 Mallinckrodt Strategic Agreement Ikaria, Inc. March 5, 2015 Exhibit 99.2 |
 Forward-Looking Statements 3/5/2015 1 Statements in this document that are not strictly historical, including statements regarding, the proposed acquisition of Ikaria, Inc., the expected timetable for completing the transaction, future financial condition and operating results, economic, business, competitive and/or regulatory factors affecting Mallinckrodt’s and Ikaria’s businesses and any other statements regarding events or developments that we believe or anticipate will or may occur in the future, may be "forward-looking" statements within the meaning of the Private Securities Litigation Reform Act of 1995, and involve a number of risks and uncertainties. There are a number of important factors that could cause actual events to differ materially from those suggested or indicated by such forward-looking statements and you should not place undue reliance on any such forward-looking statements. These factors include risks and uncertainties related to, among other things: General economic conditions and conditions affecting the industries in which Mallinckrodt and Ikaria operate; The commercial success of Mallinckrodt’s products and of INOMAX®; The parties’ ability to satisfy the acquisition agreement conditions (including required regulatory approvals) and complete the Ikaria acquisitions on the anticipated timeline or at all; Mallinckrodt’s ability to realize anticipated growth, synergies and costs savings from its recently completed acquisitions and the Ikaria acquisition; Changes in laws and regulations; Mallinckrodt’s ability to identify, acquire or close future acquisitions; Mallinckrodt’s ability to successfully integrate acquisitions of operations, technology, products and businesses generally and to realize anticipated growth, synergies and cost savings (including with respect to the Ikaria acquisition); Mallinckrodt’s ability to successfully develop or commercialize new products; Mallinckrodt’s ability to protect intellectual property rights; Mallinckrodt’s ability to receive procurement and production quotas granted by the U.S. Drug Enforcement Administration; Customer concentration; |
 Forward-Looking Statements (continued) 3/5/2015 Mallinckrodt’s reliance on certain individual products that are material to its financial performance; Cost containment efforts of customers, purchasing groups, third-party payers and governmental organizations; The reimbursement practices of a small number of public or private insurers; Limited clinical trial data for H.P. Acthar ® Gel; Complex reporting and payment obligations under healthcare rebate programs; Mallinckrodt’s ability to achieve anticipated benefits of price increases; Mallinckrodt’s ability to achieve expected benefits from restructuring activities; Complex manufacturing processes; Competition; Product liability losses and other litigation liability; Ongoing governmental investigations; Material health, safety and environmental liabilities; Retention of key personnel; Conducting business internationally; and The effectiveness of information technology infrastructure. 2 These and other factors are identified and described in more detail in the "Risk Factors" section of Mallinckrodt's Annual Report on Form 10-K for the fiscal year ended September 26, 2014. The forward-looking statements made herein speak only as of the date hereof and Mallinckrodt does not assume any obligation to update or revise any forward-looking statement, whether as a result of new information, future events and developments or otherwise, except as required by law. |
