Exhibit 99.1
|
Investor update
3 June 2016
Nexvet*TM
Transforming animal medicine
|
Forward-looking statements
This presentation and other presentations given today contain forward-looking statements. All statements, other than statements of historical facts, contained in these slides and the accompanying oral presentation, including statements regarding our future results of operations and financial position, business strategy, drug discovery, results of future studies, prospective products, product approvals, research and development costs, manufacturing facility operations and costs, timing and likelihood of success, regulatory approvals, plans and objectives of management for future operations, and future results of current and anticipated products, are forward-looking statements. These statements relate to future events or to our future financial performance and involve known and unknown risks, uncertainties and other factors which may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements. The words “anticipate,” “assume,” “believe,” “contemplate,” “continue,” “could,” “estimate,” “expect,” “goal,” “intend,” “may,” “might,” “objective,” “plan,” “potential,” “predict,” “project,” “positioned,” “seek,” “should,” “target,” “will,” “would,” or the negative of these terms or other similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. These forward-looking statements are based on current expectations, estimates, forecasts and projections about our business and the industry in which we operate and management’s beliefs and assumptions, are not guarantees of future performance or development and involve known and unknown risks, uncertainties, and other factors, including: our ability to become profitable; our expected uses of our capital; our ability to secure additional financing when needed on acceptable terms; our expectation regarding the safety and efficacy our lead product candidates; our ability to advance our lead product candidates through various stages of development, especially through pivotal safety and efficacy studies; our ability to develop future additional product candidates; our ability to obtain regulatory approval for our current or future product candidates under applicable regulatory requirements; our ability to discover, develop and manufacture biologics which involves relatively novel technology and an expensive and lengthy approval process with uncertain outcomes; our ability to successfully manufacture our own products, our reliance on third-party suppliers and distributors to supply and distribute our lead product candidates for us; the rate and degree of market acceptance and the successful commercial sale of our lead product candidates, if approved; regulatory and legal developments in the United States and foreign countries; the success of competing platforms or therapies that are or become available; our ability to attract or retain key employees, advisors or consultants; our expectations and statements regarding the potential size, opportunity and growth potential of the companion animal therapeutics market, and our ability to effectively compete and achieve significant market penetration; our ability to obtain and maintain patent protection for our lead product candidates; the implementation of our business model and strategic plans for our business, products and technology; developments and growth relating to our industry; and uncertainties and assumptions described in greater detail in our annual report on Form 10-K, quarterly report on Form 10-Q and the other documents we have filed with the Securities and Exchange Commission (the “SEC”). We do not assume any obligation to update any forward-looking statements except as required by applicable law.
