
A Revolution in Mind February 8, 2017 Exhibit 99.2 A final base shelf prospectus containing important information relating to the securities described in this document has been filed with the securities regulatory authorities in each of the provinces of Canada. A copy of the final base shelf prospectus, any amendment to the final base shelf prospectus and any applicable shelf prospectus supplement that has been filed, is required to be delivered with this document. This document does not provide full disclosure of all material facts relating to the securities offered. Investors should read the final base shelf prospectus, any amendment and any applicable shelf prospectus supplement for disclosure of those facts, especially risk factors relating to the securities offered, before making an investment decision. No securities regulatory authority has expressed an opinion about these securities and it is an offence to claim otherwise.

Legal Disclaimers This presentation contains forward-looking statements and forward-looking information as such terms are defined under applicable U.S. federal securities and Canadian securities legislation. All statements other than statements of historical fact contained in this presentation constitute forward-looking statements and forward-looking information, including, without limitation, statements containing the words “believe”, “may”, “plan”, “should”, “predict”, “potential”, “will”, “estimate”, “continue”, “anticipate”, “intend”, “expect”, “seek”, “mission”, “goal” and similar words, variations, expressions or the negative thereof. Forward-looking statements are necessarily based on estimates and assumptions made by management of the Helius Medical Technologies, Inc. (“Helius”, “we” or the “Company”) in light of experience and perception of historical trends, current conditions and expected future developments, as well as the factors management of the Company believes are appropriate. Forward-looking statements and information in this presentation include but are not limited to statements relating to: the potential results of the Company’s ongoing and planned clinical trials; the Company’s estimate of the size of the potential markets for its products; the estimated clinical, regulatory and commercial milestones and the timelines for achieving such milestones; the Company’s ability to enroll and successfully complete, clinical trials; the expected timelines for patent filings and issuance; the Company’s future manufacturing strategy; the therapeutic benefits, effectiveness and safety of the Company’s product candidates; whether the Company will receive, and the timing and costs of obtaining, regulatory clearances in the U.S., Canada, EU and Australia; regulatory developments and the regulatory environments in which the Company operates; and anticipated trends and challenges in the Company’s business and the markets in which it operates. Such statements reflect management of the Company’s current views with respect to future events and are subject to risks and uncertainties and are necessarily based upon a number of estimates and assumptions that, while considered reasonable by the Company, are inherently subject to significant business, economic, competitive, political and social uncertainties and contingencies. Many factors could cause our actual results, performance or achievements to be materially different from any future results, performance, or achievements that may be expressed or implied by such forward-looking statements, including, among others: risks related to the Company’s limited operating history; the Company being dependent on the ability and expertise of its Chief Executive Officer, Chief Medical Officer and a very limited number of employees; the Company having incurred losses since its inception and its anticipation that it will continue to incur substantial net losses for the foreseeable future and may never achieve or sustain profitability; risks relating to the Company requiring additional financing to carry out its plan of operations; the Company’s independent registered public accounting firm having included an explanatory paragraph relating to the Company’s ability to continue as a going concern in its report on the Company’s audited financial statements for the year ended March 31, 2015, as amended on January 11, 2016; the Company’s failure to maintain effective internal controls over financial reporting; risks related to the Company raising additional capital by issuing securities or through debt financing or licensing arrangements that may cause dilution to existing shareholders, restrict its operations or require the Company to relinquish proprietary rights; risks concerning the Company only having one product candidate, which is still in development, and the Company not having obtained clearance from the United States Food and Drug Administration (the “FDA”), CE Mark, TGA (Australia) or Health Canada with respect thereto; the Company’s dependence on outside scientists and third-party research institutions for its research and development in order to be able to commercialize its product candidates; the risk that if the Company fails to obtain FDA clearance for commercialization of or otherwise fails to ensure that the PoNS™ device is available for purchase by the U.S. Government by December 31, 2017, the Company will be subject to significant risk of loss of data and proprietary rights and a US$2,000,000 contract penalty payable to A&B pursuant to the Strategic Agreement with A&B; the risk that the Strategic Agreement with A&B may be terminated; risks related to the limited market awareness of the Company and its product; risks related to the neuromodulation market being new and growing but undefined; the Company’s PoNS™ technology being a new “untested” form of neurostimulation therapy and the medical community tending to be very conservative in adopting new therapies; risks related to the Company needing to expand its products beyond its single product by commercializing new product candidates, and the Company not being able to do so in a timely fashion and at expected costs, or at all; and development by others of new or improved devices or products that may result in the Company’s present and future products from becoming obsolete. This presentation speaks as of its date and we, our advisors, and our and their affiliates and representatives undertake no obligation to update, revise or correct the information contained herein, except as required by law. This presentation contains industry and market data that we obtained from independent industry publications. We have not independently confirmed this data and, although we believe it is generally reliable, it involves a number of assumptions and limitations, it is inherently imprecise, and you are cautioned not to place undue reliance on it. We have filed a registration statement on Form S-3 (including a base prospectus, Registration No. 333-215286) with the SEC for the offering to which this communication relates. This presentation has been prepared solely for use by prospective investors in connection with a proposed public offering of our Class A Common Stock and contains a summary of selected information to be contained in a prospectus supplement to be filed with the SEC, and the accompanying prospectus. Before you invest, you should read the prospectus supplement, the accompanying prospectus and the information incorporated therein by reference, including the Risk Factors set forth in those materials. When available, you may obtain these documents for free by visiting EDGAR on the SEC website www.sec.gov or by contacting Canaccord Genuity Corp., Brookfield Place, 161 Bay Street, Suite 3100, Toronto, Ontario M5J 2S1, or Raymond James Ltd., 40 King Street West, Suite 5400, Toronto, ON M5H 3Y2.

