candidates, the expected timing of the FDA review process related to our resubmitted NDA for TLANDO™, clini cal and regulatory processes and objectives, potential benefits of the Company’s product candidates, intellectual property and relate d m atters, all of which involve known and unknown risks and uncertainties. Actual results may differ materially from the forward - looking statemen ts discussed in this presentation . Accordingly, the Company cautions investors not to place undue reliance on the forward - looking statements contained in, or made in connection with, this presentation . Several factors may affect the initiation and completion of clinical trials, the potential advantages of the Company’s product candidates and the Company’s capital needs. Among other things, the projected commencement and completion of the Company’s clinical trials may be affected by difficulties or delays. We may encounter delays or other issues in the FDA appro val process, including that the FDA may determine there are deficiencies in our resubmitted NDA. We are also subject to risks related to t he possibility of an advisory committee meeting related to TLANDO™. In addition, the Company’s results may be affected by its ability to manage it s financial resources, difficulties or delays in developing manufacturing processes for its product candidates, preclinical and toxicolog y t esting and regulatory developments. Delays in clinical programs, whether caused by competitive developments, adverse events, patient en rol lment rates, regulatory issues or other factors, could adversely affect the Company’s financial position and prospects. Prior clin ica l trial program designs and results are not necessarily predictive of future clinical trial designs or results. If the Company’s product can did ates do not meet safety or efficacy endpoints in clinical evaluations, they will not receive regulatory approval and the Company will not be a ble to market them. The Company may not be able to enter into any strategic partnership agreements. The Company’s commercial success depends on i ts ability to manufacture, market and sell products without infringing the proprietary rights of third parties. Operating expense and ca sh flow projections involve a high degree of uncertainty, including variances in future spending rates due to changes in corporate pr ior ities, the timing and outcomes of clinical trials, competitive developments and the impact on expenditures and available capital from licensing an d strategic collaboration opportunities. If the Company is unable to raise additional capital when required or on acceptable terms, it m ay have to significantly delay, scale back or discontinue one or more of its drug development or discovery research programs. The Compa ny is at an early stage of development and may not ever have any products that generate significant revenue. The forward - looking statements contained in this presentation are further qualified by the detailed discussion of risks and uncertainties set forth in the Company’s a nnu al report on Form 10 - K and other periodic reports filed by the Company with the Securities and Exchange Commission, all of which can be obtai ned on the Company’s website at www.lipocine.com or on the SEC website at www.sec.gov . The forward - looking statements contained in this document represent the Company’s estimates and assumptions only as of the date of this presentation and the Company undertakes no duty or obligation to update or revise publicly any forward - looking statements contained in this presentation as a result of new information, future events or changes in the Company’s expectations. 2

Men's Health Franchise Testosterone Replacement Therapy (“TRT”) Women's Health Preterm Birth (“PTB”) Proprietary Drug Delivery Platform Significant Unmet Need In Both Therapeutic Areas 3 Unique Specialty Pharmaceutical Company Advanced Pipeline
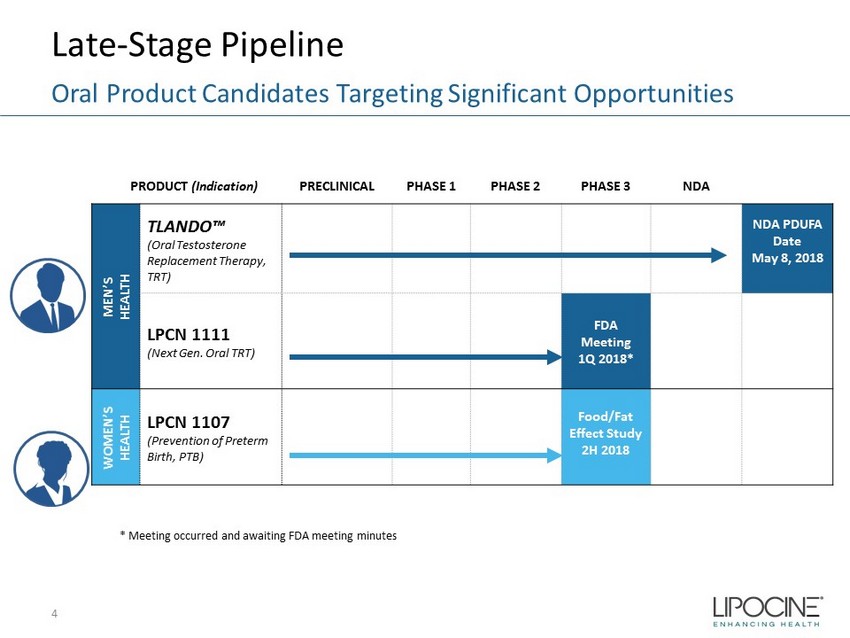
Late - Stage Pipeline 4 Oral Product Candidates Targeting Significant Opportunities PRODUCT (Indication) PRECLINICAL PHASE 1 PHASE 2 PHASE 3 NDA MEN’S HEALTH TLANDO™ (Oral Testosterone Replacement Therapy, TRT) NDA PDUFA Date May 8, 2018 LPCN 1111 (Next Gen. Oral TRT) FDA Meeting 1Q 2018* WOMEN’S HEALTH LPCN 1107 (Prevention of Preterm Birth, PTB) Food/Fat Effect Study 2H 2018 * Meeting occurred and awaiting FDA meeting minutes

Lipocine is Positioned for Success Because…… 5 Significant Advantages over Existing Approved Products LPCN 1107 • Poised to be First oral to the market • Potentially better PTB results • Orphan drug designation – Major contribution to patient care TRT Franchise TLANDO™ • Patient preferred first oral option • Positive head to head clinical results vs the $ market leader • No additional doctor visits as fixed dose LPCN 1111 (Next generation potential once - daily oral T) • Potential for sustained and enhanced revenues with QD dosing • Positive top - line Phase 2b study results Significant Technical and Commercial Expertise on the team Prevention of Preterm Birth

6

2017 2016 2014 2015 350 375 400 425 450 475 500 525 550 575 600 625 650 Sep 14 Nov 14 Jan 15 Mar 15 May 15 Jul 15 Sep 15 Nov 15 Jan 16 Mar 16 May 16 Jul 16 Sep 16 Nov 16 Jan 17 Mar 17 May 17 Jul 17 Sept 17 Nov 17 Monthly TRx (000s) FDA Label Guidance Monthly TRT TRx Trend 7 TRT Market is Approaching Heights Experienced in 2013 and Growing 2017 Gross Annual Sales - $1.7B Annual estimates of 7.0 million TRx Source: IMS database TRx = Total prescriptions
Issues with Current Non - oral TRT Options • Black Box Warning – Secondary exposure to testosterone – Pulmonary oil micro embolism (POME) and anaphylaxis shock • Inconvenient application or painful injection • Poor persistence reflects need for oral – Average days on therapy is 100 days • More than 50% of patients need dosage adjustment – Burdensome for patients due to multiple doctor visits Potential Barrier To Newly Diagnosed and Existing Patients 8

