Exhibit 99.1

Company PresentationSeptember 2020 A subsidiary of

Forward Looking Statement This presentation contains "forward-looking statements" relating to future events, and we may from time to time make other statements, regarding our outlook or expectations for future financial or operating results and/or other matters regarding or affecting Evogene Ltd. or its subsidiaries, including Canonic Ltd. (“Subsidiary” or “Company”) (collectively, “Evogene” or “we”), that are considered “forward-looking statements” as defined in the U.S. Private Securities Litigation Reform Act of 1995 (the “PSLRA”) and other securities laws. Such forward-looking statements may be identified by the use of such words as “believe”, “expect”, “anticipate”, “should”, “planned”, “estimated”, “intend” and “potential” or words of similar meaning. Canonic is using forward-looking statements in this presentation when it discusses its near-term value drivers, including statements to the effect that it will reach commercialization, its estimated market size, its short-term milestones and commercial milestones. For these statements, Evogene and Subsidiary claim the protection of the safe harbor for forward-looking statements contained in the PSLRA and other securities laws.Such statements are based on current expectations, estimates, projections and assumptions, describe opinions about future events, involve certain risks and uncertainties which are difficult to predict and are not guarantees of future performance. Therefore, actual future results, performance or achievements, and trends in the future of Subsidiary and Evogene may differ materially from what is expressed or implied by such forward-looking statements due to a variety of factors, many of which are beyond Subsidiary's and Evogene's control, including, without limitation, those described in greater detail in Evogene's Annual Report on Form 20-F and in other information it files and furnishes with the Israel Securities Authority and the U.S. Securities and Exchange Commission, including those factors under the heading “Risk Factors”.All written and oral forward-looking statements attributable to us or persons acting on our behalf are expressly qualified in their entirety by the previous statements. Except for any obligations to disclose information as required by applicable securities laws, Subsidiary and Evogene disclaim any obligation or commitment to update any information contained in this presentation or to publicly release the results of any revisions to any statements that may be made to reflect future events or developments or changes in expectations, estimates, projections and assumptions.The information contained herein does not constitute a prospectus or other offering document, nor does it constitute or form part of any invitation or offer to sell, or any solicitation of any invitation or offer to purchase or subscribe for, any securities of Evogene or Subsidiary, nor shall the information or any part of it or the fact of its distribution form the basis of, or be relied on in connection with, any action, contract, commitment or relating thereto or to the securities of Evogene or Subsidiary.The trademarks included herein are the property of the owners thereof and are used for reference purposes only. Such use should not be construed as an endorsement of the products or services of Evogene or Subsidiary. 2

OUR VISION Commercialize effective,precise and stable medicalcannabis products foroptimized therapeutic impact 3

