Corporate Presentation 2018 James Sapirstein, CEO NASDAQ: CTRV Issuer Free Writing Prospectus dated June 5, 2018 Relating to Prospectus dated June 4, 2018 Filed Pursuant to Rule 43 3 Registration Statement File No. 333 - 225041

This presentation may contain forward-looking statements within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. Such forward-looking statements are characterized by future or conditional verbs such as “may,” “will,” “expect,” “intend,” “anticipate,” believe,” “estimate” and “continue” or similar words. You should read statements that contain these words carefully because they discuss future expectations and plans, which contain projections of future results of operations or financial condition or state other forward-looking information. Such statements are only predictions and our actual results may differ materially from those anticipated in these forward-looking statements. We believe that it is important to communicate future expectations to investors. However, there may be events in the future that we are not able to accurately predict or control. Factors that may cause such differences include, but are not limited to, those discussed under Risk Factors in our periodic reports filed with the Securities and Exchange Commission, including the uncertainties associated with product development, the risk that products that appeared promising in early clinical trials do not demonstrate safety and efficacy in larger-scale clinical trials, the risk that we will not obtain approval to market our products, the risks associated with dependence upon key personnel and the need for additional financing. We do not assume any obligation to update forward-looking statements as circumstances change. This presentation does not constitute an offer or invitation for the sale or purchase of securities or to engage in any other transaction with ContraVir or its affiliates. The information in this presentation is not targeted at the residents of any particular country or jurisdiction and is not intended for distribution to, or use by, any person in any jurisdiction or country where such distribution or use would be contrary to local law or regulation. This presentation highlights basic information about ContraVir Pharmaceuticals, Inc. and the offering. ContraVir Pharmaceuticals, Inc. has filed a registration statement on Form S-1 (Registration Statement File No. 333-225041) (including a prospectus) with