Theravance Biopharma, Inc. (NASDAQ: TBPH) Second Quarter 2019 Financial Results & Business Update July 31, 2019 THERAVANCE®, the Cross/Star logo, and MEDICINES THAT MAKE A DIFFERENCE® are registered trademarks of the Theravance Biopharma group of companies. All third party trademarks used herein are the property of their respective owners. © 2019 Theravance Biopharma. All rights reserved.

Forward Looking Statements Under the safe harbor provisions of the U.S. Private Securities Litigation Reform Act of 1995, the company cautions investors that any forward-looking statements or projections made by the company are subject to risks and uncertainties that may cause actual results to differ materially from the forward-looking statements or projections. Examples of forward-looking statements in this presentation may include the current dispute with Innoviva, Inc. and TRC LLC, statements relating to the company’s strategies, plans and objectives, the company’s regulatory strategies and timing of clinical studies (including the data therefrom), the potential characteristics, benefits and mechanisms of action of the company’s product and product candidates, the potential that the company’s research programs will progress product candidates into the clinic, the company’s expectations for product candidates through development, potential regulatory approval and commercialization (including their differentiation from other products or potential products), product sales or profit share revenue and the company’s expectations for its 2019 operating loss, excluding share-based compensation. The company’s forward-looking statements are based on the estimates and assumptions of management as of the date of this presentation and are subject to risks and uncertainties that may cause the actual results to be materially different than those projected, such as risks related to the nature of the current dispute with Innoviva and TRC LLC, the uncertainty of arbitration and litigation and the possibility that an arbitration award or litigation result involving the current dispute could be adverse to the company, delays or difficulties in commencing or completing clinical studies, the potential that results from clinical or non-clinical studies indicate product candidates are unsafe or ineffective (including when our product candidates are studied in combination with other compounds), delays or failure to achieve and maintain regulatory approvals for product candidates, risks of collaborating with third parties to discover, develop and commercialize products, risks associated with establishing and maintaining sales, marketing and distribution capabilities. Other risks affecting the company are described under the heading “Risk Factors” and elsewhere in the company’s Form 10-Q filed with the Securities and Exchange Commission (SEC) on May 10, 2019, and other periodic reports filed with the SEC. 2