Over 30 years in the health sciences industry Former CEO at MediMedia Health Marketing Services Former President and CEO at GSW Worldwide Former Director of Neuroscience Marketing at Bristol-Myers Squibb Meet the Helius Management Team Philippe Deschamps President and CEO Chairman and Director 31 years in the health sciences industry Trained surgeon and pioneer of new medical technologies Has helped build several companies including medical technology, research and product-design and medical contract sales organizations Jonathan Sackier Chief Medical Officer Experienced management team with expertise in health sciences and business development 29 years in the health sciences industry Accomplished pharmaceutical/healthcare public company CAO Former Executive Director/group controller at Aptalis Pharmaceuticals Former Chief Operating Officer and CFO MM Pharmaceutical Solutions Joyce LaViscount CFO and COO 17 years in health sciences/financial industry PhD in Experimental Medicine Former Healthcare Analyst at Raymond James Partner in private equity firm advising on healthcare transactions Brian Bapty VP Strategy and Business Development
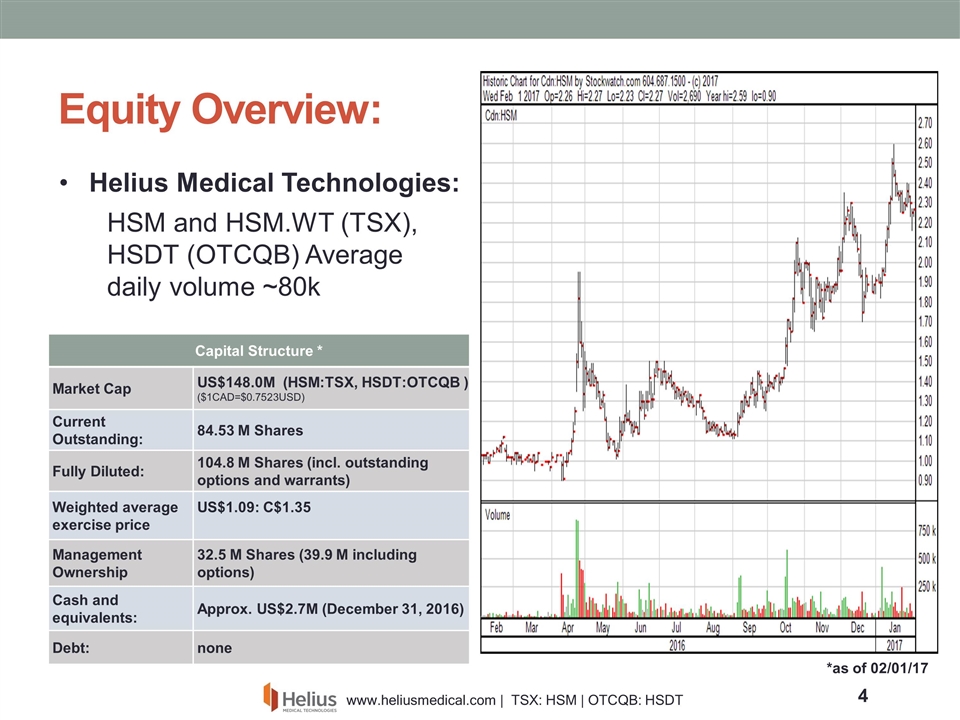
Equity Overview: *as of 02/01/17 Helius Medical Technologies: HSM and HSM.WT (TSX), HSDT (OTCQB) Average daily volume ~80k Capital Structure * Market Cap US$148.0M (HSM:TSX, HSDT:OTCQB ) ($1CAD=$0.7523USD) Current Outstanding: 84.53 M Shares Fully Diluted: 104.8 M Shares (incl. outstanding options and warrants) Weighted average exercise price US$1.09: C$1.35 Management Ownership 32.5 M Shares (39.9 M including options) Cash and equivalents: Approx. US$2.7M (December 31, 2016) Debt: none

Funding to Date TCNL Lab funding: $7.1M ($3.0M NIH grants, $4.1M in cash donations from treated subjects) – 2008-2013 Cash from Private Placement/Prospectus Offerings/Warrants: $7.0M initial investment reverse merger – Q2 2014 $1.0M convertible debenture – Q2 2014 $2.8M non-brokered private placement – Q2&Q3 2015 $7.0M A&B Company (China) strategic investment – Q4 2015 $8.0M US private placement/prospectus offering in Canada – Q2 2016 $1.4M from exercise of warrants– Q2 2016 $27.2M total funding through September 30, 2016 Non-Dilutive Commitment: US Army ($1.8M) $1.8M expense reimbursement, through September 30, 2016, from sole source contract ($3M total commitment)

