
Interim Analysis of STK-001 for the Treatment of Dravet Syndrome Stoke Therapeutics November 14, 2022 Exhibit 99.2

Agenda Introduction Eric Rojas, Head of Investor Relations Introductory Remarks Edward M. Kaye, M.D., Chief Executive Officer Phase 1/2a Interim Analysis Barry Ticho, M.D., Ph.D., Chief Medical Officer Closing Remarks Edward M. Kaye, M.D., Chief Executive Officer Q&A

Forward Looking Statements This presentation has been prepared by Stoke Therapeutics, Inc. (“Stoke” or “our”) for informational purposes only and not for any other purpose. Nothing contained in this presentation is, or should be construed as, a recommendation, promise or representation by the presenter or Stoke or any officer, director, employee, agent or advisor of Stoke. This presentation does not purport to be all-inclusive or to contain all of the information you may desire. Information provided in this presentation speaks only as of the date hereof. Stoke assumes no obligation to publicly update any information or forward-looking statement, whether written or oral, that may be made from time to time, whether as a result of new information, future developments, subsequent events, or circumstances after the date hereof, or to reflect the occurrence of unanticipated events. This presentation contains “forward-looking” statements within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995, including, but not limited to: the ability of STK-001 to treat Dravet syndrome and reduce seizures or show improvements in non-seizure comorbidities, the timing and expected progress of clinical trials, data readouts, milestones and presentations, and the timing or receipt of regulatory approval. These forward-looking statements may be accompanied by such words as “anticipate,” “believe,” “could,” “estimate,” “expect,” “intend,” “likely,” “may,” “might,” “plan,” “potential,” “possible,” “will,” “would,” and other words and terms of similar meaning. These forward-looking statements involve risks and uncertainties, as well as assumptions, which, if they do not fully materialize or prove incorrect, could cause our results to differ materially from those expressed or implied by such forward-looking statements. Forward-looking statements are subject to risks and uncertainties that may cause the company’s actual activities or results to differ significantly from those expressed in any forward-looking statement, including risks and uncertainties related to the Company’s ability to advance its product candidates, obtain regulatory approval of and ultimately commercialize its product candidates, the timing and results of preclinical and clinical trials, preliminary interim data readouts of ongoing trials showing results that change when such trials are completed, the company’s ability to fund development activities and achieve development goals, the company’s ability to protect intellectual property and other risks and uncertainties described under the heading “Risk Factors” in the Company's Annual Report on Form 10-K for the year ended December 31, 2021, its Quarterly Reports on Form 10-Q, and other documents the company files from time to time with the Securities and Exchange Commission. These forward-looking statements speak only as of the date of this presentation, and the Company undertakes no obligation to revise or update any forward-looking statements to reflect events or circumstances after the date hereof. These statements are based on our current beliefs and expectations and speak only as of the date of this presentation. We do not undertake any obligation to publicly update any forward-looking statements. By attending or receiving this presentation you acknowledge that you are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date such statements are made; you will be solely responsible for your own assessment of the market and our market position; and that you will conduct your own analysis and be solely responsible for forming your own view of the potential future performance of Stoke.

