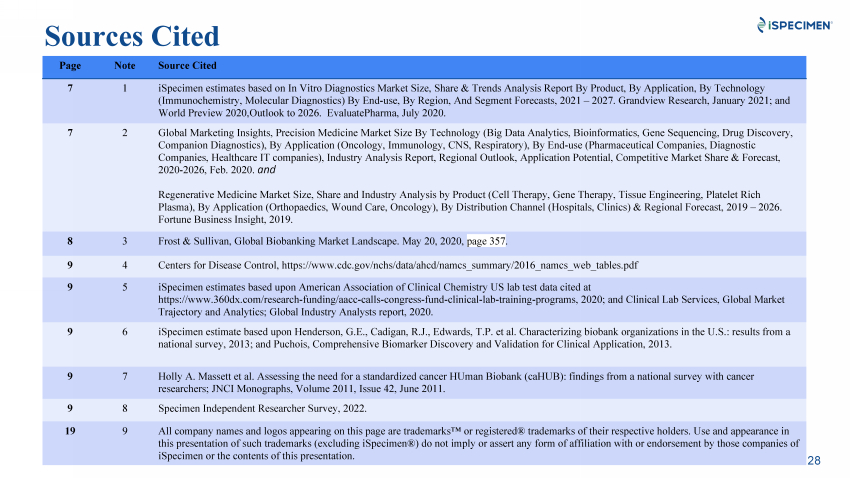
| 28 Page Note Source Cited 7 1 iSpecimen estimates based on In Vitro Diagnostics Market Size, Share & Trends Analysis Report By Product, By Application, By Technology (Immunochemistry, Molecular Diagnostics) By End-use, By Region, And Segment Forecasts, 2021 – 2027. Grandview Research, January 2021; and World Preview 2020,Outlook to 2026. EvaluatePharma, July 2020. 7 2 Global Marketing Insights, Precision Medicine Market Size By Technology (Big Data Analytics, Bioinformatics, Gene Sequencing, Drug Discovery, Companion Diagnostics), By Application (Oncology, Immunology, CNS, Respiratory), By End-use (Pharmaceutical Companies, Diagnostic Companies, Healthcare IT companies), Industry Analysis Report, Regional Outlook, Application Potential, Competitive Market Share & Forecast, 2020-2026, Feb. 2020. and Regenerative Medicine Market Size, Share and Industry Analysis by Product (Cell Therapy, Gene Therapy, Tissue Engineering, Platelet Rich Plasma), By Application (Orthopaedics, Wound Care, Oncology), By Distribution Channel (Hospitals, Clinics) & Regional Forecast, 2019 – 2026. Fortune Business Insight, 2019. 8 3 Frost & Sullivan, Global Biobanking Market Landscape. May 20, 2020, page 357. 9 4 Centers for Disease Control, https://www.cdc.gov/nchs/data/ahcd/namcs_summary/2016_namcs_web_tables.pdf 9 5 iSpecimen estimates based upon American Association of Clinical Chemistry US lab test data cited at https://www.360dx.com/research-funding/aacc-calls-congress-fund-clinical-lab-training-programs, 2020; and Clinical Lab Services, Global Market Trajectory and Analytics; Global Industry Analysts report, 2020. 9 6 iSpecimen estimate based upon Henderson, G.E., Cadigan, R.J., Edwards, T.P. et al. Characterizing biobank organizations in the U.S.: results from a national survey, 2013; and Puchois, Comprehensive Biomarker Discovery and Validation for Clinical Application, 2013. 9 7 Holly A. Massett et al. Assessing the need for a standardized cancer HUman Biobank (caHUB): findings from a national survey with cancer researchers; JNCI Monographs, Volume 2011, Issue 42, June 2011. 9 8 Specimen Independent Researcher Survey, 2022. 19 9 All company names and logos appearing on this page are trademarks™ or registered® trademarks of their respective holders. Use and appearance in this presentation of such trademarks (excluding iSpecimen®) do not imply or assert any form of affiliation with or endorsement by those companies of iSpecimen or the contents of this presentation. Sources Cited |