INTRODUCTION 4
Company Overview 5 Founded in 2019 as a spinoff from Evogene Ltd (EVGN) We aim to develop & commercialize medical cannabis products We aim to bring products all the way to the market starting from genetics to reach patients Our products under development are based on stable high yield plants with optimized therapeutic effects AI –based computational platform seeks to empower our plant breeding pipeline Commercialization is expected in early 2022, up to 4 products in the following 3 years
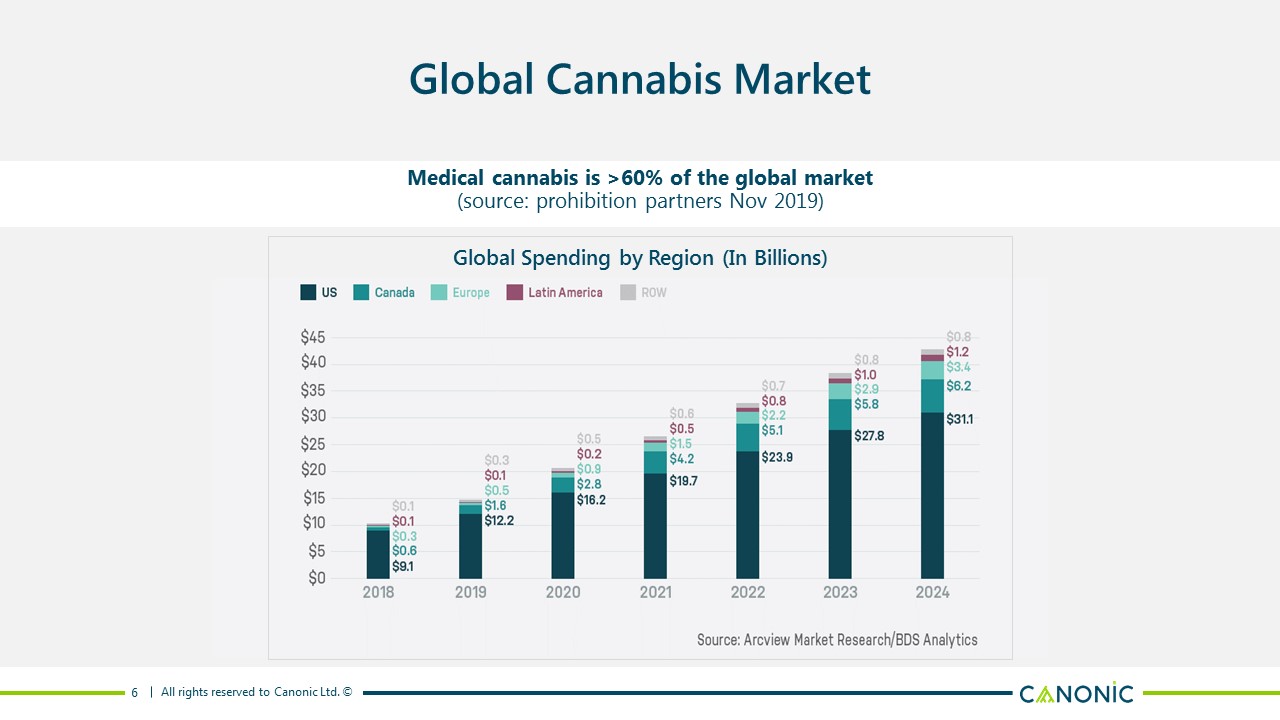
6 Global Cannabis Market Medical cannabis is >60% of the global market(source: prohibition partners Nov 2019) Global Spending by Region (In Billions)
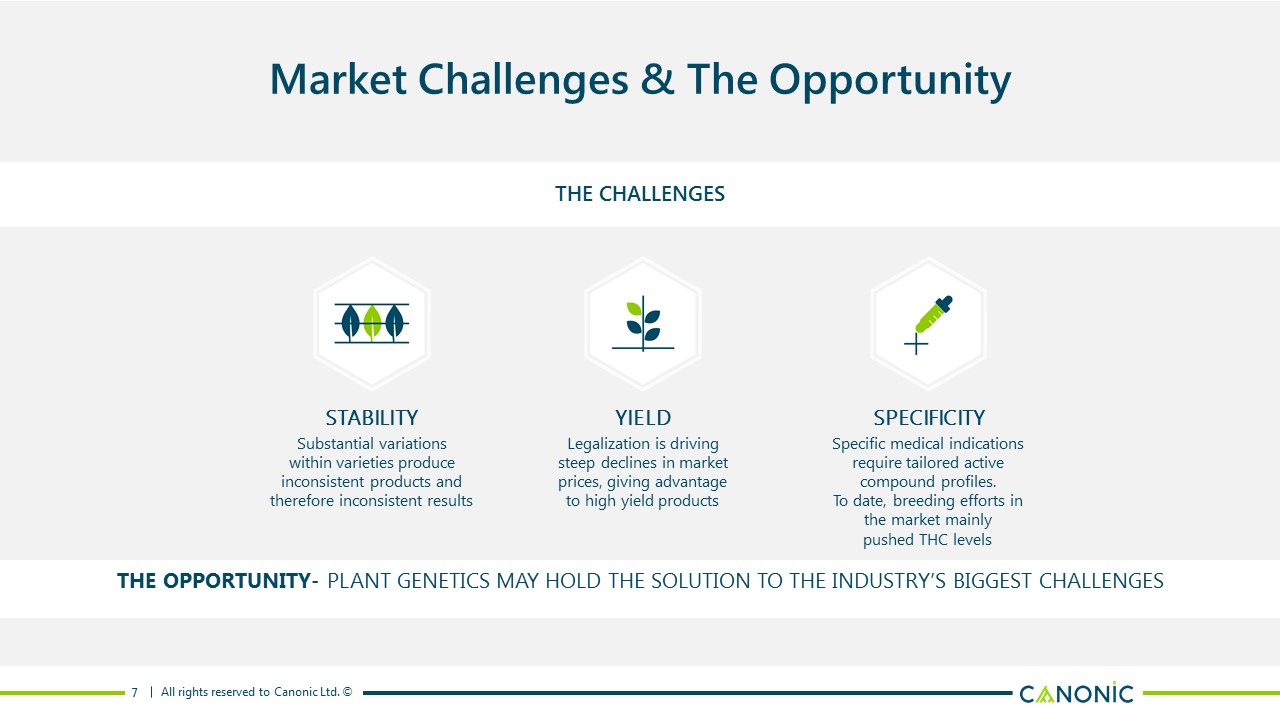
Market Challenges & The Opportunity THE CHALLENGES 7 SPECIFICITY Specific medical indicationsrequire tailored activecompound profiles.To date, breeding efforts in the market mainlypushed THC levels YIELD Legalization is driving steep declines in market prices, giving advantage to high yield products STABILITY Substantial variationswithin varieties produceinconsistent products andtherefore inconsistent results THE OPPORTUNITY- PLANT GENETICS MAY HOLD THE SOLUTION TO THE INDUSTRY’S BIGGEST CHALLENGES

