Exhibit 99.1

Investor Presentation April 2024

we are partners in energy storage Forward Looking Statements This presentation contains forward-looking statements within the meaning of the U.S. Private Securities Litigation Reform Act of 1995, Section 27A of the U.S. Securities Act of 1933, and Section 21E of the U.S. Securities Exchange Act of 1934. All statements contained in this presentation other than statements of historical fact are forward-looking statements. Such forward-looking statements may include projections regarding Lifeward’s future performance and, in some cases, may be identified by words like "anticipate," "assume," "believe," "continue," "could," "estimate," "expect," "intend," "may," "plan," "potential," "predict," "project," "future," "will," "should," "would," "seek," and similar terms or phrases. The forward-looking statements contained in this presentation are based on management's current expectations, which are subject to uncertainty, risks and changes in circumstances that are difficult to predict and many of which are outside of Lifeward’s control. Important factors that could cause Lifeward’s actual results to differ materially from those indicated in the forward-looking statements are more fully described in Lifeward’s periodic filings with the Securities and Exchange Commission (“SEC”), including the risk factors described in the section entitled "Risk Factors" in Lifeward’s annual and quarterly reports that ReWalk files with the SEC. In addition, this presentation contains estimates, projections and other information concerning market, industry and other data. Lifeward obtained this data from its own internal estimates and research and from academic and industry research, publications, surveys, and studies conducted by third parties, including governmental agencies. These data involve a number of assumptions and limitations, are subject to risks and uncertainties, and are subject to change based on various factors, including those discussed in Lifeward’s filings with the SEC. These and other factors could cause results to differ materially from those expressed in the estimates made by the independent parties and by Lifeward. While management believes such information is generally reliable, Lifeward has not independently verified any third-party information. Forward-looking statements made in this presentation are based on a combination of facts and factors currently known to management and speak only as of the date hereof. Factors or events that could cause Lifeward’s actual results to differ from the statements contained herein may emerge from time to time, and it is not possible for Lifeward to predict all of them. Except as required by law, Lifeward undertakes no obligation to publicly update any forward-looking statements, whether as a result of new information, future developments or otherwise.

https://youtu.be/-WC02eT-Yig

The Lifeward Mission: Our mission at Lifeward is to relentlessly drive innovation to change the lives of people with physical limitations or disabilities. We are committed to delivering groundbreaking solutions that empower individuals to do what they love.

5 A Leader in Pioneering Life-Changing Solutions Recent strategic achievements FDA approval of stairs / curb capability for the ReWalk exoskeleton Acquisition of AlterG, the initial innovator of Anti-Gravity Systems CMS establishes Medicare benefit category for exoskeletons First claim paid by Medicare for a ReWalk Personal Exoskeleton Market-leading innovation – First-to-market personal exoskeleton for spinal cord injury, with over 600 placements worldwide Strong cash position – We believe we have resources to fund organic growth, M&A, and internal development Strategic vision – Leverage leadership position in novel technologies to become consolidator of broad portfolio of life changing solutions for physical rehabilitation and recovery Experienced management – Multi-disciplinary leadership with successful track records in medical technology, life science tools, and biopharmaceuticals

A History of Innovative Firsts 6 First personal exoskeleton to market with FDA and CE approvals First commercial application of NASA-derived Differential Air Pressure Technology First to establish a listing for exoskeletons within the German Medical Device Directory First Anti-Gravity Treadmill to be installed in over 4,000 clinics worldwide First CE and FDA cleared Exo-Suit for stroke rehabilitation First Personal Exoskeleton to receive FDA Breakthrough Designation for use on stairs and curbs First to achieve Medicare coverage for a personal exoskeleton

Product Portfolio

8 Innovation in Motion: Portfolio Overview ReWalk Personal Exoskeleton State-of-the-art technology that enables paralyzed individuals to access the health-related benefits of walking from the comfort of their home and community setting. AlterG Anti-Gravity System NASA-derived Differential Air Pressure (DAP) technology that reduces the effects of gravity to enable users to move with calibrated support and reduced pain. MyoCycle FES Bike Easy-to-use, functional electric stimulation that allows individuals with weakened or paralyzed legs to therapeutically cycle in the clinic or home. ReStore Exo-Suit Versatile, robotic gait-training solution designed to promote restoration of functional walking patterns post-stroke.