Introductory Remarks Edward M. Kaye, M.D. Chief Executive Officer
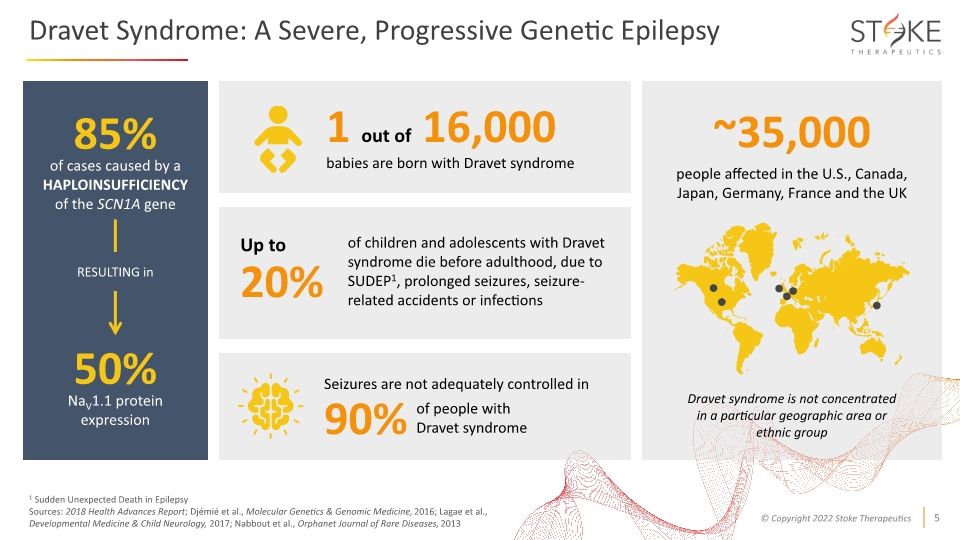
1 Sudden Unexpected Death in Epilepsy Sources: 2018 Health Advances Report; Djémié et al., Molecular Genetics & Genomic Medicine, 2016; Lagae et al., Developmental Medicine & Child Neurology, 2017; Nabbout et al., Orphanet Journal of Rare Diseases, 2013 ~35,000 Seizures are not adequately controlled in 90% of people with Dravet syndrome 85% of cases caused by a HAPLOINSUFFICIENCY of the SCN1A gene people affected in the U.S., Canada, Japan, Germany, France and the UK Dravet syndrome is not concentrated in a particular geographic area or ethnic group 50% NaV1.1 protein expression RESULTING in Dravet Syndrome: A Severe, Progressive Genetic Epilepsy

Non-Seizure Comorbidities of Dravet Syndrome Are Not Addressed by Current Therapies No Approved Disease-Modifying Therapies for Dravet Syndrome Intellectual disability Developmental delays Movement and balance issues Language and speech disturbances Growth defects Sleep abnormalities Disruptions of the autonomic nervous system Mood disorders Dravet syndrome is classified as a developmental and epileptic encephalopathy due to the developmental delays and cognitive impairment associated with the disease

* VABS = Vineland Adaptive Behavior Scales * ABC score based on Communication, Daily Living, and Socialization domains and expressed relative to normative mean of 100 Source: BUTTERFLY: An Observational Study to Investigate Cognition and Other Non-seizure Comorbidities in Children and Adolescents with Dravet Syndrome (DS) (AES 2021). Initial findings showed: Validation of standard cognitive measures for use in DS patients Substantially decreased neurocognitive abilities despite the use of multiple anti-seizure medications A gap in adaptive functioning was observed in VABS* testing Gap in overall intellectual development and adaptive function between patients and neurotypical children appears to widen with age (n=36, 2-18 year-olds). Study ongoing. Non-Seizure Comorbidities of DS are Progressive and Measurable 100 80 60 40 20 0 24 48 72 96 120 144 168 192 216 Standard Score Age (m) Adaptive Behavior Composite (ABC)* Results from the VABS Assessment

