Exhibit 99.2
USE OF CERTAIN TERMS
As used in this Offering Memorandum (as defined below), except where otherwise specified or unless the context otherwise requires:
| • | the “Issuer” refers to Mallinckrodt International Finance S.A., a Luxembourg public limited liability company (société anonyme) incorporated under the laws of Luxembourg, with its registered office at 124 boulevard de la Pétrusse, L-2330 Luxembourg and being registered with the Luxembourg trade and companies register under number B.172.865 and an indirect wholly owned subsidiary of Mallinckrodt plc; |
| • | “MCB” or the “U.S. Co-Issuer” refers to Mallinckrodt CB LLC, a Delaware limited liability company and a direct wholly owned subsidiary of the Issuer; |
| • | the “Issuers” refers to the Issuer and the U.S. Co-Issuer, collectively; |
| • | the “Exchange Offers” refer to the private offers by the Issuers, commenced on November 5, 2019, to exchange the Existing Notes for the New Notes, subject to the terms and conditions set forth in the Offering Memorandum; |
| • | the “Consent Solicitations” refer to the solicitations of consents from the holders of each series of Existing Notes to amend the indentures governing such series of Existing Notes; subject to the terms and conditions set forth in the Offering Memorandum; |
| • | the “Exchange Agreement” refers to that certain Exchange Agreement entered into by and among the Exchanging Holders and the Issuers, dated as of November 5, 2019; |
| • | “Exchanging Holders” refers to Deerfield Partners, L.P., Deerfield Special Situations Fund, L.P. and Deerfield Private Design Fund IV, L.P.; |
| • | the “Existing Notes” refers to the following series of notes issued by the Issuers: the 4.875% Senior Notes due 2020; 5.750% Senior Notes due 2022; 4.750% Senior Notes due 2023; 5.625% Senior Notes due 2023; and 5.500% Senior Notes due 2025. |
| • | the “New Notes” refers to the 10.000% Second Lien Senior Secured Notes due 2025 to be issued by the Issuers in connection with the Exchange Offer and Exchange Agreement; |
| • | the “Offering Memorandum” refers to the Issuers’ confidential offering memorandum and consent solicitation statement, dated November 5, 2019; |
| • | “Early Delivery Time” means 5:00 p.m., New York City time, on November 19, 2019; |
| • | “we,” “us,” and “our” refer to the Issuers and their respective direct and indirect subsidiaries, collectively; |
| • | “Mallinckrodt plc” refers to Mallinckrodt plc, an Irish public limited company, excluding its subsidiaries; |
| • | “Mallinckrodt,” “our company” and “the company” refer to Mallinckrodt plc, an Irish public limited company, and its subsidiaries; |
| • | “Annual Report” refers to Mallinckrodt plc’s Annual Report on Form 10-K for the fiscal year ended December 28, 2018 filed on February 26, 2019; |
| • | “Quarterly Reports” refer to Mallinckrodt plc’s Quarterly Reports on Form 10-Q for the quarterly periods ended March 29, 2019 and June 28, 2019, filed on May 7, 2019 and August 6, 2019, respectively; |
| • | “GAAP” means accounting principles generally accepted in the U.S; |
| • | “dollar” or “$” refer to the U.S. dollar; and |
| • | “U.S.” means the United States. |
Except as otherwise indicated, references in this Offering Memorandum to fiscal 2019, fiscal 2018, fiscal 2017 and fiscal 2016 are to Mallinckrodt plc’s fiscal years ending or ended December 27, 2019, December 28, 2018, December 29, 2017 and September 30, 2016. We historically reported our results based on a “52-53 week” year ending on the last Friday of September. During fiscal 2016, we changed our fiscal year-end to the last Friday in December from the last Friday in September. The change in fiscal year became effective for our 2017 fiscal year, which began on December 31, 2016 and ended on December 29, 2017. As a result of the change in fiscal year-end, the period from October 1, 2016 through December 30, 2016 is referred to herein as “the three months ended December 30, 2016”.
1
Recent Developments
Planned Spinoff of Specialty Generics Business and Subsequent Suspension of Spinoff Plans
In fiscal 2016, the Board of Directors of Mallinckrodt plc began to explore a range of strategic alternatives for the company’s Specialty Generics business. Consistent with that strategy, on December 6, 2018, Mallinckrodt plc announced its plans to spin off to Mallinckrodt plc shareholders a new independent public company that would hold Mallinckrodt plc’s Specialty Generics business. On August 6, 2019, based on market conditions and developments, including increasing uncertainties created by the opioid litigation (which is further described under the section captioned “Legal Proceedings” in Mallinckrodt plc’s Quarterly Report on Form 10-Q for the fiscal quarter ended June 28, 2019), Mallinckrodt plc announced the suspension of its previously announced plans to spin off the Specialty Generics company. Mallinckrodt plc’s long-standing goal remains to be an innovation-driven biopharmaceutical company focused on improving outcomes for underserved patients with severe and critical conditions. Accordingly, while the previously disclosed spinoff plan remains suspended, Mallinckrodt plc continues to actively consider a range of options intended to lead to the ultimate separation of the Specialty Generics business, consistent with its previously stated strategy.
Opioid Litigation Settlements
As further described under the section captioned “Legal Proceedings” in Mallinckrodt plc’s Quarterly Report on Form 10-Q for the fiscal quarter ended June 28, 2019, multiple U.S. states, counties, other governmental persons or entities and private plaintiffs have filed lawsuits against certain Mallinckrodt entities, as well as various other manufacturers, distributors, pharmacies, pharmacy benefit managers, individual doctors and/or others, asserting claims relating to defendants’ alleged sales, marketing, distribution, reimbursement, prescribing, dispensing and/or other practices with respect to prescription opioid medications, including certain of Mallinckrodt’s products. Mallinckrodt has been engaged and as of the date of this Offering Memorandum is still engaged in settlement discussions in connection with certain of these lawsuits.