Certain information contained in this presentation concerning our industry and the markets in which we operate is based on information from our management estimates and primary market research commissioned by us, as well as from industry and general publications and research, surveys and studies conducted by third parties. Management estimates are derived from publicly available information, our knowledge of our industry and assumptions based on such information and knowledge, which we believe to be reasonable. This information, however, involves a number of assumptions and limitations, and you are cautioned not to give undue weight to such information.
PETization, PETisation, Tevxen, BioNua, Nexvet, our logo and its other registered or common law trademarks, trade names or service marks appearing in this presentation are owned by us. Other trademarks, trade names or service marks appearing in this presentation are the property of their respective owners. Solely for convenience, our trademarks and tradenames appear without the ® or ™ symbols, but those references are not intended to indicate, in any way, that we will not assert, to the fullest extent under applicable law, our rights or the right of the applicable licensor to these trademarks and tradenames.
|
Our vision
“The Genentech” of animal health
A company born from a novel platform technology dedicated to market disruption via the superiority of biopharmaceuticals
|
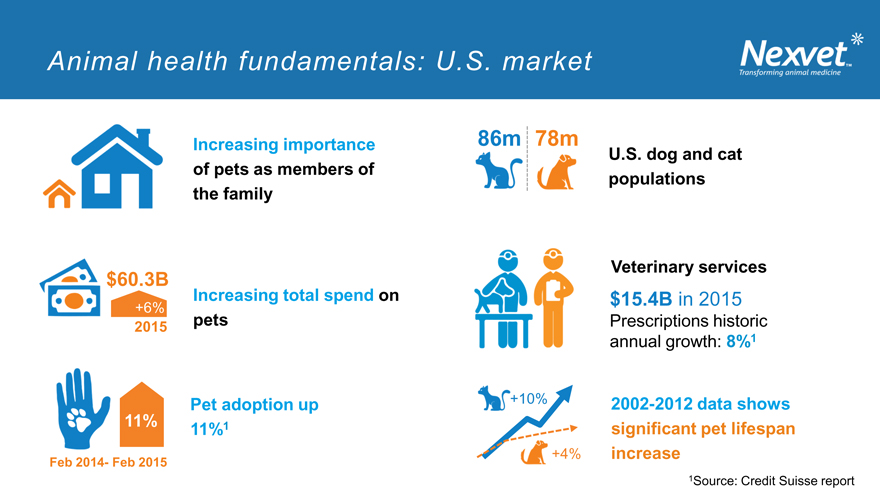
Animal health fundamentals: U.S. market
Increasing importance of pets as members of the family
$60.3B Increasing total spend on
+6%
2015 pets
Pet adoption up 11% 1 11%
Feb 2014- Feb 2015
86m 78m
U.S. dog and cat populations
Veterinary services
$15.4B in 2015
Prescriptions historic annual growth: 8%1
+10% 2002-2012 data shows significant pet lifespan +4% increase
1Source: Credit Suisse report
|
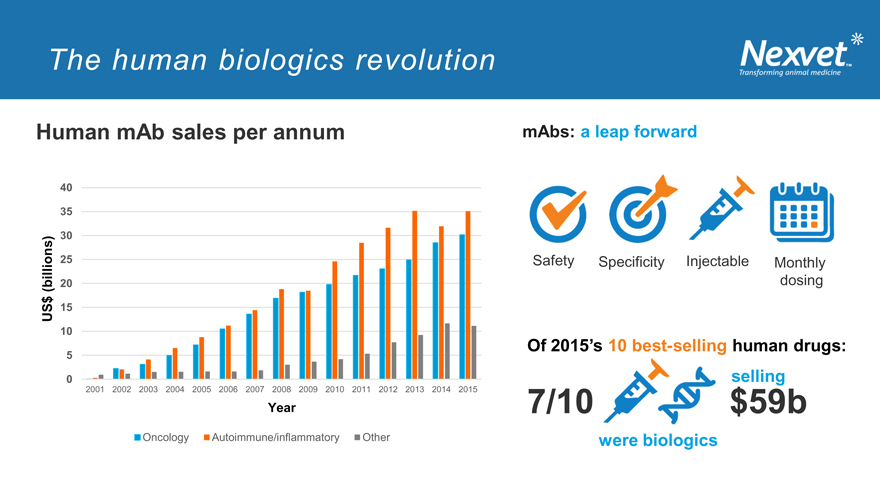
The human biologics revolution
Human mAb sales per annum
40 35 30
25
(billions) 20 $ US 15
10
5
0
2001 2002 2003 2004 2005 2006 2007 2008 2009 2010 2011 2012 2013 2014 2015
Year
Oncology Autoimmune/inflammatory Other
mAbs: a leap forward
Safety Specificity Injectable Monthly dosing
Of 2015’s 10 best-selling human drugs: selling
7/10 $59b
were biologics
|
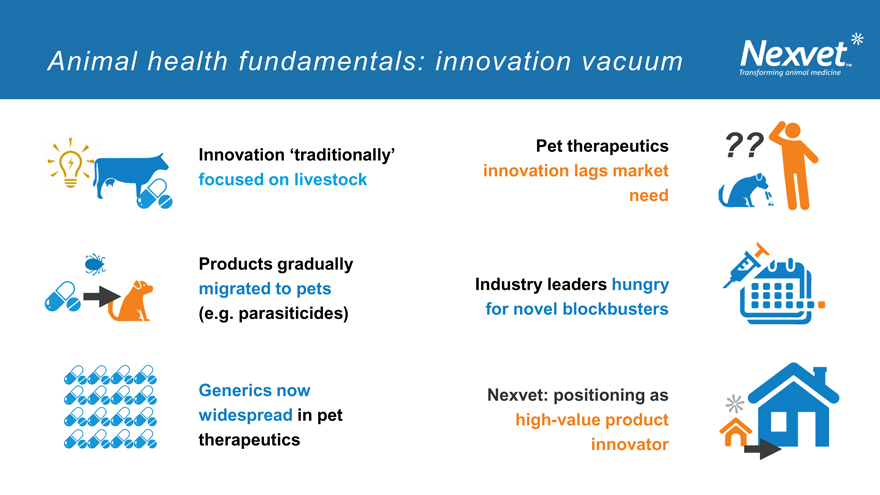
Animal health fundamentals: innovation vacuum
Innovation ‘traditionally’ focused on livestock
Products gradually migrated to pets (e.g. parasiticides)
Generics now widespread in pet therapeutics
Pet therapeutics innovation lags market need
Industry leaders hungry for novel blockbusters
Nexvet: positioning as high-value product innovator
|
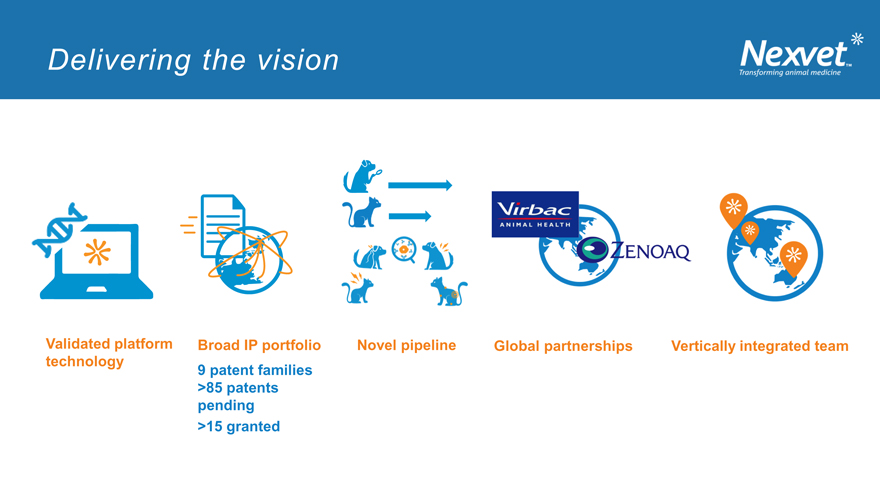
Delivering the vision
Validated platform Broad IP portfolio Novel pipeline Global partnerships Vertically integrated team
technology 9 patent families
>85 patents
pending
>15 granted
|
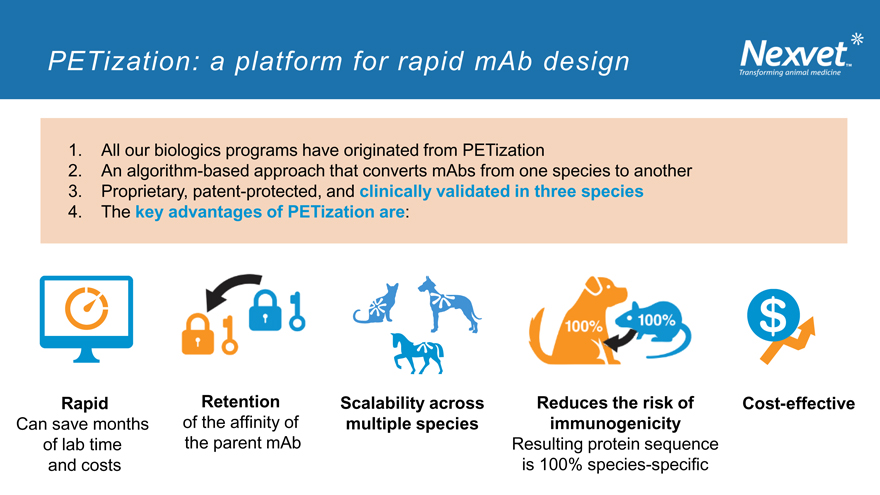
PETization: a platform for rapid mAb design
1. All our biologics programs have originated from PETization
2. An algorithm-based approach that converts mAbs from one species to another
3. Proprietary, patent-protected, and clinically validated in three species
4. The key advantages of PETization are:
Rapid
Can save months of lab time and costs
Retention of the affinity of the parent mAb
Scalability across multiple species
Reduces the risk of immunogenicity
Resulting protein sequence is 100% species-specific
Cost-effective
|
Pipeline
Therapeutic Research AND Est.
