 4Q14 BioMarin Pharmaceutical Inc. Exhibit 99.1 |
 2 Safe Harbor Statement This non-confidential presentation contains ‘forward-looking statements’ about the business prospects of BioMarin Pharmaceutical Inc., including potential future products in different areas of therapeutic research and development. Results may differ materially depending on the progress of BioMarin’s product programs, actions of regulatory authorities, availability of capital, future actions in the pharmaceutical market and developments by competitors, and those factors detailed in BioMarin’s filings with the Securities and Exchange Commission such as 10-Q, 10-K and 8-K reports. |
 3 Additional Information The offer by BioMarin to purchase ordinary shares of Prosensa Holding N.V. (“Prosensa”) contemplated by the Purchase Agreement, dated November 23, 2014, between BioMarin, Prosensa and BioMarin Falcons B.V. (the “Offer”) has not yet commenced. This communication is neither an offer to purchase nor a solicitation of an offer to sell any ordinary shares of Prosensa or any other securities. On the commencement date of the Offer, a tender offer statement on Schedule TO, including an offer to purchase, a letter of transmittal and related documents, will be filed with the United States Securities and Exchange Commission (the “SEC”). Thereafter, Prosensa will file a solicitation/recommendation statement on 14D-9 with the SEC. The offer to purchase ordinary shares of Prosensa will only be made pursuant to the offer to purchase, the letter of transmittal and related documents filed as a part of the Schedule TO. INVESTORS AND SECURITY HOLDERS OF PROSENSA ARE URGED TO READ BOTH THE SCHEDULE TO (AND THE INCLUDED OFFER TO PURCHASE) AND THE SOLICITATION/ RECOMMENDATION STATEMENT, AS THEY MAY BE AMENDED FROM TIME TO TIME, AND OTHER RELEVANT DOCUMENTS FILED WITH THE SEC WHEN THEY BECOME AVAILABLE BEFORE THEY MAKE ANY DECISION WITH RESPECT TO THE TENDER OFFER, BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION ABOUT THE TERMS OF THE OFFER, THE PROPOSED TRANSACTIONS AND THE PARTIES THERETO. The tender offer statement will be filed with the SEC by BioMarin and the solicitation/recommendation statement will be filed with the SEC by Prosensa. Investors and security holders may obtain a free copy of these statements (when available) and other documents filed with the SEC at the website maintained by the SEC at www.sec.gov or by directing such requests to the information agent for the tender offer that will be named in the tender offer statement. |