Specifically, on September 30, 2019, Mallinckrodt plc, along with its wholly owned subsidiaries Mallinckrodt LLC and SpecGx LLC, announced that it had executed a definitive settlement agreement and release (the “Settlement Agreement”) with Cuyahoga and Summit Counties in Ohio in connection with lawsuits pending in multidistrict opioid litigation (MDL) in the U.S. District Court for the Northern District of Ohio: The County of Cuyahoga, et al. v. Purdue Pharma, L.P., et al., Case No. 17-OP-45004; and The County of Summit, et al. v. Purdue Pharma, L.P., et al., Case No. 18-OP-45090. As of the date of this Offering Memorandum, Mallinckrodt has been in preliminary discussions with certain plaintiffs in other pending opioid lawsuits and is likely to have further discussions and/or enter into additional discussions with other parties in connection with opioid lawsuits. Mallinckrodt may be required to pay material amounts and/or incur other material obligations as a result of any settlements that are entered into as a result of such discussions. Further, such matters or the resolution thereof, whether through judicial process or settlement or otherwise, may make it necessary or advisable for Mallinckrodt and/or one or more of its subsidiaries to seek to restructure its or their obligations in a bankruptcy proceeding. The company is exploring a wide array of such potential outcomes as part of its contingency planning, including the impact such actions could have on its business and operations. Should a bankruptcy occur, Mallinckrodt would be subject to additional risk and uncertainties that could adversely affect Mallinckrodt’s business prospects and ability to continue as a going concern, as further described under “Risk Factors.”
U.S. Department of Health and Human Services (HHS) and Centers for Medicare and Medicaid (CMS) Litigation
On May 21, 2019, Mallinckrodt plc announced that its subsidiary, Mallinckrodt ARD LLC, had filed suit in federal district court against the U.S. Department of Health and Human Services (“HHS”) and Centers for Medicare and Medicaid Services (“CMS”, and together with HHS, the “Agency”) seeking to hold unlawful and set aside CMS’s decision to require that Mallinckrodt plc revert to the base date average manufacturer price used to calculate Medicaid drug rebates for Acthar Gel. The court held a hearing regarding this matter on August 2, 2019 and the court took the matter under advisement. While Mallinckrodt believes that its lawsuit has strong factual and legal bases, the potential for retroactive non-recurring charges could range from zero to approximately $600.0 million. See the section captioned “Legal Proceedings” in Mallinckrodt plc’s Quarterly Report on Form 10-Q for the fiscal quarter ended June 28, 2019.
ORGANIZATIONAL STRUCTURE
The following chart summarizes our corporate structure and principal amount of third-party indebtedness in millions of dollars on a pro forma basis as of the date of the Offering Memorandum after giving effect to the Exchange Offers and the Consent Solicitations and the transactions contemplated by the Exchange Agreement. The chart assumes that the holders of notes representing 50.1% of the aggregate principal amount outstanding of each series of Existing Notes validly tender such notes (and do not validly withdraw them) in each of the Exchange Offers. See “Capitalization” elsewhere in this Offering Memorandum.
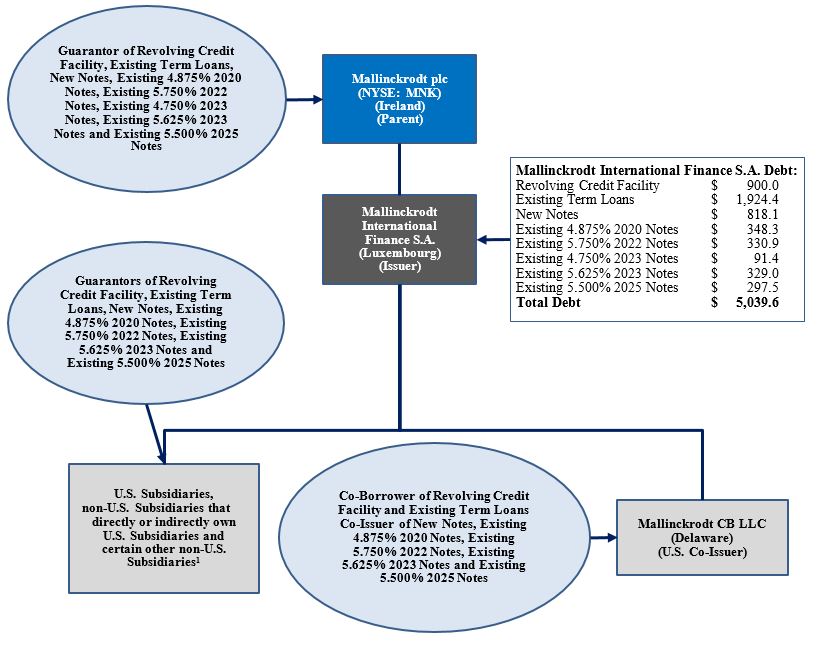
| (1) | Certain U.S. subsidiaries are additional borrowers of the Revolving Credit Facility. A U.S. subsidiary is the issuer of approximately $10 million of 9.500% Debentures due 2022 and approximately $4 million 8.000% Debentures due 2023. |
RISK FACTORS
Governmental investigations, inquiries, and regulatory actions and lawsuits brought against Mallinckrodt by government agencies and private parties with respect to the company’s historical commercialization of opioids could adversely affect Mallinckrodt’s business, financial condition, results of operations and cash flows.
As a result of greater public awareness of the public health issue of opioid abuse, there has been increased scrutiny of, and investigation into, the commercial practices of opioid manufacturers by state and federal agencies. Mallinckrodt, along with other opioid manufacturers, have been the subject of federal and state government investigations and enforcement actions, focused on the misuse and abuse of opioid medications in the U.S. Similar investigations may be initiated in the future.
In addition, a significant number of lawsuits have been filed against the company, other opioid manufacturers, distributors, and others in the supply chain by cities, counties, state Attorneys General and private persons seeking to hold Mallinckrodt and others accountable for opioid misuse and abuse. As of November 4, 2019, the cases we are aware of include, but are not limited to, approximately 2,313 cases filed by counties, cities, Native American tribes and/or other government-related persons or entities; approximately 205 cases filed by hospitals, health systems, unions, health and welfare funds or other third-party payers; approximately 104 cases filed by individuals and 14 cases filed by the Attorneys General for New Mexico, Kentucky, Rhode Island, Georgia, Florida, Alaska, New York, Hawaii, Nevada, South Dakota, New Hampshire, Louisiana, Illinois, and Idaho, with Idaho being the only state Attorney General to file in federal as opposed to state court. As of November 4, 2019, the Mallinckrodt defendants in these cases consist of Mallinckrodt plc and the following subsidiaries of Mallinckrodt plc: Mallinckrodt Enterprises LLC, Mallinckrodt LLC, SpecGx LLC, Mallinckrodt Brand Pharmaceuticals Inc., Mallinckrodt Inc., MNK 2011 Inc., and Mallinckrodt Enterprises Holdings, Inc. However, there can be no assurance that plaintiffs will not assert claims against additional Mallinckrodt plc subsidiaries in the future. The lawsuits assert a variety of claims, including, but not limited to, public nuisance, negligence, civil conspiracy, fraud, violations of the Racketeer Influenced and Corrupt Organizations Act (“RICO”) or similar state laws, violations of state Controlled Substances Act (“CSA”) or state False Claims Act, product liability, consumer fraud, unfair or deceptive trade practices, false advertising, insurance fraud, unjust enrichment and other common law and statutory claims arising from defendants’ manufacturing, distribution, marketing and promotion of opioids and seek restitution, damages, injunctive and other relief and attorneys’ fees and costs. The claims generally are based on alleged misrepresentations and/or omissions in connection with the sale and marketing of prescription opioid medications and/or an alleged failure to take adequate steps to prevent diversion. Other parties may file similar lawsuits against the company in the future.