Significant existing cash resources to fund strategic priorities2 Focus on Strategic Priorities COMMITMENT TO CREATING TRANSFORMATIONAL MEDICINES 3 TRELEGY ELLIPTA is FF/UMEC/VI or fluticasone furoate/umeclidinium/vilanterol; comprised of ICS, LAMA, and LABA, active components of Anoro (UMEC/VI). 1 TBPH holds 85% economic interest in upward-tiering royalty stream of 6.5% – 10% payable by GSK (net of TRC LLC expenses paid and the amount of cash, if any, expected to be used by TRC pursuant to the TRC LLC Agreement over the next four fiscal quarters). All statements based on publicly available information. 2 Cash, cash equivalents, and marketable securities of approximately $396M as of June 30, 2019 Opportunities to Create Transformational Medicines YUPELRI® Nebulized LAMA in COPD U.S. commercial launch underway TD-1473 Gut-selective oral JAK inhibitor for inflammatory intestinal diseases Phase 2b/3 RHEA study in ulcerative colitis ongoing; Phase 2b data planned late-2020 Phase 2 DIONE study in Crohn’s disease ongoing; data planned late-2020 Ampreloxetine NRI in symptomatic neurogenic orthostatic hypotension (nOH) Registrational Phase 3 program progressing; 4-week efficacy data expected 2H 2020 TD-8236 Lung-selective inhaled pan-JAK inhibitor for serious respiratory diseases Safety and biomarker data from Phase 1 study in healthy volunteers and asthmatics expected September 2019 Research Organ-selective research platform designed to expand therapeutic index compared to conventional systemic therapies Economic Interest TRELEGY ELLIPTA1 Single inhaler triple therapy in COPD Product launched in 36 countries, including Japan; China approval expected 4Q19 sNDA submitted to FDA supporting revised labelling on reduction in risk of all-cause mortality compared with ANORO ELLIPTA in patients with COPD Potential sNDA for asthma indication in 2H 2019

TD-1473: Gut-selective oral JAK inhibitor LATE STAGE STUDIES IN ULCERATIVE COLITIS AND CROHN’S DISEASE 1 Maintenance phase of the study will have induction responder subjects re-randomized to active doses compared to placebo at 44 weeks Crohn’s disease Ulcerative colitis Phase 2 (N=160) Dose-finding induction 12 weeks Active treatment extension 48 weeks Responders Phase 2b/3 (N=240) Dose-finding induction 8 weeks Maintenance phase1 44 weeks Phase 3 (N=640) Dose-confirming induction 8 weeks Phase 2b/3 study in UC and Phase 2 study in CD progressing; data planned late-2020 4
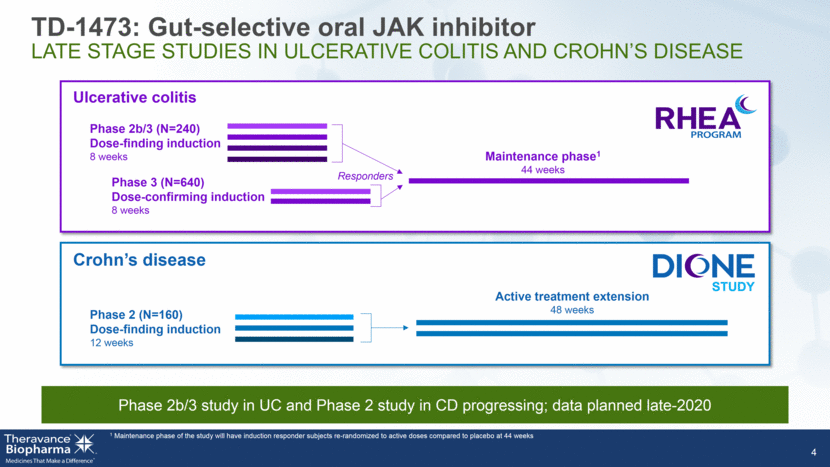
5 TD-1473 Research Vision ORGAN-SELECTIVE APPROACH DESIGNED TO EXPAND THERAPEUTIC INDEX Treat disease at site to maximize efficacy Optimize pharmacology to include potent inhibition of Tyk2 Improve upon the efficacy of a clinically validated target WARNING: SERIOUS INFECTIONS AND MALIGNANCY See full prescribing information for complete boxed warning. Tofacitinib Dose Proportional Efficacy in UC Ph 21 Patients with Clinical Response, % Tofacitinib (mg) Inhibitory potency (pKi) IL-12 and IL-23 inhibition 1 Sandborn WJ, et al. N EnglJ Med 2012;367:616-24 UC = Ulcerative colitis CD = Crohn’s disease Encouraging Phase 1b data and preclinical package (including daily dose administration for 6 & 9 months)
