Exhibit 99.1

BiondVax Pharmaceuticals Ltd Company Presentation BiondVax Pharmaceuticals Ltd. | Jerusalem, Israel | www.biondvax.com www.biondvax.com March 2021

S AFE H ARBOR S TATEMENT This presentation is not a prospectus or offer of securities for subscription or sale in any jurisdiction. All statements in this communication, other than those relating to historical facts, are "forward - looking statements" within the meaning of the United States Private Litigation Reform Act of 1995 . You can identify forward - looking statements by terms including ‘‘anticipates,’’ ‘‘believes,’’ ‘‘could,’’ ‘‘estimates,’’ ‘‘expects,’’ ‘‘intends,’’ ‘‘may,’’ ‘‘plans,’’ ‘‘potential,’’ ‘‘predicts,’’ ‘‘projects,’’ ‘‘should,’’ ‘‘will,’’ ‘‘would,’’ and similar expressions intended to identify forward - looking statements . These forward - looking statements relate to our business and financial performance and condition, as well as our plans, strategies, objectives and expectations for our business, operations and financial performance and condition . However, these forward - looking statements are not guarantees of future performance and are subject to a number of assumptions, involve known and unknown risks, uncertainties and other factors, many of which are beyond our control, that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward - looking statements . Important factors that could cause actual results to differ materially from our expectations include, among others : the risk that we may not be able to secure capital on attractive terms, if at all ; the risk that we are currently exploring new business strategies after our M - 001 universal influenza vaccine candidate failed to meet the trial’s primary and secondary efficacy endpoints and that we may not execute a strategic alternative to M - 001 ; the risk that the European Investment Bank may accelerate the loans under its finance contract with BiondVax ; risks relating to the COVID - 19 (coronavirus) pandemic ; our ability to acquire rights to additional product opportunities ; our ability to enter into collaborations on terms acceptable to us or at all ; timing of receipt of regulatory approval of our manufacturing facility in Jerusalem, if at all or when required ; and the risk that drug development involves a lengthy and expensive process with uncertain outcomes . More detailed information about the risks and uncertainties affecting the Company is contained under the heading “Risk Factors” in our prospectus supplement, which is available on the SEC’s website, www . sec . gov . and in the Company’s periodic filings with the SEC . Readers are urged to carefully review and consider the various disclosures made in the Company’s SEC reports, which are designed to advise interested parties of the risks and factors that may affect its business, financial condition, results of operations and prospects . These forward - looking statements speak only as of the date of this presentation, and we assume no obligation to update or revise these forward - looking statements for any reason, whether as a result of new information, future events or otherwise, except as required by law .

A BOUT B IOND V AX About BiondVax Pharmaceuticals (NASDAQ: BVXV) is a biopharmaceutical company focused on development, manufacturing and ultimately commercializing products for the prevention and treatment of infectious diseases and related illnesses. Experience Vaccine Development Founded 2005 . Extensive experience in vaccine development from preclinical through eight clinical trials Phase 3 clinical trial for influenza vaccine candidate in over 12,400 participants, 105 sites and seven countries While Phase 3 did not meet its primary efficacy endpoint, it was completed on time and on budget in the face of the growing threat of COVID - 19 Assets Leadership, Capital & Manufacturing Big Pharma experienced leadership team including Chairman (Alexion), CEO (Novartis and GSK), Board director (BMS), COO (Novartis) €24m European Investment Bank (EIB) non - dilutive financing agreement, the first such EIB agreement with a non - EU country >$15m cash - on - hand State - of - the - art GMP vaccine manufacturing facility in Jerusalem Activities Pipeline development Pursuing opportunities in the infectious disease space, including exploring several alternatives towards development of a robust multi - asset pipeline

B IOND V AX ’ S M ISSION AND V ISION About To become a globally trusted and preferred bio - pharmaceutical company known for fast product development of highly innovative products, offering a reliable supply of high - quality and effective prophylactic and therapeutic products for infectious diseases and related illnesses. VISION: To help build a healthier and happier world by developing, manufacturing and ultimately commercializing products for the prevention and treatment of infectious diseases and related illnesses. MISSION:
CSO & CMO ▪ PhD (Weizmann Institute of Science) ▪ Biotechnology General Ltd. ▪ Co - invented and guided BiondVax’s original vaccine candidate through 8 clinical trials including pivotal Phase 3 CFO ▪ BA Business (College of Administration) ▪ MBA (Bar Ilan University) ▪ Certified Public Accountant (CPA) ▪ Glycominds Ltd. (VP Finance) ▪ Menorah Capital Markets (Comptroller) M ANAGEMENT CEO ▪ MSc. Biotechnology Engineering ( Ben Gurion University of the Negev) ▪ MBA, Finance and Health Care Mgmt. (The Wharton School) ▪ NeuroDerm (R&D) ▪ Novartis Vaccines (Global Supply Chain) ▪ GSK Vaccines (Global Engineering) Amir Reichman MSc, MBA Tamar Ben - Yedidia PhD Uri Ben - Or CPA, MBA COO ▪ BSc. Engineering ( Afeka Tel Aviv College of Engineering) ▪ MBA (Open University of Israel) ▪ Principal bioprocess engineer ▪ Novartis (Technical Project Manager - Process) Elad Mark BSc Engineering, MBA Director of Business Development & IR ▪ Hon. BSc. (University of Toronto) ▪ MBA (Ben Gurion University of the Negev) ▪ Accenture (Business Management Consultant) ▪ BioData Ltd. (Marketing Manager) Joshua Phillipson Hon. BSc, MBA Leadership

