UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 10-Q
| ☒ | Quarterly Report Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 |
| For the quarterly period ended March 31, 2023 |
| ☐ | Transition Report Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 |
| For the transition period from ______________ to ______________ |
Commission file number: 001-38362
PROLUNG, INC.
(Exact name of registrant as specified in its charter)
| Delaware | 20-1922768 | |
(State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) |
| 350 W. 800 N., Suite 214 | ||
| Salt Lake City, Utah | 84103 | |
| (Address of principal executive offices) | (Zip Code) |
(801) 736–0729
(Registrant’s telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||
| Common | None | None |
Indicate by check mark whether the issuer (1) filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☒ No ☐
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes ☒ No ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company or an emerging growth company. See definition of “accelerated filer”, “large accelerated filer”, “smaller reporting company” and “emerging growth company” in Rule 12b-2 of the Exchange Act (Check one):
| Large accelerated filer | ☐ | Accelerated filer | ☐ | |||
| Non-accelerated filer | ☒ | Smaller reporting company | ☒ | |||
| Emerging growth company | ☐ |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 7(a)(2)(B) of the Securities Act. ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ☐ No ☒.
Indicate the number of shares outstanding of each of the issuer’s classes of common stock, as of the latest practicable date: As of May 22, 2023 the issuer had shares of common stock, $0.001 par value, outstanding.
PROLUNG, INC.
TABLE OF CONTENTS
| 2 |
PART I – FINANCIAL INFORMATION
ITEM 1. FINANCIAL STATEMENTS
ProLung, Inc.
Condensed Balance Sheets
| 2023 | 2022 | |||||||
| March 31, | December 31, | |||||||
| 2023 | 2022 | |||||||
| (Unaudited) | ||||||||
| Assets | ||||||||
| Current Assets | ||||||||
| Cash | $ | 842,131 | $ | 296,496 | ||||
| Total Current Assets | 842,131 | 296,496 | ||||||
| Property and equipment, net | 5,793 | 6,274 | ||||||
| Operating lease right of use asset | 113,026 | 123,431 | ||||||
| Intangible assets, net | 105,978 | 108,368 | ||||||
| Total Assets | $ | 1,066,928 | $ | 534,569 | ||||
| Liabilities and Stockholders’ Deficit | ||||||||
| Current Liabilities | ||||||||
| Accounts payable | $ | 124,661 | $ | 76,540 | ||||
| Accrued liabilities | 2,823,620 | 2,610,804 | ||||||
| Operating lease liability - current | 42,980 | 40,959 | ||||||
| Payable for research and development - current | 315,000 | 315,000 | ||||||
| Convertible notes payable, related party, net - current | 566,843 | 439,000 | ||||||
| Convertible notes payable - current, net | 10,607,045 | 10,018,022 | ||||||
| Derivative liability | 143,630 | - | ||||||
| Total Current Liabilities | 14,623,779 | 13,500,325 | ||||||
| Long-Term Liabilities | ||||||||
| Operating lease liability - long term | 72,279 | 83,748 | ||||||
| Total Long-Term Liabilities | 72,279 | 83,748 | ||||||
| Total Liabilities | 14,696,058 | 13,584,073 | ||||||
| Stockholders’ Deficit: | ||||||||
| Preferred stock, $ par value; shares authorized; issued and outstanding | - | - | ||||||
| Common stock, $par value; shares authorized; and shares issued and outstanding | 4,614 | 4,591 | ||||||
| Additional paid-in capital | 31,661,166 | 31,497,065 | ||||||
| Subscription receivable | - | (65,000 | ) | |||||
| Accumulated deficit | (45,294,910 | ) | (44,486,160 | ) | ||||
| Total Stockholders’ Deficit | (13,629,130 | ) | (13,049,504 | ) | ||||
| Total Liabilities and Stockholders’ Deficit | $ | 1,066,928 | $ | 534,569 | ||||
The accompanying notes are an integral part of these unaudited condensed financial statements
| 3 |
ProLung, Inc.
Condensed Statements of Operations
(Unaudited)
| 2023 | 2022 | |||||||
| For the Three Months Ended | ||||||||
| March 31, | ||||||||
| 2023 | 2022 | |||||||
| Revenues: | ||||||||
| Revenue | $ | - | $ | - | ||||
| Total revenue | - | - | ||||||
| Cost of revenue: | - | - | ||||||
| Gross margin | - | - | ||||||
| Operating expenses: | ||||||||
| Research and development expense | 197,223 | 726,711 | ||||||
| Selling, general and administrative expense | 371,441 | 703,810 | ||||||
| Total operating expenses | 568,664 | 1,430,521 | ||||||
| Loss from operations | (568,664 | ) | (1,430,521 | ) | ||||
| Other income (expense): | ||||||||
| Interest expense | (240,086 | ) | (330,903 | ) | ||||
| Total other expense | (240,086 | ) | (330,903 | ) | ||||
| Net loss | $ | (808,750 | ) | $ | (1,761,424 | ) | ||
| Basic and diluted loss per share | $ | (0.18 | ) | $ | (0.42 | ) | ||
| Weighted-average common shares outstanding, basic and diluted | 4,592,488 | 4,157,158 | ||||||
The accompanying notes are an integral part of these unaudited condensed financial statements
| 4 |
ProLung, Inc.
