
March 2018 Relieving pain…….Improving lives Exhibit 99.1

Forward Looking Statements This presentation includes forward-looking statements within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. These statements, among other things, relate to our development, launch and commercialization strategy for IV meloxicam, goals and expectations concerning IV meloxicam, future operations, prospects, plans and objectives of management. The words "anticipate", "believe", "could", "estimate", "expect", "intend", "may", "plan", "predict", "project", "will" and similar terms and phrases may be used to identify forward-looking statements in this presentation. Our operations involve risks and uncertainties, many of which are outside our control, and any one of which, or a combination of which, could materially affect our results of operations and whether the forward-looking statements ultimately prove to be correct. These forward-looking statements should be considered together with the risks and uncertainties that may affect our business and future results included in our filings with the Securities and Exchange Commission at www.sec.gov. These forward-looking statements are based on information currently available to us, and we assume no obligation to update any forward-looking statements except as required by applicable law. Non-Promotion: This presentation is intended to be non-promotional and for investor discussion purposes only. The information provided herein contains references to IV meloxicam, an investigational product. Use of IV meloxicam has not been approved by the FDA. The safety and efficacy of the investigational use of IV meloxicam has not been determined. There is no guarantee that IV meloxicam will be approved for marketing by any regulatory agency. IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA.

Company Highlights Specialty pharmaceutical company focused on hospital and related settings with late stage investigational product, IV Meloxicam, targeting management of moderate to severe pain Filed New Drug Application for IV Meloxicam in July 2017; PDUFA Action Date- May 26, 2018 Commercial teams and go to market strategy prepared Multiple therapeutics in clinical development for hospital and related settings Revenue and cash flow positive contract development and manufacturing (CDMO) business 2017 Revenue- $71.8M; 2017 EBITDA- $32.8M Cash position- $64.5M @ 12/31/17 $100 million credit facility secured in November 2017 Experienced management team with significant development, regulatory and commercial experience *EBITDA is a non-GAAP financial measure. See reconciliation on last page of presentation. IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA.
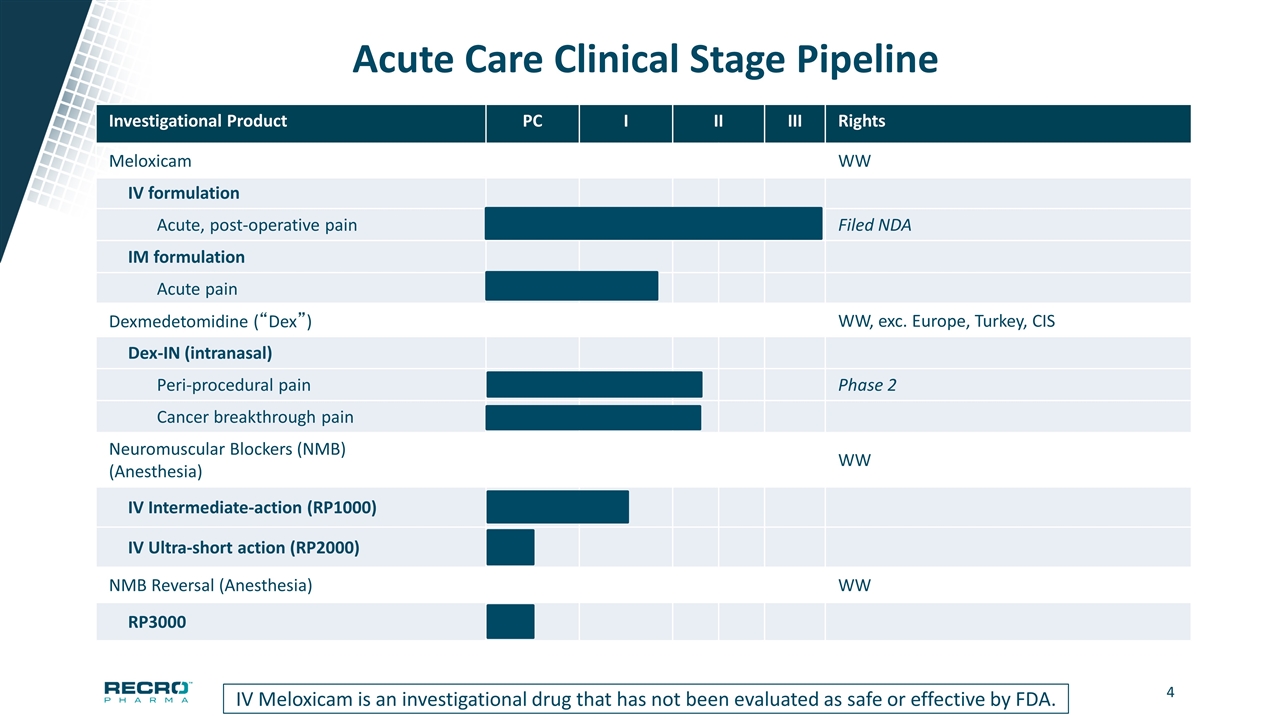
Acute Care Clinical Stage Pipeline Investigational Product PC I II III Rights Meloxicam WW IV formulation Acute, post-operative pain Filed NDA IM formulation Acute pain Dexmedetomidine (“Dex”) WW, exc. Europe, Turkey, CIS Dex-IN (intranasal) Peri-procedural pain Phase 2 Cancer breakthrough pain Neuromuscular Blockers (NMB) (Anesthesia) WW IV Intermediate-action (RP1000) IV Ultra-short action (RP2000) IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA. NMB Reversal (Anesthesia) WW RP3000