 4 BioMarin is the Industry Leader In Orphan Drugs 2014 Year-to-date: Strong Growth Across the Organization Approved Products to Drive $1B+ in Revenues in Next 2-3 Years Potentially Pivotal data for PEG PAL, BMN 701 and BMN 190 in 2015 5 Marketed Products to Generate $700M-$710M in 2014 Revenues* 5 Potential Products Currently in the Clinic * Increased from prior range of $650M-$680M in 1Q14 |
 5 “Orphan” Diseases Represent a Large Unmet Need • Orphan Drug Act in US (1983) provides incentives for research and development • Orphan drugs can receive rapid approval and high levels of reimbursement Medical Advances Create Opportunities for Orphan Drugs 1 NIH and EORDIS Many rare diseases • Collectively rare diseases impact tens of millions of people in the U.S. alone • ~ 7K rare diseases 1 ; 250+ new rare diseases discovered per annum • Innovative and effective products for Orphan diseases have pricing leverage Supportive regulatory environment and incentives Flexible pricing for certain therapies |
 6 5 Years /$300M IND Filing (or Equivalent) 5 years 5 Years/$102M 3 Years/$59M 5 Years/$100M Efficient Product Development Track Record IND to Approval is Less than Half the Industry Average Time to Approval 10 years |
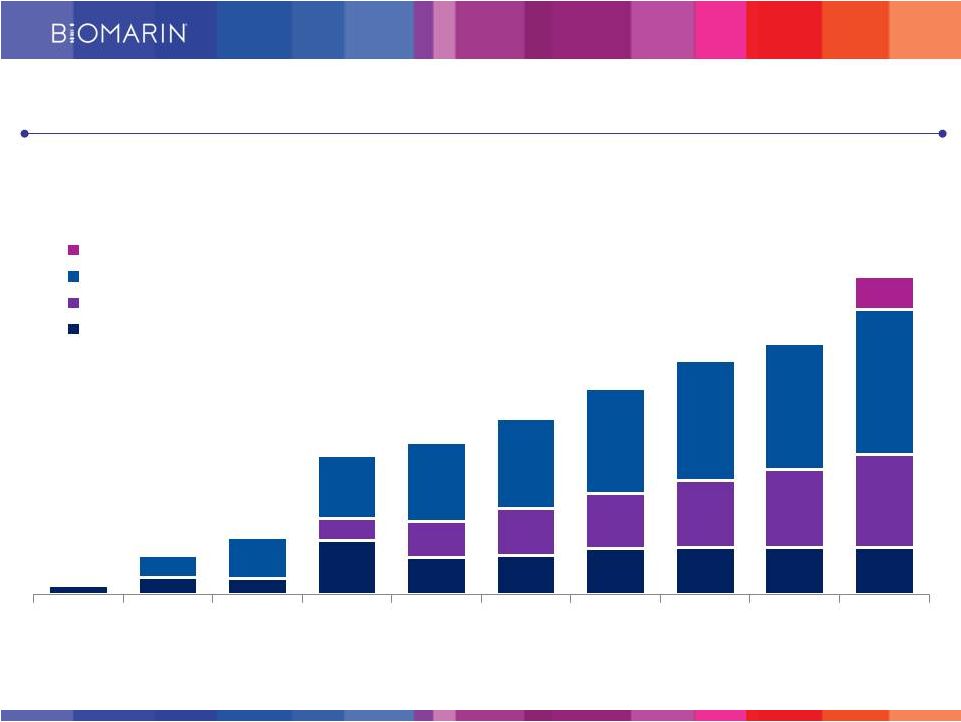 7 Core Commercial Products Expected to Continue Growing (revenues in millions) $305 - $320 $65- $70 $26 $84 $122 $297 $325 $376 $441 $501 $549 $700 -$710 2005 2006 2007 2008 2009 2010 2011 2012 2013 2014 Outlook VIMIZIM Naglazyme Kuvan Aldurazyme + Other $190 - $200 History of Strong Revenue Growth |
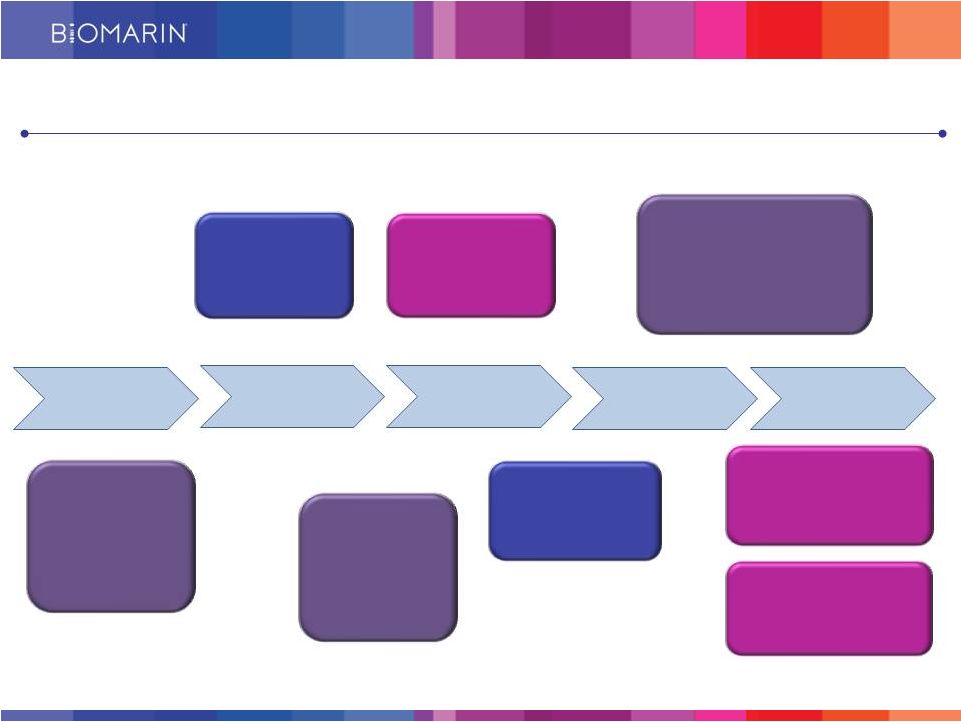 8 4Q14 BMN 190: Batten Disease Phase 1/2 enrollment complete 2Q15 BMN 111: Phase 2 data in Achondroplasia 1H15 BMN 701: Pompe Disease enrollment complete 4Q15 PEG PAL (BMN 165): Phase 3 data in PKU 2015: Advancement across the Development Portfolio 2H15 talazoparib (BMN 673): PARP inhibitor for mBC Phase 3 enrollment complete Mid 2015 BMN 250: MPS IIIB IND filing 1Q15 BMN 270: Hemophilia A IND filing 1Q15 2Q15 3Q15 4Q15 4Q14 4Q15 BMN 190: Phase 1/2 data in Batten Disease |
