 Creating a New Model in Specialty Pharmaceutical Leadership Exhibit 99.2 |
 Important Information For Investors and Shareholders 2 This communication does not constitute an offer to sell or the solicitation of an offer to buy any securities or a solicitation of any vote or approval, nor shall there be any sale of securities in any jurisdiction in which such offer, solicitation or sale would be unlawful prior to registration or qualification under the securities laws of any such jurisdiction. In connection with the proposed merger between Actavis and Forest, Actavis will file with the Securities and Exchange Commission (the “SEC”) a registration statement on Form S-4 that will include a joint proxy statement of Actavis and Forest that also constitutes a prospectus of Actavis. The definitive joint proxy statement/prospectus will be delivered to shareholders of Actavis and Forest. INVESTORS AND SECURITY HOLDERS OF ACTAVIS AND FOREST ARE URGED TO READ THE DEFINITIVE JOINT PROXY STATEMENT/PROSPECTUS AND OTHER DOCUMENTS THAT WILL BE FILED WITH THE SEC CAREFULLY AND IN THEIR ENTIRETY WHEN THEY BECOME AVAILABLE BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION. Investors and security holders will be able to obtain free copies of the registration statement and the definitive joint proxy statement/prospectus (when available) and other documents filed with the SEC by Actavis and Forest through the website maintained by the SEC at http://www.sec.gov. Copies of the documents filed with the SEC by Actavis will be available free of charge on Actavis’ internet website at www.actavis.com or by contacting Actavis’ Investor Relations Department at (862) 261- 7488. Copies of the documents filed with the SEC by Forest will be available free of charge on Forest’s internet website at www.frx.com or by contacting Forest’s Investor Relations Department at (212) 224-6713. Participants in the Merger Solicitation Actavis, Forest, their respective directors and certain of their executive officers and employees may be considered participants in the solicitation of proxies in connection with the proposed transaction. Information regarding the persons who may, under the rules of the SEC, be deemed participants in the solicitation of the Actavis and Forest shareholders in connection with the proposed merger will be set forth in the joint proxy statement/prospectus when it is filed with the SEC. Information about the directors and executive officers of Forest is set forth in its proxy statement for its 2013 annual meeting of stockholders, which was filed with the SEC on July 8, 2013 and certain of its Current Reports on Form 8-K. Information about the directors and executive officers of Actavis is set forth in its proxy statement for its 2013 annual meeting of stockholders, which was filed with the SEC on March 29, 2013 and certain of its Current Reports on Form 8-K. Additional information regarding the participants in the proxy solicitations and a description of their direct and indirect interests, by security holdings or otherwise, will be contained in the joint proxy statement/prospectus filed with the above-referenced registration statement on Form S-4 and other relevant materials to be filed with the SEC when they become available. |