IV Meloxicam Overview FDA approved as an oral, preferential COX-2 inhibitor and used in a number of indications Proprietary non-opioid, once daily IV form Incorporates Alkermes’ NanoCrystal® technology IV Meloxicam – once daily, preferential COX-2 inhibitor for moderate to severe acute pain Clinical and safety data package complete NDA submitted to FDA, July 2017 NDA accepted for review; PDUFA action date 5/26/18 Pre-Approval Inspections have been scheduled for Q1 of 2018 Formulation IP issued through 2022 and additional methods of preparation IP issued through May 2030 IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA. NanoCrystal® is a registered trademark of APIL
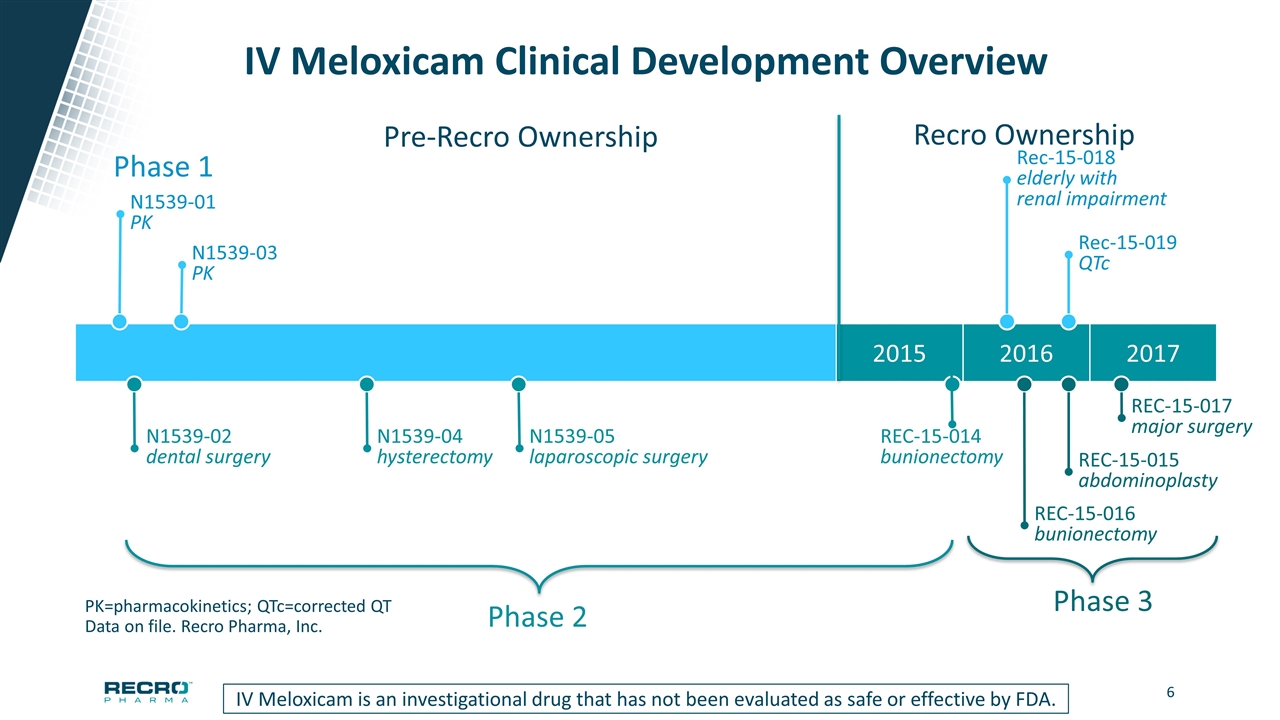
2015 2016 2017 IV Meloxicam Clinical Development Overview PK=pharmacokinetics; QTc=corrected QT Data on file. Recro Pharma, Inc. Phase 1 N1539-01 PK N1539-03 PK Rec-15-018 elderly with renal impairment Rec-15-019 QTc Phase 2 N1539-02 dental surgery N1539-04 hysterectomy N1539-05 laparoscopic surgery REC-15-014 bunionectomy Phase 3 REC-15-015 abdominoplasty REC-15-016 bunionectomy REC-15-017 major surgery Pre-Recro Ownership Recro Ownership IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA.
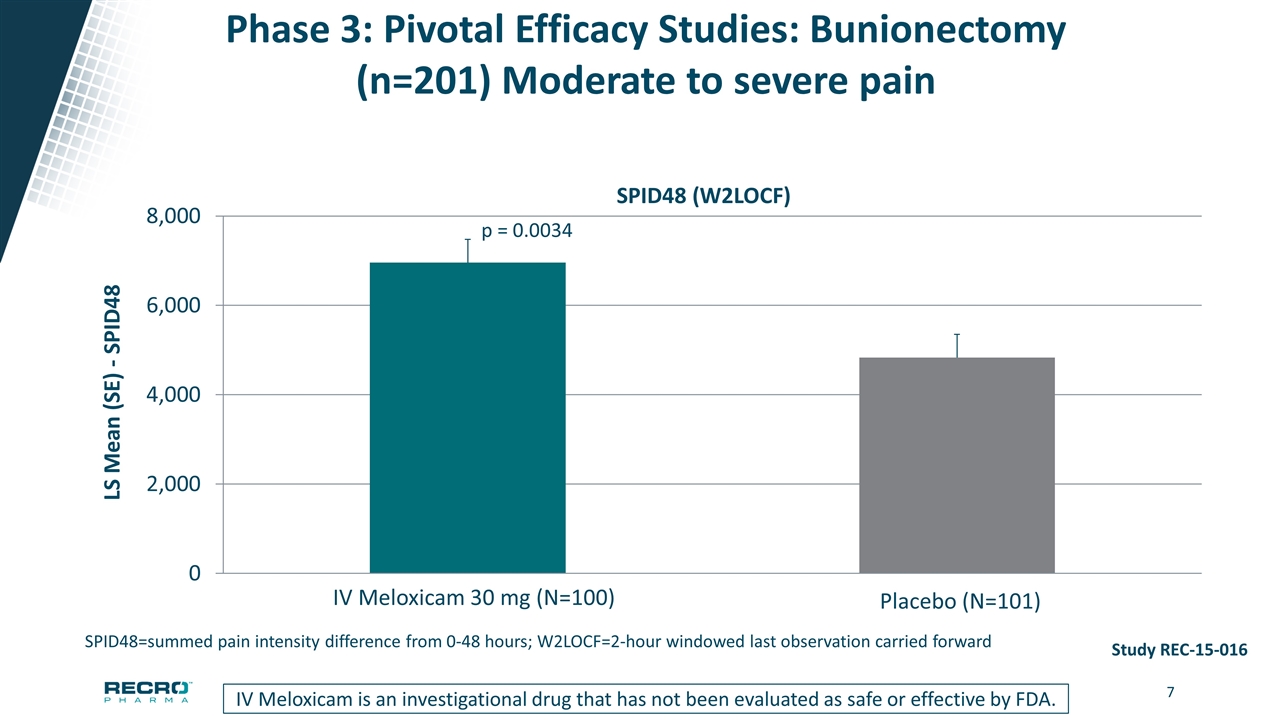
Phase 3: Pivotal Efficacy Studies: Bunionectomy (n=201) Moderate to severe pain p = 0.0034 Study REC-15-016 Meloxicam IV is an investigational drug that has not been evaluated as safe or effective by FDA SPID48 (W2LOCF) SPID48=summed pain intensity difference from 0-48 hours; W2LOCF=2-hour windowed last observation carried forward IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA.
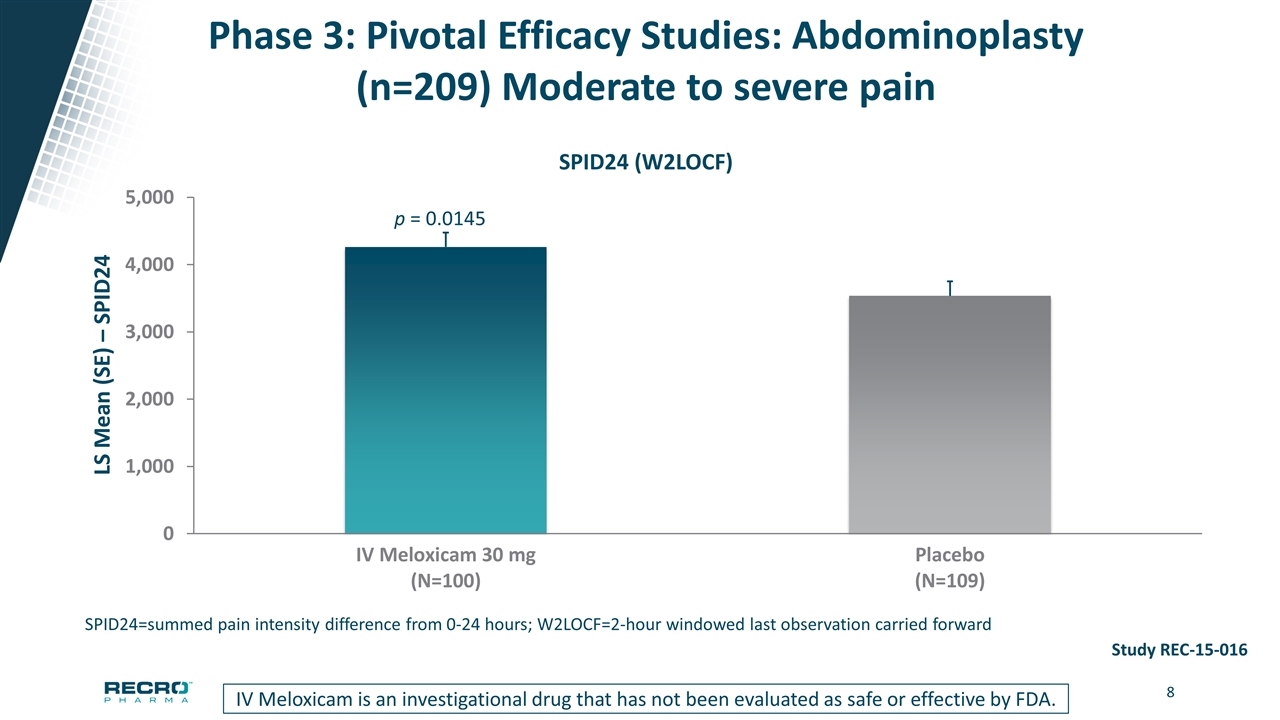
Phase 3: Pivotal Efficacy Studies: Abdominoplasty (n=209) Moderate to severe pain SPID24=summed pain intensity difference from 0-24 hours; W2LOCF=2-hour windowed last observation carried forward p = 0.0145 IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA. Study REC-15-016