Canonic TechnologyPowered By Evogene’s GeneRator AI CUTTING EDGE AI DRIVEN COMPUTATIONAL GENOMIC POWER TO TACKLE INDUSTRY CHALLENGES 8
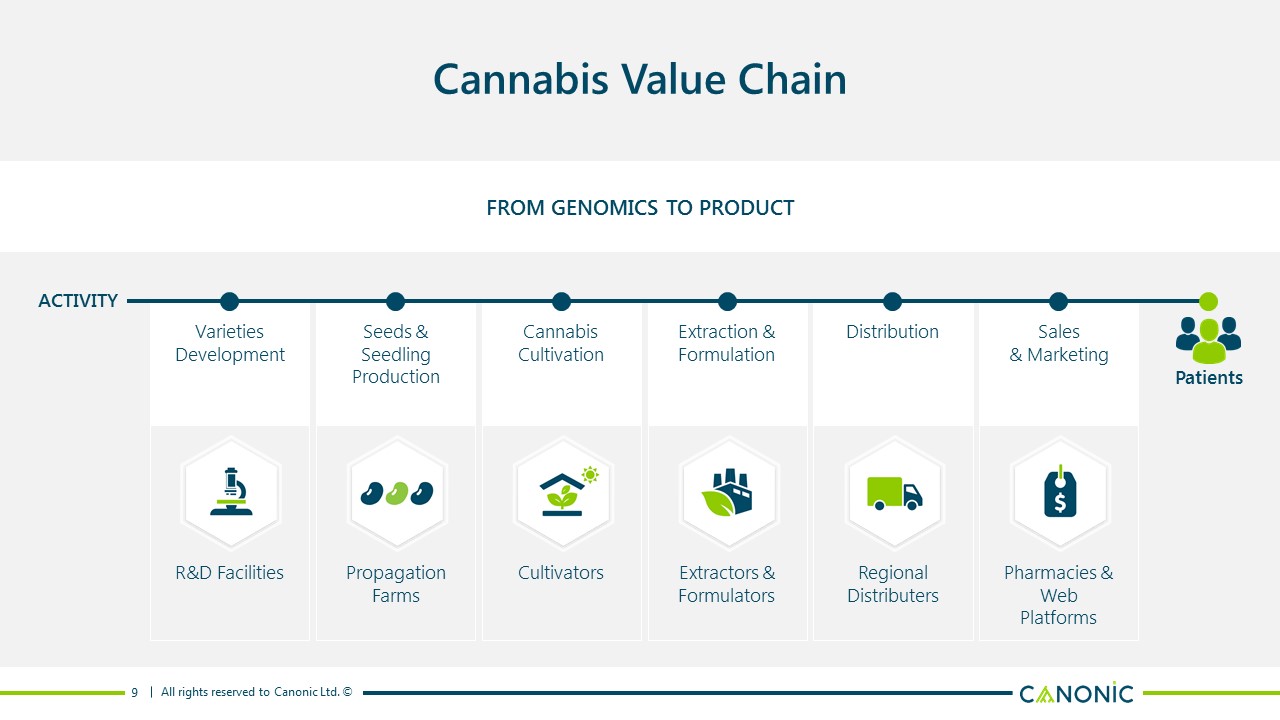
Cannabis Value Chain FROM GENOMICS TO PRODUCT 9 VarietiesDevelopment R&D Facilities Distribution RegionalDistributers Seeds & SeedlingProduction PropagationFarms Sales& Marketing Pharmacies & WebPlatforms Extraction &Formulation Extractors &Formulators CannabisCultivation Cultivators Patients ACTIVITY