Condensed Statements of Cash Flows
(Unaudited)
| 2023 | 2022 | |||||||
| For the Three Months Ended | ||||||||
| March 31, | ||||||||
| 2023 | 2022 | |||||||
| Cash flows from operating activities: | ||||||||
| Net loss | $ | (808,750 | ) | $ | (1,761,424 | ) | ||
| Adjustments to reconcile net loss to net cash flows from operating activities: | ||||||||
| Depreciation and amortization | 2,871 | 15,464 | ||||||
| Amortization of right of use asset | 10,405 | 172,961 | ||||||
| Stock-based compensation - share issuance and options/warrants | 155,868 | 437,437 | ||||||
| Amortization of loan discount | 24,372 | 126,593 | ||||||
| Change in assets and liabilities: | ||||||||
| Accounts payable | 48,121 | (6,877 | ) | |||||
| Accrued liabilities | 212,816 | 204,277 | ||||||
| Operating lease liability | (9,448 | ) | - | |||||
| Net cash flows used in operating activities | (363,745 | ) | (811,569 | ) | ||||
| Cash flows from investing activities: | ||||||||
| Net cash flows used in investing activities | - | - | ||||||
| Cash flows from financing activities: | ||||||||
| Proceeds from settlement of subscription receivable | 65,000 | - | ||||||
| Proceeds from exercise of warrants | 8,256 | 1,318,722 | ||||||
| Proceeds from convertible notes payable - related party | 150,000 | - | ||||||
| Proceeds from convertible notes payable | 686,124 | - | ||||||
| Net cash flows provided by financing activities | 909,380 | 1,318,722 | ||||||
| Net increase in cash | 545,635 | 507,153 | ||||||
| Cash at beginning of period | 296,496 | 745,003 | ||||||
| Cash at end of period | $ | 842,131 | $ | 1,252,156 | ||||
| Supplemental disclosure of cash flow information: | ||||||||
| Cash paid for interest | $ | - | $ | - | ||||
| Supplemental disclosure of non-cash investing and financing activities: | ||||||||
| Establishment of convertible note discount/derivative liability | $ | 143,630 | $ | - | ||||
| Cashless exercise of warrants | $ | 22 | $ | 52 | ||||
| Exercise of warrants where funds yet received | $ | - | $ | 670,234 | ||||
The accompanying notes are an integral part of these unaudited condensed financial statements
| 5 |
ProLung, Inc.
Condensed Statement of Stockholders’ Deficit
For the Three Months Ended March 31, 2022 and 2023
(Unaudited)
| Shares | Amount | Capital | Receivable | Deficit | Deficit | |||||||||||||||||||
| Common Stock | Additional Paid-in | Subscription | Accumulated | Total Stockholders’ | ||||||||||||||||||||
| Shares | Amount | Capital | Receivable | Deficit | Deficit | |||||||||||||||||||
| Balance, December 31, 2022 | 4,591,399 | $ | 4,591 | $ | 31,497,065 | $ | (65,000 | ) | (44,486,160 | ) | $ | (13,049,504 | ) | |||||||||||
| Exercise of warrants for cash | 1,290 | 1 | 8,255 | - | - | 8,256 | ||||||||||||||||||
| Cashless exercise of warrants | 22,300 | 22 | (22 | ) | - | - | - | |||||||||||||||||
| Settlement of subscription receivable | - | - | - | 65,000 | - | 65,000 | ||||||||||||||||||
| Stock-based compensation | - | - | 155,868 | - | - | 155,868 | ||||||||||||||||||
| Net loss | - | - | - | - | (808,750 | ) | (808,750 | ) | ||||||||||||||||
| Balance, March 31, 2023 | 4,614,989 | $ | 4,614 | $ | 31,661,166 | $ | - | $ | (45,294,910 | ) | $ | (13,629,130 | ) | |||||||||||
| Common Stock | Additional Paid-in | Subscription | Accumulated | Total Stockholders’ | ||||||||||||||||||||
| Shares | Amount | Capital | Receivable | Deficit | Deficit | |||||||||||||||||||
| Balance, December 31, 2021 | 4,122,130 | $ | 4,122 | $ | 28,571,741 | $ | - | $ | (40,126,759 | ) | $ | (11,550,896 | ) | |||||||||||
| Exercise of warrants for cash | 253,601 | 254 | 1,318,468 | - | - | 1,318,722 | ||||||||||||||||||
| Cashless exercise of warrants | 52,174 | 52 | (52 | ) | - | - | - | |||||||||||||||||
| Exercise of warrants for acquired research and development | 19,414 | 20 | 113,730 | - | - | 113,750 | ||||||||||||||||||
| Additional shares of common stock issued to warrant holders | 27,671 | 28 | 254,546 | - | - | 254,574 | ||||||||||||||||||
| Exercise of warrants funds not yet received | 128,890 | 127 | 670,107 | (670,234 | ) | - | - | |||||||||||||||||
| Issuance of common stock acquired research and development | 6,436 | 6 | 59,205 | - | - | 59,211 | ||||||||||||||||||
| Stock-based compensation - from option - warrant amortization | - | - | 182,863 | - | - | 182,863 | ||||||||||||||||||
| Net loss | - | - | - | - | (1,761,424 | ) | (1,761,424 | ) | ||||||||||||||||
| Balance, March 31, 2022 | 4,610,316 | $ | 4,609 | $ | 31,170,608 | $ | (670,234 | ) | $ | (41,888,183 | ) | $ | (11,383,200 | ) | ||||||||||
The accompanying notes are an integral part of these unaudited condensed financial statements
| 6 |
ProLung, Inc. and Subsidiary
Notes to Condensed Financial Statements
(Unaudited)
Note 1 – Organization and Summary of Significant Accounting Policies
Organization
ProLung, Inc. (the “Company”), is a Delaware corporation that was incorporated on November 22, 2004 and is doing business as (dba) “IONIQ Sciences” and “ProLung.” The Company’s headquarters are located in Salt Lake City, Utah. The Company’s business is the development of a modern solution for the early detection of multiple cancers thereby expanding the therapeutic window, significantly improving survivability, and reducing the cost of healthcare. One in two Americans will be diagnosed with cancer during their lifetime and one in five will die. Clinical literature shows that early detection can save lives and money. IONIQ Sciences operates at the confluence of its Electrical Impedance Analytics (EIA) technology or bioimpedance and artificial intelligence (AI). The Company has active projects in lung, breast and gastro-intestinal (GI) cancers. The first planned product utilizing its proprietary digital platform, the IONIQ ProLung Test™ for lung cancer, has been designated a Breakthrough Device by the U.S. FDA in February 2020. The Company submitted its de novo application to the U.S. FDA in February 2022 and received a substantive review from the FDA outlining additional information that is required for US regulatory clearance. These items include questions and requests related to biocompatibility testing, clinical data, Indications for use and labeling, cybersecurity documentation, electrical safety, special controls, human factors and usability, performance testing, risk analysis, software, sterility and shelf life. IONIQ Sciences remain fully-committed to gaining U.S. FDA regulatory de novo clearance and continue to collaborate with the US FDA so the Company can satisfy their requests and subsequently commercialize the IONIQ ProLung Test for lung cancer.
