Exhibit 99.2
PHERECYDES PHARMA
Limited company (société anonyme) with a Board of Directors and capital of €7,939,179
Registered office: 22, Boulevard Benoni Goullin – 44200 Nantes, France
493 252 266 Nantes Trade and Companies Register
2022 ANNUAL FINANCIAL REPORT
1
STATEMENT BY THE PERSON RESPONSIBLE FOR THE 2022 ANNUAL FINANCIAL REPORT
(Article 222-3-4° of the AMF General Regulation)
“I certify that the information contained in this financial report is, to the best of my knowledge, in accordance with the facts and contains no omission likely to affect its scope.”
“I certify that, to the best of my knowledge, the accounts have been prepared in accordance with the applicable set of accounting standards and give a true and fair view of the assets, liabilities, financial position and profit or loss of the Company, and that the accompanying management report presents a true and fair view of the development of the Company’s business, profit or loss and financial position, together with a description of the principal risks and uncertainties that it faces.”
Chief Executive Officer
Thibaut du Fayet
2
PHERECYDES PHARMA
Limited company (société anonyme) with a Board of Directors and capital of €7,939,179
Registered office: 22, Boulevard Benoni Goullin – 44200 Nantes, France
493 252 266 Nantes Trade and Companies Register
MANAGEMENT REPORT
FOR THE FISCAL YEAR ENDED DECEMBER 31, 2022
Dear Shareholders,
We have called this Annual General Meeting of Shareholders in accordance with the articles of association and the French law on commercial companies, to report to you on the activities of Pherecydes Pharma (hereinafter the “Company”) during the fiscal year ending December 31, 2022, the results of these activities and future outlooks, and to submit the annual financial statements for said fiscal year for your approval.
The notices of meeting required by law will be regularly sent to you, and all the documentation provided for by applicable regulations will be made available to you within the time frame established.
TABLEOFCONTENTS
CHAPTER 1 - | Activity of the Company and development of its business during the fiscal year ended December 31, 2022 | 5 | ||||
1.1 | General presentation of the Company’s activity | 5 | ||||
1.2 | Status fo the activity and analysis of the development of the business during the year 2022 | 6 | ||||
1.3 | Results of the activity, progress made and difficulties encountered | 15 | ||||
1.3.1 | Economic and financial results | 15 | ||||
1.3.2 | Results for the fiscal year ended December 31, 2022 | 16 | ||||
1.4 | Research and development activities | 16 | ||||
1.5 | Polluting or risky activities | 16 | ||||
1.6 | Main risks and uncertainties facing the company and financial risk management | 17 | ||||
1.6.1 | Liquidity risks | 17 | ||||
1.6.2 | Risks related to historical and future losses | 17 | ||||
1.6.3 | Risks of impairment of intangible assets | 18 | ||||
1.6.4 | Risks relating to shares and other financial instruments | 18 | ||||
1.6.5 | Dilution risks | 19 | ||||
1.7 | Significant events between the balance sheet date and the reporting date | 19 | ||||
1.8 | Foreseeable developments and outlook | 20 | ||||
CHAPTER 2 - | Subsidiaries and equity investments | 23 | ||||
2.1 | Subsidiaries of the Company | 23 |
3
2.2 | Acquisition of significant shareholdings in companies headquartered in France or acquisition of control of such companies | 23 | ||||
CHAPTER 3 - | Information on payment terms to suppliers and customers | 24 | ||||
CHAPTER 4 - | Total dividends distributed over the last three years | 25 | ||||
CHAPTER 5 - | Five year summary of results | 26 | ||||
CHAPTER 6 - | Breakdown of share capital and treasury shares | 27 | ||||
6.1 | Breakdown of share capital during the last fiscal year | 27 | ||||
6.2 | Securities giving access to the share capital | 27 | ||||
6.3 | Crossing of statutory ownership thresholds | 28 | ||||
6.4 | Employee shareholding | 28 | ||||
6.5 | Information on treasury shares | 29 | ||||
CHAPTER 7 - | Transactions carried out by managers on their shares | 31 | ||||
CHAPTER 8 - | Compensation and benefits granted to corporate officers and shareholding | 32 | ||||
8.1 | Compensation and benefits granted to corporate officers | 32 | ||||
8.2 | Shareholdings of corporate officers | 34 | ||||
CHAPTER 9 - | Special report on stock options and free shares | 35 | ||||
9.1 | Share subscription or purchase options | 35 | ||||
9.2 | Free share allocations | 35 | ||||
CHAPTER 10 - | Appointment of the Statutory Auditors | 38 | ||||
10.1 | Statutory Auditors | 38 | ||||
10.2 | Statutory Auditors who resigned, were dismissed or were not reappointed during the fiscal year ended December 31, 2022 | 38 | ||||
CHAPTER 11 - | List of related-party agreements and current agreements | 39 | ||||
11.1 | New related-party agreements entered into during fiscal year 2022 | 39 | ||||
11.2 | New related-party agreements entered into since the end of fiscal year 2022 | 39 | ||||
11.3 | Related-party agreements approved by the General Meeting of Shareholders, but which continue to be in effect during fiscal year 2022 | 39 | ||||
11.4 | Sureties, endorsements and guarantees given by the Company to third parties | 39 | ||||
11.5 | Agreements between a corporate officer or a shareholder holding more than 10% of the voting rights of the Company and a subsidiary, excluding current agreements | 39 | ||||
CHAPTER 12 - | Other information | 40 | ||||
CHAPTER 13 - | Report of the Board of Directors | 41 |
4
CHAPTER 1— ACTIVITY OF THE COMPANY AND DEVELOPMENT OF ITS BUSINESS DURING THE FISCAL YEAR ENDED DECEMBER 31, 2022
Below you will find the information required pursuant to Articles L.232-1 II and R.225-102 of the French Commercial Code.
| 1.1 | GENERALPRESENTATIONOFTHE COMPANY’SACTIVITY |
Founded in 2006, Pherecydes Pharma is a French biotechnology company that aims to develop new solutions to fight complicated and/or resistant bacterial infections. Pherecydes Pharma specializes in the treatment of bacterial infections with bacteriophages (or phages), natural viruses capable of fighting antibiotic-resistant bacteria.
As presented below, Pherecydes Pharma’s portfolio consists of assets for which the first phase II clinical trials began in spring 2022. With CPP (Comités de Protection des Personnes – institutional review board) approval obtained in February 2022, the Company announced the enrollment of the first patient in the PhagoDAIR phase II clinical trial on June 15, 2022.
An initial market launch of the bacteriophages is expected in 2026 (PhagoDAIR study, Staphylococcus aureus program).
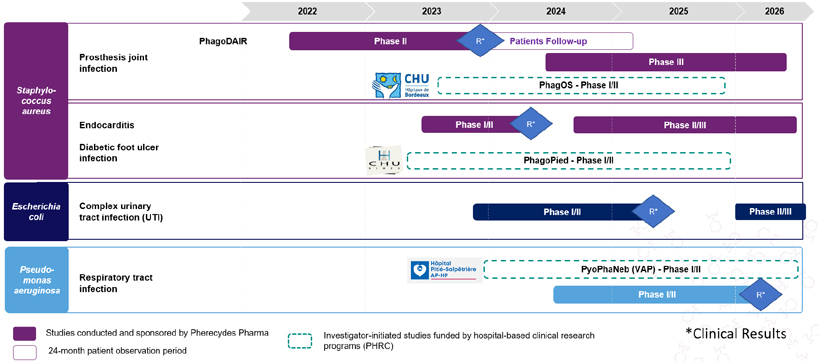
The Staphylococcus aureus program, a bacterium considered a high global priority by the World Health Organization (WHO), is the Company’s flagship program. It includes, at this stage, three clinical trials: PhagoDAIR for which Pherecydes Pharma is the sponsor, as well as PhagOS and PhagoPied, which are financed by Hospital Clinical Research Programs (“Programmes Hospitaliers de Recherche Clinique – PHRC”) and whose sponsors will be the University Hospital Centers of Bordeaux and Nîmes. The latter two trials are expected to start in the third and second quarters of 2023 respectively.
The Company enrolled its first patient on June 15, 2022 in the PhagoDAIR phase II clinical trial and received a unanimous recommendation from the Data Safety Monitoring Board (DSMB) on November 29, 2022 to continue without modification this phase II clinical study on osteoarticular infections of prostheses caused by Staphylococcus aureus (S. aureus).
5
According to the Company, PhagoDAIR is the world’s first clinical study on precision phagotherapy in this indication.
In addition to the PhagoDAIR study, the company aims to launch, as a sponsor, another clinical study (Phase 1 PK) on infective endocarditis, for which the level of medical need is very high, as it is associated with a one-year mortality rate of around 30-40%.
| 1.2 | STATUSOFTHEACTIVITYANDANALYSISOFTHEDEVELOPMENTOFTHEBUSINESSDURINGTHEYEAR 2022 |
| • | A pronounced acceleration in compassionate treatment with >75 patients treated to date |
In line with the authorizations of the French National Agency for the Safety of Medicines (Agence Nationale de Sécurité du Médicament “ANSM”), more than 75 patients have treated with Pherecydes’ precision phagotherapy since early 2021.
These therapies were administered in nine hospitals, and mainly focused on S. aureus and P. aeruginosa infections. Several indications were targeted, with a high proportion of osteoarticular infections of prostheses.
Pherecydes Pharma’s phages, administered by different routes (intra-articular, intravenous, bronchoalveolar nebulization, etc.), have demonstrated excellent tolerance with no reported side effects, and an initial clinical benefit through the control of infection for nearly 75% of evaluable patients.
Pherecydes Pharma and CEA’s PhagECOLI project receives a €2 million grant from Bpifrance
On January 3, 2022, the Company and the CEA announced the selection of the PhagECOLI project submitted as part of the Emerging Infectious Diseases and New Radiological, Biological and Chemical Threats’ Call for Expressions of Interest. This project will receive a grant from Bpifrance of €2 million – 50% of its estimated cost – of which Pherecydes Pharma will receive 80% and the CEA will receive 20%.
The PhagECOLI project draws on the complementary expertise and experience of Pherecydes Pharma and the CEA. The aim of this three-year project is to offer new treatments for hard-to-treat or resistant E. Coli infections thanks to precision phagotherapy. To provide the best approach to the therapeutic need, Pherecydes Pharma and the CEA will develop a new generation of tools to measure the efficacy of anti-E. Coli phages on the patient’s bacterial strain. These new tools could then become the basis for a brand-new generation of phagograms1 for the various target bacteria. This financial support will also allow Pherecydes Pharma to accelerate the development of its anti-E. coli phages.
