Exhibit 99.5
Transforming Immuno - Oncology Using Next - Generation Immune Cell Engagers Corporate Presentation November 2016

Forward - looking statements / safe harbor This presentation and the accompanying oral commentary contain “forward - looking” statements that involve substantial risks and uncertainties. All statements other than statements of historical facts contained in this presentation and the accompanying oral commentary, including statements regarding our future financial condition, business strategy and plans and objectives of management for future operations, are forward - looking statements. In some cases, you can identify forward - looking statements by terminology such as “believe,” “will,” “may,” “estimate,” “continue,” “anticipate,” “intend,” “should,” “plan,” “might,” “approximately,” “expect,” “predict,” “could,” “potentially” or the negative of these terms or other similar expressions. Forward - looking statements appear in a number of places throughout this presentation and the accompanying oral commentary and include statements regarding our intentions, beliefs, projections, outlook, analyses and current expectations concerning, among other things, our ongoing and planned preclinical development and clinical trials, our collaborations and development of our products in combination with other therapies, the timing of and our ability to make regulatory filings and obtain and maintain regulatory approvals for our product candidates our intellectual property position, our collaboration activities, our ability to develop commercial functions, expectations regarding clinical trial data, our results of operations, cash needs, financial condition, liquidity, prospects, future transactions, growth and strategies, the industry in which we operate, the trends that may affect the industry or us and the risks uncertainties and other factors described under the heading “Risk Factors” in Affimed’s filings with the Securities and Exchange Commission. Forward - looking statements involve known and unknown risks, uncertainties, assumptions and other factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward - looking statements. Forward - looking statements represent our management’s beliefs and assumptions only as of the date of this presentation. Except as required by law, we assume no obligation to update these forward - looking statements publicly, or to update the reasons why actual results could differ materially from those anticipated in the forward - looking statements, even if new information becomes available in the future. 2

History of cutting - edge science, innovation , and expertise • Clinical and pre - clinical pipeline based on bi - and trispecific antibodies • Eliminate tumor cells by recruiting NK - cells or T - cells • Partnerships with industry, academic, and advocacy groups • Nasdaq - listed company with 70+ employees located in Heidelberg, Germany (HQ) and affiliate offices in the U.S. (Affimed, Inc.) and in the Czech Republic ( AbCheck s.r.o .) • Raised ~$120 million gross proceeds since September 2014 3
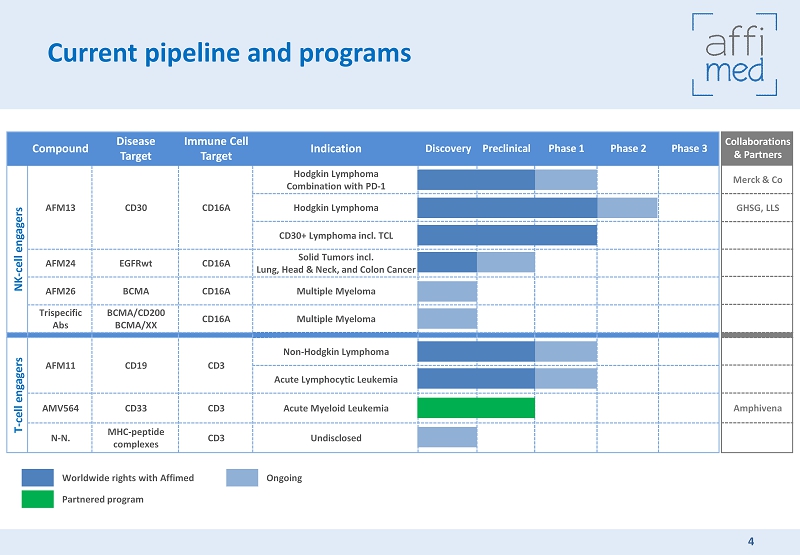
Current pipeline and programs Worldwide rights with Affimed Partnered program Compound Disease Target Immune Cell Target Indication Discovery Preclinical Phase 1 Phase 2 Phase 3 NK - cell engagers AFM13 CD30 CD16A Hodgkin Lymphoma Combination with PD - 1 Hodgkin Lymphoma CD30+ Lymphoma incl. TCL AFM24 EGFRwt CD16A Solid Tumors incl. Lung, Head & Neck, and Colon Cancer AFM26 BCMA CD16A Multiple Myeloma Trispecific Abs BCMA/CD200 BCMA/ XX CD16A Multiple Myeloma T - cell engagers AFM11 CD19 CD3 Non - Hodgkin Lymphoma Acute Lymphocytic Leukemia AMV564 CD33 CD3 Acute Myeloid Leukemia N - N. MHC - peptide complexes CD3 Undisclosed Collaborations & Partners Merck & Co GHSG, LLS Amphivena Ongoing 4
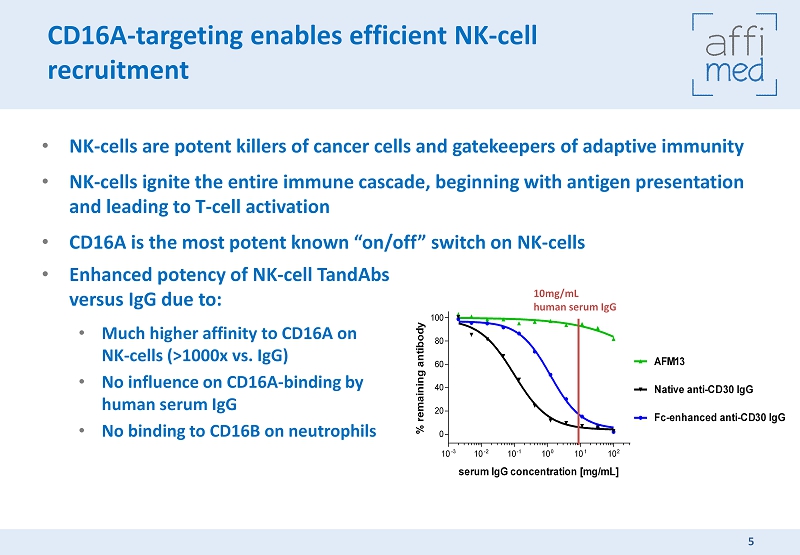
CD16A - targeting enables efficient NK - cell recruitment • NK - cells are potent killers of cancer cells and gatekeepers of adaptive immunity • NK - cells ignite the entire immune cascade, beginning with antigen presentation and leading to T - cell activation • CD16A is the most potent known “on/off” switch on NK - cells 5 10 -3 10 -2 10 -1 10 0 10 1 10 2 0 20 40 60 80 100 Native anti-CD30 IgG Fc-enhanced anti-CD30 IgG AFM13 serum IgG concentration [mg/mL] % r e m a i n i n g a n t i b o d y 10mg/mL human serum IgG • Enhanced potency of NK - cell TandAbs versus IgG due to: • Much higher affinity to CD16A on NK - cells (>1000x vs. IgG) • No influence on CD16A - binding by human serum IgG • No binding to CD16B on neutrophils