The Inventors of PoNS™ Technology TCNL Project Directors: Mitchell E. Tyler, Kurt Kaczmarek, Yuri P. Danilov Over 20 years of individual experience in their respective fields of neuroscience, biomedical science and engineering Co-discoverers of the retention effect and neurorehabilitation potential of tongue electrotactile stimulation Recognized experts in electrotactile stimulation Invented core tongue display technology TACTILE COMMUNICATION AND NEUROREHABILITATION LABORATORY (“TCNL”) UNIVERSITY OF WISCONSIN–MADISON Department of Biomedical Engineering Founded in 1992 by a pioneer of neuroplasticity, Dr. Paul Bach-Y-Rita Research center using various areas of science to study the theory and application of applied neuroplasticity, the brain’s ability to reorganize in response to new information, needs, and pathways Research objective to develop solutions for sensory and motor disorders Danilov YP et al. “New Approaches to neurorehabilitation: cranial nerve non-invasive neuromodulation (CN-NINM) technology. SPIE Proceedings. 2014. Tyler ME et al. “Non-invasive neuromodulation to improve gait and chronic multiple sclerosis: a randomized double-blind controlled pilot trial. J. NeuroEngineering and Rehabilitation. 2014. Key Publications
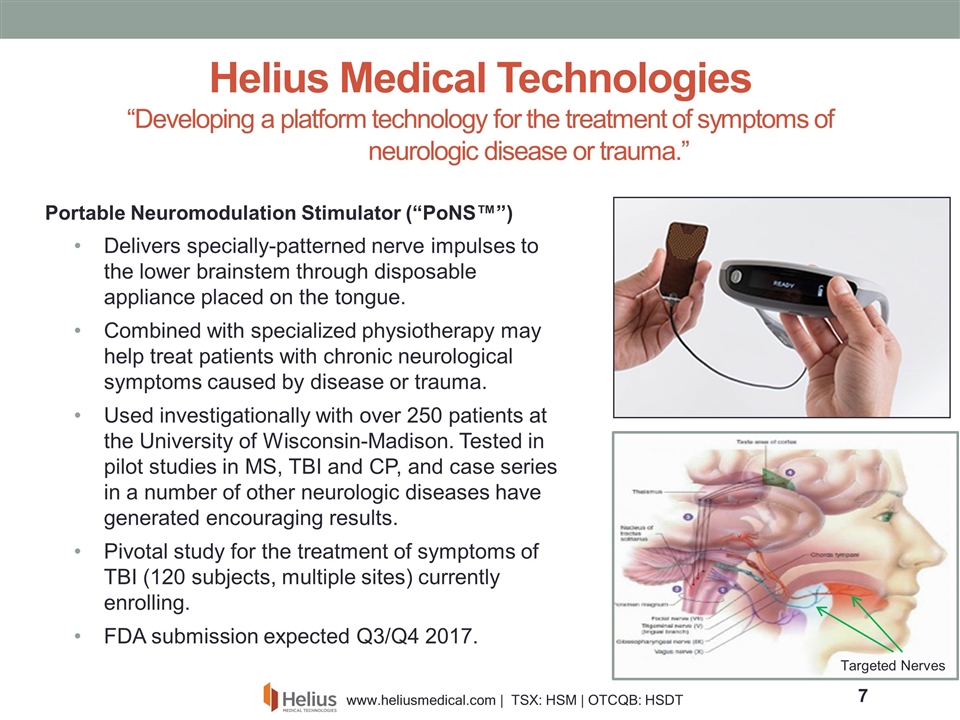
Portable Neuromodulation Stimulator (“PoNS™”) Delivers specially-patterned nerve impulses to the lower brainstem through disposable appliance placed on the tongue. Combined with specialized physiotherapy may help treat patients with chronic neurological symptoms caused by disease or trauma. Used investigationally with over 250 patients at the University of Wisconsin-Madison. Tested in pilot studies in MS, TBI and CP, and case series in a number of other neurologic diseases have generated encouraging results. Pivotal study for the treatment of symptoms of TBI (120 subjects, multiple sites) currently enrolling. FDA submission expected Q3/Q4 2017. Helius Medical Technologies “Developing a platform technology for the treatment of symptoms of neurologic disease or trauma.” Targeted Nerves
Exclusively licensed from inventors (4% royalty): 3 US Medical Method Patents Issued Skin Stimulation + physical therapy = Therapeutic outcome Oral Cavity Stimulation + physical therapy = Therapeutic outcome Oral Cavity Stimulation + cognitive therapy = Therapeutic outcome 5 US Patent Applications Allowed or Pending 3 Allowed US Applications: Treatment of Tinnitus and treatment concepts; Skin + Cognitive Exercise; Stimulation with Pulse Generator (expect to issue 2017) 2 Pending US Applications- filed July 2016: Human Performance Patents owned by Helius (no royalty): 22 US Patents Issued 2 US Patent Applications Allowed: System (Mouthpiece + Controller) including mechanical details of Controller; Methods of Manufacture (expected to issue in 2017) 4 US Patent Applications Pending 1 US Patent Application Forthcoming – to be filed Q1 2017: Mouth-Breathing Features 11 Non-US Design Patents Issued: Europe (1); Canada (7); Russia (3) 3 International Patent Applications Pending 1 Eurasian National Phase Patent Application Pending – filed in 2017: Methods of Providing Physical and Cognitive Therapy Helius patents transferred to CMS (China Medical System Holdings) in strategic sale/license and investment transaction 3 Chinese Design Patents Cornerstone Neuromodulation IP Portfolio
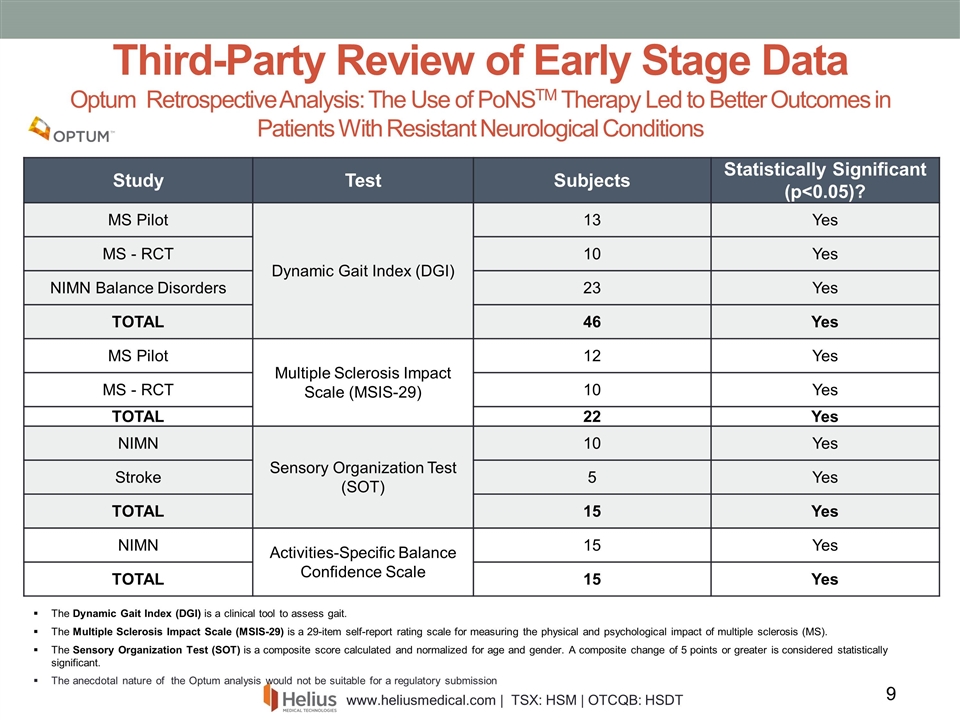
The Dynamic Gait Index (DGI) is a clinical tool to assess gait. The Multiple Sclerosis Impact Scale (MSIS-29) is a 29-item self-report rating scale for measuring the physical and psychological impact of multiple sclerosis (MS). The Sensory Organization Test (SOT) is a composite score calculated and normalized for age and gender. A composite change of 5 points or greater is considered statistically significant. The anecdotal nature of the Optum analysis would not be suitable for a regulatory submission Third-Party Review of Early Stage Data Optum Retrospective Analysis: The Use of PoNSTM Therapy Led to Better Outcomes in Patients With Resistant Neurological Conditions Study Test Subjects Statistically Significant (p<0.05)? MS Pilot Dynamic Gait Index (DGI) 13 Yes MS - RCT 10 Yes NIMN Balance Disorders 23 Yes TOTAL 46 Yes MS Pilot Multiple Sclerosis Impact Scale (MSIS-29) 12 Yes MS - RCT 10 Yes TOTAL 22 Yes NIMN Sensory Organization Test (SOT) 10 Yes Stroke 5 Yes TOTAL 15 Yes NIMN Activities-Specific Balance Confidence Scale 15 Yes TOTAL 15 Yes