the U.S. Securities and Exchange Commission (“SEC”) for the offering to which this presentation relates. The registration statement has not yet become effective. Before you invest, you should read the prospectus in the registration statement (including among other things, the risks factors described therein) and other documents the issuer has filed with the SEC for more complete information about ContraVir Pharmaceuticals, Inc. and this offering. The preliminary prospectus dated June 4, 2018 and subsequent amendments are available at the SEC website. You may get these documents for free by visiting EDGAR on the SEC website at www.sec.gov. Alternatively, ContraVir Pharmaceuticals, Inc. or any placement agent or any dealer participating in the offering will arrange to send you the prospectus if you request it by contacting Maxim Group LLC, 405 Lexington Ave., New York, NY 10174, Attn: Prospectus Department by tel: (800) 724-0761. This presentation contains statistics and other data that has been obtained from or compiled from information made available by third parties service providers. ContraVir Pharmaceuticals, Inc. has not independently verified such statistics or data. The information contained in this presentation is as of June 4, 2018, unless indicated otherwise. Safe Harbor Statement

Corporate Overview Preclinical Phase 1 Phase 2 Phase 3 TXL™ Hepatitis B Company Profile Committed to finding the right combination for a curative regimen in hepatitis B Value Drivers TXL™: Novel, target organ specific, lipid conjugate of tenofovir for treating hepatitis B Completed Phase 2a trial Commencement of second generation formulation development program Clinical Program initiated in the US FDA agreement on NDA package leveraging 505(b)(2) Regulatory Pathway CRV431: Novel, high-potency cyclophilin inhibitor for treating hepatitis B Strong preclinical proof of concept established IND submitted Clinical Program expected to be initiated Q3 2018 CRV431 Hepatitis B **Continued strong execution of HBV portfolio Development Programs**

Experienced Leadership James Sapirstein, R.Ph., M.B.A Chief Executive Officer 30+ years bio/pharma experience, including Gilead Sciences, Hoffmann-La Roche and BMS Key in the development and launch of Viread® Founder of Tobira Therapeutics (acquired by Allergan for $1.7 billion) Theresa Matkovits, Ph.D. Chief Operating Officer 20+ years of proven leadership in global drug development and