9 Our flagship product ReWalk Personal Exoskeleton Unprecedented freedom – Only personal exoskeleton enabling ambulatory access to real-world environments, including locations with stairs or curbs Natural gait – Only personal exoskeleton with six adjustable degrees of freedom for more customizable, natural walking Robust construction – Engineered and built for years of daily use to keep up with users wherever they want to go Vibrant community – A worldwide community of ReWalk Personal Exoskeleton users that share common experiences

Proven Health Benefits Health benefits reported by ReWalk users in a 2023 home-user survey8 Frequently reported benefits in literature include reductions in common comorbidities after SCI, including: Reduced spasticity Improved bowel/bladder function Reduced chronic pain Regular access to exoskeleton-assisted walking results in a multitude of health and wellness benefits for people with spinal cord injury1-8

Increasing Progress of Reimbursement/Coverage 9M US Veterans covered under VA Personal Exoskeleton policy since 2015. Over 225 systems covered through private and other insurance, on a case-by-case basis. Exoskeletons assigned to Medicare Brace benefit category effective Jan 1, 2024. First Medicare claim paid for a ReWalk Personal Exoskeleton as a medically necessary device. 65M lives covered by DGUV (workers compensation) in Germany since 2019. Contractual coverage for 30% of all Germans through Statutory and Private health insurance plans: 23.2M lives covered since 2020 Additional 2M covered lives added in 2021 11 United States Germany

SCI Market Potential to Be Determined by CMS Coverage Potential Total Addressable Market US 15,416 Germany 2,064 Global 17,480 Potential TAM $1.75B US 296,0009 Germany 74,00010 Global 7.1 Million10 Prevalence of SCI Survivors Eligible Candidates11,12 Reimbursement Pathway13,14

13 AlterG Anti-Gravity Systems Broadening our impact to more individuals than ever before Expert & proven development – NASA-derived technology to reduce the effects of gravity during rehabilitation and recovery Precision Calibration – patented algorithms enable precise measurement and real-time adjustments to ensure accurate unweighting at every step. Established Market Presence – Over 6,000 units used globally with broad indications Growth Potential – significant untapped potential in entry level market segments
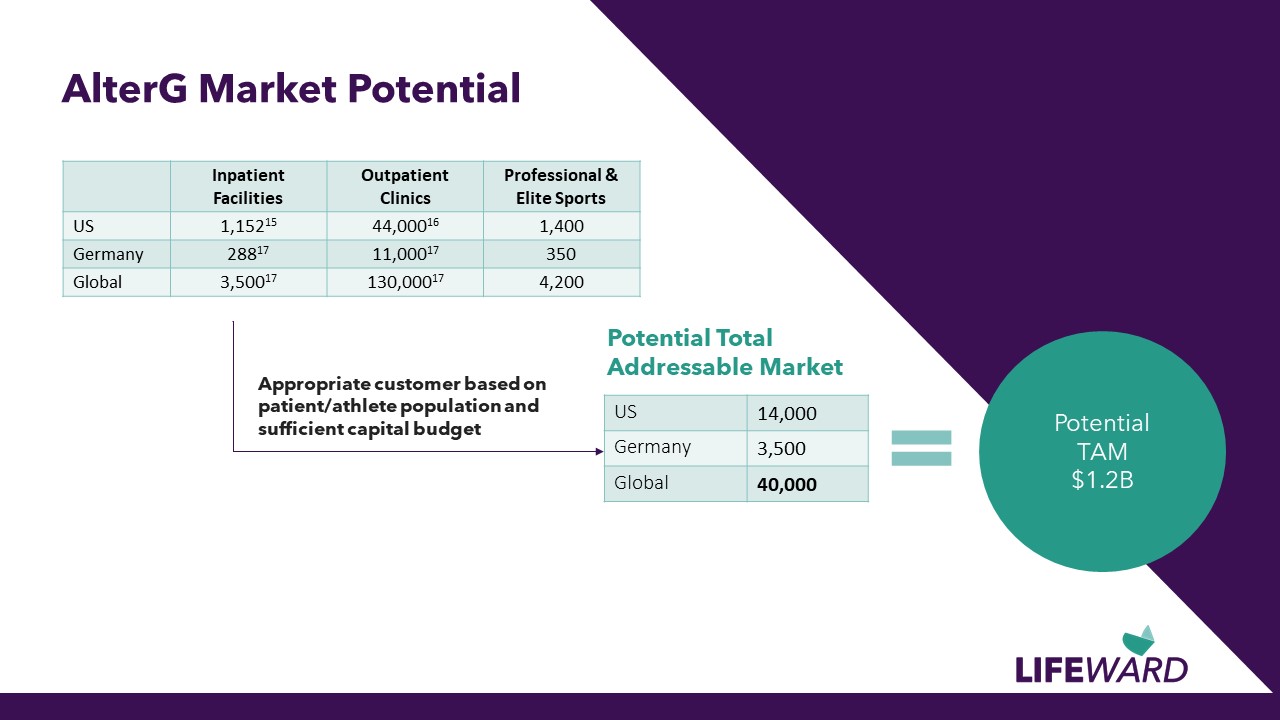
AlterG Market Potential Appropriate customer based on patient/athlete population and sufficient capital budget Potential Total Addressable Market US 14,000 Germany 3,500 Global 40,000 Potential TAM $1.2B Inpatient Facilities Outpatient Clinics Professional & Elite Sports US 1,15215 44,00016 1,400 Germany 28817 11,00017 350 Global 3,50017 130,00017 4,200