6
Favorable opinion from the institutional review board (CPP) to initiate the PhagoDAIR study
On February 7, 2022, the Company announced that it had received a favorable opinion from the Ile de France III CPP for the PhagoDAIR study, a phase II pilot clinical study in the treatment of osteoarticular infections of prostheses caused by Staphylococcus aureus.
PhagoDAIR is the world’s first phagotherapy study conducted in this indication. Its protocol was already approved by the ANSM in December 2021.
It will be conducted in France, Spain and other European countries (Germany and the Netherlands) on 64 patients with a knee or hip joint infection due to S.-aureus, divided into one group who will be treated with phagotherapy and a control group who will receive a placebo, in addition to the standard of care. The patients treated with phagotherapy will receive anti-Staphylococcus aureus phages that are active on their strain, selected thanks to Pherecydes Pharma’s phagogram. The standard of care will comprise the surgical procedure called DAIR (Debridement, Antibiotics, Implant Retention) combined with a suppressive antibiotic therapy. An assessment will be carried out 12 weeks after the phagotherapy, and patients will continue to be monitored for two years.
The favorable opinion of the CPP paves the way for the start of enrollment in the study at French centers in spring 2022. Results are expected in the summer of 2023 and follow-up will continue until the first half of 2025. Depending on the preliminary results of phase I/II, Pherecydes Pharma will conduct a phase III study on the same indication, which could start in late 2024.
Pherecydes Pharma announces a change in its governance structure to become a company with a Board of Directors
On February 24, 2022, the Company announced it was changing its governance structure by adopting a board of directors and general management instead of the existing Supervisory Board and Management Board.
The setting up of a board of directors and general management corresponds to Pherecydes Pharma’s wish to simplify its operating decision procedures and more readily respond to the rapidity, efficacy and reactivity requirements specific to the biotechnology sector while ensuring the continuity of the Company’s leadership and administration, with the mandates of the current representatives having been renewed. The Company will thus be able to confidently address the next decisive stages of its activity, in particular the implementation of the PhagoDAIR clinical study.
As a result, the ordinary and extraordinary general meeting of the shareholders, which was held on May 19, 2022, was asked to:
| • | modify the Company’s method of management and administration by adopting a board of directors and general management; |
| • | correspondingly amend the Company’s articles of association; |
7
| • | appoint the current members of the supervisory board as the first members of the board of directors. |
Following this General Meeting of Shareholders, the Board of Directors decided on general management’s method of exercise. The newly formed Board of Directors was thus asked to:
| • | approve the separation of the functions of the President of the board of directors and the chief executive officer; |
| • | appoint Mr. Guy-Charles Fanneau de La Horie, the current President of the management board, as chief executive officer; and |
| • | appoint Didier Hoch, the current President of the Supervisory Board, as President of the board of directors. |
The separation of the functions of President of the board of directors and chief executive officer is in line with the Autorité des marchés financiers‘s (AMF) 2021 report on “corporate governance and the executive compensation of listed companies” prepared pursuant to Article L.621-18-3 of the French Monetary and Financial Code.
The Company also announced that Philippe Rousseau, Chief Operating Officer, and a member of the management board, left the Company to pursue other ventures.
Pherecydes Pharma extends its intellectual property in China with a first patent granted for its anti-Pseudomonas aeruginosa phages in this territory
On March 8, 2022, the Company announced the granting of a patent for its anti-Pseudomonas aeruginosa phages by the Chinese patent office.
This second patent granted in China, following the one granted the preceding December for its anti-Staphylococcus aureus phages, gives the Company protection until the end of 2034 on this vast market.
Pherecydes Pharma and BIOASTER join forces in the treatment of bacteremia
On March 11, 2022, the Company and BIOASTER, the first Institute for Technological Innovation in Microbiology, announced the implementation of research collaboration to use phages to treat bacteremia (blood infections) caused by the S. aureus, E. Coli and Pseudomonas aeruginosa bacteria.
Bacteremia, defined as the presence of viable bacteria in the bloodstream, represents the world’s second greatest cause of bacterial infection, with around 1.5 million deaths associated with this pathology per year In the United States alone, the Centers for Disease Control and Prevention (CDC) estimates that up to 1.7 million people develop bacteremia every year. S. aureus is the most commonly identified pathogenic agent, both within and outside a hospital setting. Age-adjusted mortality assessments show that mortality due to S. aureus bacteremia is higher than that of AIDS, tuberculosis, or viral hepatitis and similar to the mortality rate of breast or prostate cancer. E. Coli and Pseudomonas aeruginosa also represent major causes of antibiotic-resistant bacteremia.
8
Within the framework of this collaboration, Pherecydes Pharma will draw on BIOASTER’s excellence in developing preclinical models that are relevant to these infections to test the efficacy of the proprietary phages selected by Pherecydes Pharma in these pathologies.
Pherecydes Pharma obtains a new patent for its anti-Pseudomonas aeruginosa phages in Europe
On March 22, 2022, the Company announced the granting of a new patent for its anti-Pseudomonas aeruginosa phages by the European Patent Office (EPO).
This new patent granted in Europe gives the Company protection until 2034 on this vast market.
Pherecydes Pharma announces a partnership with Navarrabiomed to evaluate its anti-S. aureus phages in a new indication
On March 31, 2022 Pherecydes Pharma announced the signing of a research agreement with Navarrabiomed to evaluate its anti-S. aureus phages in the treatment of endocarditis.
Endocarditis is an infection of the endocardium (inner layer of the heart), the heart valves or the aorta, which inevitably leads to hospitalization. This infection is most often caused by bacteria, mainly Staphylococcus aureus. This bacterium is among the six most deadly germs1 in antibiotic resistance and is on the WHO priority pathogens list2 for R&D of new antibiotics.
The objective of the Navarrabiomed research team is to test Pherecydes Pharma’s phages on a porcine model of S. aureus endocarditis.
This is the third indication targeted by the company’s anti-Staphylococcus aureus phages, after bone and joint infections of prostheses and diabetic foot ulcers.
Pherecydes Pharma announces the appointment of Thibaut du Fayet as Chief Operating Officer and member of the Management Board
On April 7, 2022 Pherecydes Pharma announced that Thibaut du Fayet, an expert in the biotech sector, joined the Management Board of Pherecydes Pharma as Chief Operating Officer.
Thibaut du Fayet brings to the company over 20 years of experience in the pharmaceutical and life sciences industry. After obtaining an MBA from ESSEC Business School in 1992, he was Senior Managing Director at Bossard-Gemini for six years. From 2000, he held management positions first at Rhône Poulenc as Strategy Director for four years, and then at BioMerieux where he was also Strategic Marketing Director for four years. In 2008, he joined Transgene where he held the position of VP Corporate Development, Alliances & Programs Management for 14 years.
Through his various leadership roles in development, strategy, business development, and program management, he has acquired important and diverse skills in the pharmaceutical industry.
| 1 | Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis; The Lancet, January 19, 2022 |
| 2 | https://www.who.int/fr/news/item/27-02-2017-who-publishes-list-of-bacteria-for-which-new-antibiotics-are-urgently-needed |
9
Pherecydes Pharma announces the first international approval of a compassionate treatment with its phages – The Swedish Medical Products Agency (SMPA) has given its approval to treat a case of osteoarticular infection of a prosthesis with the Company’s anti-S. aureus phages
On April 28, 2022, Pherecydes Pharma announced the first international approval of a compassionate treatment with its phages, in Sweden.
The Swedish Medical Products Agency (SMPA) has given its approval to treat a case of osteoarticular infection of a prosthesis with Pherecydes Pharma’s anti-S. aureus phages.
With the compassionate treatments that continue to be provided in France, Pherecydes Pharma has now treated more than 75 patients in this context.
The general meeting of the shareholders of Pherecydes Pharma approves the change in its governance to a company with a Board of Directors – The new Board of Directors has appointed Didier Hoch as Chief Executive Officer and Thibaut du Fayet as Chief Operating Officer
On May 23, 2022, Pherecydes Pharma announced that the Combined General Meeting of Shareholders (CGM) held on May 19, 2022 was able to deliberate, with the quorum having been reached.
The CGM adopted all the proposed resolutions, except for the twenty-third resolution which was rejected, in accordance with the Board’s recommendations.
In particular, the company’s shareholders approved the change in the company’s method of management and administration by adopting a Board of Directors and general Management.
The current members of the Supervisory Board were appointed as the first directors of Pherecydes Pharma.
The Board of Directors is thus composed of seven members, two of whom are independent.
At the end of the general meeting of shareholders, the Board of Directors adopted the following decisions:
| • | the Board of Directors has taken note of the termination of the functions of Mr. Guy-Charles Fanneau de La Horie, previously President of the Management Board, who did not wish, for personal reasons, to assume the mandate of Chief Executive Officer in the new governance; |
| • | consequently, and contrary to what had been announced, the Board has not decided to adopt, at this stage, a separation of the functions of Chief Executive Officer and President of the Board of Directors; |
| • | Mr. Didier Hoch, previously President of the Supervisory Board, was appointed Chief Executive Officer; and |
| • | Mr. Thibaut du Fayet, previously a member of the Management Board, was appointed Chief Operating Officer. |
10
The Board of Directors thanked Mr. Guy-Charles Fanneau de La Horie who has been one of the driving forces in the development of the company, both operationally and financially, notably with the company’s successful IPO in 2021. Mr. Guy-Charles Fanneau de La Horie will continue to contribute his expertise, on a transitional basis, within the framework of a service contract.
There are also plans to adopt, at a later stage, a separation of the functions of Presidentof the Board of Directors and Chief Executive Officer, and thus to appoint a new Chief Executive Officer.
Pherecydes Pharma obtains Early Access Approval (Autorisation d’Accès Compassionnel—AAC) from the ANSM for its anti-Staphylococcus aureus phages – This is the first AAC for a treatment in patients who have failed antibiotic therapy, particularly expected in the context of the fight against antibiotic resistance.
On May 30, 2022, Pherecydes Pharma announced that it had been granted Early Access Approval (AAC) from the ANSM for its anti-Staphylococcus aureus (S. aureus) phages.
The new AAC system (formerly nominative authorization for early access, or ATU), introduced in July 2021, allows certain categories of sick patients in France with no therapeutic solutions to benefit from drugs yet to be granted marketing approval (Autorisation de mise sur le marché – AMM).
As of May 30, 2022, Pherecydes Pharma had treated more than 60 patients as part of compassionate treatment under the supervision of the ANSM, excluding AAC status. These phages, administered by various routes (intra-articular, intravenous, bronchoalveolar nebulization, etc.), have demonstrated excellent tolerance with no reported side effects.
The AAC scheme allows Pherecydes Pharma to make its anti-S. aureus phages available to larger populations and thus generate the first revenue in the Company’s history.
Pherecydes Pharma announces the enrollment of the first patient in the PhagoDAIR phase II study in the treatment of osteoarticular infections of prostheses caused by Staphylococcus aureus.