Phase 3 Safety Study in Major Surgery IV Meloxicam or Placebo + Standard of Care Design 722 subjects undergoing major surgery with inpatient hospitalization for at least 24-48 hours Randomized, double-blind, placebo-controlled trial; IV meloxicam 30mg Evaluating the safety of IV Meloxicam in the setting of major surgery 31 centers in 4 countries (US, CAN, AUS, NZ) Study drug administered as a bolus Added on top of existing pain management protocols after surgery Other NSAIDS prohibited; no minimum pain score required for randomization Once daily dosing for up to 7 doses Additional dose at time of discharge at the discretion of subject and primary investigator IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA. Study REC-15-016
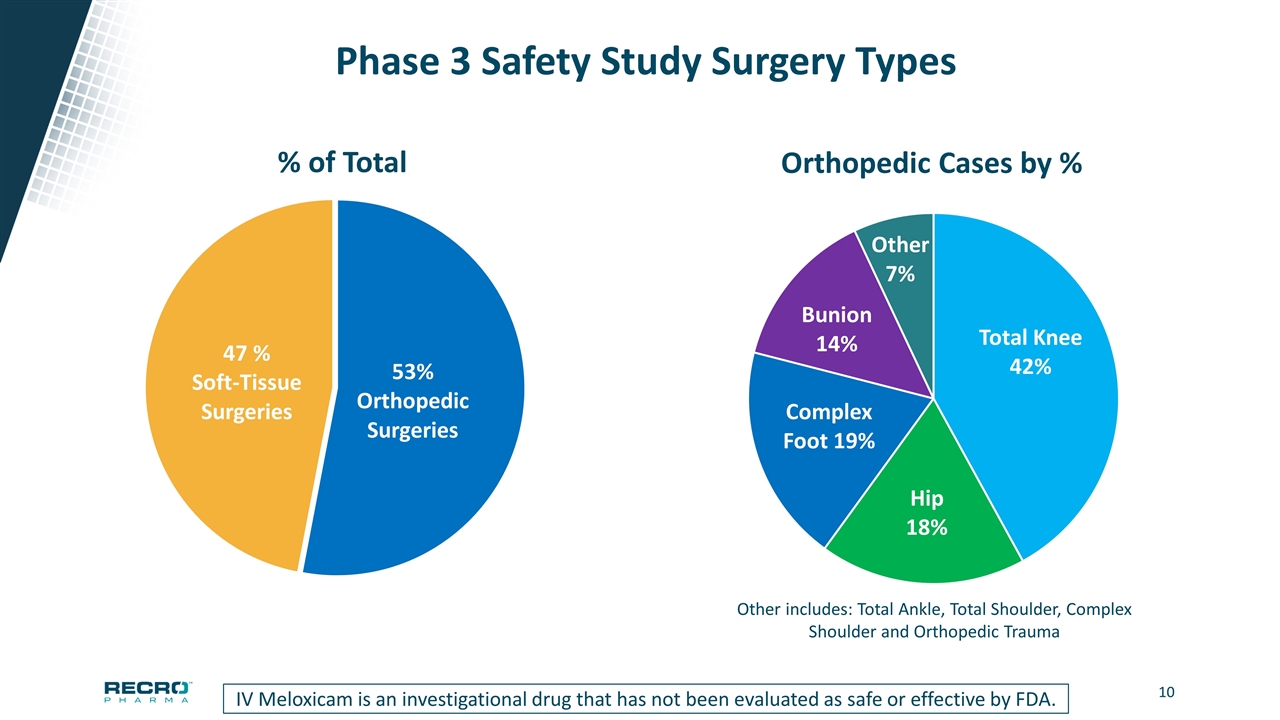
Phase 3 Safety Study Surgery Types Other includes: Total Ankle, Total Shoulder, Complex Shoulder and Orthopedic Trauma IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA.
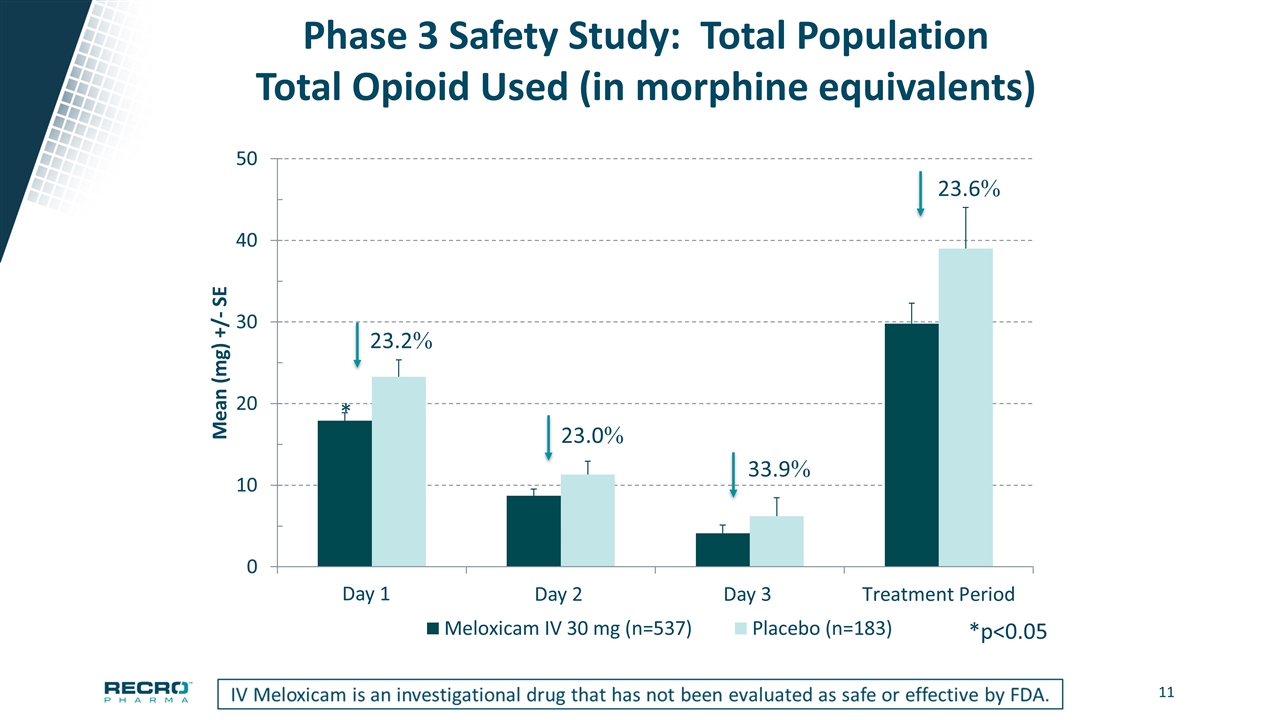
Phase 3 Safety Study: Total Population Total Opioid Used (in morphine equivalents) * *p<0.05 Day 1
