EXHIBIT 99.1

1
NASDAQ: KMDA JUNE 2015
KAMADA INVESTOR PRESENTATION
2
Investor Presentation | June 2015
Forward Looking Statement
This presentation is not intended to provide investment
or medical advice. It should be noted that some
products under development described herein have not
been found safe or effective by any regulatory agency
and are not approved for any use outside of clinical
trials.
or medical advice. It should be noted that some
products under development described herein have not
been found safe or effective by any regulatory agency
and are not approved for any use outside of clinical
trials.
This presentation contains forward-looking statements,
which express the current beliefs and expectations of
Kamada’s management. Such statements involve a
number of known and unknown risks and uncertainties
that could cause Kamada's future results, performance
or achievements to differ significantly from the results,
performance or achievements expressed or implied by
such forward-looking statements. Important factors that
could cause or contribute to such differences include
risks relating to Kamada's ability to successfully develop
and commercialize its pharmaceutical products, the
progress and results of any clinical trials, the
introduction of competing products, the impact of any
changes in regulation and legislation that could affect
the pharmaceutical industry, the difficulty of predicting
U.S. Food and Drug Administration, European
Medicines Agency and other regulatory authority
approvals, the regulatory environment and changes in
the health policies and structures of various countries,
environmental risks, changes in the worldwide
pharmaceutical industry and other factors that are
discussed in Kamada's prospectus related to this
offering.
which express the current beliefs and expectations of
Kamada’s management. Such statements involve a
number of known and unknown risks and uncertainties
that could cause Kamada's future results, performance
or achievements to differ significantly from the results,
performance or achievements expressed or implied by
such forward-looking statements. Important factors that
could cause or contribute to such differences include
risks relating to Kamada's ability to successfully develop
and commercialize its pharmaceutical products, the
progress and results of any clinical trials, the
introduction of competing products, the impact of any
changes in regulation and legislation that could affect
the pharmaceutical industry, the difficulty of predicting
U.S. Food and Drug Administration, European
Medicines Agency and other regulatory authority
approvals, the regulatory environment and changes in
the health policies and structures of various countries,
environmental risks, changes in the worldwide
pharmaceutical industry and other factors that are
discussed in Kamada's prospectus related to this
offering.
This presentation includes certain non-GAAP financial
information, which is not intended to be considered in
isolation or as a substitute for, or superior to, the
financial information prepared and presented in
accordance with GAAP. The non-GAAP financial
measures may be calculated differently from, and
therefore may not be comparable to, similarly titled
measures used by other companies. A reconciliation of
these non-GAAP financial measures to the comparable
GAAP measures is included in an appendix to this
presentation. Management uses these non-GAAP
financial measures for financial and operational decision
-making and as a means to evaluate period-to-period
comparisons. Management believes that these non-
GAAP financial measures provide meaningful
supplemental information regarding Kamada’s
performance and liquidity.
information, which is not intended to be considered in
isolation or as a substitute for, or superior to, the
financial information prepared and presented in
accordance with GAAP. The non-GAAP financial
measures may be calculated differently from, and
therefore may not be comparable to, similarly titled
measures used by other companies. A reconciliation of
these non-GAAP financial measures to the comparable
GAAP measures is included in an appendix to this
presentation. Management uses these non-GAAP
financial measures for financial and operational decision
-making and as a means to evaluate period-to-period
comparisons. Management believes that these non-
GAAP financial measures provide meaningful
supplemental information regarding Kamada’s
performance and liquidity.
Forward-looking statements speak only as of the date
they are made, and Kamada undertakes no obligation to
update any forward-looking statement to reflect the
impact of circumstances or events that arise after the
date the forward-looking statement was made. You
should not place undue reliance on any forward-looking
statement and should consider the uncertainties and
risks noted above, as well as the risks and uncertainties
more fully discussed under the heading “Risk Factors” of
Kamada’s 2014 Annual Report on Form 20-F filed with
the U.S. Securities and Exchange Commission on April
29, 2015.
they are made, and Kamada undertakes no obligation to
update any forward-looking statement to reflect the
impact of circumstances or events that arise after the
date the forward-looking statement was made. You
should not place undue reliance on any forward-looking
statement and should consider the uncertainties and
risks noted above, as well as the risks and uncertainties
more fully discussed under the heading “Risk Factors” of
Kamada’s 2014 Annual Report on Form 20-F filed with
the U.S. Securities and Exchange Commission on April
29, 2015.
3
Investor Presentation | June 2015
Kamada Overview
1. Rapidly Growing, Globally Positioned Biopharmaceutical
Company Focused on Orphan Diseases and Plasma-
Derived Protein Therapeutics
Company Focused on Orphan Diseases and Plasma-
Derived Protein Therapeutics
● Revenue and profitability with 10 marketed products
● $100M of revenues expected by 2017
2. Leader in the Development of Alpha-1 Antitrypsin (“AAT”)
Products Globally and Specific Immunoglobulin
Products Globally and Specific Immunoglobulin
● Developed and obtained FDA Approval for the first and only liquid,
ready-to-use intravenous AAT product, Glassia® for AAT deficiency
ready-to-use intravenous AAT product, Glassia® for AAT deficiency
● Selling Glassia® in selected emerging markets globally and through
Baxalta (formerly Baxter) collaboration in the U.S.
Baxalta (formerly Baxter) collaboration in the U.S.
● KamRAB for rabies prophylaxis (U.S. Phase III complete) to be
launched in U.S. through collaboration with Kedrion
launched in U.S. through collaboration with Kedrion
3. Attractive Pipeline for 5 Orphan Indications including
● AAT to treat type-1 diabetes (Phase II/III)
● AAT to treat Graft-vs-Host Disease (GVHD) (Phase I/II)
● Novel Inhaled AAT for AATD (EU Phase III completed)
● Pursuing approval in EU, MAA submission end 2015
● Ongoing Phase II in the U.S.; pathway to be discussed with FDA
4. Fully Integrated Manufacturing and Distribution
Notes: 1. As of March 31, 2015 2. Market data as of May 13, 2015
● Founded in 1990. Based in Weizmann Science Park,
Israel
Israel
● Employees: ~300 (1)
● Listed on NASDAQ since 2013 & TASE since 2005
(KMDA)
(KMDA)
● Current market capitalization: ~$157MM (2)
● Cash, cash equivalents and ST investments: $50MM(1)
● Total Debt: $7.5MM(1)
Key Statistics
4
Investor Presentation | June 2015
Integrated, Efficient, Scalable Platform Technology
Fully-Invested
Manufacturing Facility &
Marketed Products
Manufacturing Facility &
Marketed Products
● FDA approved since
2010
2010
● cGMP compliant
● Multiple countries'
certifications (U.S.,
Brazil, Israel, Mexico,
Russia)
certifications (U.S.,
Brazil, Israel, Mexico,
Russia)
● State-of-the-art clean
room environment
room environment
● Located in Beit Kama,
Israel
Israel
Proprietary, Innovative
and Patented Technology
Platform
and Patented Technology
Platform
● Patent protected:
Chromatography-based
purification process
Chromatography-based
purification process
● Enables high purity
extraction
extraction
● Ready-to-use, liquid and
stable specialty protein
therapeutics (AAT,
Albumin, Transferrin and
many others)
stable specialty protein
therapeutics (AAT,
Albumin, Transferrin and
many others)
● Enables production of
almost any human plasma
-derived specific
immunoglobulins
almost any human plasma
-derived specific
immunoglobulins
Benefits
● Enables manufacturing of
plasma-derived protein
therapeutics with differentiated
product profiles
plasma-derived protein
therapeutics with differentiated
product profiles
● Infrastructure in place to meet
future pipeline product demand
future pipeline product demand
● Expandable product platform to
additional territories and
indications
additional territories and
indications
+
=
5
Respiratory
Glassia®
Immunoglo-
bulin
bulin
KamRAB™
KamRho (D) IM
KamRho (D) IV
Other
Products
Heparin Lock Flush
Kamacaine 0.5%
Human Transferrin
Respiratory
Bramitob
Foster
Immunoglo-
bulins
bulins
IVIG 5%
Varitect
Hepatect CP
Megalotect
Zutectra
Critical
Care
Heparin sodium
Injection
Albumin
Other
Factor VIII
Factor IX
Proprietary
Products
Products
Segment
2014
Revenue:
$44MM
$44MM
Distribution
Segment
Segment
2014
Revenue:
$27MM
Alpha-1 Antitrypsin (human)
Anti-rabies immunoglobulin (human)
Rho(D) immunoglobulin (human)
Rho(D) immunoglobulin (human)
Heparin sodium
Bupivacaine HCl
Transferrin (Diagnostic grade)
Tobramycin
Beclomethasone+Formoterol
Gamma globulins (IgG) (human)
Varicella zoster immunoglobulin (human)
Hepatitis B immunoglobulin (human)
CMV immunoglobulin (human)
Hepatitis B Immunoglobulins S.C
Human serum Albumin
Coagulation Factor VIII (human)
Coagulation Factor IX (human)
Snake Antiserum
Anti-snake venom
Growing Proprietary Products Segment Through Glassia®
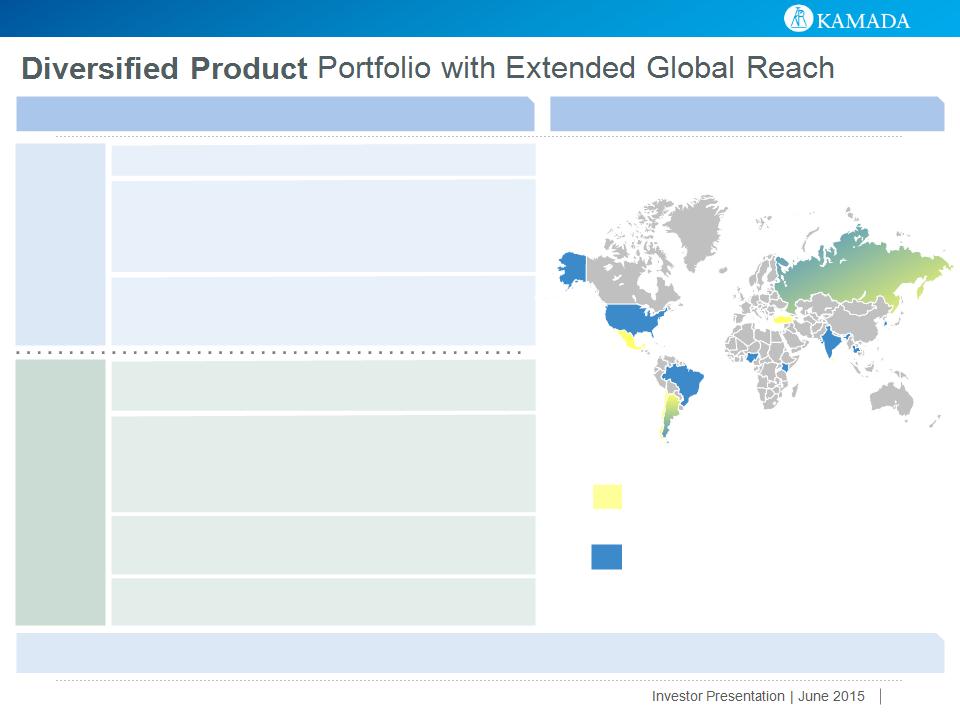
United States
Mexico
El Salvador
Brazil
Argentina
Slovenia
Croatia
Nigeria
Kenya
India
Thailand
South Korea
Russia
Turkey
Israel*
Chile
Sri Lanka
Diverse Portfolio of Predominantly Plasma-Derived Protein Therapeutics
Global Presence with Exposure to Emerging Markets
*Kamada distributes products directly in
Israel through its own salesforce
Israel through its own salesforce
Countries where Kamada currently sells certain
of its Proprietary Products through strategic or
distributor partnerships
of its Proprietary Products through strategic or
distributor partnerships
Countries where Kamada has received regulatory
approvals for certain of its Proprietary Products
approvals for certain of its Proprietary Products
Heparin sodium
6
Glassia® is a Differentiated Product
Key Product Advantages
AATD (IV) Product Sales
AATD (IV) Product Sales
W/O Milestone Revenues ( in MM$)
W/O Milestone Revenues ( in MM$)
● Glassia® is the first and only liquid, ready-to-use,
IV plasma-derived AAT product
IV plasma-derived AAT product
● No reconstitution required, reducing risk of
contamination and infection and reducing
treatment time
contamination and infection and reducing
treatment time
● Potentially reduced risk for adverse event and/or
allergic reaction due to the absence of
preservatives and stabilizing agent(s)
allergic reaction due to the absence of
preservatives and stabilizing agent(s)
● Glassia® is sold in the U.S. by Baxalta (formerly
Baxter), a leading plasma therapeutics company
Baxter), a leading plasma therapeutics company
● Significantly faster infusion rate was recently
(2014) approved by the U.S. FDA
(2014) approved by the U.S. FDA
Glassia® is sold in 8 countries,
with majority of sales in the U.S.