On June 15, 2022, Pherecydes Pharma announced the enrollment of the first patient in the PhagoDAIR phase II clinical trial.
PhagoDAIR is the world’s first study of phagotherapy conducted in osteoarticular infections of prostheses caused by Staphylococcus aureus (S. aureus). Its protocol was approved by the ANSM in December 2021 and by the CPP in February 2022.
Since then, Pherecydes Pharma has been actively recruiting patients for this clinical trial, the first of which has just been enrolled in France at the Hospices Civils de Lyon (Hospices Civils de Lyon—HCL).
The study, also conducted in other European countries, includes 64 patients with a knee or hip joint infection due to S. aureus, divided into one group treated with phagotherapy and a control group receiving a placebo, in addition to the standard of care. The patients treated with phagotherapy receive anti-S. aureus phages that are active on their strain, selected thanks to Pherecydes Pharma’s phagogram.
11
The standard of care comprises the surgical procedure called DAIR (Debridement, Antibiotics, Implant Retention) combined with a suppressive antibiotic therapy. An assessment is carried out 12 weeks after the phagotherapy, and patients continue to be monitored for two years. Follow-up of patients continues until the first half of 2025.
Depending on the preliminary results of phase II, Pherecydes Pharma will conduct a phase III study on the same indication, which could start in 2024.
Positive pre-clinical results for inhaled phagotherapy presented to the Reanimation 2022 conference – A pre-clinical study demonstrates the benefits of inhaled Pherecydes Pharma phages in treating mechanical ventilation-associated pneumonia
On June 27, 2022, Pherecydes Pharma announced that the results of a pre-clinical study conducted with its phages were presented at the Reanimation 2022 conference organized by the French Intensive Care Society (Société de Réanimation de Langue Française – SRLF) and held in Paris from June 22 to 24, 2022.
Antoine Guillon, an intensive care doctor at the University Hospital of Tours and researcher at the Research Center for Respiratory diseases at the University of Tours (Étude des Pathologies Respiratoires Inserm-Université de Tours – UMR1100), presented the results of the study during a Flash Com session entitled “Inhaled bacteriophage therapy in a porcine model of pneumonia caused by Pseudomonas aeruginosa during mechanical ventilation.”
The study was carried out within the framework of the Pneumophage project, associating the UMR1100 and the company Diffusion Technique Française, aimed at demonstrating the effectiveness of inhaled phagotherapy in treating mechanical ventilation-associated infections. To this end, the UMR1100 researchers in Tours developed a porcine model of pneumonia caused by P. aeruginosa incorporating the essential characteristics of the human disease. The work undertaken showed that the use of Pherecydes Pharma’s anti-P. aeruginosa phages significantly reduced the lung bacterial load (1.5-log reduction, p < 0.001).
The results obtained also demonstrate:
| • | the feasibility of delivering large quantities of active phages by nebulization during mechanical ventilation; |
| • | the rapid control of the infection in situ in a respiratory model close to humans. |
This demonstration in a highly translational model paves the way for pulmonary phagotherapy, notably in intensive care units where the occurrence of mechanical ventilation-associated P. aeruginosa infections is higher than in other units. The P. aeruginosa bacterium is a major cause of mechanical ventilation-associated pneumonia, a pathology with a relapse rate of 40% and a high mortality rate of around 20%.
12
Pherecydes Pharma announces an initial registration of its phagogram as an in vitro diagnostic test in accordance with EC directives – This vital tool for precision medicine thus meets regulatory requirements and clinicians’ expectations
On September 12, 2022, Pherecydes Pharma announced the registration of its phagogram as an in vitro diagnostic test (“Phagogram 1.5”) in accordance with Directive 98/79/EC.
The phagogram is an in vitro diagnostic test to verify the sensitivity of patients’ bacterial strains to Pherecydes Pharma’s phages.
Pherecydes Pharma is developing its phagogram for three target bacteria: Staphylococcus aureus, Escherichia coli and Pseudomonas aeruginosa. These bacteria, which are the most resistant and dangerous according to the WHO, alone account for more than two thirds of hospital-acquired resistant infections in industrialized countries3.
The EC registration of the Phagogram 1.5 was made possible by the setting up of a new specialized laboratory in Nantes; this new laboratory initially ensured the development and validation of the in vitro diagnostic test following a very stringent process from a technical, quality, and regulatory perspective. It is now responsible for carrying out phagograms for patients enrolled in our clinical studies and patients undergoing compassionate treatment.
The Phagogram 1.5 program is the first stage in a broader strategic program whose goal is to develop a new generation phagogram, “Phagogram 2.0,” in collaboration with the CEA (see Pherecydes Pharma press release from January 3, 2022). This program aims to significantly shorten the lead time required for a diagnosis and thus offer phagotherapy in all types of indications, whether acute or chronic.
Pherecydes Pharma announces the creation of an International Medical Advisory Board – This group of infectious disease experts will contribute to the design and implementation of the Company’s clinical development strategy in phagotherapy
On September 15, 2022, Pherecydes Pharma announced the establishment of a Medical Advisory Board comprising prominent international scientific and clinical experts in infectious diseases.
This Board will support Pherecydes Pharma in consolidating its clinical development strategy in phagotherapy. It will meet twice a year, with its first meeting on September 28, 2022.
The members of the Medical Advisory Board are:
| • | Dr. Saima Aslam, Associate Professor of Medicine, Infectious Disease Specialist at UC San Diego, California, USA. |
| • | Prof. Marc Bonten, President of the Infection & Immunity strategic research program at UMC Utrecht, the Netherlands. |
| • | Prof. Tristan Ferry, Deputy Head of the Infectious and Tropical Diseases department, Hospices Civils de Lyon, and President of the national CRIOAc scientific committee, France. |
| • | Prof. Yok-Ai Que, Associate Professor in Intensive Care Medicine at Bern University Hospital, Switzerland. |
| • | Prof. Martin Witzenrath, Head of Department of Respiratory Diseases and Intensive Care at the Charité Universitätsmedizin Berlin, Germany. |
| 3 | Opatowski M—Hospitalisations with infections related to antimicrobial-resistant bacteria from the French nationwide hospital discharge database, 2016 |
13
Pherecydes Pharma successfully completes €3.1 million fundraising
On September 22, 2022, Pherecydes Pharma announced the success of its capital increase for a total amount of €3.1 million, comprising €2.6 million from institutional investors and €0.5 million from individuals.
The Company plans to use the net proceeds of this fundraising to partially finance:
| 1) | for around 50%, the clinical development of anti-Staphylococcus aureus (S. aureus) phages notably including: |
| • | the ramping up of the PhagoDAIR phase II study in France and Europe with the opening of new clinical centers, mainly in Spain and the Netherlands, through to the key criterion assessment stage; |
| • | the preparation and initiation of a Phase II study in a clinical indication with substantial medical needs; |
| • | the launch and management of studies (PHRC) for which the Company is not the sponsor (PhagoPied, Phagos) via the conducting of Phagograms and various associated costs; |
| 2) | for around 30%, the development of anti-Pseudomonas aeruginosa phages notably including: |
| • | the initiation of a regulatory toxicology study; |
| • | the preparation and initiation of a (phase II) clinical study in an indication associated with respiratory infections with substantial medical needs; |
| 3) | for around 10%, perfecting manufacturing and quality control processes, producing GMP batches of E. Coli phages and completing preclinical development; |
| 4) | for around 10%, research work associated with the identification of a new bacterial target, as well as the Company’s other running costs. |
Pherecydes Pharma announces the positive recommendation of the DSMB for the continuation of its phase II clinical study PhagoDAIR in osteoarticular infections of prostheses caused by Staphylococcus aureus
On November 29, 2022, Pherecydes Pharma announced that it had received a unanimous recommendation from the Data Safety Monitoring Board (DSMB) to continue without modification the phase II clinical study PhagoDAIR in osteoarticular infections of prostheses due to Staphylococcus aureus (S. aureus).
The DSMB is an independent expert committee responsible for the open-label review of the PhagoDAIR study safety data and will meet twice a year during the study. Following its first meeting, the committee recommended the continuation of the study without modification.
Pherecydes Pharma: Minutes of the Combined General Meeting of Shareholders
On December 15, 2022, Pherecydes Pharma announced that the Combined General Meeting of the shareholders (CGM) was able to deliberate, with the quorum having been reached. The shareholders followed all the voting recommendations of the Board of Directors.
14
Following the Meeting, the Board of Directors adopted the following decisions:
| • | The functions of Chief Executive Officer and President of the Board of Directors were separated as planned following the CGM on May 19, 2022; |
| • | Mr. Thibaut du Fayet, previously Chief Operating Officer, was appointed Chief Executive Officer; |
| • | Mr. Didier Hoch, previously President and Chief Executive Officer, was appointed President of the Board of Directors. |
| 1.3 | RESULTSOFTHEACTIVITY,PROGRESSMADEANDDIFFICULTIESENCOUNTERED |
| 1.3.1 | Economic and financial results |
Simplified income statement (in euros) | 2022 | 2021 | ||||||
Operating income | 3,167,235 | 2,200,380 | ||||||
Operating expenses | 9,857,278 | 6,521,075 | ||||||
Operating results | (6,690,043 | ) | (4,320,694 | ) | ||||
Financial results | (63,504 | ) | (46,346 | ) | ||||
Extraordinary results | 511,125 | 189,470 | ||||||
Net results (loss) | (4,854,342 | ) | (3,189,057 | ) | ||||
On December 31, 2022, total operating income, essentially consisting of capitalized production associated with the Staphylococcus aureus (S. aureus) and Pseudomonas aeruginosa (P. aeruginosa) programs, was €3,167 thousand compared with €2,200 thousand at December 31, 2021, an increase of 44%. This growth was notably a result of the increase in R&D activity associated with these programs between the two years in question.
On December 31, 2022, operating expenses totaled €9,857 thousand over the year, up 50% from the fiscal year 2021 figure of €6,521 thousand. This growth was essentially due to the 125% increase in subcontracting charges to €2,357 thousand in 2022, and reflects the expenses related to the subcontracting of the PhagoDAIR phase II clinical trial.
The annual operating loss was €6,690 thousand as of December 31, 2022 versus a loss of €4,321 thousand at December 31, 2021, an increase of 55% between the two years, primarily as a result of the increase in operating expenses from one year to the next.
Exceptional results totaled €551 thousand at December 31, 2022 compared with €189 thousand the previous year, reflecting income from management operations related to debt write-off on the PHAGOSCLIN project.
The Research Tax Credit amounted to €1,384 thousand at December 31, 2022 versus €989 thousand the previous year.
Net loss stood at €4,854 thousand in 2022 compared with a loss of €3,189 thousand the previous year.
