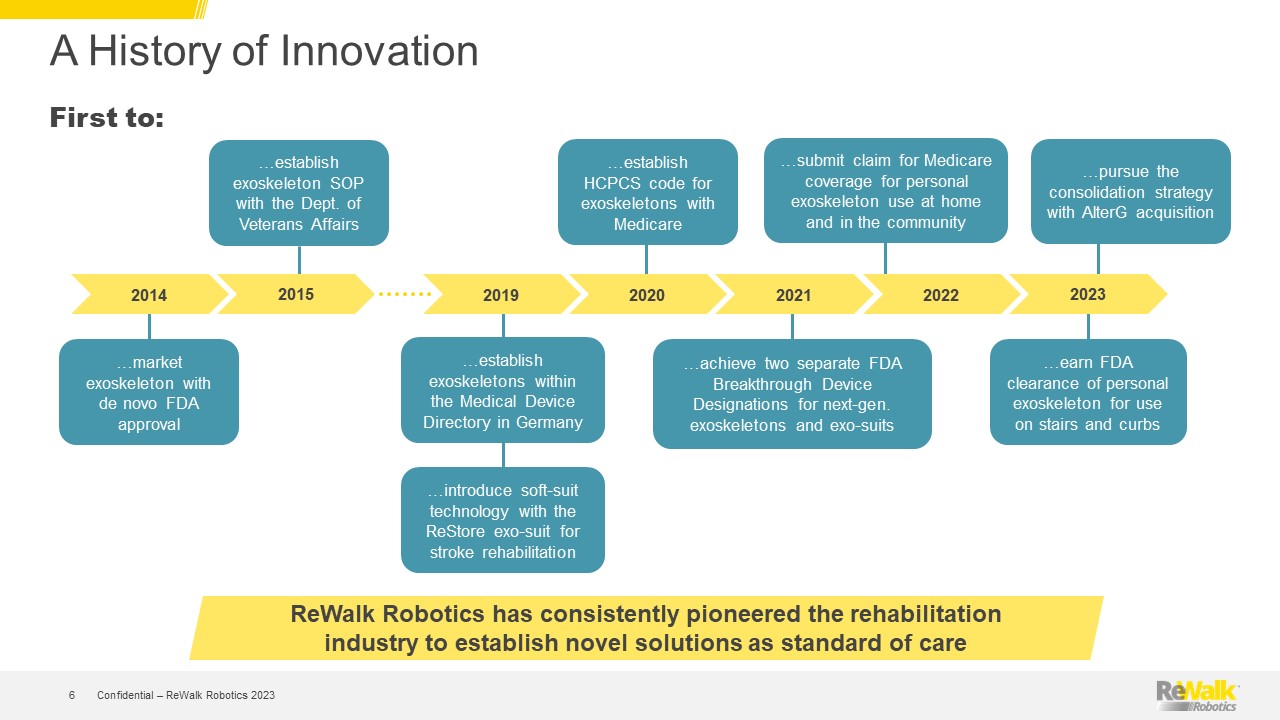
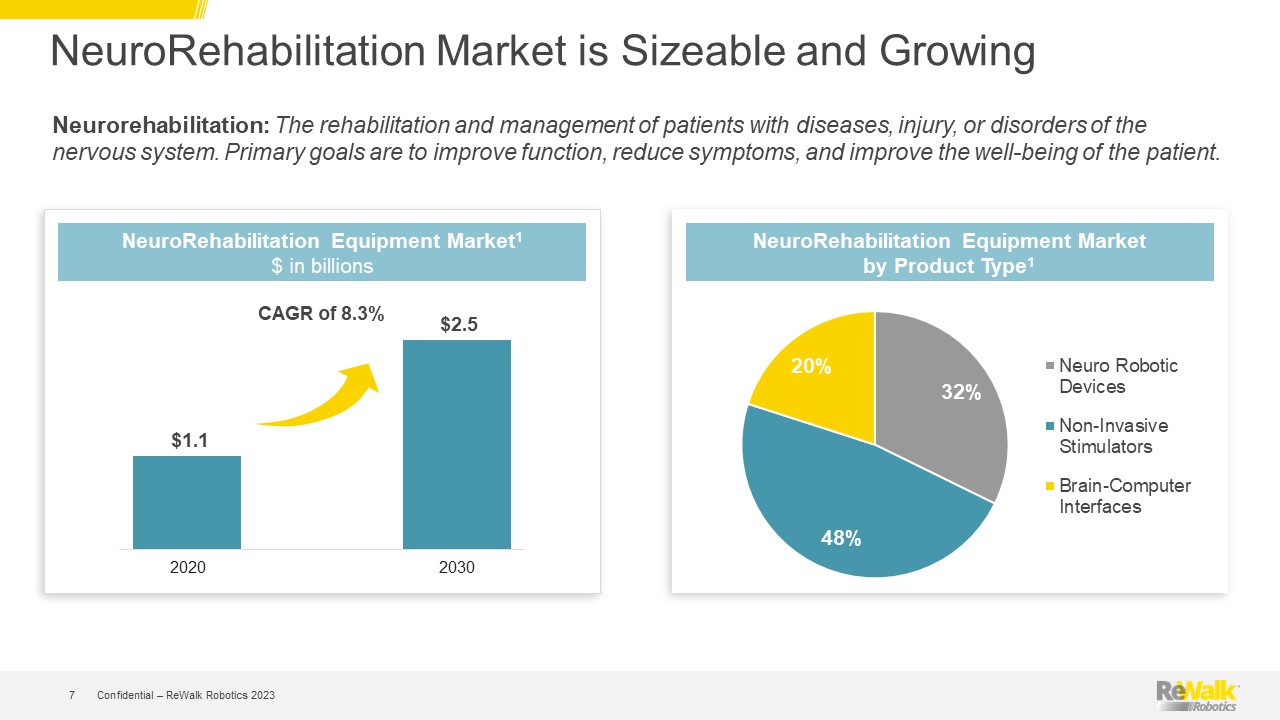
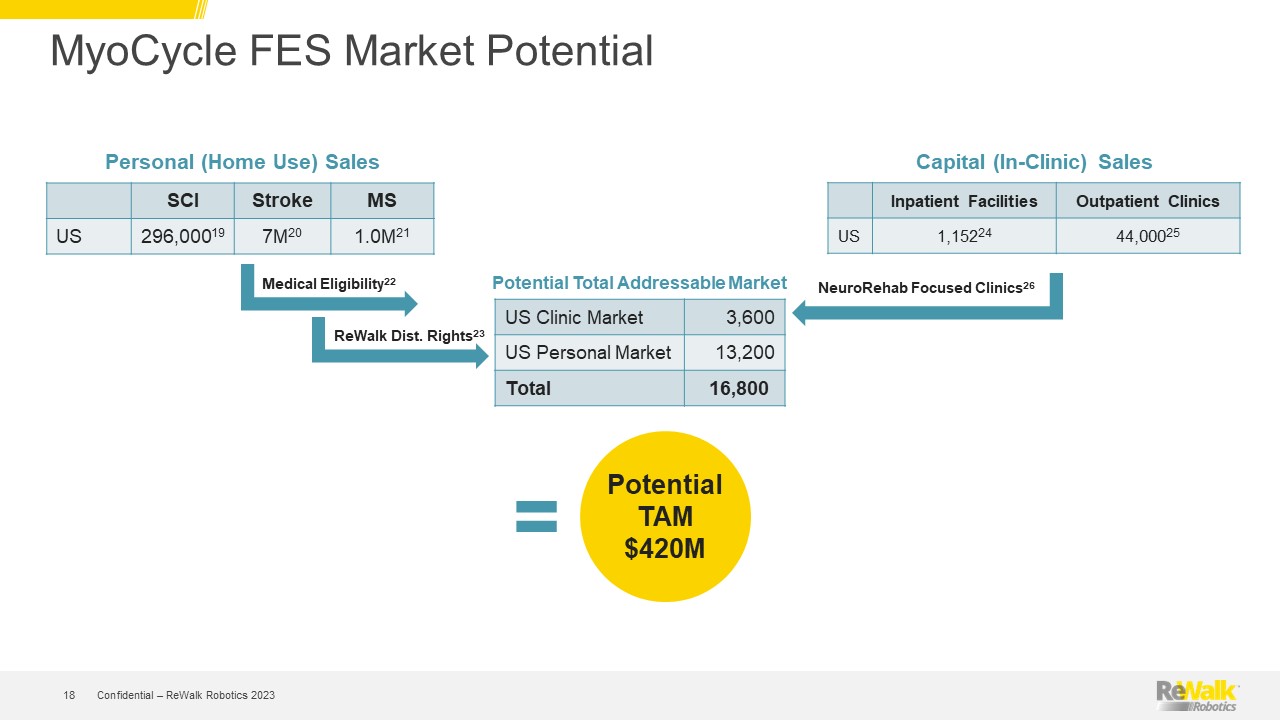
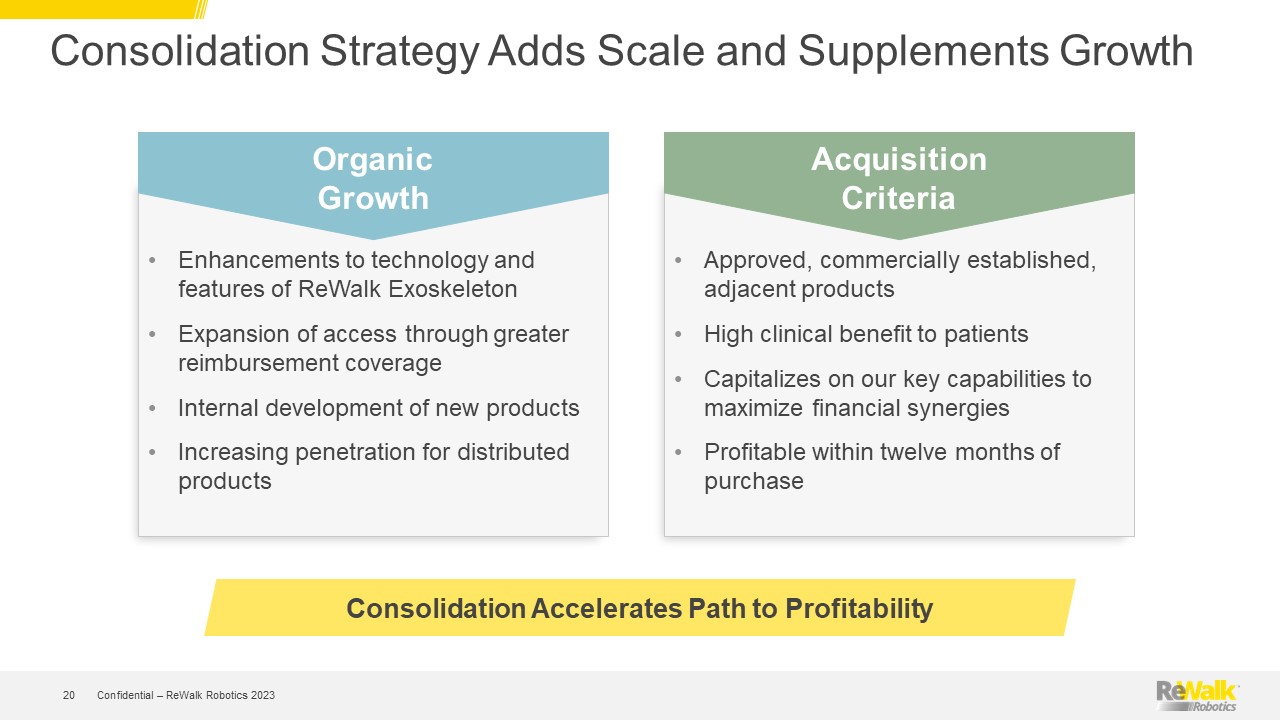
Forward Looking Statements This presentation contains forward-looking statements within the meaning of the U.S. Private Securities Litigation Reform Act of 1995, Section 27A of the U.S. Securities Act of 1933, and Section 21E of the U.S. Securities Exchange Act of 1934. All statements contained in this presentation other than statements of historical fact are forward-looking statements. Such forward-looking statements may include projections regarding ReWalk's future performance and, in some cases, may be identified by words like "anticipate," "assume," "believe," "continue," "could," "estimate," "expect," "intend," "may," "plan," "potential," "predict," "project," "future," "will," "should," "would," "seek," and similar terms or phrases. The forward-looking statements contained in this presentation are based on