7
Investor Presentation | June 2015
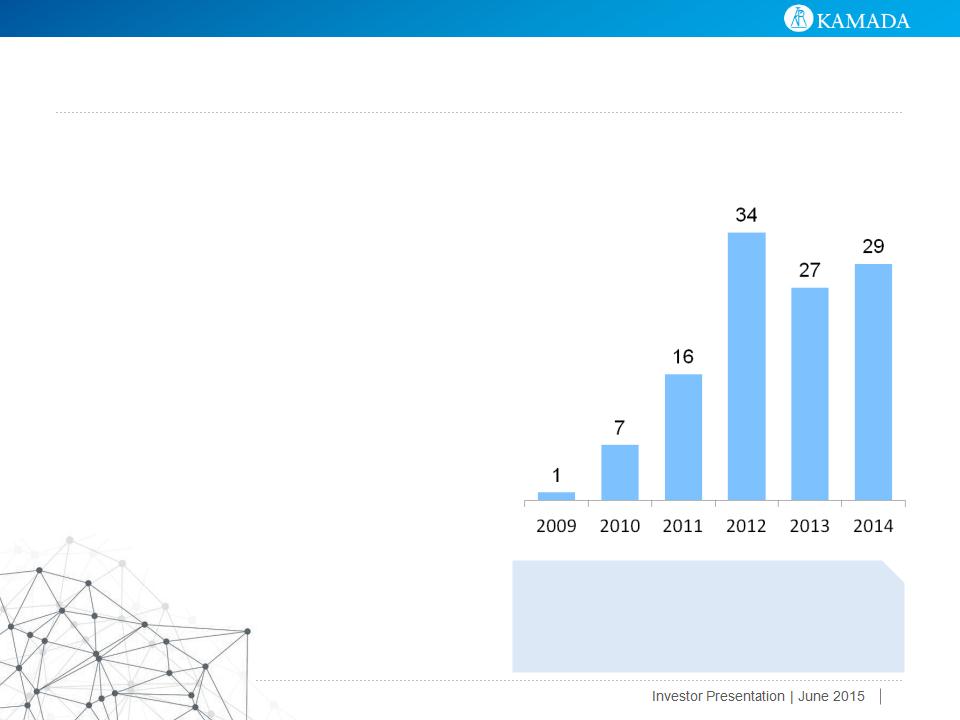
Growth of Glassia® Driven by Strategic Partnership
with Baxalta (formerly Baxter)
with Baxalta (formerly Baxter)
● Commencement: Sales to Baxalta commenced in September 2010
● Agreements: distribution, technology license and fraction IV supply
● Product: AAT IV (Glassia®), including future AAT IV
● Territories: U.S., Canada, Australia and New Zealand
● Milestone and upfront revenues: $45MM ($34.5MM received)
● Agreement recently extended:
● Baxalta to distribute Glassia® produced by Kamada through 2017
● Minimum revenues of $191MM through 2017 ($121MM already recognized through 12/31/2014)
● Royalties from sales of Glassia® produced by Baxalta expected from 2018
8
Investor Presentation | June 2015
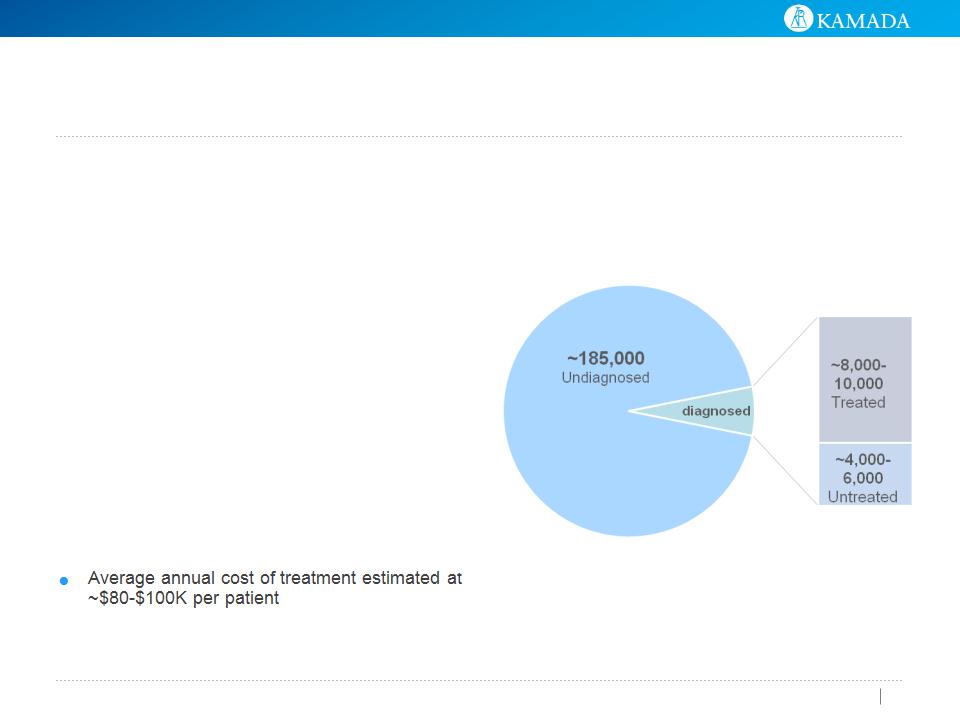
Significant Opportunity to
Expand the AATD Market
Expand the AATD Market
● Patients suffering from AAT Deficiency
(“AATD”) remain under-identified and under-
treated
(“AATD”) remain under-identified and under-
treated
● Only ~6% of cases treated in the U.S. and
~2% in EU
~2% in EU
● Simple blood test for diagnosis expected to
impact demand
impact demand
● Greater AAT use in Europe and other
geographies could further accelerate market
growth
geographies could further accelerate market
growth
● Chronic therapy creates sustainable product
opportunity
opportunity
Sustainable Market with Strong
Growth Potential
Growth Potential
AATD Prevalence: ~200,000
Yet Fewer than 5% of Potential Patients in
the U.S. and Europe are Treated
the U.S. and Europe are Treated
Source : Alpha 1 Foundation, MRB and Company estimates
9
Investor Presentation | June 2015
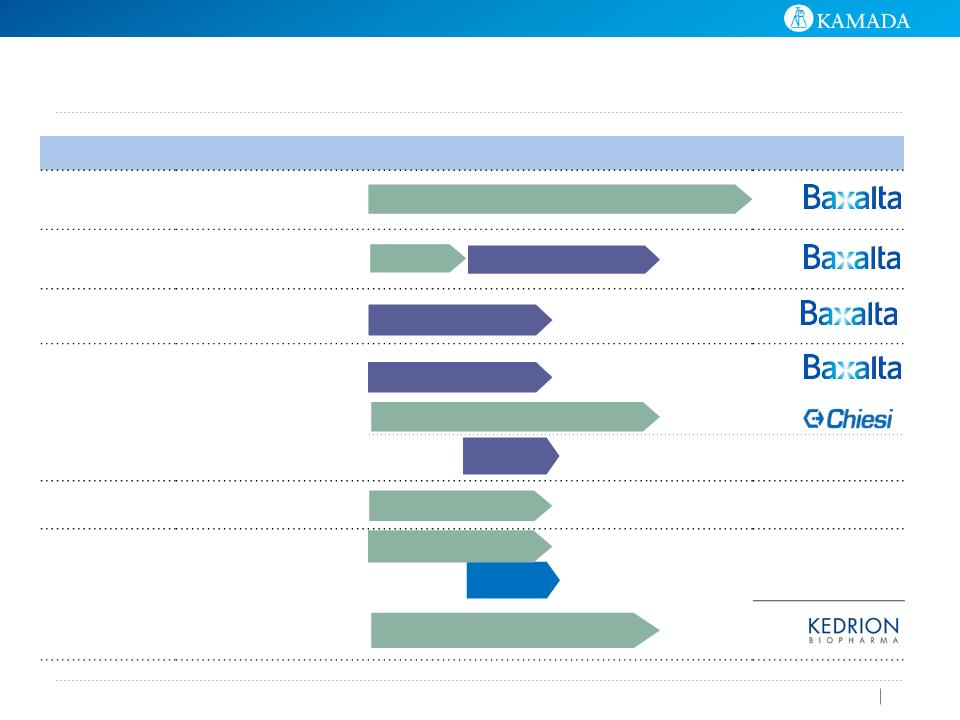
KamRAB: Human Rabies Immune Globulin
U. S. Opportunity
● Strategic agreement with Kedrion S.p.A for the clinical development and
marketing of KamRAB in the U.S.
