Exhibit 99.2

First Quarter 2022 Financial Results and Business Update May 5, 2022 THERAVANCE BIOPHARMA ® , THERAVANCE ® , the Cross/Star logo and MEDICINES THAT MAKE A DIFFERENCE ® are registered trademarks of the Theravance Biopharma group of companies (in the U.S. and certain other countries). All third party trademarks used herein are the property of their respective owners. © 2022 Theravance Biopharma. All rights reserved.

Under the safe harbor provisions of the U . S . Private Securities Litigation Reform Act of 1995 , the company cautions investors that any forward - looking statements or projections made by the company are subject to risks and uncertainties that may cause actual results to differ materially from the forward - looking statements or projections . Examples of forward - looking statements in this presentation may include the Company's goals, designs, strategies, plans and objectives, the impact of the Company’s restructuring plan, ability to provide value to shareholders, the Company's regulatory strategies and timing of clinical studies (including the data therefrom), the potential characteristics, benefits and mechanisms of action of the Company's product and product candidates, the potential that the Company's research programs will progress product candidates into the clinic or will be partnered successfully, the Company's expectations for product candidates through development and the market for products being commercialized, the Company's expectations regarding its allocation of resources, potential regulatory actions and commercialization (including differentiation from other products or potential products and addressable market), product sales or profit share revenue and the Company's expectations for its expenses, excluding share - based compensation and other financial results . The company’s forward - looking statements are based on the estimates and assumptions of management as of the date of this presentation and are subject to risks and uncertainties that may cause the actual results to be materially different than those projected, such as risks related to the impacts on the COVID - 19 global pandemic on our business, disagreements with Innoviva , Inc . and TRC LLC, the uncertainty of arbitration and litigation and the possibility that the results of these proceedings could be adverse to the Company, additional future analysis of the data resulting from our clinical trial(s), delays or difficulties in commencing, enrolling or completing clinical studies, the potential that results from clinical or non - clinical studies indicate the Company's compounds, products or product candidates are unsafe, ineffective or not differentiated, risks of decisions from regulatory authorities that are unfavorable to the Company, the feasibility of undertaking future clinical trials based on policies and feedback from regulatory authorities, dependence on third parties to conduct clinical