EXHIBIT 99.1

1
May 19th, 2015, Denver Colorado, 2015
Inhaled AAT Phase II/III
Update of Study Results
Update of Study Results

2
Forward Looking Statement
This presentation is not intended to provide investment or medical advice. It should be noted that some products under development described herein
have not been found safe or effective by any regulatory agency and are not approved for any use outside of clinical trials.
have not been found safe or effective by any regulatory agency and are not approved for any use outside of clinical trials.
This presentation contains forward-looking statements, which express the current beliefs and expectations of Kamada’s management. Such statements
involve a number of known and unknown risks and uncertainties that could cause Kamada's future results, performance or achievements to differ
significantly from the results, performance or achievements expressed or implied by such forward-looking statements. Important factors that could cause
or contribute to such differences include risks relating to Kamada's ability to successfully develop and commercialize its pharmaceutical products, the
progress and results of any clinical trials, the introduction of competing products, the impact of any changes in regulation and legislation that could affect
the pharmaceutical industry, the difficulty of predicting U.S. Food and Drug Administration, European Medicines Agency and other regulatory authority
approvals, the regulatory environment and changes in the health policies and structures of various countries, environmental risks, changes in the
worldwide pharmaceutical industry and other factors that are discussed in Kamada's prospectus related to this offering.
involve a number of known and unknown risks and uncertainties that could cause Kamada's future results, performance or achievements to differ
significantly from the results, performance or achievements expressed or implied by such forward-looking statements. Important factors that could cause
or contribute to such differences include risks relating to Kamada's ability to successfully develop and commercialize its pharmaceutical products, the
progress and results of any clinical trials, the introduction of competing products, the impact of any changes in regulation and legislation that could affect
the pharmaceutical industry, the difficulty of predicting U.S. Food and Drug Administration, European Medicines Agency and other regulatory authority
approvals, the regulatory environment and changes in the health policies and structures of various countries, environmental risks, changes in the
worldwide pharmaceutical industry and other factors that are discussed in Kamada's prospectus related to this offering.
This presentation includes certain non-GAAP financial information, which is not intended to be considered in isolation or as a substitute for, or superior
to, the financial information prepared and presented in accordance with GAAP. The non-GAAP financial measures may be calculated differently from, and
therefore may not be comparable to, similarly titled measures used by other companies. A reconciliation of these non-GAAP financial measures to the
comparable GAAP measures is included in an appendix to this presentation. Management uses these non-GAAP financial measures for financial and
operational decision-making and as a means to evaluate period-to-period comparisons. Management believes that these non-GAAP financial measures
provide meaningful supplemental information regarding Kamada’s performance and liquidity.
to, the financial information prepared and presented in accordance with GAAP. The non-GAAP financial measures may be calculated differently from, and
therefore may not be comparable to, similarly titled measures used by other companies. A reconciliation of these non-GAAP financial measures to the
comparable GAAP measures is included in an appendix to this presentation. Management uses these non-GAAP financial measures for financial and
operational decision-making and as a means to evaluate period-to-period comparisons. Management believes that these non-GAAP financial measures
provide meaningful supplemental information regarding Kamada’s performance and liquidity.
Forward-looking statements speak only as of the date they are made, and Kamada undertakes no obligation to update any forward-looking statement to
reflect the impact of circumstances or events that arise after the date the forward-looking statement was made. You should not place undue reliance on
any forward-looking statement and should consider the uncertainties and risks noted above, as well as the risks and uncertainties more fully discussed
under the heading “Risk Factors” of Kamada’s 2014 Annual Report on Form 20-F filed with the U.S. Securities and Exchange Commission on April 29,
2015.
reflect the impact of circumstances or events that arise after the date the forward-looking statement was made. You should not place undue reliance on
any forward-looking statement and should consider the uncertainties and risks noted above, as well as the risks and uncertainties more fully discussed
under the heading “Risk Factors” of Kamada’s 2014 Annual Report on Form 20-F filed with the U.S. Securities and Exchange Commission on April 29,
2015.