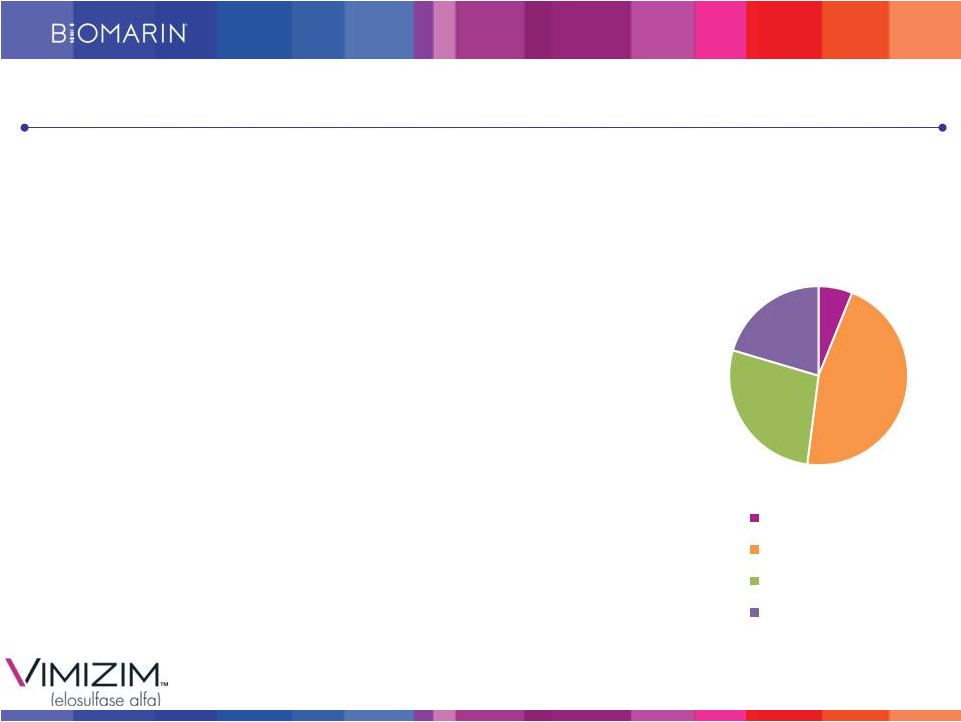 9 Patient Starts Robust Since Recent Approvals • Patients on commercial therapy in 15 countries, as of September 30, 2014 • On track to achieve FY 2014 Guidance of $65 - $70M Over 1,600 patients identified/3,000 WW (est.) Leveraging the Naglazyme Experience • Source of most of currently identified patients • Expected to prescribe and infuse VIMIZIM Peak VIMIZIM Sales Expected to Drive Profitability Patient Distribution * BioMarin estimates 5% 20% 30% 45% APAC EUMEA Latin America North America VIMIZIM: Our Next Successful Commercial Brand |
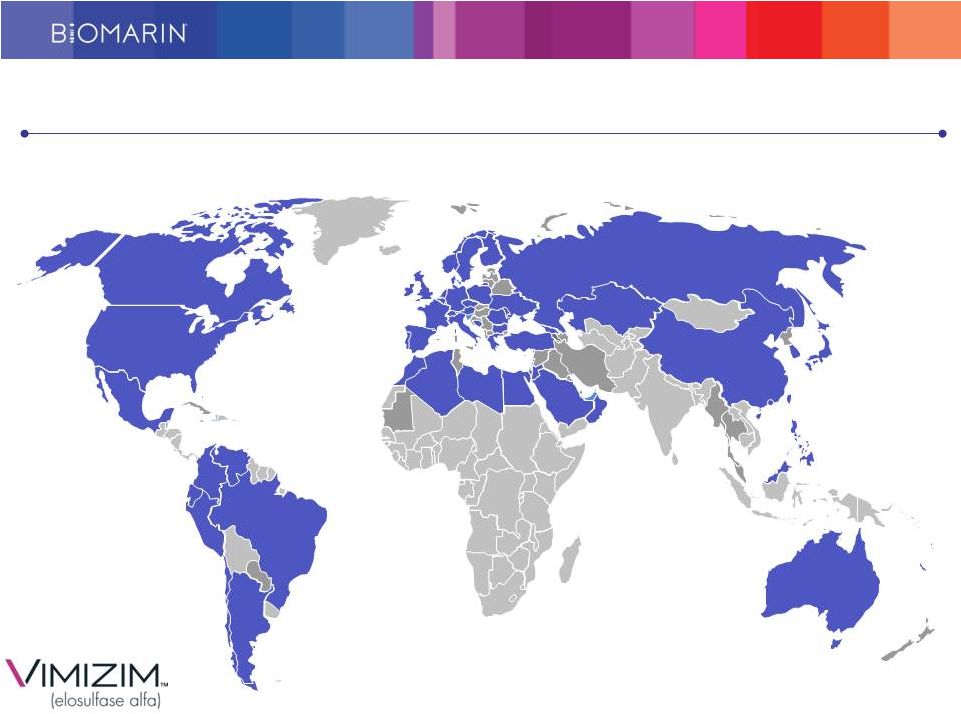 Leveraging our Worldwide Commercial Infrastructure Presence in 50 countries with 190+ FTEs 10 |
 11 VIMIZIM Approved in the U.S. and E.U. to Treat MPS IV Patient Starts Robust Since Recent Approvals FDA Approval on February 14; E.C. on April 28 Commercial roll-out considerations • FY 2014 Guidance of $65 - $70M • Reimbursement approval timing variable by market • Named patient sales open in France under ATU approval • U.S. and E.U. approval enables launch of named patient sales programs in other regions • Key is number of patient starts Leveraging our Naglazyme experience and reach • More patients seeking VIMIZIM in the U.S. than patients currently on commercial Naglazyme in the U.S. |
 12 Phenylketonuria (PKU) Represents a Significant Unmet Medical Need PKU Summary • Inherited metabolic disorder caused by deficiency of the phenylalanine hydroxylase (PAH) enzyme • PAH is required for the breakdown of the amino acid phenylalanine (Phe) into tyrosine • Elevated Phe levels are toxic to the brain • Primary treatment includes highly restrictive diet and nutritional supplementation • ~ 50,000 patients in the developed world; diagnosed through newborn screening Kuvan ® (sapropterin dihydrochloride) Oral, small molecule (6R- BH4) co-factor for PAH For BH4-responsive PKU patients Powder for oral solution recently approved PEG-PAL (phenylalanine ammonia lyase) Enzyme substitution therapy Targeted for patients with PKU age 16 and older with elevated Phe levels |