studies, delays or failure to achieve and maintain regulatory approvals for product candidates, risks of collaborating with or relying on third parties to discover, develop, manufacture and commercialize products, and risks associated with establishing and maintaining sales, marketing and distribution capabilities with appropriate technical expertise and supporting infrastructure, ability to retain key personnel, the impact of the Company’s restructuring actions on its employees, partners and others . Other risks affecting Theravance Biopharma are in the company's Form 10 - K filed with the SEC on February 28 , 2022 , and other periodic reports filed with the SEC . Forward - looking statements 2

Agenda 3 Introduction Gail B. Cohen Corporate Communications Overview Rick E Winningham Chief Executive Officer Commercial and Development Update Rhonda F. Farnum Senior Vice President, Chief Business Officer Richard A. Graham Senior Vice President, Research and Development Financial Update Andrew A. Hindman Senior Vice President, Chief Financial Officer Closing Remarks Rick E Winningham Chief Executive Officer
Rapid transition to a focused and streamlined Theravance Biopharma 4 Significant cost reduction program reduces Company size to become sustainably cash - flow positive beginning 2H 2022 Focus on leveraging expertise in developing and commercializing respiratory therapeutics Streamlined R&D investment to focus on highest value respiratory opportunities Leverage partnerships to unlock value of pipeline assets Overarching goal: maximize shareholder value

Key pillars of focused value creation plan 1. 1. Source: TBPH Analysts Consensus [8] December 9, 2021. 2. Source: Bloomberg GSK Analyst Consensus April 27, 2022. 3. TBPH h old s 85% economic interest in upward - tiering royalty stream of 6.5% – 10% payable by GSK (net of TRC expenses paid and the amount of cash, if any, expected to be used by TRC pursuant to the TRC Agree men t over the next four fiscal quarters). 75% of TRC income received is pledged to service outstanding notes, 25% of royalties retained by TBPH. Our non - recourse Triple II 9.5% Fixed Rate Term Notes are due o n or before 2035. 2. LAMA, long - acting muscarinic antagonist; PIFR, peak inspiratory flow rate. 5 YUPELRI ® Maximize growing value of YUPELRI ‣ Consensus US peak year sales of ~$400 million 1 ‣ Demonstrated growth and strong cash flow generation ‣ Unique value proposition for the only once daily nebulized LAMA ‣ PIFR - 2 study intended to strengthen competitive advantage and capture more of the addressable market ‣ Long patent life Pipeline Limited strategic investments to advance pipeline ‣ Leveraging our internal expertise in development of inhaled lung - selective agents ‣ Mid - year meeting with FDA to align on approval path for ampreloxetine ‣ Pursuing strategic collaborations across pipeline to optimize value Economic interest in GSK's TRELEGY 3 TRELEGY ‣ Consensus global peak year sales of ~$ 3.5 billion 2 ‣ Q1 2022 net sales of $ 454 million implies run rate annual sales of ~$ 1.8 billion 3 ‣ Long patent life ‣ TRELEGY - related cash flows to TBPH to increase substantially (once non - recourse note is fully repaid) 3