Canonic in the Cannabis Value Chain Quickly and cost effectively reach the end user, by leveraging proprietarygenomic expertise and outsourcing downstream elements of the value chain 10 Patients VarietiesDevelopment R&D Facilities Distribution RegionalDistributers Seeds & SeedlingProduction PropagationFarms Sales& Marketing Pharmacies & WebPlatforms Extraction &Formulation Extractors &Formulators CannabisCultivation Cultivators CONTRACTORS ACTIVITY
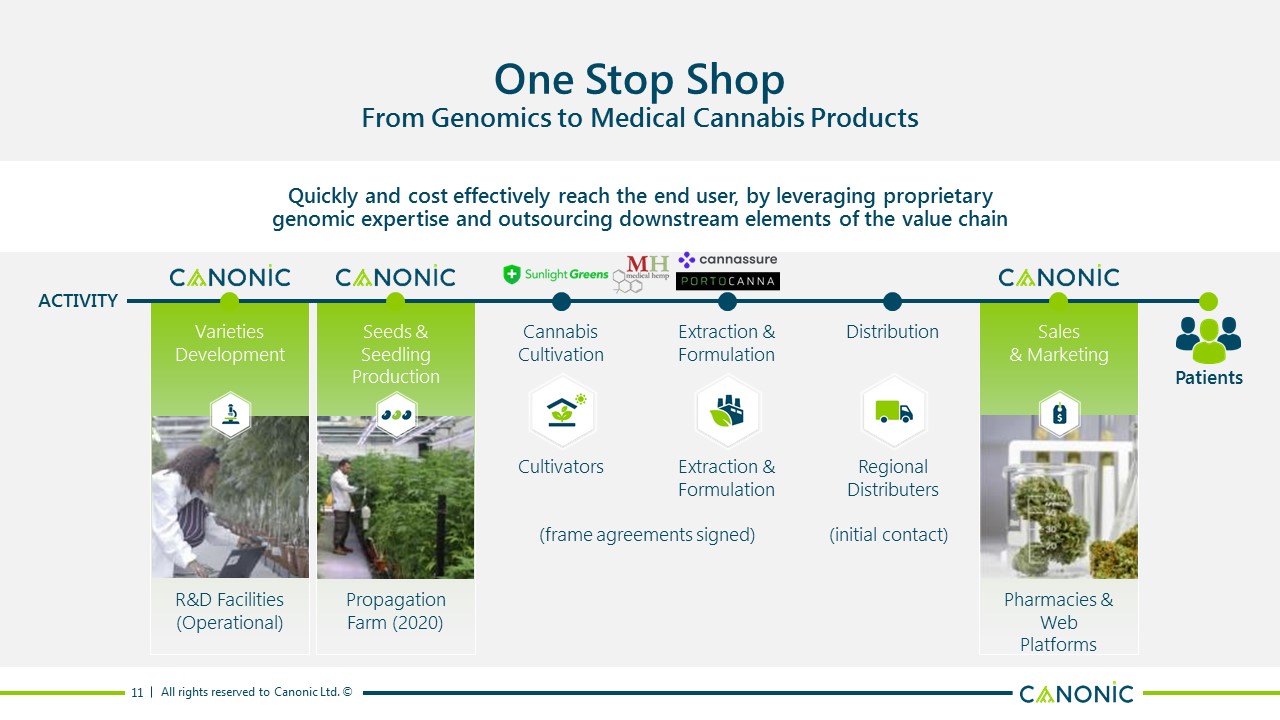
One Stop ShopFrom Genomics to Medical Cannabis Products Quickly and cost effectively reach the end user, by leveraging proprietarygenomic expertise and outsourcing downstream elements of the value chain 11 Patients VarietiesDevelopment R&D Facilities (Operational) Distribution RegionalDistributers Seeds & SeedlingProduction PropagationFarm (2020) Sales& Marketing Pharmacies & WebPlatforms Extraction &Formulation Extraction &Formulation CannabisCultivation Cultivators ACTIVITY (frame agreements signed) (initial contact)

