
JULY 2016 Relieving pain…….Improving lives Exhibit 99.1

Special Note Regarding Forward-Looking Statements This presentation includes forward-looking statements within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. These statements, among other things, relate to our business strategy, goals and expectations concerning our product candidates, future operations, prospects, plans and objectives of management. The words "anticipate", "believe", "could", "estimate", "expect", "intend", "may", "plan", "predict", "project", "will" and similar terms and phrases are used to identify forward-looking statements in this presentation. Our operations involve risks and uncertainties, including the integration of our recently acquired assets, many of which are outside our control, and any one of which, or a combination of which, could materially affect our results of operations and whether the forward-looking statements ultimately prove to be correct. These forward-looking statements should be considered together with the risks and uncertainties that may affect our business and future results included in our filings with the Securities and Exchange Commission at www.sec.gov. These forward-looking statements are based on information currently available to us, and we assume no obligation to update any forward-looking statements except as required by applicable law.

Company Highlights Multiple non-opioid therapeutics in advanced clinical development for pain conditions Progressing Phase III clinical trials for IV Meloxicam Two pivotal efficacy trials: top-line results for one pivotal study expected in Q3 2016 and the other expected in Q4 2016 Enrolling safety study of 700 patients IV/IM Meloxicam – rapid onset, long acting preferential COX-2 inhibitor with demonstrated efficacy in multiple successful Phase II post operative pain trials Dex-IN – proprietary, intranasal therapeutic pursuing peri-procedural pain with further Phase II work Revenue and cash flow positive manufacturing & royalty business Experienced management team with significant development, regulatory and commercial experience

Experienced Management and Board Gerri Henwood – President and CEO Founded Auxilium Pharmaceuticals (AUXL, NASDAQ) and IBAH (former NASDAQ Co. – acquired 1998); GSK Michael Celano – CFO Over 35 years of financial leadership experience – Kensey Nash, BioRexis, Orasure, Arthur Andersen/KPMG Randy Mack – SVP, Development Over 20 years of clinical development experience – Adolor, Auxilium, Abbott Labs and Harris Labs Stewart McCallum, MD – CMO Over 9 years of GSK Clinical experience – Development experience; past clinical Investigator, KOL and Stanford U. Fred Graff – CCO Over 20 years of successful commercial experience, including sales and marketing leadership roles at Sepracor, RPR, and MAP Pharmaceuticals Board of Directors Wayne B. Weisman – Chairman SCP VitaLife Partners Alfred Altomari CEO, Agile Therapeutics William L. Ashton Harrison Consulting Group; frmly Amgen Michael Berelowitz, M.D. Former SVP, Specialty Care Business Unit, Pfizer Winston J. Churchill SCP VitaLife Partners Gerri Henwood – CEO Abraham Ludomirski, M.D. SCP VitaLife Partners Karen Flynn President-Pharmaceutical Packaging Systems, West Pharmaceutical Services, Inc.