FDA - approved for maintenance treatment of COPD First and only once - daily, LAMA (long - acting muscarinic agent) nebulized maintenance medicine for COPD
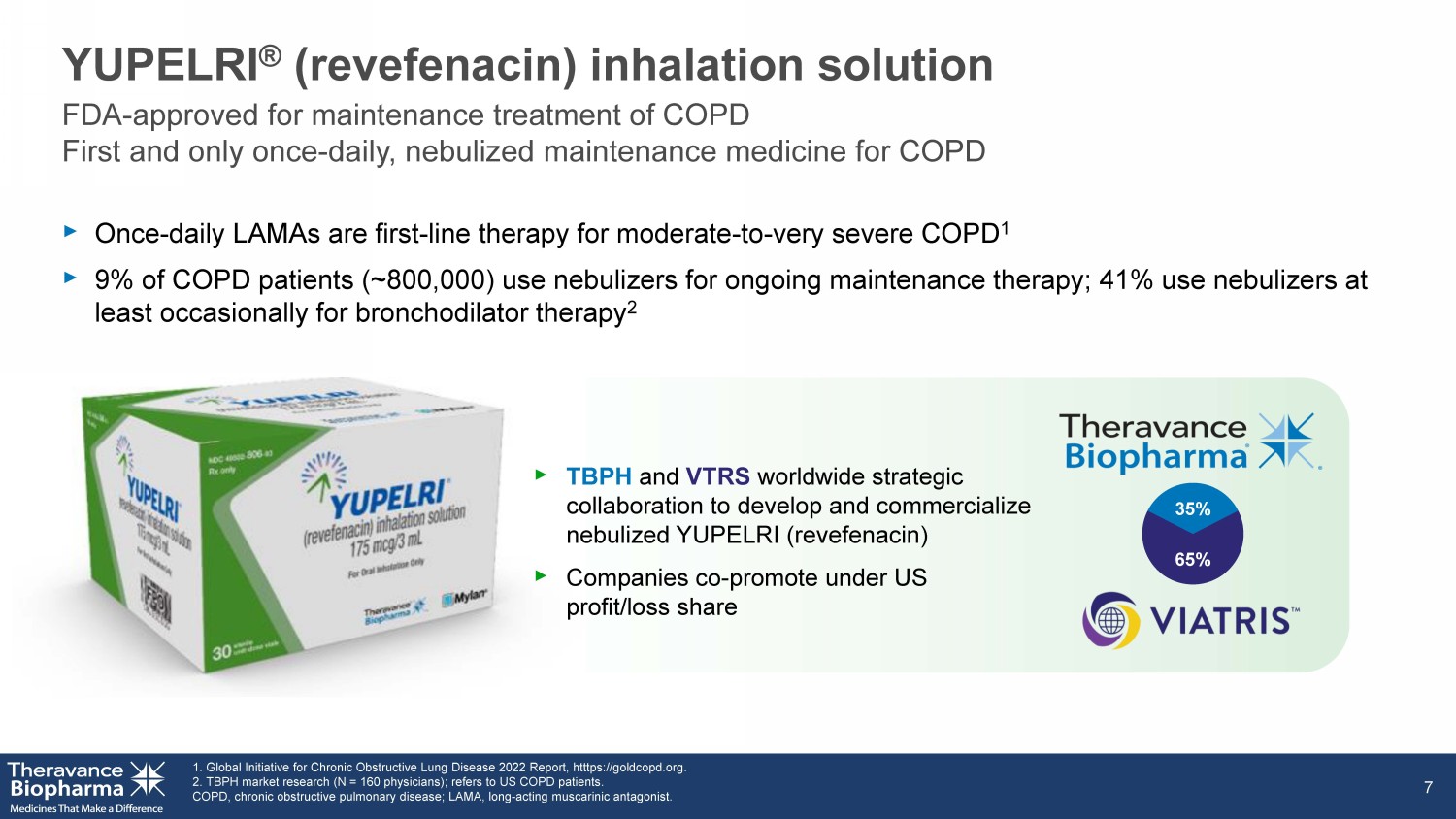
‣ Once - daily LAMAs are first - line therapy for moderate - to - very severe COPD 1 ‣ 9% of COPD patients (~800,000) use nebulizers for ongoing maintenance therapy; 41% use nebulizers at least occasionally for bronchodilator therapy 2 YUPELRI ® ( revefenacin ) inhalation solution 1. Global Initiative for Chronic Obstructive Lung Disease 2022 Report, htttps://goldcopd.org . 2. TBPH market research (N = 160 physicians); refers to US COPD patients. COPD, chronic obstructive pulmonary disease; LAMA, long - acting muscarinic antagonist. 7 ‣ TBPH and VTRS worldwide strategic collaboration to develop and commercialize nebulized YUPELRI (revefenacin) ‣ Companies co - promote under US profit/loss share 65% 35% FDA - approved for maintenance treatment of COPD First and only once - daily, nebulized maintenance medicine for COPD
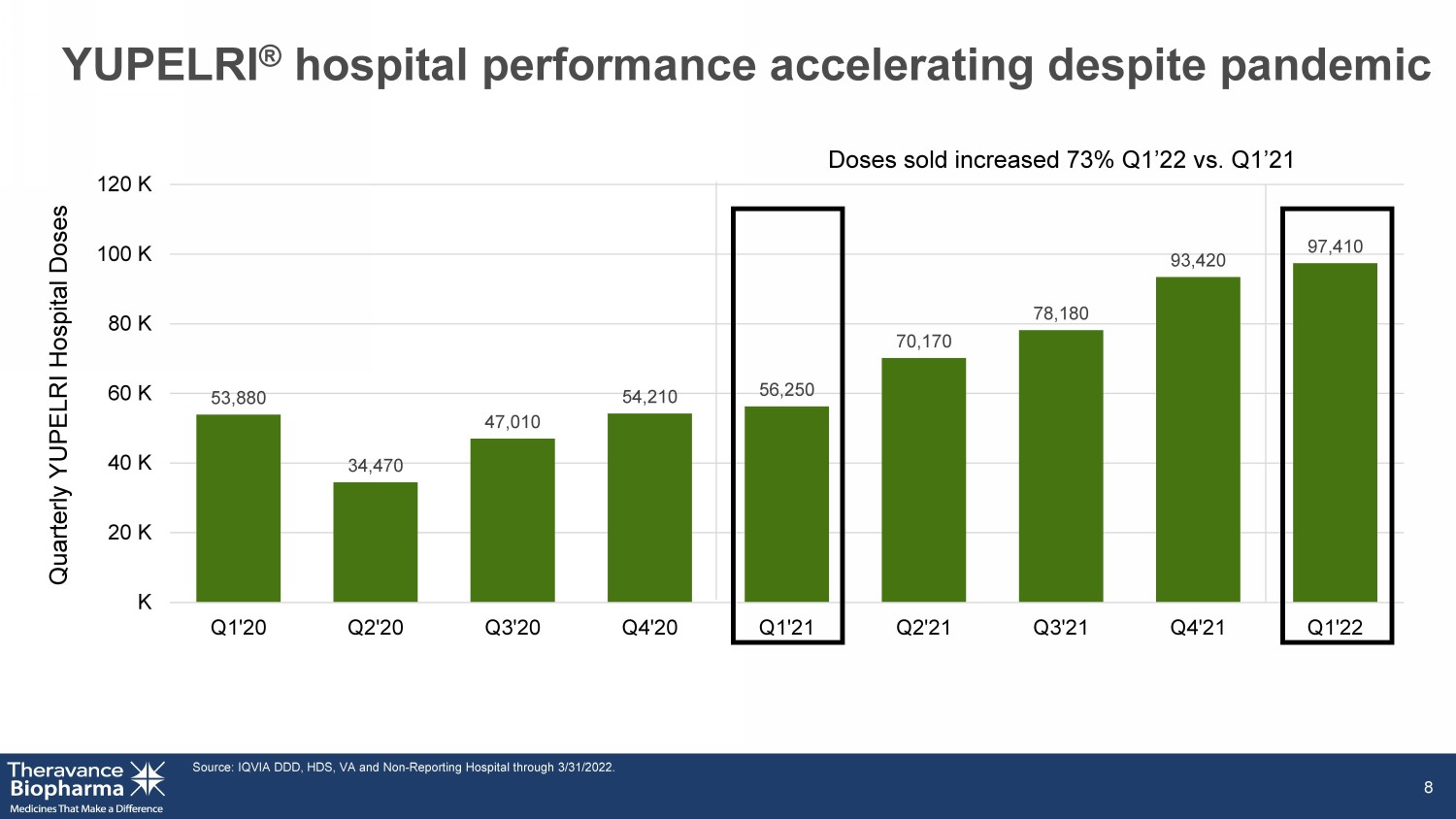
8 Quarterly YUPELRI Hospital Doses 53,880 34,470 47,010 54,210 56,250 70,170 78,180 93,420 97,410 K 20 K 40 K 60 K 80 K 100 K 120 K Q1'20 Q2'20 Q3'20 Q4'20 Q1'21 Q2'21 Q3'21 Q4'21 Q1'22 YUPELRI ® hospital performance accelerating despite pandemic Source: IQVIA DDD, HDS, VA and Non - Reporting Hospital through 3/31/2022. Doses sold increased 73% Q1’22 vs. Q1’21

YUPELRI ® hospital sales and community TRx trends Continued market share growth across both the hospital and retail channels 1.1% 2.2% 3.4% 5.4% 6.5% 6.6% 7.4% 7.8% 8.7% 9.5% 10.8% 11.3% 0% 5% 10% 15% 20% 25% 30% Q2'19 Q3'19 Q4'19 Q1'20 Q2'20 Q3'20 Q4'20 Q1'21 Q2'21 Q3'21 Q4'21 Q1'22 Hospital LA - NEB Market Share* Hospital Market Share YUPELRI LA-NEB Market Share 1. Joint VTRS/TBPH Market Research. * Hospital LA - NEB Market Share - IQVIA DDD through 3/31/2022. ** Community LA - NEB Market Share - IQVIA XPO Excl. LTC (Retail) and SolutionsRx (DME / Med B FFS) through 1/31/2022 (Q1’22 Community LA - NEB Market Share Incomplete). *** Retail TRx Volume - Symphony Health METYS Prescription Dashboard through 3/31/2022. 9 TRx volume represents retail only which is typically 33% of Retail + DME Reported DME volume, while lagged, typically follows Retail volume trends 6.1% 9.8% 13.1% 15.2% 16.3% 17.4% 18.7% 19.7% 21.4% 22.7% 23.2% 23.5% 0K 5K 10K 15K 20K 25K 0% 5% 10% 15% 20% 25% 30% Q2'19 Q3'19 Q4'19 Q1'20 Q2'20 Q3'20 Q4'20 Q1'21 Q2'21 Q3'21 Q4'21 Q1'22 Retail TRx Volume*** Community LA - NEB Market Share** Community Market Share with TRx Retail TRx YUPELRI LA-NEB Market Share Most patients who receive YUPELRI ® in the hospital are discharged with an Rx 1 **Community LA - NEB Market Share includes Retail + DME / Med B FFS through Jan’22 LA - NEB Market: YUPELRI, BROVANA, LONHALA, PERFOROMIST, arformoterol , formoterol