commercialization spanning multi-national Pharma to Emerging Biotech Global Development Team Lead for four globally marketed products John Cavan Chief Financial Officer 25+ years financial management experience at both public and private companies Extensive experience in public offerings and strategic transactions Robert Foster, Pharm.D., Ph.D. Chief Scientific Officer 30+ years of pharmaceutical discovery and development (voclosporin, Helikit) Founder of Aurinia Pharmaceuticals (formerly known as Isotechnika) and discoverer of Voclosporin

Scientific Advisory Board Carol L. Brosgart, M.D. Special Adviser of Health Care for FIPRA International, Clinical Professor of Medicine in the Divisions of Global Health Sciences Jordan Feld, M.D., M.P.H. Clinician-scientist , Western Hospital Liver Clinic & McLaughlin-Rotman Centre for Global Health Philippe Gallay, Ph.D. Cyclophilin Expert, The Scripps Research Institute Jörg Petersen, M.D., Ph.D. Professor of Medicine & Head of Liver Unit, Askepios Klinik St. George Nikolai Naoumov, M.D., Ph.D. Executive Director, Hepatology Science and Innovation, Novartis Pharma Fabien Zoulim, M.D., Ph.D. Medical Director of Hepatology Dept , Hospices Civils de Lyon and Scientific Director of the Dept of Immunology & Virology Henry Chan Lik Yuven, M.D., Head of Division of Gastroenterolgy and Hepatology, Director of Institute of Digestive Disease, Director of Center for Liver Health Jin-Lin Hou, M.D., Director and Professor of the Hepatology Unit and Department of Infectious Diseases Experienced Leadership Board of Directors Gary Jacob, Ph.D. (Chair) Executive Chairman, Synergy Pharmaceuticals Timothy M. Block, Ph.D. Co-Founder and President, Hepatitis B Foundation Tamar D. Howson, M.B.A. Former Head of Business Development at BMS Arnold S. Lippa, Ph.D. Founder of DOV Pharmaceuticals Thomas H. Adams, Ph.D. Former Chairman & CEO of Leucadia Technologies John P. Brancaccio, CPA Former CFO of Accelerated Technologies
15.4%* growth in chronic HBV patients in the U.S. through 20332 350 million chronic HBV patients worldwide1 800 thousand HBV-related deaths per year3 HBV Market Potential 1 World Health Organization 2013 2 Datamonitor 2014 3 Lozano R, et al. Lancet 2012 4 William Blair Report 2016 HBV-related ESLD or HCC responsible for >0.5-1 million deaths per year, currently represent 5-10% of liver transplant cases HBV treatments resulted in $700 million 2015 U.S. Sales4 Cumulative sales for HBV treatments in the United States, the EU Five and Japan is expected to exceed $200 billion over the next 20 years4