Target Pilot study Pivotal studies
area POC approval
Ranevetmab
~2019
Chronic pain NGF NV-02 ~2019
Chronic Dog Multiple
TNF Further studies to commence when POC complete
inflammation Cat in progress
Dog
Allergy Undisclosed Cat Multiple
Dog in progress
Cat
Immuno- PD-1 and other Dog Multiple
oncology undisclosed Dog in progress
Dog
|
Partners
Research and development Distribution and development
The Zenoaq collaboration provides:
Novel starting mAbs and IP to targets
North American and ROW rights (excl. Asia and Europe) to resulting products
The Virbac collaboration provides:
Distribution channels
ROW development and regulatory expertise
Profit-share arrangement for ranevetmab
|
Our global expertise
|
Agenda
Dr. Juergen Horn, CPDO: Clinical programs
Ray O’Connor, VP Technical Operations: BioNua
Damian Lismore, CFO: Financials and strategy
|
Development update
Juergen Horn | Chief Product Development Officer
June 2016
|
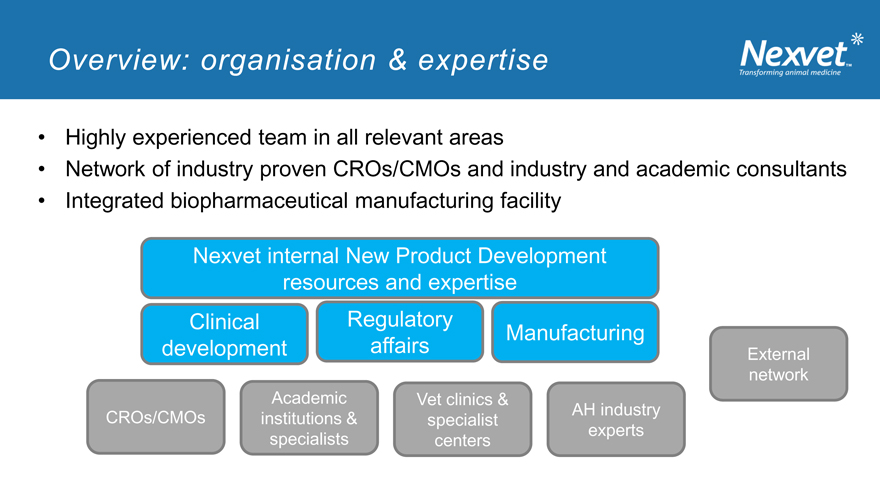
Overview: organisation & expertise
Highly experienced team in all relevant areas
Network of industry proven CROs/CMOs and industry and academic consultants
Integrated biopharmaceutical manufacturing facility
Nexvet internal New Product Development resources and expertise Clinical Regulatory Manufacturing development affairs External
network Academic Vet clinics & AH industry CROs/CMOs institutions & specialist experts specialists centers
|
BioNua: achievements since purchase
Recruitment of expert staff and update/refurbishment of facility
Installation of state-of-the-art disposable biopharmaceutical equipment
Transfer of licenses from previous owner
Successful production of first 200L demo batch
Drug product manufacturing site selected and agreement in place
Next Steps:
Put full GMP system in place and prepare for inspection
Prepare for USDA inspection and approval
Process development, validation & stability work for development programs
|
Chronic pain in dogs: first-in-class opportunity
>140m
pet dogs in U.S. and EU
U.S. small animal $284m pain drug sales 83%
2015
Daily pills:
• poor compliance ~95%
60% owners would pay for more convenient dosing
Dog population is aging: ~20% will get osteoarthritis (OA)
NSAIDs dominate despite: • label warnings • inconvenient dosing
Nearly all veterinarians would use ranevetmab1 for chronic OA pain; a majority would use it for multiple other indications
1Source: Proprietary GfK survey
|
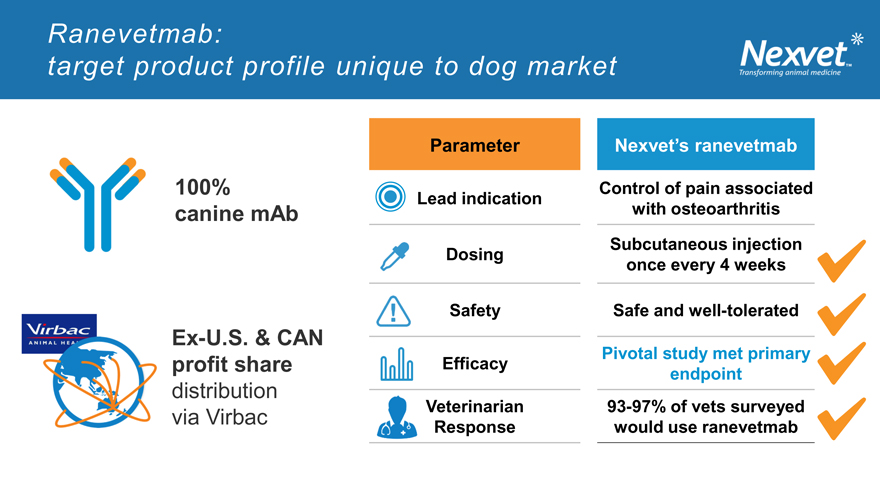
Ranevetmab: target product profile unique to dog market
Parameter Nexvet’s ranevetmab
100% Control of pain associated
Lead indication
canine mAb with osteoarthritis
Dosing Subcutaneous injection
once every 4 weeks
Safety Safe and well-tolerated
Ex-U.S. & CAN
Pivotal study met primary
profit share Efficacy endpoint
distribution
via Virbac Veterinarian 93-97% of vets surveyed
Response would use ranevetmab
|
Ranevetmab: achievements in 2015/2016
Successful completion of pivotal OA study