15 Additional Complementary Product Lines ReStore Exo-Suit for Stroke Rehabilitation Unique capabilities – only FDA and CE-cleared tool offering on-demand mechanical assistance to retrain post-stroke walking function during “push-off” (propulsion) Enhanced gait training – improves training specificity and intensity by promoting power and symmetry in forward propulsion, as well as effective paretic limb advancement Distributed Product – MyoCycle FES Broad indications – widely applicable across the physical and neurological rehabilitation spectrum, with specialized models for in-clinic and at-home use Easy to use – simple setup, with intuitive controls allowing home users and clinicians to rapidly achieve therapeutic goals Proven – FDA cleared to prevent muscle atrophy, reduce spasms, increase blood flow, and increase range of motion

Consolidation Strategy
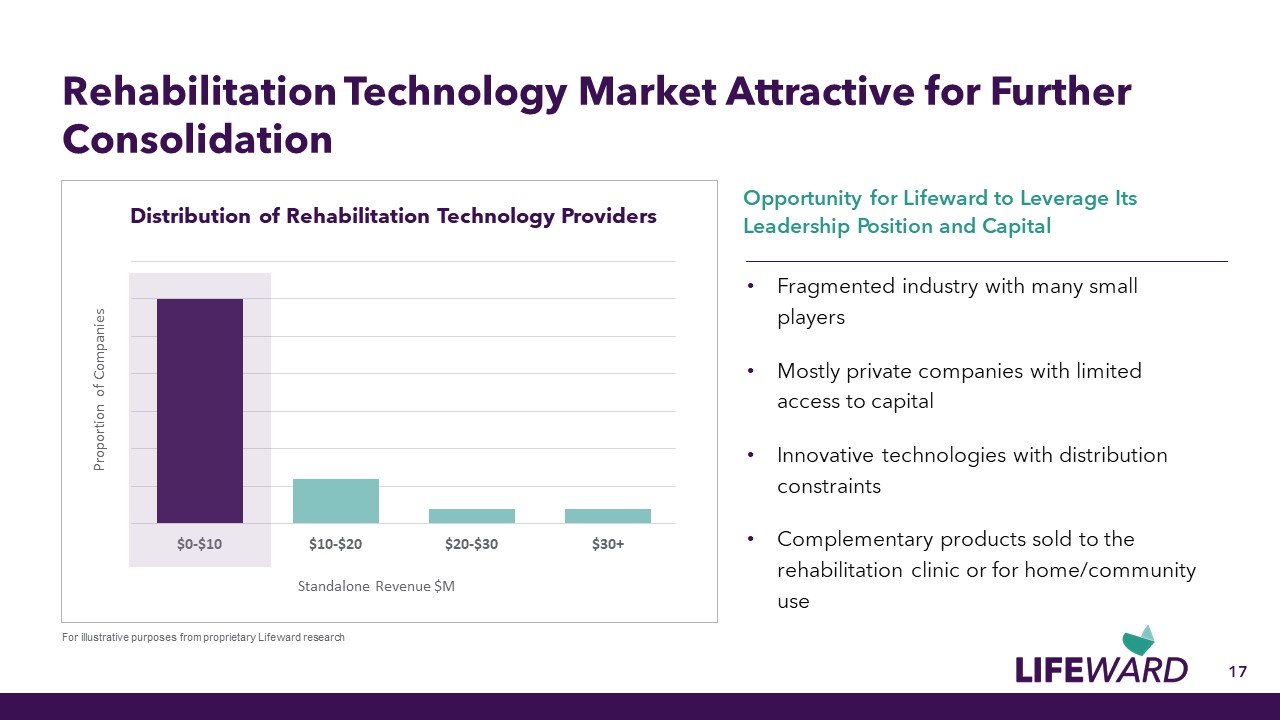
17 Rehabilitation Technology Market Attractive for Further Consolidation Fragmented industry with many small players Mostly private companies with limited access to capital Innovative technologies with distribution constraints Complementary products sold to the rehabilitation clinic or for home/community use Opportunity for Lifeward to Leverage Its Leadership Position and Capital Distribution of Rehabilitation Technology Providers For illustrative purposes from proprietary Lifeward research