Ampreloxetine: Phase 2 Study in nOH DESIGNED TO EVALUATE INITIAL AND DURABLE RESPONSE TO THERAPY 6 Purpose: To evaluate the effect of ampreloxetine in improving blood pressure and key nOH symptoms Part C: Responders in Part A eligible for open-label treatment for up to 5 months Designed to assess durability of effect Primary assessment at four weeks (Day 29) Efficacy evaluations: OHSA1 #1, standing time duration, standing systolic blood pressure Also assessed safety and pharmacokinetics of ampreloxetine 1 OHSA: Orthostatic Hypotension Symptom Assessment. OHSA #1 measures dizziness (cardinal symptom of nOH), lightheadedness, feeling faint, or feeling of impending black out. Three-part design in patients with nOH: Single ascending dose portion of ampreloxetine (up to 20 mg) Testing blood pressure response to ampreloxetine A Patients started on Part A, and responders moved to Part B and/or Part C (extension phase) Double-blind Placebo-controlled Single dose (Part A response dose) or placebo B Extension phase Open label design Up to 24 weeks (20 weeks dosing, 4 week wash out) Primary endpoint at 4 weeks C

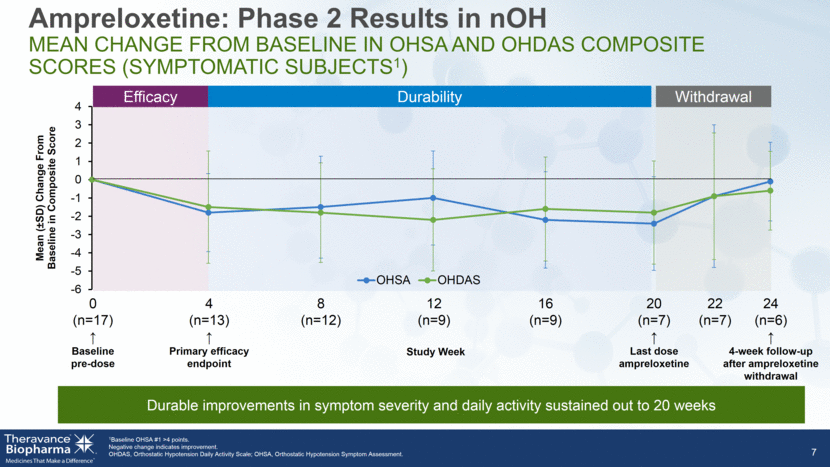
7 Ampreloxetine: Phase 2 Results in nOH Mean Change From Baseline in OHSA and OHDAS Composite Scores (Symptomatic Subjects1) 1Baseline OHSA #1 >4 points. Negative change indicates improvement. OHDAS, Orthostatic Hypotension Daily Activity Scale; OHSA, Orthostatic Hypotension Symptom Assessment. Efficacy Durability Withdrawal 0 (n=17) Baseline pre-dose 4 (n=13) Primary efficacy endpoint 8 (n=12) 12 (n=9) 16 (n=9) 20 (n=7) Last dose ampreloxetine 22 (n=7) Study Week 24 (n=6) 4-week follow-up after ampreloxetine withdrawal Durable improvements in symptom severity and daily activity sustained out to 20 weeks -6 -5 -4 -3 -2 -1 0 1 2 3 4 Mean ( ± SD) Change From Baseline in Composite Score OHSA OHDAS
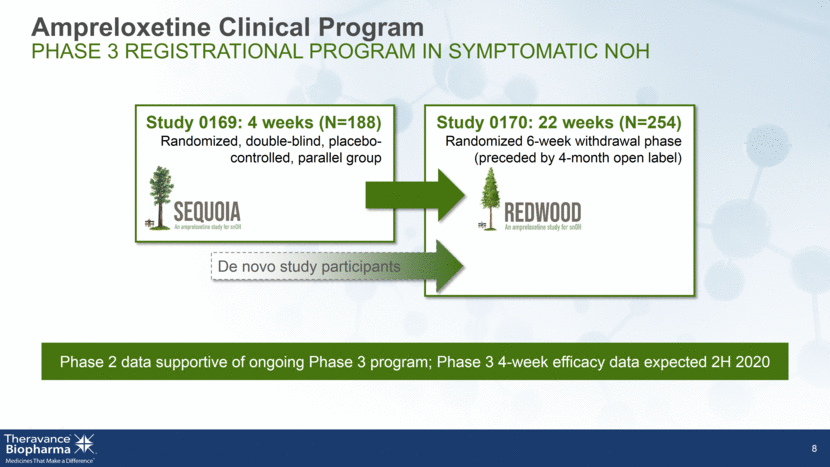
Ampreloxetine Clinical Program PHASE 3 REGISTRATIONAL PROGRAM IN SYMPTOMATIC NOH 8 Phase 2 data supportive of ongoing Phase 3 program; Phase 3 4-week efficacy data expected 2H 2020 Study 0169: 4 weeks (N=188) Randomized, double-blind, placebo-controlled, parallel group Study 0170: 22 weeks (N=254) Randomized 6-week withdrawal phase (preceded by 4-month open label) De novo study participants
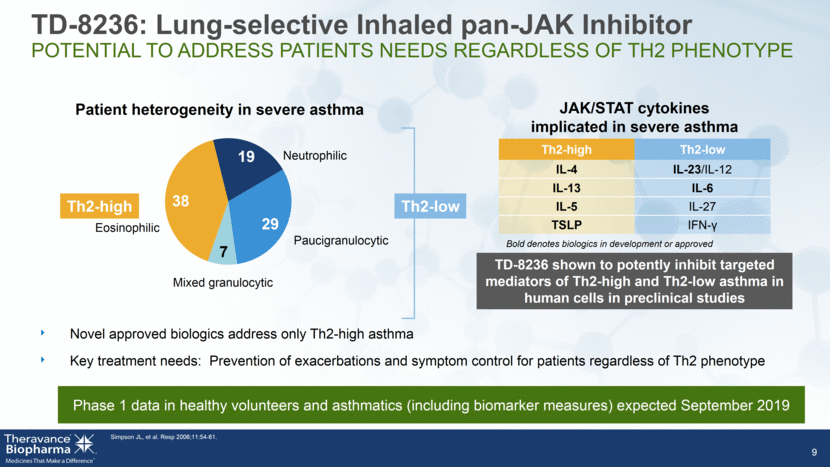
JAK/STAT cytokines implicated in severe asthma Th2-high Th2-low IL-4 IL-23/IL-12 IL-13 IL-6 IL-5 IL-27 TSLP IFN-9 Mixed granulocytic Eosinophilic Neutrophilic Paucigranulocytic Patient heterogeneity in severe asthma Th2-low Th2-high Bold denotes biologics in development or approved Novel approved biologics address only Th2-high asthma Key treatment needs: Prevention of exacerbations and symptom control for patients regardless of Th2 phenotype Simpson JL, et al. Resp 2006;11:54-61. TD-8236 shown to potently inhibit targeted mediators of Th2-high and Th2-low asthma in human cells in preclinical studies TD-8236: Lung-selective Inhaled pan-JAK Inhibitor POTENTIAL TO ADDRESS PATIENTS NEEDS REGARDLESS OF TH2 PHENOTYPE Phase 1 data in healthy volunteers and asthmatics (including biomarker measures) expected September 2019 38 19 29 7