3
Introduction Slide
Program Chair
PROF. ROBERT A. SANDHAUS
Professor of Medicine, National Jewish Health Hospital and University of Colorado
Denver, Division of Pulmonary, Critical Care and Sleep Medicine, Denver, Colorado
Denver, Division of Pulmonary, Critical Care and Sleep Medicine, Denver, Colorado
Panel Members
PROF. KENNETH R. CHAPMAN
Director, Canadian Registry for Alpha1 Anti-trypsin Deficiency
Asthma and Airway Centre, Toronto Western Hospital
University of Toronto , Toronto, Canada
PROF. GERRY MCELVANEY
Beaumont Hospital
Professor of Medicine at RCSI, Dublin, Ireland
PROF. ROBERT .A. STOCKLEY
Lung Investigation Unit, Queen Elizabeth Hospital,
Birmingham University
Birmingham, United Kingdom
DR. JAN STOLK
Leiden, The Netherlands
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015

4
Phase II/III, Double-Blind, Randomized, Placebo-Controlled,
Multicenter, International Study Evaluating the Safety and Efficacy
of Inhaled, Human, Alpha-1 Antitrypsin (AAT) in Alpha-1
Antitrypsin Deficient Patients with Emphysema
Results are presented for the double blind part of the study
Multicenter, International Study Evaluating the Safety and Efficacy
of Inhaled, Human, Alpha-1 Antitrypsin (AAT) in Alpha-1
Antitrypsin Deficient Patients with Emphysema
Results are presented for the double blind part of the study

5
Study Indication
Study Indication
Investigational
Product and
reference therapy
Product and
reference therapy
Investigational
Product and
reference therapy
Product and
reference therapy
Study Design
Study Design
● Treatment of alpha-1 antitrypsin
deficiency in subjects with clinically
demonstrable emphysema.
deficiency in subjects with clinically
demonstrable emphysema.
● Aerosolized (inhaled) human (plasma-derived)
AAT at 80 mg, 4ml inhalation X 2/day.
AAT at 80 mg, 4ml inhalation X 2/day.
● The placebo comprises the non-active
ingredients of the AAT preparation.
ingredients of the AAT preparation.
● eFlow® inhalation device- PARI Pharma GmbH.
● Phase II-III ; Double-blind; Randomized
placebo-controlled; Multicenter, intrl’ study.
placebo-controlled; Multicenter, intrl’ study.
● 168 subjects, Randomized 1:1 AAT; placebo
● 50 weeks double blind ; 50 weeks OLE
● Trial designed in accordance with EMA
scientific advise/ protocol assistance and EU
draft guidance for COPD trials
scientific advise/ protocol assistance and EU
draft guidance for COPD trials
Study Information
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015
6
Site in study
DSMB members
Sites: UK, SC, IR, SW, DK, CA, NL, GR
DSMB: IT, USA, ES
Study Information - Sites
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015

7
Primacy
Largest Study
First of its kind,
largest, IH AAT study
largest, IH AAT study
E-Diary
Use of e-Diary to
collect robust natural
history and efficacy/
safety data
collect robust natural
history and efficacy/
safety data
Efficacy
First controlled
randomized trial to
demonstrate lung
function efficacy
randomized trial to
demonstrate lung
function efficacy
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015

8
Main Inclusion / Exclusion Criteria
Inclusion
Inclusion
Exclusion
Exclusion
1. Adults with AAT deficiency
2. FEV1/FVC <70% and FEV1 < 80%
3. At least two exacerbations in the last 18 months from
screening.
screening.
4. AAT deficient subjects who are either naïve (not receiving IV
augmentation therapy) or AAT deficient subjects receiving IV
augmentation therapy.
augmentation therapy) or AAT deficient subjects receiving IV
augmentation therapy.
1. History of lung transplant; Any lung surgery within the past
two years.
two years.
2. Active smoking during the last 12 months from screening
date.
date.
3. IgA Deficiency
4. History of life threatening allergy, anaphylactic reaction, or
systemic response to human plasma derived products.
systemic response to human plasma derived products.
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015