Basis of Presentation
The accompanying condensed financial statements have been prepared by management in accordance with rules and regulations promulgated by the U.S. Securities and Exchange Commission and therefore certain information and disclosures normally included in financial statements prepared in accordance with accounting principles generally accepted in the United States of America have been condensed or omitted pursuant to such rules and regulations. In the opinion of management, the accompanying condensed financial statements contain all adjustments necessary for them to be presented fairly, with those adjustments consisting only of normal recurring adjustments. These interim financial statements should be read in conjunction with the Company’s annual financial statements included in the Company’s Annual Report on Form 10-K for the year ended December 31, 2022. The results of operations for the three months ended March 31, 2023 may not be indicative of the results to be expected for the year ending December 31, 2023.
Going Concern
The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. The Company has generated minimal revenues thus far from its operations and no revenue during the current period. Until the Company receives Food and Drug Administration (“FDA”) approval, the Company will not achieve its planned level of operations in the United States. The Company has licensed a portion of its technology to an entity located in China. The Company has incurred substantial and recurring losses to date from operations, continues to have a stockholders’ deficit and is currently dependent on debt and equity financing. These conditions raise substantial doubt about the Company’s ability to continue as a going concern for a period of one year from the issuance of these financial statements. The accompanying financial statements do not include any adjustments that might result relating to the recoverability and classification of the asset carrying amounts or the amount and classification of liabilities that might result from the outcome of this risk and uncertainty.
The ability of the Company to continue as a going concern is dependent on the Company successfully obtaining additional funding, developing products that can be sold profitably, and generating cash through operating activities. Management’s plans include issuing equity or debt securities to fund capital requirements and developing ongoing operations.
| 7 |
ProLung, Inc. and Subsidiary
Notes to Condensed Financial Statements
(Unaudited)
| 2023 | 2022 | |||||||
| For the Three Months Ended | ||||||||
| March 31, | ||||||||
| 2023 | 2022 | |||||||
| Warrants to purchase shares | 251,285 | 316,180 | ||||||
| Stock options | 933,720 | 791,588 | ||||||
| Convertible notes | 3,415,179 | 3,202,149 | ||||||
| Antidilutive securities excluded from computation of earning per share | 4,600,184 | 4,309,917 | ||||||
Recent Accounting Pronouncements
The Company has reviewed other recent accounting pronouncements and has determined that they will not significantly impact the Company’s results of operations or financial position.
Note 2 – Accrued Liabilities
Accrued liabilities consisted of the following at March 31, 2023 and December 31, 2022:
Schedule of Accrued Liabilities
| March 31, | December 31, | |||||||
| 2023 | 2022 | |||||||
| Accrued interest | $ | 2,764,288 | $ | 2,551,006 | ||||
| Accrued royalties | 17,873 | 17,873 | ||||||
| Accrued payroll and payroll taxes | 41,459 | 41,925 | ||||||
| Accrued liabilities | $ | 2,823,620 | $ | 2,610,804 | ||||
| 8 |
ProLung, Inc. and Subsidiary
Notes to Condensed Financial Statements
(Unaudited)
Note 3 – Convertible Notes Payable
$2 Million Bridge Notes - In February 2023, the company initiated a $2M bridge financing round. As of March 31, 2023, $836,124 ($150,000 from current board members) in financing has been received. These notes bear interest at 8% and are due September 30, 2023.
The Convertible Notes were convertible upon the occurrence of certain events, including upon a change in control or a next equity financing. The conversion features are described as follows:
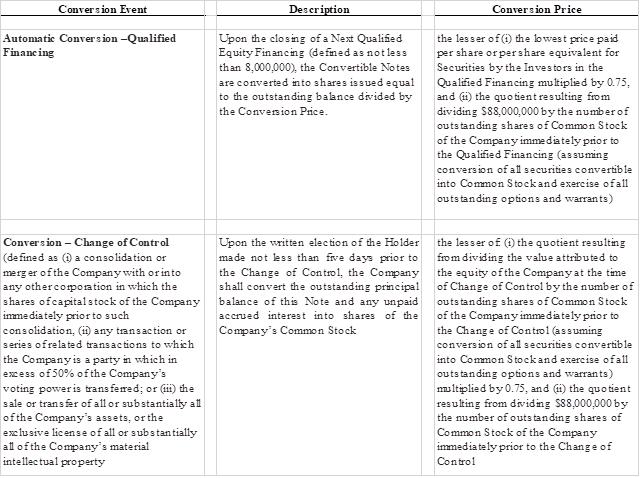
The Company determined that the terms that could result in the Bridge Notes automatically converting at 75% of the lowest per-share selling price of the stock sold by the Company in the Next Qualified Equity Financing and are deemed a redemption feature. The Company also concluded that the redemption features require bifurcation from the Bridge Notes and subsequent accounting in the same manner as a freestanding derivative.
The assigned fair value of the embedded redemption derivative liability of $143,630 (includes related party amount of $) was recorded as debt discounts to be amortized to interest expense over the respective term using the effective interest method. During the three months ended March 31, 2023, the Company recognized interest expense of $24,372 (includes related party amount of $) from the accretion of the debt discount.
Convertible Notes - In March 2022, all of the Company’s convertible notes were amended with the following new terms: (1) the maturity date was extended to September 30, 2023, (2) there is an expanded automatic contingent conversion to include a qualified financing of at least $10M and (3) added a three-month bonus interest payment in the event an automatic contingent conversion is triggered.
Notes payable are summarized as follows:
Schedule of Convertible Notes Payable
| March 31, | December 31, | |||||||
| 2023 | 2022 | |||||||
| $2 Millon Secured Bridge Notes; interest at 8%; due September 2023 (includes related party amount of $) | $ | 836,124 | $ | - | ||||
| Convertible notes payable; unsecured; interest at 8%; due September 2023 (includes related party amount of $) | 10,457,022 | 10,457,022 | ||||||
| Unamortized loan cost (includes related party amount of $) | (119,258 | ) | - | |||||
| Notes payable, net | $ | 11,173,888 | $ | 10,457,022 | ||||
| Less: current portion, net | (11,173,888 | ) | (10,457,022 | ) | ||||
| Convertible notes payable - long term, net | $ | - | $ | - | ||||
Note 4 - Derivative Liability
As described above, the redemption provisions embedded in the Bridge Notes required bifurcation and measurement at fair value as a derivative. The fair value of the Bridge Note embedded redemption derivative liability was calculated by determining the value of the debt component of the Bridge Notes at various conversion or maturity dates using a Probability Weighted Expected Return valuation method. The fair value calculation placed greater probability on the occurrence of the conversion or the maturity date scenario, with little or no weight given to other scenarios. The fair value of the embedded redemption derivative liability is significantly influenced by the discount rate, the remaining term to maturity and the Company’s assumptions related to the probability of a qualified financing or no financing prior to maturity. The Financing Date is the estimated date of an automatic conversion as the result of a Next Qualified Equity Financing.