B OARD OF D IRECTORS Leadership CEO NeuroDerm Ltd (Senior Scientist), Novartis Vaccines USA (R&D and Global Supply chain) , GSK Vaccines Belgium (Global Supply Chain and Global Engineering) Mr. Amir Reichman Director Credit Suisse First Boston (Investment Banking), Private equity and venture capital funds (Founder) Mr. Isaac Devash MBA Chairman of the Board Aentib Group (Managing Director), Pluristem (Director). Founder, director, chairman, and/or investor in over twenty biotech companies Mr. Mark Germain Director ID Biomedical (CSO), Intellivax (Founder), Walter Reed General Hospital (Consultant) Dr. George Lowell MD External Director Gamida Cell Ltd. (Nasdaq: GMDA) (President, CEO, Director), Denali Ventures LLC (VP) Dr. Yael Margolin PhD Director BioLineRx (CEO, Director), OurCrowd (Partner), Clil Medical (CEO), Vital Spark (CEO), Kitov Pharmaceuticals (Co - founder, Director) Dr. Morris C. Laster MD External Director Capacity Funding LLC (Principal) Mr. Adi Raviv MBA Director Biodar (CEO), Rodar (Founder), Israel Biotech Organization Prof. Avner Rotman PhD Director Bristol Myers Squibb (NYSE: BMY) (Senior Vice President, Corporate Strategy) Mr. Samuel Moed

S CIENTIFIC A DVISORY B OARD (SAB) Leadership ID Biomedical (CSO), Intellivax (Founder), Walter Reed General Hospital (Consultant) Dr. George Lowell Biodar (CEO), Rodar (Founder), Israel Biotech Organization Professor Avner Rotman Former VP of Weizmann Institute. Coinventor of Copaxone®. Former President of Israel Academy of Sciences. Paul Ehrlich Chair in Immunochemistry at the Weizmann Institute. Israel Prize winner. Professor Ruth Arnon Head of the SAB Professor Emeritus, Molecular Genetics, at Weizmann Institute. Winner Israel Prize for Medicine. Chief Scientist of Kadimastem . Professor Michel Revel