2015 Transformative Transaction Acquired IV/IM Meloxicam and manufacturing & royalty business from Alkermes in April 2015 $50M up-front cash payment plus working capital adjustment; Meloxicam milestones and royalties Warrants issued to Alkermes and OrbiMed Non-dilutive up-front financed by loan from OrbiMed As of May 24, 2016, paid $19.0 million in aggregate, or 38% of the original $50.0 million of senior secured term loan with free cash flow generated by the manufacturing and royalty business IV/IM meloxicam – rapid onset, long acting preferential COX-2 inhibitor for moderate to severe acute pain currently in two Phase III trials Widely prescribed, approved oral chronic pain therapeutic Multiple Phase II studies successfully completed in acute pain models (outcomes in soft tissue and hard tissue models) Dosing advantages over existing acute pain therapeutics, including longer duration of action Manufacturing, royalty and formulation business 97,000 sq. ft. facility (DEA licensed) manufactures 5 commercial products marketed by partners $52M in revenue and cash flow positive for 9 months ended Dec 2015 and $18M in revenue and cash flow positive for Q1 2016.
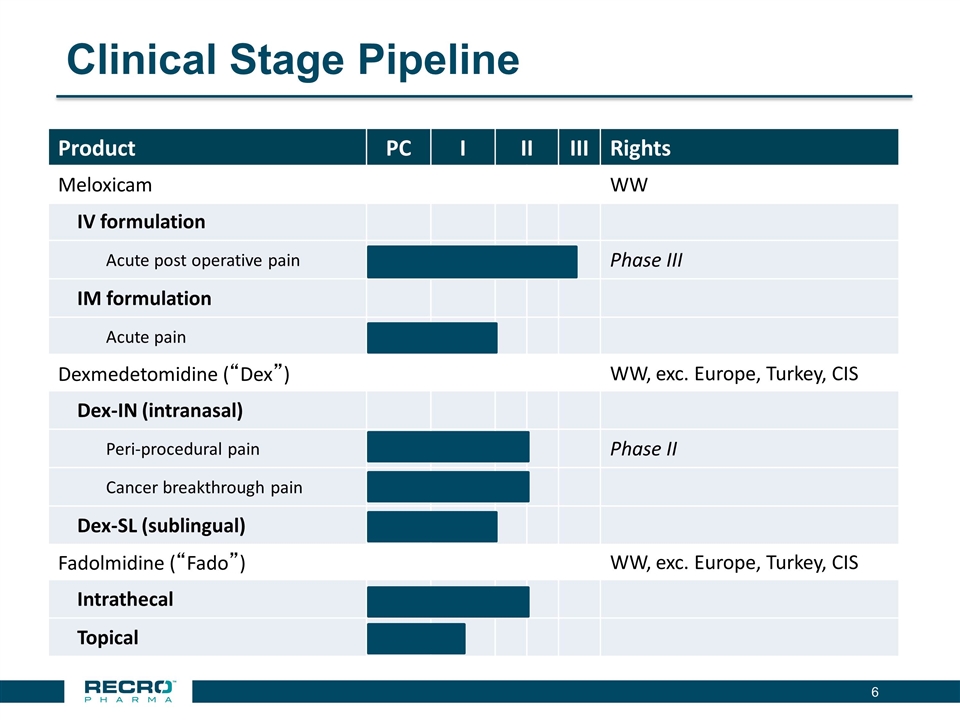
Clinical Stage Pipeline Product PC I II III Rights Meloxicam WW IV formulation Acute post operative pain Phase III IM formulation Acute pain Dexmedetomidine (“Dex”) WW, exc. Europe, Turkey, CIS Dex-IN (intranasal) Peri-procedural pain Phase II Cancer breakthrough pain Dex-SL (sublingual) Fadolmidine (“Fado”) WW, exc. Europe, Turkey, CIS Intrathecal Topical
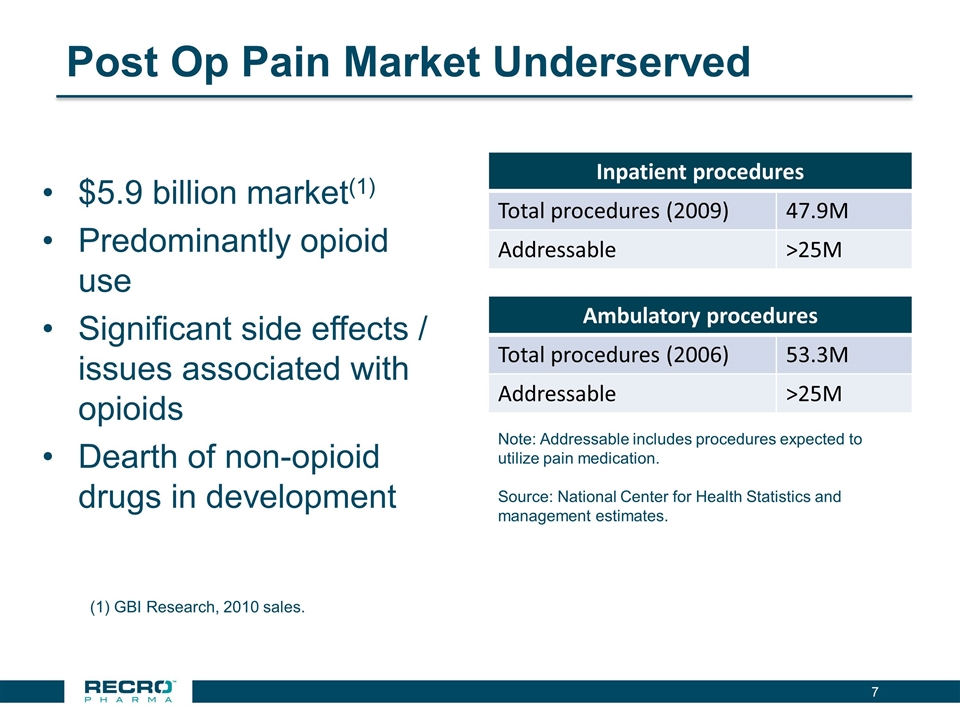
Post Op Pain Market Underserved $5.9 billion market(1) Predominantly opioid use Significant side effects / issues associated with opioids Dearth of non-opioid drugs in development Inpatient procedures Total procedures (2009) 47.9M Addressable >25M Ambulatory procedures Total procedures (2006) 53.3M Addressable >25M Note: Addressable includes procedures expected to utilize pain medication. Source: National Center for Health Statistics and management estimates. (1) GBI Research, 2010 sales.
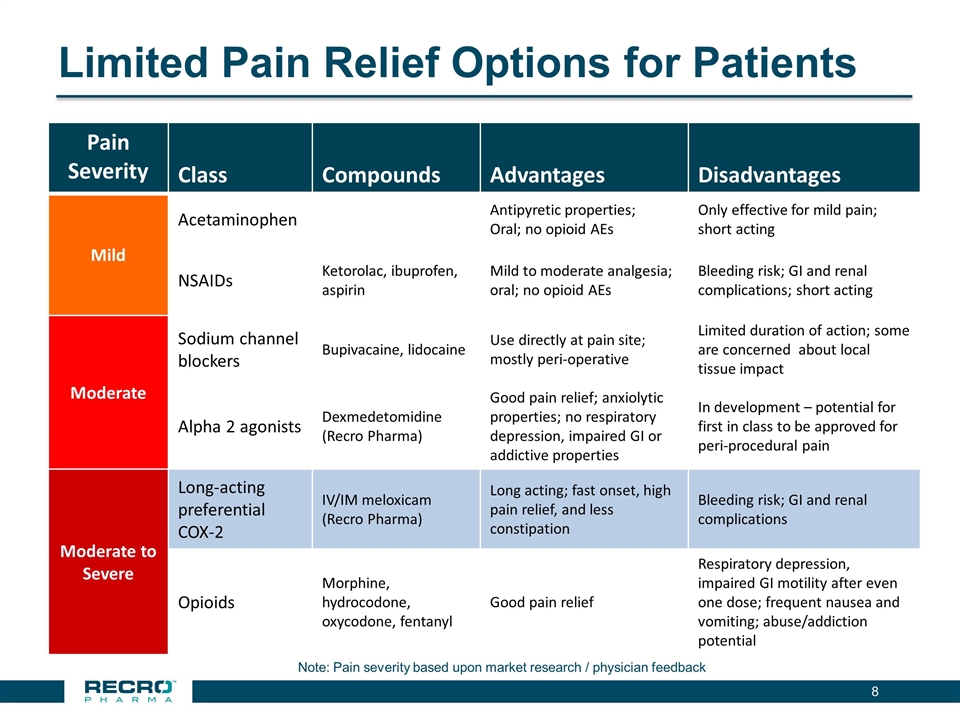
Limited Pain Relief Options for Patients Note: Pain severity based upon market research / physician feedback Pain Severity Class Compounds Advantages Disadvantages Mild Acetaminophen Antipyretic properties; Oral; no opioid AEs Only effective for mild pain; short acting NSAIDs Ketorolac, ibuprofen, aspirin Mild to moderate analgesia; oral; no opioid AEs Bleeding risk; GI and renal complications; short acting Moderate Sodium channel blockers Bupivacaine, lidocaine Use directly at pain site; mostly peri-operative Limited duration of action; some are concerned about local tissue impact Alpha 2 agonists Dexmedetomidine (Recro Pharma) Good pain relief; anxiolytic properties; no respiratory depression, impaired GI or addictive properties In development – potential for first in class to be approved for peri-procedural pain Moderate to Severe Long-acting preferential COX-2 IV/IM meloxicam (Recro Pharma) Long acting; fast onset, high pain relief, and less constipation Bleeding risk; GI and renal complications Opioids Morphine, hydrocodone, oxycodone, fentanyl Good pain relief Respiratory depression, impaired GI motility after even one dose; frequent nausea and vomiting; abuse/addiction potential