TLANDO™: Advisory Committee Meeting BRUDAC Results and Path Forward Voting Question: Is the overall benefit/risk profile of TLANDO™ acceptable to support approval as a testosterone replacement therapy? Results: 6 Yes 13 No Path Forward 9 • Voting rationale: – All 13 “No” votes cited lack of Ambulatory Blood Pressure Monitoring (“ABPM”) – Confirmation of Serum T measurement reliability (ex vivo conversion assessment) • Initiating ABPM study ahead of PDUFA date – Protocol submitted under IND for review and comments • Initiating Phlebotomy study ahead of PDUFA date to confirm no ex - vivo conversion of TU to T – Protocol submitted under IND for review and comments • Initiated legal and government affairs activities in support of TLANDO approval

Comparative Oral TU Product Profiles* TLANDO™ vs Jatenzo ™ ▪ Clinically meaningful BP increase in ABPM study ▪ Major Cardiac Adverse Events Reported in multiple studies ▪ All lipid parameters adversely changed compared with active control ▪ Mean dose: 325 mg BID (40% on 396 mg BID) ▪ 12% reported GI adverse events ▪ Atypical plasma based T measurements ▪ No ABPM data, but large clinic blood pressure (“BP”) database with no BP increase ▪ No Major Cardiac Adverse Events over 12 studies ▪ Mean dose: 225 mg BID ▪ GI effects comparable to topical gel ▪ Standard serum based T measurements 10 TLANDO™ Jatenzo ™ * From Jatenzo briefing document for Jan 9 th BRUDAC meeting

TRT and Blood Pressure Overview ▪ Long - term study underway to determine definitive link between TRT use and cardiovascular (CV) outcome, if any ▪ Blood pressure effects are surrogate marker of potential CV risk ▪ No known blood pressure effects with any approved TRT product ▪ No ABPM study requirement for any prior approved TRT product ▪ No request to perform ABPM study for TLANDO™ in any prior FDA interaction/communication (i.e., CRL, post - action meeting, DV protocol review) ▪ Clinic BP measurement is standard methodology for non - hypertensives trials and TRT products 11 * From Jatenzo briefing document for Jan 9 th BRUDAC meeting
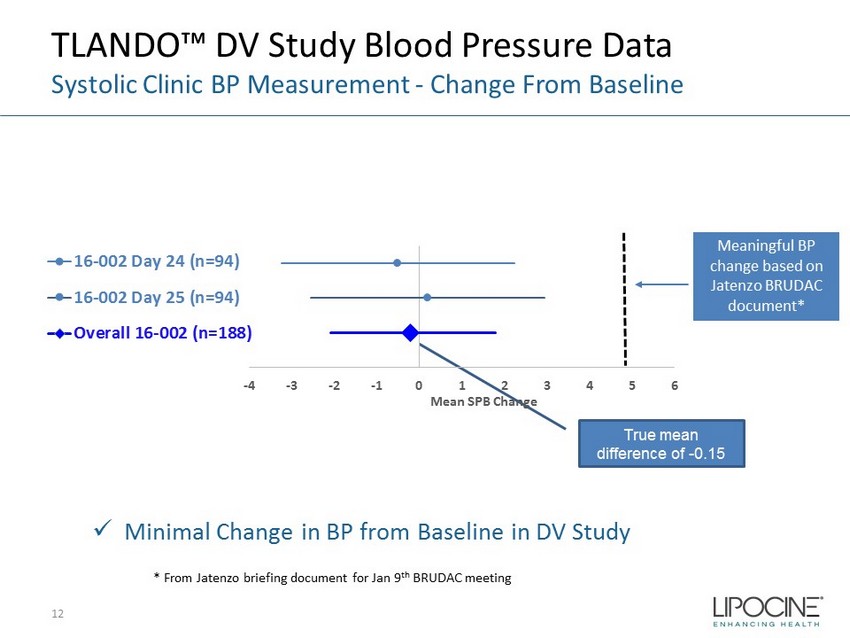
TLANDO™ DV Study Blood Pressure Data Systolic Clinic BP Measurement - Change From Baseline True mean difference of - 0.15 -4 -3 -2 -1 0 1 2 3 4 5 6 Mean SPB Change 16-002 Day 24 (n=94) 16-002 Day 25 (n=94) Overall 16-002 (n=188) Meaningful BP change based on Jatenzo BRUDAC document* x Minimal Change in BP from Baseline in DV Study * From Jatenzo briefing document for Jan 9 th BRUDAC meeting 12

-4 -3 -2 -1 0 1 2 3 4 5 6 Mean SPB Change (mmHg) 13-001 TLANDO Week 3 (n=193) 13-001 TLANDO Week 7 (n=182) 13-001 TLANDO Week 13 (n=157) 13-001 TLANDO Week 26 (n=144) 13-001 TLANDO Week 39 (n=138) 13-001 TLANDO Week 52 (n=130) 13-001 TLANDO Overall (n=944) TLANDO™ SOAR Study Blood Pressure Data Systolic Clinic BP Measurement - Change From Baseline True mean difference of - 0.62 Meaningful BP change based on Jatenzo BRUDAC document x Minimal Change in BP from baseline at each visit throughout the SOAR study 13
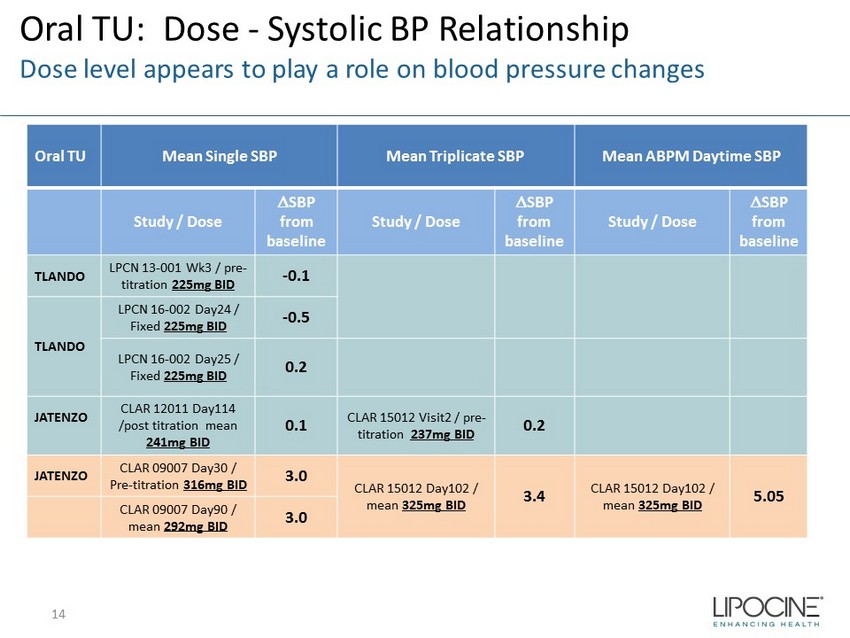
Oral TU: Dose - Systolic BP Relationship Dose level appears to play a role on blood pressure changes Oral TU Mean Single SBP Mean Triplicate SBP Mean ABPM Daytime SBP Study / Dose D SBP from baseline Study / Dose D SBP from baseline Study / Dose D SBP from baseline TLANDO LPCN 13 - 001 Wk3 / pre - titration 225mg BID - 0.1 TLANDO LPCN 16 - 002 Day24 / Fixed 225mg BID - 0.5 LPCN 16 - 002 Day25 / Fixed 225mg BID 0.2 JATENZO CLAR 12011 Day114 /post titration mean 241mg BID 0.1 CLAR 15012 Visit2 / pre - titration 237mg BID 0.2 JATENZO CLAR 09007 Day30 / Pre - titration 316mg BID 3.0 CLAR 15012 Day102 / mean 325mg BID 3.4 CLAR 15012 Day102 / mean 325mg BID 5.05 CLAR 09007 Day90 / mean 292mg BID 3.0 14