15
Financial structure and cash position
On December 31, 2022, the Company had a cash position of €1,035 thousand, versus €5,357 thousand at December 31, 2021. Shareholders’ equity stood at €7,522 thousand on December 31, 2022 compared with €9,228 thousand at December 31, 2021.
On September 22, 2022, Pherecydes Pharma successfully carried out a capital increase of €3.1 million, comprising €2.6 million from institutional and historical investors and €0.5 million from individuals, via the PrimaryBid platform.
On February 17, 2023, Pherecydes Pharma announced the completion of the capital increase announced on February 15, 2023 in connection with the publication of its proposed merger with the company ERYTECH (see press release from February 15, 2023 and section 1.7). This capital increase for €1.5 million was fully subscribed by Auriga IV Bioseeds, Ouest Ventures III and the pool of shareholders represented by Guy Rigaud, historical shareholders of Pherecydes Pharma.
The funds raised through this capital increase will provide Pherecydes Pharma with sufficient resources to finance its cash requirements until the completion of the merger expected to take place no later than June 30, 2023.
| 1.3.2 | Results for the fiscal year ended December 31, 2022 |
The Company’s result for the year was a loss of €4,854,342.477.
| 1.4 | RESEARCHANDDEVELOPMENTACTIVITIES |
The Company has developed research, pre-clinical and clinical testing programs to develop optimized bacteriophages to combat three prevalent antibiotic-resistant diseases.
The importance of its research and development activity is reflected in the predominance in the Company’s workforce of employees with high-level scientific training and dedicated to laboratory work.
As of December 31, 2022, 28 of the company’s employees are involved in research and development, representing 90% of the total workforce at that date.
| 1.5 | POLLUTINGORRISKYACTIVITIES |
Pursuant to the provisions of Article L.225-102-2 of the French Commercial Code, below you will find a description of polluting or risky activities.
The Company does not operate a classified facility and is therefore not subject to regulations on classified facilities and technological risks, but it does use hazardous chemical and biological products. Regular inspections are carried out on the Company’s laboratories. Although the Company believes that it meets its current legal obligations relating to the environment, non-compliance would expose it to criminal and administrative penalties, including the suspension or revocation of authorizations and approvals required for its activities.
16
Since the creation of the Company, no incidents have occurred in the context of polluting or risky activities. The Company therefore believes that it continues to meet the legal obligations relating to these activities in force as of the date of this report.
Compliance with applicable environmental, health and safety regulations imposes costs on the Company and may require significant investment, particularly in the future if regulatory changes require the use of new equipment or procedures.
In addition, although the Company believes that the safety procedures it implements for the storage, use, transportation and disposal of hazardous, chemical, and biological products and industrial and hospital waste are in compliance with applicable regulations, the risk of accidents or accidental contamination cannot be eliminated. In the event of an accident or contamination, the Company could be held liable, which could result in it incurring potentially significant costs to compensate the victims and remedy the damage.
| 1.6 | MAINRISKSANDUNCERTAINTIESFACINGTHECOMPANYANDFINANCIALRISKMANAGEMENT |
| 1.6.1 | Liquidity risks |
Since its founding, the Company has financed its growth by strengthening its equity through successive capital increases, loans, and obtaining public funding for innovation (grants, research tax credit, etc.).
Significant expenses have been incurred in relation to the research and development of the Company’s products and to obtaining the various regulatory approvals for their marketing. To date, this has resulted in negative cash flows from operating activities.
As of December 31, 2022, loans and debt amounted to €2.6 million, comprising:
| • | a €2.2 million government-backed loan, the first repayment of which will be made in July 2023 followed by quarterly repayments for four years; and |
| • | an innovation (R&D) loan of €0.4 million granted by BPI France in October 2020 and repaid quarterly over five years since June 2022. |
| 1.6.2 | Risks related to historical and future losses |
Since its founding at the end of 2006, the Company has recorded operating losses every year. These losses are mainly due to research and development costs, in particular the costs of strengthening the research and development teams, incurred by the Company to develop its technologies.
If the Company fails to achieve sufficient revenue growth over the next few years, it could experience further operating losses, including due to:
| • | Research and development activities, mainly; |
| • | Marketing and development costs for its products; |
| • | Its international development. |
A significant increase in these expenses could have a material adverse effect on the Company, its business, financial position, results, development, or outlook.
17
If the Company fails to generate taxable income, it will not be able to use the tax loss carryforwards generated by the Company since its founding, totaling €24,087,822 at the closing date of fiscal year 2022.
| 1.6.3 | Risks of impairment of intangible assets |
As of December 31, 2022, intangible assets represented a net amount of €9,072 thousand, and the net balance sheet total at that date stood at €13,216 thousand.
As of December 31, 2022, the Company has capitalized research and development expenses for a gross value of €5,555 thousand. The capitalized development costs relate to clearly individualized subjects with a high probability of technical success and commercial profitability or economic viability. Pherecydes Pharma has considered as eligible for capitalization the development in-progress costs of products for which the regulatory authorities have validated the Company’s good manufacturing practices or have given the Company authorization to proceed with human clinical trials and/or Early Access Approval (AAC). The net costs capitalized as assets under construction amount to €5,074 thousand. They mainly correspond to development costs for various assets:
| • | Pseudomonas aeruginosa for €547 thousand; |
| • | Staphylococcus aureus for (€5,602) thousand. |
Other intangible assets correspond to patents for an amount of €123 thousand on December 31, 2022. The Company has four patent families (two for anti-Pseudomonas aeruginosa phages, one for anti-Staphylococcus aureus phages and one for anti-E. coli phages). These patents have been granted in some jurisdictions and are pending in others.
In accordance with accounting methods, these intangible assets are recorded at their initial acquisition cost minus accumulated amortization (straight-line amortization over 20 years) and, where applicable, impairment losses.
The change in value of the Company’s intangible assets depends on its development plan and the success or failure of its programs. Should the ANSM decide to not grant AACs for the anti-Pseudomonas aeruginosa phages, or if it becomes impossible for the Company to make progress on these phages during the clinical development process, it would result in an impairment of these intangible assets. The Company would then have to review the value of these assets and record an impairment corresponding to the total value or a partial amount of these assets. Each program will need to be reviewed independently as part of this evaluation: the failure of one program does not automatically mean the failure of the other. An impairment of these assets could have a material financial impact, resulting in a decrease in the size of the balance sheet and a significant loss on the income statement.
| 1.6.4 | Risks relating to shares and other financial instruments |
The Company does not hold any equity investments or marketable securities.
18
| 1.6.5 | Dilution risks |
As part of its employee profit-sharing policy, the Company has regularly issued financial instruments giving access to its capital (start-up stock options, free shares, etc.).
At December 31, 2022, the following dilution would result if holders were to exercise these securities giving access to capital:
| Number of shares comprising the share capital | In the event of all start-up stock options being exercised | In the event of the definitive acquisition of all the free shares allocated | TOTAL | |||||||||||||
Number ofshares created | 7,221,477 | 615,450 | 58,523 | 673,973 | ||||||||||||
Share capital after issue of new shares | N/A | 7,836,927 | 7,280,000 | 7,895,450 | ||||||||||||
Dilution | N/A | 7.85 | % | 0.80 | % | 8.54 | % | |||||||||
As part of its policy of motivating its employees and corporate officers, the Company may issue new financial instruments giving access to its capital in the future.
In addition, in the context of financing its business development, the Company may issue or be required to issue new securities on the market and thus increase its share capital to finance all or part of its operating needs.
The issue of these financial instruments or new shares would result in additional, potentially significant dilution for the Company’s shareholders.
| 1.7 | SIGNIFICANTEVENTSBETWEENTHEBALANCESHEETDATEANDTHEREPORTINGDATE |
ERYTECH and PHERECYDES announce a strategic combination to create a global leader in phagotherapy
On February 15, 2023, Pherecydes Pharma and ERYTECH Pharma (“ERYTECH”; Nasdaq & Euronext: ERYP), a clinical-stage biopharmaceutical company developing innovative therapies by encapsulating therapeutic drug substances inside red blood cells, announced their proposed strategic combination intended to create a global player in extended phagotherapy and accelerate the development of a portfolio of drug candidates targeting pathogenic bacteria and potential other indications of high unmet medical needs.
The strategic combination would build on the complementary expertise and capabilities of both companies to accelerate the development of extended phagotherapies for antimicrobial resistance, in particular via the PhagoDAIR phase II study conducted by PHERECYDES, as well as other anti-infective fields and therapeutic areas with high unmet medical needs.
19
The cash runway of the new entity resulting from the merger would extend into the third quarter of 2024, with a consolidated cash position of approximately €41 million as of December 31, 2022, and would enable funding of existing and novel programs through multiple clinical milestones.
This transaction would pave the way for the expansion of PHERECYDES development programs in antimicrobial resistance and complete the strategic review process announced by ERYTECH on several occasions since November 2021.
The transaction expected to close at the end of the second quarter of 2023.
The transaction, supported by the key shareholders of PHERECYDES and ERYTECH, will result in former PHERECYDES shareholders holding approximately 49% of the combined entity.
ERYTECH and PHERECYDES intend to merge their operations and relocate all teams to ERYTECH’s premises in Lyon, France, where they will benefit from being located within a major European center for infectious diseases.
Following the announcement of its proposed merger with Erytech, Pherecydes Pharma completes a €1.5 million capital increase reserved to its historical shareholders
On February 17, 2023, Pherecydes Pharma announced the completion of the capital increase announced on February 15, 2023 in connection with the publication of its proposed merger with the Company Erytech.
This capital increase for €1.5 million was fully subscribed by Auriga IV Bioseeds, Ouest Ventures III and the pool of shareholders represented by Guy Rigaud, historical shareholders of Pherecydes Pharma.
The funds raised through this capital increase will provide Pherecydes Pharma with sufficient resources to finance its cash requirements until the completion of the merger expected to take place no later than June 30, 2023.
| 1.8 | FORESEEABLEDEVELOPMENTSANDOUTLOOK |
During fiscal years 2023–2024, the Company intends to complete its merger with ERYTECH, pursue the development of its various assets and, accelerate the commercial ramp-up of precision phagotherapy.
Completion of the merger with ERYTECH Pharma to become a global leader in phagotherapy
As of today’s date, the memorandum of understanding has been unanimously approved by the Boards of Directors of ERYTECH and PHERECYDES.
The Extraordinary General Meetings of Shareholders (“EGM”) of ERYTECH and PHERECYDES will be called to vote on the Proposed Merger. Said meeting is currently expected to be held at the end of the first half of 2023 (or at the beginning of the second half of 2023).
20
The resolutions to approve the Proposed Merger will require, within each company, the positive vote of two thirds of the shareholders present or represented. The ERYTECH EGM will also be called to approve the name change of the new entity, to mark the beginning of a new development stage for the combined activities.