Enhances Lifeward’s position as a leader in innovative physical medicine and rehabilitation technologies that enable mobility and wellness Strategy Adds novel technology – Differential Air Pressure – with proven commercial success and potential for further applications and growth Technology Creates comprehensive commercial sales team focusing on clinics and hospitals, as well as sales to individuals for use in the home Scale Combined commercial sales team to capitalize on CMS’ newly-established Medicare reimbursement coverage of exoskeletons for home use Execution Key Attributes of AlterG Acquisition
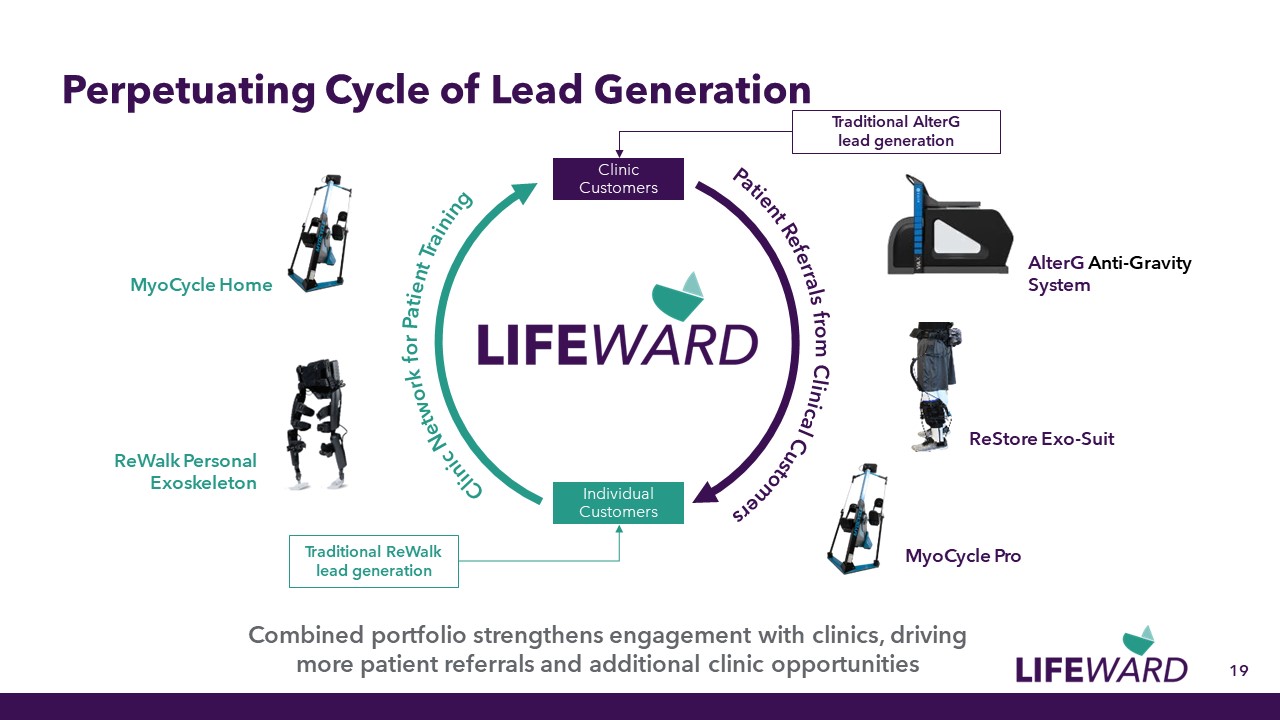
Perpetuating Cycle of Lead Generation 19 AlterG Anti-Gravity System MyoCycle Pro ReStore Exo-Suit MyoCycle Home Clinic Network for Patient Training ReWalk Personal Exoskeleton Patient Referrals from Clinical Customers Combined portfolio strengthens engagement with clinics, driving more patient referrals and additional clinic opportunities Clinic Customers Individual Customers Traditional ReWalk lead generation Traditional AlterG lead generation