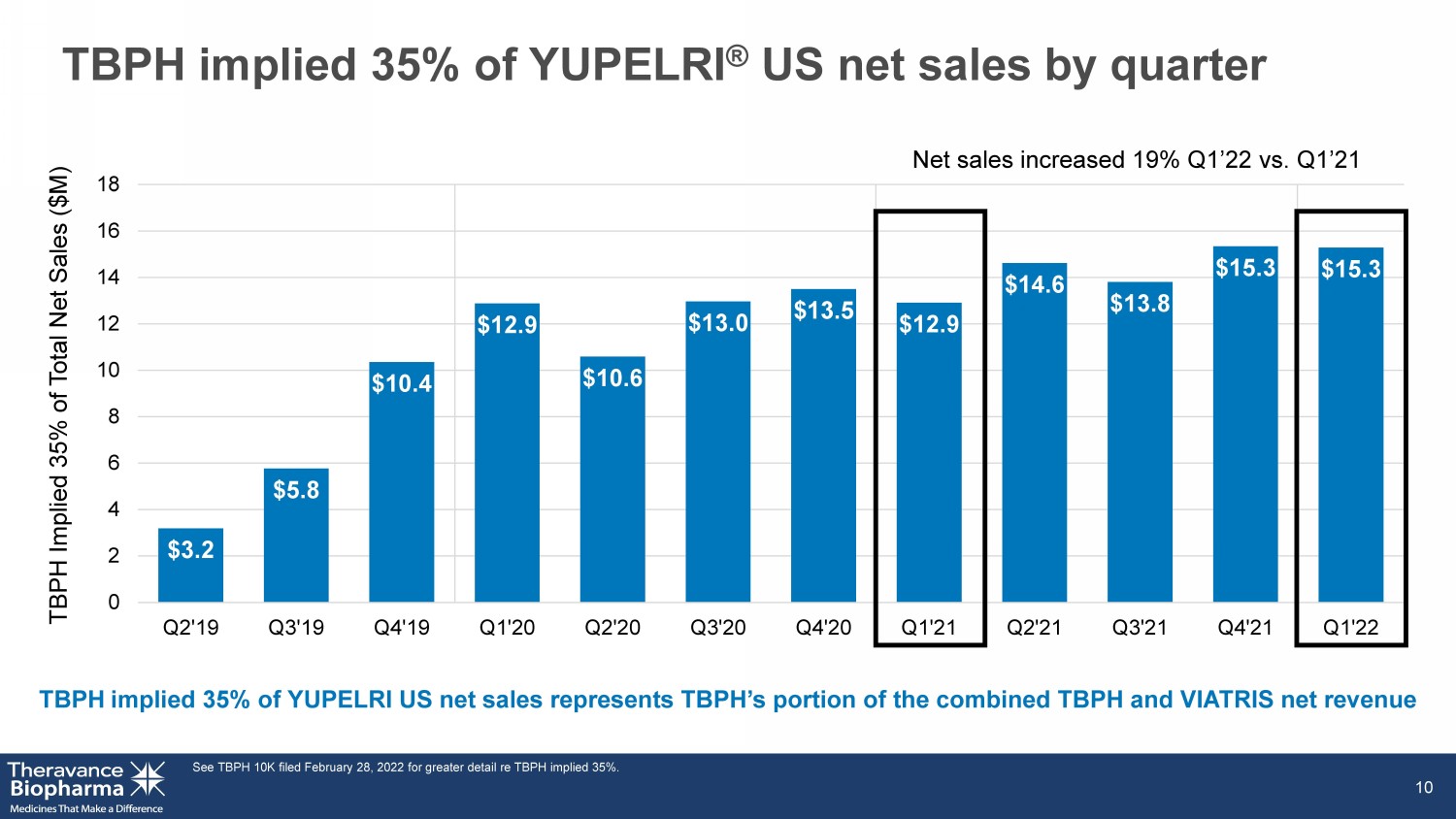
TBPH implied 35% of YUPELRI ® US net sales by quarter See TBPH 10K filed February 28, 2022 for greater detail re TBPH implied 35%. 10 TBPH implied 35% of YUPELRI US net sales represents TBPH’s portion of the combined TBPH and VIATRIS net revenue TBPH Implied 35% of Total Net Sales ($M) $3.2 $5.8 $10.4 $12.9 $10.6 $13.0 $13.5 $12.9 $14.6 $13.8 $15.3 $15.3 0 2 4 6 8 10 12 14 16 18 Q2'19 Q3'19 Q4'19 Q1'20 Q2'20 Q3'20 Q4'20 Q1'21 Q2'21 Q3'21 Q4'21 Q1'22 Net sales increased 19% Q1’22 vs. Q1’21

A new focused and streamlined Theravance Biopharma 1. TBPH estimate derived from integrating multiple data sources 2. TBPH holds 85% economic interest in upward - tiering royalty st ream of 6.5% – 10% payable by GSK (net of TRC expenses paid and the amount of cash, if any, expected to be used by TRC pursuant to the TRC Agreement over the next four fiscal quarters). 75% of TRC income received is p led ged to service outstanding notes, 25% of royalties received retained by TBPH. All statements concerning TRELEGY ELLIPTA based on publicly available information. ALK5i, transforming growth factor β receptor I kinase inhibitor; COPD, chronic obstructive pulmonary disease; FF/UMEC/VI, fluticasone furoate/umeclidinium/ vilanterol; JAKi , JAK inhibitor; LAMA, long - acting muscarinic antagonist ; nOH , neurogenic orthostatic hypotension; NRI, norepinephrine reuptake inhibitor; PIFR, peak inspiratory flow rate. 1 1 Program Indication US Patients 1 Research Phase 1 Phase 2 Phase 3 Filed Marketed Phase 4 Collaborator Commercial Asset YUPELRI ® ( revefenacin ) LAMA COPD patients with suboptimal PIFR >8mm Pipeline Assets Nezulcitinib (TD - 0903) Inhaled JAKi Acute and chronic lung inflammation, fibrotic disease >32mm Inhaled JAKi Asthma ~25mm Inhaled ALK5i Idiopathic pulmonary fibrosis ~140k Ampreloxetine (TD - 9855) NRI S ymptomatic nOH ~350k Economic Interests TRELEGY 2 FF/UMEC/VI COPD > 8mm GSK & Innoviva , Inc. Asthma ~25mm Skin - selective JAKi Dermatological diseases >8mm Phase 3 Phase 2 Marketed Phase 4 PIFR - 2 Study Marketed Phase 1 Marketed Phase 1 Phase 1
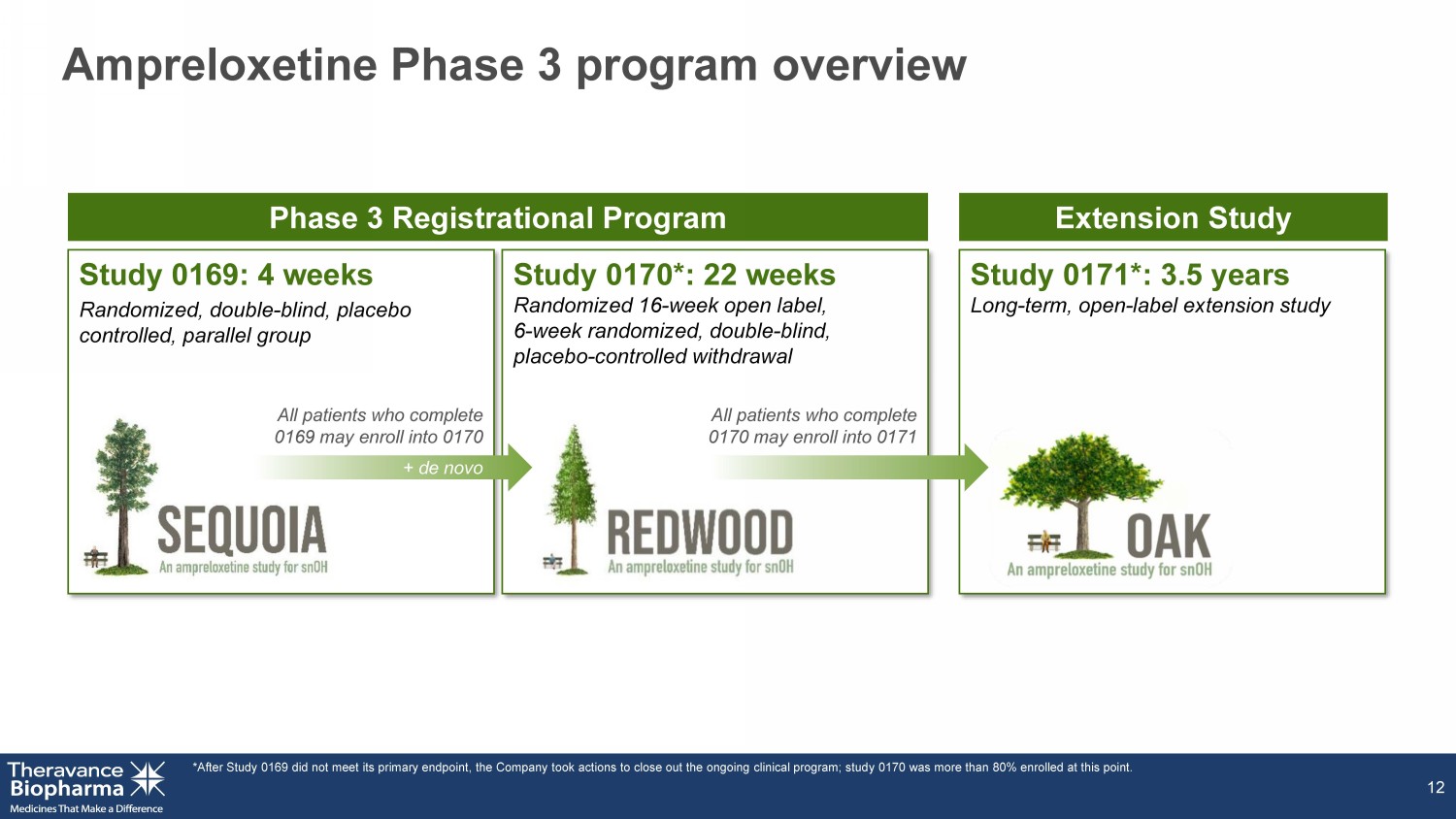
Ampreloxetine Phase 3 program overview 12 Study 0169: 4 weeks Randomized, double - blind, placebo controlled, parallel group Phase 3 Registrational Program Extension Study All patients who complete 0169 may enroll into 0170 All patients who complete 0170 may enroll into 0171 + de novo Study 0170*: 22 weeks Randomized 16 - week open label, 6 - week randomized, double - blind, placebo - controlled withdrawal Study 0171*: 3.5 years Long - term, open - label extension study *After Study 0169 did not meet its primary endpoint, the Company took actions to close out the ongoing clinical program; stud y 0 170 was more than 80% enrolled at this point.