9
Ⅱ
● The time from
randomization to the
first event-based
exacerbation with a
severity of moderate
or severe.
randomization to the
first event-based
exacerbation with a
severity of moderate
or severe.
● Time to first event-
based exac. (mild,
moderate or severe)
based exac. (mild,
moderate or severe)
● Severity of the first
event-based exac.
event-based exac.
● Rate of event-based
exac.
exac.
Primary
Primary
Secondary
Secondary
Safety
Safety
Study Endpoints
● Adverse Events
● Lung Function
● Vital Signs
● Physical Exam
● ECG
● Laboratory Evaluations
Ⅰ
Regulatory guidance as to efficacy indicated:
Importance of secondary endpoint including rate and severity of exacerbation as well as
review of totality of the data arising from the trial
review of totality of the data arising from the trial
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015

10
Analysis of the data revealed: primary
endpoint was not met
endpoint was not met
Lung function analysis and first
exacerbation severity †† statistical
significant changes
exacerbation severity †† statistical
significant changes
Kamada approached EMA and
presented the data
presented the data
EMA confirmed for this ODD
review of post-hoc analysis and
totality of the data irrespective
of not meeting primary endpoint
totality of the data irrespective
of not meeting primary endpoint
What has changed?
1
2
3
4
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015

11
Lung
Function
Function
Exacerbation
symptoms
symptoms
Safety
Analysis Strategy
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015