As a company that first began processing opioids in the 1890s, Mallinckrodt understands the utility of these products and that they are safe and effective when taken as appropriately prescribed. The company is deeply committed to diversion control efforts, has sophisticated systems in place to identify suspicious orders, and engages in significant due diligence and ongoing monitoring of customers. While the company is vigorously defending itself in these matters, the nature and scope of these matters is unique, and current public perceptions of the public health issue of opioid abuse, together with the manner in which other defendants in those cases resolve opioid-related lawsuits and other actions, may present challenges to favorable resolution of these claims. Accordingly, it is not feasible to predict the ultimate outcome of these investigations, enforcement actions and lawsuits. The allegations against the company may negatively affect Mallinckrodt’s business in various ways, including through harm to the company’s reputation. Mallinckrodt will continue to incur significant legal costs in defending these matters and could in the future be required to pay significant amounts as a result of fines, penalties, settlements or judgments, potentially in excess of established accruals. The company may be unable to obtain or maintain insurance in the future on acceptable terms or with adequate coverage against potential liabilities or other losses. Any such potential liabilities or losses could also require the company to seek financing, which may not be available on terms acceptable to the company, or at all, when required. As of the date of this Offering Memorandum, Mallinckrodt has been in preliminary discussions with certain plaintiffs in pending opioid lawsuits and is likely to have further discussions and/or enter into additional discussions with other parties in connection with opioid lawsuits and/or incur other material obligations. Mallinckrodt may be required to pay material amounts and/or incur other material obligations as a result of any settlements that are entered into as a result of such discussions.
Such matters or the resolution thereof, or increase in accruals thereof, could have a material adverse effect on Mallinckrodt’s business, financial condition, results of operations and cash flows. Further, such matters or the resolution thereof, whether through judicial process or settlement or otherwise, may make it necessary or advisable for Mallinckrodt and/or one or more of its subsidiaries to seek to restructure its or their obligations in a bankruptcy proceeding. The company is exploring a wide array of such potential outcomes as part of its contingency planning, including the impact such actions could have on its business and operations. Should a bankruptcy occur, the company would be subject to additional risks and uncertainties that could adversely affect Mallinckrodt’s business prospects and ability to continue as a going concern, including, but not limited to by causing increased difficulty obtaining and maintaining commercial relationships on competitive terms with customers, suppliers and other counterparts; increased difficulty retaining and motivating key employees, as well as
attracting new employees; diversion of management’s time and attention to dealing with bankruptcy and restructuring activities rather than focusing exclusively on business operations; incurrence of substantial costs, fees and other expenses associated with bankruptcy proceedings; and loss of ability to maintain or obtain sufficient financing sources for operations or to fund any reorganization plan and meet future obligations. Mallinckrodt would in that event also be subject to risks and uncertainties caused by the actions of creditors and other third parties who have interests that may be inconsistent with Mallinckrodt’s plans.
In addition, legislative, regulatory or industry measures to address the misuse of prescription opioid medications may also affect the company’s business in ways that the company is not able to predict. For example, the State of New York enacted the Opioid Stewardship Act (“OSA”), which went into effect on July 1, 2018 and established an aggregate $100 million annual assessment on sales of certain opioid medications in New York. The OSA was successfully challenged, and on December 19, 2018, the U.S. District Court for the Southern District of New York ruled that the OSA was unconstitutional and enjoined its enforcement. On January 17, 2019, the State of New York appealed this ruling. The litigation is still pending. In April 2019, the State of New York passed its 2020 budget, which amended the OSA so that if the OSA decision is reversed on appeal, the OSA would apply only to the sale or distribution of certain opioids in New York for 2017 and 2018 and, effective July 1, 2019, imposed an excise tax on certain opioids. Furthermore, other states are considering similar legislation that could require entities to pay an assessment or tax on the sale or distribution of opioid medications in those states and may vary in the assessment or tax amounts and the means of calculation from the OSA. If other state or local jurisdictions successfully enact such legislation and the company is not able to mitigate the impact on the company’s business through operational changes or commercial arrangements, such legislation in the aggregate may have a material adverse effect on Mallinckrodt’s business, financial condition, results of operations and cash flows. See the risk factor “Extensive laws and regulations govern the industry in which Mallinckrodt operates, and any failure to comply with such laws and regulations, including any changes to those laws and regulations may materially adversely affect the company” for more information.
Furthermore, in the current climate, stories regarding prescription drug abuse and the diversion of opioids and other controlled substances are frequently in the media. Unfavorable publicity regarding the use or misuse of opioid drugs, the limitations of abuse-deterrent formulations, the ability of drug abusers to discover previously unknown ways to abuse the company’s products, public inquiries and investigations into prescription drug abuse, litigation, or regulatory activity regarding sales, marketing, distribution or storage of opioids could have a material adverse effect on Mallinckrodt’s reputation and impact on the results of litigation.
Finally, various government entities, including Congress, state legislatures or other policy-making bodies have in the past and may in the future hold hearings, conduct investigations and/or issue reports calling attention to the opioid crisis, and may mention or criticize the perceived role of manufacturers, including Mallinckrodt, in the opioid crisis. Similarly, press organizations have and likely will continue to report on these issues, and such reporting may result in adverse publicity for the company, resulting in reputational harm.
Mallinckrodt previously identified a material weakness in the company’s internal control over financial reporting, which has now been remediated by management. If Mallinckrodt fails to maintain an effective system of internal controls over financial reporting, Mallinckrodt may not be able to report its financial results timely and accurately, which could adversely affect the company’s business or the market price of the company’s ordinary shares.