Boldly Restoring Genetic Health Addressing the underlying cause of severe diseases by upregulating protein expression with RNA-based medicines Executing in the clinic with STK-001, the first potential disease-modifying approach for the treatment of Dravet syndrome Advancing to the clinic with STK-002, the first potential disease-modifying approach for the treatment of Autosomal Dominant Optic Atrophy (ADOA) Expanding our pipeline through internal discovery and collaboration
Interim Analysis of Phase 1/2a MONARCH and ADMIRAL Studies of STK-001 Barry Ticho, M.D., Ph.D. Chief Medical Officer
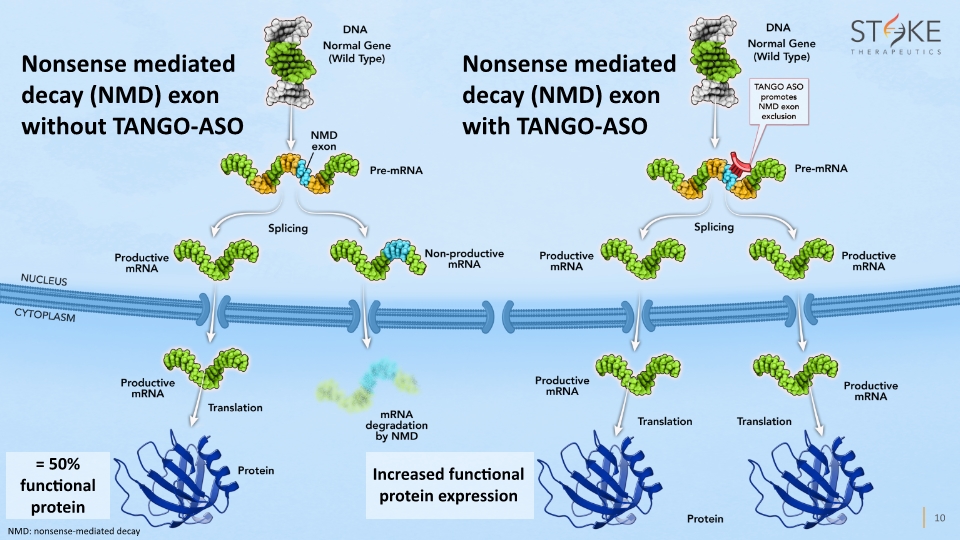
Nonsense mediated decay (NMD) exon without TANGO-ASO Increased functional protein expression Nonsense mediated decay (NMD) exon with TANGO-ASO NMD: nonsense-mediated decay
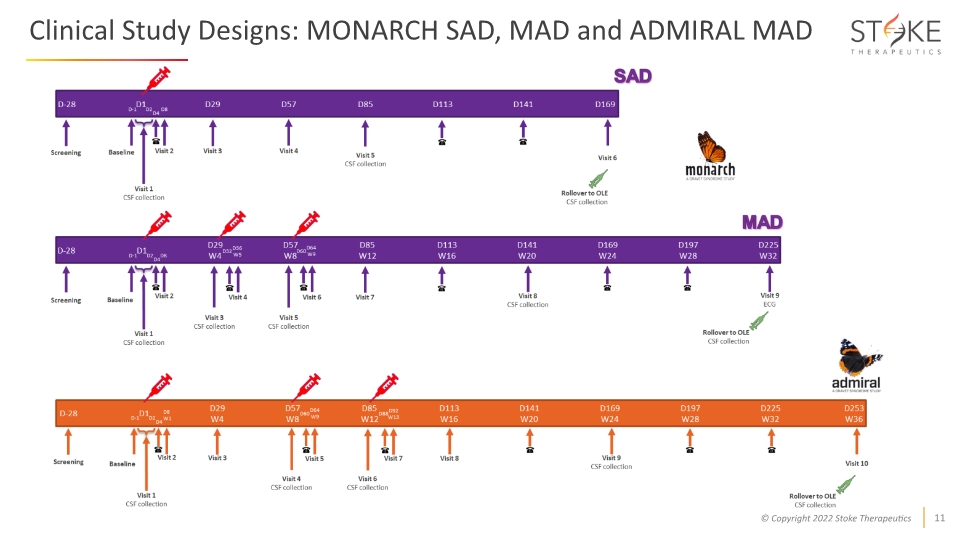
Clinical Study Designs: MONARCH SAD, MAD and ADMIRAL MAD
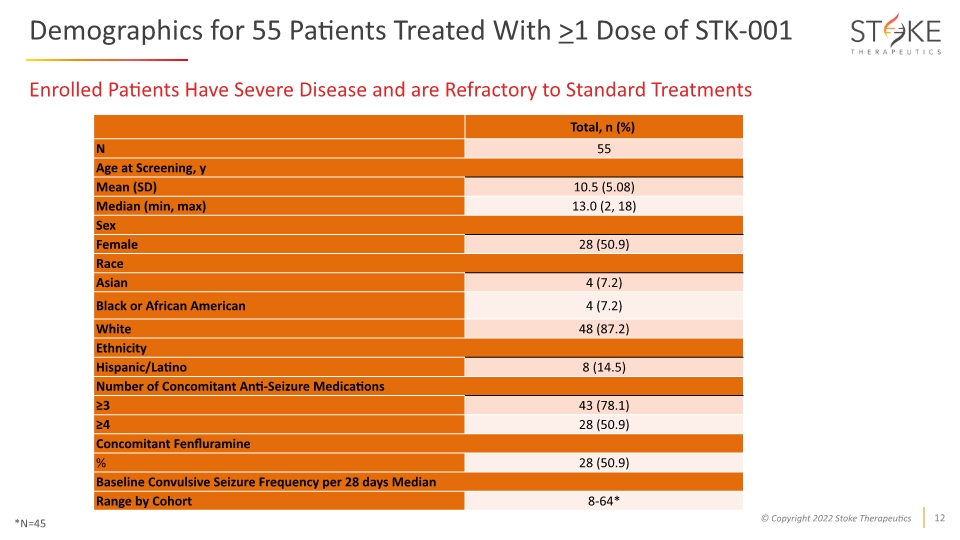
Demographics for 55 Patients Treated With >1 Dose of STK-001 Enrolled Patients Have Severe Disease and are Refractory to Standard Treatments *N=45