Oral TU Blood Pressure Effects Summary ▪ No significant increase in clinic measured BP with TLANDO™ in multiple visits across multiple studies at the indicated dosing regimen of 225 mg BID ▪ Dosing regimen appears to play a role on any blood pressure effects ▪ Clinic BP and ABPM measurements are similar* ▪ Multiple visit clinic measurements more likely to identify any BP trend 15 *From Jatenzo briefing document for Jan 9 th BRUDAC meeting

16 LPCN 1111 QD TLANDO

LPCN 1111: Next - Generation Oral TRT • Novel prodrug of testosterone for oral delivery • Once - daily potential expected to sustain and improve market share of oral T franchise • Once - daily feasibility established in Phase 2a and 2b clinical trials – Single - daily oral dose provides T levels in eugonadal range • Development status Next steps: – Preclinical toxicity study complete, data analysis on - going – FDA meeting in 1Q 2018 to discuss results of preclinical toxicity study and the Phase 3 study design 17 Potential Once - Daily Dosing

18
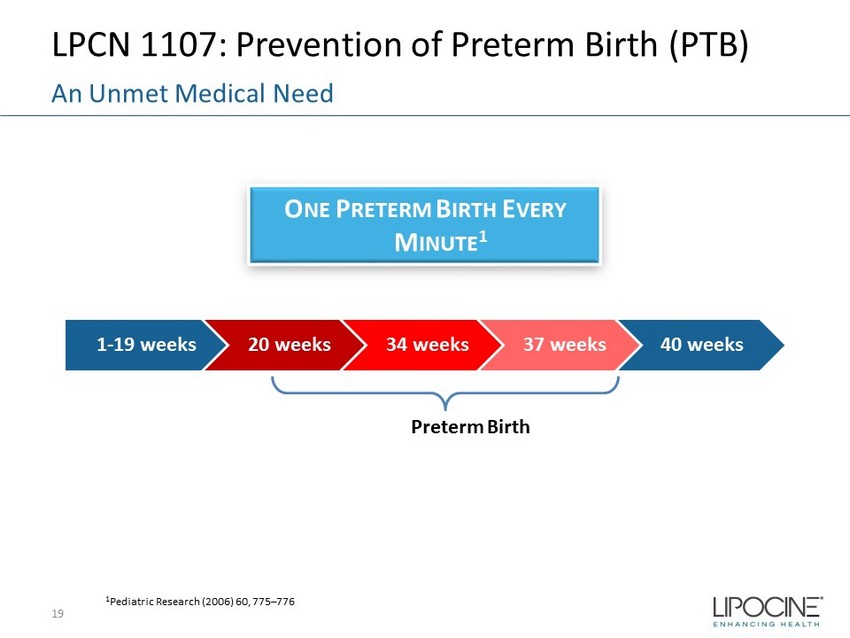
LPCN 1107: Prevention of Preterm Birth (PTB) 19 An Unmet Medical Need 1 Pediatric Research (2006) 60, 775 – 776 Preterm Birth 1 - 19 weeks 20 weeks 34 weeks 37 weeks 40 weeks O NE P RETERM B IRTH E VERY M INUTE 1
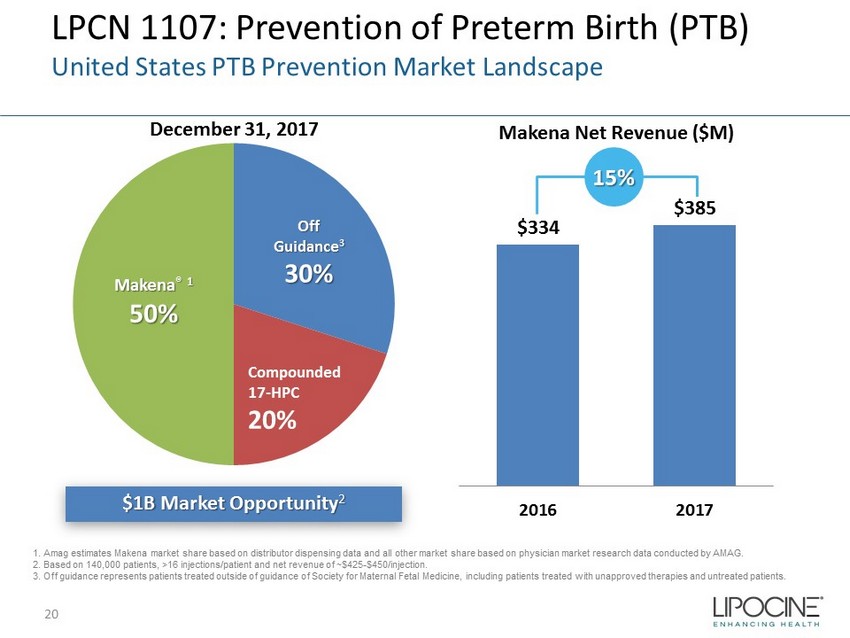
LPCN 1107: Prevention of Preterm Birth (PTB) United States PTB Prevention Market Landscape $1B Market Opportunity 2 December 31, 2017 Makena Net Revenue ($M) Makena ® 1 50% Off Guidance 3 30% Compounded 17 - HPC 20% $334 $385 2016 2017 15% 1. Amag estimates Makena market share based on distributor dispensing data and all other market share based on physician market resea rc h data conducted by AMAG. 2. Based on 140,000 patients, >16 injections/patient and net revenue of ~$425 - $450/injection. 3. Off guidance represents patients treated outside of guidance of Society for Maternal Fetal Medicine, including patients tr eat ed with unapproved therapies and untreated patients. 20

LPCN 1107: First Oral PTB Candidate 21 Characteristics Of The Only Approved Product for PTB IM HPC, Makena ® : Current preterm birth standard of care ▪ 20 - 25% patients below reported better efficacy HPC level threshold ▪ Total of 18 - 22 injections – Viscous oily weekly injection – Injection takes up to 1 min – Weekly visit to/by health care provider – ~35% of patients experienced injection site pain during clinical trial – ~17% of patients reported site swelling - much greater than placebo during clinical trial Makena 21 gauge needle
LPCN 1107 - Oral HPC ▪ Potential for superior efficacy with Phase 3 target dose ▪ No patient discomfort upon administration ▪ Steady state achieved in 7 days ▪ Orphan drug designation – Major contribution to patient care LPCN 1107: First Oral PTB Candidate 22 Addresses Unmet Need

LPCN 1107: Advancing to Phase 3 Readiness Phase 3 Special Protocol Assessment – Progress ▪ Concurrence with FDA to date: – Study Design Elements • Single Phase 3 study • Open label, active comparator, two parallel arms (1:1 randomization) • General inclusion and exclusion criteria and treatment duration • LPCN 1107 dose of 800 mg BID – Endpoints and Analysis • Primary endpoint of proportion of PTB < 37 weeks • Non - inferiority margin of 7% • Secondary endpoint: Neonatal mortality and morbidity composite index • Interim analysis with ability to resize the study – Study size: 500 to 1000 subjects per arm ▪ Open Items – Data from food effect study to inform dosing instructions – Align on approach to fulfill infant follow up data requirement ▪ Next Steps – Continue Interactions with FDA on Phase 3 protocol via Special Protocol Assessment – Conduct Food/Fat Effect Study in 2H 2018 in preparation of Phase 3 study 23