As disclosed in Mallinckrodt plc’s Form 10-K for the fiscal year 2018, Mallinckrodt previously identified a material weakness in the company’s internal control over financial reporting related to review and approval controls over future cash flow forecasts used to develop certain management estimates, including those related to goodwill and other intangible assets. This control deficiency did not result in a material misstatement of the company’s current or prior period consolidated financial statements. As described in Mallinckrodt plc’s Form 10-Q for the fiscal quarter ended June 28, 2019, during the three months ended March 29, 2019, management of Mallinckrodt, under the oversight of the Mallinckrodt executive leadership team and those charged with governance, completed the remedial actions below to improve the company’s internal control over financial reporting and remediated the design of the material weakness:
| • | Continued to emphasize the importance of, and monitor the sustained compliance with, the execution of our internal controls over financial reporting through, among other activities, numerous meetings and trainings. |
| • | Enhanced, and will continue to enhance, the design of internal controls governing oversight and evaluation of future cash flow forecasts used to develop certain management estimates, including those related to goodwill and other intangible assets. |
| • | Tested the design effectiveness of the enhanced internal controls by performing them to re-evaluate the appropriateness, and test the accuracy, of information used to develop future cash flow forecasts in 2018. |
| • | Concluded the enhanced controls were designed effectively and developed a plan to implement them to support future cash flow forecasts in 2019. |
Although Mallinckrodt has remediated this material weakness in its internal controls over financial reporting, any failure to maintain effective internal control over financial reporting or disclosure controls and procedures could adversely affect the company’s ability to record, process and report financial information accurately, and to prepare financial statements within required time periods, which could subject Mallinckrodt to litigation or investigations requiring management resources and payment of legal and other expenses and could result in negative publicity or other negative actions that could harm investor confidence in the company’s financial statements. If any or all of these events occur, it could have a material adverse effect on Mallinckrodt’s business, financial condition, results of operations and cash flows or adversely affect the market price of the company’s ordinary shares.
Sales of Mallinckrodt’s products are affected by, and the company may be negatively impacted by any changes to, the reimbursement practices of governmental health administration authorities, private health coverage insurers and other third-party payers. In addition, reimbursement criteria or policies and the use of tender systems outside the U.S. could reduce prices for the company’s products or reduce the company’s market opportunities.
Sales of Mallinckrodt’s products depend, in part, on the extent to which the costs of the company’s products are reimbursed by governmental health administration authorities, private health coverage insurers and other third-party payers. The ability of patients to obtain appropriate reimbursement for products and services from these third-party payers affects the selection of products they purchase and the prices they are willing to pay. In the U.S., there have been, and the company expects there will continue to be, a number of state and federal proposals that limit the amount that third-party payers may pay to reimburse the cost of drugs, for example with respect to Acthar Gel. The company believes the increasing emphasis on managed care in the U.S. has and will continue to put pressure on the usage and reimbursement of Acthar Gel. Mallinckrodt’s ability to commercialize the company’s products depends, in part, on the extent to which reimbursement for the costs of these products is available from government healthcare programs, such as Medicaid and Medicare, private health insurers and others. The company cannot be certain that, over time, third-party reimbursements for the company’s products will be adequate for Mallinckrodt to maintain price levels sufficient for realization of an appropriate return on the company’s investment.
Reimbursement of highly-specialized products, such as Acthar Gel, is typically reviewed and approved or denied on a patient-by-patient, case-by-case basis, after careful review of details regarding a patient’s health and treatment history that is provided to the insurance carriers through a prior authorization submission, and appeal submission, if applicable. During this case-by-case review, the reviewer may refer to coverage guidelines issued by that carrier. These coverage guidelines are subject to on-going review by insurance carriers. Because of the large number of carriers, there are a large number of guideline updates issued each year.
Furthermore, demand for new products may be limited unless Mallinckrodt obtains reimbursement approval from governmental and private third-party payers prior to introduction. Reimbursement criteria, which vary by country, are becoming increasingly stringent and require management expertise and significant attention to obtain and maintain qualification for reimbursement.
In addition, a number of markets in which Mallinckrodt operates have implemented or may implement tender systems in an effort to lower prices. Under such tender systems, manufacturers submit bids which establish prices for products. The company that wins the tender receives preferential reimbursement for a period of time. Accordingly, the tender system often results in companies underbidding one another by proposing low pricing in order to win the tender. Certain other countries may consider implementation of a tender system. Even if a tender system is ultimately not implemented, the anticipation of such could result in price reductions. Failing to win tenders, or the implementation of similar systems in other markets leading to price declines, could have a material adverse effect on Mallinckrodt’s competitive position, business, financial condition, results of operations and cash flows.
Mallinckrodt is unable to predict what additional legislation or regulation or changes in third-party coverage and reimbursement policies may be enacted or issued in the future or what effect such legislation, regulation and policy changes would have on the company’s business. In May 2019, CMS issued a decision requiring that Mallinckrodt revert to the base date average manufacturer price used to calculate Medicaid drug rebates for Acthar Gel. Mallinckrodt subsequently filed suit in federal district court against the Agency seeking to hold unlawful and set aside this decision. Mallinckrodt plans to vigorously defend its position. If Mallinckrodt is unsuccessful in its efforts to set aside CMS’s decision, Medicaid net sales of Acthar Gel could be substantially eliminated and Mallinckrodt’s efforts to continue building on its investment in non-sales and marketing activities to modernize Acthar Gel could be significantly undermined.
Mallinckrodt may be unable to protect the company’s intellectual property rights, intellectual property rights may be limited or the company may be subject to claims that the company infringes on the intellectual property rights of others.
Mallinckrodt relies on a combination of patents, trademarks, trade secrets, proprietary know-how, market exclusivity gained from the regulatory approval process and other intellectual property to support the company’s business strategy, most notably in relation to Acthar Gel, Ofirmev® (acetaminophen) injection (“Ofirmev”), Inomax® (nitric oxide) gas, for inhalation (“Inomax”), Therakos® photopheresis (“Therakos”) and Amitiza products. However, Mallinckrodt’s efforts to protect the company’s intellectual property rights may not be sufficient. If Mallinckrodt does not obtain sufficient protection for the company’s intellectual property, or if Mallinckrodt is unable to effectively enforce the company’s intellectual property rights, or if there is a change in the way courts and regulators interpret the laws, rules and regulations applicable to the company’s intellectual property, Mallinckrodt’s competitiveness could be impacted, which could adversely affect the company’s competitive position, business, financial condition, results of operations and cash flows.
The composition patent for Acthar Gel has expired and the company has no patent-based market exclusivity with respect to any indication or condition the company might target. Mallinckrodt relies on trade secrets and proprietary know-how to protect the commercial viability and value of Acthar Gel. The company currently obtains such protection, in part, through confidentiality and proprietary information agreements. These agreements may not provide meaningful protection or adequate remedies for proprietary technology in the event of unauthorized use or disclosure of confidential and proprietary information. The parties may not comply with or may breach these agreements. Furthermore, Mallinckrodt’s trade secrets may otherwise become known to, or be independently developed by, competitors.