TAS protocol concurred
Meetings with regulators (U.S., EU): clarity on requirements
Successful introduction of new purification step to improve yield even further
Successful transfer from CMOs for process development: robust upstream process established
Next Steps:
Submission of effectiveness technical section to FDA-CVM (end June 2016)
Transfer to BioNua for scale up, process validation & stability (from May 2016)
Manufacture clinical trial material and initiate pivotal TAS study (H2.2016)
|
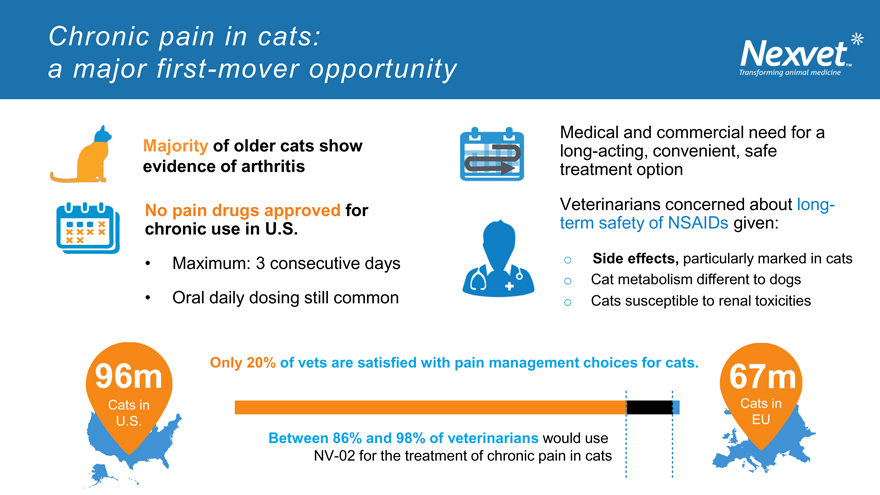
Chronic pain in cats: a major first-mover opportunity
Medical and commercial need for a Majority of older cats show long-acting, convenient, safe evidence of arthritis treatment option
No pain drugs approved for Veterinarians concerned about long-chronic use in U.S. term safety of NSAIDs given:
Maximum: 3 consecutive days o Side effects, particularly marked in cats
o Cat metabolism different to dogs
Oral daily dosing still common o Cats susceptible to renal toxicities
96m Only 20% of vets are satisfied with pain management choices for cats. 67m
Cats in Cats in U.S. EU
Between 86% and 98% of veterinarians would use NV-02 for the treatment of chronic pain in cats
|
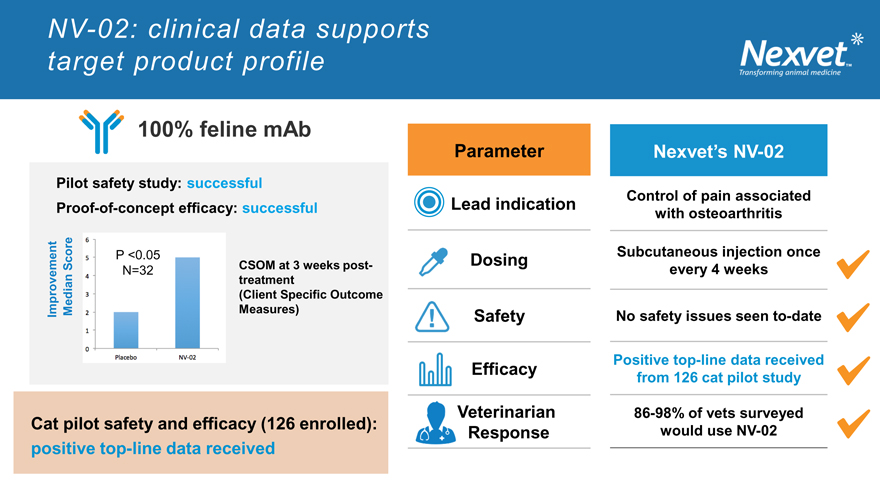
NV-02: clinical data supports target product profile
100% feline mAb
Pilot safety study: successful
Proof-of-concept efficacy: successful
P <0.05
Score CSOM at 3 weeks post-
N=32 treatment
(Client Specific Outcome Improvement Median Measures)
Cat pilot safety and efficacy (126 enrolled): positive top-line data received
Parameter Nexvet’s NV-02
Lead indication Control of pain associated
with osteoarthritis
Dosing Subcutaneous injection once
every 4 weeks
Safety No safety issues seen to-date
Positive top-line data received
Efficacy from 126 cat pilot study
eterinarian 86-98% of vets surveyed
Response would use NV-02
|
NV-02: achievements 2015/16
Successful completion of pilot OA study
TAS protocol submitted to FDA-CVM for concurrence
Regulatory confirmation that NV-02 follows same principles as NV-01
Successful transfer from CMOs to BioNua: first 200L demo batch successfully produced
Next Steps:
Protocol concurrence for pivotal efficacy and TAS studies by FDA-CVM (Q3.2016)
Initiate pivotal efficacy and TAS studies (H2.2016)
Manufacturing process validation and stability (H2.2016 onwards)
|
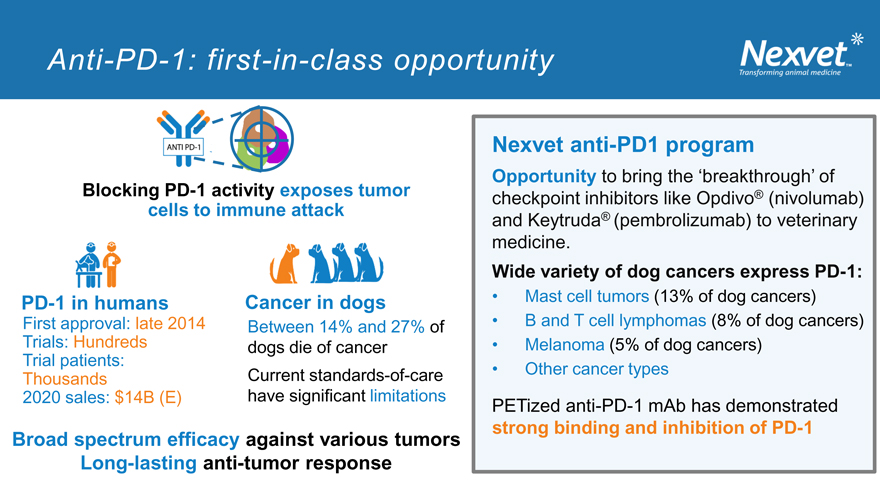
Anti-PD-1: first-in-class opportunity
`
Blocking PD-1 activity exposes tumor cells to immune attack
PD-1 in humans Cancer in dogs
First approval: late 2014 Between 14% and 27% of Trials: Hundreds dogs die of cancer Trial patients: Current standards-of-care Thousands 2020 sales: $14B (E) have significant limitations
Broad spectrum efficacy against various tumors Long-lasting anti-tumor response
Nexvet anti-PD1 program
Opportunity to bring the ‘breakthrough’ of checkpoint inhibitors like Opdivo® (nivolumab) and Keytruda® (pembrolizumab) to veterinary medicine.