12

13
AAT (N=85) | Placebo (N=83) | |
Males Females | 51 (60.0%) 34 (40.0%) | 49 (59.0%) 34 (41.0%) |
Mean age ± SD (years) Age ≥ 60 years | 56.5 ± 9.9 38 (44.7%) | 54.4 ± 10.3 26 (31.3%) |
Race: Caucasian | 79 (100%) | 75 (100%) |
BMI (kg/m2): mean ± SD BMI <20 | 25.8 ± 4.6 8 (9.4%) | 26.3 ± 5.5 4 (4.8%) |
Oxygen users | 18 (21.2%) | 10 (12.0%) |
FEV1 (L): mean ± SD | 1.32 ± 0.49 | 1.33 ± 0.53 |
FEV1% (%): mean ± SD | 42.8 ± 14.8 | 41.8 ± 14.7 |
DLCO (mMol/min/kPa): mean ± SD | 4.23 ± 1.61 | 4.59 ± 1.96 |
Baseline Characteristics
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015
14
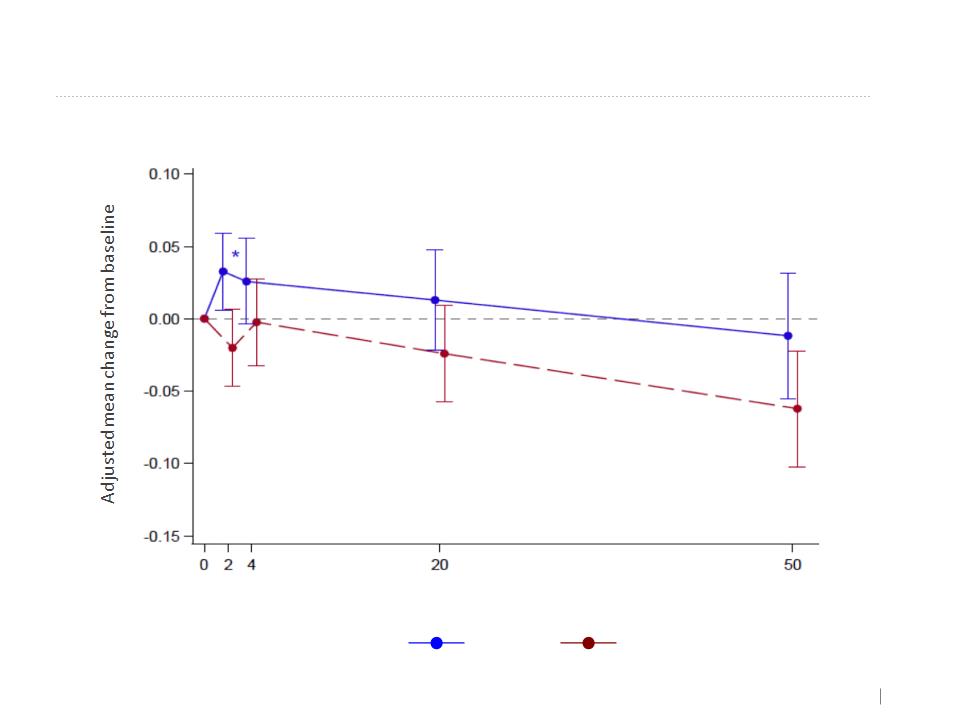
Spirometry Measures (MMRM)
FEV1 (L) - MMRM
Week
Placebo
AAT
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015
Overall effect
+15ml
+15ml
Overall effect
-27ml
-27ml
P=0.0268
15
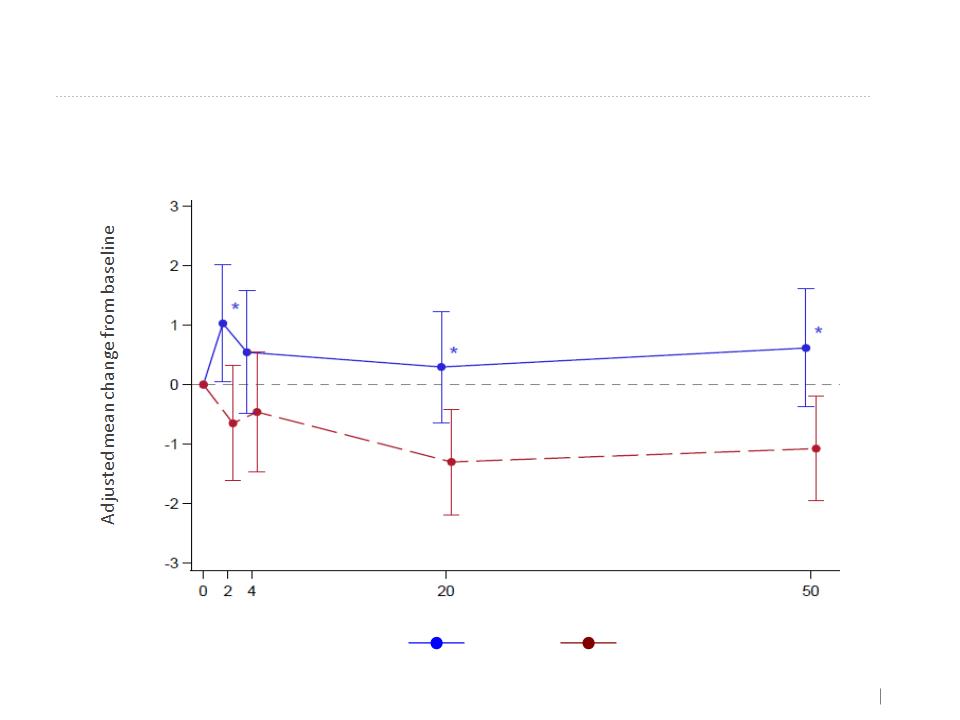
Spirometry Measures (MMRM)
FEV1/SVC - MMRM
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015
Placebo
AAT
Overall effect
+0.623
+0.623
Overall effect
-0.8715
-0.8715
P=0.0074

16
Lung Function | Least Squares Means (SE) (Changes at Week 50 from Baseline) | P-Value (Changes at Week 50) | Least Squares Means (SE) (overall treatment effect) | P-Value (Overall Effect) | ||
AAT (N= 84) | Placebo (N= 81) | AAT (N= 84) | Placebo (N= 81) | |||
FEV1 (L) | -12mL -0.01183 (0.02196) | -62mL -0.06216 (0.02036) | 0.0956 | +15mL 0.01503 (0.01338) | -27mL -0.02718 (0.01322) | 0.0268 |
FEV1 (% of predicted) | -0.1323 (0.6649) | -1.6205 (0.6140) | 0.1032 | 0.5404 (0.4451) | -0.6273 (0.4425) | 0.0658 |
FEV1/SVC (%) | 0.6183 (0.5015) | -1.0723 (0.4455) | 0.0132 | 0.6230 (0.3931) | -0.8715 (0.3804) | 0.0074 |
SE in brackets
MMRM = Mixed Model Repeated Measure
Spirometry Measures (MMRM)
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015