Upcoming Milestones 24 Driving Value in 2018 Event Expected Timing TLANDO™ Initiate ABPM study Initiate Phlebotomy study PDUFA goal date 1H 2018 1H 2018 May 8, 2018 LPCN 1111 FDA meeting to discuss Phase 3 clinical trial 1Q 2018* LPCN 1107 Conduct Food/Fat Effect clinical study 2H 2018 * Meeting occurred and awaiting FDA meeting minutes
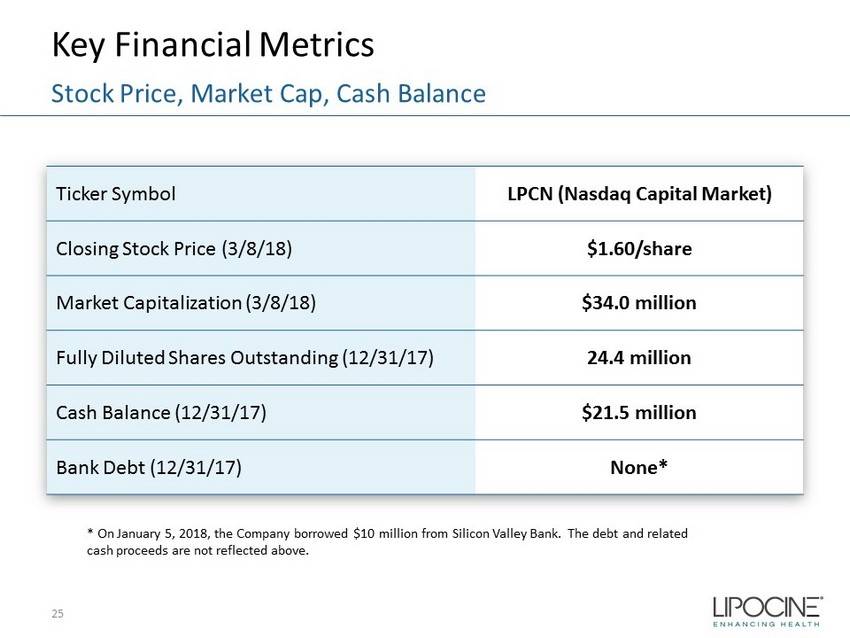
Key Financial Metrics 25 Stock Price, Market Cap, Cash Balance Ticker Symbol LPCN (Nasdaq Capital Market) Closing Stock Price (3/8/18) $1.60/share Market Capitalization (3/8/18) $34.0 million Fully Diluted Shares Outstanding (12/31/17) 24.4 million Cash Balance (12/31/17) $21.5 million Bank Debt (12/31/17) None* * On January 5, 2018, the Company borrowed $10 million from Silicon Valley Bank. The debt and related cash proceeds are not reflected above.
MEN’S HEALTH Testosterone Replacement Therapy (“TRT”) Franchise • TLANDO™ : Potential first oral TRT option - Differentiated product targeting ~$2.0 billion established US TRT market - Poised to meet and exceed unmet need • LPCN 1111 : Next generation potential once - daily oral TRT option - Positive top - line Phase 2b study results WOMEN’S HEALTH LPCN 1107: Orphan designated oral alternative for the prevention of preterm birth • EOP2 meeting completed • Phase 3 protocol submitted to FDA via Special Protocol Assessment Lipocine Investment Highlights 26 Innovative Technology with Advanced Products Proven Proprietary Technology Platform

Appendix 27

28 28

TLANDO™: Potential First Oral Option Profile Demonstrated Clinically with Target Label Regimen ▪ Met primary endpoint - 80% response rate in “worst - case analysis” vs. FDA requirement of 75% ▪ Met key secondary endpoint - No eligible subjects with T levels >2500 ng/ dL ▪ Other secondary endpoints generally consistent with approved products Efficacy ✔ ▪ 525 unique hypogonadal men exposure ▪ Well tolerated in 52 week exposure - AE profile comparable to active control, including GI - No cardiac, hepatic or drug related SAEs ▪ No apparent correlation of the observed Cmax excursions - ADRs - AEs - Meaningful changes in critical lab parameters Safety ▪ Preferred oral option - No risk of accidental T transference - Non - invasive - Less cumbersome - Less burdensome - Simpler to prescribe - Fewer doctor visits - Easier for patients to properly use Clear Benefits 29