marketing of KamRAB in the U.S.
● U.S. Phase II/III clinical trial completed, with data expected during 2015
● Expect to file Biological License Application with the FDA in beginning of 2016
● U.S. launch expected by 2016/17
● In the U.S., there are ~40,000 post-exposure prophylaxis treatments
administered each year, representing an ~$100 million market opportunity
administered each year, representing an ~$100 million market opportunity
● Currently, only one significant provider of anti-rabies immunoglobulin exists
Out of U.S.: Product marketed by Kamada in 10 countries
● The product has been marketed since 2003
● WHO estimates ~10 million people worldwide require medical treatment
against rabies each year after being bitten by an animal suspected of rabies
infection
against rabies each year after being bitten by an animal suspected of rabies
infection
Kamada’s human rabies immune globulin is a post-exposure
prophylaxis (PEP) for rabies.
prophylaxis (PEP) for rabies.
10
Investor Presentation | June 2015
High Value Pipeline Focused on Orphan Indications
Product | Indication | Phase I | Phase II | Phase III | Market | Partners |
Intravenous AAT | AAT Deficiency | U.S.: | ||||
D1-AAT (IV) | Type 1 Diabetes* | U.S.: | ||||
G1-AAT (IV) | GVHD* | U.S.: | ||||
L1-AAT (IV) | Lung transplant | U.S.: | ||||
Inhaled AAT | AAT Deficiency* | EU: | ||||
B1-AAT (IH) | Bronchiectasis* | |||||
C1-AAT (IH) | Cystic Fibrosis* | |||||
KamRAB (IM) | Prophylaxis for Rabies | U.S.: | ||||
Phase III Completed
(LPO)
Ph II/III In Process
FDA Approved (2010)
Completed
Ph I/II In Process
U.S.: IND
Approved
U.S.: Ph II
In Process
EU: Completed
Completed
Completed
* Orphan drug designation
Ph I/II in Initiation

11
Inhaled AAT to Treat AATD
12
Investor Presentation | June 2015
Inhaled AAT for AATD: Completed Pivotal Phase II/III
Trials in Europe and on going Phase II in the U.S.
Trials in Europe and on going Phase II in the U.S.
Phase II / III EU
Phase II U.S.
Description
● Randomized; Over 160 AATD subjects, majority
are treatment naïve
are treatment naïve
● Double blind, placebo controlled, randomized
● Multi center international study: Western EU (UK,
IR, SC, SW, NL, DK, GR) and Canada
IR, SC, SW, NL, DK, GR) and Canada
● 80% power to detect a difference between the two
groups at 1 year
groups at 1 year
● Powered for 20% difference between the two
groups
groups
● Power is based on number of events collected
during the study
during the study
● Randomized; Sample size of ~36-40 subjects
● Double blind, placebo controlled, randomized
Route &
Dosage
Form
Dosage
Form
● Inhalation of human AAT, 160mg total, twice
daily ~10-15 minutes; eFlow® device
daily ~10-15 minutes; eFlow® device
● Inhalation of human AAT; two dosage groups
(80mg and 160mg daily); eFlow® device
(80mg and 160mg daily); eFlow® device
Clinical
Endpoints
Endpoints
● Exacerbation events (Primary: time to first
moderate/severe, Secondary (among others):
rate, severity of first event; Lung Function)
moderate/severe, Secondary (among others):
rate, severity of first event; Lung Function)
● Primary: Concentration of AAT in ELF
● Secondary: safety and tolerability, Concentration
AAT in serum, ELF inflammatory analytes
AAT in serum, ELF inflammatory analytes
Duration
● 50 wk treatment in DB period; daily treatment
● 50 wk open label extension ; daily treatment
● Study completed
● 12 weeks double blind +
● 12 weeks open label extension
● Study initiated in 1Q2014
13
Inhaled AAT Phase II/III Trial:
Summary of the Results
Summary of the Results
Results demonstrate:
1.Primary and secondary endpoints didn’t demonstrate statistical significant
difference.
difference.
2.Efficacy in lung function (statistically significant)
3.Change in the nature of exacerbations (reduction in number of Type 1-
exacerbations (trend) and reduction in dyspnea score (statistically significant) for
first exacerbation
exacerbations (trend) and reduction in dyspnea score (statistically significant) for
first exacerbation
4.Safe and tolerable drug
Submission of MAA is planned by end of 2015 on the basis of:
1.Orphan designated drug
2.Demonstrated efficacy in lung function
3.Unmet patient need - Clinical primacy in efficacy data for IH AAT and AATD in
general
general
4.EMA confirmed review of post-hoc analysis and totality of the data irrespective of
not meeting primary endpoint
not meeting primary endpoint
5.Pre-existing cases of approved drugs of similar nature (ODD, post hoc analyses
and existing patient un met need)
and existing patient un met need)
Investor Presentation | June 2015
14
Investor Presentation | June 2015
Inhaled AAT Phase II/III Trial Results:
Spirometry Measures (MMRM*)
Spirometry Measures (MMRM*)
*MMRM = Mixed Model Repeated Measure, SE in brackets
Lung Function | Least Squares Means (SEM) (Changes at Week 50 from Baseline) | P-Value* (Changes at Week 50) | Least Squares Means (SEM) (overall treatment effect) | P-Value* (Overall Effect) | ||
AAT (N= 84) | Placebo (N= 81) | AAT (N= 84) | Placebo (N= 81) | |||
FEV1 (L) | -12mL -0.01183 (0.02196) | -62mL -0.06216 (0.02036) | 0.0956 | +15mL 0.01503 (0.01338) | -27mL -0.02718 (0.01322) | 0.0268 |
FEV1 (% of predicted) | -0.1323 (0.6649) | -1.6205 (0.6140) | 0.1032 | 0.5404 (0.4451) | -0.6273 (0.4425) | 0.0658 |
FEV1/SVC (%) | 0.6183 (0.5015) | -1.0723 (0.4455) | 0.0132 | 0.6230 (0.3931) | -0.8715 (0.3804) | 0.0074 |
15
Investor Presentation | June 2015
In the Words of the Key Opinion Leaders
“The study results demonstrated primarily that the overall treatment effect on lung
functions, is of significant clinical value. This study is the first study ever that is indicative
of inhaled AAT’s ability to potentially reduce lung inflammation as expressed by its
preservation of lung function and the changes shown in symptoms.”
functions, is of significant clinical value. This study is the first study ever that is indicative
of inhaled AAT’s ability to potentially reduce lung inflammation as expressed by its
preservation of lung function and the changes shown in symptoms.”
Prof. Jan Stolk, MD, Department of Pulmonology, Leiden University Medical Center, Principal Investigator of the Phase
2/3 clinical trial and acting Chairman of the Alpha 1 International Registry (AIR)
2/3 clinical trial and acting Chairman of the Alpha 1 International Registry (AIR)
“These new analyses confirm the clinically-meaningful lung function improvement seen
with inhaled AAT patients in this study. These results are impressive and underscore the
initial findings from this study. In my opinion, inhaled AAT has shown to be an efficacious
treatment for this orphan disease.”
with inhaled AAT patients in this study. These results are impressive and underscore the
initial findings from this study. In my opinion, inhaled AAT has shown to be an efficacious
treatment for this orphan disease.”
acceptance of the inhaled route of administration of alpha-
1 antitrypsin augmentation therapy, which could be a real
breakthrough for AATD patients.”