PRODUCTS 12
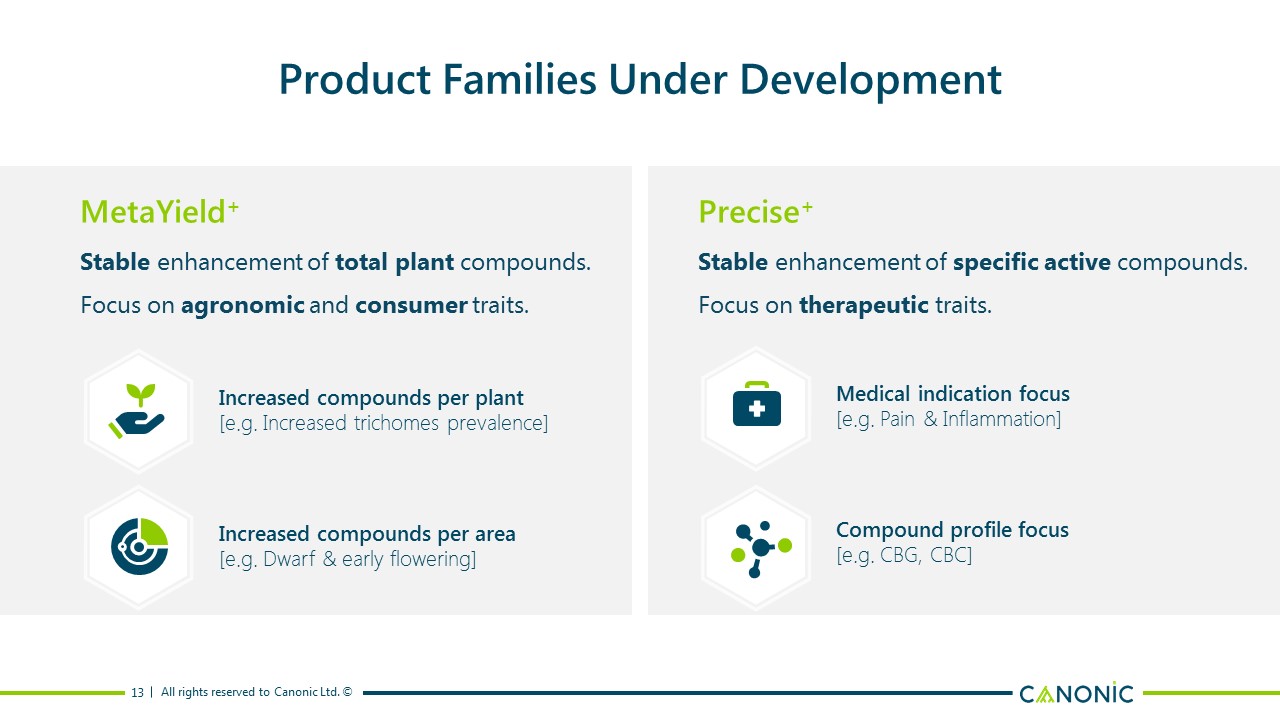
Product Families Under Development 13 Increased compounds per area [e.g. Dwarf & early flowering] Increased compounds per plant[e.g. Increased trichomes prevalence] MetaYield+ Stable enhancement of total plant compounds.Focus on agronomic and consumer traits. Compound profile focus [e.g. CBG, CBC] Medical indication focus [e.g. Pain & Inflammation] Precise+ Stable enhancement of specific active compounds.Focus on therapeutic traits.

MetaYield+ : First Commercial Product 14 Canonic expects to commercialize its first varieties during 2022 High THC PRODUCT NAME- NOA 1 Status:product development Expected launch in Israel:early 2022 Relevant market segment:T20/C5 High yield Big inflorescence Dense trichomes Target markets: Israel (170M$*) & EU *Company estimate, based on current market sale prices and data published by Israeli Health Ministry regarding number of medical cannabis users in Israel