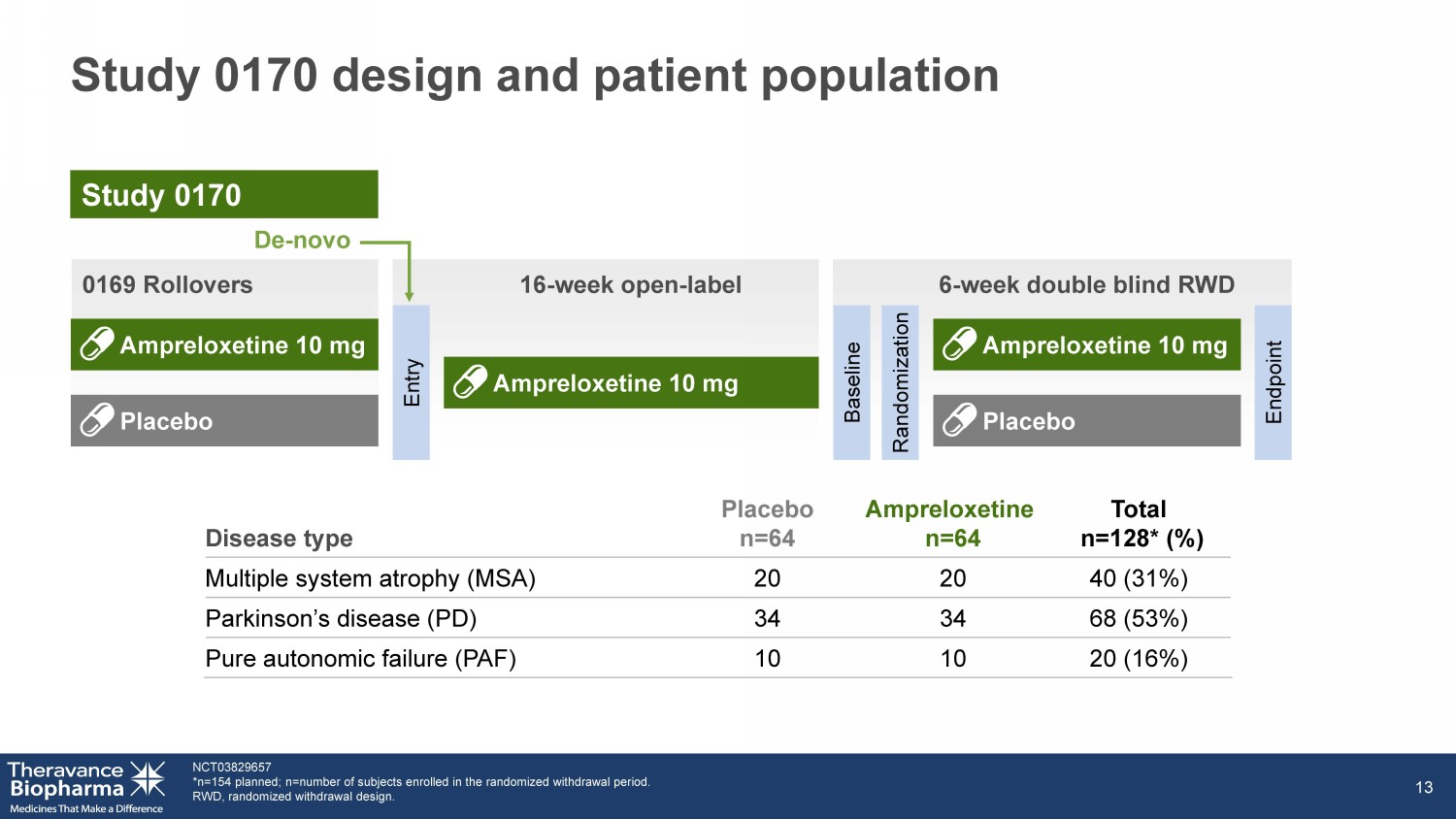
Study 0170 design and patient population NCT03829657 *n=154 planned; n =number of subjects enrolled in the randomized withdrawal period. RWD, randomized withdrawal design. 13 Disease type Placebo n=64 Ampreloxetine n=64 Total n=128* (%) Multiple system atrophy (MSA) 20 20 40 (31%) Parkinson’s disease (PD) 34 34 68 (53%) Pure autonomic failure (PAF) 10 10 20 (16%) Entry Endpoint 6 - week double blind RWD 0169 Rollovers 16 - week open - label Baseline Randomization De - novo Ampreloxetine 10 mg Placebo Ampreloxetine 10 mg Ampreloxetine 10 mg Placebo Study 0170
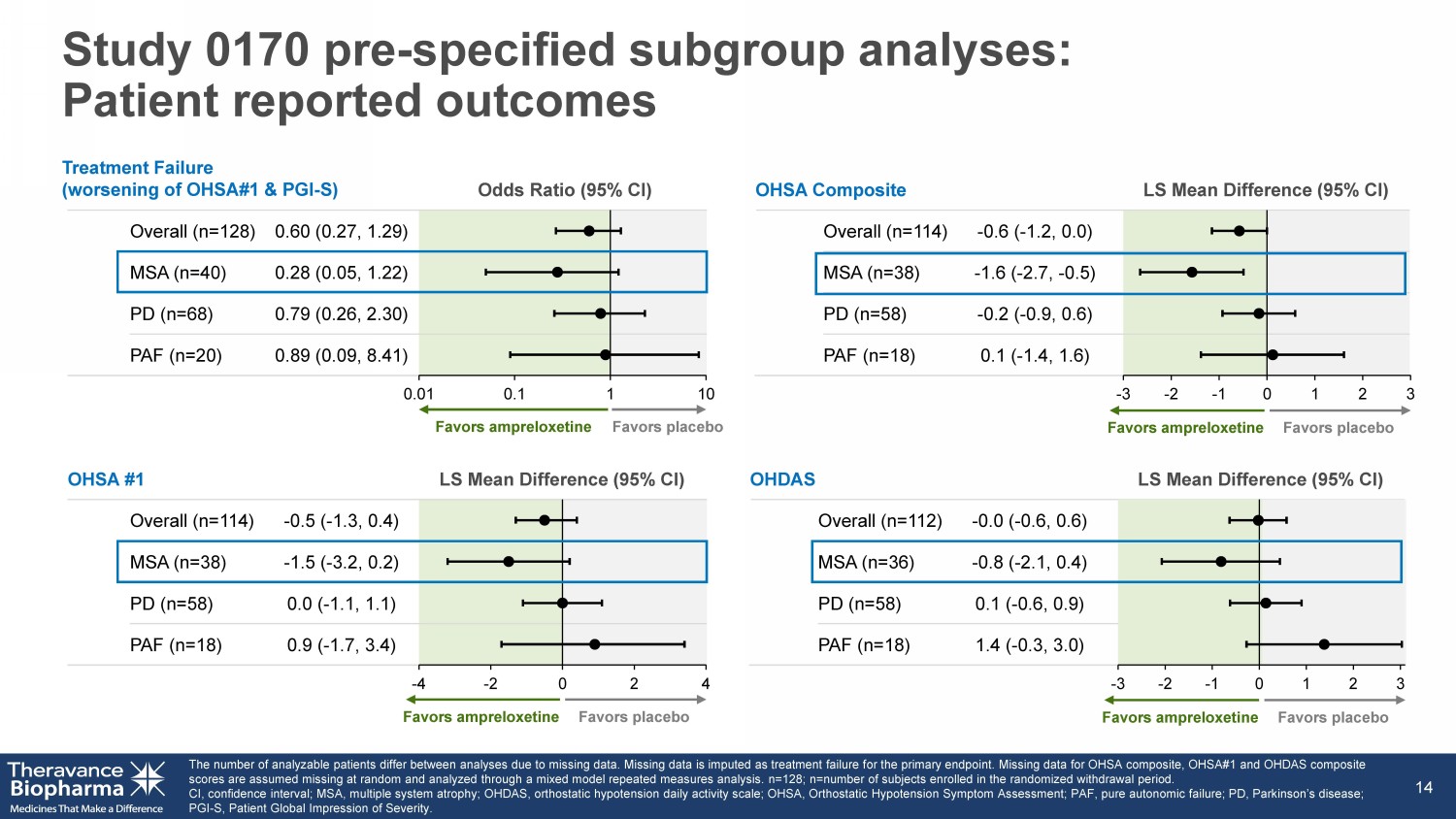
OHDAS LS Mean Difference (95% CI) Overall (n=112) - 0.0 ( - 0.6, 0.6) MSA (n=36) - 0.8 ( - 2.1, 0.4) PD (n=58) 0.1 ( - 0.6, 0.9) PAF (n=18) 1.4 ( - 0.3, 3.0 ) Odds Ratio (95% CI) Overall (n=128 ) 0.60 (0.27, 1.29) MSA (n=40) 0.28 (0.05, 1.22) PD (n=68) 0.79 (0.26, 2.30) PAF (n=20) 0.89 (0.09, 8.41) OHSA #1 LS Mean Difference (95% CI) Overall (n=114) - 0.5 ( - 1.3, 0.4) MSA (n=38) - 1.5 ( - 3.2, 0.2) PD (n=58) 0.0 ( - 1.1, 1.1) PAF (n=18) 0.9 ( - 1.7, 3.4) Study 0170 pre - specified subgroup analyses: Patient reported outcomes The number of analyzable patients differ between analyses due to missing data. Missing data is imputed as treatment failure f or the primary endpoint. Missing data for OHSA composite, OHSA#1 and OHDAS composite scores are assumed missing at random and analyzed through a mixed model repeated measures analysis. n=128; n =number of subjects enrolled in the randomized withdrawal period. CI, confidence interval; MSA, multiple system atrophy; OHDAS, orthostatic hypotension daily activity scale; OHSA, Orthostatic Hy potension Symptom Assessment; PAF, pure autonomic failure; PD, Parkinson’s disease; PGI - S, Patient Global Impression of Severity. 14 0.01 0.1 1 10 -4 -2 0 2 4 Favors ampreloxetine Favors placebo Favors ampreloxetine Favors placebo Treatment Failure (worsening of OHSA#1 & PGI - S) OHSA Composite LS Mean Difference (95% CI) Overall (n=114) - 0.6 ( - 1.2, 0.0) MSA (n=38) - 1.6 ( - 2.7, - 0.5) PD (n=58) - 0.2 ( - 0.9, 0.6) PAF (n=18) 0.1 ( - 1.4, 1.6) -3 -2 -1 0 1 2 3 Favors ampreloxetine Favors placebo -3 -2 -1 0 1 2 3 Favors ampreloxetine Favors placebo

LS Mean (95% CI) OHSA Symptom composite score - 1.6 ( - 2.7, - 0.5) Dizziness - 1.5 ( - 3.2, 0.2) Vision - 1.7 ( - 3.2, - 0.3) Weakness - 0.7 ( - 2.3, 0.9) Fatigue - 1.5 ( - 3.1, 0.1) Trouble concentrating - 1.8 ( - 3.3, - 0.4) Head/neck discomfort - 2.2 ( - 3.7, - 0.7) OHDAS Daily activities composite score - 0.8 ( - 2.1, 0.4) Standing short time - 2.0 ( - 3.6, - 0.4) Standing long time - 0.3 ( - 1.8, 1.2) Walking short time - 0.7 ( - 2.4, 1.0) Walking long time 0.4 ( - 1.2, 2.1) OHQ composite score - 1.2 ( - 2.3, - 0.2) -4 -2 0 2 4 Study 0170 OHQ questionnaire composite scores and individual items for MSA patients CI, confidence interval; MSA, multiple system atrophy; OHDAS, orthostatic hypotension daily activity scale; OHQ, orthostatic hyp otension questionnaire; OHSA, Orthostatic Hypotension Symptom Assessment. Individual item score analyses are post - hoc, except for dizziness. 15 Favors ampreloxetine Favors placebo