Certain patents related to the use of therapeutic nitric oxide for treating or preventing bronchoconstriction or reversible pulmonary vasoconstriction expired in 2013. Prior to their expiration, Mallinckrodt depended, in part, upon these patents to provide the company with exclusive marketing rights for the company’s product for some period of time. Since then, the company has obtained additional patents, which expire at various dates through 2036, including patents on methods of identifying patients at risk of serious adverse events when nitric oxide is administered to patients with particular heart conditions. Such methods have been approved by the FDA for inclusion in the Warnings and Precautions sections of the Inomax label. Other patents are on inhaled nitric oxide gas delivery systems as well as methods of using such systems, and on use of nitric oxide gas sensors. The Paragraph IV patent litigation trial against Praxair Distribution, Inc. and Praxair, Inc. (collectively “Praxair”) to prevent the marketing of its potential infringing nitric oxide drug product delivery system prior to the expiration of the patents covering Inomax was held in March 2017 and a decision was rendered September 5, 2017 that ruled five patents invalid and six patents not infringed. Mallinckrodt appealed the decision to the Court of Appeals for the Federal Circuit, which upheld the lower court’s decision on August 27, 2019. Mallinckrodt filed a petition for en banc review at the Federal Circuit on September 26, 2019. While Praxair received FDA approval of their Abbreviated New Drug Application (“ANDA”) for their Noxivent nitric oxide and clearance of their 510(k) for their NOxBOXi device on October 2, 2018, the Noxivent product received an AA-rating and the Noxivent label states that Noxivent must be delivered using the NOxBOXi device. The Court of Appeals’ decision with respect to the Praxair litigation ultimately could result in the launch of a competitive nitric oxide product before the expiration of the last of the patents listed in the FDA Orange Book, which could adversely affect the company’s ability to successfully maximize the value of Inomax and have an adverse effect on the company’s competitive position, business, financial condition, results of operations and cash flows.
The active ingredient in Ofirmev is acetaminophen. Patent protection is not available for the acetaminophen molecule itself in the territories licensed to Mallinckrodt, which include the U.S. and Canada. As a result, competitors who obtain the requisite regulatory approval can offer products with the same active ingredient as Ofirmev so long as the competitors do not infringe any process or formulation patents that the company has in-licensed from Bristol-Myers Squibb and its licensor, New Pharmatop LLC and any method-of-use patents that the company subsequently obtained. The latest expiration date of the in-licensed patents is 2021 whereas the latest expiration date of the subsequently obtained Company-owned patents is 2032. Settlement agreements have been reached in association with certain challenges to the in-licensed patents, which allow for generic competition to Ofirmev in December 2020, or earlier under certain circumstances.
Mallinckrodt’s Therakos products focus on extracorporeal photopheresis, which is an autologous immune cell therapy that is indicated in the U.S. for skin manifestations of cutaneous T-cell lymphoma (“CTCL”) and is available for several additional indications in markets outside the U.S. In the extracorporeal photopheresis (“ECP”) process, blood is drawn from the patient, separating white blood cells from plasma and red blood cells (which are immediately returned to the patient). The separated white blood cells are treated with an Ultraviolet-A (“UVA”) light activated drug, UVADEX®, followed by UVA radiation in the photopheresis instrument, prior to being returned to the patient. Patents related to the methoxsalen composition have expired. Therakos historically manufactured two photopheresis systems, the CELLEX® Photopheresis System (“CELLEX”), which is the only FDA-approved closed ECP system, and the UVAR XTS® Photopheresis System (“UVAR XTS”). While the company no longer manufactures the UVAR XTS system, disposable, sterile kits are still supplied to customers for each of the systems. The kits are single use and discarded after a treatment. Certain key patents related to the UVAR XTS system, disposable kit and overall photopheresis method expire in 2020. Key patents related to the CELLEX system, disposable kit and overall
photopheresis method expire in 2023. The company continues to pursue additional patentable enhancements to the Therakos ECP system. Patent applications were filed in 2016 relating to improvements to the CELLEX system, disposable kit and overall photopheresis method, that, if approved, may offer patent protection through approximately 2036.
Mallinckrodt’s pending patent applications may not result in the issuance of patents, or the patents issued to or licensed by the company in the past or in the future may be challenged or circumvented by competitors. Existing patents may be found to be invalid or insufficiently broad to preclude the company’s competitors from using methods or making or selling products similar or identical to those covered by the company’s patents and patent applications. Regulatory agencies may refuse to grant Mallinckrodt the market exclusivity that the company was anticipating, or may unexpectedly grant market exclusivity rights to other parties. In addition, the company’s ability to obtain and enforce intellectual property rights is limited by the unique laws of each country. In some countries it may be particularly difficult to adequately obtain or enforce intellectual property rights, which could make it easier for competitors to capture market share in such countries by utilizing technologies and product features that are similar or identical to those developed or licensed by Mallinckrodt. Competitors also may harm the company’s sales by designing products that mirror the capabilities of the company’s products or technology without infringing the company’s patents, including by coupling separate technologies to replicate what the company’s products accomplish through a single system. Competitors may diminish the value of the company’s trade secrets by reverse engineering or by independent invention. Additionally, current or former employees may improperly disclose such trade secrets to competitors or other third parties. Mallinckrodt may not become aware of any such improper disclosure, and, in the event the company does become aware, Mallinckrodt may not have an adequate remedy available to the company.
Mallinckrodt operates in an industry characterized by extensive patent litigation, and the company may from time to time be a party to such litigation.
The pursuit of or defense against patent infringement is costly and time-consuming and the company may not know the outcomes of such litigation for protracted periods of time. Mallinckrodt may be unsuccessful in the company’s efforts to enforce the company’s patent or other intellectual property rights. In addition, patent litigation can result in significant damage awards, including the possibility of treble damages and injunctions. Additionally, the company could be forced to stop manufacturing and selling certain products, or the company may need to enter into license agreements that require Mallinckrodt to make significant royalty or up-front payments in order to continue selling the affected products. Given the nature of the company’s industry, the company is likely to face additional claims of patent infringement in the future. A successful claim of patent or other intellectual property infringement against Mallinckrodt could have a material adverse effect on the company’s competitive position, business, financial condition, results of operations and cash flows.
The healthcare industry has been under increasing scrutiny from governments, legislative bodies and enforcement agencies related to sales, marketing and pricing practices, and changes to, or non-compliance with, relevant policies, laws, regulations or government guidance may result in actions that could adversely affect Mallinckrodt’s business.