Wide variety of dog cancers express PD-1:
Mast cell tumors (13% of dog cancers)
B and T cell lymphomas (8% of dog cancers)
Melanoma (5% of dog cancers)
Other cancer types
PETized anti-PD-1 mAb has demonstrated strong binding and inhibition of PD-1
|
USDA: achievements 2015/16
Candidate anti-PD-1 PETized mAbs identified
USDA jurisdiction granted for anti-PD1 mAbs
Identified experienced regulatory and manufacturing partner for USDA programs
Product Development Plan established for first USDA development program
Next Steps:
Initiate PKPD/safety and proof-of-concept studies
Initiate USDA manufacturing strategy
Prepare BioNua for USDA inspection
|
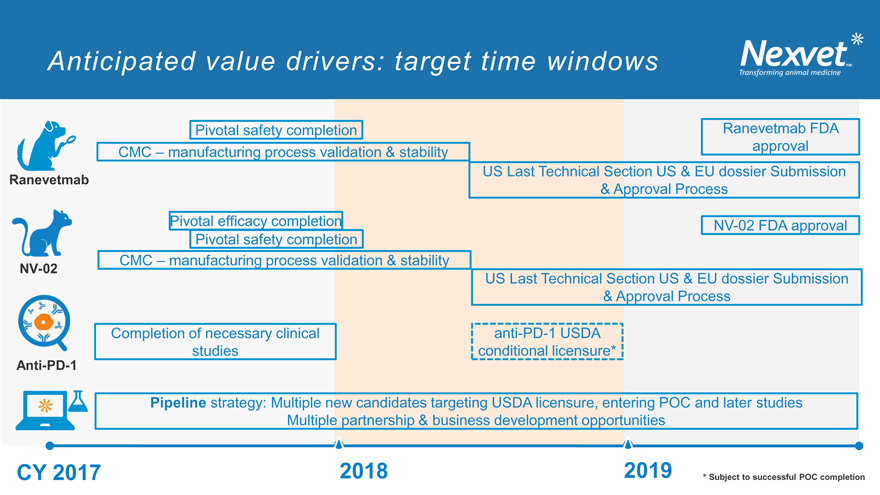
Anticipated value drivers: target time windows
Pivotal safety completion Ranevetmab FDA CMC – manufacturing process validation & stability approval US Last Technical Section US & EU dossier Submission
Ranevetmab
& Approval Process
Pivotal efficacy completion NV-02 FDA approval Pivotal safety completion CMC – manufacturing process validation & stability
NV-02
US Last Technical Section US & EU dossier Submission
& Approval Process
Completion of necessary clinical anti-PD-1 USDA studies conditional licensure*
Anti-PD-1
Pipeline strategy: Multiple new candidates targeting USDA licensure, entering POC and later studies Multiple partnership & business development opportunities
v v
CY 2017 2018 2019 * Subject to successful POC completion
|
Establishment of internal drug substance manufacturing capabilities: BioNua
Ray O’Connor | VP Technical Operations
June 2016
|
Strategic importance of BioNua
1. Dedicated CMOs for veterinary mAbs do not exist
2. There are substantial challenges with sharing veterinary and human manufacturing space
3. There is currently shortage of global biopharma manufacturing capacity
4. Regulators require strong linkage between clinical material and commercial manufacture
5. Lowers development and commercial manufacture costs
6. Our own products will always be first priority
|
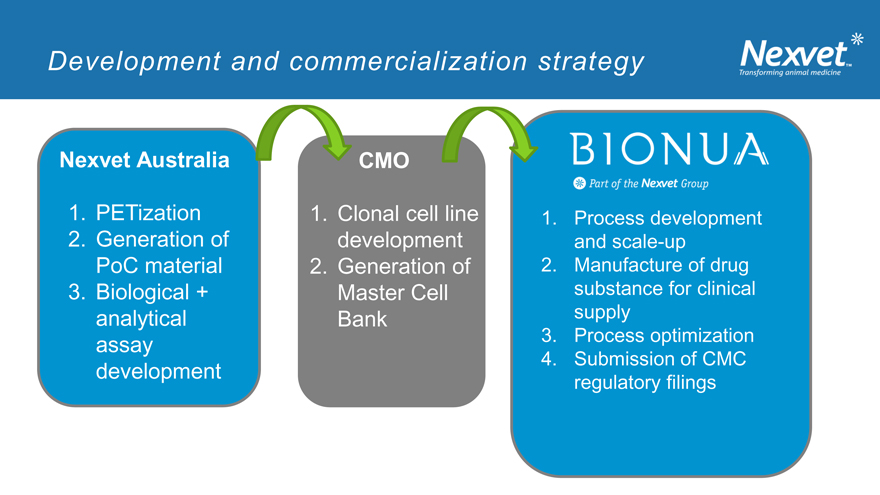
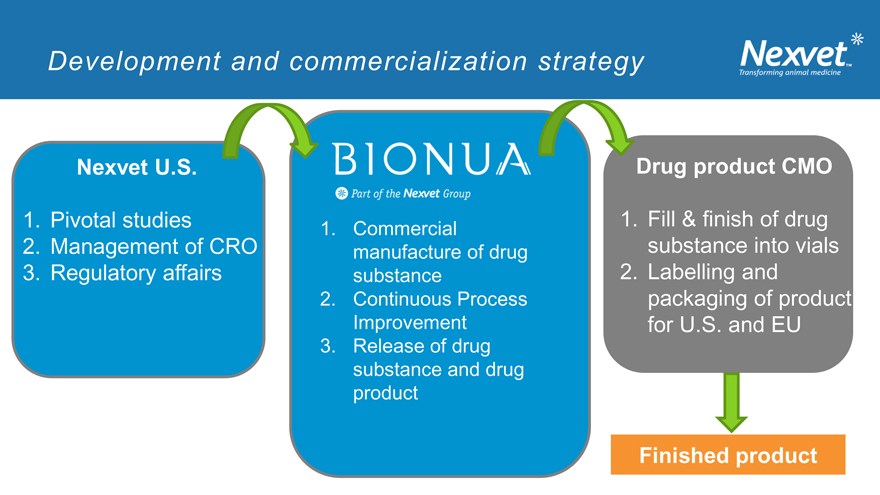
Development and commercialization strategy
Nexvet Australia CMO
1. PETization 1. Clonal cell line 1. Process development
2. Generation of development and scale-up
PoC material 2. Generation of 2. Manufacture of drug
3. Biological + Master Cell substance for clinical
analytical Bank supply
assay 3. Process optimization
4. Submission of CMC
development
regulatory filings
|
Development and commercialization strategy
Nexvet U.S. Drug product CMO
1. Pivotal studies 1. Fill & finish of drug
1. Commercial
2. Management of CRO manufacture of drug substance into vials
3. Regulatory affairs substance 2. Labelling and
2. Continuous Process packaging of product Improvement for U.S. and EU
3. Release of drug substance and drug product
Finished product
|
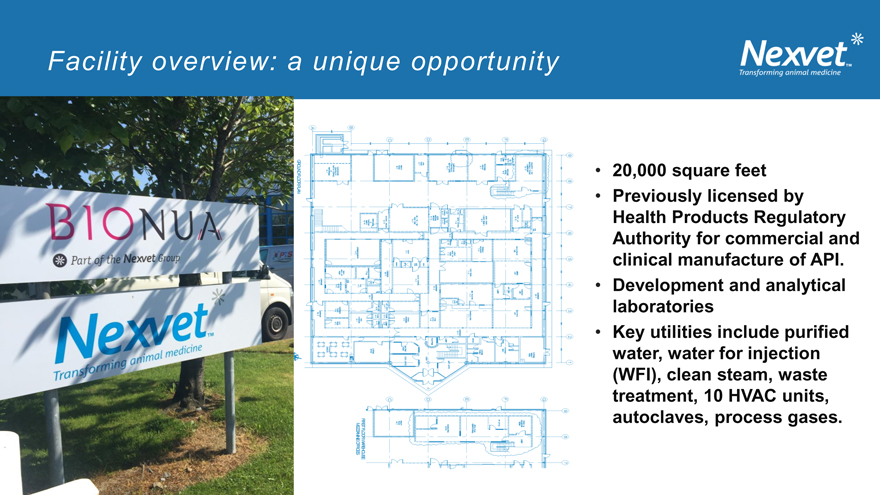
Facility overview: a unique opportunity
• 20,000 square feet
• Previously licensed by Health Products Regulatory Authority for commercial and clinical manufacture of API.