 Actavis Cautionary Statement Regarding Forward-Looking Statements 3 Statements contained in this communication that refer to Actavis’ estimated or anticipated future results, including estimated synergies, or other non-historical facts are forward-looking statements that reflect Actavis’ current perspective of existing trends and information as of the date of this communication. Forward looking statements generally will be accompanied by words such as “anticipate,” “believe,” “plan,” “could,” “should,” “estimate,” “expect,” “forecast,” “outlook,” “guidance,” “intend,” “may,” “might,” “will,” “possible,” “potential,” “predict,” “project,” or other similar words, phrases or expressions. Such forward-looking statements include, but are not limited to, statements about the benefits of the Forest acquisition, including future financial and operating results, Actavis’ or Forest’s plans, objectives, expectations and intentions and the expected timing of completion of the transaction. It is important to note that Actavis’ goals and expectations are not predictions of actual performance. Actual results may differ materially from Actavis’ current expectations depending upon a number of factors affecting Actavis’ business, Forest’s business and risks associated with acquisition transactions. These factors include, among others, the inherent uncertainty associated with financial projections; restructuring in connection with, and successful closing of, the Forest acquisition; subsequent integration of the Forest acquisition and the ability to recognize the anticipated synergies and benefits of the Forest acquisition; the ability to obtain required regulatory approvals for the transaction (including the approval of antitrust authorities necessary to complete the acquisition), the timing of obtaining such approvals and the risk that such approvals may result in the imposition of conditions that could adversely affect the combined company or the expected benefits of the transaction; the ability to obtain the requisite Forest and Actavis shareholder approvals; the risk that a condition to closing of the Forest acquisition may not be satisfied on a timely basis or at all; the failure of the proposed transaction to close for any other reason; risks relating to the value of the Actavis shares to be issued in the transaction; the anticipated size of the markets and continued demand for Actavis’ and Forest’s products; the impact of competitive products and pricing; access to available financing (including financing for the acquisition or refinancing of Actavis or Forest debt) on a timely basis and on reasonable terms; the risks of fluctuations in foreign currency exchange rates; the risks and uncertainties normally incident to the pharmaceutical industry, including product liability claims and the availability of product liability insurance on reasonable terms; the difficulty of predicting the timing or outcome of pending or future litigation or government investigations; periodic dependence on a small number of products for a material source of net revenue or income; variability of trade buying patterns; changes in generally accepted accounting principles; risks that the carrying values of assets may be negatively impacted by future events and circumstances; the timing and success of product launches; the difficulty of predicting the timing or outcome of product development efforts and regulatory agency approvals or actions, if any; market acceptance of and continued demand for Actavis’ and Forest’s products; costs and efforts to defend or enforce intellectual property rights; difficulties or delays in manufacturing; the