 13 Guideline highlights: • Treatment of PKU should be life long • Treatment should include pharmacotherapy • Every PKU patient should be offered a trial of Kuvan • Encourages medical insurance to provide coverage for both medications and medical foods regardless of age Treatment Guidelines for PKU Just Published Beneficial to BioMarin PKU Franchise |
 14 14 PEG PAL Market Opportunity is Adult PKU Patients Average Price for Adult Patients on Kuvan $180K/yr *BioMarin estimates ~ 10,000* Total U.S. PKU Patients >18 years old ~ 850 Currently on Kuvan ~ 6,000 Not in clinic ~ 1,500 in PKU clinic ~ 1,850 in BMRN data base ~ 9,150 not on Kuvan |
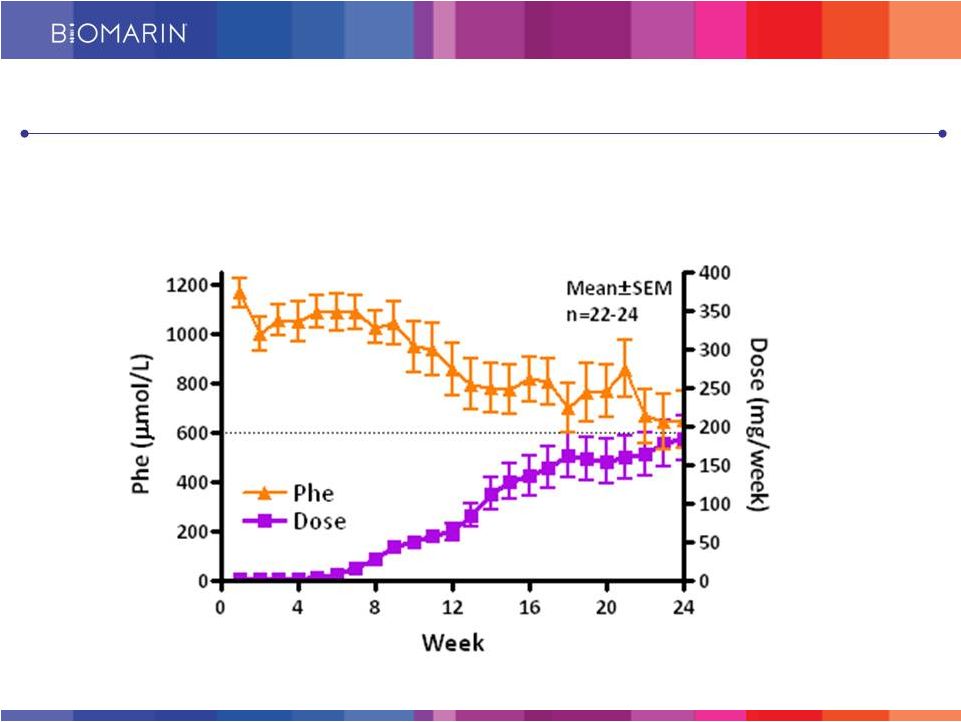 15 PEG PAL Meaningfully Reduces Phe Levels in Patients Approximately 50% Reduction within 24 weeks in Most Recent Phase 2 Study With No Diet Restrictions |
 16 PEG PAL Pre-filled Syringe For Self Administration Plunger Finger flange Needle safety guard Better Compliance and Easier to Use |
 17 PEG PAL Approval Plan Pivotal Study Underway; Data 4Q15 301 Open Label Study 302 Pivotal Study Endpoints • Targets fast responders for pivotal study enrollment • Open-label, multi-center study to access safety and tolerability of PEG PAL by naïve adult PKU patients • Requires pre-specified reduction in blood Phe for entry • Double-blind, placebo controlled, randomized discontinuation study to evaluate the efficacy and safety of PEG PAL by adults with PKU • N=120 • Primary endpoint: Phe lowering • Secondary endpoint: neurocognitive benefit (features of inattention) |
 18 PARP Inhibitor Talazoparib (BMN 673) 100x More Efficient at Trapping PARP-DNA Complexes* Phase 1/2 gBRCA Breast Cancer • RECIST response rate currently 50% (7/14) patients on Phase 3 dose of 1mg/day • Median PFS is estimated to be 31 weeks • Median duration of response is currently estimated to be 40.3 weeks, with 8 patients still on therapy as of SABC 2013 gBRCA Ovarian Cancer • RECIST response rate currently 44% (11/25) patients on Phase 3 dose of 1mg/day Small Cell Lung Cancer • 2 patients have had partial responses on Phase 3 dose of 1mg/day • Expansion phase target enrollment is 22 patients * by Drs. Yves Pommier, Junko Murai, Shar-yin N. Huang, Amèlie Renaud, et al., “Stereospecific PARP trapping by BMN 673 and comparison with olaparib and rucaparib”, Molecular Cancer Therapeutics; Published Online First December 19, 2013; doi:10.1158/1535-7163.MCT-13-0803 |