Robert A. Sandhaus, Ph.D., M.D., FCCP, Founder and Director
of the Alpha1-Antitrypsin Deficiency Program at National Jewish Health
in Denver, Colorado, and the Clinical Director of the Alpha-1 Foundation
“
16
Investor Presentation | June 2015
Inhaled AAT Phase II/III Trial Results:
Spirometry Measures (MMRM)
Spirometry Measures (MMRM)
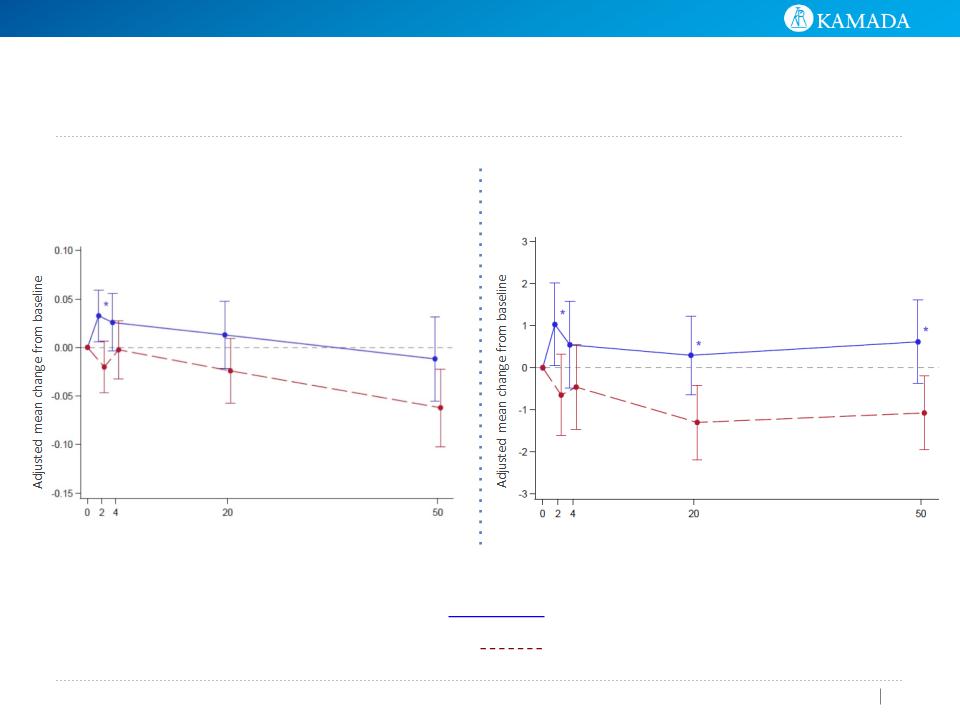
FEV1 (L) - MMRM
Week
FEV1/SVC - MMRM
Week
Overall effect +15ml
Overall effect -27ml
P=0.0268
Overall effect +0.623
Overall effect -0.8715
P=0.0074
Treatment Group
AAT
Placebo
17
Investor Presentation | June 2015
Inhaled AAT Phase II/III Trial:
Nature of the First Exacerbation
Nature of the First Exacerbation
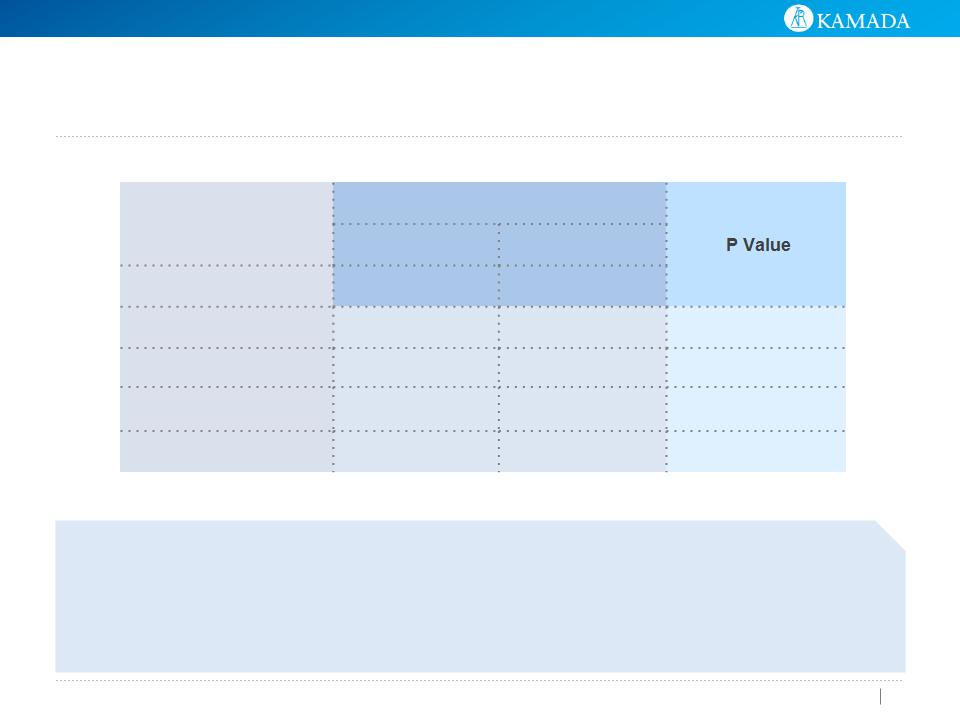
ITT | N (%) | ||
AAT | Placebo | ||
Type/Category | N=85 | N=83 | |
Type I | 16 (18.8%) | 26 (31.3%) | 0.0614 |
Type II | 23 (27.1%) | 12 (14.5%) | 0.0444 |
Type III | 34 (40.0%) | 33 (39.8%) | 0.9746 |
None | 12 (14.1%) | 12 (14.5%) | 0.9498 |
AAT may change the nature of the exacerbation
(Potential change from Type I to Type II)
Type I+II è Type I exacerbation stands for 41% within total of type I+
II exacerbations for AAT group vs. 68% for placebo group.
II exacerbations for AAT group vs. 68% for placebo group.
18
Investor Presentation | June 2015
Inhaled AAT Phase II/III Trial:
Symptom Score MMRM
Symptom Score MMRM
*Adjustment to age, oxygen, BMI, Country, Treatment Duration
Symptom | Exac. Type | Days | MMRM Least Square Means | P-Value* | |
AAT N=73 | Placebo N=71 | ||||
Dyspnea | 0-10 | 11.9464 | 12.2548 | 0.0243 | |
0-14 | 11.5803 | 11.7832 | 0.0817 | ||
Sputum Volume | 0-10 | 1.2748 | 1.3837 | 0.0334 | |
0-14 | 1.2367 | 1.3206 | 0.0595 | ||
Sputum Color | 0-10 | 2.1566 | 2.0137 | 0.0502 | |
0-14 | 2.0240 | 1.8393 | 0.0032 | ||
Analysis of First (Types I+II+III) Exacerbation Severity for each major Symptom
(during 0-10 and 0-14 days of the exacerbation event)
(during 0-10 and 0-14 days of the exacerbation event)
During first exacerbation, AAT group significantly improves
dyspnea and sputum volume symptoms
dyspnea and sputum volume symptoms
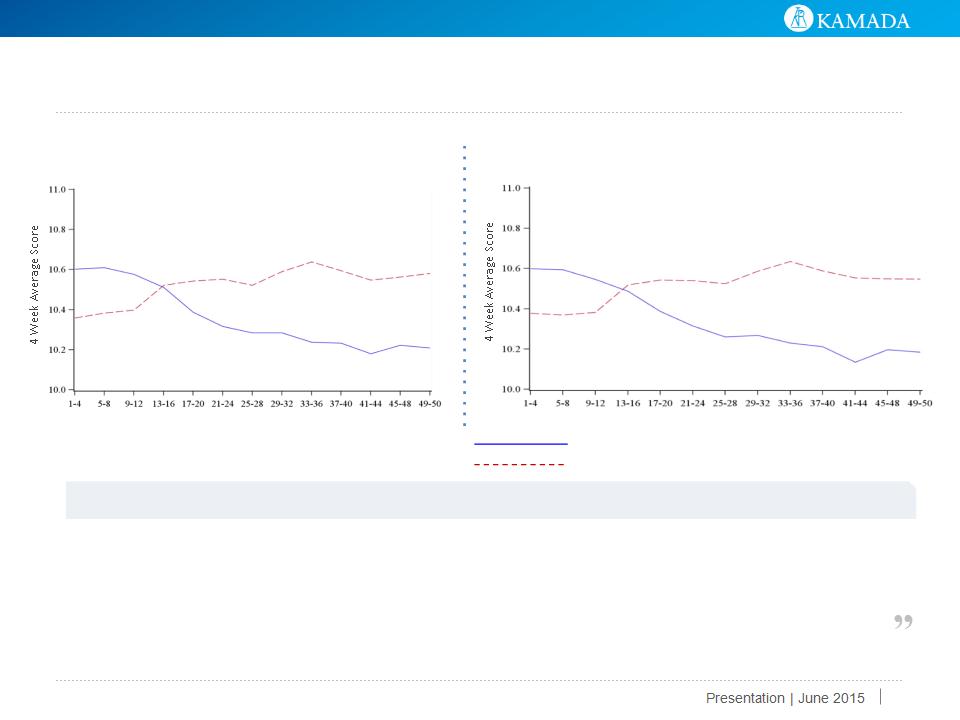
19
Inhaled AAT Phase II/III Trial Results
Continuous Symptom Score: Dyspnea
Continuous Symptom Score: Well Being
Dyspnea 4 Week Moving Average Graphs
Week
AAT
Placebo
Well Being 4 Week Moving Average Graphs
Week
Improvement trend in favor of AAT group, No statistical significance
“This study has enlightened our understanding about the course of exacerbation events, specifically with respect
to its composite symptoms, exacerbation severity and frequency with linkage to patients’ baseline disease.
Importantly, the improvements seen in well-being and dyspnea in the inhaled AAT treated patients suggest that
in addition to lung function improvements, these patients are seeing important improvement in their symptoms,
which are correlated to quality of life.”
to its composite symptoms, exacerbation severity and frequency with linkage to patients’ baseline disease.
Importantly, the improvements seen in well-being and dyspnea in the inhaled AAT treated patients suggest that
in addition to lung function improvements, these patients are seeing important improvement in their symptoms,
which are correlated to quality of life.”
Prof. R.A. Stockley, M.D., Professor of Medicine at Birmingham University and Medical Director of the Lung Resource Centre,
Queen Elizabeth Hospital, Birmingham, U.K. and a principal investigator of the European Phase 2/3 study.
Queen Elizabeth Hospital, Birmingham, U.K. and a principal investigator of the European Phase 2/3 study.
Improvement trend in favor of AAT group - not statistical significant
“

20
Investor Presentation | June 2015
Inhaled AAT: Moving Forward
● Compilation of an MAA dossier
● EMA submission (centralized
procedure) end of 2015
procedure) end of 2015
EMA: EU Front
● Approach U.S.-FDA with results in
H2 2015to obtain guidance on the
clinical/ regulatory pathway for
licensing the IH AAT by Kamada in
the U.S.
H2 2015to obtain guidance on the
clinical/ regulatory pathway for
licensing the IH AAT by Kamada in
the U.S.