Ambitious clinical and pre-clinical development strategy
| • | Expansion of the PhagoDAIR phase II study and preparation for phase III |
The Company plans to expand its ongoing PhagoDAIR phase II study in patients with knee and hip joint infections due to Staphylococcus aureus (S. aureus) by opening new clinical centers in Europe, with results expected in the first quarter of 2024.
The Company will also prepare the protocol for the phase III study that will follow the completion of the PhagoDAIR phase II study.
| • | Expansion of Pherecydes’ clinical phagotherapy portfolio |
The Company plans to expand its clinical portfolio with two additional phase II studies funded by the Company, one in patients with S. aureus endocarditis, which is expected to begin in mid-2023, and the second in patients with complex urinary tract infections due to Escherichia coli (E. coli), which is expected to begin in the first quarter of 2024. The Company plans to potentially open clinical centers in the United States for these two studies.
| • | Creation of a research and development strategy based on ERYTECH Pharma platforms and expertise |
In particular, the Company plans to develop drug-delivery solutions using red blood cells or red blood cell-derived vesicles from ERYTECH Pharma’s platforms (the ERYCAPS and ERYCEV platforms, respectively).
The Company also intends to leverage ERYTECH Pharma’s formulation and oncology expertise to support phage- and endolysin-based therapeutic approaches, drawing on Erytech’s know-how in protein engineering and in anti-infective fields such as antimicrobial resistance and beyond (such as food, cosmetics, and animal health), or the development of novel carriers.
| • | Expansion of the PHERECYDES portfolio |
The Company plans to expand its portfolio with two new phages, complementary to the three already existing (S. aureus, P. aeruginosa, E. Coli). These phages are critical in developing a comprehensive target portfolio in the fight against resistant bacterial infections.
International development and strong financial visibility
The Company plans to open new clinical study centers in Europe for the PhagoDAIR phase II clinical study, including Spain, the Netherlands, Germany, and the United Kingdom.
21
The Company intends to strengthen its Scientific and Clinical Committee with new members of an international caliber.
The Company will also continue its efforts with the European Medicine Agency (EMA) and the US Food and Drug Administration (FDA).
The Company will capitalize on ERYTECH’s presence in the United States to facilitate access to North American clinical and regulatory players with a view to its future phase II and III clinical developments. This presence in the United States will benefit the Company’s financial visibility with US investors.
International industrial strategy
In the medium term, the Company will seek to consolidate its strategic industrial partnerships and establish complementary industrial solutions to protect itself against the risk of clinical batch supply shortages.
At the same time, the Company will pursue an international industrial roll-out strategy to support its global development, in line with its clinical development.
22
CHAPTER 2— SUBSIDIARIES AND EQUITY INVESTMENTS
| 2.1 | SUBSIDIARIESOFTHE COMPANY |
As of December 31, 2022, the Company has no subsidiaries.
| 2.2 | ACQUISITIONOFSIGNIFICANTSHAREHOLDINGSINCOMPANIESHEADQUARTEREDIN FRANCEORACQUISITIONOFCONTROLOFSUCHCOMPANIES |
In accordance with Article L.233-6 of the French Commercial Code, we hereby inform you that during the fiscal year ended December 31, 2022, the Company did not make any equity investments in any company with its registered office in France.
23
CHAPTER 3— INFORMATION ON PAYMENT TERMS TO SUPPLIERS AND CUSTOMERS
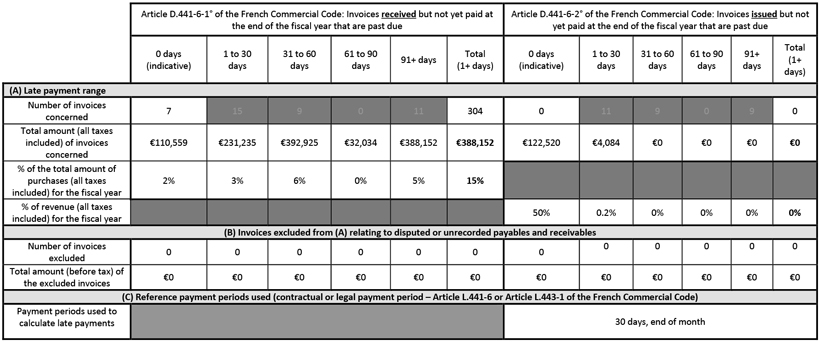
Below is a table showing the balance of the Company’s trade account payables and receivables by due date as of December 31, 2022, in accordance with Articles L.441-14 and D.441-6 of the French Commercial Code:
24
CHAPTER 4— TOTAL DIVIDENDS DISTRIBUTED OVER THE LAST THREE YEARS
The Company has not distributed any dividends in the last three fiscal years.
The Company plans to use all available funds to finance its activities and growth, and therefore does not intend to distribute dividends in the near future.
25
CHAPTER 5— FIVE YEAR SUMMARY OF RESULTS
Below you will find the five year summary of results required by Article R.225-102 para. 2 of the French Commercial Code:
Breakdown of Indications/Periods | 12/31/2022 | 12/31/2021 | 12/31/2020 | 12/31/2019 | 12/31/2018 | |||||||||||||||
Duration of fiscal year | 12 months | 12 months | 12 months | 12 months | 12 months | |||||||||||||||
I—Financial situation at the end of the fiscal year |
| |||||||||||||||||||
a) Share capital | 7,211,477 | 5,852,308 | 4,230,687 | 3,293,694 | 2,784,042 | |||||||||||||||
b) Number of shares issued | 7,221,477 | 1,621,621 | ||||||||||||||||||
c) Number of bonds convertible into shares | ||||||||||||||||||||
II—Overall results of actual operations | ||||||||||||||||||||
a) Revenue excluding taxes | ||||||||||||||||||||
b) Income before taxes, depreciation, amortization and provisions | 5,797,999 | -3,949,499 | -1,996,472 | -1,974,371 | -1,447,205 | |||||||||||||||
c) Income tax | -1,388,080 | -988,514 | -413,732 | -1,028,138 | -367,344 | |||||||||||||||
d) Income after taxes but before depreciation, amortization and provisions | -4,409,919 | -2,960,985 | -1,582,740 | -946,233 | -1,079,861 | |||||||||||||||
e) Income after taxes, depreciation, amortization and provisions | -4,854,342 | -3,189,057 | -1,395,340 | -1,221,868 | -2,419,726 | |||||||||||||||
f) Amount of distributed income | ||||||||||||||||||||
g) Employee shareholding | ||||||||||||||||||||
III—Results of operations per share | ||||||||||||||||||||
a) Income after taxes but before depreciation or amortization | ||||||||||||||||||||
b) Income after taxes, depreciation, amortization and provisions | ||||||||||||||||||||
c) Dividend paid to each share | ||||||||||||||||||||
IV—Personnel: |
| |||||||||||||||||||
a) Number of employees | ||||||||||||||||||||
b) Payroll amount | 2,330,672 | 2,015,555 | 1,341,644 | 1,600,316 | 1,151,093 | |||||||||||||||
c) Amounts paid for employee benefits | 990,671 | 893,130 | 578,951 | 693,375 | 514,182 | |||||||||||||||
26
CHAPTER 6—BREAKDOWN OF SHARE CAPITAL AND TREASURY SHARES
| 6.1 | BREAKDOWNOFSHARECAPITALDURINGTHELASTFISCALYEAR |
The table below shows the breakdown of the Company’s capital and voting rights as of December 31, 2022:
| Number of shares and voting rights at December 31, 2022 | ||||||||||||||||
Shareholders | Shares held | Percentage of capital | Number of voting rights | Percentage of voting rights | ||||||||||||
Members of the Board of Directors and general management | 4,483 | 0.06 | % | 4,483 | 0.06 | % | ||||||||||
Elaia Capital | 1,491,037 | 20.65 | % | 1,491,037 | 20.65 | % | ||||||||||
Go Capital | 1,023,103 | 14.17 | % | 1,023,103 | 14.17 | % | ||||||||||
Tikehau ACE Capital | 1,384,564 | 19.17 | % | 1,384,564 | 19.17 | % | ||||||||||
Omnes Capital | 301,494 | 4.17 | % | 301 ,494 | 4.17 | % | ||||||||||
Participations Besancon | 243,819 | 3.38 | % | 243,819 | 3.38 | % | ||||||||||
GR Pool | 484,645 | 6.71 | % | 484,645 | 6.71 | % | ||||||||||
Treasury shares | 22,057 | 0.31 | % | 22,057 | 0.31 | % | ||||||||||
Public | 2,266,275 | 31.38 | % | 2,266,275 | 31.38 | % | ||||||||||
|
|
|
|
|
|
|
| |||||||||
Total | 7,221,477 | 100 | % | 7,221,477 | 100 | % | ||||||||||
|
|
|
|
|
|
|
| |||||||||
| * | Given the termination of the functions of Mr. Guy-Charles Fanneau de La Horie, previously President of the Management Board, on May 23, 2022, and of Mr. Philippe Rousseau, previously a member of the Management Board, on March 31, 2022, the Company’s shares that they held as of December 31, 2022 are not taken into account in this number of shares. This number corresponds to the shares held personally by Mr. Guy Rigaud, not included in the “GR Pool” line below, and by Mr. Didier Hoch, President of the Board of Directors. The shares held by Elaia Capital and Go Capital are indicated in the corresponding lines of the table. |
As of December 31, 2022 and to the best of the Company’s knowledge, no shareholder other than those mentioned in the table above held more than 5% of the Company’s capital and voting rights.
Each share gives the right to one vote. There is no limit to the number of votes that each shareholder may cast. There are no double voting rights.
| 6.2 | SECURITIESGIVINGACCESSTOTHESHARECAPITAL |
The breakdown of the securities giving access to the Company’s capital and valid at December 31, 2022 are set out in the table below. In total, these securities give the right to subscribe to or issue 673,973 new shares, i.e., 8.53% of the capital existing at December 31, 2022.
27
Summary table of start-up stock options on December 31, 2022.
Potential share capital | Number | Parity | Maximum number of shares to be issued | |||||||||
2017 start-up stock option | — | 1 for 1 | — | |||||||||
2018 start-up stock option | 4,200 | 1 for 1 | 4,200 | |||||||||
2019 start-up stock option | 29,538 | 1 for 1 | 29,538 | |||||||||
2020 start-up stock option | 371,927 | 1 for 1 | 371,927 | |||||||||
2021 start-up stock option | 209,785 | 1 for 1 | 209,785 | |||||||||
2022 start-up stock option | — | 1 for 1 | — | |||||||||
Number of potential shares | 615,450 | |||||||||||
|
| |||||||||||
Number of existing shares | 7,221,477 | |||||||||||
|
| |||||||||||
Overall dilution | 7.854264 | % | ||||||||||
|
| |||||||||||
Summary table of free shares on December 31, 2022.
| Date of allocation | Number of shares allocated | In vesting period | In holding period | |||||||||||||
2022 free share allocation | May 19, 2022 | 58,523 | 58,523 | 0 | ||||||||||||
| 6.3 | CROSSINGOFSTATUTORYOWNERSHIPTHRESHOLDS |
As the Company’s shares have been admitted to trading and listed since February 5, 2021, the provisions of Article L.233-7 of the French Commercial Code relating to the crossing of ownership thresholds have only been applicable since that date.