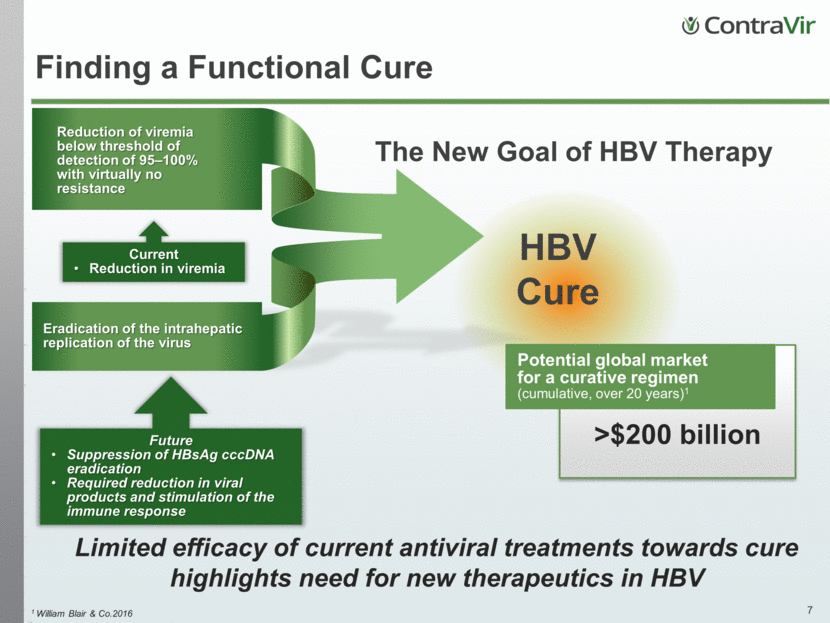
Reduction of viremia below threshold of detection of 95–100% with virtually no resistance Finding a Functional Cure HBV Cure Eradication of the intrahepatic replication of the virus Current Reduction in viremia >$200 billion Potential global market for a curative regimen (cumulative, over 20 years)1 The New Goal of HBV Therapy Future Suppression of HBsAg cccDNA eradication Required reduction in viral products and stimulation of the immune response 1 William Blair & Co.2016 Limited efficacy of current antiviral treatments towards cure highlights need for new therapeutics in HBV
First Generation Tenofovir Tenofovir Disoproxil (TDF) Brand name: Viread® Bioavailability is about 25% (fasting), with systemic exposure increased by 40% with high fat meals Light meals do not significantly impact the PK of TDF TDF is unstable and high systemic blood levels of TFV linked to kidney and bone toxicities Currently Marketed Tenofovir Compounds Second Generation Tenofovir Tenofovir Aladenamide (TAF) Brand name: Vemlidy® Must be taken with food to enhance absorption across the gut TAF exposures (AUC) increased by 1.65 fold with food (e.g., 25 mg TAF with food equivalent exposure to approximately 40 mg fasting) Increases lipids / abnormalities over that observed with TDF Less efficacious than TDF in high viral load patients Serum amylase elevations Hepatic flares Effective in treatment of chronic HBV and HIV Nucleotide analog reverse transcriptase inhibitor

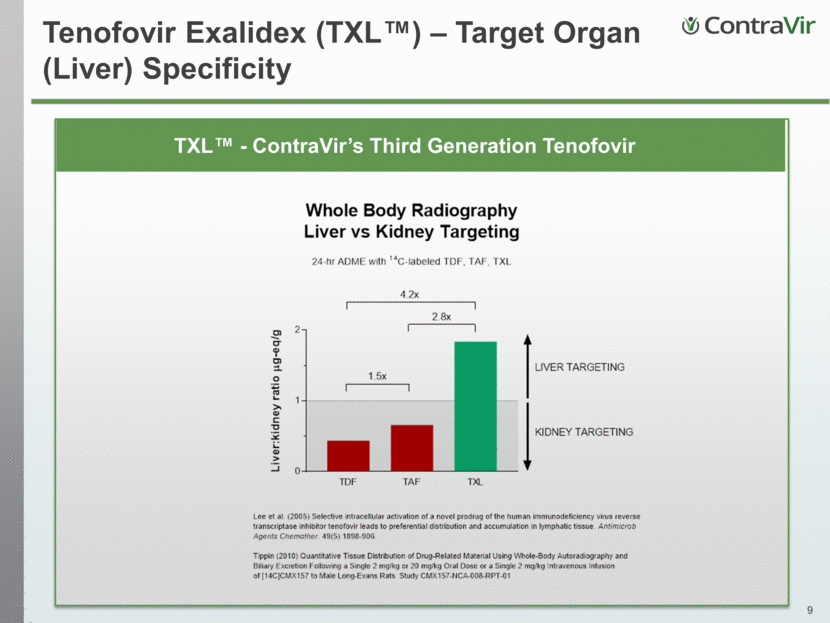
TXL™ - ContraVir’s Third Generation Tenofovir Tenofovir Exalidex (TXL™) – Target Organ (Liver) Specificity
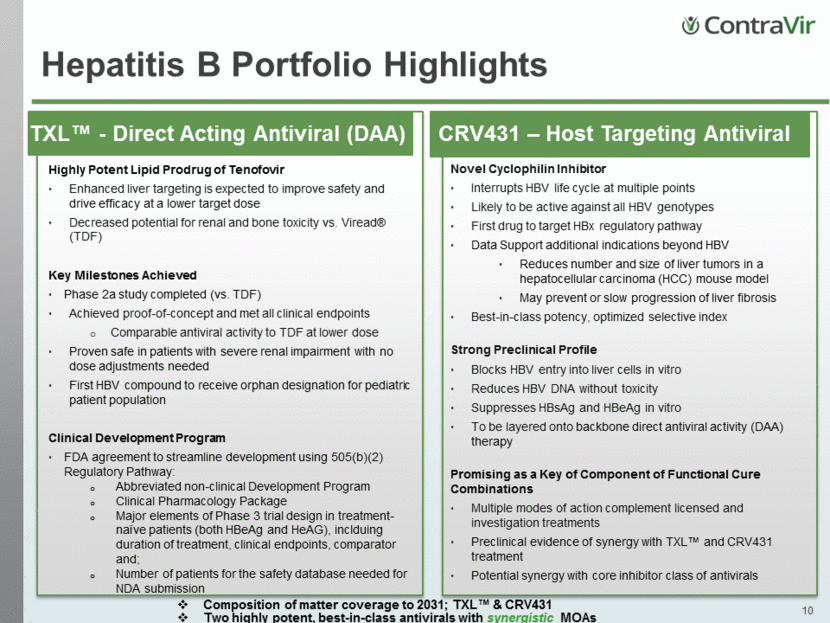
Hepatitis B Portfolio Highlights TXL™ - Direct Acting Antiviral (DAA) Highly Potent Lipid Prodrug of Tenofovir Enhanced liver targeting is expected to improve safety and drive efficacy at a lower target dose Decreased potential for renal and bone toxicity vs. Viread® (TDF) Key Milestones Achieved Phase 2a study completed (vs. TDF) Achieved proof-of-concept and met all clinical endpoints Comparable antiviral activity to TDF at lower dose Proven safe in patients with severe renal impairment with no dose adjustments needed First HBV compound to receive orphan designation for pediatric patient population Clinical Development Program FDA agreement