TLANDO™ BRUDAC Briefing Document FDA Blood Pressure Conclusion • “The 52 - week blood pressure data are more reassuring, but are limited by the non - duplicative (single morning) cuff pressure readings, open - label trial design and 38% dropout rate in the Tlando arm. Based on these considerations, FDA cardiologists concluded that we cannot definitively exclude a blood pressure effect with Tlando and recommend that the Applicant perform an ambulatory blood pressure monitoring study to further assess the effects of Tlando on blood pressure and heart rate.” 30
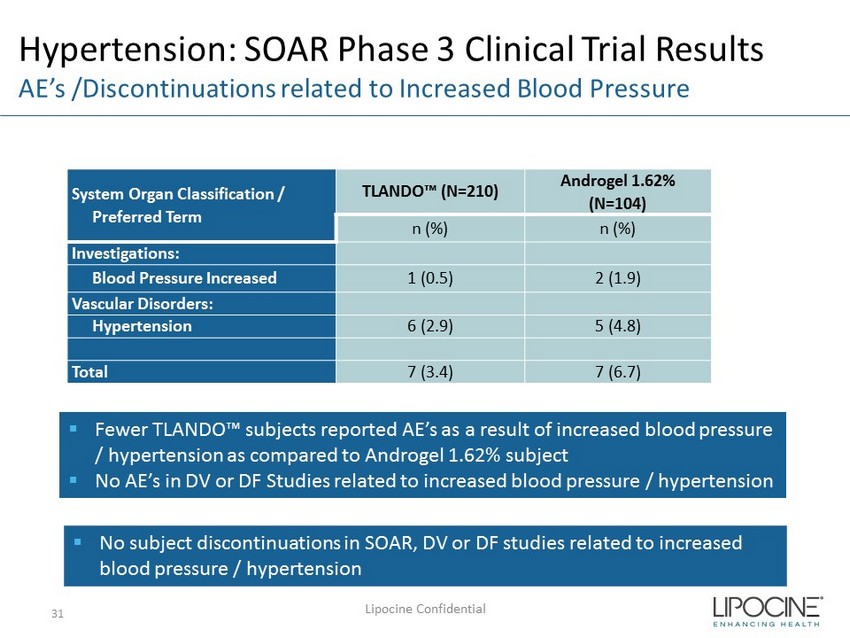
Hypertension: SOAR Phase 3 Clinical Trial Results AE’s /Discontinuations related to Increased Blood Pressure System Organ Classification / Preferred Term TLANDO™ (N=210) Androgel 1.62% (N=104) n (%) n (%) Investigations: Blood Pressure Increased 1 (0.5) 2 (1.9) Vascular Disorders: Hypertension 6 (2.9) 5 (4.8) Total 7 (3.4) 7 (6.7) Lipocine Confidential 31 ▪ Fewer TLANDO™ subjects reported AE’s as a result of increased blood pressure / hypertension as compared to Androgel 1.62% subject ▪ No AE’s in DV or DF Studies related to increased blood pressure / hypertension ▪ No subject discontinuations in SOAR, DV or DF studies related to increased blood pressure / hypertension
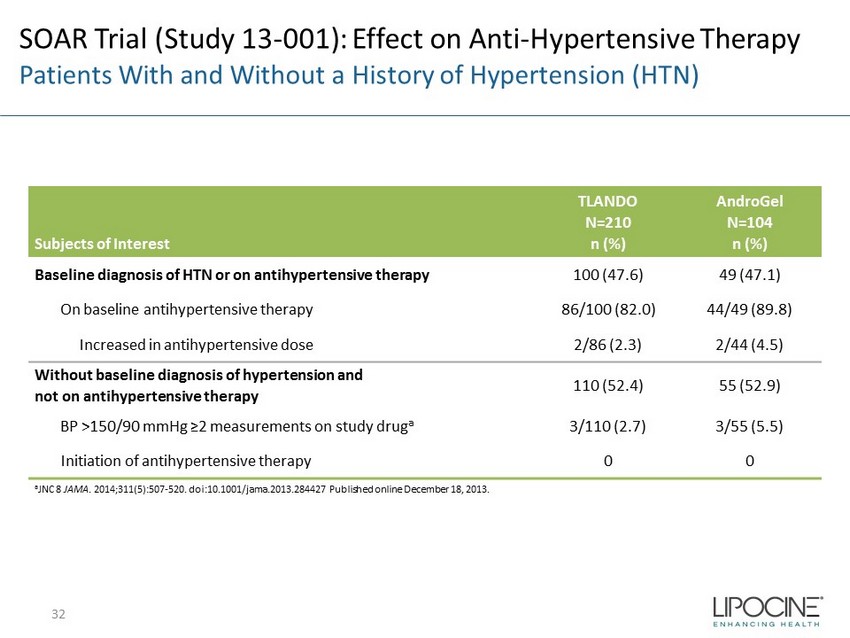
SOAR Trial (Study 13 - 001): Effect on Anti - Hypertensive Therapy Patients With and Without a History of Hypertension (HTN) Subjects of Interest TLANDO N=210 n (%) AndroGel N=104 n (%) Baseline diagnosis of HTN or on antihypertensive therapy 100 (47.6) 49 (47.1) On baseline antihypertensive therapy 86/100 (82.0) 44/49 (89.8) Increased in antihypertensive dose 2/86 (2.3) 2/44 (4.5) Without baseline diagnosis of hypertension and not on antihypertensive therapy 110 (52.4) 55 (52.9) BP >150/90 mmHg ≥2 measurements on study drug a 3/110 (2.7) 3/55 (5.5) Initiation of antihypertensive therapy 0 0 a JNC 8 JAMA . 2014;311(5):507 - 520. doi:10.1001/jama.2013.284427 Published online December 18, 2013. 32
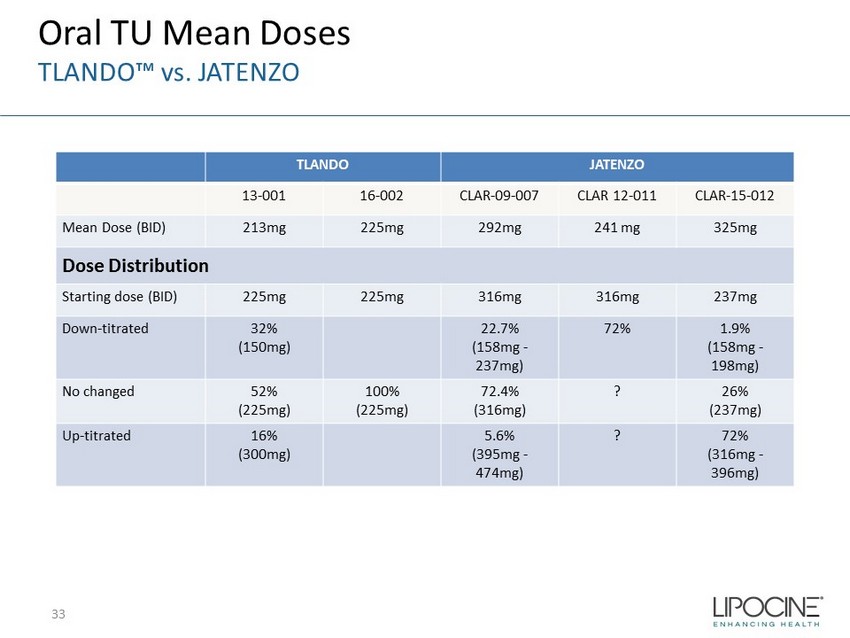
TLANDO JATENZO 13 - 001 16 - 002 CLAR - 09 - 007 CLAR 12 - 011 CLAR - 15 - 012 Mean Dose (BID) 213mg 225mg 292mg 241 mg 325mg Dose Distribution Starting dose (BID) 225mg 225mg 316mg 316mg 237mg Down - titrated 32% (150mg) 22.7% (158mg - 237mg) 72% 1.9% (158mg - 198mg) No changed 52% (225mg) 100% (225mg) 72.4% (316mg) ? 26% (237mg) Up - titrated 16% (300mg) 5.6% (395mg - 474mg) ? 72% (316mg - 396mg) 33 Oral TU Mean Doses TLANDO™ vs. JATENZO

Jatenzo ™ Duplicative Clinic Blood Pressure Results* Over Time 34 0.2 0.8 2.8 1.8 - 0.5 - 0.7 Dose Level Appears to Have a Role in Blood Pressure Increase Mean Dose: 237mg Mean Dose: 325mg x Minimal Change in BP from Baseline at Visit 2 *From Jatenzo briefing document for Jan 9 th BRUDAC meeting