YUPELRI®: Formal Commercial Launch Underway FDA-APPROVED FOR THE MAINTENANCE TREATMENT OF COPD 10 YUPELRI® (revefenacin) inhalation solution. Approved for the maintenance treatment of patients with COPD. COPD = Chronic Obstructive Pulmonary Disease. 1 Global Strategy for Diagnosis, Management, and Prevention of COPD. 2 Suboptimal Inspiratory Flow Rates Are Associated with COPD and All Cause Readmissions. Loh et al., Annals of ATS 2017. First and only once-daily bronchodilator delivered in a nebulizer Higher of two doses approved: 175 mcg once daily, for use with any standard jet nebulizer Unmet need for nebulized LAMA therapy Once-daily LAMAs are first-line therapy for moderate to severe COPD 1 No once-daily nebulized LAMAs available previously; only available in handheld devices Nebulized therapy associated with reduced hospital readmissions in low PIFR patients 2

Compelling Need for Once-Daily Nebulized LAMA ENDURING PATIENT NICHE AND SIGNIFICANT MARKET OPPORTUNITY 11 Enduring patient niche 9% of COPD patients currently use nebulizers for ongoing maintenance therapy 1 >100M patient treatment days in nebulized COPD segment 2 41% of COPD patients use nebulizers at least occasionally for bronchodilator therapy 1 Pricing in branded LA nebulized segment ~ 2x handheld Spiriva 2 Significant market opportunity YUPELRI® may be complementary to existing nebulized LABA treatments Mylan partnership brings commercial strength in nebulized segment 1 TBPH market research (N = 160 physicians); Refers to US COPD patients. 2 IMS Health information service: NSP for period MAT May, 2015. Excludes nebulized SABAs. IMS expressly reserves all rights, including rights of copying, distribution and republication.

Partnership with Mylan Brings Commercial Strength in Nebulized Opportunity 12 HD = hospital discharge HCPs = health care providers Targeting HCPs at key intersections in the patient’s disease management process Hospital is an important site of care for patients with worsening of COPD symptoms Theravance Biopharma’s established hospital-focused sales force is targeting the inpatient setting Theravance Biopharma partners with institutions to transition appropriate patients from hospital to home on YUPELRI® Mylan’s role is to ensure patients remain on YUPELRI® for maintenance therapy in the outpatient setting Combined sales infrastructures cover Hospital, Hospital Discharge and Home Health settings HD +

YUPELRI® Launch Update ENCOURAGING INITIAL MARKET RESPONSE 13 42 Wins (equates to 136 accounts) ~93 Reviews Scheduled (~405 potential accounts) 100% medical support requests fulfilled <30 days FORMULARY Field force productivity goals exceeded ~7,000 patients prescribed (thru 2Q19) PATIENT 100% Medicare Part B 1 ~46% Commercial Permanent J-CODE issued (effective July 1, 2019) ACCESS Majority of YUPELRI® volume flows through durable medical equipment (DME) channel 2; remaining volume flows through hospitals, retail and long-term care pharmacies WAC: $1,030 per month (or ~$34 per day) 1 For patients with supplemental insurance 2 Approximately 3 month lag in data capture 13