MetaYield+ : Second Commercial Product 15 Canonic expects to commercialize its first varieties during 2022 High THC PRODUCT NAME- NOA 2 Status:product development Expected launch in Israel:early 2022 Relevant market segment:T20/C5 High yield Big inflorescence *Company estimate, based on current market sale prices and data published by Israeli Health Ministry regarding number of medical cannabis users in Israel Target markets: Israel (170M$*) & EU
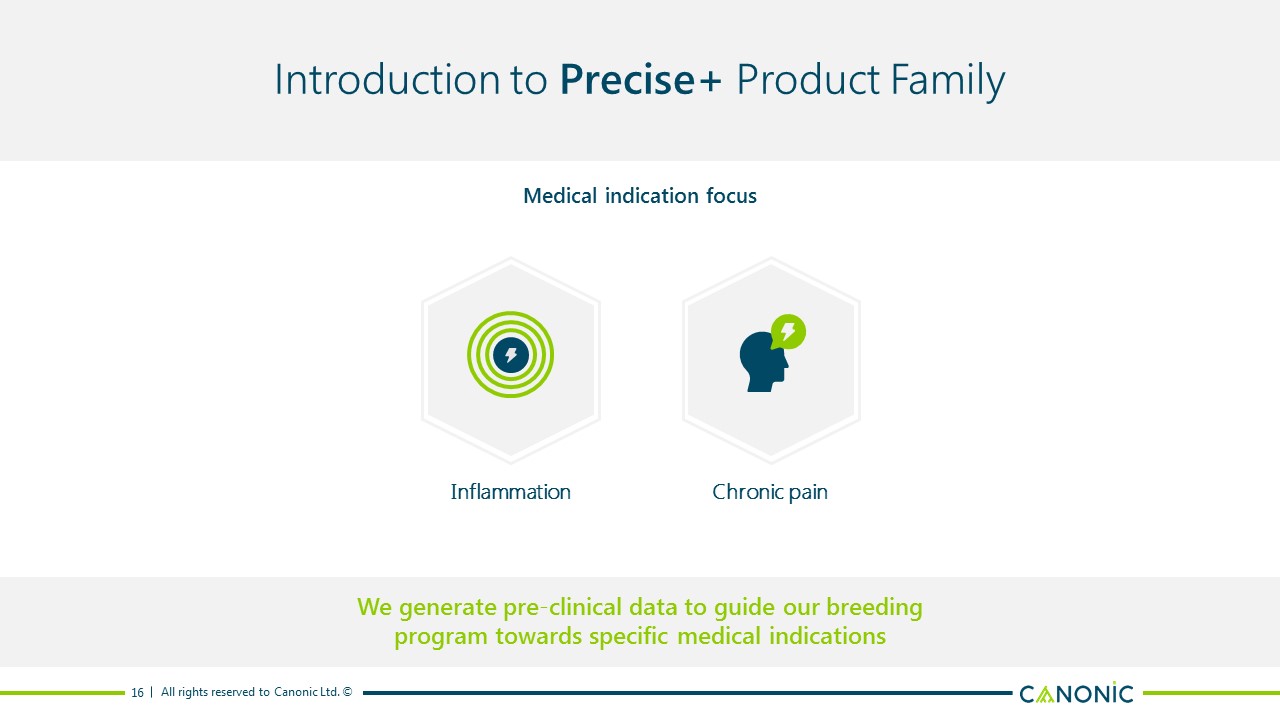
Introduction to Precise+ Product Family 16 Medical indication focus We generate pre-clinical data to guide our breeding program towards specific medical indications Chronic pain Inflammation

Next Generation Products 17 Canonic is developing and validating its varieties in collaboration with leading research centers Indication: Inflammation Product 1 Pre clinical study: Hadassah medical center Status: product development Expected launch in Israel: 2023 Indication: Pain Product 2 Pre clinical study: Migal research center Status: product development Expected launch in Israel: 2023 Target market size in Israel (2023):217 M$* Target market size in Israel (2023):70M$* *Company estimate, based on current market sale prices and data published by Israeli Health Ministry regarding number of medical cannabis users in Israel per medical indication