Results from MS Pilot Study Multiple Sclerosis (MS) pilot study evaluating the putative additive beneficial effects of PoNS™ device stimulation when combined with physiotherapy* - Trial performed at the Montreal Neurological Institute and Hospital and Concordia University’s PERFORM Center 14 subjects participated in the study: All received physiotherapy, and 7 received stimulation via the PoNS device, while 7 were in the sham group (no stimulation) After 14 weeks, the study was un-blinded and subjects in the sham arm were crossed over onto an active device for a further 12 weeks of therapy Subjects were assessed for changes in a number of physical, physiologic, and psychologic endpoints including: balance (SOT score), gait (DGI score) and physiological changes related to neuroplasticity (BOLD signal) as measured by functional MRI. * See appendix
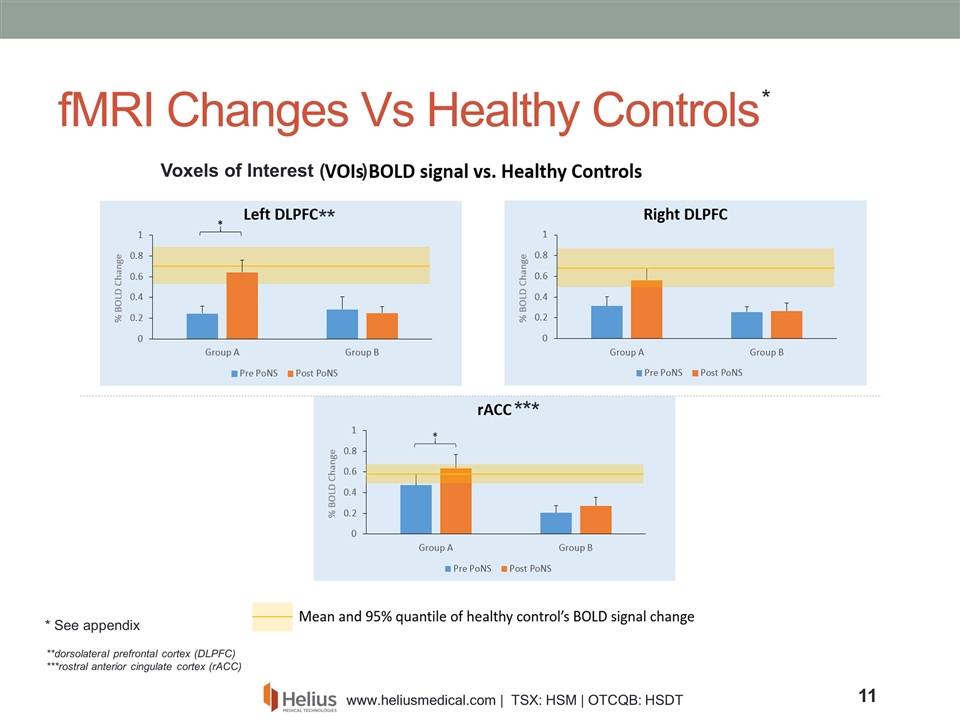
fMRI Changes Vs Healthy Controls Active Arm Active Arm Active Arm Placebo Arm Placebo Arm Placebo Arm Voxels of Interest ( ) **dorsolateral prefrontal cortex (DLPFC) ***rostral anterior cingulate cortex (rACC) * * * * * * * See appendix
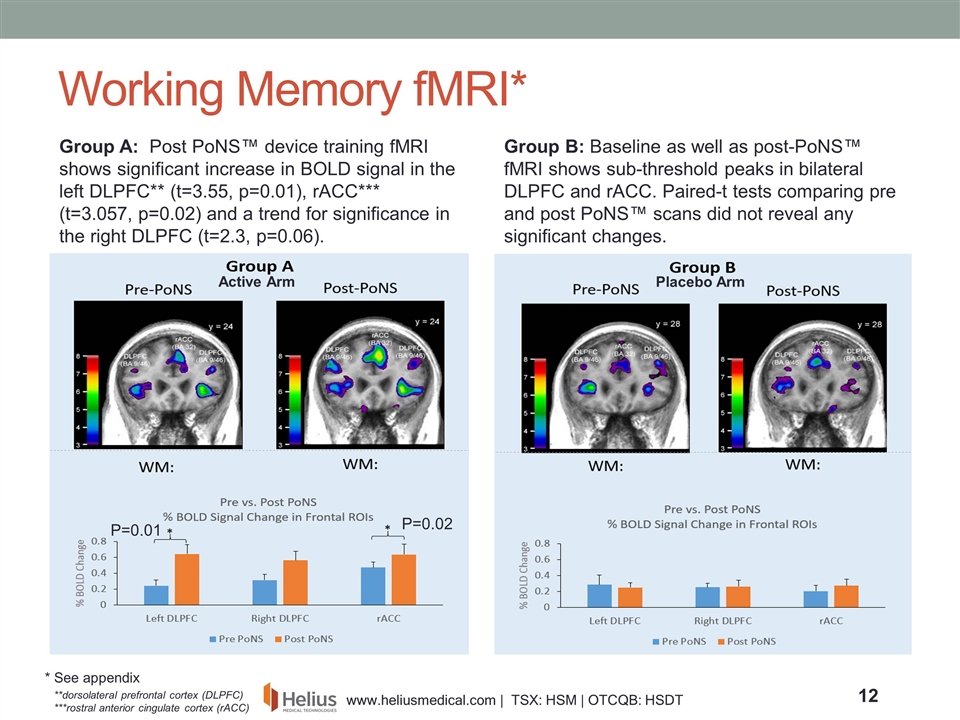
Active Arm Placebo Arm Group A: Post PoNS™ device training fMRI shows significant increase in BOLD signal in the left DLPFC** (t=3.55, p=0.01), rACC*** (t=3.057, p=0.02) and a trend for significance in the right DLPFC (t=2.3, p=0.06). Working Memory fMRI* **dorsolateral prefrontal cortex (DLPFC) ***rostral anterior cingulate cortex (rACC) Group B: Baseline as well as post-PoNS™ fMRI shows sub-threshold peaks in bilateral DLPFC and rACC. Paired-t tests comparing pre and post PoNS™ scans did not reveal any significant changes. P=0.01 P=0.02 * See appendix
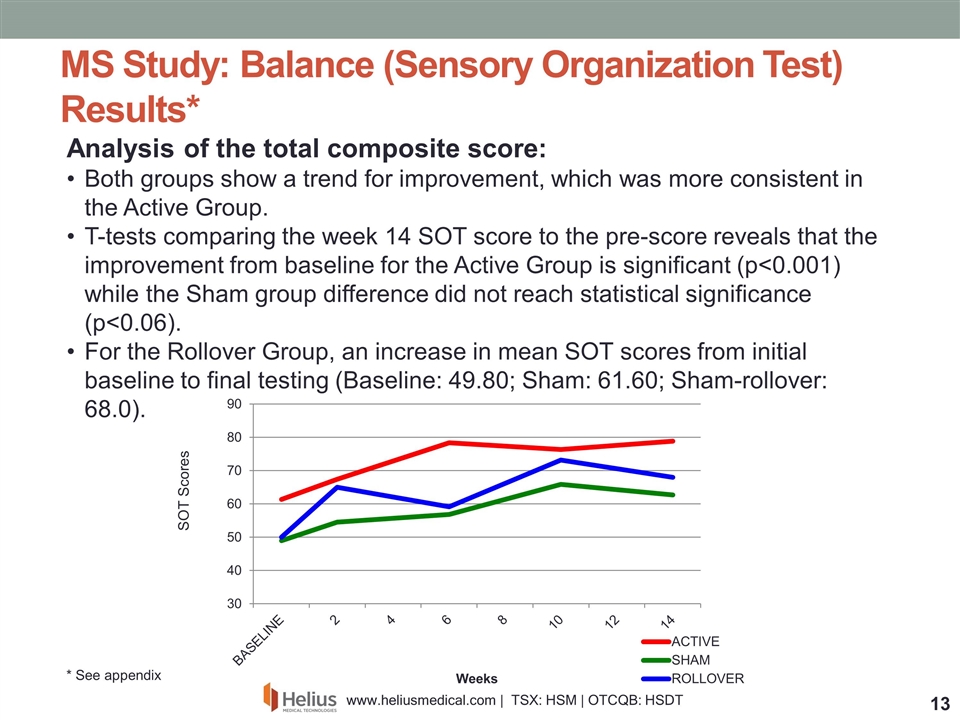
Analysis of the total composite score: Both groups show a trend for improvement, which was more consistent in the Active Group. T-tests comparing the week 14 SOT score to the pre-score reveals that the improvement from baseline for the Active Group is significant (p<0.001) while the Sham group difference did not reach statistical significance (p<0.06). For the Rollover Group, an increase in mean SOT scores from initial baseline to final testing (Baseline: 49.80; Sham: 61.60; Sham-rollover: 68.0). MS Study: Balance (Sensory Organization Test) Results* SOT Scores * See appendix
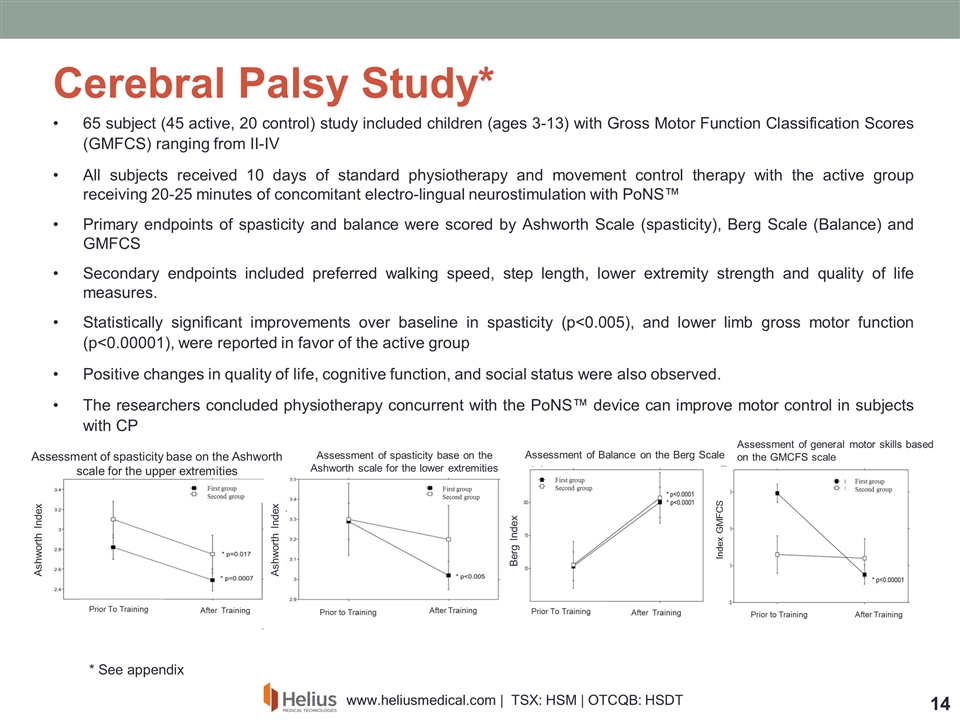