 19 Talazoparib: Phase 3 Trial Underway Enrolling metastatic breast cancer, gBRCA mutation carriers • Two or fewer lines of prior chemotherapy in metastatic setting • Myriad Genetics’ BRCAnalysis ® test being used to select patients Study Design: Two arms • 1 mg talazoparib once daily • Physician’s choice of capecitabine, gemcitabine, eribulin, or vinorelbine n = 430, 2:1 randomization • Powered for a 0.67 hazard ratio to improve median PFS from 16 weeks in the control arm to 24 weeks in the drug arm Primary endpoint: Progression-free survival Secondary endpoint: Overall survival Enrollment Target: Complete enrollment in 2H15 |
 20 Talazoparib Opportunities for Program Expansion Broadening Breast Cancer Program • Neoadjuvant setting Patients Beyond BRCA • Homologous recombination repair defects (HRD) • Research Collaboration with Myriad Genetics Other Tumor Types • SCLC – Ph 1/2 data is preliminary, but encouraging • Ovarian – Ph 1/2 data supports Phase 3 program Talazoparib in Combination |
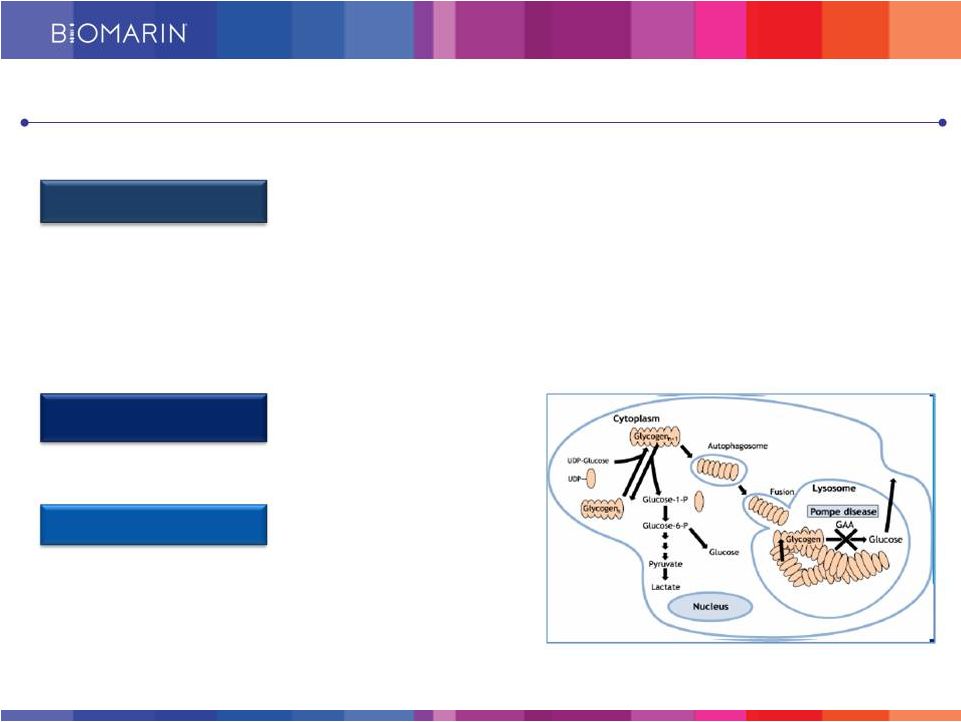 21 BMN 701 for Late Onset Pompe Disease Current Marketed Therapies Have Limited Efficacy Pompe Disease Market Standard of Care • Caused by a deficiency of acid alfa glucosidase (GAA) • GAA breaks down glycogen (stored glucose) • Glycogen accumulates in muscles due to low/absent GAA in Pompe • Respiratory muscle weakness causes respiratory impairment, leading cause of mortality and morbidity • 3,000 to 6,000 patients in the developed world • Myozyme/Lumizyme generating ~ $700M in global sales • Suboptimal uptake into the cells • Unmet medical need remains |
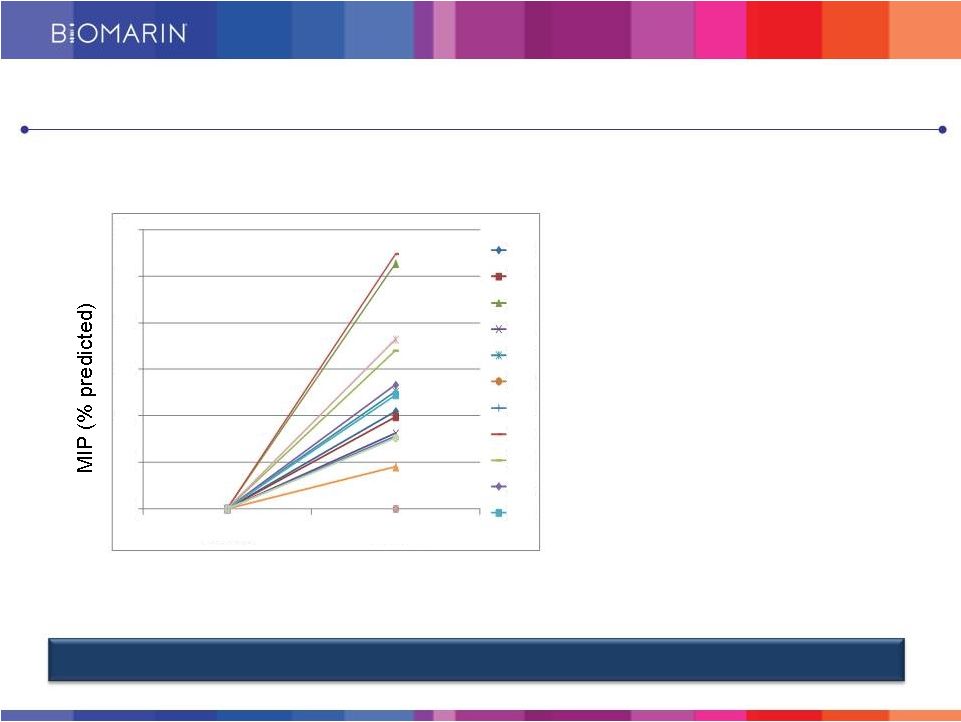 Compelling Clinical Activity Seen in Phase 1/2 Study Clinical Activity of BMN 701 Demonstrated in Prior Phase 1/2 Study POM-001, n = 16 (20 mg/kg cohort) Respiratory muscle strength 6MWT • High responders: 3 out of 16 patients demonstrated >75 m increase in 6MWT over baseline (18.8%) • Mean increase of 22.3 m in 6MWT over baseline 22 0 5 10 15 20 25 30 Baseline Week 24 1007 1008 1009 1010 1011 1012 1013 1014 1015 1016 1017 |