Deep and Talented Leadership Team 20 Larry JasinskiChief Executive Officer Mike LawlessChief Financial Officer Charles RemsbergChief Sales Officer Miri ParienteVice President, Operations, Regulatory & Quality David Hexner Vice President, Research & Development Ami KraftExecutive Advisor to the CEO Almog AdarVice President, Finance Jeannine LynchVice President, Strategy & Market Access Kathleen O'DonnellVice President, Marketing & New Business Development Judy KulaVice President, Customer Service & Human Resources

Financial Summary 21 Cash $28 million Cash & cash equivalents (as of December 31, 2023) Debt No debt Cash Runway Path to profitability by 2026 with existing cash balance Well-Positioned for Funding Internal Growth and Integration Strategy

we are partners in energy storage 2024 Milestones & Catalysts Medicare benefit category for Personal Exoskeletons effective January 1, 2024 First Medicare claim paid for a ReWalk Personal Exoskeleton as a medically necessary device Established Medicare payment rate for Personal Exoskeletons New Product Introduction to advance lead in innovation and performance 510k submission for FDA clearance of the next-generation system to improve features and reliability
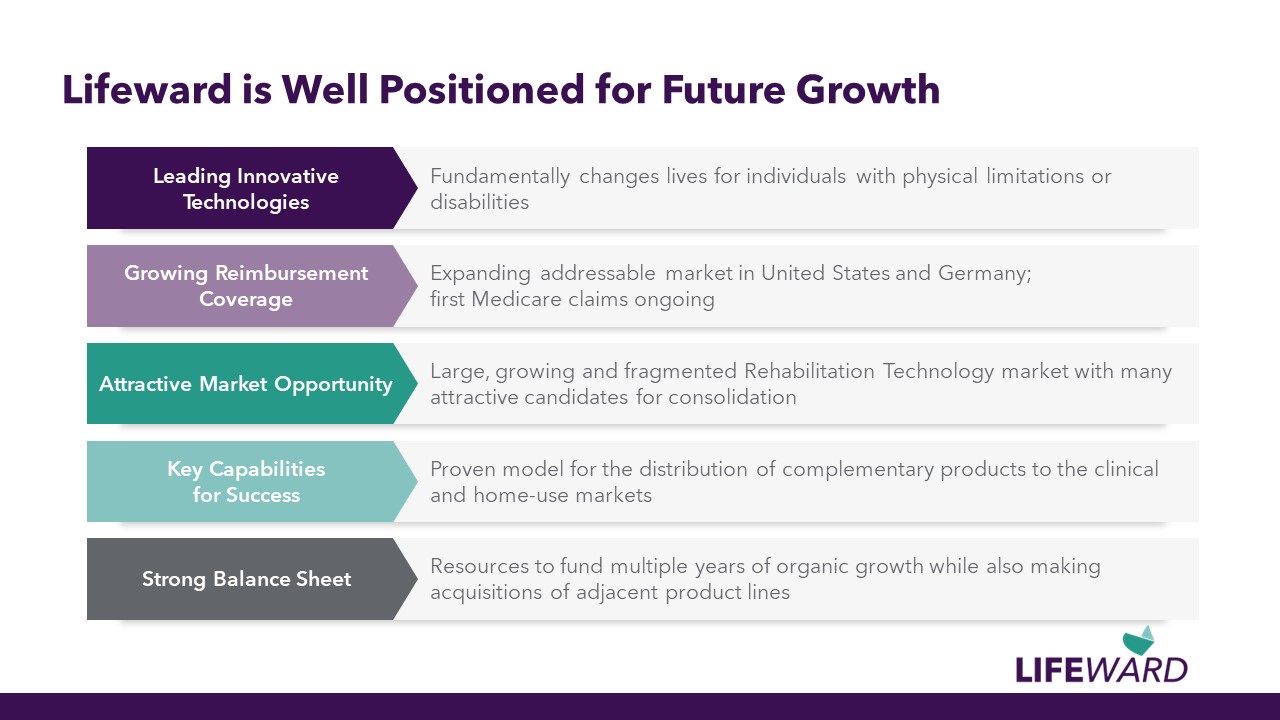
Expanding addressable market in United States and Germany; first Medicare claims ongoing Lifeward is Well Positioned for Future Growth Large, growing and fragmented Rehabilitation Technology market with many attractive candidates for consolidation Fundamentally changes lives for individuals with physical limitations or disabilities Leading Innovative Technologies Proven model for the distribution of complementary products to the clinical and home-use markets Resources to fund multiple years of organic growth while also making acquisitions of adjacent product lines Growing Reimbursement Coverage Attractive Market Opportunity Key Capabilities for Success Strong Balance Sheet