AFM13 : A first - in - class CD16A - targeting NK - cell engager • Most advanced NK - cell engager in clinical development • Clinical/PD activity in heavily pretreated HL patients • Tumor shrinkage in 8/13 (62%) and PRs in 3/13 (23%) patients treated with just 4 weekly doses of at least 1.5 mg/kg • Favorable safety profile, offering opportunities for combination with wide range of other drugs • Combination of AFM13 with checkpoint modulators induced crosstalk between innate/adaptive immunity eliciting an integrated immune response (PDX model ) • Highest synergy measured for combination with PD - 1 6 Tumor cell TandAb NK - cell CD16A CD30

AFM13: Clinical development • Phase 1b trial in r/r HL in combination with Merck’s KEYTRUDA® ( pembrolizumab ) initiated in May 2016, recruitment ongoing • Update anticipated by YE/2016 or in Q1/2017 • Phase 2a IST led by the German Hodgkin Study Group (GHSG) in r/r HL was initiated in 2015, recruitment ongoing • Study design currently in the process of being amended • Expansion of clinical activities for AFM13 currently evaluated • CD30 - positive indications, such as TCL, ALCL • Combination with adoptive NK - cell transfer 7

AFM24: Affimed’s first - in - class NK - cell engager targeting solid tumors • EGFRwt/CD16A - specific NK - cell engager • Potential solid tumor indications include lung, H&N, colon cancers • Differentiation from Cetuximab • More potent cytotoxic activity in vitro (low pM ) • Tumor cell killing including cells expressing the proto - oncogene ras • Virtually no competition of NK - cell binding by circulating IgG • Cross - reactive to EGFRwt and CD16A target structures in cynomolgus monkey • GMP - manufacturing in progress • IND - enabling toxicology studies initiated with anticipated update in H1/2017 8 8