65 subject (45 active, 20 control) study included children (ages 3-13) with Gross Motor Function Classification Scores (GMFCS) ranging from II-IV All subjects received 10 days of standard physiotherapy and movement control therapy with the active group receiving 20-25 minutes of concomitant electro-lingual neurostimulation with PoNS™ Primary endpoints of spasticity and balance were scored by Ashworth Scale (spasticity), Berg Scale (Balance) and GMFCS Secondary endpoints included preferred walking speed, step length, lower extremity strength and quality of life measures. Statistically significant improvements over baseline in spasticity (p<0.005), and lower limb gross motor function (p<0.00001), were reported in favor of the active group Positive changes in quality of life, cognitive function, and social status were also observed. The researchers concluded physiotherapy concurrent with the PoNS™ device can improve motor control in subjects with CP Cerebral Palsy Study* Assessment of spasticity base on the Ashworth scale for the upper extremities Assessment of spasticity base on the Ashworth scale for the lower extremities Prior To Training Prior to Training After Training After Training Prior To Training Prior to Training After Training After Training Assessment of Balance on the Berg Scale Assessment of general motor skills based on the GMCFS scale Ashworth Index Ashworth Index Index GMFCS Berg Index First group Second group First group Second group First group Second group First group Second group * See appendix
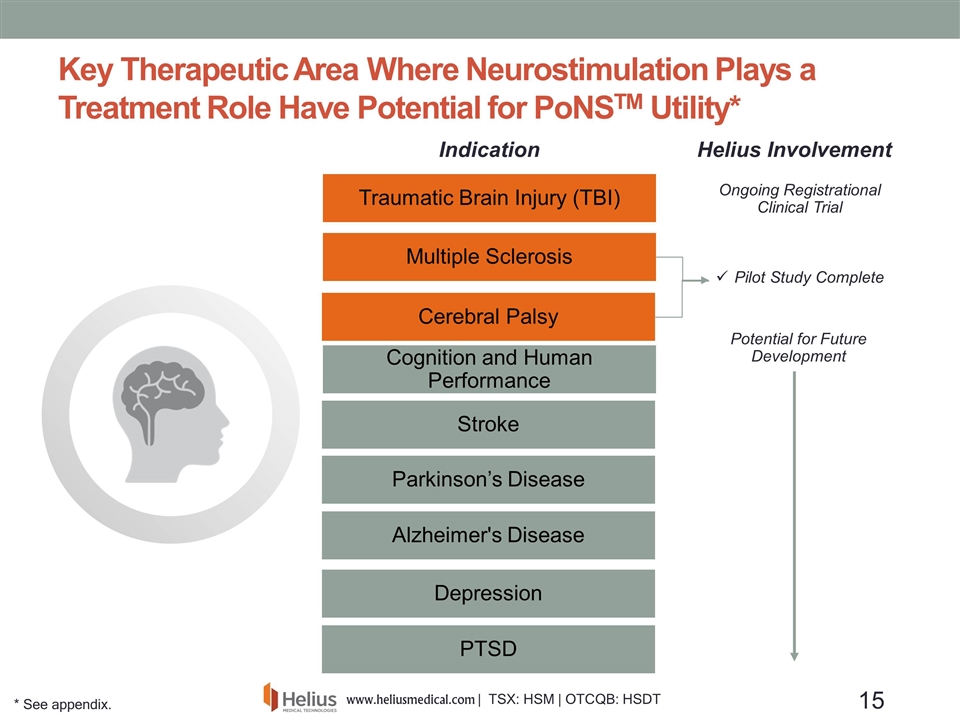
Key Therapeutic Area Where Neurostimulation Plays a Treatment Role Have Potential for PoNSTM Utility* Multiple Sclerosis Traumatic Brain Injury (TBI) Parkinson’s Disease Stroke Alzheimer's Disease Depression PTSD Cerebral Palsy Helius Involvement Indication Pilot Study Complete Ongoing Registrational Clinical Trial Potential for Future Development Cognition and Human Performance * See appendix.
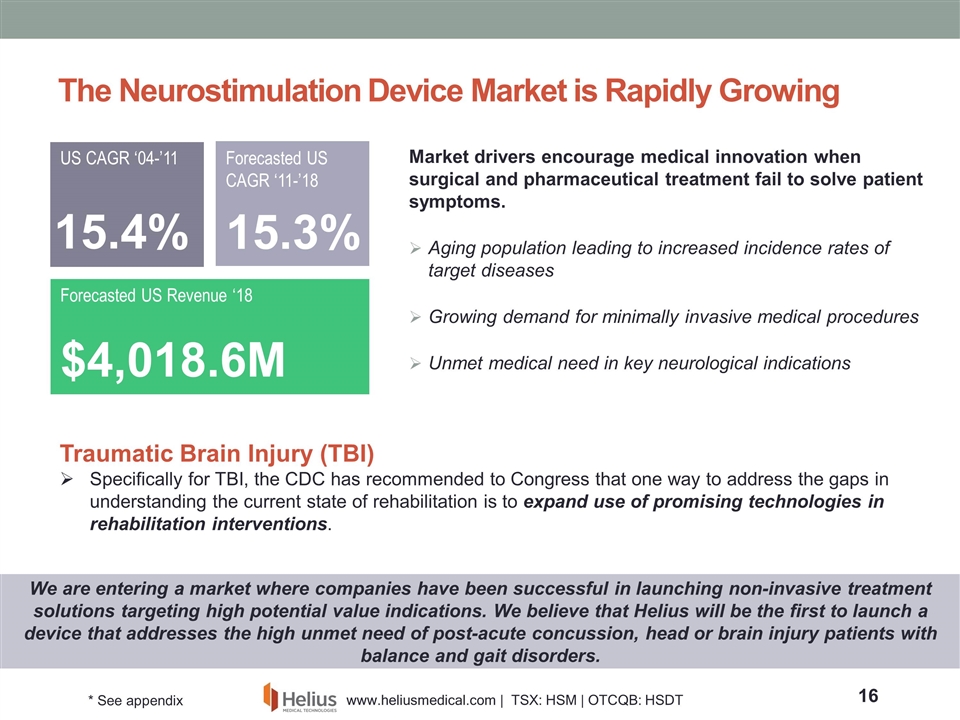
The Neurostimulation Device Market is Rapidly Growing Market drivers encourage medical innovation when surgical and pharmaceutical treatment fail to solve patient symptoms. Aging population leading to increased incidence rates of target diseases Growing demand for minimally invasive medical procedures Unmet medical need in key neurological indications $4,018.6M Forecasted US Revenue ‘18 US CAGR ‘04-’11 15.4% Forecasted US CAGR ‘11-’18 15.3% We are entering a market where companies have been successful in launching non-invasive treatment solutions targeting high potential value indications. We believe that Helius will be the first to launch a device that addresses the high unmet need of post-acute concussion, head or brain injury patients with balance and gait disorders. Traumatic Brain Injury (TBI) Specifically for TBI, the CDC has recommended to Congress that one way to address the gaps in understanding the current state of rehabilitation is to expand use of promising technologies in rehabilitation interventions. * See appendix