17
Lung Function | Least Squares Means (SE) (Changes at Week 50 from Baseline) | P-Value (Changes at Week 50) | Least Squares Means (SE) (overall treatment effect) | P-Value (Mixed Linear Model - Overall Treatment Effect) | ||
AAT (N= 84) | Placebo (N= 81) | AAT (N= 84) | Placebo (N= 81) | |||
DLCO | -0.2704 (0.07713) | -0.3054 (0.07182) | 0.7407 | -0.2011 (0.05585) | -0.1640 (0.05577) | 0.6401 |
DLCO (% of predicted) | -2.9103 (0.9058) | -3.5785 (0.8459) | 0.5920 | -2.1459 (0.6721) | -1.8723 (0.6734) | 0.7748 |
DLCO/VA | -0.02858 (0.01359) | -0.02464 (0.01299) | 0.8349 | -0.02672 (0.01061) | -0.00953 (0.01071) | 0.2580 |
DLCO/VA (% of predicted) | -2.1951 (0.9686) | -1.8049 (0.9232) | 0.7720 | -2.0143 (0.7777) | -0.7094 (0.7851) | 0.2415 |
SE in brackets
No Difference Between Groups
Diffusing Capacity (MMRM)
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015

19
2
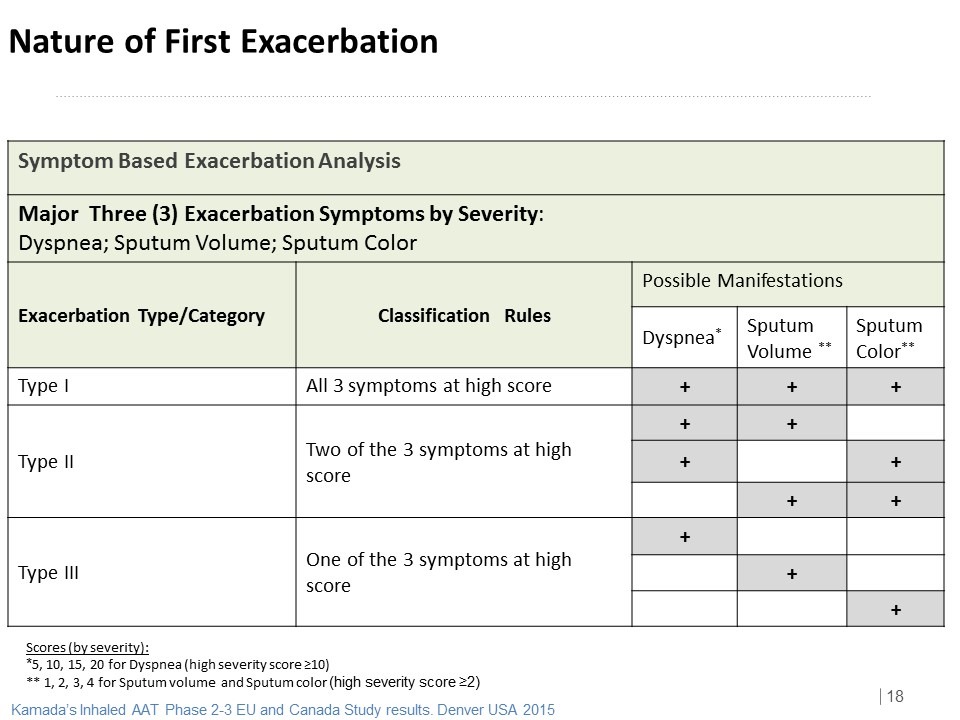
ITT | N (%) | P Value | |
AAT | Placebo | ||
Type/Category | N=85 | N=83 | |
Type I | 16 (18.8%) | 26 (31.3%) | 0.0614 |
Type II | 23 (27.1%) | 12 (14.5%) | 0.0444 |
Type III | 34 (40.0%) | 33 (39.8%) | 0.9746 |
None | 12 (14.1%) | 12 (14.5%) | 0.9498 |
AAT may change the nature of the Exacerbation (Potential change from Type I to Type II)
Type I+II è Type I exacerbation stands for 41% within total of type I+ II exacerbations for AAT
group vs. 68% for placebo group.
Type I+II è Type I exacerbation stands for 41% within total of type I+ II exacerbations for AAT
group vs. 68% for placebo group.
Nature of the First Exacerbation
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015