to streamline development using 505(b)(2) Regulatory Pathway: Abbreviated non-clinical Development Program Clinical Pharmacology Package Major elements of Phase 3 trial design in treatment-naïve patients (both HBeAg and HeAG), inclduing duration of treatment, clinical endpoints, comparator and; Number of patients for the safety database needed for NDA submission CRV431 – Host Targeting Antiviral Novel Cyclophilin Inhibitor Interrupts HBV life cycle at multiple points Likely to be active against all HBV genotypes First drug to target HBx regulatory pathway Data Support additional indications beyond HBV Reduces number and size of liver tumors in a hepatocellular carcinoma (HCC) mouse model May prevent or slow progression of liver fibrosis Best-in-class potency, optimized selective index Strong Preclinical Profile Blocks HBV entry into liver cells in vitro Reduces HBV DNA without toxicity Suppresses HBsAg and HBeAg in vitro To be layered onto backbone direct antiviral activity (DAA) therapy Promising as a Key of Component of Functional Cure Combinations Multiple modes of action complement licensed and investigation treatments Preclinical evidence of synergy with TXL™ and CRV431 treatment Potential synergy with core inhibitor class of antivirals Composition of matter coverage to 2031; TXL™ & CRV431 Two highly potent, best-in-class antivirals with synergistic MOAs
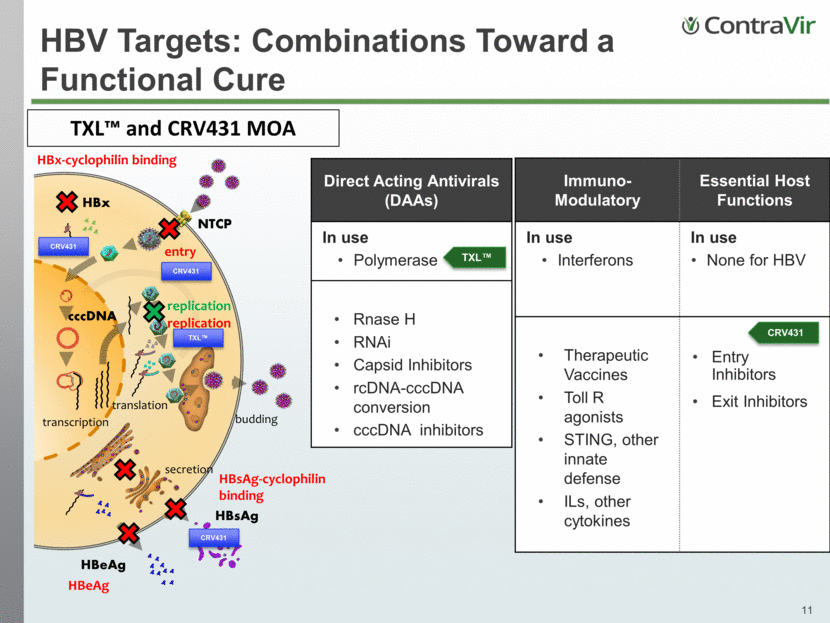
HBV Targets: Combinations Toward a Functional Cure Direct Acting Antivirals (DAAs) In use Polymerase Rnase H RNAi Capsid Inhibitors rcDNA-cccDNA conversion cccDNA inhibitors TXL™ Immuno- Modulatory Essential Host Functions In use Interferons In use None for HBV Therapeutic Vaccines Toll R agonists STING, other innate defense ILs, other cytokines Entry Inhibitors Exit Inhibitors TXL™ and CRV431 MOA HBsAg cccDNA HBeAg NTCP HBx replication transcription entry budding translation secretion HBx-cyclophilin binding HBsAg-cyclophilin binding replication HBeAg CRV431 CRV431 CRV431 TXL™ CRV431
TXL™ De-Risked Development Asset Safety Profile Favorable Pharmacokinetics Compared to Viread® Profile consistent with target organ specificity (liver) resulting in fewer “off-target” effects Significantly lower levels of systemically circulating tenofovir in blood Potent Antiviral Activity at Lower Doses 50 mg/day TXL™ comparable to Viread® 300 mg/day Ongoing formulation optimization to enhance liver targeting to lower mg potency