As of December 31, 2022, the Company had not received any ownership threshold crossing declarations pursuant to the provisions of Article L.233-7 of the French Commercial Code.
| 6.4 | EMPLOYEESHAREHOLDING |
Below you will find the information required pursuant to Article L.225-102 of the French Commercial Code.
As of December 31, 2022, the Company’s employees held 0.007% of the Company’s capital on a non-diluted basis and 4.03% on a fully diluted basis.
We inform you that no company savings plan has been established for the benefit of the Company’s employees.
The Company did not acquire any shares during the fiscal year for allocation to employees, pursuant to Article L.225-208 of the French Commercial Code.
28
| 6.5 | INFORMATIONONTREASURYSHARES |
Following the admission to trading and the first listing of the Company’s shares on the Euronext Growth Paris market, the Company has entered into a liquidity contract with Portzamparc as of February 5, 2021 for a period of one year, renewable by tacit agreement.
This liquidity contract is executed within the framework of the share buyback program authorized by the fourteenth (14th) resolution of the Company’s Ordinary and Extraordinary General Meeting of Shareholders of December 24, 2020, and within the framework of the program authorized by the thirteenth (13th) resolution of the Company’s Ordinary and Extraordinary General Meeting of Shareholders of May 19, 2022.
This program has the following objectives, in order of priority:
| • | to promote the liquidity and boost the price of the Company’s securities on the Euronext Growth Paris market, or on any other market, through the intermediary of an investment service provider acting independently within the framework of a liquidity contract in compliance with the code of ethics recognized by the French Financial Markets Authority ( AMF); |
| • | to remit the repurchased securities in payment or exchange within the framework of a merger, spin-off or contribution; |
| • | to allocate shares to employees or corporate officers of the Company and of French or foreign companies or groupings affiliated with it, in accordance with legal and regulatory conditions, in particular within the framework of profit-sharing schemes, employee shareholding plans or company savings plans, stock option plans, or by means of free share allocations or any other condition permitted by regulations; |
| • | to allocate the shares repurchased upon the exercise of rights attached to securities giving the right, through redemption, conversion, exchange, presentation of a warrant or any other means, to existing shares to be issued by the Company; |
| • | where applicable, to allocate shares repurchased within the framework of the implementation of any market practice that may be accepted by the AMF and that complies with the regulations in force at the time of the effective buyback of the shares, it being specified that, in such case, the Company would inform its shareholders by means of a press release. |
The duration of the program is a maximum of 18 months from the ordinary general meeting of the shareholders of May 19, 2022. The program will expire either on the date on which any General Meeting of Shareholders of the Company adopts a new share buyback program or, failing that, on November 19, 2023.
The maximum authorized percentage of share capital that may be repurchased is 10%. The number of shares acquired by the Company with a view to their holding and subsequent remittance in payment or exchange within the framework of a merger, spin-off or contribution may not exceed 5% of its share capital.
The maximum unit purchase price is €20, i.e., a maximum theoretical amount devoted to the buyback program of €11,708,680.00 based on the maximum percentage of 10%, excluding trading fees.
29
This number of shares and the purchase price limits may be adjusted, where necessary, by the Board of Directors in the event of financial transactions by the Company or decisions affecting the share capital.
As of December 31, 2022, the following resources were included in the liquidity account:
| • | 22,057 Pherecydes Pharma shares; |
| • | €43,066.35. |
30
CHAPTER 7—TRANSACTIONS CARRIED OUT BY MANAGERS ON THEIR SHARES
Pursuant to the provisions of Articles 223-22 A and 223-26 of the AMF’s General Regulations, we hereby inform you that the following transactions on the Company’s securities were carried out by executive directors during the fiscal year:
None.
31
| CHAPTER | 8—COMPENSATION AND BENEFITS GRANTED TO CORPORATE OFFICERS AND SHAREHOLDING |
| 8.1 | COMPENSATIONANDBENEFITSGRANTEDTOCORPORATEOFFICERS |
The tables below show the compensation and benefits in kind paid to corporate officers by the Company during the fiscal years ended December 31, 2022 and December 31, 2021:
Summary table of all compensation, options and free shares granted to executive directors
A summary of all compensation due and paid to the members of the Board of Directors of the Company during the fiscal years ended December 31, 2022 and December 31, 2021 is presented below:
Name | 2022 | 2021 | ||||||||||||||||||||||
| Fixed compensation due and paid (in euros) | Variable compensation due and paid (in euros) | Benefits in kind due and paid (in euros) | Fixed compensation due and paid (in euros) | Variable compensation due and paid (in euros) | Benefits in kind due and paid (in euros) | |||||||||||||||||||
Guy-Charles Fanneau de La Horie President of the Management Board(1) | 99,764 | 97,389 | 2,307 | 235,200 | 37,632 | 5,538 | ||||||||||||||||||
Philippe Rousseau Member of the Management Board(2) | 39,201 | 89,977 | 156,830 | 67,720 | — | |||||||||||||||||||
Didier Hoch Chief Executive Officer(3) | 161,504 | |||||||||||||||||||||||
Thibaut du Fayet Chief Executive Officer(4) | 131,249 | |||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
| |||||||||||||
TOTAL | 431,718 | 187,366 | 2,307 | 392,031 | 100,352 | 5,538 | ||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
| |||||||||||||
| (1) | Mr. Guy-Charles Fanneau de La Horie was appointed member of the Management Board and President of the Management Board by the Supervisory Board at its meeting on December 1, 2015, from January 4, 2016 until May 23, 2022. |
| (2) | Mr. Philippe Rousseau was appointed member of the Management Board by the Supervisory Board at its meeting on June 24, 2020 and left the company on March 31, 2022. |
| (3) | Mr. Didier Hoch was appointed member of the Supervisory Board and President of the Supervisory Board on January 16, 2019; the Ordinary and Extraordinary General Meeting of Shareholders of May 19, 2022 changed the Company’s method of management and administration by adopting a Board of Directors and general management; Mr. Hoch was appointed Chief Executive Officer by the Board of Directors on May 23, 2022 until December 15, 2022. |
| (4) | Mr. Thibaut du Fayet was appointed Chief Operating Officer and member of the Management Board on April 7, 2022, and the Pharma General Meeting of Shareholders on May 19, 2022 approved the change in governance to a company with a Board of Directors. The new Board of Directors appointed Thibaut du Fayet as Chief Operating Officer as of May 23, 2022. On December 15, 2022, at the end of the Combined General Meeting of Shareholders on December 15, 2022, the Board of Directors appointed Mr. Thibaut du Fayet as Chief Executive Officer. |
32
Summary table of attendance fees and other compensation received by non-executive corporate officers
A summary of all attendance fees and other compensation paid to members of the Company’s Supervisory Board for the fiscal years ended December 31, 2021 and December 31, 2022 is presented below:
NON-EXECUTIVE CORPORATE OFFICERS | Fiscal year 2021 | Fiscal year 2022 | ||||||||||||||
| Amounts allocated | Amounts paid | Amounts allocated | Amounts paid | |||||||||||||
Maryvonne Hiance, director (1) | ||||||||||||||||
Compensation (fixed, variable) | € | 16,643 | € | 16,643 | € | 28,711 | € | 17,710 | ||||||||
Other compensation | — | — | — | — | ||||||||||||
Elaia Partners, represented by Franck Lescure, director (2) | ||||||||||||||||
Compensation (fixed, variable) | € | 0 | € | 0 | € | 0 | € | 0 | ||||||||
Other compensation | € | 0 | € | 0 | € | 0 | € | 0 | ||||||||
Go Capital, represented by Leila Nicolas, director (3) | ||||||||||||||||
Compensation (fixed, variable) | € | 0 | € | 0 | € | 0 | € | 0 | ||||||||
Other compensation | € | 0 | € | 0 | € | 0 | € | 0 | ||||||||
Guy Rigaud, director (4) | ||||||||||||||||
Compensation (fixed, variable) | € | 0 | € | 0 | € | 0 | € | 0 | ||||||||
Other compensation | € | 0 | € | 0 | € | 0 | € | 0 | ||||||||
Robert Sebbag, director (5) | ||||||||||||||||
Compensation (fixed, variable) | € | 7.989 | € | 7.989 | € | 15,221 | € | 9,221 | ||||||||
Other compensation | € | 0 | € | 0 | — | — | ||||||||||
Eric Leire, director (6) | ||||||||||||||||
Compensation (fixed, variable) | € | 10.794 | € | 10.794 | € | 19.026 | € | 11.526 | ||||||||
Other compensation | € | 0 | € | 0 | — | — | ||||||||||
|
|
|
|
|
|
|
| |||||||||
Total | € | 96,887 | € | 96,887 | € | 62,598 | € | 38,458 | ||||||||
|
|
|
|
|
|
|
| |||||||||
33
| (1) | Ms. Maryvonne Hiance was appointed member of the Supervisory Board and President of the Supervisory Board on January 16, 2019; the Ordinary and Extraordinary General Meeting of Shareholders of May 19, 2022 appointed the current members of the Supervisory Board as the first directors. |
| (2) | Elaia Partners was appointed member of the Supervisory Board on May 28, 2019; the Ordinary and Extraordinary General Meeting of Shareholders of May 19, 2022 appointed the current members of the Supervisory Board as the first directors. |
| (3) | Go Capital was appointed member of the Supervisory Board on December 22, 2017; the Ordinary and Extraordinary General Meeting of Shareholders of May 19, 2022 appointed the current members of the Supervisory Board as the first directors. |
| (4) | Mr. Guy Rigaud was appointed member of the Supervisory Board on December 24, 2020; the Ordinary and Extraordinary General Meeting of Shareholders of May 19, 2022 appointed the current members of the Supervisory Board as the first directors. |
| (5) | Mr. Robert Sebbag was appointed member of the Supervisory Board on December 24, 2020; the Ordinary and Extraordinary General Meeting of Shareholders of May 19, 2022 appointed the current members of the Supervisory Board as the first directors. |
| (6) | Mr. Eric Leire was appointed member of the Supervisory Board on December 24, 2020; the Ordinary and Extraordinary General Meeting of Shareholders of May 19, 2022 appointed the current members of the Supervisory Board as the first directors. |
| 8.2 | SHAREHOLDINGSOFCORPORATEOFFICERS |
As of December 31, 2022, the shareholding of each officer in the share capital of the Company is as follows:
Officers | Shares | Voting rights | ||||||||||||||
| Number | % | Number | % | |||||||||||||
| Chief Executive Officer | ||||||||||||||||
Thibaut du Fayet | 0 | 0 | % | 0 | 0 | % | ||||||||||
| Members of the Board of Directors | ||||||||||||||||
Didier Hoch | 334 | N/S | 334 | N/S | ||||||||||||
Maryvonne Hiance | 0 | 0 | % | 0 | 0 | % | ||||||||||
Go Capital Represented by Ms. Leila Nicolas | 1,023,103 | 14.17 | % | 1,023,103 | 14.17 | % | ||||||||||
Elaia Partners Represented by Mr. Franck Lescure | 1,491,037 | 20.65 | % | 1,491,037 | 20.65 | % | ||||||||||
Guy Rigaud | 3,997 | 0.05 | % | 3,997 | 0.05 | % | ||||||||||
Eric Leire | 0 | 0 | % | 0 | 0 | % | ||||||||||
Robert Sebbag | 0 | 0 | % | 0 | 0 | % | ||||||||||
34
CHAPTER 9—SPECIAL REPORT ON STOCK OPTIONS AND FREE SHARES
Dear Shareholders,
This report is presented to you:
| • | pursuant to the provisions of Article L.225-184 of the French Commercial Code concerning transactions relating to share subscription or purchase options, and |
| • | pursuant to the provisions of Article L.225-197-4 of the French Commercial Code concerning transactions relating to free shares. |
| 9.1 | SHARESUBSCRIPTIONORPURCHASEOPTIONS |
Allocation of share subscription options during the fiscal year ended December 31, 2022
No share subscription or purchase options were allocated during the fiscal year ended December 31, 2022.