30,000/year active duty soldiers withTBI³ 200,000 retired soldiers diagnosed with TBI4 20-30% of new cases result in chronic symptoms5 Traumatic Brain Injury (TBI) Military Athletic / Civilian Common Types of TBI due to Military Activity: Explosive blast injury Overpressure Penetrating injury Diffuse axonal injury 2.1 million people with balance disorder related to non severe TBI² Causes of Civilian TBI: Blunt trauma Motor vehicle accident Sports related injury Assaults 1.7M new cases of TBI reported in U.S. each year6 20-30% of new cases result in chronic symptoms5 3.2 - 5.3M living with TBI related disability7 1,2,3,4,5,6,7 see appendix

Funding From U.S. Army Medical Research and Material Command CRADA with the U.S. Army Medical Research and Material Command effective February 2013 U.S. Army commits non-dilutive funding and resources for PoNS™ research $1.8M+ in expense reimbursement contributed to the project to date U.S. Army provides regulatory support, facilities and personnel as needed December 2015 modification extends CRADA through December 2017 Expands PoNS™ research into fully-funded tinnitus, PTSD, sleep disturbances and pain studies if the initial TBI trial results are positive Sole Source Cost-Share Contract executed July 2015 for TBI Trial Significant financial support for TBI clinical and registrational trial Helius sponsor of regulatory and clinical development