Opportunity for YUPELRI® (revefenacin) in China POTENTIAL TO ADDRESS LARGE AND UNDERSERVED COPD PATIENT POPULATION 14 Expansion of development and commercialization agreement Mylan granted exclusive development and commercialization rights to revefenacin in China and adjacent territories Theravance Biopharma eligible to receive: $18.5 million upfront payment Up to $54 million in additional potential development and sales milestones Tiered royalties on net sales, if approved Mylan responsible for all aspects of development and commercialization in partnered regions Significant market opportunity COPD affects ~100 million individuals in China1 ~43% of COPD patients suffer from moderate to very severe forms of disease2 COPD is one of the top three causes of death in China3 and presents significant financial burden to healthcare system2 Theravance Biopharma and Mylan strategic collaboration In 2015, the companies established a strategic collaboration to develop and commercialize nebulized revefenacin products for COPD and other respiratory diseases Theravance Biopharma eligible to receive up to $259 million in potential development and sales milestone payments, as well as profit-sharing arrangement with Mylan on US sales and tiered royalties on ex-US sales Theravance Biopharma retains worldwide rights delivered through other dosage forms, including metered dose inhaler and dry powder inhaler (MDI/PDI) 1C. Wang, J. Xu, L. Yang et al., “Prevalence and risk factors of chronic obstructive pulmonary disease in China (the China Pulmonary Health [CPH] study): a national cross-sectional study,” The Lancet, vol. 391, no. 10131, pp. 1706–1717, 2018. 2 Fang L, Gao P, Bao H, et al., “Chronic obstructive pulmonary disease in China: a nationwide prevalence study,” Lancet Respir Med 2018; 6: 421–430. 3 Yin P, Wang H, Vos T, et al., “A subnational analysis of mortality and prevalence of COPD in China From 1990 to 2013: Findings from the global burden of disease study 2013,” Chest. 2016;150:1269–1280. 14

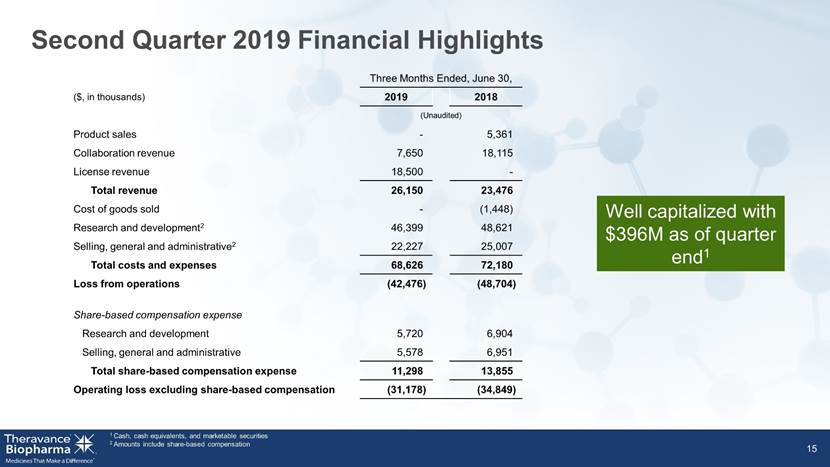
1 Cash, cash equivalents, and marketable securities 2 Amounts include share-based compensation 15 Three Months Ended, June 30, ($, in thousands) 2019 2018 (Unaudited) Product sales - 5,361 Collaboration revenue 7,650 18,115 License revenue 18,500 - Profit sharing revenue - - Total revenue 26,150 23,476 Cost of goods sold - (1,448) Research and development1 46,399 48,621 Selling, general and administrative1 22,227 25,007 Total costs and expenses 68,626 72,180 Loss from operations (42,476) (48,704) Share-based compensation expense Research and development 5,720 6,904 Selling, general and administrative 5,578 6,951 Total share-based compensation expense 11,298 13,855 Operating loss excluding share-based compensation (31,178) (34,849) Second Quarter 2019 Financial Highlights Well capitalized with $396M as of quarter end1
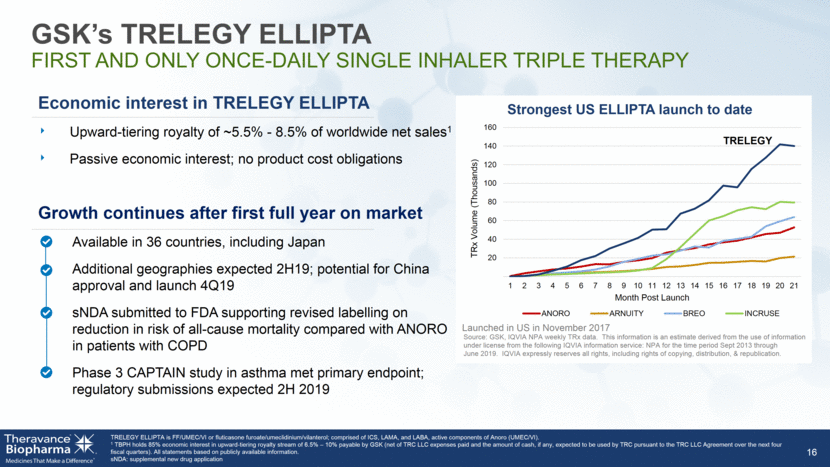