The Derivative Liability is considered a Level 3 recurring financial instrument and will be revalidated every period. Currently, the derivative liability is the only Level 3 financial instrument the Company has.
Equity Incentive Plan
In April 2017, the Board, contingent on shareholder approval, approved the ProLung, Inc. Stock Incentive Plan (the “Plan”). The shareholders approved the Plan in July 2017. The Plan authorizes the Board compensation Committee to grant incentive stock options, non-incentive stock options, stock bonuses, restricted stock, and performance-based awards to directors, officers and employees and non-employee agents, consultants, advisers and independent contractors of the Company or any parent or subsidiary of the Company. The total number of initial shares of Common Stock authorized for issuance under the Plan was shares; the authorized shares will automatically increase on January 1st of each year, for ten consecutive years, commencing on January 1, 2018, by the lesser of (i) shares of Common Stock (i.e., 8% of the shares of the shares originally authorized to be issued), or (ii) such number of shares of common stock (if any) the Board may earlier designate in writing. In April 2022, the Board authorized an additional issuance of shares on 5/1/2022, shares on 1/1/2023, and to be issued 1/1/2024.
| 9 |
ProLung, Inc. and Subsidiary
Notes to Condensed Financial Statements
(Unaudited)
As part of an agreement for their service during the three months ended March 31, 2023 current Board members and advisors accepted the issuance of options at an exercise price of $ per option. These options vested upon issuance. The fair value of these options was $6.29 per option or $ and was expensed upon grant. The fair value was computed using the Black Scholes method using the following weighted-average assumptions:
| 2023 | ||||
| Expected life | years | |||
| Exercise price | $ | |||
| Expected volatility | % to | % | ||
| Weighted average volatility | % | |||
| Expected dividends | n/a | |||
| Risk-free interest rate | % | |||
Summary of Option Activity
| Weighted | Aggregate | |||||||||||||
| Weighted | Average | Intrinsic | ||||||||||||
| Shares | Average | Remaining | Value of | |||||||||||
| Under | Exercise | Contractual | Vested | |||||||||||
| Options | Price | Life | Options | |||||||||||
| Outstanding at December 31, 2022 | 917,096 | $ | 6.01 | years | ||||||||||
| Issued | 16,624 | $ | - | |||||||||||
| Exercised | - | $ | - | |||||||||||
| Forfeited/Expired/Adjustment | - | $ | - | |||||||||||
| Outstanding at March 31, 2023 | 933,720 | $ | 6.08 | years | $ | 3,530,341 | ||||||||
| Vested at March 31, 2023 | 842,086 | $ | 5.95 | years | $ | 3,292,168 | ||||||||
The Company recorded an expense of $ and $ for the three months ended March 31, 2023 and 2022 related to the amortization of options issued under the plan. The remaining unrecognized expense of $ will be recognized through June 2026 (weighed average years).
Schedule of Share-based Compensation Expense
| For the Three Months Ended | ||||||||
| March 31, | ||||||||
| 2023 | 2022 | |||||||
| Research and development expense | $ | 9,061 | $ | 177,952 | ||||
| Selling, general and administrative expense | 146,807 | 432,446 | ||||||
| Total share-based compensation | $ | 155,868 | $ | 610,398 | ||||
| 10 |
ProLung, Inc. and Subsidiary
Notes to Condensed Financial Statements
(Unaudited)
Note 6 – Common Stock Warrants
During the three months ended March 31, 2023, warrant holders exercised warrants as follows:
| - | warrants were exercised for $8,256 in cash proceeds. | |
| - | 47,186 warrants were exercised in a cashless exercise whereby shares with a value of $9.86 per share were used to settle the exercise price and the remaining shares were issued to the warrant holders. |
A summary of warrant activity for the three months ended March 31, 2023 is presented below:
Schedule of Warrant Activity
| Weighted | Aggregate | |||||||||||||
| Weighted | Average | Intrinsic | ||||||||||||
| Shares | Average | Remaining | Value of | |||||||||||
| Under | Exercise | Contractual | Vested | |||||||||||
| Warrants | Price | Life | Warrants | |||||||||||
| Outstanding at December 31, 2022 | 299,761 | $ | 5.16 | |||||||||||
| Issued | - | $ | - | |||||||||||
| Exercised | (48,476 | ) | $ | 5.23 | ||||||||||
| Expired/Forfeited | - | $ | - | |||||||||||
| Outstanding at March 31, 2023 | 251,285 | $ | 5.15 | years | $ | 1,219,890 | ||||||||
Note 7 – Stockholders’ Equity
During the three months ended March 31, 2023, as discussed in Note 6, warrants were exercised for the issuance of common stock. Also, $65,000 in cash was received during the three months ended March 31, 2023 in settlement of the subscription receivable.
Note 8 – Commitments and Contingencies
Leases
In September 2022, the Company renegotiated their office space lease from a month-to-month lease to a long-term operating lease agreement. The lease agreement is for 36 months requiring monthly payments as follows:
Schedule of Operating Lease
| Beginning Date | Ending Date | Payment | ||||||
| September 2022 | - | August 2023 | $ | 3,960 | ||||
| September 2023 | - | August 2024 | $ | 4,356 | ||||
| September 2024 | - | August 2025 | $ | 4,487 | ||||
On January 1, 2022, the Company adopted Topic 842, Leases which requires a lessee to record a right-of-use asset and a corresponding lease liability at the inception of the lease initially measured at the present value of the lease payments. The Company determined that the fair value of the lease asset and liability at the inception of the lease using a discount rate of 8% which was the cost of capital for the Company.