• Whole blood collected in plain serum tubes • TU dissolved in ethanolic PBS solution is spiked into freshly collected blood • Allowed to clot for 30 minutes at room temperature • Centrifuged for 15 minutes to harvest serum • TU and T concentrations are measured from the serum samples Serum Normal Clotting Time: 8 to 15 minutes Freshly collected blood in serum tubes (per clinical practice) TLANDO™: Ex - Vivo TU to T Conversion Additional Analysis 35
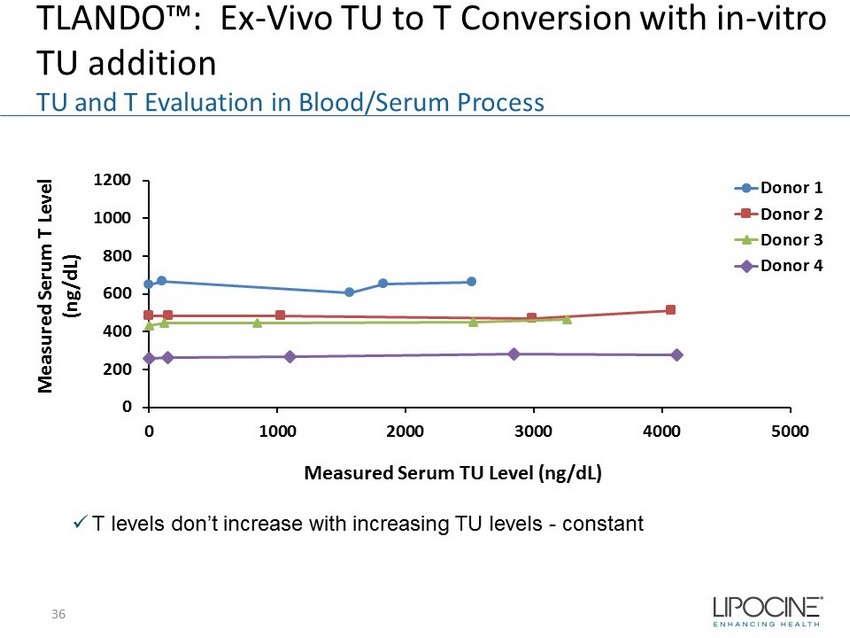
0 200 400 600 800 1000 1200 0 1000 2000 3000 4000 5000 Measured Serum T Level (ng/dL) Measured Serum TU Level (ng/dL) Donor 1 Donor 2 Donor 3 Donor 4 TLANDO™: Ex - Vivo TU to T Conversion with in - vitro TU addition TU and T Evaluation in Blood/Serum Process x T levels don’t increase with increasing TU levels - constant 36
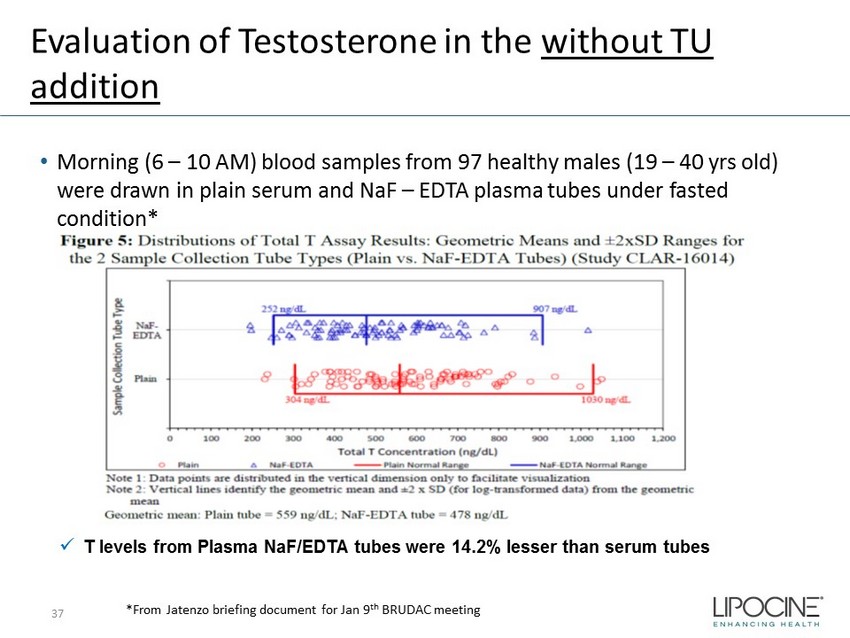
Evaluation of Testosterone in the without TU addition • Morning (6 – 10 AM) blood samples from 97 healthy males (19 – 40 yrs old) were drawn in plain serum and NaF – EDTA plasma tubes under fasted condition* x T levels from Plasma NaF /EDTA tubes were 14.2% lesser than serum tubes *From Jatenzo briefing document for Jan 9 th BRUDAC meeting 37

Evaluation of Testosterone post Oral TU administration • 14 blood samples from 8 hypogonadal subjects were drawn in plain serum and NaF – EDTA plasma tubes following a single oral dose of 316mg TU* x Cavg T levels from Plasma NaF /EDTA tubes were 13% lesser than serum tubes *From Jatenzo briefing document for Jan 9 th BRUDAC meeting 38 Similar differences between enzyme inhibited plasma and plain serum both in presence and absence of TU suggesting TU do not play a role in the difference

• No conversion of TU to T occurs ex - vivo during blood collection and serum sample processing • Treatment outcomes confirm reliability of T measurements – Pharmacological effects (e.g., LH/FSH suppression) – Testosterone metabolite levels (e.g., E2) TLANDO™: Ex - Vivo TU to T Conversion Serum Testosterone Assay Data Are Reliable Conclusion: 39

HPC Concentration and PTB Rates 40 40

PTB Prevalence CONFIDENTIAL Born Too Soon, Global Action Report on Preterm Birth, WHO, 2012 1. India 2. China 3. Nigeria 4. Pakistan 5. Indonesia 6. United States of America 7. Bangladesh 8. Philippines 9. Dem. Rep. of Congo 10. Brazil 10 Countries Account for 60% of the World’s Preterm Births by Rank: 41