AFM26: Affimed’s novel candidate for multiple myeloma targeting BCMA • Therapeutic rationale • Current treatments fail to achieve MRD negativity in majority of multiple myeloma (MM) patients ; most patients eventually relapse • MM is characterized by high M - protein serum levels (up to 170mg/mL) • Competition by serum IgG is known to strongly impair ADCC activity of mAbs • AFM26: BCMA - targeting TandAb introducing a novel MoA • NK - cell binding of candidates unaffected by circulating IgG, indicating potential for NK - cell activation in the presence of M - protein • High affinity to target and NK - cells • Potential benefit for majority of patients, incl. NDMM (+/ - ASCT) and RRMM 9
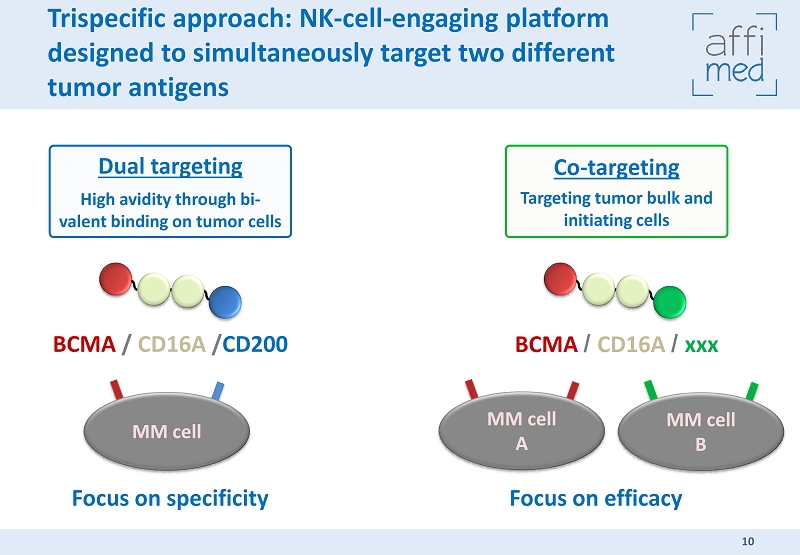
Co - targeting Targeting tumor bulk and initiating cells Dual targeting High avidity through bi - valent binding on tumor cells Trispecific approach: NK - cell - engaging platform designed to simultaneously target two different tumor antigens BCMA / CD16A / CD200 BCMA / CD16A / xxx MM cell MM cell A MM cell B Focus on specificity Focus on efficacy 10

T - cell TandAbs differentiation from other therapies Efficacy • High affinity binding for effective killing of cells with low target expression (AFM11 50 - fold more potent than Blincyto at low T - cell numbers) PD • Significantly lower dosing as compared to IgGs (ng/kg vs. mg/kg) • No receptor saturation required for full activation of T - cells Safety • Very narrow therapeutic window requires careful patient management (e.g. interruption of dosing effective way of resolving side effects) • No non - specific activation of T - cells (no binding to other immune cells e.g. via FcRN or Fc g receptors) 11

• T - cell engaging TandAb targeting CD19/CD3 • Phase 1 dose escalation in NHL patients ongoing • Study amended in late 2015 • New sites opened in the U.S. and planned in Eastern Europe to address slower - than - expected recruitment • Phase 1 dose escalation in r/r ALL patients initiated in Q3/2016 • Study to be conducted in Eastern Europe, Russia and Israel • Next progress update in H1/2017 12 AFM11: High potency, very low COGS

AMV564: A CD33/CD3 T - cell - engaging TandAb developed by Amphivena • High unmet need in AML • Very low cure rates with 5 - year DFS of 15% (>60 yrs ) to 40% (<60 yrs ) • No effective salvage therapies • AMV564 • Potent and selective cytotoxic activity, robust tumor growth inhibition • Corroborative evidence of direct correlation between binding affinity and potency • No non - specific T - cell activation in preclinical models likely due to absent binding to other immune cells (e.g. FcRN , Fc g receptors) • IND approval in July 2016, Amphivena plans to initiate a Phase 1 study • VC - syndicate including Affimed supports clinical development 13 13

Novel platform: TandAbs mediate specific and dose - dependent lysis of MHC - target positive cells • TandAbs identified which specifically recognize target MHC - peptide but not control MHC - peptides • TandAb Candidate • Anti - HLA - A2/peptide T - cell TandAb • Specific killing of endogenous tumor cells only • E xcellent biophysical properties Cell Lines TandAb Candidate Control Abs HLA - A2 Peptide Mean EC 50 [ pM ] + + 236.6 3.8 + + 155.9 98.1 + - no 1.6 + - no 0.6 + - no 1.8 + - no 1.4 - + no 47.0 - + no 1.5 - - no 0.5 - - no 0.2 - - no 40.8 - - no 10.7 - - no 4.8 14

Q3 2016 Cash Flow statement • Cash reach is projected into Q1/2018 15 In thousands of € For the nine months ended September 30, 2016 Cash and Cash equivalents beginning of period 76,740 FX - related changes to Cash and Cash equivalents (655) Net cash used in operating activities ( 25.546 ) Cash used in investing activities ( 13,767 ) Cash and Cash equivalents end of period 35,693 Financial assets * end of period 13,440 Cash and cash equivalents and financial assets* end of period 49,133 * short - term deposits

16 Path forward Maximize value from pipeline and technologies • Leverage AFM13 for CD30 - positive lymphoma • Salvage settings enable rapid clinical development and cost - efficient M&S structure • Investigation of AFM13 both as monotherapy and in combination with anti PD - 1 to reduce development risk • Expand the application of NK - cell platform to solid tumors • Use pipeline and technologies to create value through both next - generation products and partnership opportunities • Develop AFM11 through Phase 2 POC studies • Advance EGFRwt TandAb AFM24 in solid tumors such as lung, head and neck, and colon cancer (EGFRvIII TandAbs AFM21/AFM22 as backup candidates) • Develop TandAb and Trispecific Ab in multiple myeloma • Leverage additional options for AFM13, e.g. in combination with adoptive NK - cells