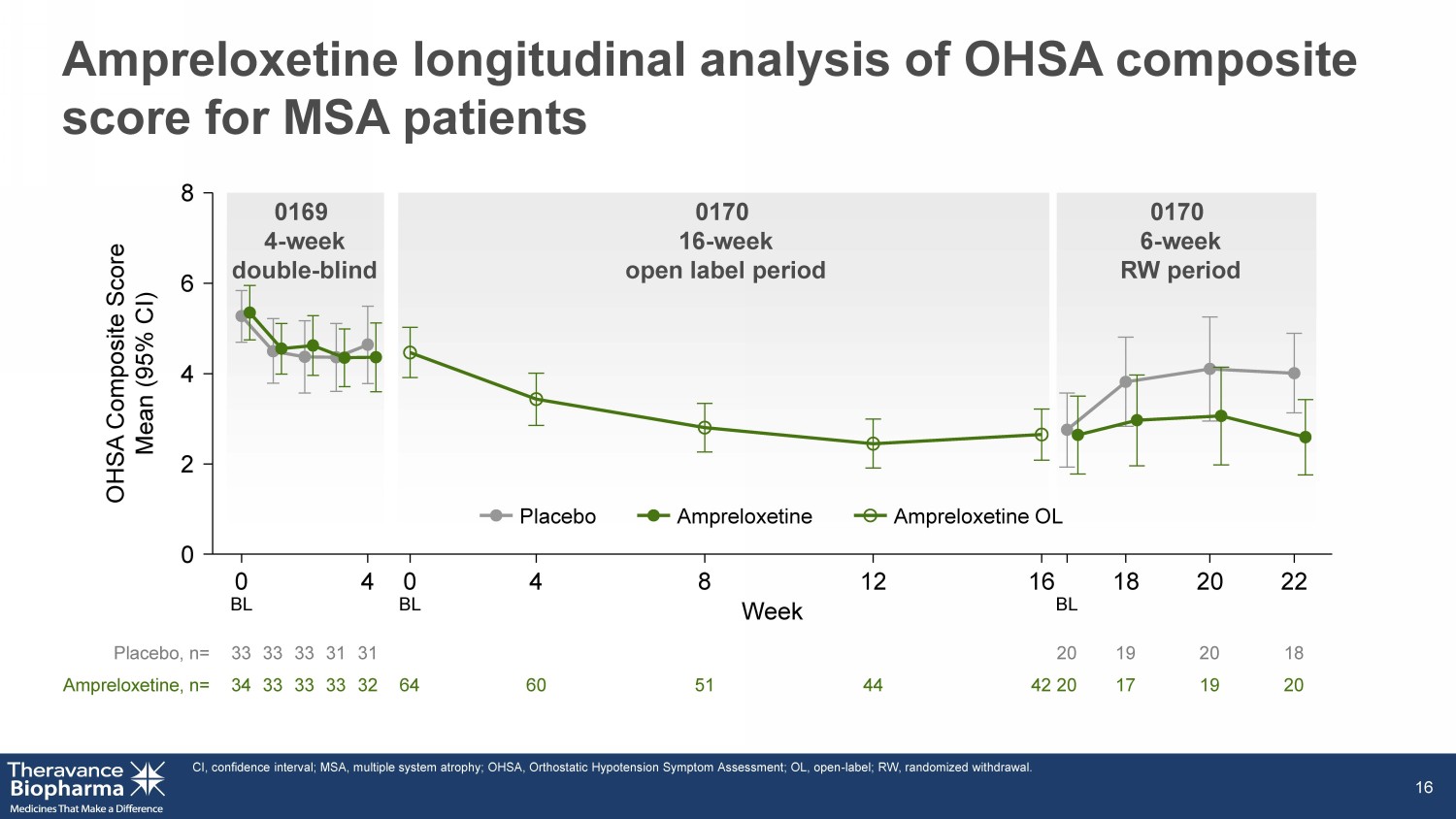
Ampreloxetine longitudinal analysis of OHSA composite score for MSA patients 16 33 34 33 33 33 33 31 33 31 32 64 60 51 44 42 20 20 19 17 20 19 18 20 0 0 4 0 4 8 12 16 18 20 22 2 4 6 8 0169 4 - week double - blind 0170 6 - week RW period 0170 16 - week open label period Placebo, n= Ampreloxetine, n= Week OHSA Composite Score Mean (95% CI) CI, confidence interval; MSA, multiple system atrophy; OHSA, Orthostatic Hypotension Symptom Assessment; OL, open - label; RW, ran domized withdrawal. BL BL BL Ampreloxetine OL Ampreloxetine Placebo

Economic interest GSK’s TRELEGY ELLIPTA (FF/UMEC/VI): First and only once - daily single inhaler triple therapy

Economic interest in GSK’s TRELEGY 1. TBPH holds 85% economic interest in upward - tiering royalty stream of 6.5% – 10% payable by GSK (net of TRC expenses paid and the amount of cash, if any, expected to be used by TRC pursuant to the TRC Agreement over the next four fiscal quarters). 75% of TRC income received is pledged to service outstanding notes, 25% of royalties retained by TBPH. Our non - recourse Triple II 9.5% Fixed Rate Term Notes are due on or before 2035. All statements concerning TRELEGY based on publicly available information. TRELEGY is FF/UMEC/VI or flutic aso ne furoate/umeclidinium/vilanterol; comprised of inhaled corticosteroid, long - acting muscarinic receptor antagonist, and long - acting β2 agonists, active components of Anoro (UMEC/VI). 1 8 TRELEGY Upward - tiering royalties of ~5.5 – 8.5% of global net sales 1 Q1 global net sales of $454M Year - over - year sales growth of 33% from the same period in 2021 0 100 200 300 400 500 600 1 3 5 7 9 11 13 15 17 19 21 23 25 27 29 31 33 35 37 39 41 43 45 47 49 51 53 Monthly TRx Volume (Thousands) Month Post Launch Strongest US ELLIPTA Launch ANORO ELLIPTA ARNUITY ELLIPTA BREO ELLIPTA INCRUSE ELLIPTA Launched in US in November 2017 Source: GSK, Symphony Health Metys monthly TRx data for the time period Sept'13 to Mar ’22. TRELEGY Mortality Ad Comm TRELEGY Asthma Approval BREO Asthma Approval TRELEGY
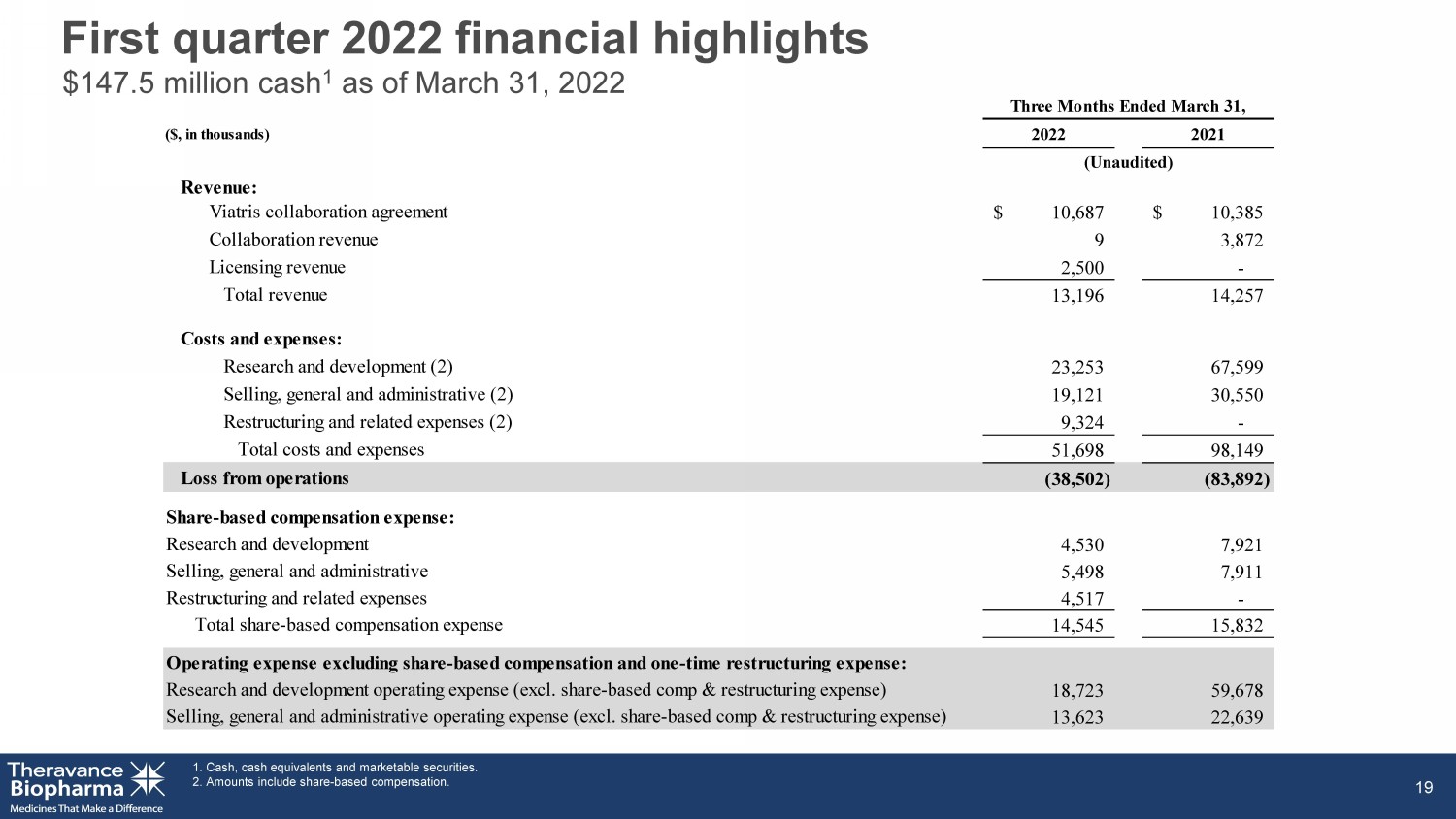