20
Symptom Score MMRM Analysis of First (Types I+II+III)
Exacerbation Severity for each major Symptom
(during 0-10 and 0-14 days of the exacerbation event)
Exacerbation Severity for each major Symptom
(during 0-10 and 0-14 days of the exacerbation event)
*Adjustment to age, oxygen, BMI, Country, Treatment Duration
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015
Symptom | Exac. Type | Days | MMRM Least Square Means | P-Value* | |
AAT N=73 | Placebo N=71 | ||||
Dyspnea | All Types (I, II, III) | 0-10 | 11.9464 | 12.2548 | 0.0243 |
0-14 | 11.5803 | 11.7832 | 0.0817 | ||
Sputum Volume | 0-10 | 1.2748 | 1.3837 | 0.0334 | |
0-14 | 1.2367 | 1.3206 | 0.0595 | ||
Sputum Color | 0-10 | 2.1566 | 2.0137 | 0.0502 | |
0-14 | 2.0240 | 1.8393 | 0.0032 | ||
During first Exacerbation,
AAT group improves significantly Dyspnea and Sputum volume symptoms
AAT group improves significantly Dyspnea and Sputum volume symptoms
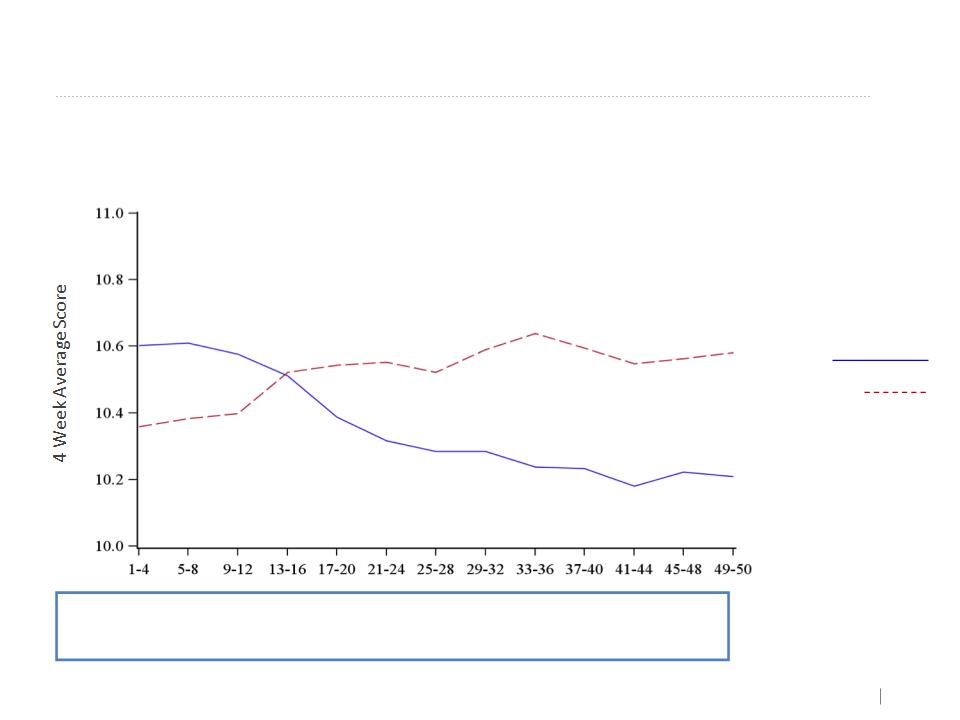
21
Continuous Symptom Score - Dyspnea
Dyspnea 4 Week Moving Average Graphs
Week
Treatment group
AAT
Placebo
Improvement trend in favor of AAT group
No statistical significance
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015