Technology 18

End-to-End Technologies 19 Developing in Israel’s top cannabis R&D facilities Large genetic & breeding collections In house propagation State of the art phenotypic tools Fully equipped labs & tissue culture rooms In-house Advanced-breeding World leading computational capabilities
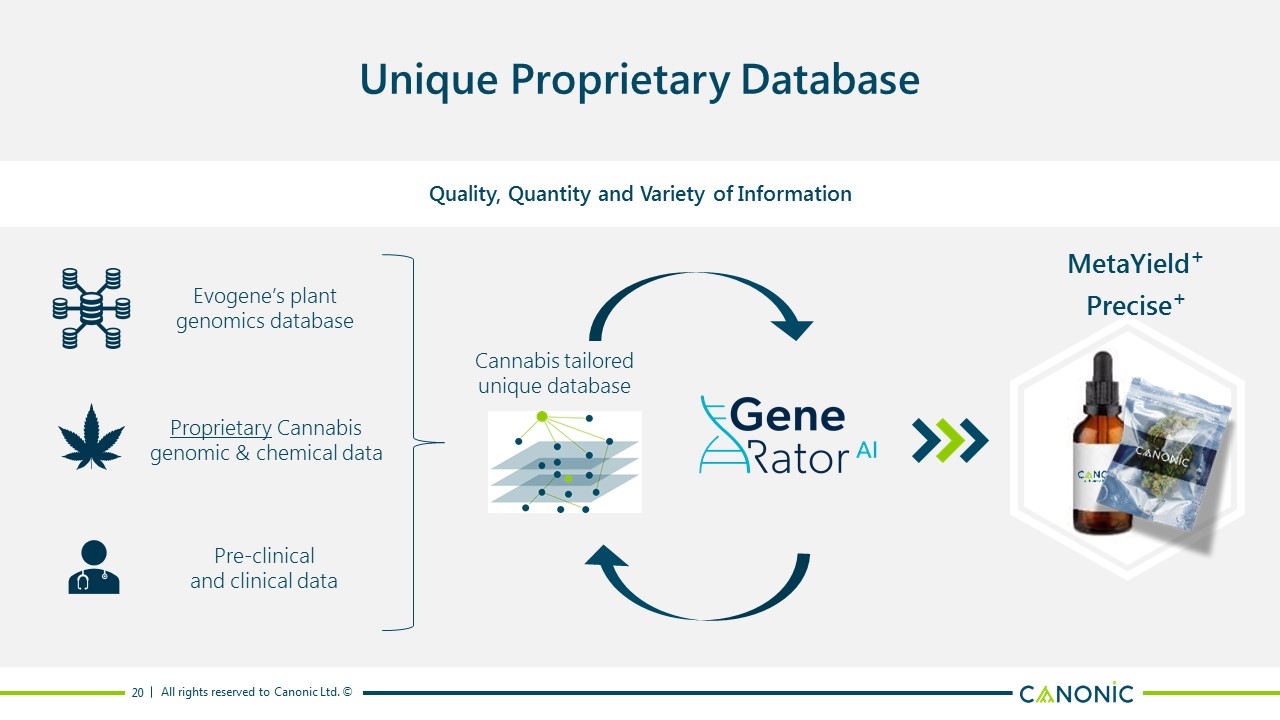
Unique Proprietary Database Quality, Quantity and Variety of Information Evogene’s plant genomics database Proprietary Cannabis genomic & chemical data Pre-clinical and clinical data Cannabis tailored unique database MetaYield+ 20 Precise+

- Advanced Breeding Technologies Re-engineering traditional Cannabis breeding- Computational Technology 21 Computational genomics Proprietary Comparative Genomics ATTTCCGCACAAGGATTAGT Novel Genes & Markers (IP protected) ATCGCTTCACATAGGAAGT Functional Cannabinoids patterns THC / CBD / CBG / …… High throughput plant breeding Pre-Clinical trials Computed phenotyping Genetic markers assisted selection MetaYield+ Precise+ Unique database

Cutting-Edge Technology Addressing Market Challenges 22 Stability Specificity Yield Reduced genetic variation Diverse extracts for pre-clinical assays Increased trichome density

Summary 23

Guy Adler Product manager & business development Team 24 Dr. Arnon HeymanCEO Dr. Inbal Dangoor Head of R&D Aviv FilhaberAgronomist Dr. Michi BrogPlant breeder Barel TalTechnician

Team 25 Ofer Haviv Chairman of the board Board of directors Evogene CEO and President Raanan CohenBusiness strategy Former CEO Koor industries Dr. Yossi TamCannabinoid clinical research Director, Multidisciplinary Center for Cannabinoid Research of the Hebrew university Dr. Yariv BrotmanPlant metabolic pathways Ben Gurion University & Max Planck institute of Molecular Plant Physiology Dorit KreinerFinance Evogene CFO Nirit ElyovichMarketing strategy Senior marketing specialist with over 20 years of B2C experience in various markets Shai LeviatovPlant breeding Commercial varieties breeding for more than 30 years in top leading multinational seeds companies. Adv. Itay Rosenferld Business strategy Advisors Cannabis entrepreneur and former BOD member of Intelicanna (TLV:INTL)