and continue expanding strong safety profile Lower dose should lead to further improved safety profile relative to Viread® and Vemlidy® Strong Candidate to be Partner of Choice Expected to be stand-alone product and backbone to functional cure based on strong safety and efficacy profile Potential to be economically more attractive for potential partners as a Phase 3-ready asset May not be required to be taken with food Potential to offer “best-in-class” target organ specificity (liver) Well-tolerated up to 100 mg daily, with no safety signal No dose adjustments for patients with severe renal impairment Potential for decreased bone and kidney toxicity
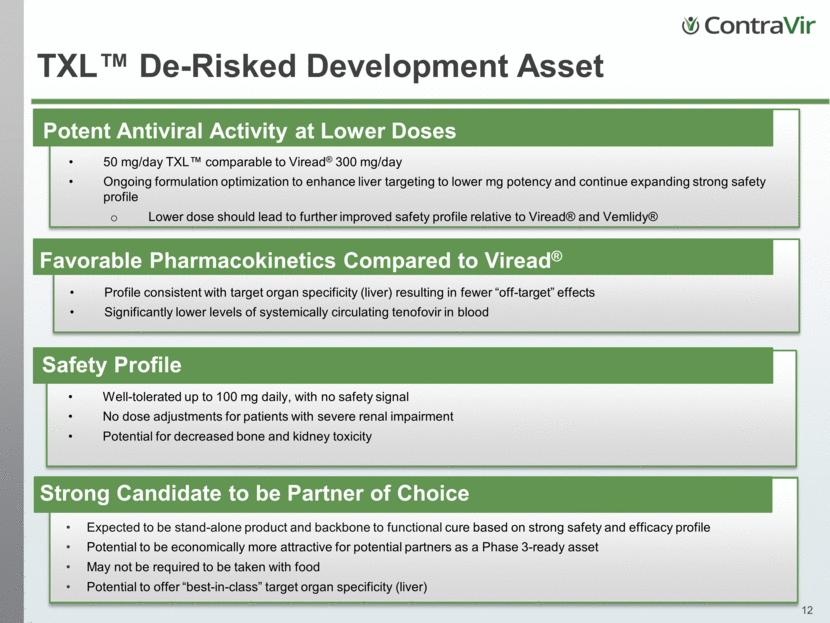
Strong Drug Profile Best-in-class potency against HBV Widest selective index of any known CPI Addresses Key HBV Endpoints Inhibits viral entry Reduces intracellular HBV DNA Reduces HBsAg, pgRNA and HBeAg May prevent liver disease progression (e.g. fibrosis, HCC) CRV431: Novel, Highly Potent Cyclophilin Inhibitor Positioned to be leading host-targeting cyclophilin inhibitor (CPI) Interrupts HBV life cycle at multiple points (HBsAg, HBx and pgRNA) Expected to have activity against all HBV genotypes HBV-Optimized Cyclosporine A Derivative (Non-Immunosuppressive) Sponsored by NIH – NIAID. Study conducted at Institute for Antiviral Research, Utah State University. Wide Selective Index CRV431 High toxicity Low toxicity Low HBV potency High HBV potency alisporivir “YELLOW LIGHT” “RED LIGHT” “GREEN LIGHT” Sangamide(s)
HBV Combination Platform Comparison of expected additive effect versus observed synergistic effect Combining Complementary MOAs TXL™ directly inhibits HBV replication CRV431 blocks HBV from entering liver cells; interrupts viral life cycle at multiple points through interactions with host molecules Highly potent, potentially best-in-class antivirals with synergistic MOAs against hepatitis B Snapshot of Synergy in AD38 cells (in vitro) 1 10 100 1000 0 50 100 Drug nM % I n h i b i t i o n H B V D N A Observed TXL + CRV431 combination synergy Expected additive inhibition CRV431 alone TXL alone Moderate to strong synergy observed in 3 different cell lines: AD38, DE19 and DES19

CRV431 and Liver Disease Chronic HBV infection frequently leads to liver fibrosis, cirrhosis, and liver cancer (hepatocellular carcinoma; HCC) Chronic HBV infection is the leading cause of HCC (approximately 50% of HCC cases), which results in approximately 350,000 deaths globally per year1 In addition to antiviral therapies, new treatments are needed to reduce the liver injury and disease that arises from chronic viral infection 1 World J Hepatol 2015