During the three months ended March 31, 2023, the Company made payments resulting in a $9,448 reduction in the lease liability. As of March 31, 2023 the lease liability amounted to $115,259. Topic 842 requires recognition in the statement of operations of a single lease cost, calculated so that the cost of the lease is allocated over the lease term, generally on a straight-line basis. The right-of-use asset on March 31, 2023 was $113,026, net of amortization of $10,405. The remaining term of the lease is 29 months on March 31, 2023.
Maturities of the Company’s lease liability is as follows:
Schedule of Maturities Lease Liability
| Year Ending December 31, | ||||
| 2023 (Remaining) | $ | 37,620 | ||
| 2024 | 52,796 | |||
| 2025 | 35,896 | |||
| Less: Interest | (11,053 | ) | ||
| Total | $ | 115,259 | ||
The above liability is presented on the accompanying balance sheet as follows:
Schedule of Right-of-Use Asset and Lease Liabilities
| Lease liability - short-term | $ | 42,980 | ||
| Lease liability - long-term | $ | 72,279 |
Research and Development Agreement
On July 29, 2019, the Company amended a license agreement dated April 10, 2013 between the Company and ProLung Biotech Wuxi / ProLung China (Wuxi). The original agreement allowed Wuxi to utilize the Company’s technology in China in return for royalty payments based on Wuxi’s revenues. Wuxi has yet to earn any revenue but has been conducting clinical trials. The license agreement was amended whereby Wuxi will provide the Company its clinical trial data, know-how and improvements which the Company will use outside the greater China area. This amendment further requires full collaboration (i.e., protocols and methodologies) between the two entities. In consideration for such trial data and know-how, the Company will make cash payments to Wuxi of up to $575,000 and issue up to shares of common stock upon the completion of certain events.
Through March 31, 2023, shares had been issued based on conditions being met. The final shares will be issued once the final milestone is met. Through March 31, 2023, $210,000 in payments had been made plus $50,000 in equipment with the remaining $315,000 currently payable.
Note 9 – Subsequent Events
Bridge Financing
Since March 31, 2023, The Company has raised an additional $100,000 in Bridge Financing through the issuance of Convertible Debt.
| 11 |
ITEM 2. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The following discussion and analysis should be read in conjunction with the financial statements and related notes that appear elsewhere in this Quarterly Report on Form 10-Q (this “Report”) and the Annual Report on Form 10-K for the year ended December 31, 2022 (the “2022 Form 10-K”) of ProLung, Inc. (the “Company”).
The statements contained in this Report that are not purely historical are forward-looking statements. Our forward-looking statements include, but are not limited to, statements regarding our expectations, hopes, beliefs, intentions, or strategies regarding the future. In addition, any statements that refer to projections, forecasts, or other characterizations of future events or circumstances, including any underlying assumptions, are forward-looking statements. The words “anticipates,” “believe,” “continue,” “could,” “estimate,” “expect,” “intend,” “may,” “might,” “plan,” “possible,” “potential,” “predict,” “project,” “should” and “would,” as well as similar expressions, may identify forward-looking statements, but the absence of these words does not mean a statement is not forward looking. The forward-looking statements contained in this Report are based on our current expectations and beliefs concerning future developments and their potential effects on us. There can be no assurance that future developments affecting us will be those that we have anticipated. These forward-looking statements involve a number of risks, uncertainties, or assumptions, many of which are beyond our control that may cause actual results or performance to be materially different from those expressed or implied by these forward-looking statements. Important factors that could cause these differences include the following:
| ● | We are a development stage company with limited revenue and no assurance of earning significant revenue over the long term. | |
| ● | We will need significant capital to execute our business plan, particularly as we continue to seek clearance from the FDA to market our IONIQ ProLung Test. | |
| ● | We are dependent upon financings to fund our operations and may be unable to continue as a going concern. | |
| ● | We have issued indebtedness and, if we are unable to repay or refinance it, our creditors could foreclose on our assets and force us into bankruptcy. | |
| ● | We are in the early stages of commercialization, and our IONIQ ProLung Test may never receive marketing approval from the FDA or achieve commercial market acceptance. | |
| ● | Our future growth depends, in part, on our ability to penetrate foreign markets, where we would be subject to additional regulatory burdens and other risks and uncertainties. | |
| ● | We are reliant on a single product and if we are not successful in commercializing the IONIQ ProLung Test and are unable to develop additional products, our business will not succeed. | |
| ● | We are subject to litigation risk for product liability if our IONIQ ProLung Test is not effective. | |
| ● | We may incur substantial product liability expenses due to manufacturing or design defects, or the use or misuse of our products. | |
| ● | We are subject to the risk of product recalls if our products are defective. | |
| ● | We may not obtain any, or adequate, third-party coverage and reimbursement for our prospective customers. | |
| ● | The absence of, or limits on, reimbursements may affect our revenues and our ability to achieve profitability. | |
| ● | If the IONIQ ProLung Test is not accepted by physicians and patients, we will be unable to achieve market acceptance. |
| 12 |
| ● | We are a small company and may be unable to compete with competitive technologies. | |
| ● | We are dependent upon our suppliers to safely and timely manufacture our products. | |
| ● | We are dependent upon third parties for marketing and other aspects of our business. | |
| ● | Any clinical trials that we conduct, including our ongoing trial, may not be completed on schedule, or at all, or may be more expensive than we expect, which could prevent or delay regulatory authorization(s) of our products or impair our financial position. | |
| ● | We engage in related party transactions, which result in a conflict of interest involving our management. | |
| ● | Our clinical studies, including our ongoing clinical study, may produce unfavorable results. | |
| ● | Our success depends upon our ability to effectively market our products. | |
| ● | We are dependent on key personnel, whose employment may be terminated by the Company or the employee at any time, which could cause significant disruption in our business and lead to significant expenses. | |
| ● | We must obtain regulatory clearance or approval in the US and other markets to be able to commence marketing and sales in those markets. | |
| ● | Even if we receive regulatory clearance or approval for the IONIQ ProLung Test, we still may not be able to successfully commercialize it and the revenue that we generate from its sales, if any, may be limited. | |
| ● | If we obtain FDA clearance or approval, we will be subject to Medical Device Reporting (MDR). | |
| ● | Recently proposed healthcare reform measures could hinder or prevent the commercial success of our products. | |
| ● | We will be subject to healthcare fraud and abuse law regulations. | |