 23 BMN-701 Phase 2/3 Trial Patients • Single-arm Phase 2/3 switching trial in late-onset Pompe patients previously treated with alglucosidase alfa • Primary endpoint: MIP Next Steps MIP and MEP • ~ 55%-79% of adults with Pompe disease demonstrate respiratory impairment 1 • Annually, patients average decreases in MIP of 3.2% points (p=0.018) and MEP of 3.8% points (p=0.01) 2 • 45% of patients in the Pompe registry require respiratory support 3 • ~ 8% increase in likelihood of ventilation following diagnosis 4 ( 1 Schuller et al 2011, van der Beek 2011; 2 van der Beek 2011; 3 Byrne et al 2011; 4 Toscano/Schoser 2012) Study Designed to Assess Respiratory Muscle Strength |
 24 BMN 111 for Achondroplasia Analog of C-type natriuretic peptide; positive regulator of bone growth Achondroplasia Phase 1 Findings Phase 2 Plans • Most common form of dwarfism • Autosomal dominant activating mutation in the fibroblast growth factor receptor 3 (FGFR3) • 18K - 24K patients in the U.S./EU, estimated addressable market ~ 25% • Generally well-tolerated, all AEs were of mild severity • Systemic exposure similar at doses observed to cause growth in healthy and disease model animals • Global Phase 2 study in children 5-12 years old has begun |
 25 BMN 111 for Achondroplasia Phase 2 Study Initiated Cohort 1: 2.5µg/kg 8 patients for 6 months 1 111-901 subjects roll into 111-202 cohorts after completion of 6-months of baseline growth velocity data Cohort 2: 7.5µg/kg 8 patients for 6 months Cohort 3: 15µg/kg 8 patients for 6 months Open Label Extension Safety Study Baseline Growth Natural History Study 1 Phase 2 BMN 111-202 BMN 111-901 BMN 111-203 |
 26 • Enzyme replacement therapy • Treats neurological symptoms by delivering the TPP1 enzyme centrally via ICV • Enzyme widely distributes in central nervous system BMN 190 for Late Infantile CLN2 Batten Disease Potential Rapid Development Path for a Devastating Childhood Disease Batten Disease BMN 190 • Autosomal recessive disease caused by mutations in the CLN2 gene • Disease onset ages 2-4, death by ages 8-12 • Cognitive impairment, visual failure, seizures, and deteriorating motor development, leading to a vegetative state • ~ 400-600 cases worldwide Phase 1/2 • Initiated Phase 1/2 in 3Q13 • All patients receive active treatment every two weeks via 2-4 hour infusion • Potential for rapid approval filing – single study may be sufficient if data is compelling |