Regulatory Pathway FDA deemed the study of the PoNSTM for mild-moderate TBI a ‘non-significant risk (NSR) device study’ under the IDE regulations Assessed the study as not posing a significant risk to human subjects FDA guidance points to 120-day regulatory review upon submission for de novo clearance for Class II FDA indicated that a de novo request for classification into Class II for the mild-moderate TBI indication would be an appropriate path to seek marketing authorization Balance disorder related to non-severe TBI FDA reviewed and provided feedback on the registrational trial protocol Primary endpoint is improvement in balance at week 5 as measured by Sensory OrganizationTest (SOT) Concurrent to FDA filing, seeking EU CE Mark, Health Canada MDL and TGA approval ISO 13485 received in December 2016 from LRQA an independent organization to review companies quality systems

Clinical Study Title A double-blind, randomized, sham-controlled study of the safety and effectiveness of the PoNS™ device for cranial nerve noninvasive neuromodulation (“CN-NINM”) training in subjects with a chronic balance deficit due to mTBI. Indication Chronic balance deficit due to non-severe TBI Start Date August 2015 Expected Completion Q2:17 Description Helius as sponsor launched a Pivotal Phase III clinical trial in conjunction with US Army Medical Research and Material Command at: Montreal NeuroFeedback Centre (Montreal, QB) Oregon Health and Science University (Portland, OR) Orlando Regional Medical Center (Orlando, FL) HealthTech Connex (Surrey, BC) VCU (Richmond, VA) MedStar National Rehabilitation Center (Washington, D.C.) University of Wisconsin, Madison (February 2017) Patient Enrollment 120 patient double-blind, active control study Primary endpoint is improvement in chronic balance deficit at 5 weeks Long Term Treatment Study (Fully Enrolled) to end on May 28, 2017 Tactile Communication Neurorehabilitation Laboratory at University of Wisconsin-Madison Sponsored by US Army 44 patients (active/sham; 14-weeks active treatment, 12-week washout) PoNS™ Ongoing Clinical Trial in TBI

Manufacturing Progress Class II, 510k FDA submissions largely focus on quality systems and design and manufacturing history files Current expected FDA submission: Q3/Q4 2017 We expanded our development and manufacturing capabilities by contracting with Cambridge Consulting to: assume responsibility for DV and documentation support for FDA submission assist in identification of, and transition to, scale manufacturer Ximedica has agreed to continue with manufacturing our clinical and testing supply for FDA submission and potentially early commercialization devices

(1) Current plan based on availability of funding, among other factors Anticipated PoNS™ Clinical Milestones Pre-clinical Pilot Study Begin FDA Reg. Trial Complete FDA Reg. Trial Submit FDA Filing Obtain Clearance/ Approval PoNS™ 4.0 Device | Cranial Nerve Non-Invasive Neuromodulation + Physical Therapy CLINICAL STAGE PROGRAMS Traumatic Brain Injury Q3:15 Q2:17 Q3/4:17 Q1:18 Pre-clinical Pilot Study Begin FDA Reg. Trial Complete FDA Reg. Trial Submit FDA Filing Obtain Clearance/ Approval Multiple Sclerosis (1) Q2:17 Q2:18 Q3:18 Q4:18 PoNS™ 4.0 Device | Cranial Nerve Non-Invasive Neuromodulation + Exercise Confirmatory Study CLINICAL STAGE PROGRAMS Expanded Protocol Validate Endpoint Cognition related neurological disease pilot (1) Traumatic Brain Injury Q3:15 Q2:17 Q3/Q4:17 Q1:18 Cognition Q1:17 Q2:17 Q3/Q4:17 Cognition (no FDA required) Q1:17 Q2:17 N/A N/A Multiple Sclerosis Q2:17 Q2:18 Q3:18 Q4:18
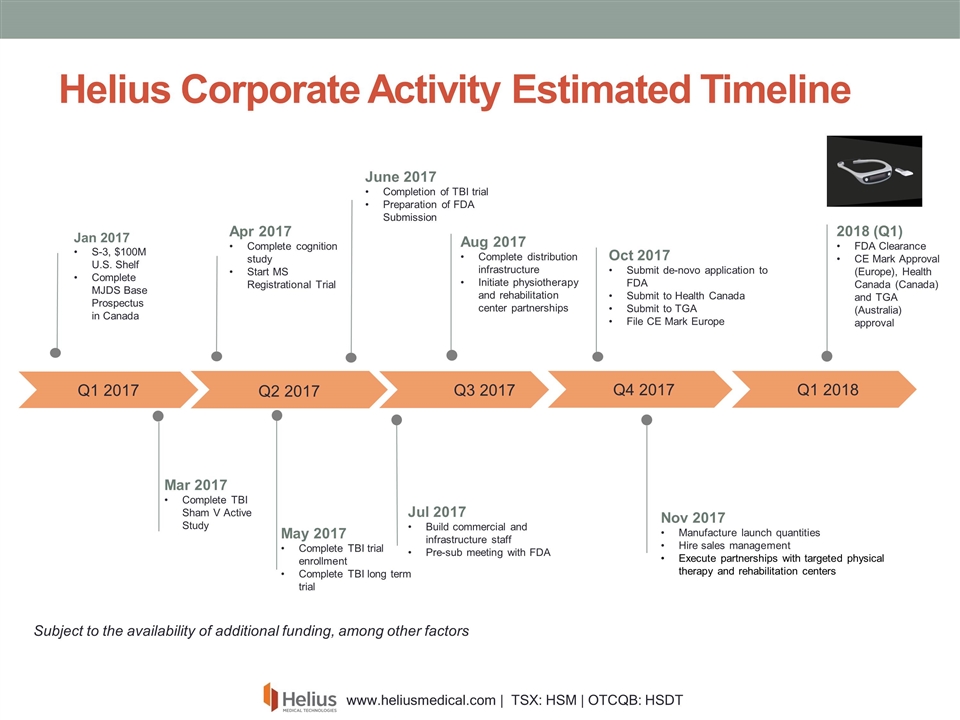
Aug 2017 Complete distribution infrastructure Initiate physiotherapy and rehabilitation center partnerships Helius Corporate Activity Estimated Timeline Q1 2017 Jan 2017 S-3, $100M U.S. Shelf Complete MJDS Base Prospectus in Canada Mar 2017 Complete TBI Sham V Active Study Apr 2017 Complete cognition study Start MS Registrational Trial June 2017 Completion of TBI trial Preparation of FDA Submission May 2017 Complete TBI trial enrollment Complete TBI long term trial Q2 2017 Q3 2017 Jul 2017 Build commercial and infrastructure staff Pre-sub meeting with FDA Nov 2017 Manufacture launch quantities Hire sales management Execute partnerships with targeted physical therapy and rehabilitation centers Oct 2017 Submit de-novo application to FDA Submit to Health Canada Submit to TGA File CE Mark Europe Q4 2017 2018 (Q1) FDA Clearance CE Mark Approval (Europe), Health Canada (Canada) and TGA (Australia) approval Q1 2018 Subject to the availability of funding Subject to the availability of additional funding, among other factors