FDA: U.S. Front
Kamada is committed to the AATD patient community to bring the IH AAT into
the market place and provide an adequate, safe and efficacious answer to
current unmet medical need of these orphan patients.
the market place and provide an adequate, safe and efficacious answer to
current unmet medical need of these orphan patients.

21
AAT to Treat Type 1 Diabetes
22
Investor Presentation | June 2015
AAT (IV) is a Promising Potential Treatment
for Newly Diagnosed Type ‐1 Diabetes Patients
for Newly Diagnosed Type ‐1 Diabetes Patients
Type-1 Diabetes
occurs when the immune
system attacks and destroys
beta cells in the pancreas
occurs when the immune
system attacks and destroys
beta cells in the pancreas
Studies have shown
That AAT protects
beta cell islets
Preservation of beta cells
correlates with reduced risk
of long term complications
correlates with reduced risk
of long term complications
● More than 10 million suffer
from T1D globally
from T1D globally
● 100,000 new patients
diagnosed annually
diagnosed annually
● In the U.S. alone: 3 million
patients, with 30,000 new
patients diagnosed annually
patients, with 30,000 new
patients diagnosed annually
● Delays the onset of autoimmune
diabetes
diabetes
● Reduces the incidence of
diabetes
diabetes
● Inhibits insulitis and beta-cell
apoptosis
apoptosis
● Decreases beta-cell inflammation
● DCCT* indicated that patients
with C-peptide on MMTT ≥0.2
pmol/mL were less likely to
complicate of retinopathy and
hypoglycemia (Greenbaum et al
2012)
with C-peptide on MMTT ≥0.2
pmol/mL were less likely to
complicate of retinopathy and
hypoglycemia (Greenbaum et al
2012)
● Higher / sustained levels of C-
peptide correlate with reduced
incidences of the microvascular
complications (Steffes et al 2013)
peptide correlate with reduced
incidences of the microvascular
complications (Steffes et al 2013)
*Diabetes Control and Complications Trial
FDA Guidance: “We acknowledge the evidence from the DCCT and other studies that have
demonstrated clinical benefits in patients who achieve better glucose control, in terms of
delaying the chronic complications of diabetes”**
demonstrated clinical benefits in patients who achieve better glucose control, in terms of
delaying the chronic complications of diabetes”**
**FDA Guidance, 2008
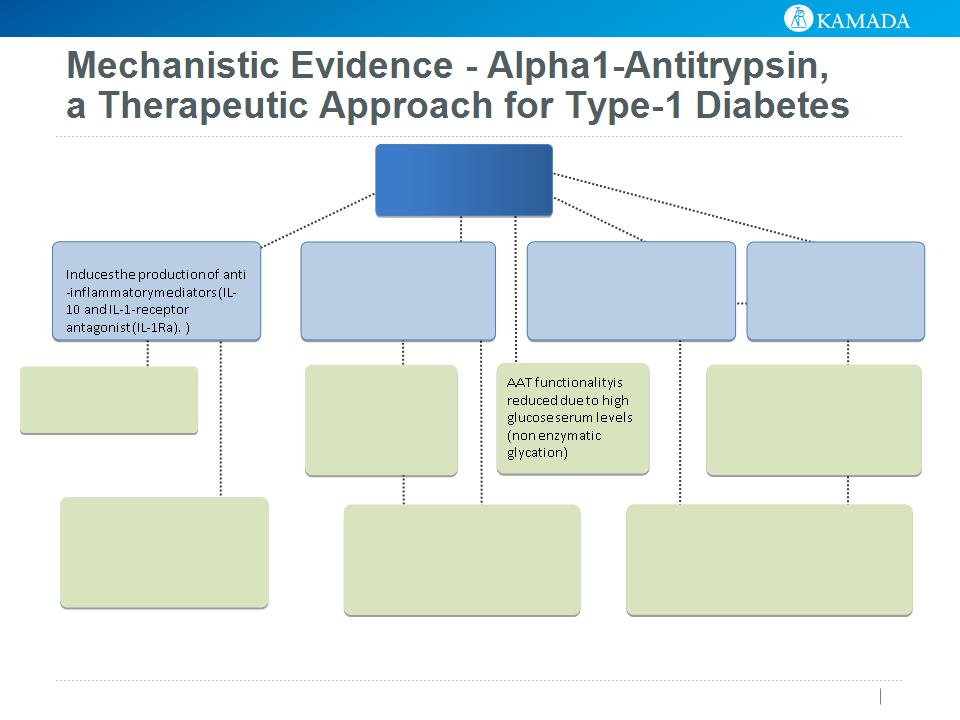
23
Investor Presentation | June 2015
AAT
Anti Inflammatory
Blocks pro-Inflammatory
mediators incl. IL-1b, IL-6,
IL-8 and TNFa
mediators incl. IL-1b, IL-6,
IL-8 and TNFa
Immunomodulatory
AAT promotes Tregs and
modifies dendritic cell
maturation towards a
tolerance-inducing profile
modifies dendritic cell
maturation towards a
tolerance-inducing profile
Tissue protective -
Protects from cell death,
blocks apoptosis
blocks apoptosis
Regulatory T cell
differentiation
differentiation
AAT promotes Tregs
differentiation
differentiation
Blocks ‘danger’ molecules
AAT binds to gp96 (involved in
authoimmune attack) and diminishes gp96-
induced islet injury
authoimmune attack) and diminishes gp96-
induced islet injury
Increases insulin release
AAT increases cAMP levels
(required for insulin release).
cAMP also induces IL-10
release
(required for insulin release).
cAMP also induces IL-10
release
Protease Inhibitor
Inhibits proteins that activate
inflammation: Elastase, trypsin
& PR3 and has tissue-
protective effects
inflammation: Elastase, trypsin
& PR3 and has tissue-
protective effects
Modifies dendritic cells:
AAT modifies dendritic cell
maturation towards a tolerance-
inducing profile
maturation towards a tolerance-
inducing profile
Protect islets from injury
AAT protects islet cells from
IL-1b/IFNg-induced injury and
reduces the levels of released
nitric oxide
reduces the levels of released
nitric oxide
Reference: Fleixo-Lima et al. Mechanistic Evidence in Support of Alpha1-Antitrypsin as a
Therapeutic Approach for Type 1 Diabetes. J Diabetes Sci Technol. 2014.
Therapeutic Approach for Type 1 Diabetes. J Diabetes Sci Technol. 2014.
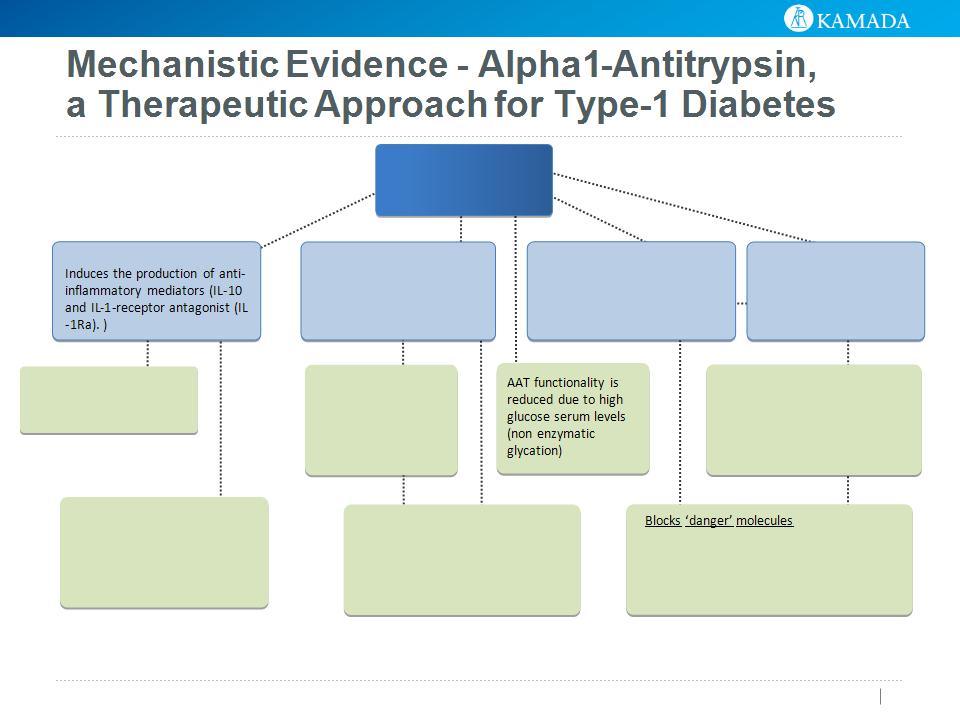
23
Investor Presentation | June 2015
AAT
Anti Inflammatory
Blocks pro-Inflammatory
mediators incl. IL-1b, IL-6,
IL-8 and TNFa
mediators incl. IL-1b, IL-6,
IL-8 and TNFa
Immunomodulatory
AAT promotes Tregs and
modifies dendritic cell
maturation towards a
tolerance-inducing profile
modifies dendritic cell
maturation towards a
tolerance-inducing profile
Tissue protective -
Protects from cell death,
blocks apoptosis
blocks apoptosis
Regulatory T cell
differentiation
differentiation
AAT promotes Tregs
differentiation
differentiation
AAT binds to gp96 (involved in
authoimmune attack) and diminishes
gp96-induced islet injury
authoimmune attack) and diminishes
gp96-induced islet injury
Increases insulin release
AAT increases cAMP levels
(required for insulin release).
cAMP also induces IL-10
release
(required for insulin release).
cAMP also induces IL-10
release
Protease Inhibitor
Inhibits proteins that activate
inflammation: Elastase, trypsin
& PR3 and has tissue-protective
effects
inflammation: Elastase, trypsin
& PR3 and has tissue-protective
effects
Modifies dendritic cells:
AAT modifies dendritic cell
maturation towards a tolerance-
inducing profile
maturation towards a tolerance-
inducing profile
Protect islets from injury
AAT protects islet cells from
IL-1b/IFNg-induced injury and
reduces the levels of released
nitric oxide
reduces the levels of released
nitric oxide
Reference: Fleixo-Lima et al. Mechanistic Evidence in Support of Alpha1-Antitrypsin as a
Therapeutic Approach for Type 1 Diabetes. J Diabetes Sci Technol. 2014.