management's current expectations, which are subject to uncertainty, risks and changes in circumstances that are difficult to predict and many of which are outside of ReWalk's control. Important factors that could cause ReWalk's actual results to differ materially from those indicated in the forward-looking statements are more fully described in ReWalk’s periodic filings with the Securities and Exchange Commission (“SEC”), including the risk factors described in the section entitled "Risk Factors" in ReWalk's annual and quarterly reports that ReWalk files with the SEC. In addition, this presentation contains estimates, projections and other information concerning market, industry and other data. ReWalk obtained this data from its own internal estimates and research and from academic and industry research, publications, surveys, and studies conducted by third parties, including governmental agencies. These data involve a number of assumptions and limitations, are subject to risks and uncertainties, and are subject to change based on various factors, including those discussed in ReWalk’s filings with the SEC. These and other factors could cause results to differ materially from those expressed in the estimates made by the independent parties and by ReWalk. While management believes such information is generally reliable, ReWalk has not independently verified any third-party information. Forward-looking statements made in this presentation are based on a combination of facts and factors currently known to management and speak only as of the date hereof. Factors or events that could cause ReWalk's actual results to differ from the statements contained herein may emerge from time to time, and it is not possible for ReWalk to predict all of them. Except as required by law, ReWalk undertakes no obligation to publicly update any forward-looking statements, whether as a result of new information, future developments or otherwise. Confidential – ReWalk Robotics 2023