Helius Medical Technologies, Inc. │41 University Drive, Suite 400 │Newtown, PA 18940 T: 215 809-2018 │E: info@heliusmedical.com │W: www.heliusmedical.com Investor Contact: Brian Bapty T: 604 652-3950 bbapty@heliusmedical.com

Appendix and References

Scientific Advisory Board Jonathan Sackier, M.D., Chairman Scientific Advisory Board Ron Alterman, M.D., M.B.A. Harvard professor Neurosurgeon at Beth Israel (BIDMC) Expertise in movement rehabilitation Carl Hauser, M.D. Director of Trauma at BIDMC Visiting professor of Surgery at Harvard Medical School Scott Parazynski, M.D. Former NASA astronaut Inventor/leader in the medical device/research fields Catherine Cho, M.D. MSCR Assistant Professor in the Department of Neurology at The Icahn School of Medicine at Mount Sinai Jennifer Sweet, M.D. Department of Neurological Surgery University Hospitals Case Medical Center D. James Surmeier, M.D. Chair of the Department of Physiology and Director of Parkinson’s Disease Research Center at Northwestern University Reggie Edgerton, M.D., Ph.D Professor in the Departments of Neurobiology, Integrative Biology and Physiology and Neurosurgery at UCLA Member of the Brain Research Institute Rick Celebrini, Ph.D Physiotherapist, Founder of Fortius Institute, Retired Canadian professional soccer player Canadian Medical team member at 3 Olympic games Head of Sports Medicine and Science for the Vancouver Whitecaps FC Gale Pollock, R.N., Former Commander of the US Army Medical Command Acting Surgeon General of the Army Fellow at the American College of Healthcare Executives, American Academy of Nursing and National Board of Corporate Directors.

Governance – Board of Directors Philippe Deschamps Chairman of the Board, Company CEO Vice Admiral Ed Straw (retired) Director Former head of the Defense Logistics Agency at DOD Blane Walter Director Partner at Talisman Capital; Former Chairman CEO, InVentiv Health Inc. Dr. Huaizheng Peng Director General Manager, International Operations for China Medical Systems Mitch Tyler Director Co-inventor of the PoNSTM device Tom Griffin Director, Chair of the Audit Committee

References Slide 10: Results from MS Pilot Study Helius press release November 15, 2015. Slide 11/12: Results from MS Pilot Study American Congress of Rehabilitation Medicine (ACRM) has accepted a submission from Helius for a panel discussion – “PoNS™ Therapy: non-invasive investigational cranial nerve neuromodulation to augment therapeutic interventions” – at ACRM’s 93rd Annual Conference (October 30 to November 4, 2016, in Chicago, IL) In January 2017, we received confirmation that the manuscript “Non-invasive tongue stimulation combined with intensive cognitive and physical rehabilitation induces neuroplastic changes in patients with multiple sclerosis: a multimodal neuroimaging study” was accepted for publication in the Journal: Multiple Sclerosis Journal: Experimental, Translational and Clinical. Publication data not set. Slide 13: Results from MS Pilot Study In January 2017, we received confirmation that the manuscript “Non-invasive tongue stimulation combined with intensive cognitive and physical rehabilitation induces neuroplastic changes in patients with multiple sclerosis: a multimodal neuroimaging study” was accepted for publication in the Journal: Multiple Sclerosis Journal: Experimental, Translational and Clinical. Publication data not set. Slide 14: Cerebral Palsy Study Published (in Russian ) “Journal of Restorative Medicine and Rehabilitation” (http://www.vvmr.ru/). Results of the study were presented in an oral session at the International Conference for Innovation in Angio-Neurology held in Moscow on September 23-24, 2016 (http://www.altaastra.com/2016/07/angioneurology), (certified English translation available) Company Press Release Sept 6, 2016

References Slide 18: Traumatic Brain Injury (“TBI”) Finkelstein E, Corso P, Miller T and Associates. The Incidence and Economic Burden of Injuries in the United States. New York (NY): Oxford University Press; 2006. Addressable market: 5.3 million people with chronic disability multiplied by 40% having a balance disorder tied to TBI. http://dvbic.dcoe.mil/dod-worldwide-numbers-tbi http://www.ncsl.org/documents/statefed/health/TBI_Vets2013.pdf http://www.msktc.org/tbi/factsheets/Balance-Problems-After-Traumatic-Brain-Injury http://www.cdc.gov/traumaticbraininjury/pdf/BlueBook_factsheet-a.pdf http://www.cdc.gov/traumaticbraininjury/pdf/TBI_Report_to_Congress_Epi_and_Rehab-a.pdf Slide 16:The Neurostimulation Device Market is Continuously and Rapidly Growing GBI Research: Neurostimulation Devices Market to 2018; https://www.cdc.gov/traumaticbraininjury/pdf/tbi_report_to_congress_epi_and_rehab-a.pdf Slide 15:Key therapeutic Areas Where Neurostimulation Plays a Treatment Role Have Potential for PoNSTM Utility Multiple Sclerosis - http://www.nationalmssociety.org/About-the-Society/MS-Prevalence TBI - http://www.cdc.gov/traumaticbraininjury/pdf/TBI_Report_to_Congress_Epi_and_Rehab-a.pdf Parkinson’s - http://www.pdf.org/en/parkinson_statistics Stroke - http://www.cdc.gov/stroke/facts.htm Alzheimer’s - https://www.alz.org/downloads/facts_figures_2014.pdf Depression - http://www.nimh.nih.gov/health/statistics/prevalence/major-depression-among-adults.shtml - http://www.cdc.gov/nchs/fastats/depression.htm PTSD - http://www.adaa.org/about-adaa/press-room/facts-statistics - http://www.ptsd.va.gov/professional/PTSD-overview/epidemiological-facts-ptsd.asp ADHD - http://www.adhd-institute.com/burden-of-adhd/epidemiology/ - http://www.cdc.gov/nchs/fastats/adhd.htm - http://www.ncbi.nlm.nih.gov/pubmed/16585449 Chronic Pain - http://www.painmed.org/patientcenter/facts_on_pain.aspx




