IV/IM Meloxicam

IV/IM Meloxicam Overview FDA approved, oral preferential COX-2 inhibitor used in a wide variety of indications Proprietary long acting injectable form for moderate to severe acute pain Incorporates Alkermes’ NanoCrystalTM technology IV/IM meloxicam – long acting preferential COX-2 inhibitor for moderate to severe acute pain currently in two Phase III trials Positive Phase II bunionectomy, hysterectomy and dental pain studies with demonstrated efficacy Prior to start of Phase III, had a successful end of Phase II meeting with the FDA Formulation IP issued through 2022 and additional methods of preparation IP issued through May 2030 NanoCrystal® is a registered trademark of Alkermes plc.
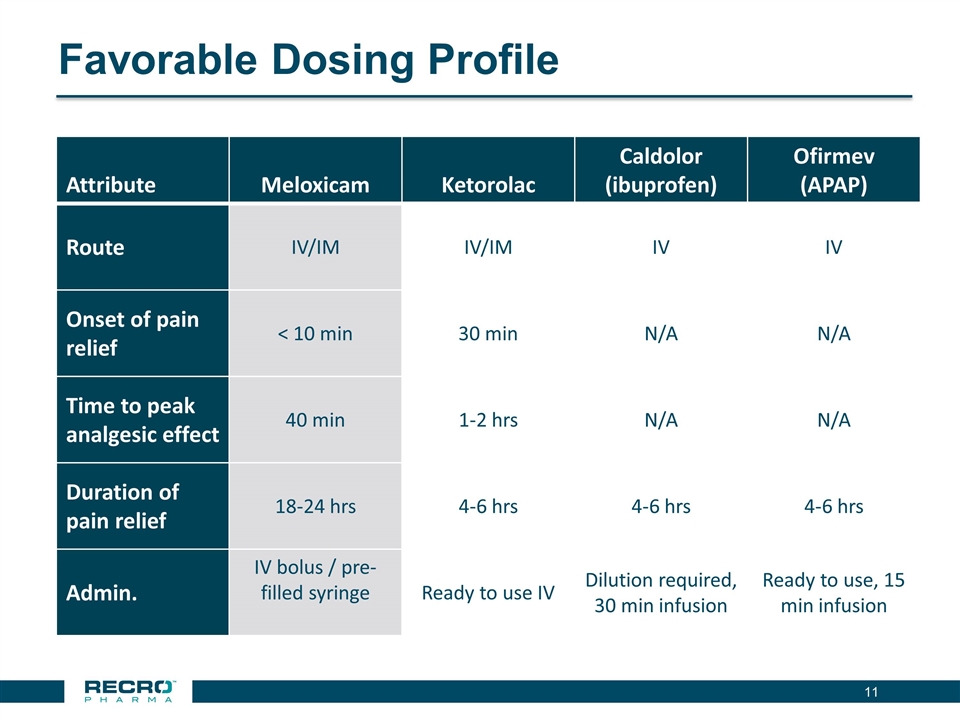
Favorable Dosing Profile Attribute Meloxicam Ketorolac Caldolor (ibuprofen) Ofirmev (APAP) Route IV/IM IV/IM IV IV Onset of pain relief < 10 min 30 min N/A N/A Time to peak analgesic effect 40 min 1-2 hrs N/A N/A Duration of pain relief 18-24 hrs 4-6 hrs 4-6 hrs 4-6 hrs Admin. IV bolus / pre-filled syringe Ready to use IV Dilution required, 30 min infusion Ready to use, 15 min infusion