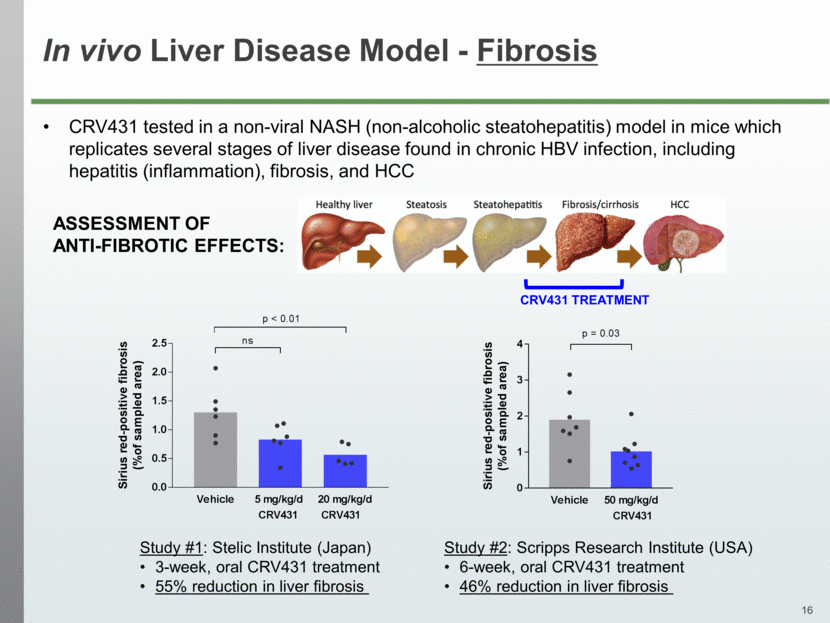
In vivo Liver Disease Model - Fibrosis CRV431 tested in a non-viral NASH (non-alcoholic steatohepatitis) model in mice which replicates several stages of liver disease found in chronic HBV infection, including hepatitis (inflammation), fibrosis, and HCC Study #1: Stelic Institute (Japan) 3-week, oral CRV431 treatment 55% reduction in liver fibrosis CRV431 TREATMENT ASSESSMENT OF ANTI-FIBROTIC EFFECTS: Study #2: Scripps Research Institute (USA) 6-week, oral CRV431 treatment 46% reduction in liver fibrosis Vehicle 5 mg/kg/d 20 mg/kg/d 0.0 0.5 1.0 1.5 2.0 2.5 S i r i u s r e d - p o s i t i v e f i b r o s i s ( % o f s a m p l e d a r e a ) CRV431 CRV431 ns p < 0.01 Vehicle 50 mg/kg/d 0 1 2 3 4 S i r i u s r e d - p o s i t i v e f i b r o s i s ( % o f s a m p l e d a r e a ) CRV431 p = 0.03
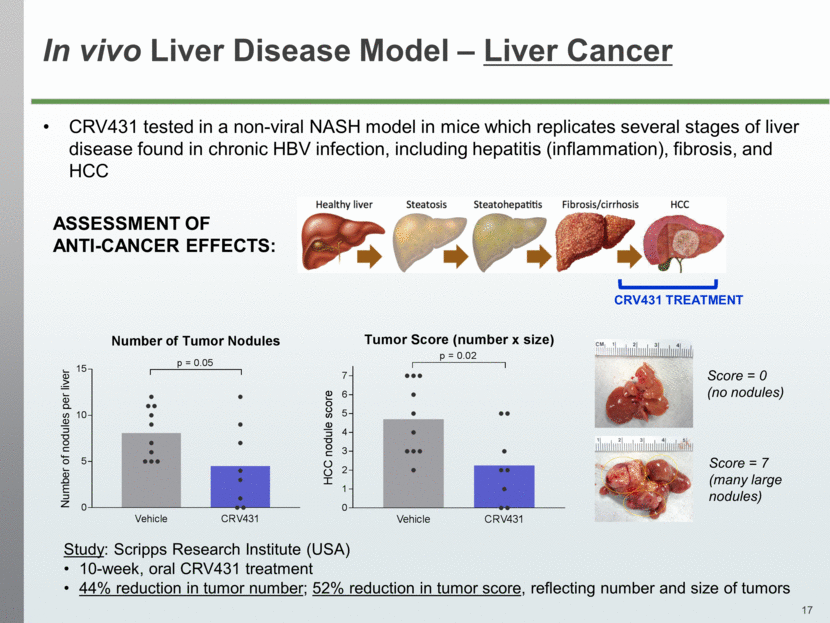
In vivo Liver Disease Model – Liver Cancer CRV431 tested in a non-viral NASH model in mice which replicates several stages of liver disease found in chronic HBV infection, including hepatitis (inflammation), fibrosis, and HCC CRV431 TREATMENT ASSESSMENT OF ANTI-CANCER EFFECTS: Study: Scripps Research Institute (USA) 10-week, oral CRV431 treatment 44% reduction in tumor number; 52% reduction in tumor score, reflecting number and size of tumors Score = 0 (no nodules) Score = 7 (many large nodules) Vehicle CRV431 0 5 10 15 Number of Tumor Nodules N u m b e r o f n o d u l e s p e r l i v e r p = 0.05 Vehicle CRV431 0 1 2 3 4 5 6 7 Tumor Score (number x size) H C C n o d u l e s c o r e p = 0.02
ContraVir Value Drivers Strategic Focus Continue to drive streamlined development of TXL™ and CRV431 as essential elements of one or more curative combinations HBV Program: TXL™ – De-risked Phase 3-Ready Asset CRV431 – Advancing into Clinical Development in the US Combination therapy has a greater chance of achieving a functional cure Strong composition of matter patents for both compounds Leveraging TFV database with 505(b)(2) Regulatory Pathway for streamlined development and commercialization Experienced Leadership Significant expertise in global drug development and commercialization of targeted antivirals

399 Thornall Street, 1st Floor Edison, NJ 08837