22
Well Being 4 Week Moving Average Graphs
Week
AAT
Placebo
Improvement trend in favor of AAT group
No statistical significance
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015
Continuous Symptom Score - Well Being
Treatment group

23
Month | AAT | Placebo |
1 | 1.75 | 1.14 |
2 | 0.78 | 0.69 |
3 | 0.96 | 0.71 |
4 | 0.87 | 0.53 |
5 | 0.71 | 0.9 |
6 | 0.78 | 0.47 |
7 | 0.63 | 0.51 |
8 | 0.6 | 0.59 |
9 | 0.63 | 0.74 |
10 | 0.78 | 0.53 |
11 | 0.37 | 0.51 |
12 | 0.83 | 0.71 |
Safety: Mean AE per Patient by Month
month
• There were no AE indicating immunogenicity
and/or clinical indication of bronchospasms
and/or clinical indication of bronchospasms
• No specific AE pattern
• Most AEs relate to underlying disease
• No Anaphylactic reactions
• Nature of AEs was similar between groups.
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015

24
Strengths vs. Constraints
• No improvement in
time to first
moderate/severe exac.
time to first
moderate/severe exac.
• No improvement in
rate
rate
• Spirometry efficacy
• Exacerbation nature effect
• Safe and tolerable
• Primacy in clinical efficacy
• Unmet medical need
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015

25
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015
In Summary
1. Efficacy in lung function (statistically significant)
2. Change in the nature of exacerbations(reduction in number of Type 1-
exacerbations (trend) and reduction in dyspnea score (statistically significant)
exacerbations (trend) and reduction in dyspnea score (statistically significant)
3. Safe and tolerabledrug
4. Orphan designated drug
5. Unmet patient need- Clinical primacy in efficacy data for IH AAT and
AATD in general
AATD in general

26
• Compilation of an MAA dossier
• EMA submission (centralized
procedure) end of 2015
procedure) end of 2015
EMA -EU Front
• Approach US-FDA with results
in H2 2015to obtain guidance
on the clinical/ regulatory
pathway for licensing the IH
AAT by Kamada in the US.
in H2 2015to obtain guidance
on the clinical/ regulatory
pathway for licensing the IH
AAT by Kamada in the US.
FDA -US Front
Moving Forward
Kamada is committed to the AATD patient community to bring the IH AAT
into the market place and provide an adequate, safe and efficacious answer
to current unmet medical need of these orphan patients.
into the market place and provide an adequate, safe and efficacious answer
to current unmet medical need of these orphan patients.
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015

27
To the entire
Kamada team
Kamada team
To our DSMB
Dr. Marc Miravitlles
Dr. Maurizio Luisetti
Prof. Victor DeGruttola
Prof. Victor DeGruttola
Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015
To our study investigators
Dr. Jan Stolk
Prof. Rob Stockley
Prof. Kenneth Chapman
Prof. Gerry McElvaney
Prof. William McNee
Dr. Eeva Piitulainen
Prof. Dr. Claus Vogelmeier
Dr. Kevin Elwood
Dr. Abboud Raja
Dr. Niels Seersholm
Dr. Michael Runold
Prof. Nick Hopkinsons
To our study nurses &
coordinators
coordinators
To Dr. Pablo Fernandez,
our Medical advisor
our Medical advisor
To AIR Group
To our study CRO,
QP, labs, logistics
and other
vendors
QP, labs, logistics
and other
vendors
SPECIAL THANKS TO…

28
Thank you