availability and pricing of third party sourced products and materials; successful compliance with governmental regulations applicable to Actavis’ and Forest’s facilities, products and/or businesses; changes in the laws and regulations affecting, among other things, pricing and reimbursement of pharmaceutical products; changes in tax laws or interpretations that could increase Actavis’ consolidated tax liabilities; the loss of key senior management or scientific staff; and such other risks and uncertainties detailed in Actavis’ periodic public filings with the Securities and Exchange Commission, including but not limited to Actavis’ Annual Report on form 10-K for the year ended December 31, 2012 and from time to time in Actavis’ other investor communications. Except as expressly required by law, Actavis disclaims any intent or obligation to update or revise these forward-looking statements. |
 Forest Cautionary Statement Regarding Forward-Looking Statements 4 This release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Such forward-looking statements include, but are not limited to, statements about the benefits of the acquisition of Forest by Actavis, including future financial and operating results, Forest’s or Actavis’ plans, objectives, expectations and intentions and the expected timing of completion of the transaction. It is important to note that Forest’s goals and expectations are not predictions of actual performance. Actual results may differ materially from Forest’s current expectations depending upon a number of factors affecting Forest’s business, Actavis’ business and risks associated with acquisition transactions. These factors include, among others, the inherent uncertainty associated with financial projections; restructuring in connection with, and successful closing of, the acquisition; subsequent integration of the companies and the ability to recognize the anticipated synergies and benefits of the acquisition; the ability to obtain required regulatory approvals for the transaction (including the approval of antitrust authorities necessary to complete the acquisition), the timing of obtaining such approvals and the risk that such approvals may result in the imposition of conditions that could adversely affect the combined company or the expected benefits of the transaction; the ability to obtain the requisite Forest and Actavis shareholder approvals; the risk that a condition to closing of the acquisition may not be satisfied on a timely basis or at all; the failure of the proposed transaction to close for any other reason; risks relating to the value of the Actavis shares to be issued in the transaction; access to available financing (including financing for the acquisition or refinancing of Forest or Actavis debt) on a timely basis and on reasonable terms; the difficulty of predicting FDA approvals, the acceptance and demand for new pharmaceutical products, the impact of competitive products and pricing, the timely development and launch of new products, and the risk factors listed from time to time in Forest Laboratories’ Annual Report on Form 10-K, Quarterly Reports on Form 10-Q and any subsequent SEC filings. Forest assumes no obligation to update forward-looking statements contained in this release to reflect new information or future events or developments. |
 Presentation Overview • Transaction Overview • An Innovative New Model in Specialty Pharmaceutical Leadership • Forest Overview • Summary 5 |