 27 BMN 250 for MPS IIIB (Sanfilippo B syndrome) – NAGLU-IGF2 MPS IIIB BMN 250 Next Steps • Lysosomal storage disease, caused by a deficiency in the enzyme alpha-N- acetylglucosaminidase (NAGLU) • Symptoms appear between ages 2-6 years and include behavior disorders, intellectual deterioration, sleep disorders, mild dysmorphism; death typically occurs between 30-40 years • Estimated 1,000-2,000 patients in developed world • Complete IND-enabling studies • IND filing and initiation of Phase 1/2 trial in mid-2015 • Enzyme replacement therapy using recombinant NAGLU with GILT tag • Delivered directly to the brain • Leverages BioMarin know-how and established IP 27 IND Candidate Announced 1Q14 |
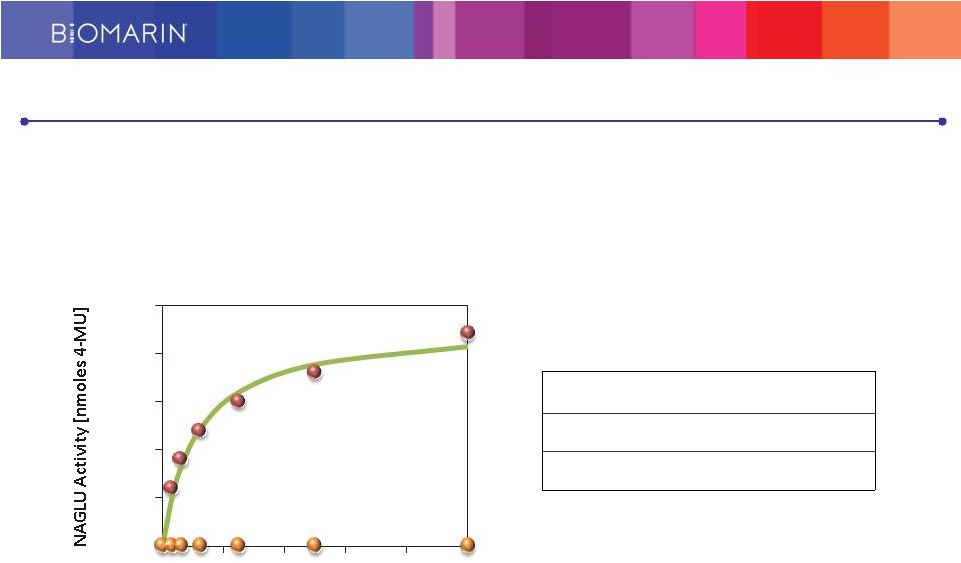 rhNAGLU-IGF2 is Effectively Taken up by MPS IIIB fibroblasts via Glycosylation Independent Lysosomal Targeting (GILT) Pathway K uptake [nM] rhNAGLU-IGF2 5.4 ± 0.94 rhNAGLU No uptake rhNAGLU-IGF2 = recombinant human alpha-N-acetylglucosaminidase fusion protein with insulin-like growth factor 2 BMN 250 Effectively Taken Up in MPS IIIB Fibroblasts 0 0.05 0.1 0.15 0.2 0.25 0 10 20 30 40 50 NAGLU Protein [nM] Cell uptake of rhNAGLU-IGF2 28 |
 29 Potential Gene Therapy Solution for Hemophilia A BMN 270 for Hemophilia A -- AAV Factor VIII Vector Hemophilia A Standard of Care Next Steps • Hereditary disease resulting from a deficiency of clotting factor VIII • Severe hemophilia is a progressive disease • 90,000 patients in BioMarin commercial territories • For the 60% of severe patients, prophylactic regimen of factor VIII replacement therapy consisting of intravenous infusions 3x/week • Many receiving replacement therapy still have bleeding events and debilitating joint damage • Complete IND-enabling studies in 2014 • IND filing and initiate Phase 1/2 trial in 1Q15 |
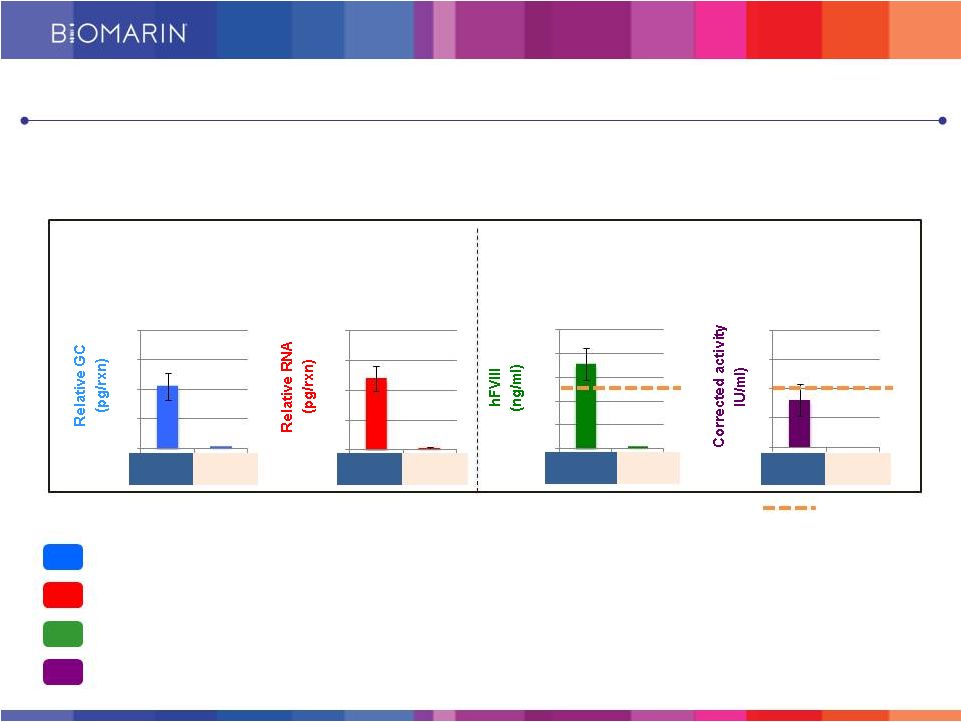 30 DNA RNA Protein Activity AAV FVIII #2 Vehicle AAV FVIII #2 Vehicle AAV FVIII #2 Vehicle AAV FVIII #2 Vehicle Assessment in Liver Tissue in Factor VIII Deficient Mice Assessment in Plasma in Factor VIII Deficient Mice BMN 270 Restores Factor VIII Plasma Concentration to Levels Projected to be Adequate for Normal Clotting in Humans Findings: Vector DNA delivered successfully to the liver DNA is