Phase III Acute Postoperative Pain Studies Progressing with two Phase III trials in acute postoperative pain following abdominoplasty surgery and bunionectomy surgery Placebo vs IV Meloxicam (30mg) every 24 hours Standard analgesia study design Pain Intensity assessments (SPID24 = Primary Endpoint Soft Tissue Study; SPID48 = Primary Endpoint Hard Tissue Study) Rescue medication Time to onset Currently enrolling Phase III 700 patient safety study
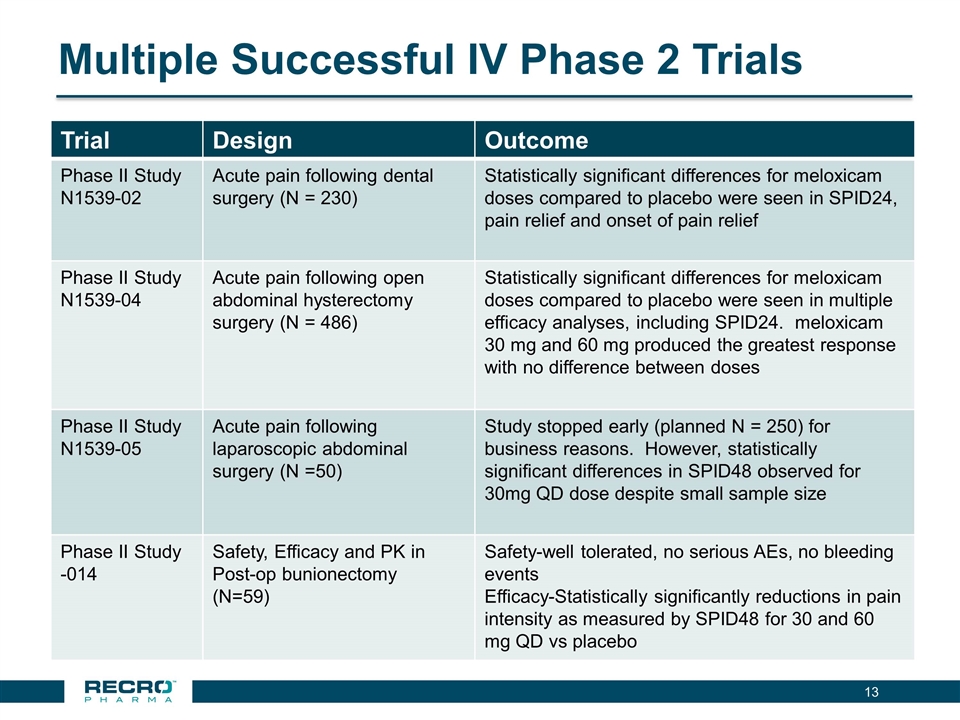
Multiple Successful IV Phase 2 Trials Trial Design Outcome Phase II Study N1539-02 Acute pain following dental surgery (N = 230) Statistically significant differences for meloxicam doses compared to placebo were seen in SPID24, pain relief and onset of pain relief Phase II Study N1539-04 Acute pain following open abdominal hysterectomy surgery (N = 486) Statistically significant differences for meloxicam doses compared to placebo were seen in multiple efficacy analyses, including SPID24. meloxicam 30 mg and 60 mg produced the greatest response with no difference between doses Phase II Study N1539-05 Acute pain following laparoscopic abdominal surgery (N =50) Study stopped early (planned N = 250) for business reasons. However, statistically significant differences in SPID48 observed for 30mg QD dose despite small sample size Phase II Study -014 Safety, Efficacy and PK in Post-op bunionectomy (N=59) Safety-well tolerated, no serious AEs, no bleeding events Efficacy-Statistically significantly reductions in pain intensity as measured by SPID48 for 30 and 60 mg QD vs placebo
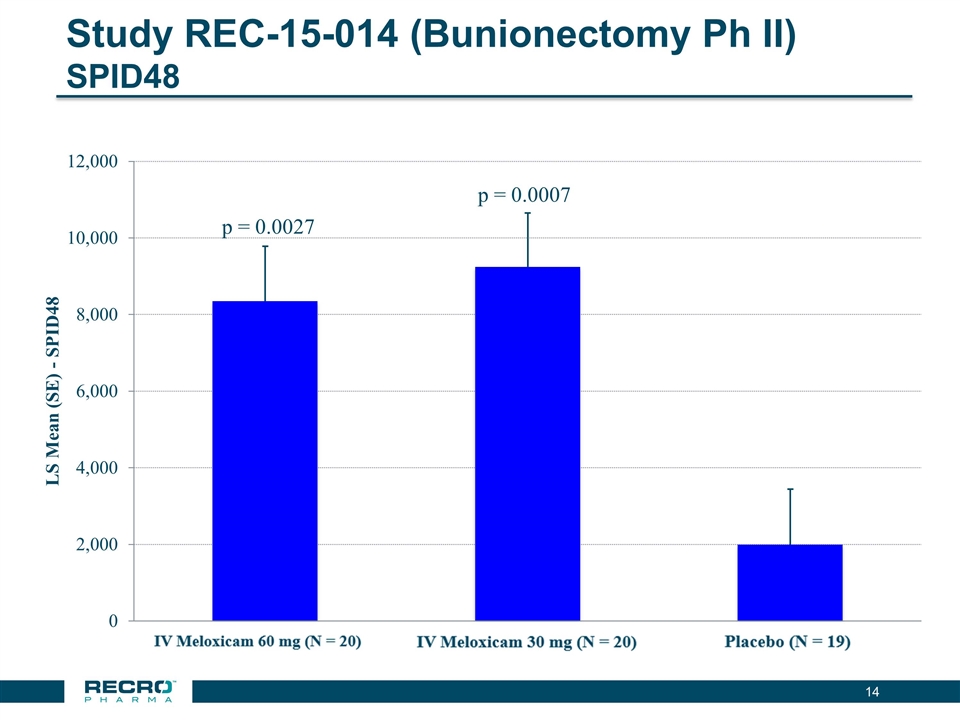
Study REC-15-014 (Bunionectomy Ph II) SPID48 p = 0.0007 p = 0.0027 14
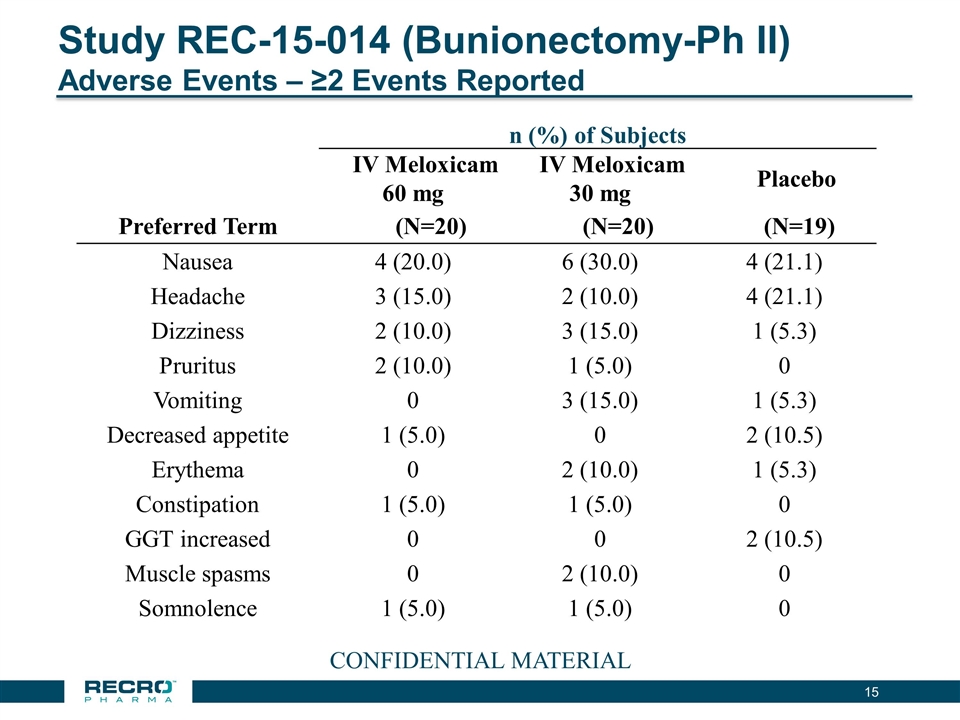
Study REC-15-014 (Bunionectomy-Ph II) Adverse Events – ≥2 Events Reported 15 n (%) of Subjects IV Meloxicam 60 mg IV Meloxicam 30 mg Placebo Preferred Term (N=20) (N=20) (N=19) Nausea 4 (20.0) 6 (30.0) 4 (21.1) Headache 3 (15.0) 2 (10.0) 4 (21.1) Dizziness 2 (10.0) 3 (15.0) 1 (5.3) Pruritus 2 (10.0) 1 (5.0) 0 Vomiting 0 3 (15.0) 1 (5.3) Decreased appetite 1 (5.0) 0 2 (10.5) Erythema 0 2 (10.0) 1 (5.3) Constipation 1 (5.0) 1 (5.0) 0 GGT increased 0 0 2 (10.5) Muscle spasms 0 2 (10.0) 0 Somnolence 1 (5.0) 1 (5.0) 0 CONFIDENTIAL MATERIAL