| ● | Our business is subject to complex and evolving U.S. and international laws and regulation regarding privacy and data protection. Many of these laws and regulations are subject to change and uncertain interpretation and could result in claims, changes to our business practices, penalties, increased cost of operations, or declines in user growth or engagement, or otherwise harm our business. | |
| ● | IONIQ Sciences clinical study designs have not been reviewed by the FDA, and there is a risk that the FDA will not agree with our study designs or results. | |
| ● | We may be unable to protect our intellectual property rights, which are important to the potential value of our products and company. | |
| ● | We may incur significant costs and liability if we infringe, or are accused of infringing on, the intellectual property rights of others. | |
| ● | We may need to market the IONIQ ProLung Test under a different name in the EU to avoid the risk of trademark infringement. | |
| ● | If outstanding warrants are exercised, or Convertible Debentures are converted, stockholders will be diluted. | |
| ● | Our officers and directors have significant voting power and may take actions that may not be in the best interests of other stockholders. | |
| ● | Our common stock is not quoted or traded in any market, limiting liquidity opportunities for investors. |
| 13 |
| ● | Provisions in our charter documents and under Delaware law could discourage a takeover that stockholders may consider favorable and may lead to entrenchment of management. | |
| ● | We are subject to various regulatory regimes, and may be adversely affected by inquiries, investigations and allegations that we have not complied with governing rules and laws. | |
| ● | If a market develops for our common stock, we expect the market price to be volatile and trading in our common stock to be of limited volume. | |
| ● | We have never paid, and do not intend to pay in the future, dividends on our common stock. | |
| ● | Although we are capable of internally manufacturing to meet foreseeable demand, we may at some time be dependent upon contract manufacturers to safely and timely manufacture our products. | |
| ● | There is no guarantee that FDA clearance will lead to the IONIQ ProLung Test being approved by payors for reimbursement. | |
| ● | Our IONIQ ProLung Test may produce false positive and false negative results. |
In addition, please review the other, and more detailed, risk factors discussed in our 2022 Form 10-K.
Should one or more of these risks or uncertainties materialize, or should any of our assumptions prove incorrect, actual results may vary in material respects from those projected in these forward-looking statements.
Forward-looking statements speak only as of the date they are made. You should not put undue reliance on any forward-looking statements. We assume no obligation to update forward-looking statements to reflect actual results, changes in assumptions, or changes in other factors affecting forward-looking information, except to the extent required by applicable securities laws. If we do update one or more forward-looking statements, no inference should be drawn that we will make additional updates with respect to those or other forward-looking statements.
Overview
We are a medical technology company with a mission to dramatically improve the cancer landscape with a modern solution for the early detection of multiple cancers thereby expanding the therapeutic window, significantly improving survivability, and reducing the cost of healthcare. One in two Americans will be diagnosed with cancer during their lifetime and one in five will die. Clinical literature shows that early detection can save lives and money. We operate at the confluence of our Electrical Impedance Analytics (EIA) technology and artificial intelligence (AI). We are developing an advanced multi-cancer screening technology for early detection that will expand the therapeutic window, dramatically improve survivability and reduce the cost of healthcare. The first planned product utilizing our proprietary digital platform, the IONIQ ProLung Test™ for lung cancer, has been designated a Breakthrough Device by the U.S. FDA in February 2020. We submitted our de novo application to the U.S. FDA in February 2022. and we received a substantive review from the FDA outlining additional information that is required for US regulatory clearance. These items include questions and requests related to biocompatibility testing, clinical data, Indications for Use and labeling, cybersecurity documentation, electrical safety, special controls, human factors and usability, performance testing, risk analysis, software, sterility and shelf life. We remain fully-committed to gaining U.S. FDA regulatory de novo clearance and continue to collaborate with the US FDA so that we can satisfy their requests and subsequently commercialize the IONIQ ProLung Test for lung cancer.
Our non-invasive, rapid and radiation-free IONIQ ProLung Test has been developed to assess the risk of malignancy in lung nodules found in the chest by a Computed Tomography “CT” scan, which is currently the primary method used in the United States (“US”) for screening lung cancer. Lung cancer is the leading cause of cancer death in the US and the world according to American Cancer Society and World Health Organization. Earlier detection makes a substantial improvement in survival in individuals at high risk of lung cancer. Timely identification of malignancy is essential for patients and their families. Currently, patients often wait from three months to three and one-half years to have the risk of malignancy assessed through periodic CT scan surveillance. Until malignancy is determined to be likely, invasive biopsy and treatment are typically delayed. Current statistics reflect an average 17% survival rate at five years for those diagnosed with lung cancer.
| 14 |
We believe the IONIQ ProLung Test, in conjunction with the discovery of a nodule by CT scan, provides a more rapid assessment of the risk of malignancy, which must be determined prior to biopsy. Since a lung biopsy is invasive and may require life threatening thoracic surgery, physicians, patients, and insurance companies typically delay biopsy and therapy until the risk of malignancy outweighs the risk of further diagnostic procedures. For these patients, the delay can reduce the time available to treat the tumor and may cause sustained emotional trauma.
Results of Operations
The following discussion is included to describe our financial position and results of operations. The financial statements and notes thereto contain detailed information that should be referred to in conjunction with this discussion.
Revenues and Cost of Revenue. During the three months ended March 31, 2023 and March 31, 2022 we had no revenues or cost of revenues.
Operating Expenses. Total operating expense for the three months ended March 31, 2023 was $568,664 compared to the total operating expenses for the three months ended March 31, 2022 of $1,430,521, representing an decrease of $861,857. Operating expenses have been classified by management as either research and development or selling, general and administrative based on an assignment of certain expenses directly to these classifications or based on management’s allocation of certain expenses between these classifications.
The overall decrease in operating expense is primarily due to equity issuances for purchased research and development and expense recorded for additional shares issued to warrant holders during 2022.
Research and Development Expense. Research and development expense for the three months ended March 31, 2023, was $203,454, compared to research and development expense of $726,711 for the three months ended March 31, 2022; representing a decrease of $523,257. This increase was mostly due to research and development expenses from the acquisition of certain intellectual property from two entities paid for in cash and equity instruments.