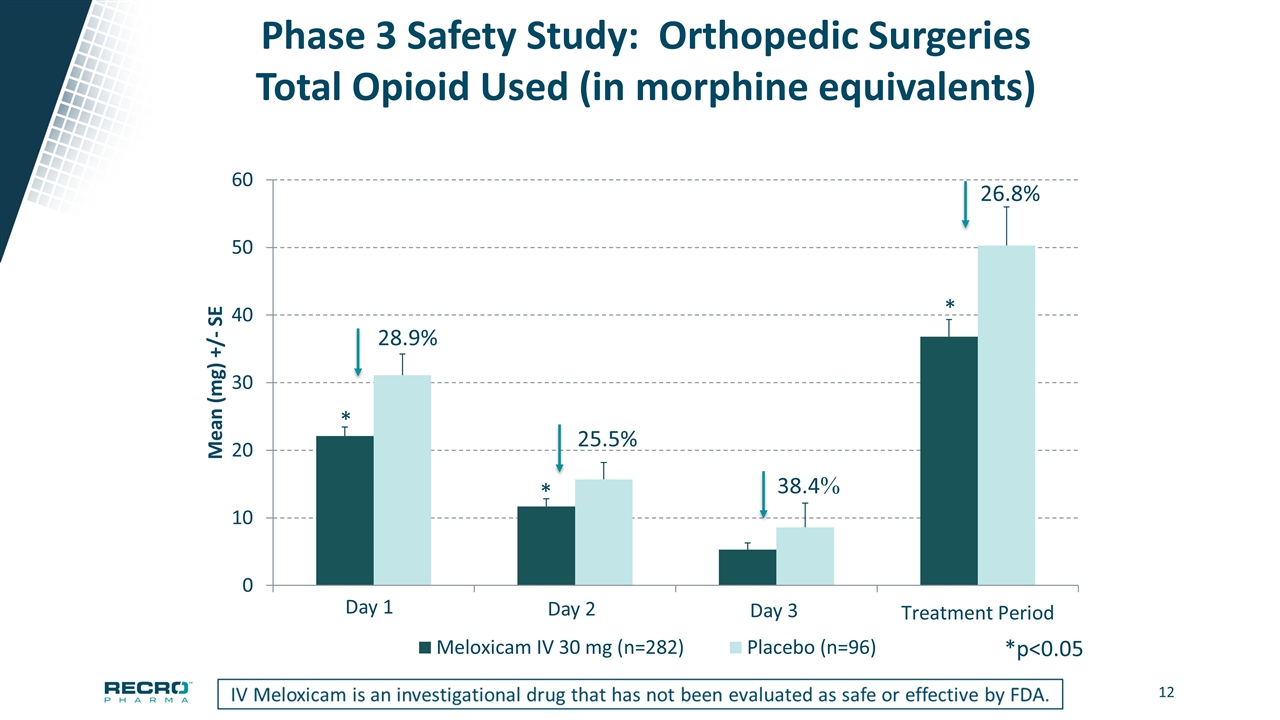
Phase 3 Safety Study: Orthopedic Surgeries Total Opioid Used (in morphine equivalents) 28.9% 25.5% *p<0.05 * * * Day 2 Day 3
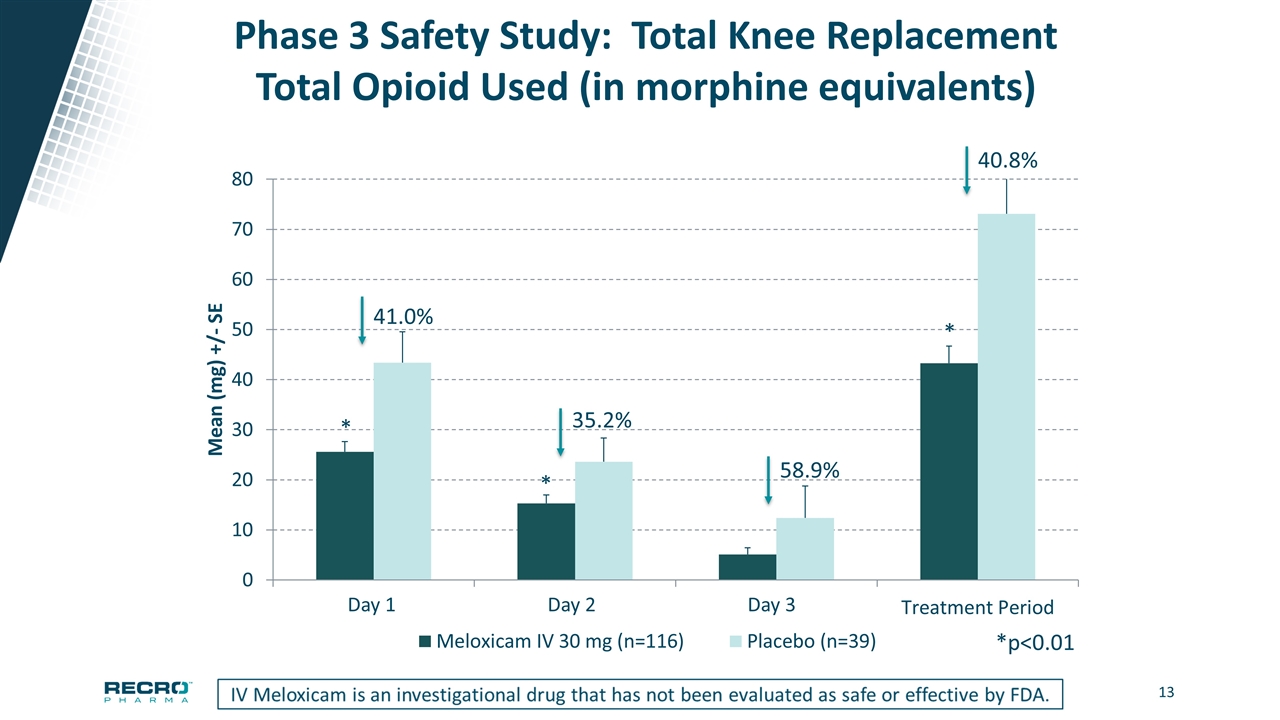
Phase 3 Safety Study: Total Knee Replacement Total Opioid Used (in morphine equivalents) 41.0% 35.2% *p<0.01 * * * 58.9% 40.8% Day 1 Day 2 Day 3
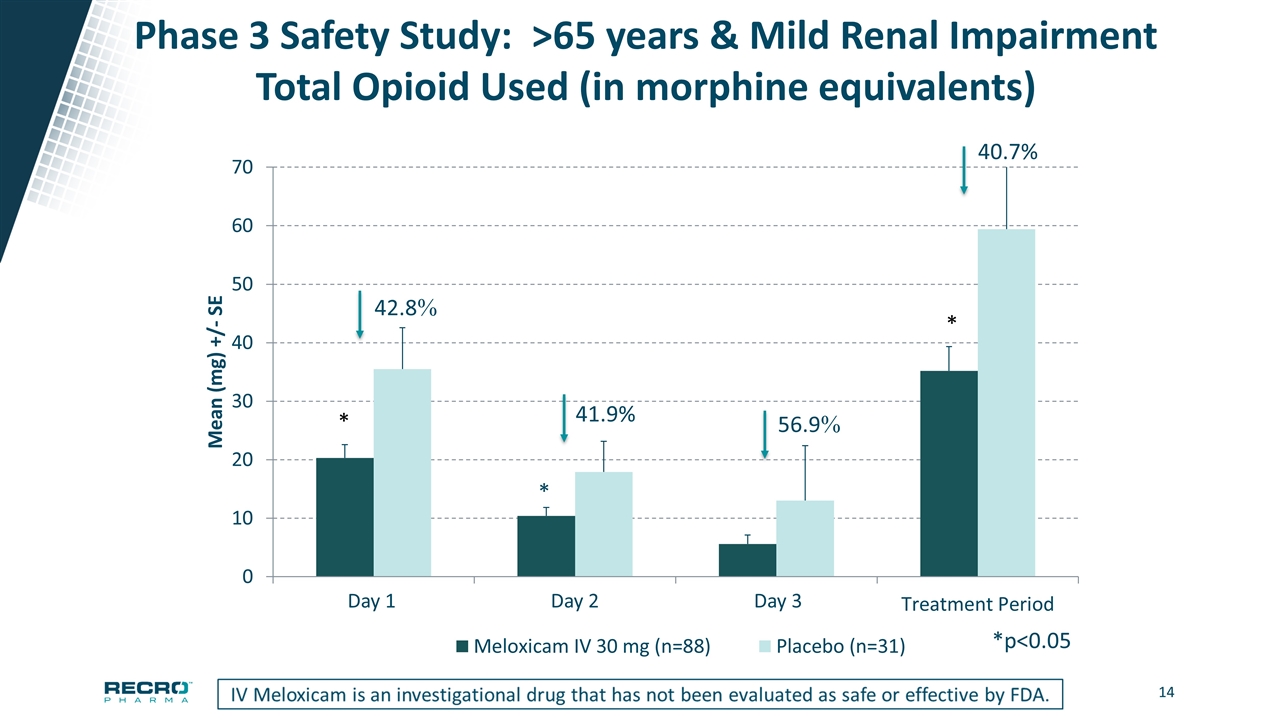
Phase 3 Safety Study: >65 years & Mild Renal Impairment Total Opioid Used (in morphine equivalents) *p<0.05 Day 1 Day 2 Day 3
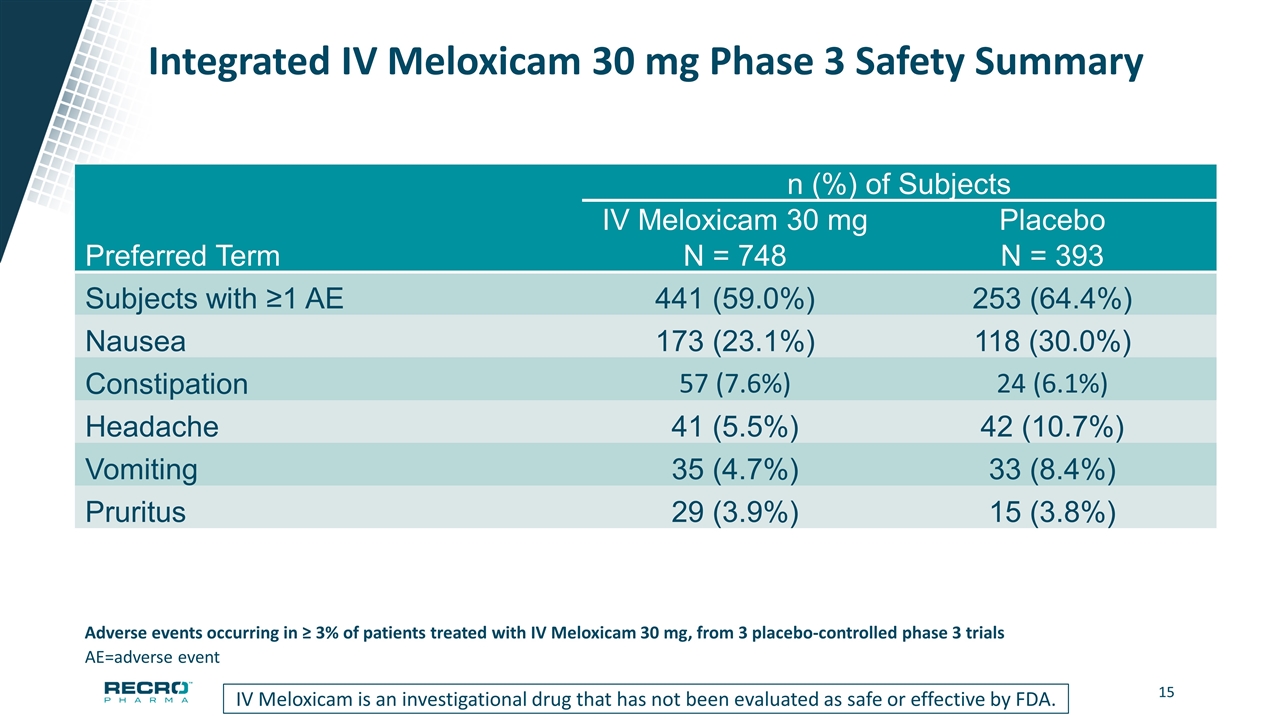
Integrated IV Meloxicam 30 mg Phase 3 Safety Summary n (%) of Subjects IV Meloxicam 30 mg Placebo Preferred Term N = 748 N = 393 Subjects with ≥1 AE 441 (59.0%) 253 (64.4%) Nausea 173 (23.1%) 118 (30.0%) Constipation 57 (7.6%) 24 (6.1%) Headache 41 (5.5%) 42 (10.7%) Vomiting 35 (4.7%) 33 (8.4%) Pruritus 29 (3.9%) 15 (3.8%) AE=adverse event Adverse events occurring in ≥ 3% of patients treated with IV Meloxicam 30 mg, from 3 placebo-controlled phase 3 trials IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA.