Phase II Abdominal Hysterectomy Study Multicenter, single-dose, randomized, double-blind, placebo- & active-controlled study in Eastern Europe In double-blind period, single doses of: All patients received placebo, IV Morphine (10-15 mg), Meloxicam 5 mg, 7.5 mg, 15 mg, 30 mg, 60 mg After 24 hours, open-label Meloxicam was available Standard analgesia study design Pain Intensity assessments (SPID24 = Primary Endpoint) Pain Relief Rescue mediation Time to onset
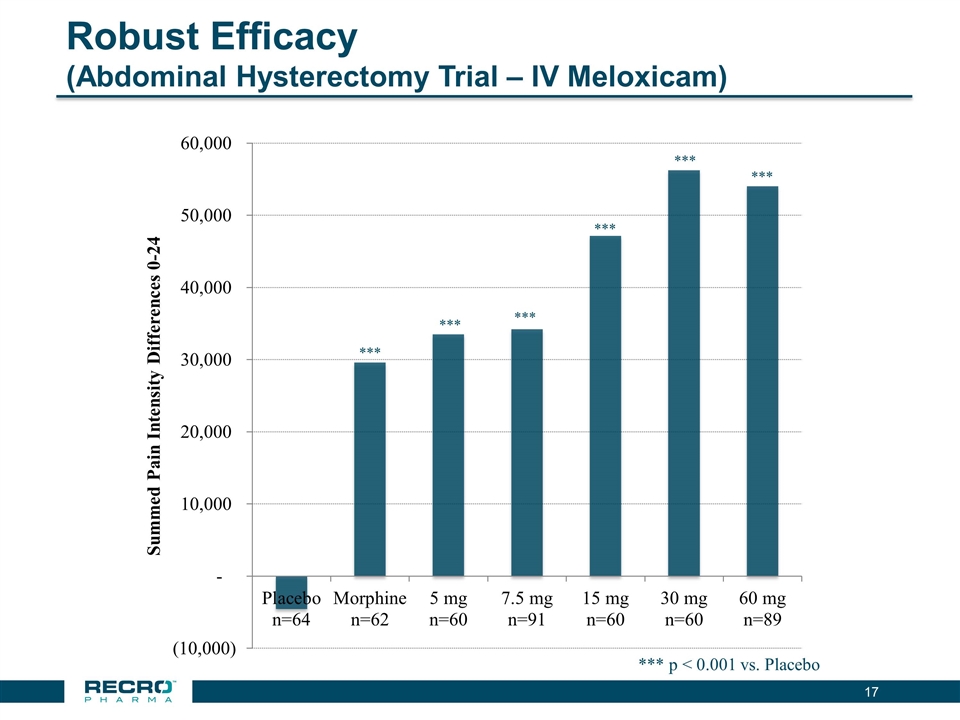
Robust Efficacy (Abdominal Hysterectomy Trial – IV Meloxicam) *** p < 0.001 vs. Placebo *** *** *** *** *** ***
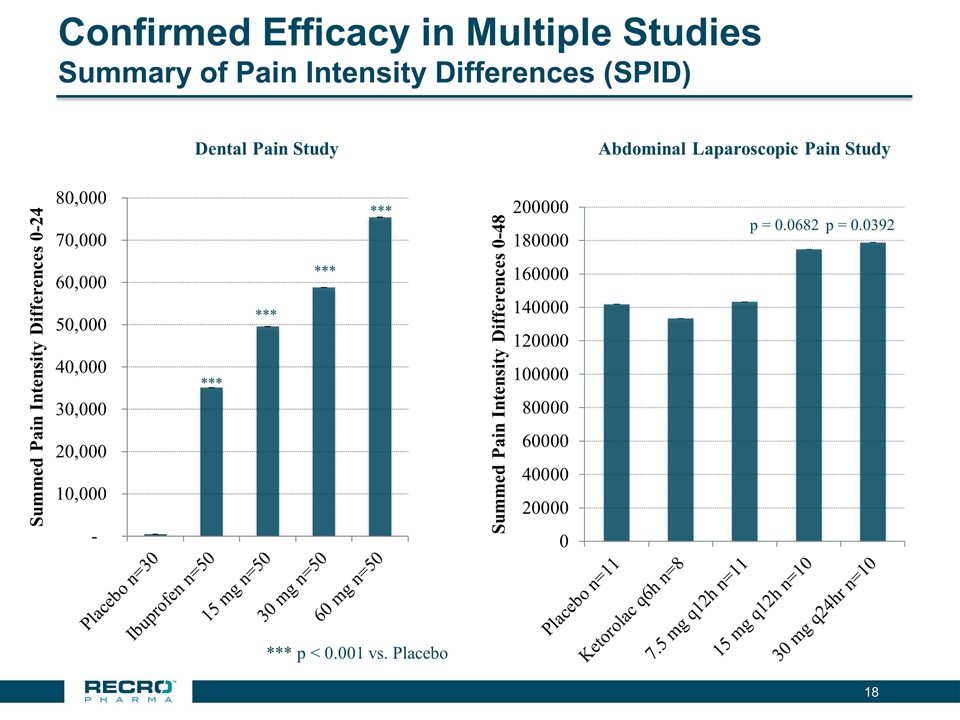
Confirmed Efficacy in Multiple Studies Summary of Pain Intensity Differences (SPID) *** p < 0.001 vs. Placebo *** *** *** *** Dental Pain Study p = 0.0682 p = 0.0392 Abdominal Laparoscopic Pain Study