In the U.S. over the past several years, a significant number of pharmaceutical and biotechnology companies have been subject to inquiries and investigations by various federal and state regulatory, investigative, prosecutorial and administrative entities in connection with the promotion of products for unapproved uses and other sales, marketing and pricing practices, including the U.S. Department of Justice (“DOJ”) and various other agencies including the Office of the Inspector General within the Department of Health and Human Services, the FDA, the Federal Trade Commission and various state Attorneys General offices. These investigations have alleged violations of various federal and state laws and regulations, including claims asserting antitrust violations, violations of the U.S. Federal Food, Drug and Cosmetic Act (“FFDCA”), the False Claims Act, the Prescription Drug Marketing Act, anti-kickback laws, data and patient privacy laws, export and import laws, consumer protection laws and other alleged violations in connection with the promotion of products for unapproved uses, pricing and Medicare and/or Medicaid reimbursement. The DOJ and the SEC have also increased their focus on the enforcement of the Foreign Corrupt Practices Act of 1977 (“FCPA”), particularly as it relates to the conduct of pharmaceutical companies.
Many of these investigations originate as “qui tam” actions under the False Claims Act. Under the False Claims Act, any individual can bring a claim on behalf of the government alleging that a person or entity has presented a false claim, or caused a false claim to be submitted, to the government for payment. The person bringing a “qui tam” suit is entitled to a share of any recovery or settlement. Qui tam suits, also commonly referred to as “whistleblower suits,” are often brought by current or former employees. In a qui tam suit, the government must decide whether to intervene and prosecute the case. If the government declines to intervene and prosecute the case, the individual may pursue the case alone. If the FDA or any other governmental agency initiates an enforcement action against Mallinckrodt or if the company is the subject of a qui tam suit and it is determined that the company violated prohibitions relating to the promotion of products for unapproved uses in connection with past or future activities, the company could be subject to substantial civil or criminal fines or damage awards and other sanctions such as the possible exclusion from federal healthcare programs including Medicare and Medicaid, consent decrees and corporate integrity agreements pursuant to which the company’s activities would be subject to ongoing scrutiny and
monitoring to ensure compliance with applicable laws and regulations. Any such fines, awards or other sanctions could have an adverse effect on Mallinckrodt’s competitive position, business, financial condition, results of operations and cash flows.
Specific to Mallinckrodt’s business, in September 2012, prior to the company’s acquisition of Questcor Pharmaceuticals, Inc. (“Questcor”) in August 2014, a subpoena was received from the U.S. Attorney’s Office (“USAO”) for the Eastern District of Pennsylvania, requesting documents pertaining to an investigation of its promotional practices. On or about March 8, 2019, the U.S. District Court for the Eastern District of Pennsylvania unsealed two qui tam actions involving the allegations under investigation by the USAO for the Eastern District of Pennsylvania. The DOJ intervened in both actions, which have since been consolidated. In September 2019, Mallinckrodt executed a settlement agreement to resolve the portion of the investigation and the litigation involving promotional practices for $15.4 million. If any of Mallinckrodt’s current practices related to the legacy Questcor business are found to be unlawful, the company will have to change those practices, which could have a material adverse effect on the company’s business, financial condition and results of operations. Further, if as a result of this investigation or litigation the company is found to have violated one or more applicable laws, the company could be subject to a variety of fines, penalties, and related administrative sanctions, and Mallinckrodt’s business, financial condition, results of operations and cash flows could be materially adversely affected.
In addition, there has recently been enhanced scrutiny of company-sponsored patient assistance programs, including insurance premium and co-pay assistance programs and donations to third-party charities that provide such assistance. If the company is deemed to have failed to comply with relevant laws, regulations or government guidance in any of these areas, the company could be subject to criminal and civil sanctions, including significant fines, civil monetary penalties and exclusion from participation in government healthcare programs, including Medicare and Medicaid, actions against executives overseeing the company’s business, and burdensome remediation measures. As discussed above, the USAO for the Eastern District of Pennsylvania is investigating this issue and the U.S. District Court for the Eastern District of Pennsylvania has unsealed two qui tam actions involving the allegations that are the subject of this investigation. In addition, in December 2016, the company received a subpoena from the USAO for the District of Massachusetts requesting documents related to the company’s support of 501(c)(3) organizations that provide financial assistance to patients and documents concerning the company’s provision of financial assistance to patients prescribed Acthar Gel. Other companies have disclosed similar inquiries. The company is cooperating with this inquiry. It is possible that any actions taken by the DOJ or one of the USAOs as a result of this inquiry or any future action taken by federal or local governments, legislative bodies and enforcement agencies on this subject could result in civil penalties or injunctive relief, negative publicity or other negative actions that could harm Mallinckrodt’s reputation, and could reduce demand for the company’s products and/or reduce coverage of the company’s products, including by federal healthcare programs such as Medicare and Medicaid and state health care, which would negatively impact sales of the company’s products. If any or all of these events occur, it could have an adverse effect on Mallinckrodt’s business, financial condition, results of operations and cash flows.
The DEA regulates the availability of controlled substances, including API, drug products under development and marketed drug products. At times, the procurement and manufacturing quotas granted by the DEA may be insufficient to meet Mallinckrodt’s needs.
The DEA is the U.S. federal agency responsible for domestic enforcement of the CSA. The CSA classifies drugs and other substances based on identified potential for abuse. Schedule I controlled substances, such as heroin and LSD, have a high abuse potential and have no currently accepted medical use; thus, they cannot be lawfully marketed or sold. Schedule II controlled substances include molecules such as oxycodone, oxymorphone, morphine, fentanyl, and hydrocodone. The manufacture, storage, distribution and sale of these controlled substances are permitted, but highly regulated. The DEA regulates the availability of API, products under development and marketed drug products that are in the Schedule II category by setting annual quotas. Every year, the company must apply to the DEA for manufacturing quota to manufacture API and procurement quota to manufacture finished dosage products. Given that the DEA has discretion to grant or deny the company’s manufacturing and procurement quota requests, the quota the DEA grants may be insufficient to meet Mallinckrodt’s needs. In 2018, manufacturing and procurement quotas granted by the DEA were sufficient to meet the company’s sales and inventory requirements on most products. In November 2017, the DEA reduced the amount of almost every Schedule II opiate and opioid medication that may be manufactured in the U.S. in 2018 by 20% and could take similar actions in the future. In December 2018, the DEA reduced the amount of the six most frequently misused opioids that may be manufactured in the U.S. in calendar year 2019 by an average of 10% as compared to the 2018 amount. On September 13, 2019, the DEA proposed that benzylfentanyl and 4-anilinopiperidine be controlled as list I chemicals under the CSA. On September 17, 2019, the DEA proposed to designate norfentanyl as an immediate precursor (i.e., a substance from which another is formed) for fentanyl and to make it a Schedule II controlled substance under the CSA. The DEA could take similar actions in the future. Future delay or refusal by the DEA to grant, in whole or in part, the company’s quota requests could delay or result in stopping the manufacture of the company’s marketed drug products, new product launches or the conduct of bioequivalence studies and clinical trials. Such delay or refusal also could require Mallinckrodt to allocate marketed drug products among the company’s customers.