Next Steps for IV Meloxicam Filed NDA for IV Meloxicam in July 2017; Received FDA acceptance for review in September 2017 (PDUFA Action Date- May 26, 2018) Total treated subjects across NDA: approximately 1400 meloxicam patients Largest Phase 3, double-blind, placebo-controlled, non-opioid study evaluating an IV pain product in a post-operative setting Initiated Phase 3b studies Orthopedic surgery (total knee)- opioid consumption/pain intensity; health economics Colorectal surgery- opioid consumption; length of stay; health economics Execute commercialization and launch plan pending FDA approval IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA.

Go To Market Strategy
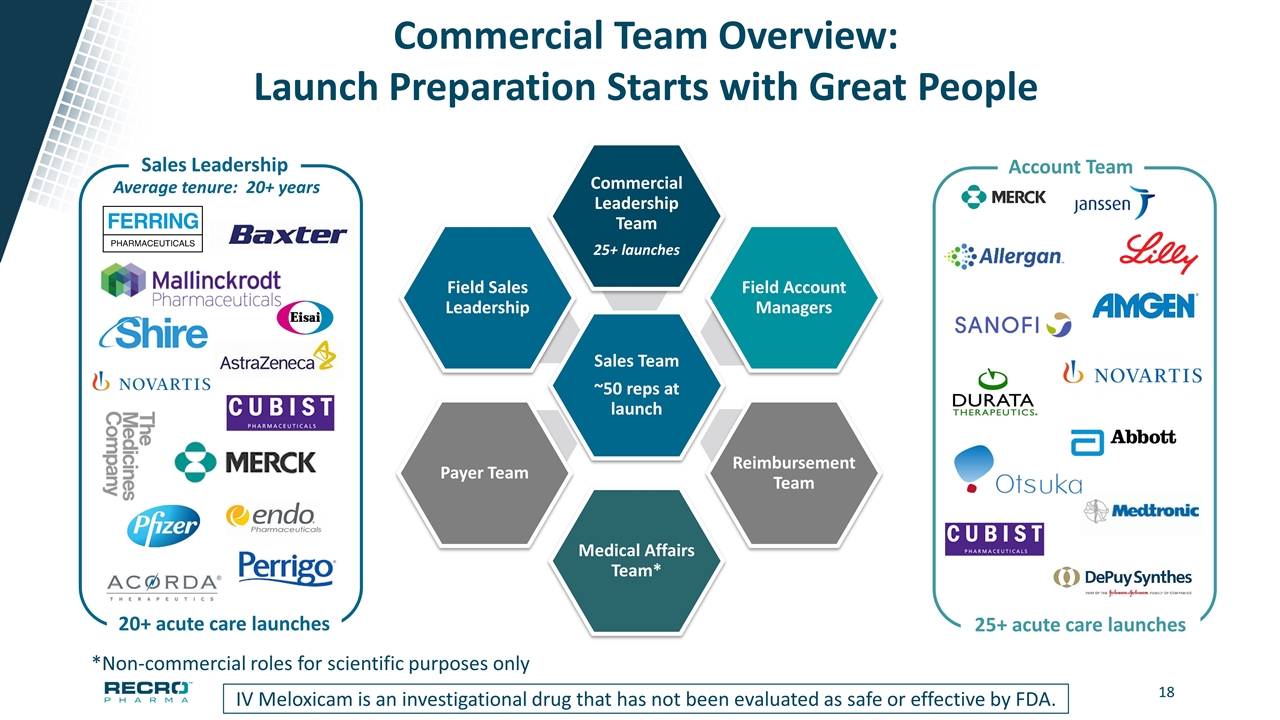
Commercial Team Overview: Launch Preparation Starts with Great People IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA. Sales Team ~50 reps at launch Commercial Leadership Team 25+ launches Field Account Managers Reimbursement Team Medical Affairs Team* Payer Team Field Sales Leadership Account Team 25+ acute care launches Sales Leadership 20+ acute care launches *Non-commercial roles for scientific purposes only Average tenure: 20+ years
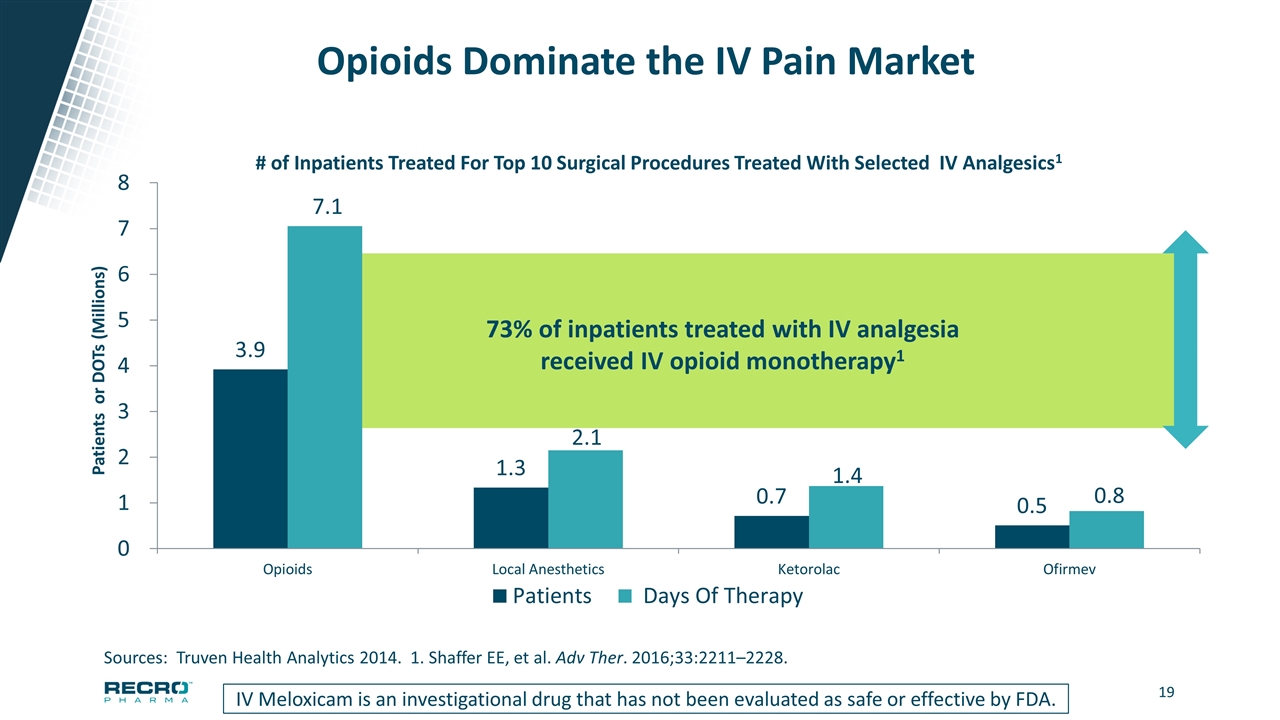
Opioids Dominate the IV Pain Market IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA. Sources: Truven Health Analytics 2014. 1. Shaffer EE, et al. Adv Ther. 2016;33:2211–2228. 73% of inpatients treated with IV analgesia received IV opioid monotherapy1

Incidence of and Risk Factors for Chronic Opioid Use Among Opioid-Naive Patients in the Post-operative Period In opioid-naive patients, many surgical procedures are associated with an increased risk of chronic opioid use in the post-operative period1 6%-10% of opioid naïve surgical patients will use opioids persistently2 Incidence of new persistent opioid use was similar between minor and major surgery, 5.9% and 6.5% respectively Incidence of new persistent opioid use in the nonoperative control group was only 0.4% IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA. 1. Sun EC, et al: JAMA Intern Med. Published online July 11, 2016. 2. Brummett CM, Waljee JF, Goesling J, et al. New Persistent Opioid Use After Minor and Major Surgical Procedures in US Adults. JAMA Surg. 2017;e170504. doi:10.1001/jamasurg.2017.0504