Therapeutic Approach for Type 1 Diabetes. J Diabetes Sci Technol. 2014.
24
Investor Presentation | June 2015
Clinical Development for Newly Diagnosed
Type‐1 Diabetes: New Exciting Prospects
Type‐1 Diabetes: New Exciting Prospects
Phase I/II Open Label Study to evaluate the safety, tolerability and efficacy of AAT on
beta cell preservation and glycemic control on newly diagnosed T1D pediatric patients
beta cell preservation and glycemic control on newly diagnosed T1D pediatric patients
● AUC% for C-peptide decreased 23% from baseline vs. ~40-50% expected decrease after 12-15M from diagnosis (1)
● Specific diabetes antibody levels decreased in all groups from baseline to study completion, a decrease that may indicate
an Immune modulatory effect.
an Immune modulatory effect.
● At end-of-study, 38% of patients decreased insulin dose.
● All subjects completed the study. No Serious AEs occurred. AEs were mild and mostly infusion-related (fatigue,
headache)
headache)
End-of-study slope analysis of C-peptide[max] and
C-peptide[AUC] revealed no significant changes
from baseline
C-peptide[AUC] revealed no significant changes
from baseline
HbA1C data indicated that almost all patients reached
glycemic control
glycemic control
1. Greenbaum et al 2012
25
Diabetes Extension Clinical Study:
Interim Report #2
Interim Report #2
HbA1c
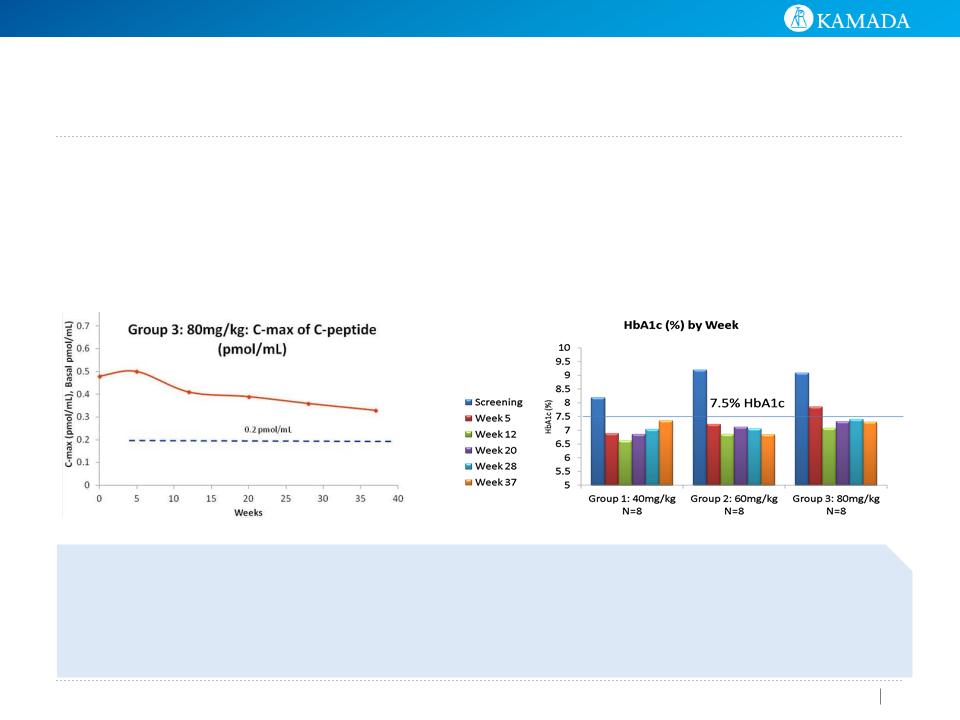
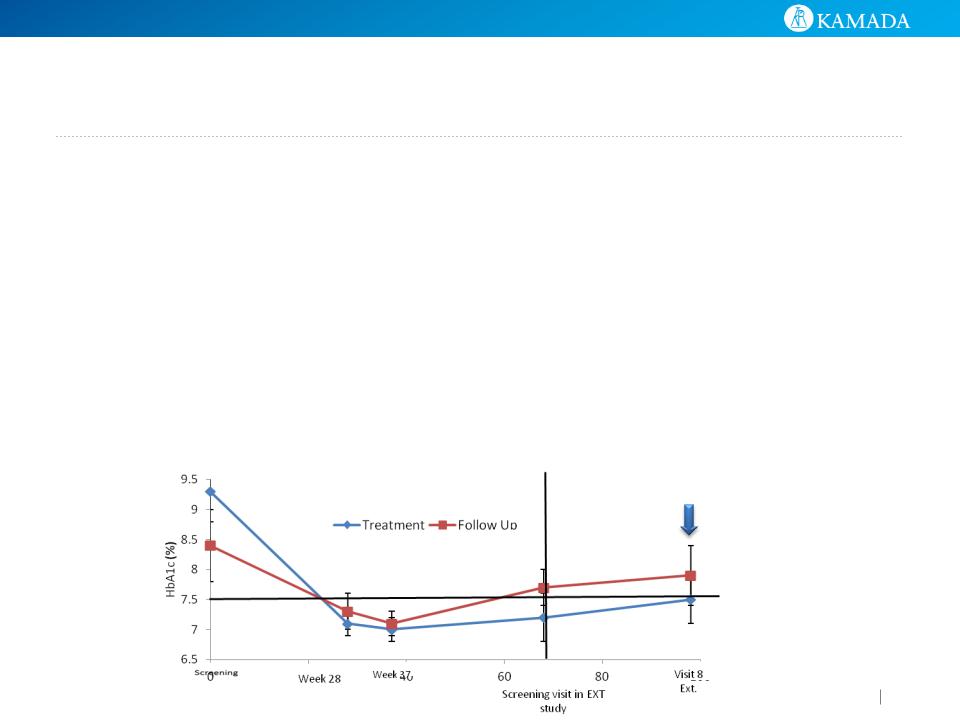
● Treated patients had an avg HbA1C of 7.5%, vs 7.9% for
the follow-up patients
the follow-up patients
● 60% of treated patients had HbA1C levels lower or equal
to 7.5% vs. 44% of follow-up patients
to 7.5% vs. 44% of follow-up patients
● Differences are not statistically significant - study was not
powered for efficacy
powered for efficacy
External Insulin Consumption and Safety
● Median insulin intake- treated patients 0.6 IU/kg/d vs to
1.00 IU/kg/d for follow-up patients (p = 0.025)
1.00 IU/kg/d for follow-up patients (p = 0.025)
● No safety issues were reported during this interim review
of trial data
of trial data
HbA1c (%) by Visit and Study Arm
After avg 26 months
from diagnosis
from diagnosis
19 subjects enrolled : the treatment arm
(n=10), follow-up arm (n=9)
(n=10), follow-up arm (n=9)
Data is presented 26 months (avg) post
T1D diagnosis- following 6 additional
AAT infusions
T1D diagnosis- following 6 additional
AAT infusions
C- Peptide
● Mean peak C-peptide level, was 0.40 pmol/ml in
the treatment group
the treatment group
● 60% of treated patients had a level ≥ 0.2
pmol/ml.
pmol/ml.
● C- peptide not collected for follow-up patients
Investor Presentation | June 2015
26
Investor Presentation | June 2015
Newly Diagnosed Type‐1 Diabetes Currently
Ongoing Phase II/III Clinical Trial
Ongoing Phase II/III Clinical Trial
Study objective: To evaluate the efficacy and safety
of human, Alpha-1 Antitrypsin (AAT) in the treatment
of new onset Type 1 Diabetes
of human, Alpha-1 Antitrypsin (AAT) in the treatment
of new onset Type 1 Diabetes
Design: Two doses, placebo controlled, randomized
with ~190 pediatric and young adult patients
with ~190 pediatric and young adult patients
Expected Duration: Two years
Endpoints: In accordance with FDA / EMA guidance
for clinical trials evaluating beta-cell preservation [c
peptide parameters, HbA1C, hypoglycemic events and
insulin daily dose]
for clinical trials evaluating beta-cell preservation [c
peptide parameters, HbA1C, hypoglycemic events and
insulin daily dose]
Pivotal, Phase II/III, Double-
Blind, Randomized, Placebo-
Controlled, Multicenter Study

27
AAT to Treat Graft versus Host Disease
28
Investor Presentation | June 2015
Graft versus Host Disease (GVHD):
The Major Issue in Stem Cell Transplantation
The Major Issue in Stem Cell Transplantation
● Deadly side effects:
● ~20% of transplanted patients’ deaths are caused by GvHD complications
● ~70% mortality in patients with grade III/IV GvHD
● ~50% of patients are non responsive to steroids
● Searching for an effective treatment
● Standard of care prophylaxis exhibits poor efficacy/severe AE’s
(Glucocorticoids)
(Glucocorticoids)
● No FDA approved specific drug for GvHD indication
● Estimated market size: ~ $700 million
Donor’s immune cells (the graft) recognize the recipient (the host) as
“Non-self“. The transplanted immune cells attack the host's body cells.
29
Investor Presentation | June 2015
Proof-of-Concept Study with AAT (IV)
for Graft-Versus-Host Disease (GVHD)
for Graft-Versus-Host Disease (GVHD)
This proof-of-concept study may serve as a potential platform,
to expand the use of AAT beyond GVHD, to other transplantations, based
on a similar mechanism of action
to expand the use of AAT beyond GVHD, to other transplantations, based
on a similar mechanism of action
Dose: 4 dose groups - 15 day regimen. Doses given
on days: 1,3,5,7, 9, 11, 13 and 15
on days: 1,3,5,7, 9, 11, 13 and 15
Primary End Points: % of patients at each dosing
cohort who experience no toxicity and in whom GVHD
is stable or improved
cohort who experience no toxicity and in whom GVHD
is stable or improved
Secondary End Points - AAT levels, cytokine levels,
infection rate, progression of GVHD, SAEs.
infection rate, progression of GVHD, SAEs.