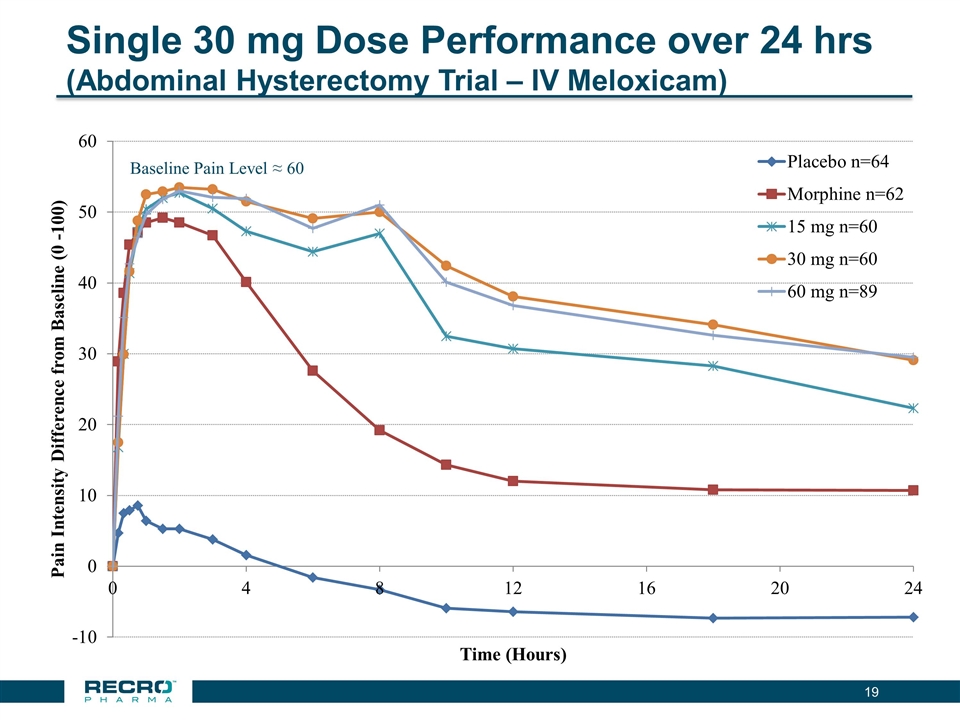
Single 30 mg Dose Performance over 24 hrs (Abdominal Hysterectomy Trial – IV Meloxicam) Baseline Pain Level ≈ 60
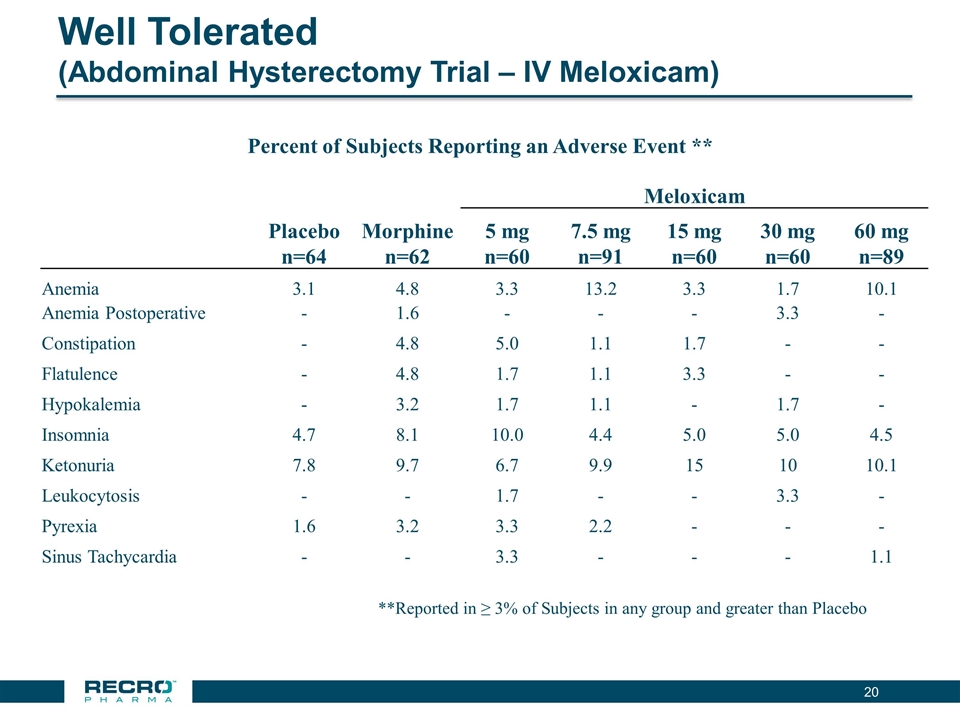
Well Tolerated (Abdominal Hysterectomy Trial – IV Meloxicam) **Reported in ≥ 3% of Subjects in any group and greater than Placebo Meloxicam Placebo n=64 Morphine n=62 5 mg n=60 7.5 mg n=91 15 mg n=60 30 mg n=60 60 mg n=89 Anemia 3.1 4.8 3.3 13.2 3.3 1.7 10.1 Anemia Postoperative - 1.6 - - - 3.3 - Constipation - 4.8 5.0 1.1 1.7 - - Flatulence - 4.8 1.7 1.1 3.3 - - Hypokalemia - 3.2 1.7 1.1 - 1.7 - Insomnia 4.7 8.1 10.0 4.4 5.0 5.0 4.5 Ketonuria 7.8 9.7 6.7 9.9 15 10 10.1 Leukocytosis - - 1.7 - - 3.3 - Pyrexia 1.6 3.2 3.3 2.2 - - - Sinus Tachycardia - - 3.3 - - - 1.1 Percent of Subjects Reporting an Adverse Event **