• Development and analytical laboratories
• Key utilities include purified water, water for injection (WFI), clean steam, waste treatment, 10 HVAC units, autoclaves, process gases.
|
Acquisition process
Full due diligence was performed from May to September 2015
Engineering review determined that facility was well maintained and utilities were in good working order
A review of the quality systems indicated that the facility had a good track record with the HPRA (Irish Regulators – equivalent of FDA). Ex-FDA inspectors had inspected the site previously and deemed it suitable as an FDA facility
There were no staff legacy issues and a new team could be hired
Review of past overhead costs indicated that overheads were relatively stable and in line with a plant of this size
|
Manufacturing and development capabilities
Current capacity is approximately 30 x 200L batches per year
Facility is capable of scaling up to 1,000L disposable bioreactor
The bridge between research to development and onto commercial-scale manufacturing has been established using 250mL and 10L bioreactors
|
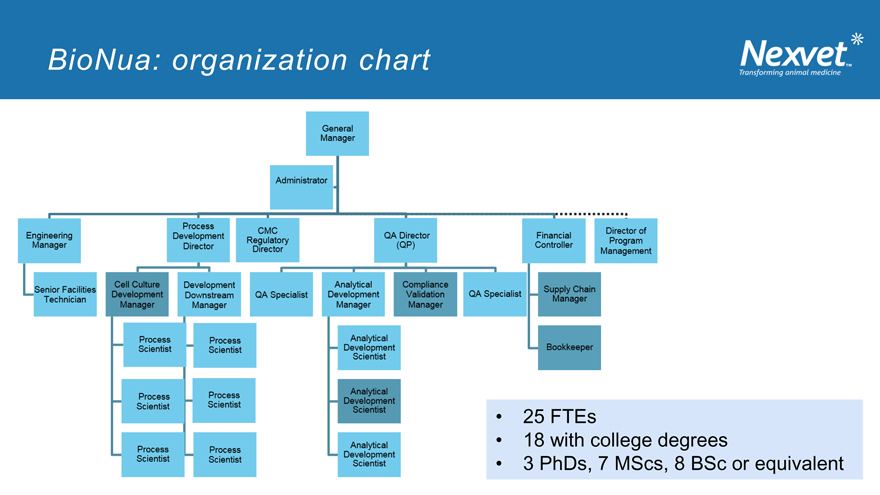
BioNua: organization chart
25 FTEs
18 with college degrees
3 PhDs, 7 MScs, 8 BSc or equivalent
|
Accelerated start-up schedule delivered
A traditional start-up schedule
0 month 18 months
Delivered BioNua start-up schedule
0 month 9 months
Start-up strategy
Experienced team
Leveraged the previous operator’s infrastructure
Purchased high quality disposable equipment: similar to previous CMO
Keeping It Simple
Produced first 200L-scale NV-02 batch
|
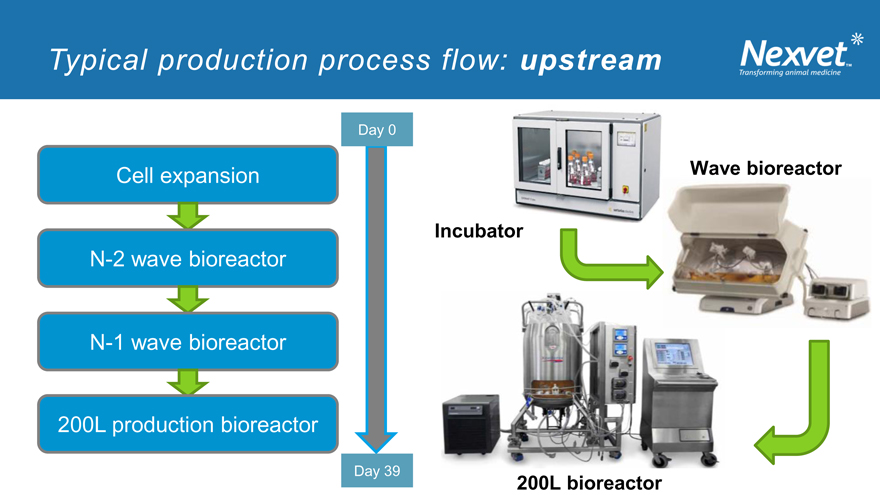
Typical production process flow: upstream
Day 0
Cell expansion Wave bioreactor
Incubator
N-2 wave bioreactor
N-1 wave bioreactor
200L production bioreactor
Day 39
200L bioreactor
|
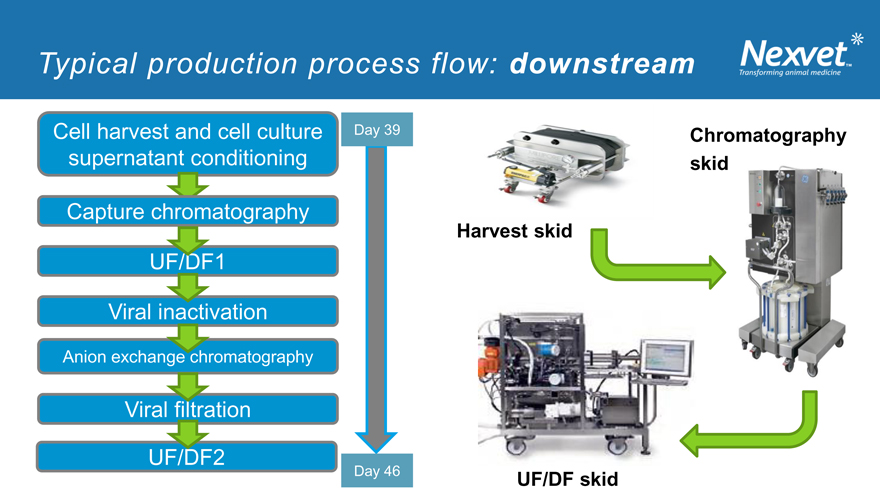
Typical production process flow: downstream
Cell harvest and cell culture Day 39 Chromatography supernatant conditioning skid
Capture chromatography
Harvest skid
UF/DF1
Viral inactivation
Anion exchange chromatography
Viral filtration
UF/DF2
Day 46 UF/DF skid
|
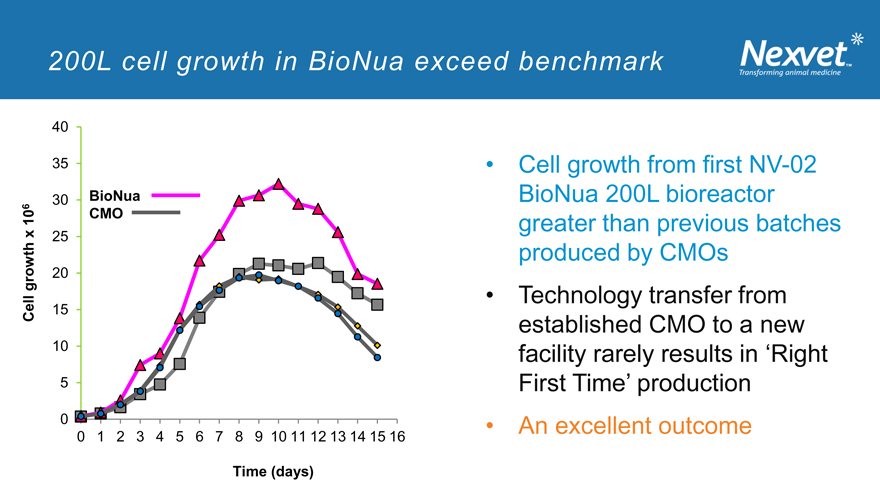
200L cell growth in BioNua exceed benchmark
40
35
30 BioNua 6 10 CMO x
25 growth 20 Cell 15 10
5
0
0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16
Time (days)
Cell growth from first NV-02 BioNua 200L bioreactor greater than previous batches produced by CMOs