Short Term Milestones 26 2020 Conduct pre-clinical studies to support the development of Canonic’s medical cannabis products.Demonstrate yield improvement incannabis lines under development.Engagement with commercial partners for cultivation and production. 2021 Pre-commercial activity with firstcannabis variety towards commercialization in 2022.Demonstrate clinical effects of Canonic varieties based on dedicated research with a medical institution.
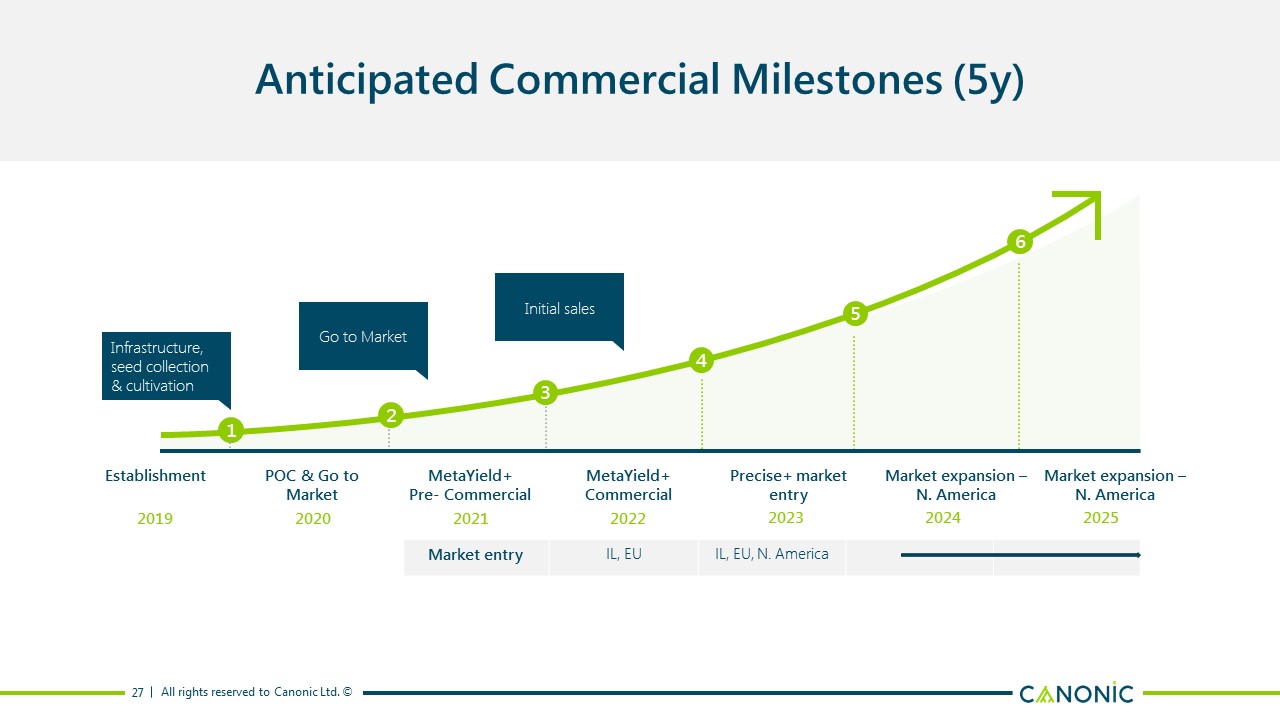
Anticipated Commercial Milestones (5y) 27 Establishment POC & Go to Market MetaYield+ Pre- Commercial MetaYield+ Commercial Precise+ market entry Market expansion – N. America 2019 2020 2021 2022 Market expansion – N. America 1 2 3 4 5 6 Infrastructure, seed collection & cultivation Go to Market Initial sales 2023 2024 2025 Market entry IL, EU IL, EU, N. America

Summary 28 Canonic aims to develop and commercialize medical grade Cannabis through a revolutionary genomic approach Exclusive access to Evogene’s CPB technology platform Commercial partners in place for cultivation and production (IL & EU) Initial commercialization expected in 2022 Infrastructure in place – from seed to sale Leading academic partnerships for unique variety development

THANK YOU