GSK’s TRELEGY ELLIPTA FIRST AND ONLY ONCE-DAILY SINGLE INHALER TRIPLE THERAPY 16 Economic interest in TRELEGY ELLIPTA Upward-tiering royalty of ~5.5% - 8.5% of worldwide net sales1 Passive economic interest; no product cost obligations Growth continues after first full year on market Available in 36 countries, including Japan Additional geographies expected 2H19; potential for China approval and launch 4Q19 sNDA submitted to FDA supporting revised labelling on reduction in risk of all-cause mortality compared with ANORO in patients with COPD Phase 3 CAPTAIN study in asthma met primary endpoint; regulatory submissions expected 2H 2019 TRELEGY ELLIPTA is FF/UMEC/VI or fluticasone furoate/umeclidinium/vilanterol; comprised of ICS, LAMA, and LABA, active components of Anoro (UMEC/VI). 1 TBPH holds 85% economic interest in upward-tiering royalty stream of 6.5% – 10% payable by GSK (net of TRC LLC expenses paid and the amount of cash, if any, expected to be used by TRC pursuant to the TRC LLC Agreement over the next four fiscal quarters). All statements based on publicly available information. sNDA: supplemental new drug application 20 40 60 80 100 120 140 160 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 TRx Volume (Thousands) Month Post Launch Strongest US ELLIPTA launch to date ANORO ARNUITY BREO INCRUSE TRELEGY Launched in US in November 2017 Source: GSK, IQVIA NPA weekly TRx data. This information is an estimate derived from the use of information under license from the following IQVIA information service: NPA for the time period Sept 2013 through June 2019. IQVIA expressly reserves all rights, including rights of copying, distribution, & republication.
Key programs drive near- and long-term value-creating events Focus on Strategic Priorities COMMITMENT TO CREATING TRANSFORMATIONAL MEDICINES 17 TRELEGY ELLIPTA is FF/UMEC/VI or fluticasone furoate/umeclidinium/vilanterol; comprised of ICS, LAMA, and LABA, active components of Anoro (UMEC/VI). 1 TBPH holds 85% economic interest in upward-tiering royalty stream of 6.5% – 10% payable by GSK (net of TRC LLC expenses paid and the amount of cash, if any, expected to be used by TRC pursuant to the TRC LLC Agreement over the next four fiscal quarters). All statements based on publicly available information. Opportunities to Create Transformational Medicines YUPELRI® Nebulized LAMA in COPD U.S. commercial launch underway TD-1473 Gut-selective oral JAK inhibitor for inflammatory intestinal diseases Phase 2b/3 RHEA study in ulcerative colitis ongoing; Phase 2b data planned late-2020 Phase 2 DIONE study in Crohn’s disease ongoing; data planned late-2020 Ampreloxetine NRI in symptomatic neurogenic orthostatic hypotension (nOH) Registrational Phase 3 program progressing; 4-week efficacy data expected 2H 2020 TD-8236 Lung-selective inhaled pan-JAK inhibitor for serious respiratory diseases Safety and biomarker data from Phase 1 study in healthy volunteers and asthmatics expected September 2019 Research Organ-selective research platform designed to expand therapeutic index compared to conventional systemic therapies Economic Interest TRELEGY ELLIPTA1 Single inhaler triple therapy in COPD Product launched in 36 countries, including Japan; China approval expected 4Q19 sNDA submitted to FDA supporting revised labelling on reduction in risk of all-cause mortality compared with ANORO ELLIPTA in patients with COPD Potential sNDA for asthma indication in 2H 2019