First quarter 2022 financial highlights 1. Cash, cash equivalents and marketable securities. 2. Amounts include share - based compensation. 19 $147.5 million cash 1 as of March 31, 2022 ($, in thousands) Revenue: Viatris collaboration agreement $ 10,687 $ 10,385 Collaboration revenue 9 3,872 Licensing revenue 2,500 - Total revenue 13,196 14,257 Costs and expenses: Research and development (2) 23,253 67,599 Selling, general and administrative (2) 19,121 30,550 Restructuring and related expenses (2) 9,324 - Total costs and expenses 51,698 98,149 Loss from operations (38,502) (83,892) Share-based compensation expense: Research and development 4,530 7,921 Selling, general and administrative 5,498 7,911 Restructuring and related expenses 4,517 - Total share-based compensation expense 14,545 15,832 Operating expense excluding share-based compensation and one-time restructuring expense: Research and development operating expense (excl. share-based comp & restructuring expense) 18,723 59,678 Selling, general and administrative operating expense (excl. share-based comp & restructuring expense) 13,623 22,639 Three Months Ended March 31, 2022 2021 (Unaudited)
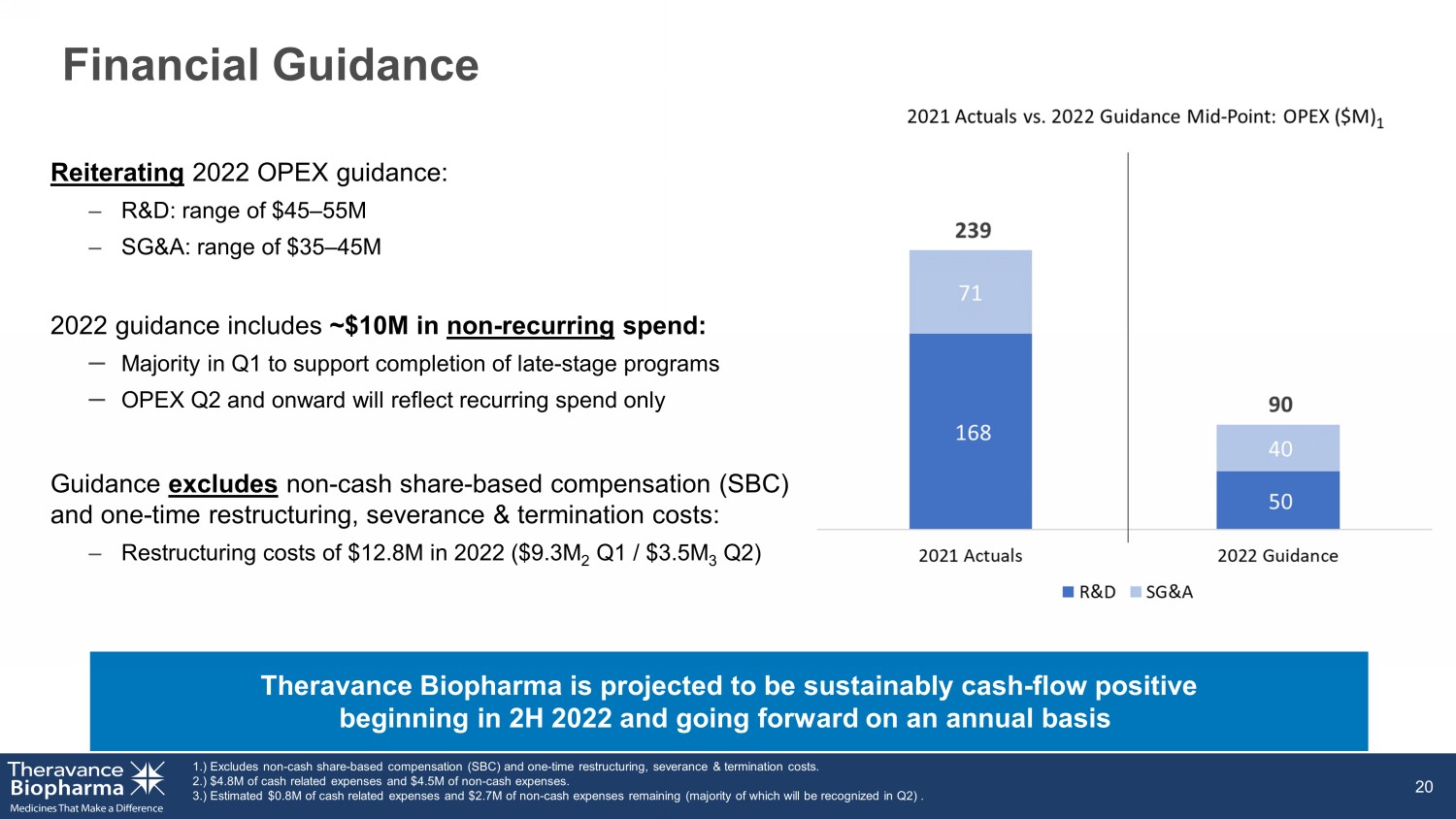
Financial Guidance 20 Reiterating 2022 OPEX guidance: – R&D: range of $45 – 55M – SG&A: range of $35 – 45M 2022 guidance includes ~$10M in non - recurring spend: – Majority in Q1 to support completion of late - stage programs – OPEX Q2 and onward will reflect recurring spend only Guidance excludes non - cash share - based compensation (SBC) and one - time restructuring, severance & termination costs: – Restructuring costs of $12.8M in 2022 ($9.3M 2 Q1 / $3.5M 3 Q2) Theravance Biopharma is projected to be sustainably cash - flow positive beginning in 2H 2022 1.) Excludes non - cash share - based compensation (SBC) and one - time restructuring, severance & termination costs. 2.) $4.8M of cash related expenses and $4.5M of non - cash expenses. 3.) Estimated $0.8M of cash related expenses and $2.7M of non - cash expenses remaining (majority of which will be recognized in Q 2) .
Rapid transition to a focused and streamlined Theravance Biopharma 21 Significant cost reduction program reduces Company size to become sustainably cash - flow positive beginning 2H 2022 Focus on leveraging expertise in developing and commercializing respiratory therapeutics Streamlined R&D investment to focus on highest value respiratory opportunities Leverage partnerships to unlock value of pipeline assets Overarching goal: maximize shareholder value

Q&A Session Rick E Winningham Chairman and Chief Executive Officer Andrew A. Hindman Senior Vice President, Chief Financial Officer Rhonda F. Farnum Senior Vice President, Chief Business Officer Richard A. Graham Senior Vice President, Research and Development

