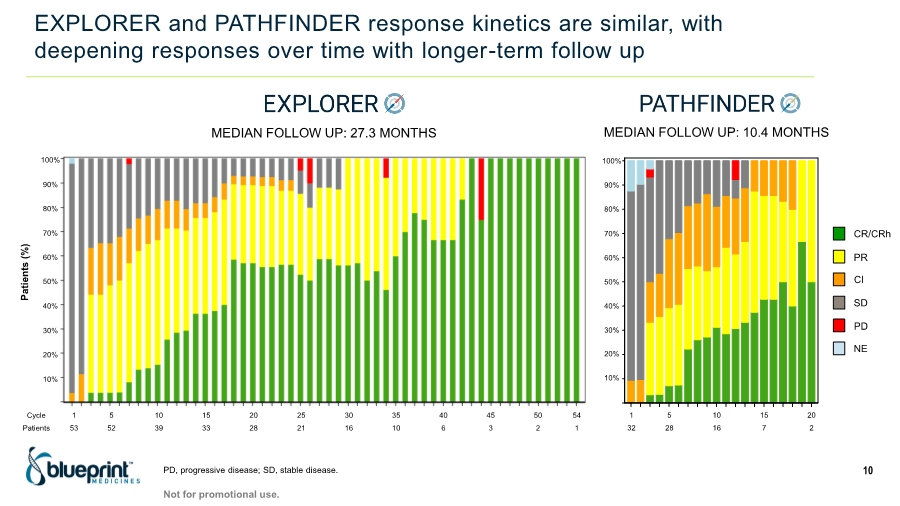
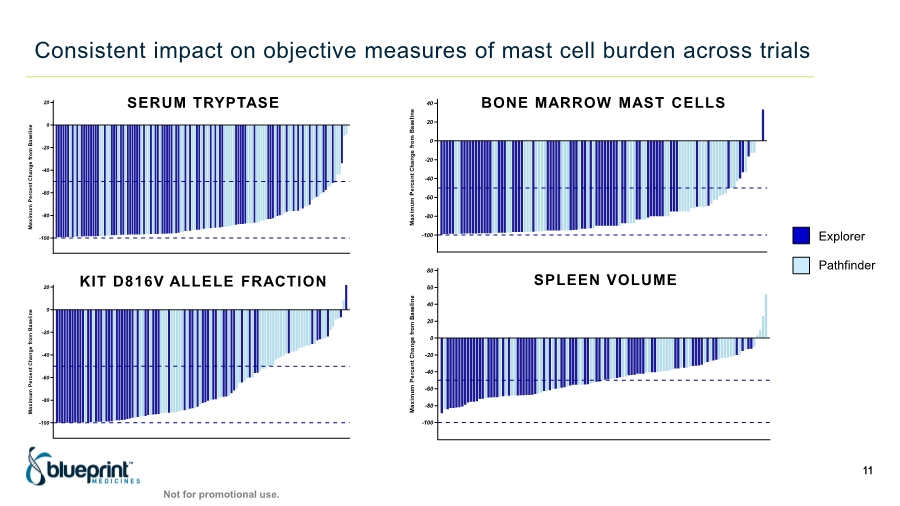
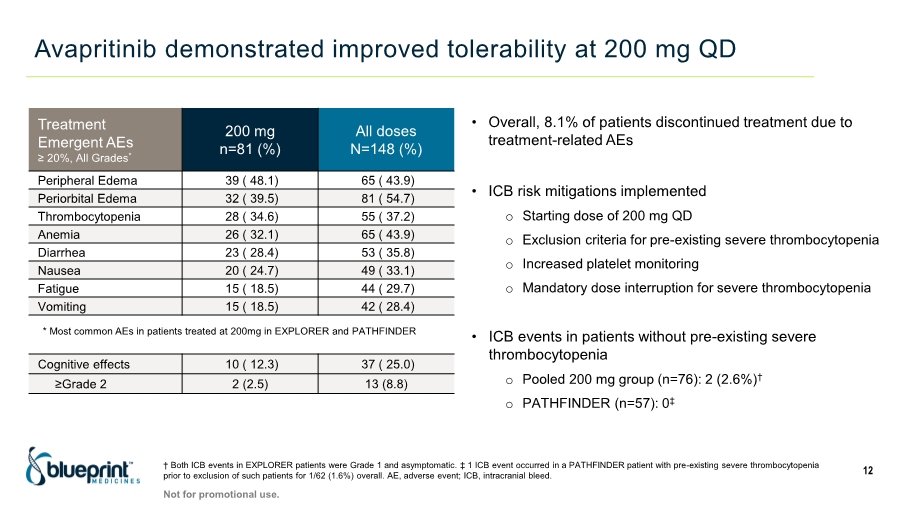
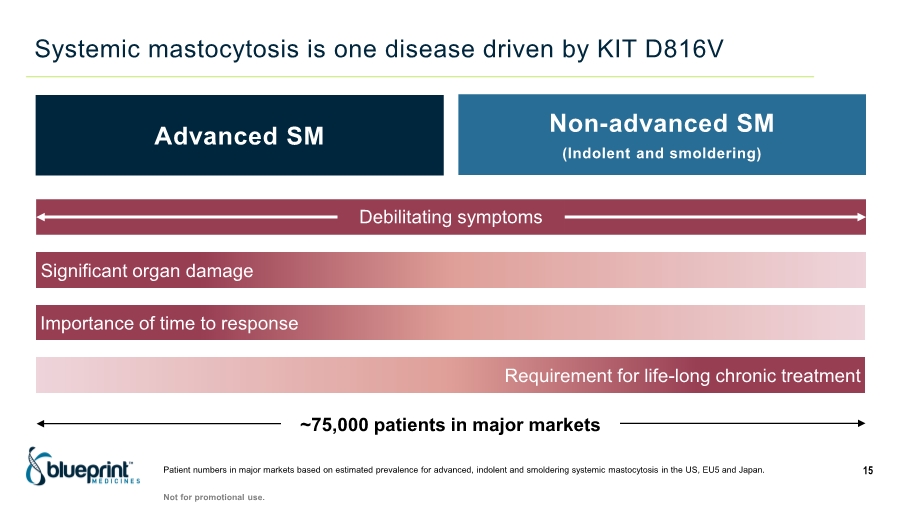
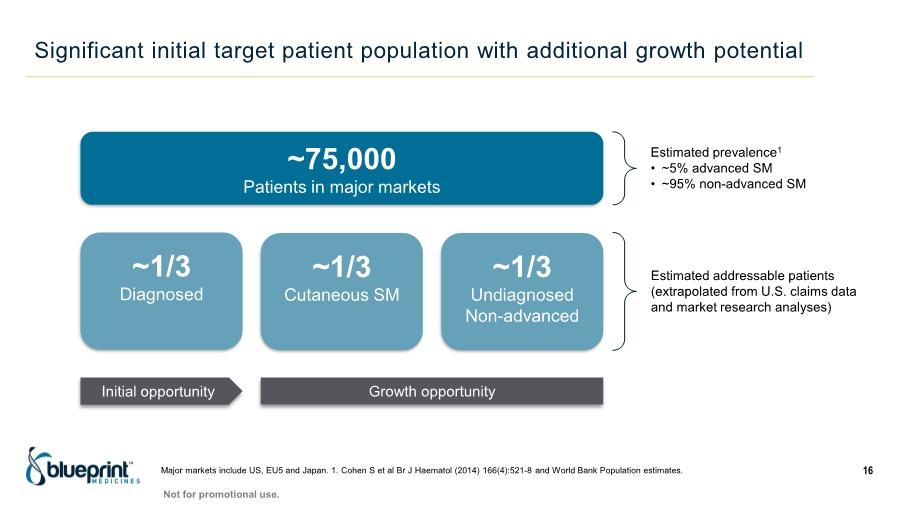
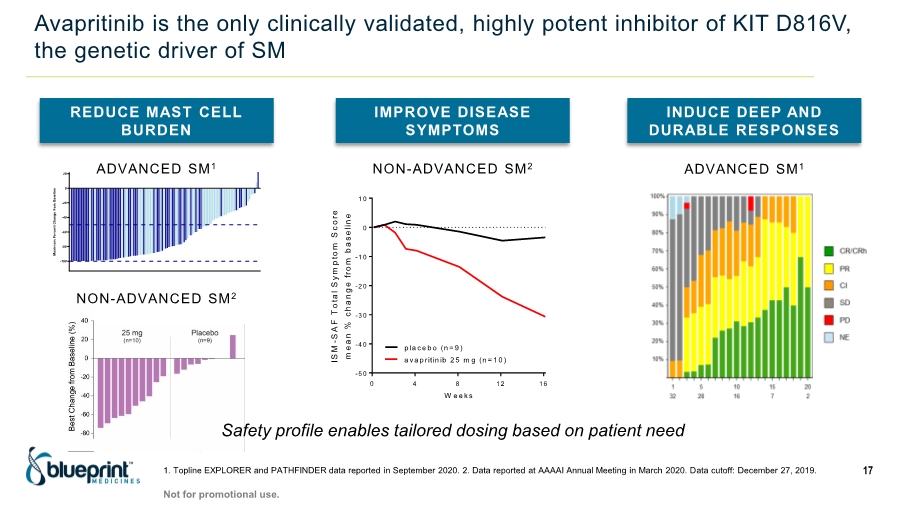
| Forward-looking statements 2 This presentation contains forward-looking statements as defined in the Private Securities Litigation Reform Act of 1995, as amended. The words “aim,” “may,” “will,” “could,” “would,” “should,” “expect,” “plan,” “anticipate,” “intend,” “believe,” “estimate,” “predict,” “project,” “potential,” “continue,” “target” and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. In this presentation, forward-looking statements include, without limitation, statements regarding plans, timelines and expectations for interactions with the U.S. Food and Drug Administration (“FDA”) and other regulatory authorities; plans and timelines for submitting an supplemental New Drug Application to the FDA for AYVAKIT™ (avapritinib) for the treatment of advanced systemic mastocytosis (“SM”); expectations regarding the potential benefits of AYVAKIT in treating patients with SM; and the strategy, goals and anticipated milestones, business plans and focus of Blueprint Medicines Corporation (the “Company”). The Company has based these forward-looking statements on management’s current expectations, assumptions, estimates and projections. While the Company believes these expectations, assumptions, estimates and projections are reasonable, such forward-looking statements are only predictions and involve known and unknown risks, uncertainties and other important factors, many of which are beyond the Company’s control and may cause actual results, performance or