These factors, along with any delay or refusal by the DEA to provide customers who purchase API from the company with sufficient quota, could have a material adverse effect on Mallinckrodt’s competitive position, business, financial condition, results of operations and cash flows.
Mallinckrodt’s reporting and payment obligations under the Medicare and Medicaid rebate programs, and other governmental purchasing and rebate programs, are complex. Any determination of failure to comply with these obligations or those relating to healthcare fraud and abuse laws could have a material adverse effect on the company’s business.
The regulations regarding reporting and payment obligations with respect to Medicare and Medicaid reimbursement programs, and rebates and other governmental programs, are complex. Because Mallinckrodt’s processes for these calculations and the judgments used in making these calculations involve subjective decisions and complex methodologies, these accruals may have a higher inherent risk for material changes in estimates. In addition, they are subject to review and challenge by the applicable governmental agencies, and it is possible that such reviews could result in material adjustments to amounts previously paid. See “Sales of Mallinckrodt’s products are affected by, and the company may be negatively impacted by any changes to, the reimbursement practices of governmental health administration authorities, private health coverage insurers and other third-party payers. In addition, reimbursement criteria or policies and the use of tender systems outside the U.S. could reduce prices for the company’s products or reduce the company’s market opportunities.”
Any governmental agencies that have commenced, or may commence, an investigation of Mallinckrodt relating to the sales, marketing, pricing, quality or manufacturing of pharmaceutical products could seek to impose, based on a claim of violation of fraud and false claims laws or otherwise, civil and/or criminal sanctions, including fines, penalties and possible exclusion from federal healthcare programs including Medicare and Medicaid. Some of the applicable laws may impose liability even in the absence of specific intent to defraud. Furthermore, should there be ambiguity with regard to how to properly calculate and report payments, and even in the absence of any such ambiguity, a governmental authority may take a position contrary to a position the company has taken, and may impose civil and/or criminal sanctions. For example, from time to time, state attorneys general have brought cases against the company that allege generally that the company and numerous other pharmaceutical companies reported false pricing information in connection with certain drugs that are reimbursable under Medicaid, resulting in overpayment by state Medicaid programs for those drugs, and generally seek monetary damages and attorneys’ fees. Any such penalties or sanctions that the company might become subject to in this or other actions could have a material adverse effect on Mallinckrodt’s competitive position, business, financial condition, results of operations and cash flows.
The terms of the agreements that govern Mallinckrodt’s indebtedness restrict Mallinckrodt’s current and future operations, particularly the company’s ability to respond to changes or to pursue the company’s business strategies.
The agreements that govern the terms of Mallinckrodt’s indebtedness contain (and the indentures governing the New Notes will contain) a number of restrictive covenants that impose significant operating and financial restrictions on the company and may limit the company’s ability to engage in acts that may be in Mallinckrodt’s long-term best interest, including limitations or restrictions on the company’s ability to:
| • | incur, assume or guarantee additional indebtedness; |
| • | declare or pay dividends, make other distributions with respect to equity interests, or purchase or otherwise acquire or retire equity interests; |
| • | make any principal payment on, or redeem or repurchase, subordinated debt; |
| • | make loans, advances or other investments; |
| • | sell or otherwise dispose of assets, including capital stock of subsidiaries; |
| • | incur liens; |
| • | enter into transactions with affiliates; |
| • | enter into sale and lease-back transactions; and |
| • | consolidate or merge with or into, or sell all or substantially all of Mallinckrodt’s assets to, another person or entity. |
In addition, the restrictive covenants in the Existing Senior Secured Credit Facilities require Mallinckrodt to comply with a financial maintenance covenant in certain circumstances. Mallinckrodt’s ability to satisfy this financial maintenance covenant
can be affected by events beyond the company’s control and the company cannot assure you that Mallinckrodt will be able to comply.
A breach of the covenants under the agreements that govern the terms of any of Mallinckrodt’s indebtedness could result in an event of default under the applicable indebtedness. Such default may allow the creditors to accelerate the related debt and may result in the acceleration of any other debt to which a cross-acceleration or cross-default provision applies. In addition, an event of default under the Existing Senior Secured Credit Facilities would permit the lenders under such facilities to terminate all commitments to extend further credit thereunder. Furthermore, if Mallinckrodt is unable to repay the amounts due and payable under the Existing Senior Secured Credit Facilities or the New Notes, those lenders or investors will be able to proceed against the collateral granted to them to secure that indebtedness. If Mallinckrodt’s debtholders accelerate the repayment of the company’s borrowings, the company may not have sufficient assets to repay that indebtedness.
As a result of these restrictions, Mallinckrodt may be:
| • | limited in how Mallinckrodt conducts the company’s business; |
| • | unable to raise additional debt or equity financing to operate during general economic or business downturns; or |
| • | unable to compete effectively, execute the company’s growth strategy or take advantage of new business opportunities. |
These restrictions may affect Mallinckrodt’s ability to grow in accordance with the company’s plans.
If the conditions to the Exchange Offers are not met, the Exchange Offers may not be completed, in which case the Issuers’ liquidity may be limited and the Issuers may be unable to pay principal and interest on the Existing Notes when due.
Consummation of each Exchange Offer is subject to the satisfaction or waiver of a number of conditions. Each of the Issuers and Mallinckrodt is highly leveraged. In the event that the Exchange Offers and the transactions contemplated by the Exchange Agreement are not completed, the Issuers and Mallinckrodt will remain highly leveraged, which may result in the inability to pay principal and interest on the Existing Notes when due, which may result in you not realizing a full recovery on your investment in the Existing Notes, or an event of default under the terms of the Existing Notes, which may lead to acceleration of our other indebtedness by the holders thereof.
If the proposed amendments to the indentures governing the Existing Notes become operative, the future subsidiaries of Mallinckrodt plc will not be required to guarantee the Existing Notes and such future subsidiaries may incur significant indebtedness, and the Issuers and the existing subsidiary guarantors of the Existing Notes may make investments in or transfer assets to such non-guarantor subsidiaries.