Selling, General and Administrative Expense. Selling, general and administrative expense for the three months ended March 31, 2023 was $365,210 compared to selling, general and administrative of $703,810 for the three months ended March 31, 2022; representing a decrease of $338,600. This decrease was primarily due to shares issued to warrant holders for additional services partially offset by increased amortization of stock based compensation for employee options.
Other Expense. Other expense for the three months ended March 31, 2023 was $240,086 as compared to $330,903 for the three months ended March 31, 2022 representing an decrease of $90,817. This increase was solely due interest expense. During the three months ended March 31, 2022 we completed amortizing cash and non-cash loan costs related to our convertible debt. The Bridge Notes we issued during 2023 had no direct loan costs but did have a discount related to the derivative liability redemption feature.
Liquidity and Capital Resources
The following is a summary of our key liquidity measures at March 31, 2023 and December 31, 2022:
| March 31, | December 31, | |||||||
| 2023 | 2022 | |||||||
| Cash | $ | 842,131 | $ | 296,496 | ||||
| Current assets | 842,131 | 296,496 | ||||||
| Current liabilities | (14,623,779 | ) | (13,500,325 | ) | ||||
| Working (deficit) capital | $ | (13,781,648 | ) | $ | (13,203,829 | ) | ||
| 15 |
We need additional capital to continue our operations. During the three months ended March 31, 2023, we received $836,124 from the issuance of Convertible Bridge Notes and $73,256 from the exercise of warrants the settlement of our subscription receivable. In order for us to continue operations we will need additional capital which will require us to issue equity securities, debt securities and rights to acquire equity securities. Given our early stage of development, we may be unable to raise sufficient capital when needed and, in any case, will likely be required to pay a high price for capital.
Our future capital requirements and adequacy of available funds will depend on many factors including:
| ● | Our ability to find a commercial market for our IONIQ ProLung Test and obtain needed regulatory clearance | |
| ● | Our financial results; | |
| ● | The cost and availability of capital generally; and | |
| ● | The occurrence of unexpected adverse expenses or events. |
Cash provided by (used in) operating, investing and financing activities
Cash provided by (used in) operating, investing and financing activities for the Three Months Ended March 31, 2023 and 2022 is as follows:
| Three Months Ending | ||||||||
| 2023 | 2022 | |||||||
| Operating activities | $ | (363,745 | ) | $ | (811,569 | ) | ||
| Investing activities | - | - | ||||||
| Financing activities | 909,380 | 1,318,722 | ||||||
| Net increase (decrease) in cash | $ | 545,635 | $ | 507,153 | ||||
Operating Activities
For the three months ended March 31, 2023, the differences between our net loss and net cash used in operating activities were due to net non-cash charges totaling $193,516 for stock-based compensation, amortization and depreciation.
For the three months ended March 31, 2023, the differences between our net loss and net cash used in operating activities were due to net non-cash charges totaling $752,455 for stock-based compensation, share issuances, amortization of debt discount and depreciation.
Financing Activities
During the three months ended March 31, 2023, cash flows from financing activities totaled $909,380. This was from the issuance of Convertible Bridge Notes ($836,124), settlement of subscription receivable ($65,000) and the exercise of warrants (8,256).
During the three months ended March 31, 2022, cash flows from financing activities totaled $1,318,722. This was solely due to the exercise of warrants for cash.
Critical Accounting Policies and Estimates
The accompanying discussion and analysis of our financial condition and results of operations are based upon our financial statements, which have been prepared in accordance with U.S. generally accepted accounting principles (GAAP). The preparation of financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the reported amounts of assets, liabilities and contingencies as of the date of the financial statements and reported amounts of revenues and expenses during the reporting periods. We evaluate our estimates on an on-going basis. We base our estimates on historical experience and on other assumptions that are believed to be reasonable under the circumstances. However, future events may cause us to change our assumptions and estimates, which may require adjustment. Actual results could differ from these estimates. We have determined that for the periods reported in this Quarterly Annual Report on Form 10-Q the following accounting policies and estimates are critical in understanding our financial condition and results of operations.
| 16 |
Long-lived Assets – Long-lived assets, including property and equipment, and intangible assets are tested for recoverability whenever events or changes in circumstances indicate that their carrying amount may not be recoverable. When such events occur, we compare the sum of the undiscounted cash flows expected to result from the use and eventual disposition of the asset or asset group to the carrying amount of the long-lived asset or asset group. If this comparison indicates that there is an impairment, the amount of the impairment is calculated based on fair value.
Stock-based Compensation – The Company measures the cost of employee and consulting services received in exchange for an award of equity instruments based on the grant-date fair value of the award. The awards issued are valued using a fair value-based measurement method. The resulting cost is recognized over the period during which an employee or consultant is required to provide services in exchange for the award, usually the vesting period.
Off Balance Sheet Arrangements
The Company has not had any off-balance sheet arrangements.
ITEM 3. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
This item is not applicable to the Company because the Company is a smaller reporting company.
ITEM 4. CONTROLS AND PROCEDURES
Evaluation of Disclosure Controls and Procedures
Our management, with the participation of our chief executive officer and chief financial officer, evaluated the effectiveness of our disclosure controls and procedures pursuant to Rule 13a-15 under the Securities Exchange Act of 1934, as amended (the “Exchange Act”), as of March 31, 2023. In designing and evaluating the disclosure controls and procedures, management recognized that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving the desired control objectives. In addition, the design of disclosure controls and procedures must reflect the fact that there are resource constraints and that management is required to apply its judgment in evaluating the benefits of possible controls and procedures relative to their costs.
Based on that evaluation, our chief executive officer concluded as of March 31, 2023 that our disclosure controls and procedures were not effective to ensure that the information required to be disclosed in the reports filed or submitted by us under the Exchange Act was recorded, processed, summarized and reported within the requisite time periods and that such information was accumulated and communicated to our chief executive officer, as appropriate to allow for timely decisions regarding required disclosure.