PTB Risk Factors • Prior spontaneous PTB • Having a short cervix • African American ethnicity • Age <18 years or >40 years • Problem with uterus or cervix - mature cervical dilation • Certain infections during pregnancy (UTI, vaginal, uterine) • High blood pressure, stress, diabetes or being underweight or overweight • Short inter - pregnancy interval (6 - 18 months) • Smoking, drinking alcohol or Illicit drug use CONFIDENTIAL 42
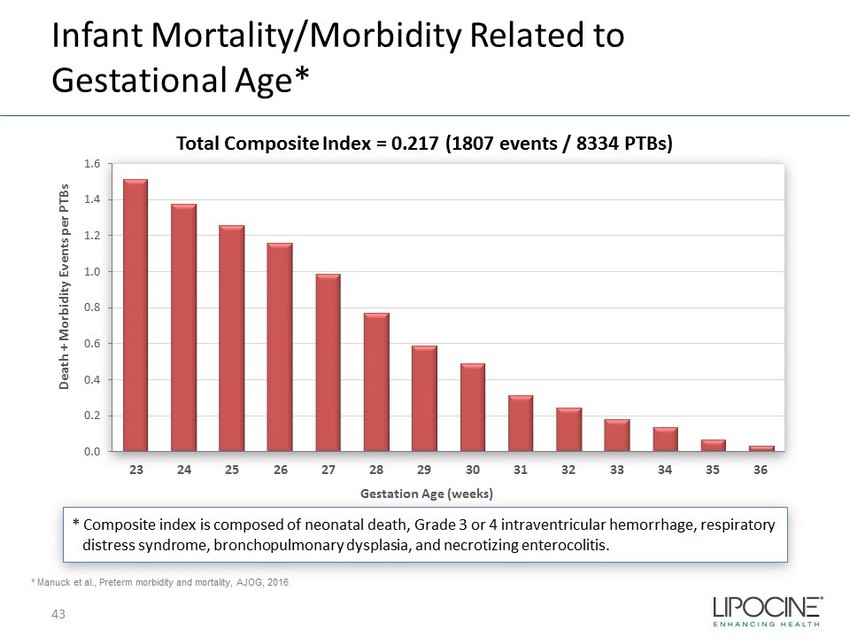
Infant Mortality/Morbidity Related to Gestational Age* * Composite index is composed of neonatal death, Grade 3 or 4 intraventricular hemorrhage, respiratory distress syndrome, bronchopulmonary dysplasia, and necrotizing enterocolitis. 0.0 0.2 0.4 0.6 0.8 1.0 1.2 1.4 1.6 23 24 25 26 27 28 29 30 31 32 33 34 35 36 Death + Morbidity Events per PTBs Gestation Age (weeks) * Manuck et al., Preterm morbidity and mortality, AJOG, 2016 Total Composite Index = 0.217 (1807 events / 8334 PTBs) 43
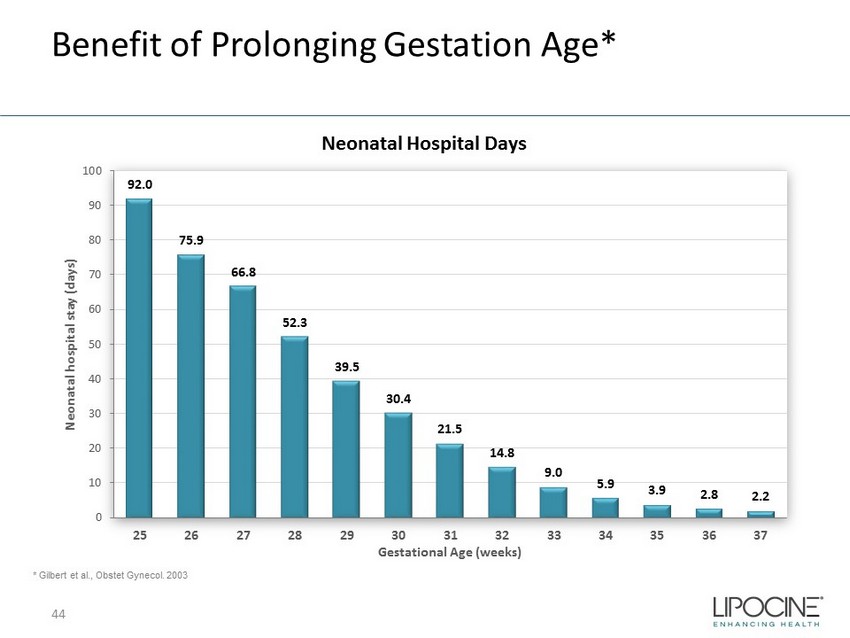
Benefit of Prolonging Gestation Age* * Gilbert et al., Obstet Gynecol. 2003 Neonatal Hospital Days 92.0 75.9 66.8 52.3 39.5 30.4 21.5 14.8 9.0 5.9 3.9 2.8 2.2 0 10 20 30 40 50 60 70 80 90 100 25 26 27 28 29 30 31 32 33 34 35 36 37 Neonatal hospital stay (days) Gestational Age (weeks) 44