Exercise by beneficiaries of share subscription options during the fiscal year ended December 31, 2022
No share subscription or purchase options were exercised during the fiscal year ended December 31, 2022.
| 9.2 | FREESHAREALLOCATIONS |
Free share allocation during the fiscal year ended December 31, 2022
The ordinary and extraordinary general meeting of the shareholders of May 19, 2022, in its twentieth (20th) resolution, authorized the Board of Directors, in accordance with the provisions of Articles L.225-197-1 et seq. of the French Commercial Code, to proceed with a free allocation of the Company’s shares, either existing or to be issued, on one or more occasions, to the staff members that it determines from among the eligible corporate officers and employees of the Company and of companies or groupings affiliated with it under the terms of Article L.225-197-2 of said code.
Under this authorization, the Board of Directors unanimously decided at its meeting on May 19, 2022 to allocate a total of 58,523 free shares to be issued by the Company.
Plan | Number of shares allocated | Vesting date | Transferability date | |||
| May 19, 2022 | 58,523 | 25% of the free share allocations will vest on May 19, 2023; 25% of the free share allocations will vest on May 19, 2024; 25% of the free share allocations will vest on May 19, 2025; The balance will vest on May 19, 2026. | One year from the expiration of the vesting period of each tranche. |
35
Free shares granted to each of the corporate officers
Beneficiaries | Number of shares allocated | Date of allocation | Vesting period | Holding period | ||||||||||||
Thibaut du Fayet | 58,523 | May 19, 2022 | 1 year | 1 year | ||||||||||||
36
Vesting of free shares during the fiscal year ended December 31, 2022
During the fiscal year ended December 31, 2022, the Board of Directors of the Company noted the acquisition by Mr. Guy-Charles Fanneau de La Horie of 2,688 free shares and by Mr. Philippe Rousseau of 4,480 free shares, all of which were allocated on February 4, 2021.
The holding period for all these free shares vested during the fiscal year ended December 31, 2022 ended on February 4, 2023.
37
CHAPTER 10—APPOINTMENT OF THE STATUTORY AUDITORS
| 10.1 | STATUTORY AUDITORS |
Primary statutory auditors
PricewaterhouseCoopers Audit
Represented by Mr. Cédric Mazille
63, Rue de Villiers
92208 Neuilly-sur-Seine, France
Appointed by the initial Articles of Association of December 12, 2006 and reappointed by the Ordinary and Extraordinary General Meeting of Shareholders of May 28, 2020 for a term of six fiscal years, i.e. until the end of the General Meeting of Shareholders called in 2026 to approve the financial statements for the fiscal year ended December 31, 2025.
Alternate statutory auditor
Mr. Patrice Morot
63, Rue de Villiers
92208 Neuilly-sur-Seine, France
Appointed by the Ordinary and Extraordinary General Meeting of Shareholders of May 28, 2020 for a term of six fiscal years, i.e. until the end of the General Meeting of Shareholders called in 2026 to approve the financial statements for the fiscal year ended December 31, 2025.
| 10.2 | STATUTORY AUDITORSWHORESIGNED,WEREDISMISSEDORWERENOTREAPPOINTEDDURINGTHEFISCALYEARENDED DECEMBER 31, 2022 |
None.
38
CHAPTER 11—LIST OF RELATED-PARTY AGREEMENTS AND CURRENT AGREEMENTS
Pursuant to the provisions of Articles L. 225-38 et seq. of the French Commercial Code, below you will find a list of related-party agreements and current agreements for the fiscal year ended December 31, 2022.
We hereby inform you that the functions of the individuals concerned by the agreements described in this chapter are considered as of the date of signature of said agreements.
To the best of the Company’s knowledge, there are no service contracts between the Company and its corporate officers providing for the granting of benefits other than those described in this chapter.
| 11.1 | NEWRELATED-PARTYAGREEMENTSENTEREDINTODURINGFISCALYEAR 2022 |
None.
| 11.2 | NEWRELATED-PARTYAGREEMENTSENTEREDINTOSINCETHEENDOFFISCALYEAR 2022 |
None.
| 11.3 | RELATED-PARTYAGREEMENTSAPPROVEDBYTHE GENERAL MEETINGOF SHAREHOLDERS,BUTWHICHCONTINUETOBEINEFFECTDURINGFISCALYEAR 2022 |
None.
| 11.4 | SURETIES,ENDORSEMENTSANDGUARANTEESGIVENBYTHE COMPANYTOTHIRDPARTIES |
None.
| 11.5 | AGREEMENTSBETWEENACORPORATEOFFICERORASHAREHOLDERHOLDINGMORETHAN 10%OFTHEVOTINGRIGHTSOFTHE COMPANYANDASUBSIDIARY,EXCLUDINGCURRENTAGREEMENTS |
None.
39
CHAPTER 12—OTHER INFORMATION
In accordance with the provisions of Articles 39-4 and 223 quater of the French General Tax Code, we hereby inform you that the Company’s financial statements for the past year do not contain any expenses that are not deductible from taxable income.
Pursuant to the provisions of Article L.232-6 of the French Commercial Code, we hereby inform you that no changes have been made to the method of presentation of the annual financial statements, nor to the valuation methods used in comparison with the previous fiscal year.
40
CHAPTER 13—REPORT OF THE BOARD OF DIRECTORS
In accordance with the provisions of Article L.225-68 paragraph 6 of the French Commercial Code, in Appendix 1 of this management report you will find the report on corporate governance prepared by the Board of Directors of the Company pursuant to the aforementioned provisions.
We hope that the above will meet with your approval.
Nantes, France,
March 30, 2023
Chief Executive Officer
Thibaut du Fayet
41
PHERECYDES PHARMA
Limited company (société anonyme) with a Board of Directors and capital of €7,939,179
Registered office: 22, Boulevard Benoni Goullin – 44200 Nantes, France
493 252 266 Nantes Trade and Companies Register
REPORTOFTHE BOARDOF DIRECTORSONCORPORATEGOVERNANCE
Dear Shareholders,
This report is hereby presented to you pursuant to Articles L. 225-37 et seq. and L. 22-10-8 et seq. of the French Commercial Code and concerns corporate governance, as well as the observations of the Board of Directors of Pherecydes Pharma (hereinafter the “Company”) and the financial statements for the fiscal year ended December 31, 2022.
We hereby remind you that the Company’s shares are admitted to trading on the Euronext Growth Paris market, an organized multilateral trading facility. As a result, in accordance with the provisions of Article L.225-37 of the French Commercial Code, this report includes all the information required by Articles L.225-37 et seq. and L.22-10-8 et seq. of said Code for limited companies (sociétés anonymes) whose securities are not listed on a regulated market.
CHAPTER 14—REPORT ON CORPORATE GOVERNANCE
| 14.1 | LISTOFOFFICES |
| 14.1.1 | Chief Executive Officer |
As of December 31, 2022, the Company has a Chief Executive Officer, Mr. Thibaut du Fayet (55 years old). He was unanimously appointed Chief Executive Officer by the Board of Directors of the Company at its meeting of December 15, 2022 for an indefinite term.
Mr. Thibaut du Fayet, Chief Executive Officer (55 years old)
Thibaut du Fayet brings to the Company over 20 years of experience in the pharmaceutical and life sciences industry. After obtaining an MBA from ESSEC Business School in 1992, he was Senior Managing Director at Bossard-Gemini for six years. From 2000, he held management positions first at Rhône Poulenc as Strategy Director for four years, and then at BioMerieux where he was also Strategic Marketing Director for four years. In 2008, he joined Transgene where he held the position of VP Corporate Development, Alliances & Programs Management for 13 years.
Through his various leadership roles in development, strategy, business development, and program management, he has acquired important and diverse skills in the pharmaceutical industry.
42
Mr. Thibaut du Fayet was appointed a member of the former Management Board by the former Supervisory Board at its meeting of April 5, 2022, to replace Mr. Philippe Rousseau for a term of four (4) years, i.e., until the end of the General Meeting of Shareholders called in 2026 to approve the financial statements for the fiscal year ended December 31, 2025.
On May 19, 2022, the Ordinary General Meeting of the Company’s Shareholders resolved to adopt a method of management and administration with a Board of Directors and general management. The meeting therefore dismissed all the members of its Management Board, including Mr. Thibaut du Fayet.
On May 23, 2022, the Board of Directors of the Company unanimously appointed Mr. Thibaut du Fayet as Chief Operating Officer of the Company for an indefinite term. On December 15, 2022, the Board of Directors of the Company unanimously appointed Mr. Thibaut du Fayet as Chief Executive Officer of the Company for an indefinite term.