Multiple Doses of STK-001 up to 45mg Well-Tolerated 27% (15/55) of patients experienced a TEAE related to study drug; All AEs related to study drug were mild or moderate 22% (12/55) of patients had a treatment-emergent SAE none related to study drug Most common TEAEs were vomiting, headache, and seizure 33% (18/55) of patients experienced CSF protein elevation (>50mg/dL) after dosing; no clinical manifestations were observed No new significant neurologic exam findings or lower extremity weakness emerged related to study drug All Adverse Events Related to Study Drug Were Mild or Moderate Data Cut-off date: August 11, 2022
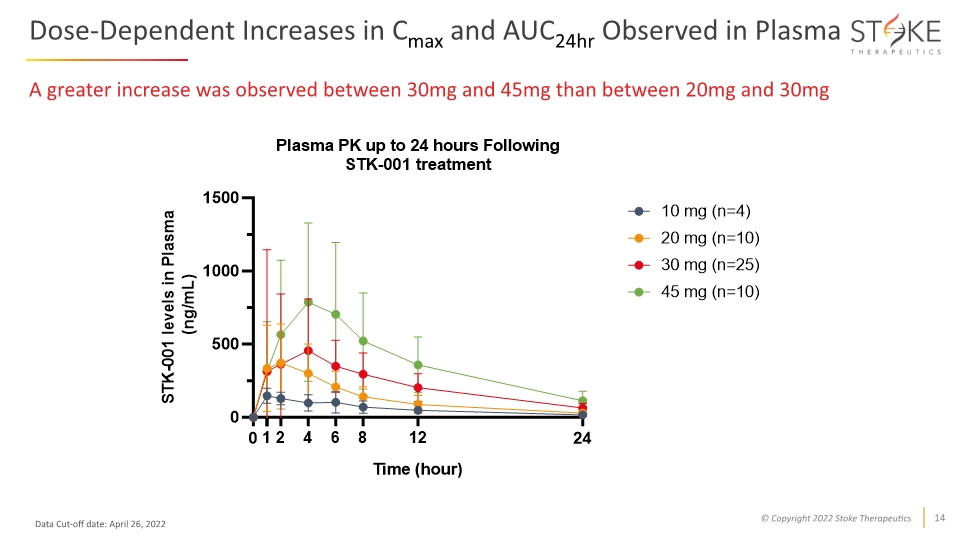
Dose-Dependent Increases in Cmax and AUC24hr Observed in Plasma A greater increase was observed between 30mg and 45mg than between 20mg and 30mg Data Cut-off date: April 26, 2022