Next Steps for IV Meloxicam Complete Phase III Pivotal Study in hard and soft tissue models Top-line results for one pivotal study expected in Q3 2016; the other expected in Q4 2016 Complete safety studies to meet adequate exposures / special populations Currently enrolling Total across above studies: est. 1,100 patients expected to be enrolled

Dexmedetomidine (“Dex”)

Dex Has Demonstrated Analgesia & Safety Alpha 2 agonist (non-opioid) Injectable form (Precedex) marketed by Hospira in US as sedative Multiple studies demonstrating analgesia of alpha 2 agonists Intranasal formulation in clinical development for peri-procedural pain In-licensed non-IV rights from Orion Worldwide rights except Europe, Turkey, and CIS Multiple studies demonstrate Dex pain relief and safety profile Including our completed placebo controlled trials Expect strong IP position Pending IP coverage could run through 2030 Expect to file 505(b)(2) NDA after completion of Ph III

Positive Dex-IN Ph II Results (REC-14-013 – Post Op Day 1 Dosing) Randomized, placebo controlled Phase II bunionectomy study (168 patients) Randomized, placebo controlled study 50 mcg of Dex-IN or placebo every 6 hours Primary endpoint – SPID48 (p=0.0214) Oral opioid rescue therapy allowed 6 patients discontinued for lack of efficacy (3 in each treatment group) and 1 patient due to serious adverse event of hypotension Most common adverse events observed in the study were: blood pressure decrease / hypotension nausea (similar incidences to placebo) nasal discomfort and headache Adverse event of bradycardia was reported in 3 subjects in the Dex-IN treatment group
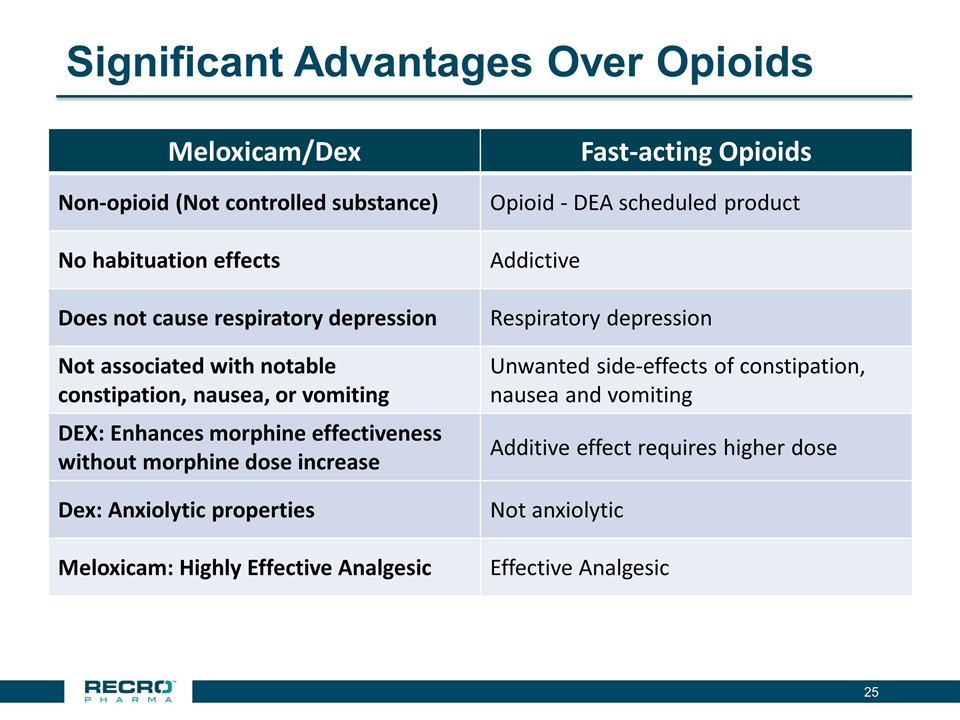
Significant Advantages Over Opioids Meloxicam/Dex Fast-acting Opioids Non-opioid (Not controlled substance) Opioid - DEA scheduled product No habituation effects Addictive Does not cause respiratory depression Respiratory depression Not associated with notable constipation, nausea, or vomiting Unwanted side-effects of constipation, nausea and vomiting DEX: Enhances morphine effectiveness without morphine dose increase Additive effect requires higher dose Dex: Anxiolytic properties Not anxiolytic Meloxicam: Highly Effective Analgesic Effective Analgesic
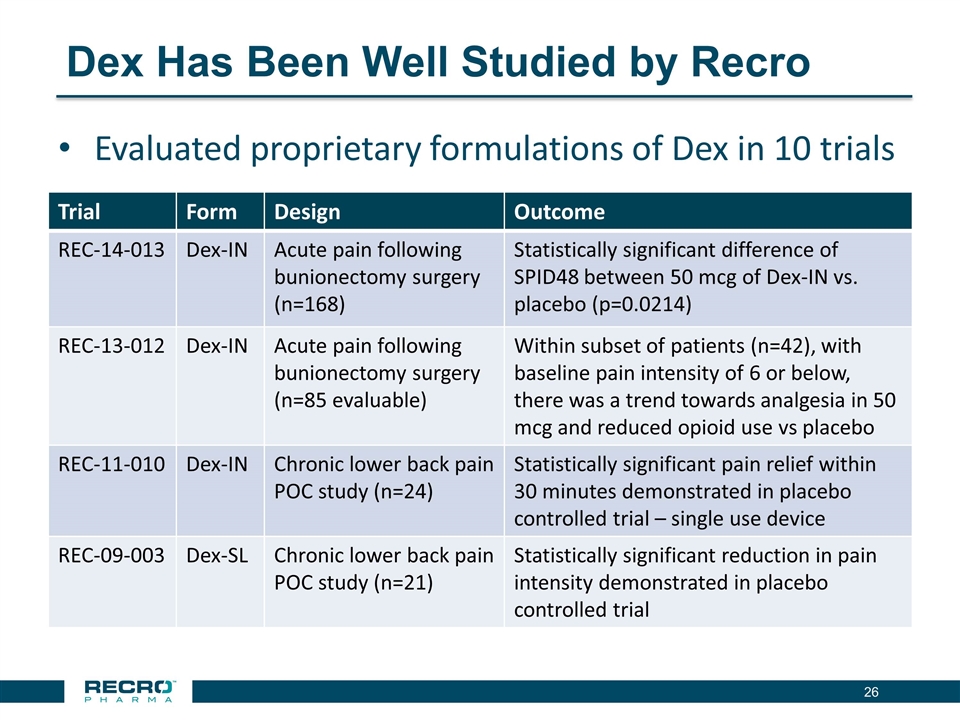
Dex Has Been Well Studied by Recro Evaluated proprietary formulations of Dex in 10 trials Trial Form Design Outcome REC-14-013 Dex-IN Acute pain following bunionectomy surgery (n=168) Statistically significant difference of SPID48 between 50 mcg of Dex-IN vs. placebo (p=0.0214) REC-13-012 Dex-IN Acute pain following bunionectomy surgery (n=85 evaluable) Within subset of patients (n=42), with baseline pain intensity of 6 or below, there was a trend towards analgesia in 50 mcg and reduced opioid use vs placebo REC-11-010 Dex-IN Chronic lower back pain POC study (n=24) Statistically significant pain relief within 30 minutes demonstrated in placebo controlled trial – single use device REC-09-003 Dex-SL Chronic lower back pain POC study (n=21) Statistically significant reduction in pain intensity demonstrated in placebo controlled trial
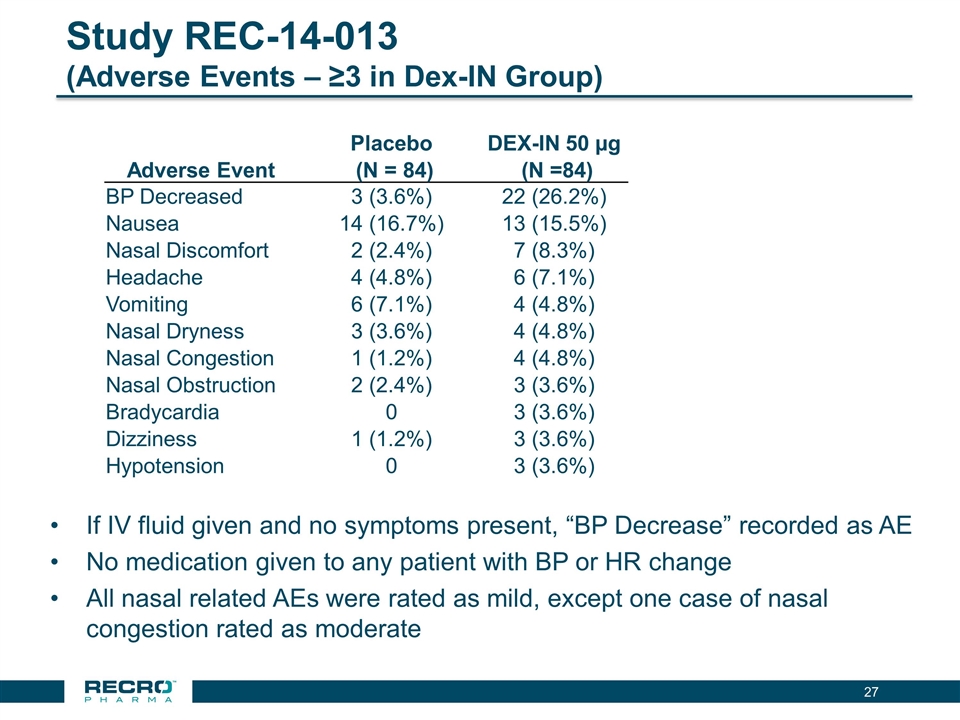
Study REC-14-013 (Adverse Events – ≥3 in Dex-IN Group) Placebo DEX-IN 50 µg Adverse Event (N = 84) (N =84) BP Decreased 3 (3.6%) 22 (26.2%) Nausea 14 (16.7%) 13 (15.5%) Nasal Discomfort 2 (2.4%) 7 (8.3%) Headache 4 (4.8%) 6 (7.1%) Vomiting 6 (7.1%) 4 (4.8%) Nasal Dryness 3 (3.6%) 4 (4.8%) Nasal Congestion 1 (1.2%) 4 (4.8%) Nasal Obstruction 2 (2.4%) 3 (3.6%) Bradycardia 0 3 (3.6%) Dizziness 1 (1.2%) 3 (3.6%) Hypotension 0 3 (3.6%) If IV fluid given and no symptoms present, “BP Decrease” recorded as AE No medication given to any patient with BP or HR change All nasal related AEs were rated as mild, except one case of nasal congestion rated as moderate