High PTB Medical Costs • 12% of all US pregnancies 1 (475 - 500K) result in PTB (< 37 weeks) - a leading cause of neonatal mortality and morbidity • First year medical costs for PTB infants are ~ 10x higher than for full term infants 2 • 28% of preterm births are to women with histories of early delivery 45 ≥ $26 Billion Economic Impact 3 1. CDC (2010) 2. J. Maternal - Fetal and Neonatal Medicine, Dec. 2006, 19(12), 773 – 782 3 . Institute of Medicine of the National Academies. Jul.200
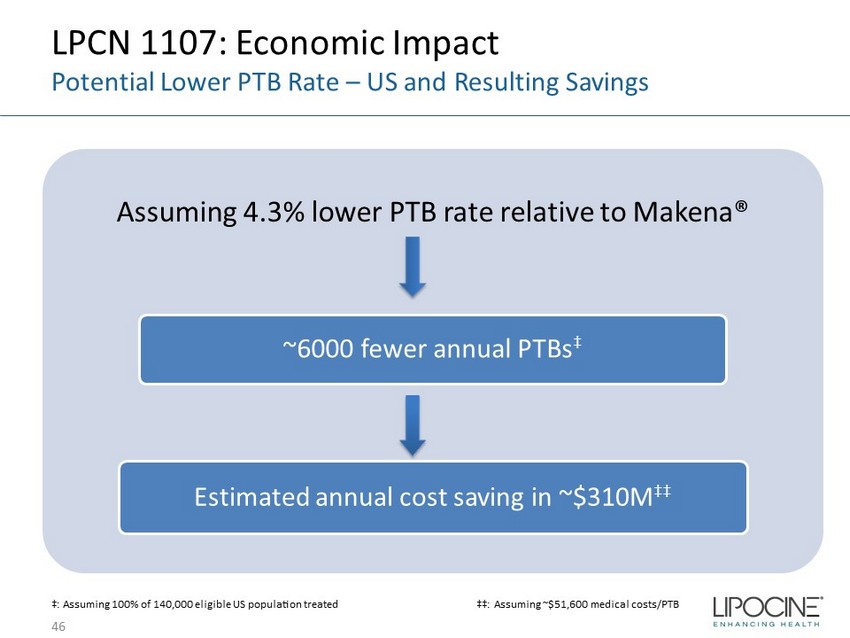
LPCN 1107: Economic Impact Potential Lower PTB Rate – US and Resulting Savings Assuming 4.3% lower PTB rate relative to Makena® ~6000 fewer annual PTBs ‡ Estimated annual cost saving in ~$310M ‡‡ 46 ‡: Assuming 100% of 140,000 eligible US population treated ‡‡: Assuming ~$51,600 medical costs/PTB
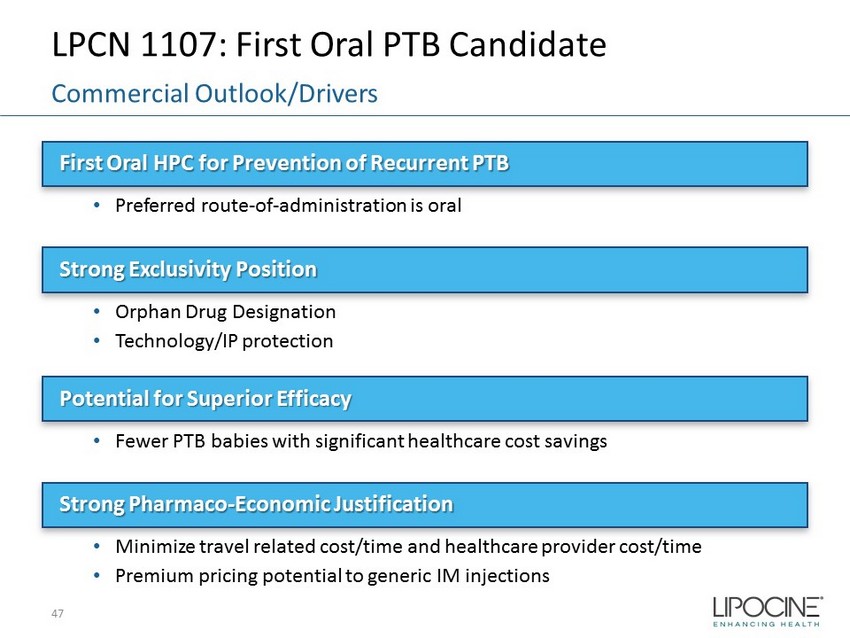
LPCN 1107: First Oral PTB Candidate First Oral HPC for Prevention of Recurrent PTB • Preferred route - of - administration is oral Strong Exclusivity Position • Orphan Drug Designation • Technology/IP protection Potential for Superior Efficacy • Fewer PTB babies with significant healthcare cost savings Strong Pharmaco - Economic Justification • Minimize travel related cost/time and healthcare provider cost/time • Premium pricing potential to generic IM injections 47 Commercial Outlook/Drivers
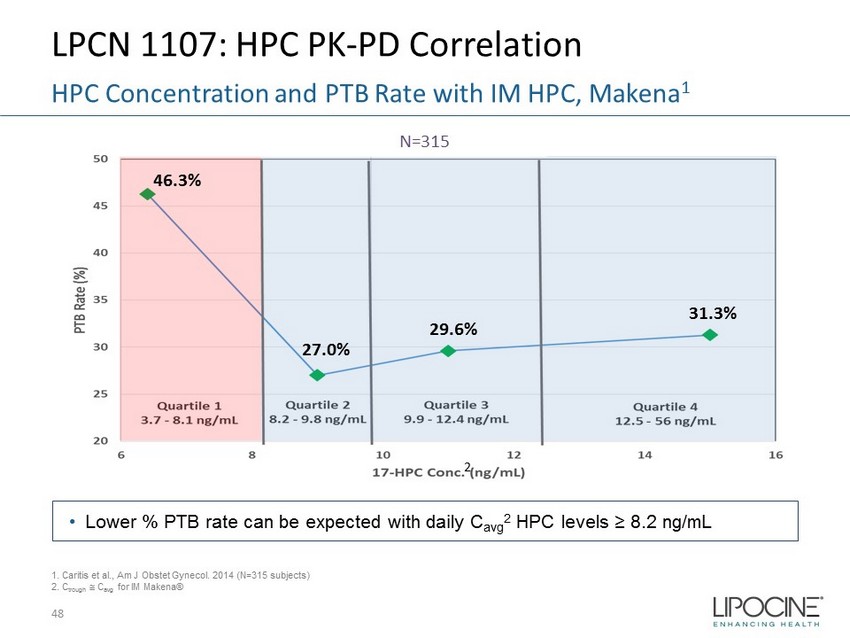
LPCN 1107: HPC PK - PD Correlation 48 HPC Concentration and PTB Rate with IM HPC, Makena 1 N=315 1. Caritis et al., Am J Obstet Gynecol. 2014 (N=315 subjects) 2. C trough C avg for IM Makena® • Lower % PTB rate can be expected with daily C avg 2 HPC levels ≥ 8.2 ng/mL 2 46.3% 27.0% 29.6% 31.3%
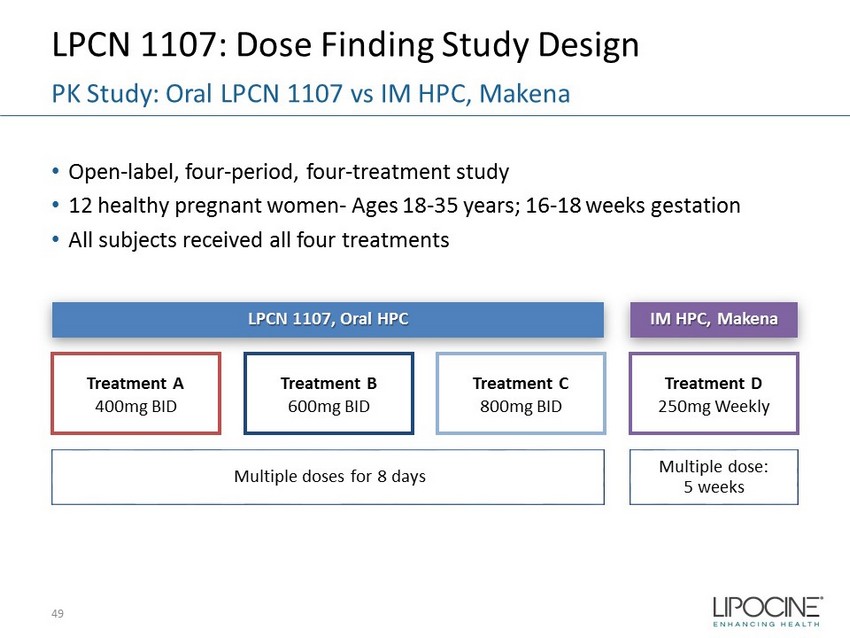
LPCN 1107: Dose Finding Study Design • Open - label, four - period, four - treatment study • 12 healthy pregnant women - Ages 18 - 35 years; 16 - 18 weeks gestation • All subjects received all four treatments 49 PK Study: Oral LPCN 1107 vs IM HPC, Makena Treatment D 250mg Weekly Treatment C 800mg BID Treatment B 600mg BID Treatment A 400mg BID IM HPC, Makena LPCN 1107, Oral HPC Multiple dose: 5 weeks Multiple doses for 8 days
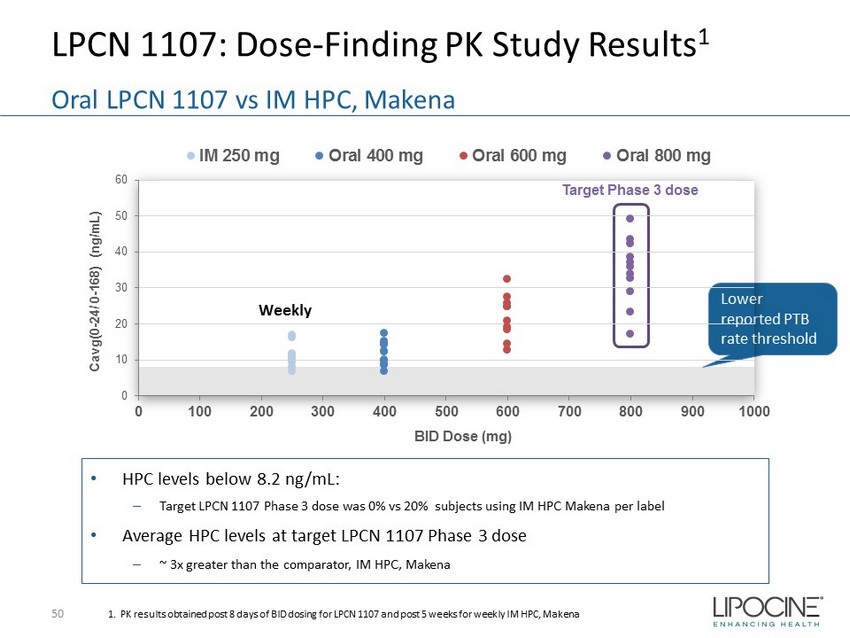
LPCN 1107: Dose - Finding PK Study Results 1 50 Oral LPCN 1107 vs IM HPC, Makena 1. PK results obtained post 8 days of BID dosing for LPCN 1107 and post 5 weeks for weekly IM HPC, Makena Lower reported PTB rate threshold • HPC levels below 8.2 ng/mL: – Target LPCN 1107 Phase 3 dose was 0% vs 20% subjects using IM HPC Makena per label • Average HPC levels at target LPCN 1107 Phase 3 dose – ~ 3x greater than the comparator, IM HPC, Makena Target Phase 3 dose 0 10 20 30 40 50 60 0 100 200 300 400 500 600 700 800 900 1000 Cavg(0 - 24/ 0 - 168) (ng/mL) BID Dose (mg) IM 250 mg Oral 400 mg Oral 600 mg Oral 800 mg Weekly

















































