Free signup for more
- Track your favorite companies
- Receive email alerts for new filings
- Personalized dashboard of news and more
- Access all data and search results
Filing tables
Filing exhibits
SCTL similar filings
- 5 Feb 15 Recro Pharma to Present at the 17th Annual BIO CEO & Investor
- 3 Feb 15 Recro Pharma Enters into Common Stock Purchase Agreement with Aspire Capital
- 19 Dec 14 Departure of Directors or Certain Officers
- 10 Nov 14 Recro Pharma Reports Third Quarter 2014 Financial Results
- 30 Oct 14 Recro Pharma Announces Dosing of First Patient in Phase II REC-14-013 Clinical Trial of Dex-IN for Treatment of Acute Pain on Day 1 Following Surgery
- 9 Sep 14 Recro Pharma to Present at the 2014 Aegis Healthcare & Technology Conference
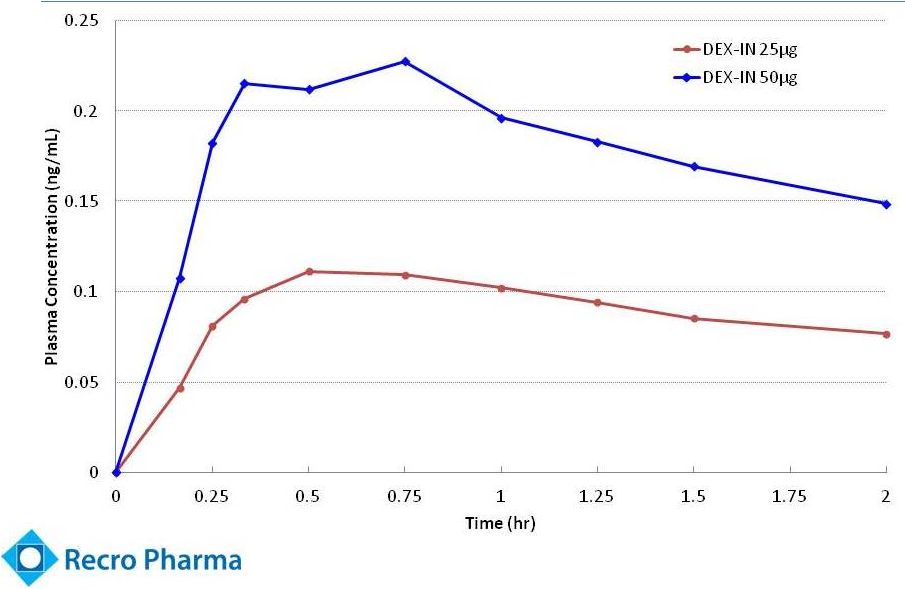
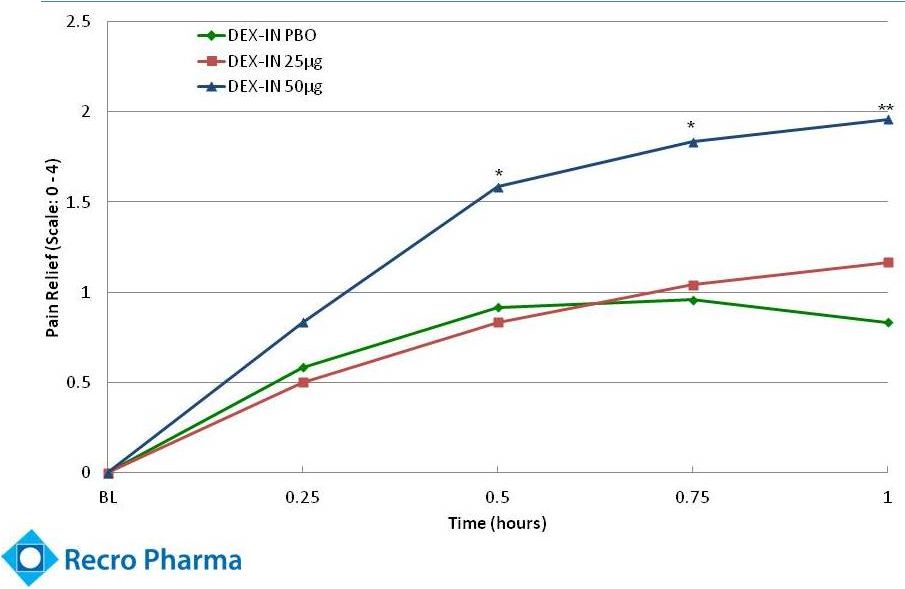
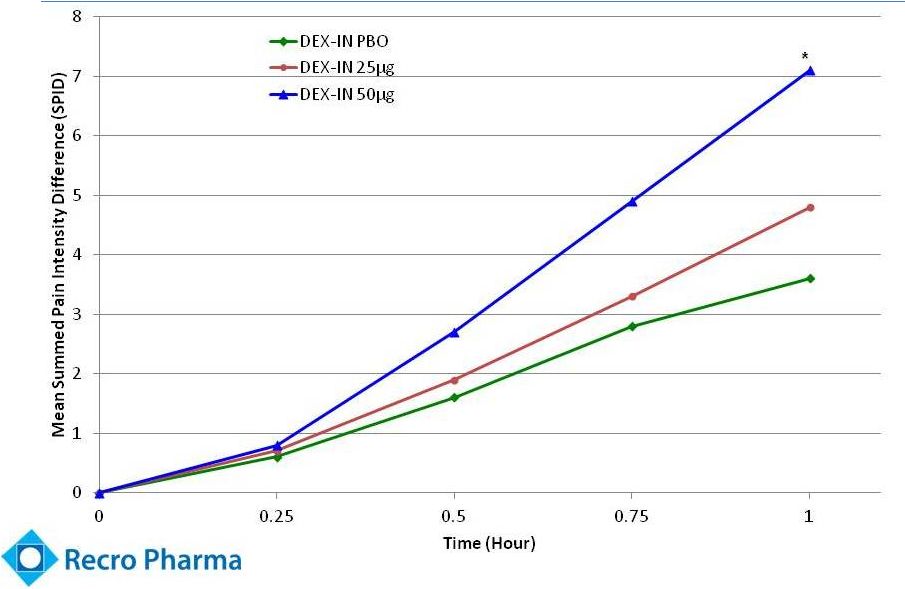
- 4 Sep 14 Recro Pharma Provides Clinical Strategy Update Following Interim Analysis for Phase IIb Clinical Trial of Dex-IN
Filing view
External links