Multiple Guidelines Recommend NSAIDs as Part of Multimodal Analgesia (MMA) IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA. Numerous clinical practice guidelines recommend multimodal approaches to pain management—including NSAIDs—to provide better pain control while reducing opioids and related adverse events *The American Academy of Orthopaedic Surgeons and The Joint Commission recommend MMA, but do not specifically recommend NSAIDs.
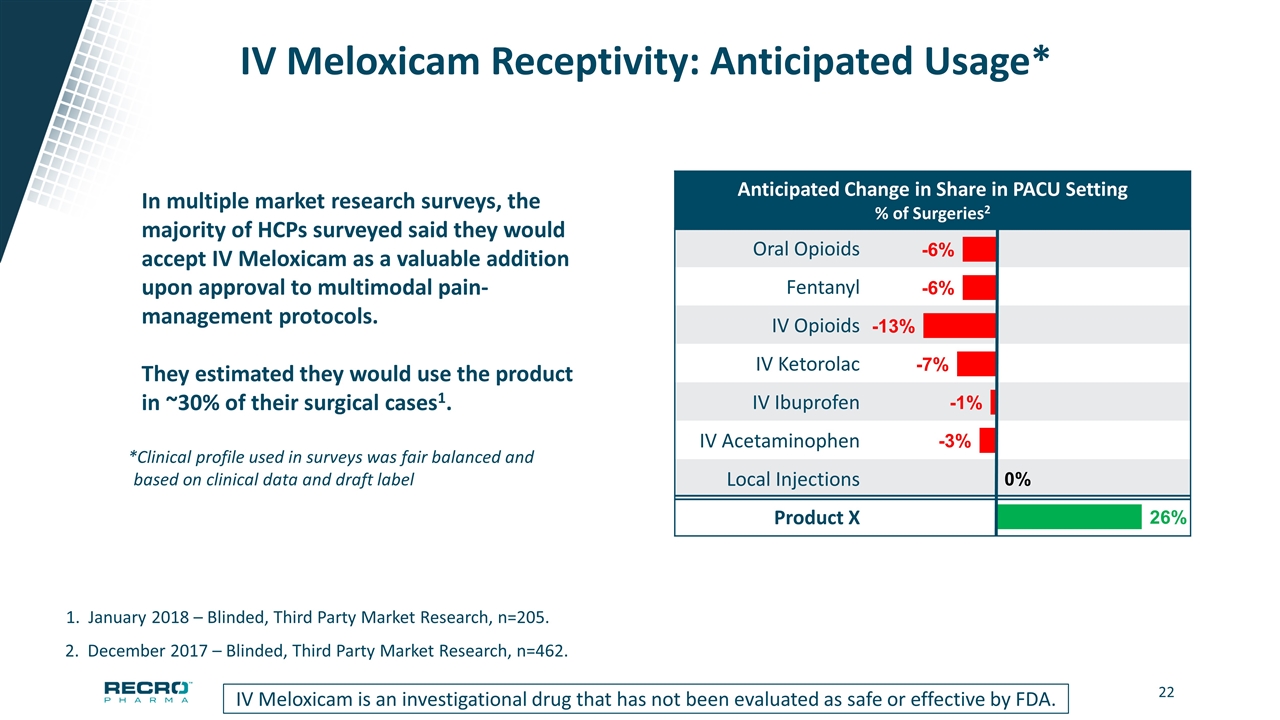
IV Meloxicam Receptivity: Anticipated Usage* IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA. 2. December 2017 – Blinded, Third Party Market Research, n=462. 1. January 2018 – Blinded, Third Party Market Research, n=205. In multiple market research surveys, the majority of HCPs surveyed said they would accept IV Meloxicam as a valuable addition upon approval to multimodal pain-management protocols. They estimated they would use the product in ~30% of their surgical cases1. Anticipated Change in Share in PACU Setting % of Surgeries2 Oral Opioids Fentanyl IV Opioids IV Ketorolac IV Ibuprofen IV Acetaminophen Local Injections Product X *Clinical profile used in surveys was fair balanced and based on clinical data and draft label
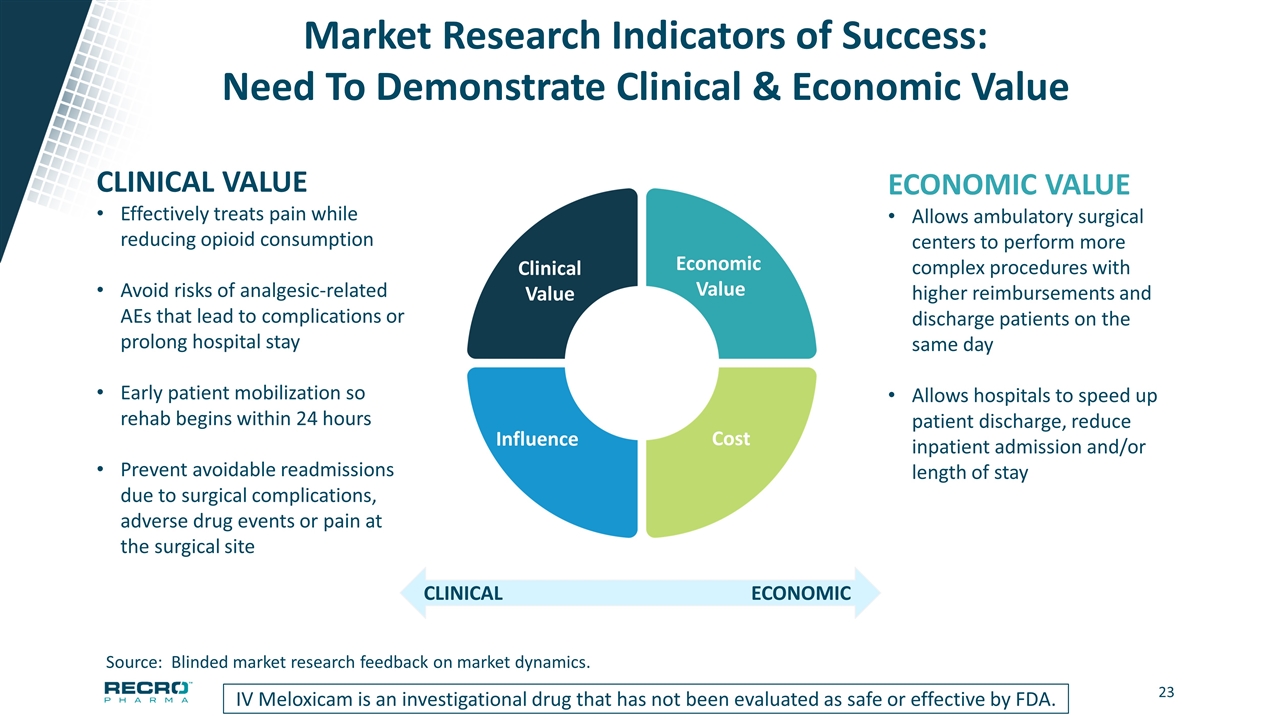
Market Research Indicators of Success: Need To Demonstrate Clinical & Economic Value IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA. CLINICAL VALUE Effectively treats pain while reducing opioid consumption Avoid risks of analgesic-related AEs that lead to complications or prolong hospital stay Early patient mobilization so rehab begins within 24 hours Prevent avoidable readmissions due to surgical complications, adverse drug events or pain at the surgical site ECONOMIC VALUE Allows ambulatory surgical centers to perform more complex procedures with higher reimbursements and discharge patients on the same day Allows hospitals to speed up patient discharge, reduce inpatient admission and/or length of stay ECONOMIC CLINICAL Source: Blinded market research feedback on market dynamics. Influence Clinical Value Cost Economic Value

A Non-Opioid Alternative: IV Meloxicam Key Learnings IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA. Once-daily IV that delivers 24-hour relief for the management of moderate to severe pain Opioid reduction up to 72 hours with once daily dosing Reduced opioid use in patients over age 65 with mild renal impairment Acts rapidly, within 10-15 minutes post dosing Preferential COX-2 NSAID with safety and tolerability evaluated in more than 1500 surgical patients Source: Most appealing attributes on IV Meloxicam based on multiple market research activities *Clinical profile used in market research was fair balanced and based on clinical data and draft label Customer Insights & Learnings from over 1,000 HCPs