transcriptionally active - human Factor VIII RNA is detected in the liver tissue Human Factor VIII protein can be detected at near normal levels in the plasma Factor VIII clotting action is detected at near normal levels Normal levels 0.0 0.2 0.4 0.6 0.8 0 50 100 150 200 250 0.0 0.5 1.0 1.5 2.0 0.0 0.4 0.8 1.2 1.6 Next IND: FVIII Gene Therapy for Hemophilia A |
 31 Manufacturing Capabilities with Room to Grow • Novato facility meets projected commercial needs through at least 2017 for Aldurazyme, Naglazyme, Vimizim and PEG-PAL • Shanbally, Ireland facility acquired in 2011 • Build-out has begun to support Vimizim peak demand and additional products |
 32 2014: Updated Financial Guidance Revenue Guidance ($ in millions) Provided Updated Item August 26, 2014 October 23, 2014 Total BioMarin Revenues $680 to $700 $700 to $710 Naglazyme Net Product Revenue $305 to $320 Unchanged Kuvan Net Product Revenue $180 to $200 $190 to $200 VIMIZIM $60 to $70 $65 to $70 Selected Income Statement Guidance ($ in millions, except percentages) Provided Updated Item August 26, 2014 October 23, 2014 Cost of Sales (% of Total Revenue) 16.5% to 17.5% Unchanged R&D Expense $460 to $480 $455 to $470 SG&A Expense $280 to $295 Unchanged Non-GAAP Net Loss $(60) to $(80) $(50) to $(65) GAAP Net Loss $(180) to $(195) $(160) to $(175) |
 33 Multiple Value-Drivers Across the Portfolio Approved Products to Drive $1B+ in Revenues in Next 2-3 Years Pivotal Data for PEG PAL, BMN 701 and BMN 190 Expected by 4Q15 Early Data Expected for BMN 111 and BMN 270 by 4Q15 Development Portfolio of 7 New Potential Products BioMarin is the Industry Leader in Orphan Drug Development 2014: On a Path Toward Profitability |
 34 PEG PAL (BMN 165) for PKU Results from Pivotal Phase 3 4Q 2015 Submission of BLA to the FDA 1Q 2016 Talazoparib (BMN 673) for mBC Enrollment completion of Phase 3 2H 2015 BMN 701 for Pompe Enrollment completion of Phase 2/3 trial 1H 2015 BMN 111 for Achondroplasia Data on first 3 cohorts in Phase 2 trial 2Q 2015 BMN 190 for Batten Disease Enrollment completion of Phase 1/2 trial 4Q 2014 Results from Phase 1/2 trial 2H 2015 BMN 270 for Hemophilia A IND/CTA filing 1Q 2015 BMN 250 for MPS IIIB IND/CTA filing Mid-2015 Multiple Portfolio Events in 2014 and 2015 |
 35 THANK YOU |