CSF Exposure was Measurable up to Six Months Following Multiple Doses of STK-001 in MONARCH study Slight increases in CSF values were observed from 20mg to 30mg Data Cut-off date: April 27, 2022

Majority of Patients Treated with Multiple Doses (20mg or 30mg) in MONARCH Reach Pharmacologically Active Brain Levels Clinical Data for 20mg and 30mg MAD Align with Model Simulations from Patient CSF Levels Solid line: Median of the predicted concentrations, Shaded area: 95th confidence interval (2.5th – 97.5th percentile) of the predicted concentrations, Open squares: Extrapolated concentrations. The 20mg MAD and 30mg MAD plots are for STK-001-DS-101 study. On Y axis, the dashed black lines indicate pharmacologically active concentration range Mean Brain without thalamus Pharmacologically active range Data Cut-off date: April 27, 2022

55% Median Reduction in Convulsive Seizure Frequency Observed in Patients Treated With Three Doses of STK-001 (45mg) Across the multiple dose cohorts (20mg, 30mg, 45mg), 74% (20/27) of patients experienced reductions in seizures Similar seizure reduction was observed among patients taking or not taking concomitant fenfluramine (>50% of patients were taking concomitant fenfluramine)
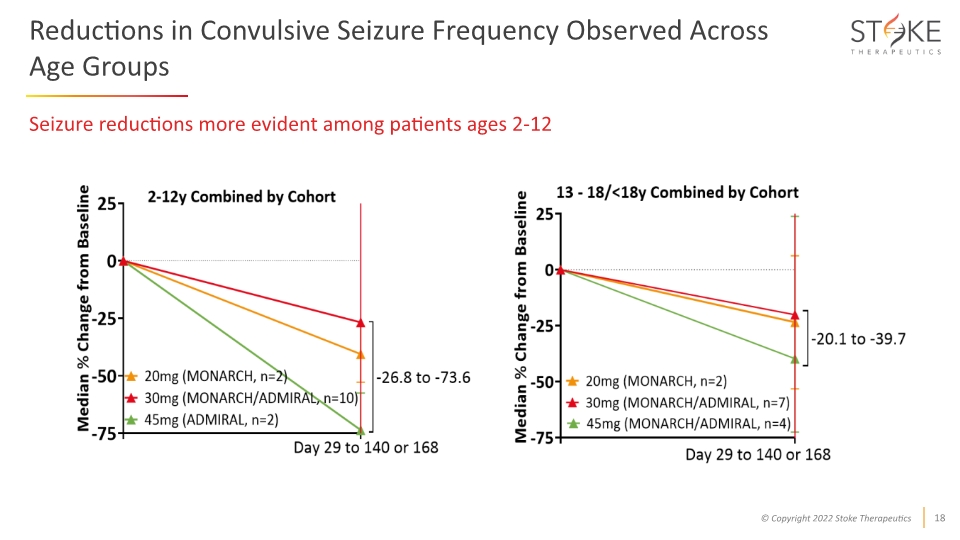
Reductions in Convulsive Seizure Frequency Observed Across Age Groups Seizure reductions more evident among patients ages 2-12