Launching in the Acute Care Space IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA. Traditional Hospital Launches Focus on the Institutions Our Approach is to Focus on the Physician and Their Settings of Care Abbreviations: IPAT = Inpatient, HOPD = Hospital Outpatient Department, ASCs = Ambulatory Surgical Centers PHYSICIAN Hospital IPAT HOPD ASC REGULATORY APPROVAL FORMULARY ACCESS PHYSICIAN USAGE
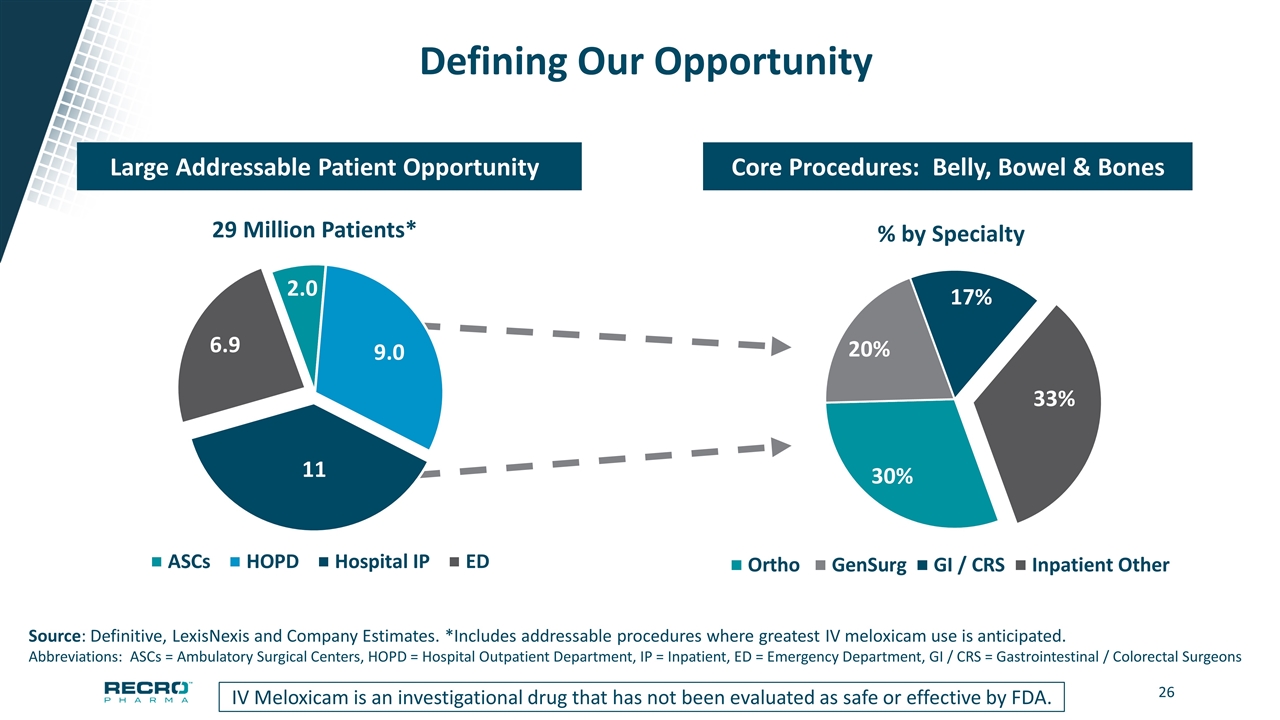
Defining Our Opportunity IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA. Core Procedures: Belly, Bowel & Bones Large Addressable Patient Opportunity Source: Definitive, LexisNexis and Company Estimates. *Includes addressable procedures where greatest IV meloxicam use is anticipated. Abbreviations: ASCs = Ambulatory Surgical Centers, HOPD = Hospital Outpatient Department, IP = Inpatient, ED = Emergency Department, GI / CRS = Gastrointestinal / Colorectal Surgeons
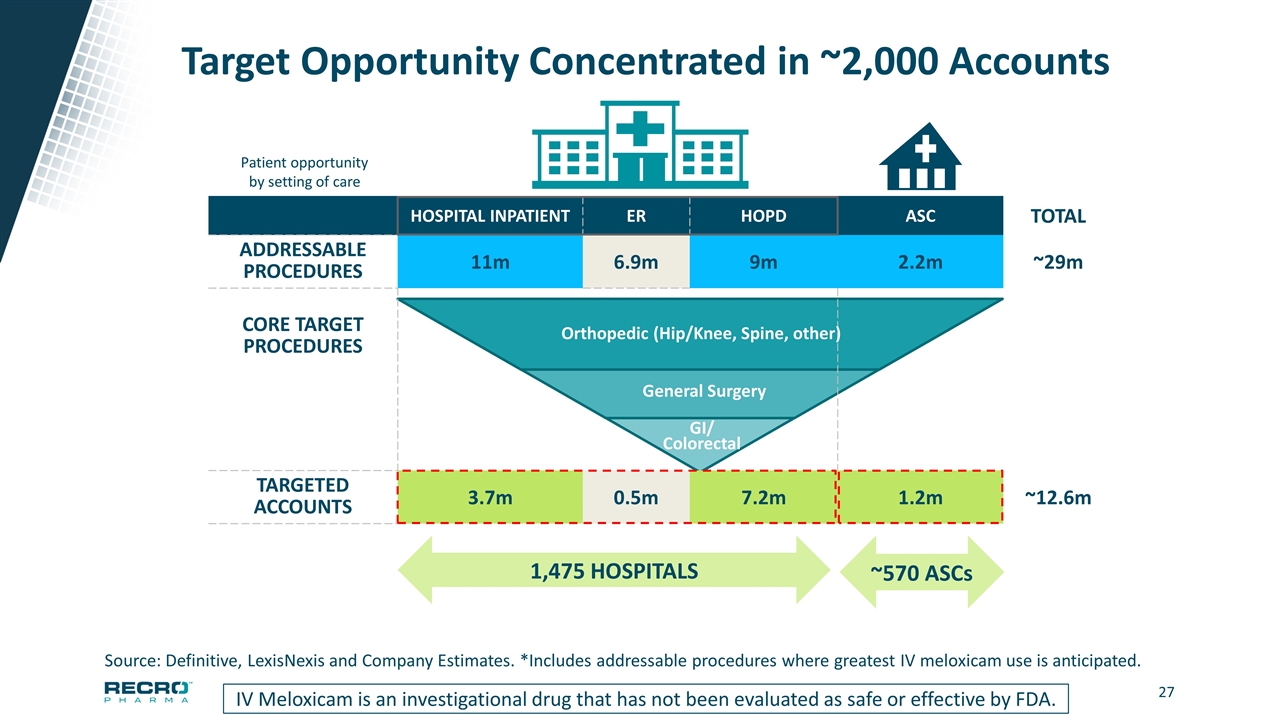
Target Opportunity Concentrated in ~2,000 Accounts IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA. HOSPITAL INPATIENT ER HOPD ASC TOTAL ADDRESSABLE PROCEDURES 11m 6.9m 9m 2.2m ~29m CORE TARGET PROCEDURES TARGETED ACCOUNTS 3.7m 0.5m 7.2m 1.2m ~12.6m 1,475 HOSPITALS ~570 ASCs Source: Definitive, LexisNexis and Company Estimates. *Includes addressable procedures where greatest IV meloxicam use is anticipated. GI/ Colorectal General Surgery Orthopedic (Hip/Knee, Spine, other) Patient opportunity by setting of care
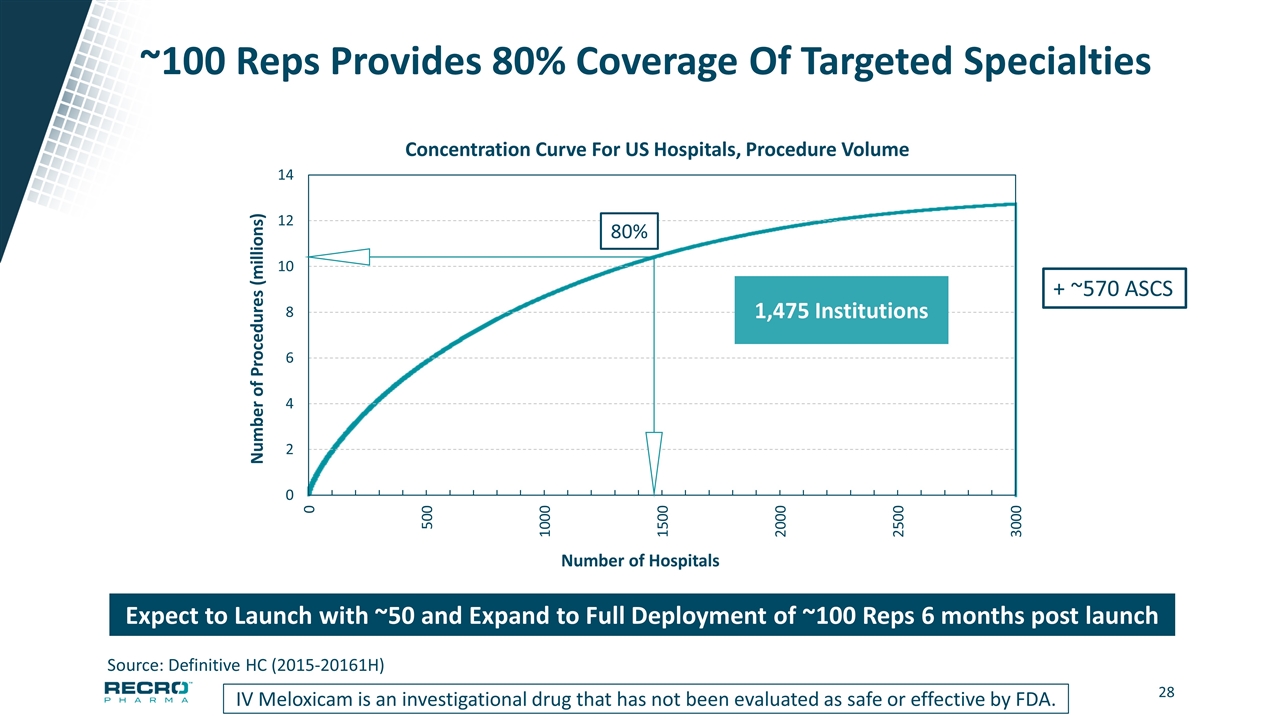
~100 Reps Provides 80% Coverage Of Targeted Specialties IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA. Source: Definitive HC (2015-20161H) Number of Procedures (millions) Number of Hospitals 80% 1,475 Institutions Expect to Launch with ~50 and Expand to Full Deployment of ~100 Reps 6 months post launch + ~570 ASCS
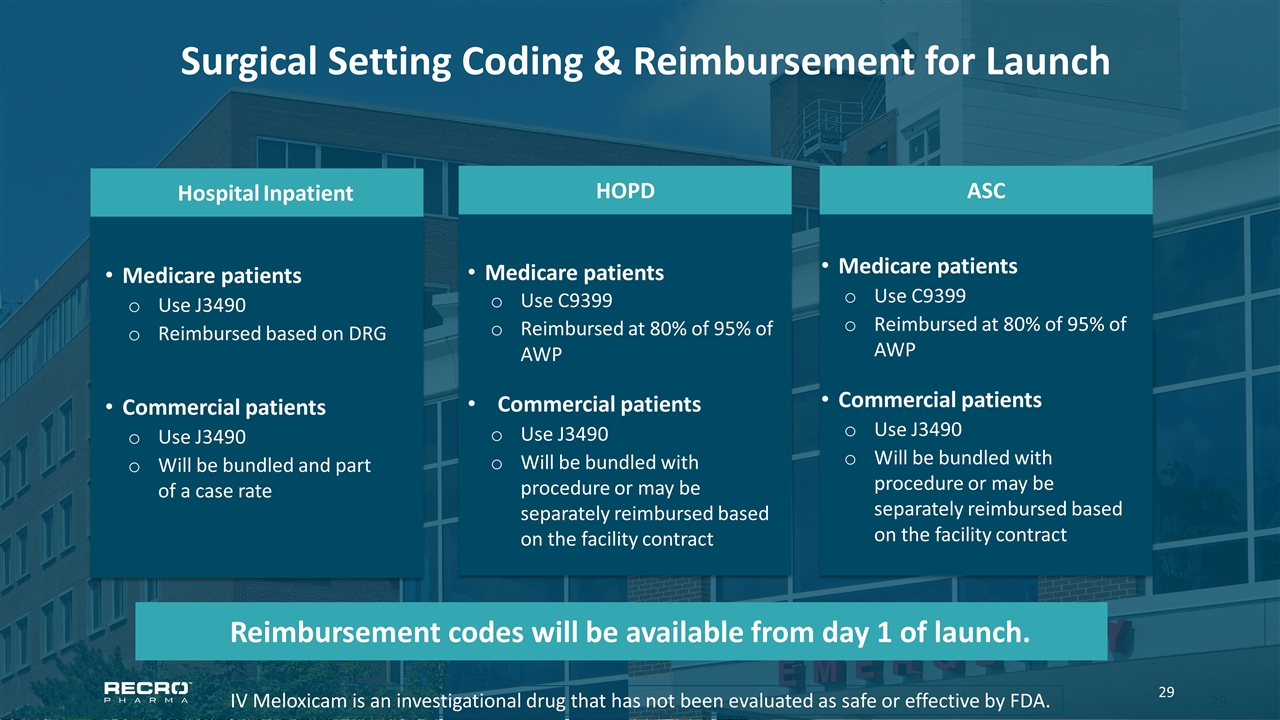
Surgical Setting Coding & Reimbursement for Launch IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA. ASC Medicare patients Use C9399 Reimbursed at 80% of 95% of AWP Commercial patients Use J3490 Will be bundled with procedure or may be separately reimbursed based on the facility contract HOPD Medicare patients Use C9399 Reimbursed at 80% of 95% of AWP Commercial patients Use J3490 Will be bundled with procedure or may be separately reimbursed based on the facility contract Hospital Inpatient Medicare patients Use J3490 Reimbursed based on DRG Commercial patients Use J3490 Will be bundled and part of a case rate Reimbursement codes will be available from day 1 of launch.