B IOND V AX V ALUE P ROPOSITION Value Proposition BiondVax’s assets, capabilities and experiences position us well for in - licensing, acquisitions, and joint ventures & rapid clinical development and manufacturing of new infectious disease therapeutic and prophylactic technologies. Manufacturing, quality, international regulatory and clinical trial knowledge and experience. An existing vaccines / recombinant protein GMP manufacturing facility in Jerusalem. Industry state - of - the - art quality and manufacturing processes. Brought novel influenza vaccine candidate from R&D at Weizmann Institute through to 12,400 participant, 7 country, two influenza season pivotal Phase 3 clinical trial. Big Pharma & biotech experienced leadership team including Chairman (Alexion, Neurocrine , Incyte, ChromaDex and others), CEO (Novartis and GSK), Board director (BMS), COO (Novartis). New CEO with extensive vaccines engineering background from GSK Vaccines, Belgium; has been involved in large capital expenditure projects building vaccines drug substance (DS) and drug product (DP) manufacturing sites in Belgium, Italy, Germany, Hungary and USA. New CEO experienced with global responsibilities for vaccines supply chain management. Twelve years of international experience (USA and Europe). Current COO with experience in building biological manufacturing sites - recombinant proteins site in China and mABs for Novartis Singapore.
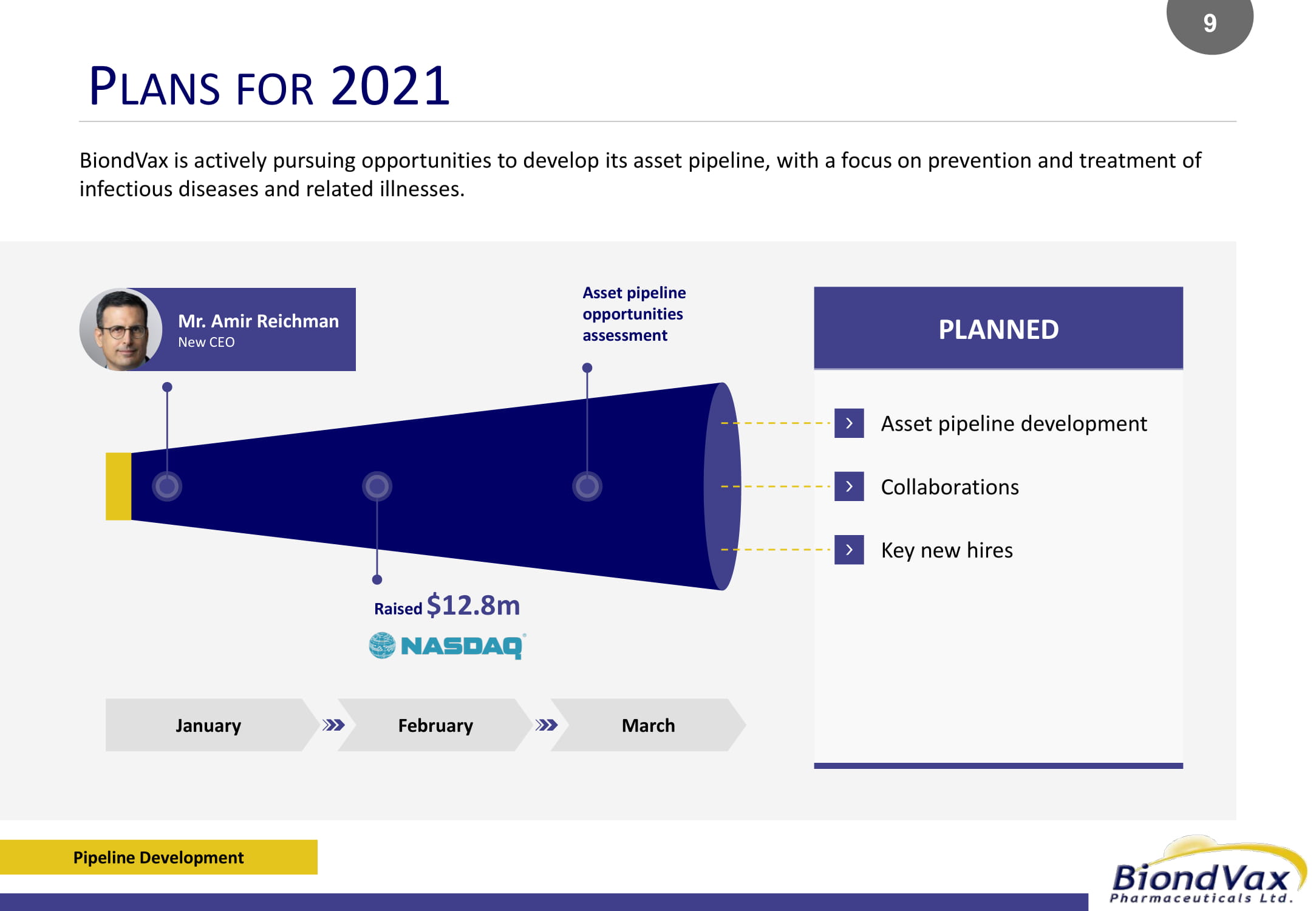
PLANNED P LANS FOR 2021 Pipeline Development BiondVax is actively pursuing opportunities to develop its asset pipeline, with a focus on prevention and treatment of infectious diseases and related illnesses. Mr. Amir Reichman New CEO Asset pipeline opportunities assessment Raised $12.8m Asset pipeline development Collaborations Key new hires January February March

M ANUFACTURING F ACILITY : 10 - 40 M DOSE C APACITY May serve as pilot plant for larger drug substance facility BiondVax GMP Manufacturing Facility Area ~ 1850 m 2 ( 20,000 ft 2 ) Approved cGMP by Israel Ministry of Health (MOH) & EU QP Designed to meet FDA and EMA requirements Currently setup to produce recombinant protein products System and equipment design and verification ASTM E 2500 Standard Capacity in bulk: 10 - 40 m doses, depending on process technology CFR Part 21, Part 11 & GAMP 5 Upstream, downstream and media/buffer prep rooms BiondVax ’ s pharmaceutical manufacturing facility JBP building | Hadassah Ein Kerem Campus | Jerusalem, Israel

M ODULAR & A GILE GMP M ANUFACTURING BiondVax GMP Manufacturing Facility Single - use equipment enables: Adaptable manufacturing processes and products Quicker manufacturing lead times for new products Agile response to emerging infectious disease threats Faster time - to - market for new vaccine and therapeutic products

S UMMARY F INANCIAL D ATA & C AP T ABLE Financials CAP TABLE ADS Outstanding % Ordinary ADS 14,332,146 96% Options + RSUs 642,052 4% Fully Diluted Shares Outstanding 14,974,197 100% • Updated February 12 , 2021 • Voluntarily delisted from Tel Aviv Stock Exchange January 22 , 2018 . (ADS - Shares 1:40 ratio) Lean structure with about 20 employees €24M EIB non - dilutive co - funding agreement Over $ 15 M cash on hand as of March 1 , 2021 . Burn rate about $ 500 K/month Government of Israel support from Ministry of Economy and Industry ’ s Israel Investment Center, and royalty - based grants from the Israel Innovation Authority (formerly Office of the Chief Scientist) American Depository Shares ticker: BVXV

BiondVax Pharmaceuticals Ltd BiondVax Pharmaceuticals Ltd. | Jerusalem, ISRAEL | www.biondvax.com Contact Information: Joshua Phillipson, Investor Relations j.phillipson@biondvax.com + 972 - 8 - 930 - 2529 www.biondvax.com