Phase 1/2a Trials of STK-001 for Dravet Syndrome are Ongoing Design Enrollment and dosing ongoing (30mg) Enrollment and dosing ongoing (45mg) Change in seizure frequency, overall clinical status, and quality of life Parallel studies in the US & UK evaluating children and adolescents ages 2 to 18 years old Evaluation of STK-001 (up to 45mg*) Evaluation of STK-001 (up to 70mg) Primary Endpoint Secondary Endpoint Open-Label Extension Safety and tolerability of SAD and MAD dose levels Safety and tolerability of MAD dose levels MAD @45mg: Dosing ongoing Status MAD @70mg: Dosing ongoing *Doses >45mg remain on FDA partial clinical hold Sources: Interim Safety, PK, and CSF Exposure Data from the Phase 1/2a MONARCH Study of STK-001, an Antisense Oligonucleotide (ASO), in Children and Adolescents with Dravet Syndrome (AES 2021). ADMIRAL: A UK Study of the Safety and Pharmacokinetics of Antisense Oligonucleotide STK-001 in Children and Adolescents with Dravet Syndrome (AES 2021) Characterize human pharmacokinetics (PK) and cerebrospinal fluid (CSF) drug exposure
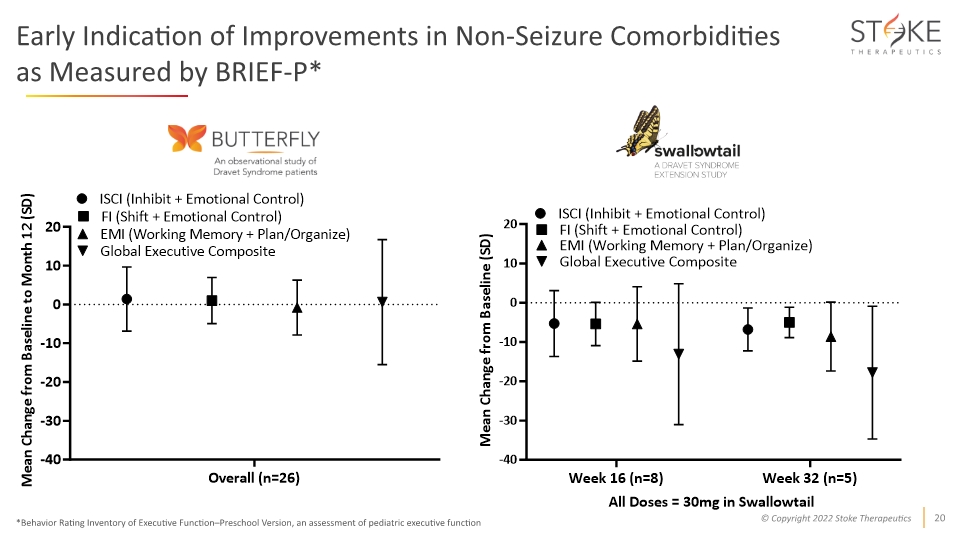
Early Indication of Improvements in Non-Seizure Comorbidities as Measured by BRIEF-P* *Behavior Rating Inventory of Executive Function–Preschool Version, an assessment of pediatric executive function

Conclusions and Next Steps STK-001 on track as the first potential disease-modifying treatment for Dravet syndrome Multiple doses of STK-001 up to 45mg were well-tolerated Majority of patients treated with multiple doses of STK-001 experienced a reduction in seizures The effects were more pronounced at higher doses and among younger patients Continued progress toward identifying optimal dose level and frequency with dose-dependent responses observed between 30mg and 45mg Phase 1/2a studies ongoing, including an expanded 45mg MAD cohort in MONARCH and dosing in the 70mg MAD cohort in ADMIRAL. Expansion of 70mg cohort planned, pending safety review. Preliminary analysis from a small cohort of patients in SWALLOWTAIL (30mg) showed reductions in seizure frequency were maintained and an early indication of improvements in non-seizure comorbidities as measured by BRIEF-P Additional data anticipated in 2023 from the 45mg and 70mg multiple dose cohorts

Closing Remarks Edward M. Kaye, M.D. Chief Executive Officer

Acknowledgements

Q&A