Contract Development and Manufacturing (CDMO) Business Overview Gainesville

Gainesville CDMO Facility Gainesville, GA, Facility Located on 148 acres of land, the facility has approximately 97,000 square feet of space Plant began operations early 90’s Includes 288 pallet vault built in accordance with DEA specifications Operates under cGMP standards Two level facility FDA/EMA/DEA Licensed Development, Scale-up and full scale manufacturing Manufactures product for U.S., European and other markets Solid oral-dosage forms (capsule, tablets, coated and un-coated) commercial capacity

CDMO Overview CDMO facility 97,000 + sq. ft. solid oral dosage manufacturing cGMP DEA licensed; ~190 employees Revenues include product sales, royalties and profit sharing Positive cash flow providing debt service and non-dilutive financing source for Company development and operating activities Service capabilities Formulation, process development and optimization Process scale-up Clinical supply and validation Commercial supply Ritalin LA Once daily ADHD treatment marketed by Novartis Focalin XR ADHD treatment marketed by Novartis Verapamil/Verelan CV/High blood pressure treatment marketed by Teva and Lannett Zohydro ER Extended release hydrocodone marketed by Pernix Abuse deterrent form launched

Strong CDMO Business Performance Revenues include product sales, royalties, profit sharing and R&D services 2018 Guidance- Revenues of approximately $70M, and EBITDA* of approximately $30M 2018 revenue will be affected by new accounting revenue recognition standard Additional capacity for new product opportunities Positive cash flow for debt service obligations, as well as cash flow to contribute to the funding of Company operating activities, including product development and commercialization. *EBITDA is a non-GAAP financial measure (see reconciliation on last page presentation) CDMO Segment ($millions) 2017 (unaudited) Revenues $71.8 CDMO Segment Operating Income $25.4 EBITDA* $32.8

$100 Million November 2017 Credit Facility Five-year, interest-only term loan at LIBOR +9.75%; Issuance of 348,664 warrants at $8.60/share Three tranches $60 million at closing $20 million upon FDA approval of IV Meloxicam subject to certain financial liquidity conditions $20 million available after Recro demonstrates early IV Meloxicam commercial traction Financial flexibility to aid our transformation into a commercial-stage enterprise Substantial reduction of our cost of capital Provides funding for IV Meloxicam FDA approval $45 million milestone Interest-only structure allows for reinvestment of CDMO’s cash flows in value accretive commercial activities Repaid existing OrbiMed credit facility with payment of $31.7 million, plus transaction fees IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA.

Company Highlights Specialty pharmaceutical company focused on hospital and related settings with late stage investigational product, IV Meloxicam, targeting management of moderate to severe pain Filed New Drug Application for IV Meloxicam in July 2017; PDUFA Action Date- May 26, 2018 Commercial teams and go to market strategy prepared Multiple therapeutics in clinical development for hospital and related settings Revenue and cash flow positive contract development and manufacturing (CDMO) business 2017 Revenue- $71.8M; 2017 EBITDA- $32.8M Cash position- $64.5M @ 12/31/17 $100M credit facility secured in November 2017 Experienced management team with significant development, regulatory and commercial experience *EBITDA is a non-GAAP financial measure. See reconciliation on last page of presentation. IV Meloxicam is an investigational drug that has not been evaluated as safe or effective by FDA.

Reconciliation of Non-GAAP Financial Measures (unaudited) CDMO Business ($millions) 2017 2018 Estimate Operating Income $25.4 $22.4 Depreciation $4.8 $5.0 Amortization of intangible assets $2.6 $2.6 EBITDA $32.8 $30.0 To supplement our financial results determined by U.S. generally accepted accounting principles ("GAAP"), we have also disclosed in the table below the following non-GAAP information for our Contract Development and Manufacturing Organization (CDMO): earnings before interest, taxes, depreciation and amortization ("EBITDA"). We believe this non-GAAP financial measure is helpful in understanding our Manufacturing Business as it is useful to investors in allowing for greater transparency of supplemental information used by management. EBITDA is used by investors, as well as management in assessing our performance. Non-GAAP financial measures should be considered in addition to, but not as a substitute for, reported GAAP results. Further, Non-GAAP financial measures, even if similarly titled, may not be calculated in the same manner by all companies, and therefore should not be compared.



