YUPELRI ® ( revefenacin ) inhalation solution YUPELRI ® inhalation solution is indicated for the maintenance treatment of patients with chronic obstructive pulmonary disease (COPD) . Important Safety Information (US) YUPELRI is contraindicated in patients with hypersensitivity to revefenacin or any component of this product . YUPELRI should not be initiated in patients during acutely deteriorating or potentially life - threatening episodes of COPD, or for the relief of acute symptoms, i . e . , as rescue therapy for the treatment of acute episodes of bronchospasm . Acute symptoms should be treated with an inhaled short - acting beta 2 - agonist . As with other inhaled medicines, YUPELRI can produce paradoxical bronchospasm that may be life - threatening . If paradoxical bronchospasm occurs following dosing with YUPELRI, it should be treated immediately with an inhaled, short - acting bronchodilator . YUPELRI should be discontinued immediately and alternative therapy should be instituted . YUPELRI should be used with caution in patients with narrow - angle glaucoma . Patients should be instructed to immediately consult their healthcare provider if they develop any signs and symptoms of acute narrow - angle glaucoma, including eye pain or discomfort, blurred vision, visual halos or colored images in association with red eyes from conjunctival congestion and corneal edema . Worsening of urinary retention may occur . Use with caution in patients with prostatic hyperplasia or bladder - neck obstruction and instruct patients to contact a healthcare provider immediately if symptoms occur . Immediate hypersensitivity reactions may occur after administration of YUPELRI . If a reaction occurs, YUPELRI should be stopped at once and alternative treatments considered . The most common adverse reactions occurring in clinical trials at an incidence greater than or equal to 2 % in the YUPELRI group, and higher than placebo, included cough, nasopharyngitis, upper respiratory infection, headache and back pain . Coadministration of anticholinergic medicines or OATP 1 B 1 and OATP 1 B 3 inhibitors with YUPELRI is not recommended . YUPELRI is not recommended in patients with any degree of hepatic impairment . OATP, organic anion transporting polypeptide. 23

About YUPELRI ® ( revefenacin ) inhalation solution YUPELRI ® ( revefenacin ) inhalation solution is a once - daily nebulized LAMA approved for the maintenance treatment of COPD in the US . Market research by Theravance Biopharma indicates approximately 9 % of the treated COPD patients in the US use nebulizers for ongoing maintenance therapy . 1 LAMAs are a cornerstone of maintenance therapy for COPD and YUPELRI ® is positioned as the first once - daily single - agent bronchodilator product for COPD patients who require, or prefer, nebulized therapy . YUPELRI ® ’s stability in both metered dose inhaler and dry powder device formulations suggest that this LAMA could also serve as a foundation for novel handheld combination products . 1. TBPH market research (N=160 physicians); refers to US COPD patients. COPD, chronic obstructive pulmonary disease; LAMA, long - acting muscarinic antagonist. 24