we are partners in energy storage THANK YOU!

we are partners in energy storage References References Tamburella et al. Journal of Neuroengineering and Rehabilitation (2022) Asselin et al., Arch Phys Med Rehab (2021) Gorman et al., J Clin Med (2021) Duddy et al., Sensors (2021) Shackleton et al., J Rehab Med (2019) Juszczak et al., Topics Spin Cord Inj Rehab (2018) Faulkner et al., Journ Spinal Cord Med (2021) Knezevic et al., Arch Phys Med Rehab (2021) ReWalk user survey, 2023 N=41 National Spinal Cord Injury Statistical Center, Facts and Figures at a Glance. Birmingham, AL: University of Alabama at Birmingham, 2021. Estimates based on scaling US statistics for total German and Global population sizes. 31% meet LOI eligibility criteria based on 2020 NSCISC annual report: www.nscisc.uab.edu/public/2020%20Annual%20Report%20-%20Complete%20Public%20Version.pdf Of LOI-eligible, an estimated 30% meet additional eligibility criteria based on aggregate of 50% screen failure rate in literature: https://pubmed.ncbi.nlm.nih.gov/32800962/ plus ReWalk’s historical experience related to additional limiters (e.g. available transportation and time to attend training, motivation, companion availability, etc.) which are not accurately reflected within research populations. 56% of US SCI prevalence market cite Medicare or Medicaid as primary 30% of German Market covered for exoskeleton coverage by insurance contracts

we are partners in energy storage References (cont’d) References (continued) MedPAC 2021 Report: www.medpac.gov/wp-content/uploads/2021/10/mar21_medpac_report_ch9_sec.pdf www.ibisworld.com/industry-statistics/number-of-businesses/physical-therapy-rehabilitation-centers-united-states/ (accessed Jan 2023) Estimates based on scaling US statistics for total German and Global population sizes