Fadolmidine (“Fado”)

Fado Effective in Phase II for Pain Relief Alpha 2 agonist more potent at the alpha 2c receptor than Dex >20 fold less potent at the alpha 1b receptor than clonidine Fado has demonstrated analgesia in multiple animal models Positive Phase II analgesia study in bunionectomy patients Intrathecal route of administration WW rights to all human uses except Europe, Turkey and CIS NCE patent w/ expected extension to 2021

Clinical Pipeline Intellectual Property IV/IM meloxicam – formulation IP through 2022 and additional methods of preparation IP through May 2030 Dex applications for methods for treating/preventing pain through intranasal and sublingual formulations without significant sedation Fado IP in-licensed from Orion Composition of matter Method of administration for analgesia Treatment and prevention of hypotension and shock Pro-Drug Regulatory exclusivity 505(b)(2) – 3 years (Meloxicam, Dex-IN, Dex-SL) 505(b)(1) – NCE, 5 years (Fado)

Gainesville Overview

US Based Manufacturing Facility

Manufacturing & Royalty Overview Manufacturing facility 97,000 + sq. ft. solid oral dosage manufacturing cGMP DEA licensed ~170 employees Service capabilities Formulation, process development and optimization Process scale-up Clinical supply and validation Commercial supply Ritalin LA Once daily ADHD treatment marketed by Novartis Focalin XR ADHD treatment marketed by Novartis Verelan / verapamil CV/High blood pressure treatment marketed by Actavis and Lannett Zohydro ER Extended release hydrocodone marketed by Pernix Launched in 2014 Abuse deterrent form launched

Strong Historical Manufacturing Performance Zohydro ER – abuse deterrent form launched Additional capacity for new product opportunities Positive cash flow expected to cover debt service obligations and excess cash flows to repay loan principal as per terms of credit agreement. *EBITDA is a non-GAAP financial metric. (millions) 9 months ended Dec. 31, 2015 (Pre-Acquisition) (audited) 12 months ended Mar. 31, 2016 (unaudited) Revenues $52.0 $69.7 EBITDA* $24.0 $31.7

Company Highlights Multiple non-opioid therapeutics in advanced clinical development for pain conditions Progressing Phase III clinical trials for IV Meloxicam Two pivotal efficacy trials: top-line results for one pivotal study expected in Q3 2016 and the other expected in Q4 2016 Enrolling safety study of 700 patients IV/IM Meloxicam – long acting preferential COX-2 inhibitor with demonstrated efficacy in multiple successful Phase II post operative pain trials Dex-IN – proprietary, intranasal therapeutic pursuing peri-procedural pain with further Phase II work Revenue and cash flow positive manufacturing & royalty business Experienced management team with significant development, regulatory and commercial experience

OrbiMed Debt Financing $50 million up-front payment funded via a five-year senior secured term loan with OrbiMed Interest at LIBOR + 14.0%, with a 1.0% LIBOR floor OrbiMed received warrants to purchase an aggregate of 3% of Recro's outstanding common stock (on a fully diluted basis) as of the closing of the transaction. As of May 24, 2016, paid $19.0 million in aggregate, or 38% of the original $50.0 million from free cash flow generated by the manufacturing and royalty business
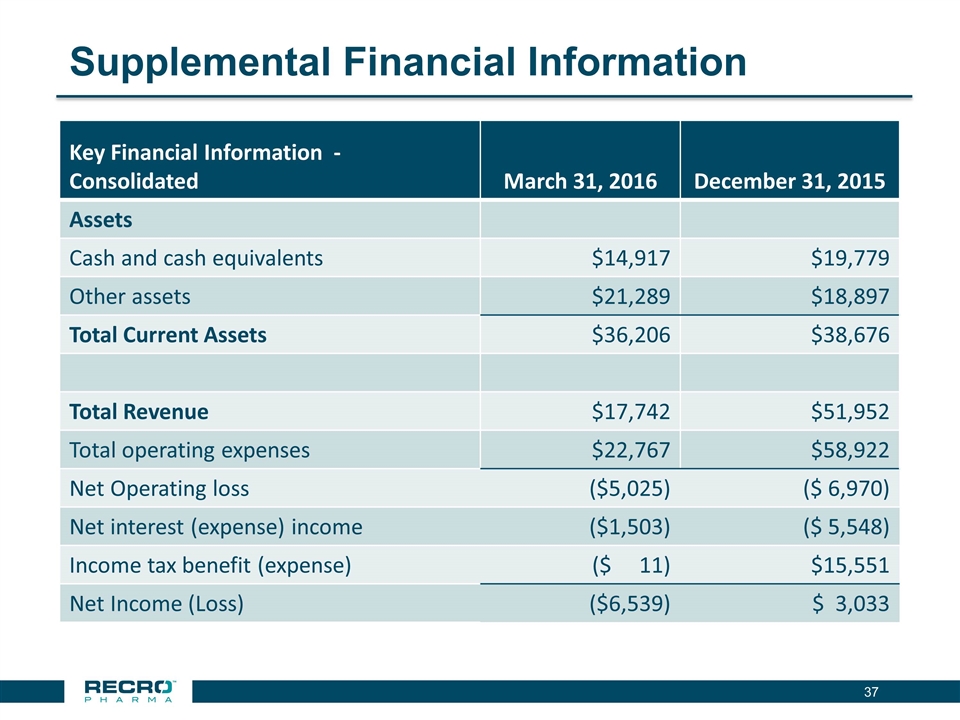
Supplemental Financial Information Key Financial Information - Consolidated March 31, 2016 December 31, 2015 Assets Cash and cash equivalents $14,917 $19,779 Other assets $21,289 $18,897 Total Current Assets $36,206 $38,676 Total Revenue $17,742 $51,952 Total operating expenses $22,767 $58,922 Net Operating loss ($5,025) ($ 6,970) Net interest (expense) income ($1,503) ($ 5,548) Income tax benefit (expense) ($ 11) $15,551 Net Income (Loss) ($6,539) $ 3,033