Technology transfer from established CMO to a new facility rarely results in ‘Right First Time’ production
An excellent outcome
|
Summary
1. Facility became available April 2015, and was a highly attractive acquisition
2. It became clear that securing this facility for Nexvet would ensure:
A smoother regulatory approval process for our products
Continuity of supply
Lower COGs
Lower development costs for future products
3. Process scientists, QA team, and support staff recruited to support development and manufacturing
4. All Upstream and Downstream process equipment (and supporting analytical): purchased, validated and proven to deliver
5. First drug substance NV-02 batch commenced on April 2016 and completed at the end of May 2016
|
Financing and corporate structure
Damian Lismore | Chief Financial Officer
June 2016
|
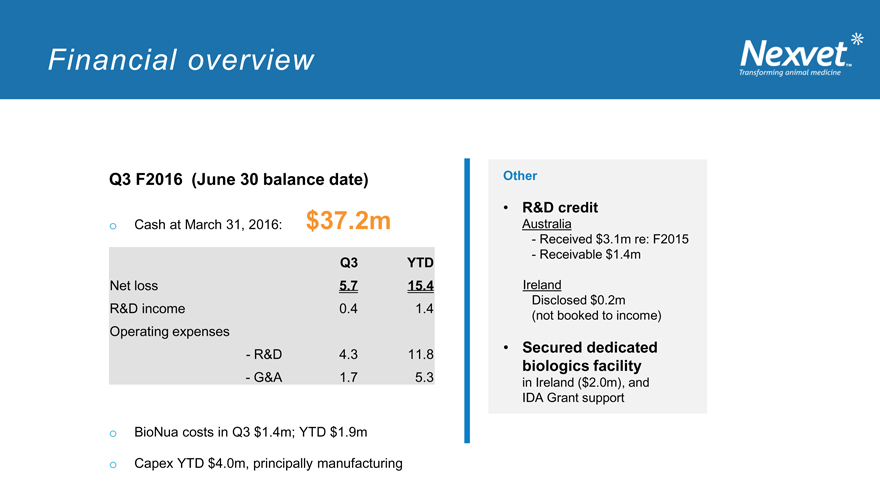
Financial overview
Q3 F2016 (June 30 balance date) Other
R&D credit o Cash at March 31, 2016: $37.2m Australia
- Received $3.1m re: F2015
- Receivable $1.4m
Q3 YTD
Net loss 5.7 15.4 Ireland
Disclosed $0.2m
R&D income 0.4 1.4
(not booked to income)
Operating expenses
• Secured dedicated
- R&D 4.3 11.8 biologics facility
- G&A 1.7 5.3 in Ireland ($2.0m), and IDA Grant support
o BioNua costs in Q3 $1.4m; YTD $1.9m
o Capex YTD $4.0m, principally manufacturing
|
BioNua snapshot
Overview of transaction o Facility historically operated under Investigational Medicines Product License and a cGMP Manufacturing License o Paid €1.75m ($2.0m) o Entered a 10 year lease—$118k p.a. initially, market assessments, with purchase option after 5 years o Initial capex requirement ~ $3m
Indicative cost profile when operational (not Year 1) o Staff costs $3m-$4m o Overhead costs ~$1.1m o BioNua development cost v External CMO
~ $3.0m saving per program (~30% savings)
~ substantial savings on commercial COGS expected
Incentives
IDA financial support via cash grant subject to agreed milestones
BioNua will be R&D eligible at 25% of net qualifying spend (i.e. after deducting IDA grant income)
|
IDA grant overview
Overview
o IDA Ireland support of over $2m cash grant over 4 years o Support for equipment, people and project-specific manufacturing o Payments contingent on milestone achievements o IDA Ireland retains re-imbursement rights in all agreements
Cash flow and reporting