achievements to differ materially from those expressed or implied by any forward-looking statements. These risks and uncertainties include, without limitation, risks and uncertainties related to the impact of the COVID-19 pandemic to the Company’s’ business, operations, strategy, goals and anticipated milestones, including the Company’s ongoing and planned research and discovery activities, ability to conduct ongoing and planned clinical trials, clinical supply of current or future drug candidates, commercial supply of current or future approved products, and launching, marketing and selling current or future approved products; the Company’s ability and plans in establishing a commercial infrastructure, and successfully launching, marketing and selling current or future approved products, including AYVAKIT and GAVRETO™ (pralsetinib); the Company’s ability to successfully expand the approved indications for AYVAKIT and GAVRETO or obtain marketing approval for AYVAKIT and GAVRETO in additional geographies in the future; the delay of any current or planned clinical trials or the development of the Company’s current or future drug candidates; the Company’s advancement of multiple early-stage efforts; the Company’s ability to successfully demonstrate the safety and efficacy of its drug candidates and gain approval of its drug candidates on a timely basis, if at all; the preclinical and clinical results for the Company’s drug candidates, which may not support further development of such drug candidates; actions of regulatory agencies, which may affect the initiation, timing and progress of clinical trials; Blueprint Medicines’ ability to develop and commercialize companion diagnostic tests for its current and future drug candidates; and the success of Blueprint Medicines’ current and future collaborations, partnerships or licensing arrangements, including the Company’s global collaboration with Roche for the development and commercialization of GAVRETO. These and other risks and uncertainties are described in greater detail under “Risk Factors” in the Company’s filings with the Securities and Exchange Commission (“SEC”), including its most recent Annual Report on Form 10-K, as supplemented by its most recent Quarterly Report on Form 10-Q and any other filings it has made or may make with the SEC in the future. The Company cannot guarantee future results, outcomes, levels of activity, performance, developments, or achievements, and there can be no assurance that its expectations, intentions, anticipations, beliefs, or projections will result or be achieved or accomplished. The forward-looking statements in this presentation are made only as of the date hereof, and except as required by law, the Company undertakes no obligation to update any forward-looking statements contained in this presentation as a result of new information, future events or otherwise. This presentation also contains estimates, projections and other statistical data made by independent parties and by the Company relating to market size and growth and other data about the Company’s industry. These data involve a number of assumptions and limitations, and you are cautioned not to give undue weight to such estimates. In addition, projections, assumptions and estimates of the Company’s future performance and the future performance of the markets in which the Company operates are necessarily subject to a high degree of uncertainty and risk. |