 Proposed Transaction Terms • Equity and Cash transaction valued at approximately $25 B - 25% Premium to Forest closing price as of 2/14/2014 - 70/30 equity and cash split - $26.04 in cash + 0.3306 share of ACT/share of FRX • Pro Forma Forest ownership of ~35% shares of Actavis • Cash portion funded through existing cash and new debt • Anticipated to close by mid-year 2014 subject to: - Subject to approval of both Actavis and Forest shareholders - Customary conditions including legal and regulatory approval (HSR) 6 |
 Creating an Innovative New Model in Specialty Pharmaceutical Leadership Enhanced size and scale Broad & diverse portfolio with multiple blockbuster therapeutic franchises Balanced portfolio of branded and generic pharmaceuticals Exceptionally strong global commercial capabilities create high value to customers More than $1 billion investment in R&D driving strong organic growth Strong free cash flow generation Efficient tax structure and solid balance sheet Drive robust organic growth accelerated by smart business development 7 |
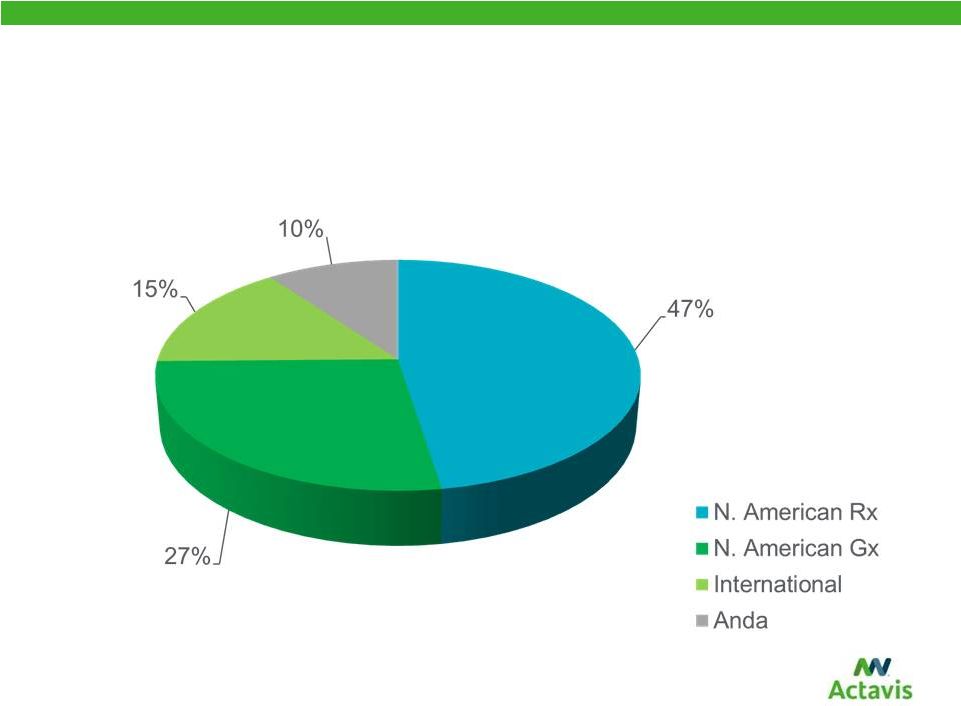 Balanced Revenue Portfolio of North American Brand, North American Generic, International and Distribution Based on 2014 proforma combined revenue 8 |
 Financially Compelling Combination • Approximately $15 billion annual revenue generation — Approximately 50/50 brand/generic • Expected double-digit accretion to non-GAAP earnings, including synergies, in 2015 and 2016 • Greater than $4 billion annual free cash flow in 2015 — Expect to maintain investment grade credit ratings • Estimated ~$1 billion in annual pre-tax operational and tax synergies within three years of transaction close 9 • Strong cash flow drives rapid deleveraging to under 3.5x debt to pro forma adjusted EBITDA by the end of 2014 |
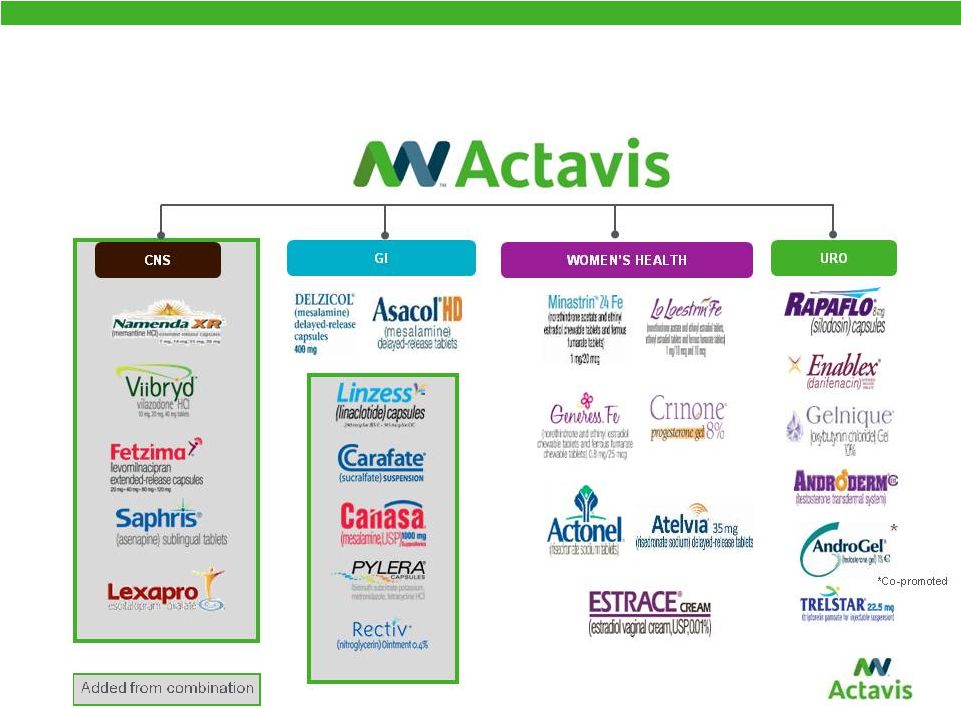 Broad & Diverse Portfolio with Multiple Blockbuster Therapeutic Franchises 10 |
 Emerging and Sustainable Portfolios in New Categories CARDIOVASCULAR DERM INFECTIOUS DISEASE RESPIRATORY Added from combination 11 CYSTIC FIBROSIS |
 Exceptionally Strong Commercial Capabilities Create High Value to Customers • Enhanced profile: size, scale and product diversification bring high value to customers – physicians, hospitals, health plans and distribution channels 12 • World-class global commercial organization competing across multiple market segments – Brands, generics, biosimilars, and OTCs • Better positioned with Forest’s strong primary care sales force to drive increased sales of Actavis Specialty Brands • Strong global operations providing high-quality reliable supply |
 Continued Strong Investment in R&D with over $1 Billion Expected in Year One • Continued investment in strong pipeline assets across all therapeutic categories • Continued strong investment in generic R&D to maintain strong global pipeline - Continue to invest in differentiated products including respiratory, injectables and ophthalmics for all markets - Continued focus on important FTF assets in the US - Continued commitment to the development of biosimilars • Forest will add more than a half dozen near- and mid-term R&D products to Actavis’ robust development portfolio. - Five products at the NDA stage of development, including treatments for Alzheimer’s disease, cardiovascular disease, infectious disease, as well as Schizophrenia and bipolar disorders and, and treatments for COPD 13 |