o Cash: normally paid annually, subject IDA audit o Grant income recognition will be lumpy, low initially – until first cash receipt o Expect regular disclosure on status
|
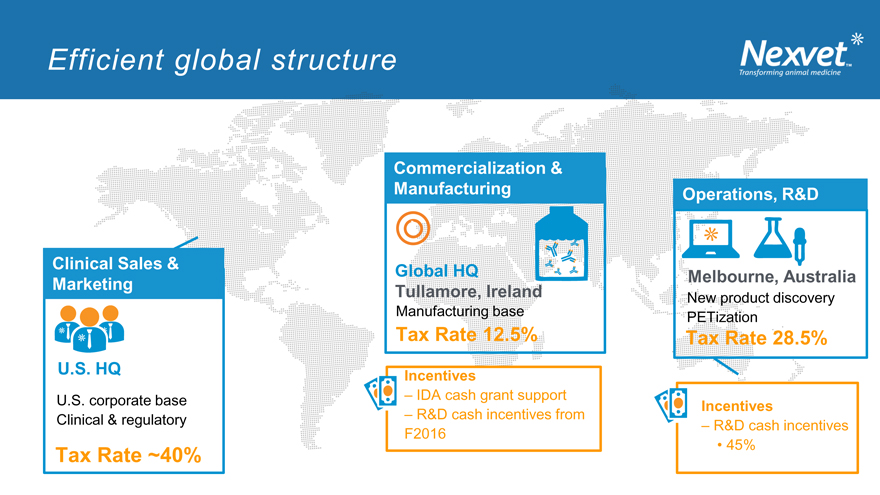
Efficient global structure
Clinical Sales & Marketing
U.S. HQ
U.S. corporate base Clinical & regulatory
Tax Rate ~40%
Commercialization & Manufacturing
`
Global HQ
Tullamore, Ireland
Manufacturing base
Tax Rate 12.5%
Incentives
– IDA cash grant support
– R&D cash incentives from F2016
Operations, R&D
Melbourne, Australia
New product discovery PETization
Tax Rate 28.5%
Incentives
– R&D cash incentives
45%
|
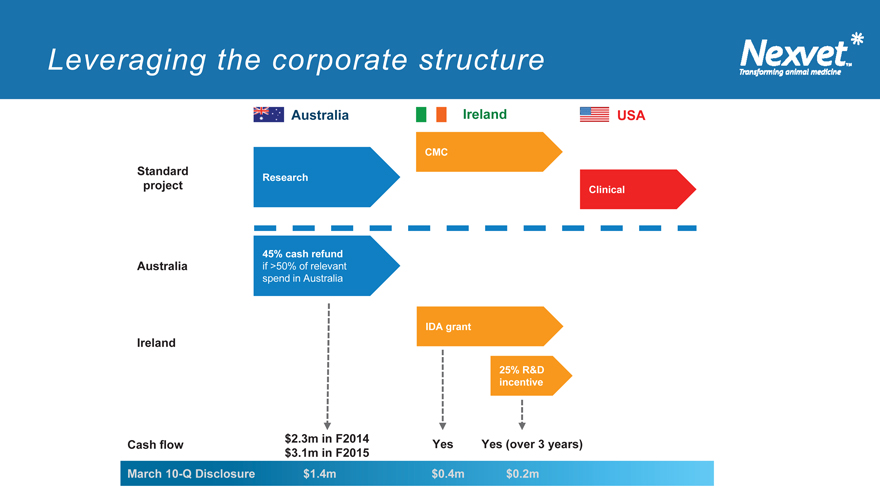
Leveraging the corporate structure
Australia Ireland USA
CMC
Standard
Research project Clinical
45% cash refund
Australia if >50% of relevant spend in Australia
IDA grant
Ireland
25% R&D incentive
$2.3m in F2014
Cash flow Yes Yes (over 3 years)
$3.1m in F2015
March 10-Q Disclosure $1.4m $0.4m $0.2m
|
Summary
Strong cash position as of March 31,
2016: Nexvet has gained a quality manufacturing facility
$37.2m Internalizing manufacturing reduces development costs and COGS
Leveraging skillsets, favorable tax incentives and Nexvet positioned for
non-dilutive grants pipeline growth in cost-effective manner
|
Summary
Dr. Mark Heffernan | CEO
June 2016
|
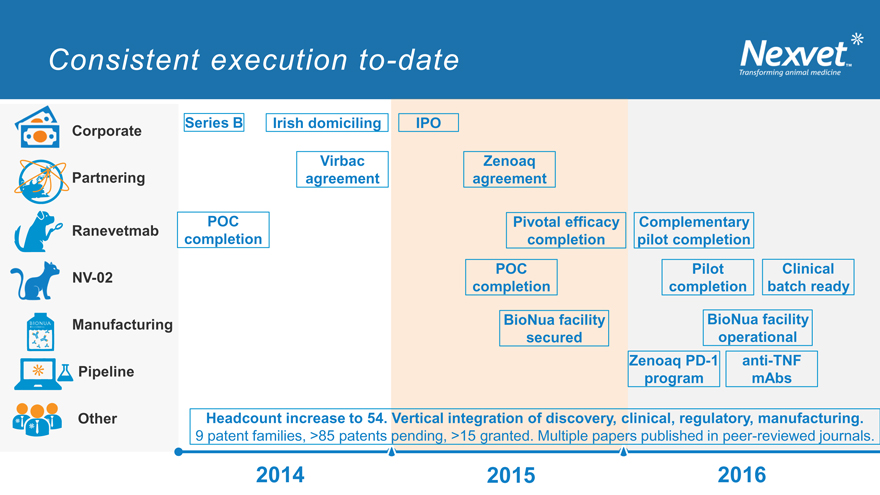
Consistent execution to-date
Series B Irish domiciling IPO Corporate
Virbac Zenoaq Partnering agreement agreement
POC Pivotal efficacy Complementary Ranevetmab completion completion pilot completion
POC Pilot Clinical NV-02 completion completion batch ready
Manufacturing BioNua facility BioNua facility secured operational Zenoaq PD-1 anti-TNF Pipeline program mAbs
Other Headcount increase to 54. Vertical integration of discovery, clinical, regulatory, manufacturing.
9 patent families, >85 patents pending, >15 granted. Multiple papers published in peer-reviewed journals.
2014 v v 2016 2015
|
Upcoming catalysts
NV-02 pilot study completion (Q2): DELIVERED
Bionua operational (Q2): DELIVERED
Ranevetmab pivotal data presentation (Q2)
Ranevetmab TAE submission (Q2)
Anticipated NV-02 pivotal safety and efficacy study initiation (2H) Manufacture clinical material and initiate pivotal TAS study (2H) Deliver on PoC study data from pipeline programmes (2H) Partnering potential
|
Nexvet*TM
Transforming animal medicine