The proposed amendments to the indentures governing the Existing Notes would eliminate the requirement that any of Mallinckrodt plc’s future subsidiaries become guarantors of the Existing Notes. As a result, the Existing Notes will be structurally subordinated to any indebtedness of any such future subsidiaries of Mallinckrodt plc, including, as applicable, the New Notes. The indentures governing the Notes (as amended by the proposed amendments) will not limit the transfer of assets to, or investments in, any such non-guarantor subsidiaries. There can be no assurance that the Issuers and the subsidiary guarantors of the Existing Notes will not transfer significant amounts of assets to, or make significant investments in, such non-guarantor subsidiaries, or any other persons.
We will incur significant costs in conducting the Exchange Offers and Consent Solicitations.
The Exchange Offers and Consent Solicitations have resulted, and will continue to result, in significant costs to us, including advisory and professional fees paid in connection with evaluating our alternatives under the Existing Notes and pursuing the Exchange Offers and Consent Solicitations.
USE OF PROCEEDS
The Issuers will not receive any cash proceeds from the Exchange Offers. The Existing Notes tendered to and accepted by the Issuers in connection with the terms and conditions of the Exchange Offers will be concurrently retired and cancelled and will not be reissued.
CAPITALIZATION
The following table sets forth Mallinckrodt’s debt capitalization as of the date of the Offering Memorandum (i) on an actual basis, (ii) pro forma for the consummation of the Exchange Offers (assuming 50.1% participation in each of the Exchange Offers prior to the Early Delivery Time) and the consummation of the transactions contemplated by the Exchange Agreement, and (iii) pro forma for the consummation of the Exchange Offers (assuming 100% participation in each of the Exchange Offers prior to the Early Delivery Time) and the consummation of the transactions contemplated by the Exchange Agreement. The information in this table should be read in conjunction with the consolidated financial statements and the related notes contained in the Annual Report and the Quarterly Reports, incorporated by reference in this Offering Memorandum.
As of the date of this Offering Memorandum Actual | Pro Forma for Exchange Offers (50.1%)(1) | Pro Forma for Exchange Offers (100%) | ||||
| (in millions) | (in millions) | (in millions) | ||||
| Debt (Aggregate Principal): | ||||||
| Existing Senior Secured Credit Facilities | ||||||
| Revolving Credit Facility | $900.0 | $900.0 | $900.0 | |||
| Existing Term Loans | 1,924.4 | 1,924.4 | 1,924.4 | |||
| New Notes offered pursuant to the Exchange Offers | — | 818.1 | 1,118.3 | |||
| Existing 4.875% 2020 Notes | 698.0 | 348.3 | — | |||
| Existing 5.750% 2022 Notes | 663.2 | 330.9 | 358.9 | |||
| Existing 4.750% 2023 Notes | 350.1 | 91.4 | 91.4 | |||
| Existing 5.625% 2023 Notes | 659.4 | 329.0 | 303.5 | |||
| Existing 5.500% 2025 Notes | 596.1 | 297.5 | 282.0 | |||
Other Mallinckrodt debt(2) | 14.8 | 14.8 | 14.8 | |||
| Variable-rate receivables securitization | — | — | — | |||
| Total debt | $5,806.0 | $5,054.4 | $4,993.3 | |||
Note: Debt amounts presented are face values and do not deduct unamortized discount or debt issuance costs.
(1) Assumes all principal of Exchanging Holders is tendered and accepted, plus additional tenders if necessary to reach 50.1% participation for each series. |
(2) Includes approximately $10 million of 9.500% Debentures due 2022 and approximately $4 million 8.000% Debentures due 2023.
CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING STATEMENTS
Statements in this Offering Memorandum that are not strictly historical, including statements regarding future financial condition and operating results, economic, business, competitive and/or regulatory factors affecting Mallinckrodt’s businesses, plans for the Specialty Generics business (including the suspension of the previously announced plans to spin off that business), and any other statements regarding events or developments the company believes or anticipates will or may occur in the future, may be “forward-looking” statements within the meaning of the Private Securities Litigation Reform Act of 1995, and involve a number of risks and uncertainties.
There are a number of important factors that could cause actual events to differ materially from those suggested or indicated by such forward-looking statements and you should not place undue reliance on any such forward-looking statements. These factors include risks and uncertainties related to, among other things: general economic conditions and conditions affecting the industries in which Mallinckrodt operates; the commercial success of Mallinckrodt’s products; conditions that could necessitate an evaluation of Mallinckrodt’s intangible assets for possible impairment; changes in laws and regulations; Mallinckrodt’s ability to successfully integrate acquisitions of operations, technology, products and businesses generally and to realize anticipated growth, synergies and cost savings; Mallinckrodt’s and Mallinckrodt’s licensers’ ability to successfully develop or commercialize new products; Mallinckrodt’s and Mallinckrodt’s licensers’ ability to protect intellectual property rights; Mallinckrodt’s ability to receive procurement and production quotas granted by the U.S. Drug Enforcement Administration (“DEA”); customer concentration; Mallinckrodt’s reliance on certain individual products that are material to its financial performance; cost containment efforts of customers, purchasing groups, third-party payers and governmental organizations; the reimbursement practices of a small number of public or private insurers; pricing pressure on certain of Mallinckrodt’s products due to legal changes or changes in insurers’ reimbursement practices resulting from recent increased public scrutiny of healthcare and pharmaceutical costs; limited clinical trial data for Acthar® Gel (repository corticotropin injection) (“Acthar Gel”); complex reporting and payment obligations under healthcare rebate programs; Mallinckrodt’s ability to navigate price fluctuations; future changes to U.S. and foreign tax laws; Mallinckrodt’s ability to achieve expected benefits from restructuring activities; complex manufacturing processes; competition; product liability losses and other litigation liability; ongoing governmental investigations; material health, safety and environmental liabilities; retention of key personnel; conducting business internationally; the effectiveness of information technology infrastructure; cybersecurity and data leakage risks; Mallinckrodt’s substantial indebtedness and its ability to generate sufficient cash to reduce its indebtedness; actions, including any future actions, taken with respect to the Specialty Generics business; and Mallinckrodt’s ability to complete the Exchange Offers, the Consent Solicitations and the transactions contemplated by the Exchange Agreement, including the expected timing of completion of the Exchange Offers and receipt of requisite consents in the Consent Solicitations.
These and other factors are identified and described in more detail in the “Risk Factors” section of this Offering Memorandum. The forward-looking statements made herein speak only as of the date hereof. Neither Mallinckrodt plc nor the Issuers assume any obligation to update or revise any forward-looking statement, whether as a result of new information, future events and developments or otherwise, except as required by law.