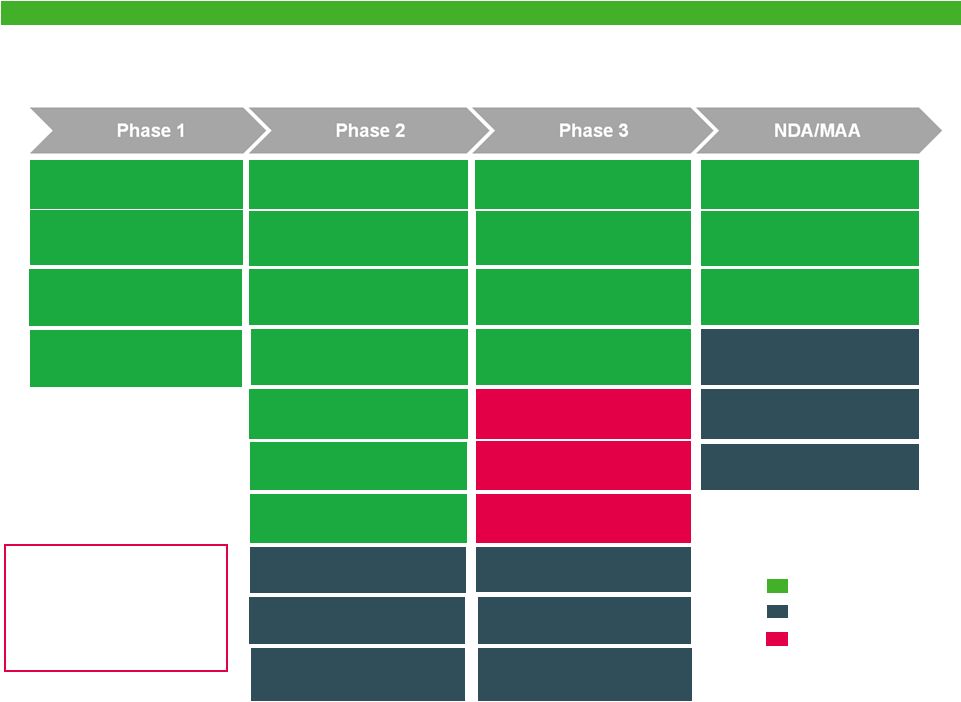 Combined Specialty Brands Pipeline Actavis Biologic Forest Colvir Albaconazole VVC Esmya®-Fibroids (US) Diafert™ WC3011 E2 Vaginal Cream Metronidazole 1.3% Vaginal Gel E4/Progestin OC Levosert™ Contraception Progestin Only Patch Amg/Act Herceptin® WC3043 Udenafil ED WC3055 Udenafil BPH Rapaflo® NextGen WC3035 Sarecycline WC2055 Doxycycline NextGen Oxybutynin Hyperhidrosis Albaconazole Onychomycosis WC3079 Delzicol® NextGen WC3046 Delzicol® 800mg Cariprazine (Bipolar Depression) Cebranopadol (Pain Management) TRV027 (Acute Heart Failure) TUDORZA®-formoterol (COPD) Ceftazidime-avibactam (Infectious Disease) BYSTOLIC®-valsartan (Hypertension) NAMENDA XR®-donepezil (Alzheimer's Disease) Cariprazine (CRL) (Schizophrenia, BP Mania) Amg/Act Avastin® rFSH VIIBRYD® (GAD) NOTE: Additional important products in preclinical development including biosimilars to Rituxin ® and Erbitux ® through Actavis collaboration with Amgen 14 |
 Overview 15 |
 BUSINESS development DRUG Marketed Drugs NDA EXECUTION AN EXCEPTIONAL COMPANY ENGINE commercial Specialty pharmaceutical company focused on large primary care and subspecialty markets Key strategy building blockbuster therapeutic line calls to create economies and relevance 16 Forest – Overview and Strategy development |
 Recent Strategic Initiatives Have Rejuvenated Forest Commenced Project Rejuvenate to achieve $500 M in cost reductions Acquired Saphris for $240 M Leveraged balance sheet with $1.2 B in first ever bond offering in December and $1.8 B offering in January for Aptalis acquisition Achieved NAMENDA XR ® Coverage at 9 of Top 10 Part D Plans Launched FETZIMA™ for Depression Filed NAMENDA ® Pediatric Written Request (PWR) Completed $2.9 B acquisition of Aptalis Notified FDA of intent to discontinue NAMENDA ® , Focus on NAMENDA 17 XR ® |
 Forest Has Strong Drug Development Capabilities and is Focused on Commercial Execution • Next 9 strategy executed well • 7 regulatory approvals since 2009 • History of first cycle approvals • New products contributed 44% of sales and grew 59% year- over-year in most recent quarter 18 |
 Recent Acquisition of Aptalis • Aptalis is an excellent strategic fit Diversifies Forest offerings in key therapeutic areas GI franchise in the US complements Linzess business CF franchise in Europe complements Colobreathe business Significantly improves Forest profitability in Canada • Aptalis products are growing and expected to contribute ~$700 M to Forest sales • Forest expects to realize significant synergies – $125 million in pre-tax synergies • Deal closed January 31, 2014 19 • Deal is immediately & highly accretive to Forest non-GAAP EPS |
 Summary of a Transformational Combination 20 |
 ~$1 Billion in Annual Synergies • ~$1 Billion Pre-Tax Operational and Tax Synergies • Majority of the synergies are expected to take place in first 12 months following close – Total synergies realized in first 3 years after acquisition close • Excludes any manufacturing and revenue synergies • Tax synergies approximately 10% of total • Pro forma combined tax rate is expected to be slightly below 16% for 2015 21 |
 Next Steps • Successful Completion of Transaction Requires: – Approval by shareholders of both companies – Regulatory review and approval including Hart-Scott- Rodino review 22 • Anticipated close mid-year 2014 • Management teams from both companies to immediately begin pre-integration activities to maximize potential at close |
 23 |