 Diversifies portfolio with durable, high-value, branded asset Builds reputation, reach and impact of Mallinckrodt and INOmax ® (Inhaled Nitric Oxide) in hospitals: Extends footprint to respiratory therapies in NICUs 1 Increases value for clinicians, hospitals with comprehensive drug- device-service platform $2.3 billion agreement to fully acquire Ikaria, Inc. Mallinckrodt expands hospital platform with Ikaria, Inc. 3/5/2015 INOmax is a registered trademark of INO Therapeutics LLC. 1 Neonatal Intensive Care Unit Adds potential diversity in nephrology rare disease pipeline with terlipressin (for injection) Builds upon Mallinckrodt expertise in drug- device development, regulatory management and manufacturing to support future development 3 |
 Transaction highlights 3/5/2015 Consideration Acquisition of Ikaria, Inc. for approximately $2.3 billion Financial Impact Transaction expected to add at least $150 million in net sales and be accretive to Mallinckrodt’s fiscal year 2015 adjusted diluted earnings per share by at least $0.25 per share 1 Structure All cash Financing Financing expected to include cash on hand, use of Mallinckrodt’s $250 million revolver and debt Timing Closing expected early third fiscal quarter, subject to customary closing conditions 1 Assuming close early in fiscal year 2015 third quarter 4 |
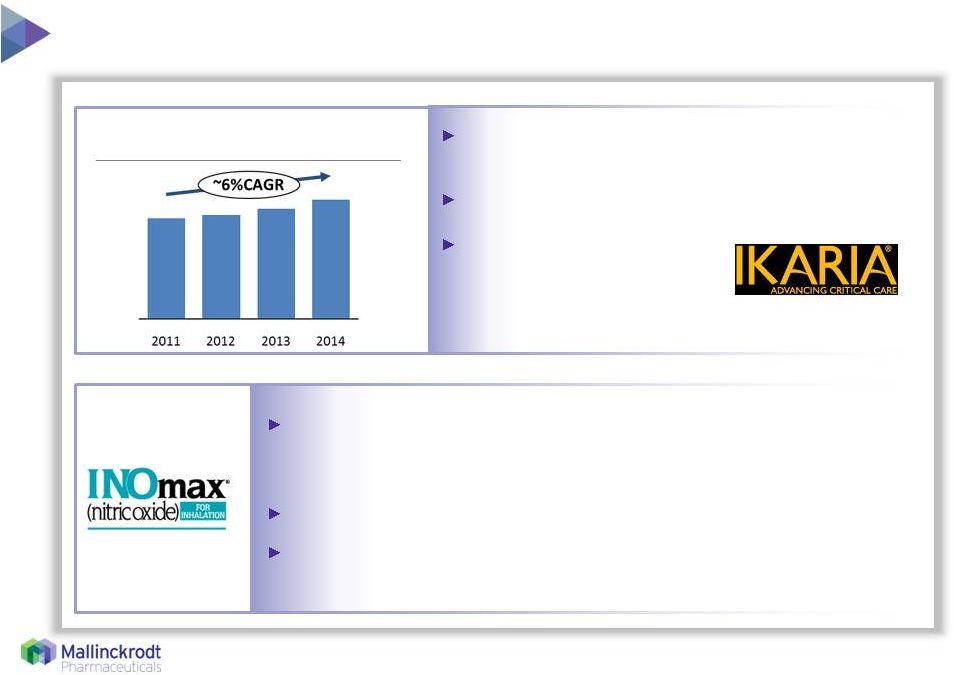 5 Innovative, integrated drug/device/service offerings to support critically ill infants in NICUs Steady growth driven by U.S. market 1 Calendar year revenue Ikaria, Inc. – growth opportunity in hospital critical care 3/5/2015 Global Revenue 1 INOmax/INOmax DS IR ® , the only FDA-approved treatment for neonates born at or near term suffering from hypoxic respiratory failure (HRF) due to persistent pulmonary hypertension Only FDA-inspected U.S. nitric oxide drug manufacturing facility Delivery systems validated with 79 commonly used ICU ventilatory devices Multi-year contracts and TotalCare ™ service foster deep relationships in ICUs |
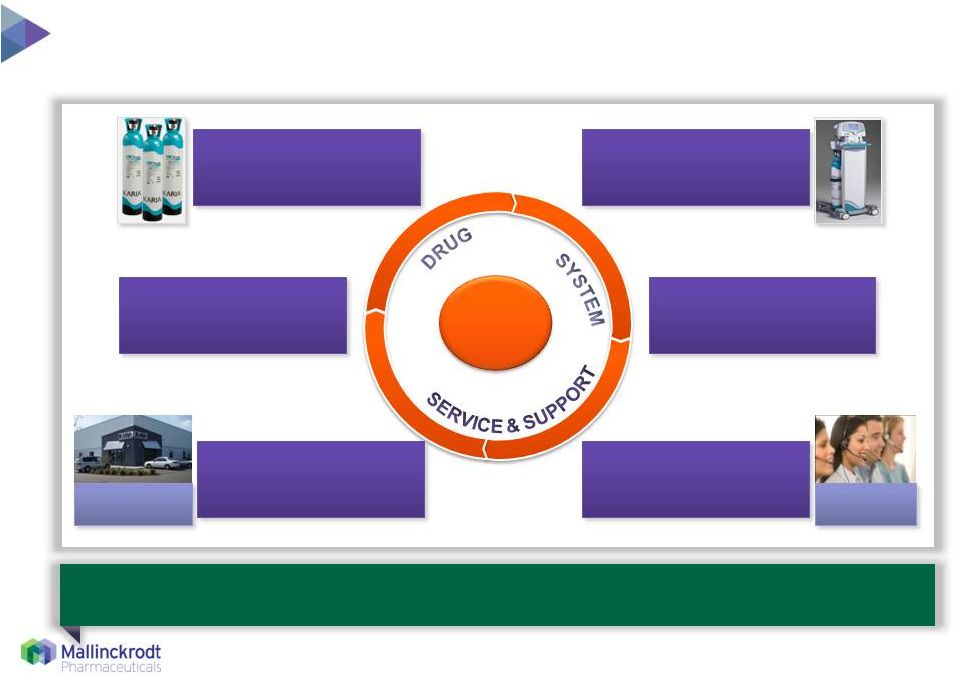 6 COG Integrated INOmax drug/device/service package is core to success 3/5/2015 5 regional service centers 5 regional service centers INOmax drug INOmax delivery system Regular preventative service & maintenance (QA/QC) Extensive distribution, including emergency shipments 24/7/365 technical & medical support Customer Care Customer Care Ongoing physician/clinician training Highly Satisfied Customer Mallinckrodt has commercial reach and scale necessary to expand access and realize the full value of INOmax in the marketplace |
 7 Transaction continues to deliver on strategy, builds value 3/5/2015 Value creation Immediately accretive to fiscal year 2015 Diversifies Mallinckrodt revenue and operating income with sustainable, growing, highly profitable business Portfolio fit Adds innovative, difficult-to-replicate asset to Mallinckrodt portfolio Extends hospital presence; establishes presence in NICU Builds potential in Mallinckrodt’s autoimmune and rare disease nephrology pipeline with terlipressin (for injection) Value for INOmax Builds brand recognition beyond the ICU Broadens access Supports drug/device innovation with Mallinckrodt development and regulatory expertise |