The Company did not maintain effective disclosure controls and procedures as defined by the framework issued by COSO. Specifically, the Company did not effectively segregate certain accounting duties due to the small size of the Company’s accounting staff. In order to mitigate these material weaknesses regular meetings are held with the audit committee and the audit committee approves all audit functions. If at any time, we determine a new control can be implemented to mitigate these risks at a reasonable cost, it is implemented as soon as possible.
Changes in Internal Control over Financial Reporting
There has been no change in our internal control over financial reporting that occurred in the three months ended March 31, 2023 that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
| 17 |
PART II—OTHER INFORMATION
ITEM 1. LEGAL PROCEEDINGS
None.
ITEM 1A. RISK FACTORS
We are a smaller reporting company and, as a result, are not required to provide the information under this item.
ITEM 2. UNREGISTERED SALES OF EQUITY SECURITIES AND USE OF PROCEEDS.
The offer and sale of the Notes, and shares of common stock issuable upon conversion of the Note (the “Conversion Shares”) have been effected in reliance upon the exemptions for sales of securities set forth in Rule 506(c) under the Securities Act, based upon the following: (a) we have confirmed in a manner consistent with the requirements of Rule 506(c) that each investor is an “accredited investor,” as defined in Rule 501 promulgated under the Securities Act, (b) each investor has represented to us that the investor has such background, education and experience in financial and business matters as to be able to evaluate the merits and risks of an investment in the securities; (c) the investors have been provided with certain disclosure materials and all other information requested with respect to our company; (d) the investors have acknowledge that all Notes and Conversion Shares being purchased are “restricted securities” for purposes of the Securities Act, and agreed to transfer such securities only in a transaction registered under the Securities Act or exempt from registration under the Securities Act; (e) there are restrictions on transfer on the Notes, and any Conversion Shares are subject to restrictions and a legend, providing that the respective security can be transferred only if subsequently registered under the Securities Act or in a transaction exempt from registration under the Securities Act; and (f) a Form D has been filed with respect to the offering.
The Company also had the following unregistered common shares issuances
| - | 1,290 warrants were exercised for $8,256 in cash proceeds. | |
| - | 47,186 warrants were exercised in a cashless exercise whereby 24,886 shares with a value of $9.86 per share were used to settle the exercise price and the remaining 22,300 shares were issued to the warrant holders. |
ITEM 3. DEFAULTS UPON SENIOR SECURITIES
None.
ITEM 4. MINE SAFETY DISCLOSURES
Not applicable.
ITEM 5. OTHER INFORMATION
Utah Division of Securities
On April 23, 2019, the Utah Division of Securities (the “Division”) filed a Notice of Agency Action and an Order to Show Cause before the Division of Securities of the Department of Commerce of the State of Utah against the Company, Jared Bauer (Bauer) and former Board Members (Clark Campbell, Tim Treu, Todd Morgan and Robert Raybould).
| 18 |
In January 2020, the Division issued a Stipulation and Consent Order which set forth the following: 1) the Company agrees to settle the matter with the Division by way of the Stipulation and Consent Order; 2) the Stipulation and Consent Order fully resolves all claims the Division has against the Company pertaining to the Order to Show Cause; 3) the Division, the Company and Bauer, agree to promptly file a stipulation and joint motion to dismiss the Company and Bauer from this administrative action, with respect to Count 1 against the Company and Bauer (the only claim brought against Bauer); 4) In or about April 2014, the Company Board of Directors circulated a consent agreement regarding the issuance of 72,763 Company stock certificates to select members of the Company Board of Directors in connection with “financing services provided” by those members; 5) In or about April 2014, the Company issued stock grants of 27,000 shares to Robert W. Raybould, 2,044 shares to Steve Eror, 7,969 shares to Tim Treu; 24,188 shares to Clark Campbell; and 12,188 shares to Todd Morgan; 6) Subsequent to issuance of those shares, ProLung was informed by counsel of potential consequences for Pro Lung employing unlicensed agents and individuals receiving the shares as compensation directly for sale of securities without a securities license, as opposed to receiving shares as compensation for generalized board service. Subsequently, no further shares were issued as compensation for fundraising. Mr. Eror returned his shares to the Company. However, Raybould, Treu, Campbell and Morgan did not return their shares to the Company. The Company did not disclose the potential licensing violation until on or about December 3, 2018, in its Note Purchase Agreements.
As set forth by the Company in its Form 8-K dated November 27, 2019, Campbell, Treu, Morgan, and Raybould entered into Stipulation and Consent Orders wherein they returned shares of stock to the Company’s treasury and paid fines to the Division of Securities.
On January 9, 2020, the Division entered an order as follows: 1) entering certain Findings and Conclusions by the Division, which ProLung admitted via a Stipulation and Consent Order; 2) ordering ProLung to cease and desist from violating Utah Uniform Securities Act (the “Act”) and to comply with the requirements of the Act in all future business in the state of Utah; 3) ordering ProLung to disclose the contents of the order to investors and prospective investors in all future capital raising efforts and disclosure documents of ProLung; and 4) Ordering ProLung to pay a fine of $55,000 to the Division. Through March 31, 2021, the Company had paid the fine in full.
ITEM 6. EXHIBITS
Exhibit Number | Description | |
| 3.1 | Third Amended and Restated Certificate of Incorporation, as amended by Certificate of Amendment dated October 10, 2017(1) | |
| 3.2 | Amended and Restated By-Laws(1) | |
| 31.1 | Certification Pursuant to Rule 13a-14 and 15d-14 under the Securities Exchange Act of 1934, as Amended* | |
| 32.1 | Certification Pursuant to 18 U.S.C. Section 1350, as Adopted Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002* | |
| 101 INS | Inline XBRL Instance Document* | |
| 101 SCH | Inline XBRL Schema Document* | |
| 101 CAL | Inline XBRL Calculation Linkbase Document* | |
| 101 LAB | Inline XBRL Labels Linkbase Document* | |
| 101 PRE | Inline XBRL Presentation Linkbase Document* | |
| 101 DEF | Inline XBRL Definition Linkbase Document* | |
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document) |
* Filed herewith
(1) Incorporated by reference from our Current Report on Form 8-K filed with the SEC on July 19, 2017.
| 19 |
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the Registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| PROLUNG, Inc. | |||
| May 22, 2023 | By: | /s/ Jared Bauer | |
| Date | Jared Bauer, Chief Executive Officer | ||
| 20 |