In cooperation with Baxalta conducted at the Fred
Hutchinson Cancer Research Center in Seattle,
Washington
Hutchinson Cancer Research Center in Seattle,
Washington
Phase I/II study open label
of 24 patients with steroid-
resistant GVHD following
allogeneic bone-marrow stem
cell transplant
of 24 patients with steroid-
resistant GVHD following
allogeneic bone-marrow stem
cell transplant
.
30
Investor Presentation | June 2015
First Cohort Results Show that AAT May Potentially Exert a
Protective Effect on the Bowel Mucosa in Gut GVHD
Protective Effect on the Bowel Mucosa in Gut GVHD
Preliminary results are
encouraging, and further
exploration of AAT therapy in
extended phase II and randomized
trials as therapy of steroid
refractory acute GVHD or as first
line therapy are warranted
encouraging, and further
exploration of AAT therapy in
extended phase II and randomized
trials as therapy of steroid
refractory acute GVHD or as first
line therapy are warranted
29
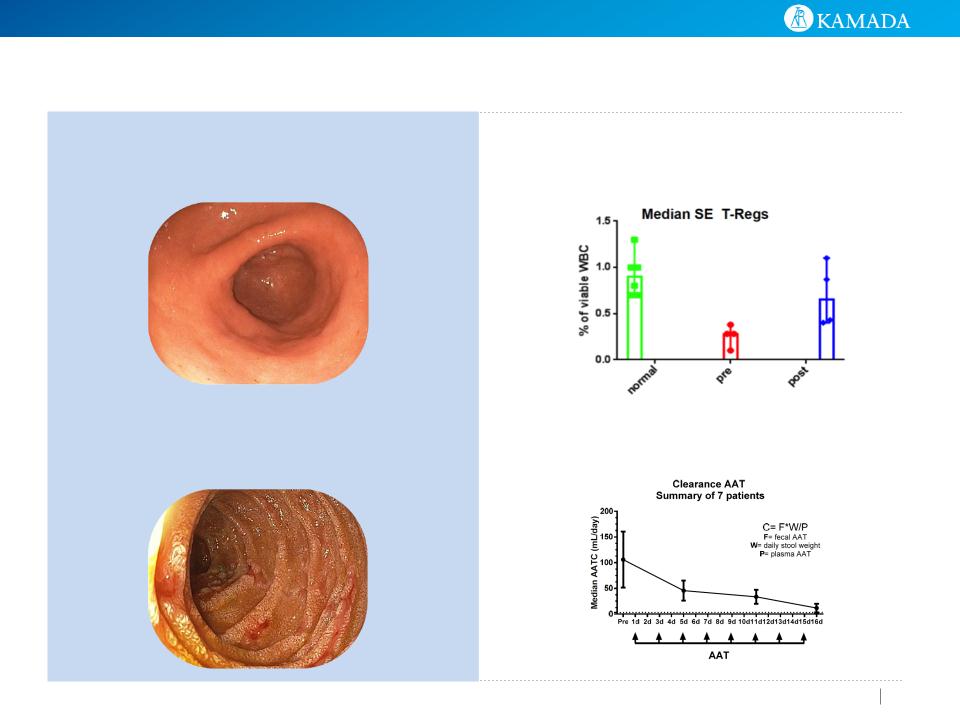
Study results have indicated that AAT may potentially exert healing of the
bowel mucosa in gut GVHD slowing/stopping the disease progression and re-
modulation of the immune attack.
bowel mucosa in gut GVHD slowing/stopping the disease progression and re-
modulation of the immune attack.
Stool AAT levels showed a decrease
in intestinal AAT loss, as measured
by AAT clearance and endoscopic
evaluation suggesting healing of the
bowel mucosa
in intestinal AAT loss, as measured
by AAT clearance and endoscopic
evaluation suggesting healing of the
bowel mucosa
Continuous administration of
AAT as salvage therapy for
steroid resistant gut GVHD is
feasible approach without
clinically toxicity
AAT as salvage therapy for
steroid resistant gut GVHD is
feasible approach without
clinically toxicity
31
Investor Presentation | June 2015
Phase I/II Clinical Study Interim Report
Loss of AAT in stool is an
expression of intestinal injury.
Before
Duodenits Suspect severe
upper and lower GvHD
After 8 doses of AAT
Moderate mucosal denudement
and edema noted throughout the duodenum.
FACS Analysis pre
and post AAT therapy

32
Investor Presentation | June 2015
Financials
33
Investor Presentation | June 2015
Compelling Investment Driven
by Multiple Pillars of Growth
by Multiple Pillars of Growth
Existing
Anchor
Products
Anchor
Products
§ Profitable unit
§ Sales in 15
countries
countries
§ Predictable,
stable business
($0.5B)*
Existing Anchor
Products
Products
+
Glassia®
(AAT-IV) in U.S.
(AAT-IV) in U.S.
+
Inhaled AAT for
AATD in
Europe & U.S.
AATD in
Europe & U.S.
+
New Geographies
+
Additional
Unencumbered
Pipeline Products
Unencumbered
Pipeline Products
Glassia®
(AAT-IV)
(AAT-IV)
in U.S.&ROW
§ Estimated only ~5%
of cases treated in
U.S.
of cases treated in
U.S.
§ Annual therapy costs
~$80K - $100K per
patient
~$80K - $100K per
patient
§ Partnered with Baxter
solely for IV products
in the U.S.
(agreement also
covers Canada,
Australia and New
Zealand)
solely for IV products
in the U.S.
(agreement also
covers Canada,
Australia and New
Zealand)
§ Key geographies
retained
retained
(100K pts.,
$0.75- 1B)*
New
Geographies
Geographies
§ Potential to sell
existing and new
products into new
geographies
existing and new
products into new
geographies
§ Rabies Ig to U.S. and
additional territories
additional territories
§ Capital-efficient
strategy minimizes
outlay required by
Kamada
strategy minimizes
outlay required by
Kamada
($0.5B)*
Additional
Unencumbered
Pipeline Products
Unencumbered
Pipeline Products
§ D1-AAT (IV):
Type-1 diabetes in
Phase I/II
(Unencumbered
outside of U.S.,
Canada, Australia and
New Zealand)
(100K pts.,$3.5-5B)*
Type-1 diabetes in
Phase I/II
(Unencumbered
outside of U.S.,
Canada, Australia and
New Zealand)
(100K pts.,$3.5-5B)*
§ G1-AAT (IV)
GVHD phase I/II
in process
($0.5-1B)*
($0.5-1B)*
§ C1-AAT (IH):
Cystic fibrosis
completed Phase II
(Unencumbered)
(100K pts.,$0.5-1B)*
Cystic fibrosis
completed Phase II
(Unencumbered)
(100K pts.,$0.5-1B)*
§ B1-AAT (IH):
Bronchiectasis
completed Phase II
(Unencumbered)
(600K pts.,$2B)*
Bronchiectasis
completed Phase II
(Unencumbered)
(600K pts.,$2B)*
Inhaled AAT for
AATD in
Europe & U.S.
AATD in
Europe & U.S.
§ Estimated only ~2% of
cases treated in
Europe
cases treated in
Europe
§ Estimated only ~5% of
cases treated in U.S.
cases treated in U.S.
§ Orphan drug
designation in U.S. and
EU
designation in U.S. and
EU
§ Partnered with Chiesi
for Inhaled AAT for
AATD in Europe only
for Inhaled AAT for
AATD in Europe only
§ Distribution (no
technology out-
licensed in Europe)
technology out-
licensed in Europe)
§ Unencumbered in U.S.
(200K pts.,$1-2B)*
The Kamada
Pillars
Pillars
* Estimated market potential

34
Investor Presentation | June 2015
Strong Financial Profile with Revenue Growth
and Expanding Profitability
and Expanding Profitability
● Stable, profit generating revenue stream from marketed
products
products
● Strategic partnership model results in efficient operating
expenses
expenses
● Baxalta purchase obligations provides stable revenue through 2017 and
royalties thereafter
royalties thereafter
● Kedrion partnership for Rabies Ig expected to increase revenues and
profitability from 2017 and on
profitability from 2017 and on
● Better product mix expected to improve gross margin
● Pipeline products expected to accelerate revenue growth
● Profits from marketed products to fund part of clinical development
programs
programs
● Low capital expenditure to support infrastructure meeting
future demand
future demand
● Preferred tax treatment under Israeli law
35
Investor Presentation | June 2015
Sustained and Rapid Growth has Made Kamada
EBITDA Positive Within 3 Years of Growth
EBITDA Positive Within 3 Years of Growth
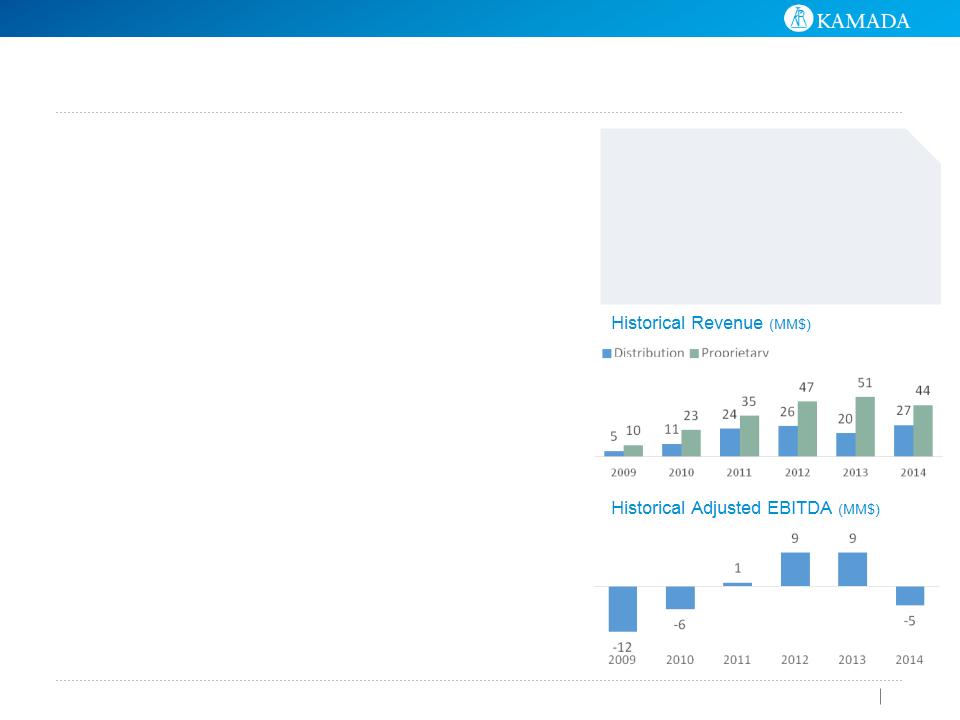
$MM | FY2009 | FY2010 | FY2011 | FY2012 | FY2013 | FY2014 |
Proprietary Products | 10 | 23 | 35 | 47 | 51 | 44 |
Growth | 130% | 54% | 32% | 9% | (14%) | |
Distribution | 4 | 11 | 24 | 26 | 20 | 27 |
Growth | 187% | 110% | 8% | (23%) | 35% | |
Total Revenues | 14 | 34 | 59 | 73 | 71 | 71 |
Growth | 146% | 73% | 22% | (3%) | 0% | |
Gross Profit | (3) | 6 | 17 | 23 | 26 | 16 |
R&D | (9) | (9) | (12) | (12) | (13) | (16) |
S&M and G&A | (5) | (7) | (7) | (7) | (10) (2) | (10) |
Net Profit (Loss) | (21) | (14) | (4) | 0.3 | 0.4 | (11) |
Adjusted EBITDA (1) | (12) | (6) | 1 | 9 | 9 | (5) |
Note
1. See Appendix for a reconciliation of Adjusted EBITDA to IFRS Net Profit (Loss)
2. Includes one time IPO related expenses of $1.4 M
36
Investor Presentation | June 2015
Consistent Track Record of Execution
U.S. FDA approval for Glassia® | |
Strategic agreement with Baxalta & First Glassia® sale in the U.S. | |
Strategic agreement for Rabies in the U.S. with Kedrion | |
Anti-Snake Venom launch | |
Strategic agreement with Chiesi for Inhaled AAT for AATD in EU | |
Newly diagnosed type-1 diabetes Phase II trial completed | |
Initiation of Phase II/III for type-1 diabetes | |
Initiation of U.S. Phase II for Inhaled AAT for AATD | |
Initiation of U.S. Phase I/II study of Glassia in GVHD | |
Completion of EU Phase II/III Inhaled AAT for AATD trial | |
Completion of U.S. Phase III Rabies Ig | |
U.S. & EU Orphan Drug Designation for Glassia to treat GVHD | |
Increased sales, profitability and production capacity |
2010
2015
May

37
Milestone Date
Investor Presentation | June 2015
Future Milestones and Value Creation
Phase III Rabies Ig trial (U.S.) results | 2H15 |
Interim data from GVHD trial | 2H15 |
MAA submission for Inhaled AAT for AATD | 2H15 |
Completion of Phase II for Inhaled AAT for AATD trial (U.S.) | 2H15 |
Strategic agreements | 2015 |
Initiation of Phase II lung transplantation trial | 2015 |
BLA submission for the Rabies lg in the U.S. | 2016 |
Initiation of Phase lll for intrevenous AAT for GVHD | 2016 |
Interim report for Phase II/III for type-1 diabetes trial | 2016 |
Rabies product launch in the U.S. (if approved) | 2016/7 |
Inhaled AAT for AATD launch (EU) (if approved) | 2016/7 |
Reaching $100 million of annual revenues | 2017 |
Double the number of Glassia patients WW | 2018 |
2H15
2018

38
Investor Presentation | June 2015
Kamada Investment Highlights
39
Investor Presentation | June 2015
Kamada Investment Highlights
• Rapidly Growing, Globally Positioned Biopharmaceutical
Company
Company
Focused on Orphan Diseases and Plasma Derived Protein Therapeutics
• Flagship Product Glassia® Approved for Alpha-1 Antitrypsin
Deficiency Disorder
Deficiency Disorder
Has a Unique and Differentiated Product Profile and Represents an Exciting
Growth Opportunity
Growth Opportunity
• Valuable R&D Pipeline Focused on Various Orphan
Indications
Indications
• Validating Strategic Partnerships with Industry Leaders
Baxalta, Chiesi, Kedrion and Pari Pharma
Baxalta, Chiesi, Kedrion and Pari Pharma

40
THANK YOU
Investor Presentation | June 2015

41
Investor Presentation | June 2015
APPENDIX
42
Investor Presentation | June 2015
Inhaled AAT Phase II/III trial:
Symptom Based Exacerbation Analysis
Symptom Based Exacerbation Analysis
Major Three (3) Exacerbation Symptoms by Severity: Dyspnea; Sputum Volume; Sputum Color | ||||
Exacerbation Type/Category | Classification Rules | Possible Manifestations | ||
Dyspnea* | Sputum Volume ** | Sputum Color** | ||
Type I | All 3 symptoms at high score | + | + | + |
Type II | Two of the 3 symptoms at high score | + | + | |
+ | + | |||
+ | + | |||
Type III | One of the 3 symptoms at high score | + | ||
+ | ||||
+ | ||||
Scores (by severity):
*5, 10, 15, 20 for Dyspnea (high severity score ≥10)
** 1, 2, 3, 4 for Sputum volume and Sputum color (high severity score ≥2)
*Kamada’s Inhaled AAT Phase 2-3 EU and Canada Study results. Denver USA 2015
43
Investor Presentation | June 2015
Conditional Approval Guidance & Precedence
EMEA/509951/2006
GUIDELINE ON THE SCIENTIFIC APPLICATION AND THE PRACTICAL ARRANGEMENTS NECESSARY TO IMPLEMENT
COMMISSION REGULATION (EC) No 507/2006 ON THE CONDITIONAL MARKETING AUTHORISATION FOR
MEDICINAL PRODUCTS FOR HUMAN USE FALLING WITHIN THE SCOPE OF REGULATION (EC) No 726/2004
COMMISSION REGULATION (EC) No 507/2006 ON THE CONDITIONAL MARKETING AUTHORISATION FOR
MEDICINAL PRODUCTS FOR HUMAN USE FALLING WITHIN THE SCOPE OF REGULATION (EC) No 726/2004
EMEA Guidance
Arzerra - GSK
http://www.bloomberg.com/apps/news?pid=newsarchive&sid=aaAMTmwslwq4
Cometriq - Exelixis
http://www.exelixis.com/investors-media/press-releases
Translarna PTC Therapeutics
http://ir.ptcbio.com/releasedetail.cfm?ReleaseID=888466
Deltyba - Otsuka
http://www.otsuka.co.jp/en/company/release/2013/1125_02.html
Sirturo - Johnson & Johnson
http://www.investor.jnj.com/releasedetail.cfm?ReleaseID=831021
Precedence for Conditional Approval
44
Investor Presentation | June 2015
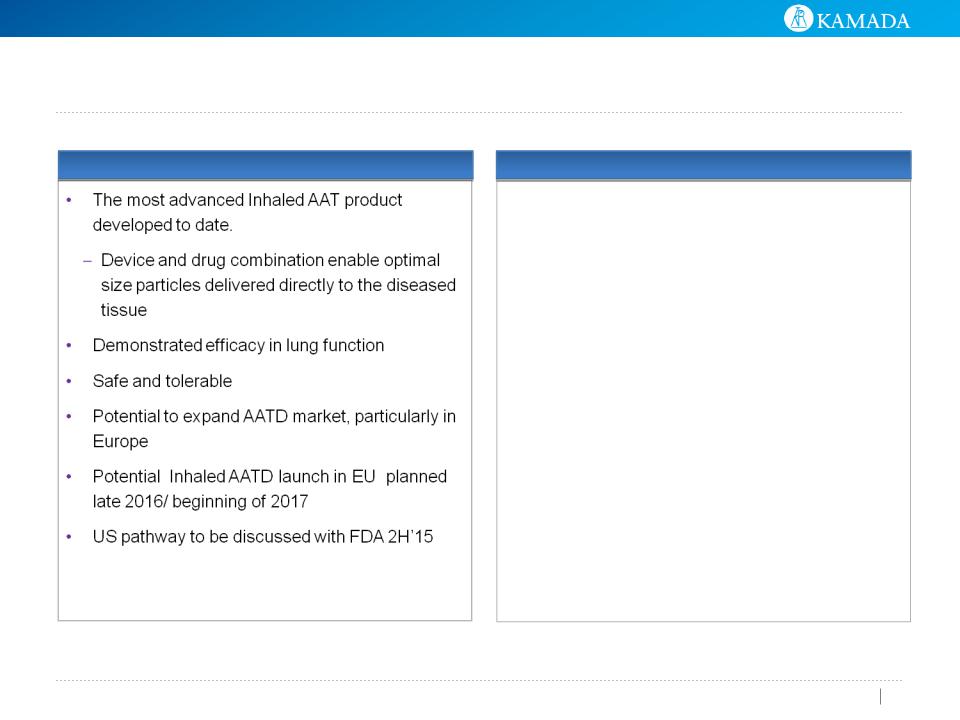
Inhaled AAT Is A Significant Opportunity
• Chiesi distribution agreement as of August 2012
• Agreement: Chiesi responsible for S&M, patient ID,
and reimbursement
and reimbursement
• Product: AAT for AATD Inhaled only
• Territories: EU and Turkey
• Milestone revenues: $60MM upfront, regulatory and
sales
sales
• Distributor price
• Minimum purchases from 2nd yr following receipt of
regulatory and reimbursement approvals, ~$120MM
for first 4 years, subject to actual price after
regulatory approval
regulatory and reimbursement approvals, ~$120MM
for first 4 years, subject to actual price after
regulatory approval
Strategic Partnership with Chiesi
Inhaled AAT Highlights