Other offices and positions held as of December 31, 2022
Positions | Companies | Start dates | ||
| None |
Other offices and positions held over the last five years
Positions | Companies | Dates | ||
| Within the Company | ||||
| Chief Operating Officer | Pherecydes Pharma | 2022 | ||
| Member of the Management Board | Pherecydes Pharma | 2022 | ||
| Outside the Group | ||||
| None | ||||
Number of shares held as of December 31, 2022: 0 | ||||
| 14.1.2 | BOARDOF DIRECTORS |
Composition of the Board of Directors
As of December 31, 2022, the Board of Directors of the Company is composed as follows:
Name | Office | Age | Independent | Member of | Member of the | Date of | Expiration | |||||||
Didier Hoch | President of the Board of Directors | 66 | 2022 | 2026 | ||||||||||
Maryvonne Hiance | Director | 74 | ✓ | 2022 | 2026 |
43
Name | Office | Age | Independent | Member of | Member of the | Date of | Expiration | |||||||
Go Capital – represented by Leila Nicolas | Director | 42 | ✓ | 2022 | 2026 | |||||||||
Elaia Partners – represented by Franck Lescure | Director | 54 | 2022 | 2026 | ||||||||||
Guy Rigaud | Director | 75 | ✓ | 2022 | 2026 | |||||||||
Robert Sebbag | Director | 72 | ✓ | 2022 | 2026 | |||||||||
Eric Leire | Director | 65 | ✓ | ✓ | 2022 | 2026 |
| (1) | In accordance with recommendation no. 9 of the Middlenext corporate governance code for small and mid-sized companies of September 2021. |
Profiles of the members of the Board of Directors
Didier Hoch, President of the Board of Directors (66 years old)
Didier Hoch, M.D. is a director of a listed company, OSE Immunotherapeutics, and was previously a member of the board of DBV Technologies and Genticel. From 2011 to 2013, he was President of Pevion Biotech, and from 2013 to 2018, President of the Biovision Forum and of the startup incubator “Big Booster.” From 2000 to 2010, he was President of Sanofi-Pasteur-MSD, a joint venture company between Sanofi & Merck dedicated to vaccines. Didier Hoch also held various managerial positions at Rhône Poulenc Rorer, then Aventis (“VP Middle East-Africa”). He was also President of the vaccine manufacturers association “Vaccine Europe” and President of the Biotechnology Committee of LEEM (Les Entreprises du médicament).
Mr. Didier Hoch was co-opted as a member of the former Supervisory Board by the Supervisory Board at its meeting of January 16, 2019, to replace Mr. Michel Joli for the remainder of the latter’s term of office, i.e., until the end of the General Meeting of Shareholders called in 2022 to approve the financial statements for the fiscal year ended December 31, 2021. The Ordinary and Extraordinary General Meeting of Shareholders of December 24, 2020 decided to reduce the term of office of members of the Supervisory Board from six to four years. As a result, the Ordinary and Extraordinary General Meeting of Shareholders of December 24, 2020 decided to renew his term of office of member of the Supervisory Board for a period of four years, i.e. until the end of the General Meeting of Shareholders called in 2024 to approve the financial statements for the fiscal year ended December 31, 2023.
44
The former Supervisory Board, at its meeting on January 16, 2019, decided to appoint Mr. Didier Hoch as President of the Supervisory Board for the duration of his term of office of member of the Supervisory Board, and he was reappointed by the Supervisory Board at its meeting on December 24, 2020.
On May 19, 2022, the Ordinary General Meeting of the Company’s Shareholders resolved to adopt a method of management and administration with a Board of Directors and general management. As a result, the meeting dismissed all the members of its Supervisory Board, including Mr. Didier Hoch, whose terms of office of both member of the Supervisory Board and of President of the Supervisory Board were terminated.
In addition, the same meeting of May 19, 2022 appointed Mr. Didier Hoch as a director of the Company for a term of four (4) years, i.e. until the end of the General Meeting of Shareholders to be held in 2026 to approve the financial statements for the fiscal year ended December 31, 2025.
On May 19, 2022, the directors of the Company unanimously appointed Mr. Didier Hoch as President of the Board of Directors for the duration of his term of office as a member of the Board of Directors. Mr. Didier Hoch was unanimously appointed Chief Executive Officer by the Board of Directors on May 23, 2022. He held the position of Chief Executive Officer until December 15, 2022.
Other offices and positions held as of December 31, 2022
Positions | Companies | Start dates | ||
| Director | Ose Immuno Therapeutics | 2011 | ||
| Co-President | Charity for the Underground University Foundation | 2018 | ||
| President of the Strategic Board | Goliver Therapeutics | 2019 |
Other offices and positions held over the last five years
Positions | Companies | Dates | ||
| Within the Company | ||||
| Chief Executive Officer | Pherecydes Pharma | 2022 | ||
| President of the Supervisory Board | Pherecydes Pharma | 2019–2022 | ||
| Outside the Group | ||||
| Director | Germitec | 2014–2019 | ||
| Director | DBV Technologies | 2012–2016 | ||
| Director | Genticel | 2011–2017 | ||
| Advisory Scientific Board member | Myasterix/Curavac | 2014–2018 | ||
| President of Biovision and Big Booster | Fondation pour l’Université de Lyon | 2012–2018 | ||
Number of shares held as of December 31, 2022: 334 | ||||
45
Maryvonne Hiance, member of the Board of Directors (74 years old)
Previously President and co-founder of Effimune, Maryvonne Hiance is a nuclear engineer who managed a nuclear neutron program at FRAMATOME (Areva) for 14 years. She also previously held managerial positions at various innovative companies in Biotechnology for more than 20 years: SangStat Atlantic (the parent company Sangstat medical corporation was acquired by industrialist Genzyme in 2003 for its portfolio of products in immune suppression and transplantation). She also managed the innovative companies DrugAbuse Sciences and TcLand. Maryvonne Hiance founded and led Strategic Ventures, a consulting firm involved in helping technology companies. She was a member of the Strategic Innovation Council and advisor to the French Minister of SMEs and Industry. She is currently Vice-President of France Biotech.
Ms. Maryvonne Hiance was co-opted as a member of the former Supervisory Board by the Supervisory Board at its meeting of January 16, 2019, to replace Mr. Jean-Pierre Loza for the remainder of the latter’s term of office, i.e., until the end of the General Meeting of Shareholders called in 2019 to approve the financial statements for the fiscal year ended December 31, 2018. The Ordinary and Extraordinary General Meeting of Shareholders of June 28, 2019 resolved to reappoint Ms. Maryvonne Hiance as a member of the Board of Directors for a period of six years. The Ordinary and Extraordinary General Meeting of Shareholders of December 24, 2020 decided to reduce the term of office of members of the Supervisory Board from six to four years and noted, as a result, that the term of office of Ms. Maryvonne Hiance as a member of the Supervisory Board will expire at the end of the General Meeting of Shareholders called in 2023 to approve the financial statements for the fiscal year ended December 31, 2022.
The former Supervisory Board, at its meeting on January 16, 2019, decided to appoint Ms. Maryvonne Hiance as Vice-President of the Supervisory Board for the duration of her term of office of member of the Supervisory Board.
On May 19, 2022, the Ordinary General Meeting of the Company’s Shareholders resolved to adopt a method of management and administration with a Board of Directors and general management. As a result, the meeting dismissed all the members of its Supervisory Board, including Ms. Maryvonne Hiance, whose terms of office of both member of the Supervisory Board and of Vice-President of the Supervisory Board were terminated.
In addition, the same meeting of May 19, 2022 appointed Ms. Maryvonne Hiance as a director of the Company for a term of four (4) years, i.e. until the end of the General Meeting of Shareholders to be held in 2026 to approve the financial statements for the fiscal year ended December 31, 2025.
Other offices and positions held as of December 31, 2022
Positions | Companies | Start dates | ||
| Vice-Chairwoman, strategy director | Ose Immuno Therapeutics | 2016 | ||
| Vice-President | France Biotech | 2019 | ||
| President of HealthTECH For Care | France Biotech | 2019 | ||
| President | Olgram | 2020 |
46
Other offices and positions held over the last five years
Positions | Companies | Dates | ||
| Within the Company | ||||
| Vice-President of the Supervisory Board | Pherecydes Pharma | 2019–2022 | ||
| Outside the Group | ||||
| President | France Biotech | 2016–2019 | ||
| Director | Apave | 2018–2020 | ||
Number of shares held as of December 31, 2022: 0 | ||||
Mr. Franck Lescure, permanent representative of Elaia Partners, member of the Board of Directors (54 years old)
Franck Lescure joined Elaia Partners to bring his expertise in life sciences investment after more than 20 years of experience in early-stage biotech startups. Franck Lescure began his career as a scientist at Genset, one of the first French biotech startups. In 1997, he joined the Health subsidiary of the Air Liquide Group as a product manager. He started his career as an investor at Crédit Lyonnais Private Equity and then became a Partner in Life Sciences at Auriga Partners.
The company Elaia Partners was co-opted as a member of the former Supervisory Board by the Supervisory Board at its meeting of May 28, 2019, to replace the company Auriga Partners for the remainder of the latter’s term of office, i.e., until the end of the General Meeting of Shareholders called in 2021 to approve the financial statements for the fiscal year ended December 31, 2020. The Ordinary and Extraordinary General Meeting of Shareholders of December 24, 2020 decided to reduce the term of office of members of the Supervisory Board from six to four years. As a result, the Ordinary and Extraordinary General Meeting of Shareholders of December 24, 2020 decided to renew its term of office of member of the Supervisory Board for a period of four years, i.e. until the end of the General Meeting of Shareholders called in 2024 to approve the financial statements for the fiscal year ended December 31, 2023. Mr. Franck Lescure has been the permanent representative of Elaia Partners on the Company’s Supervisory Board since its co-option on May 28, 2019.
On May 19, 2022, the Ordinary General Meeting of the Company’s Shareholders resolved to adopt a method of management and administration with a Board of Directors and general management. The meeting therefore dismissed all the members of its Supervisory Board, including the company Elaia Partners.
In addition, the same meeting of May 19, 2022 appointed the company Elaia Partners as a director of the Company for a term of four (4) years, i.e. until the end of the General Meeting of Shareholders to be held in 2026 to approve the financial statements for the fiscal year ended December 31, 2025.
Mr. Franck Lescure has been the permanent representative of Elaia Partners on the Board of Directors of the Company since its appointment on May 19, 2022.
47
Other offices and positions held as of December 31, 2022
Positions | Companies | Start dates | ||
| Permanent representative of Elaia Partners | Flash Therapeutics | 2016 | ||
| Permanent representative of Elaia Partners | Honing Biosciences | 2020 | ||
| Permanent representative of Elaia Partners | Pili | 2019 | ||
| Permanent representative of Elaia Partners | Pylote | 2015 | ||
| Permanent representative of Elaia Partners | Aviwell | 2021 |
Other offices and positions held over the last five years
Positions | Companies | Dates | ||
| Within the Company | ||||
| Member of the Supervisory Board | Pherecydes Pharma | 2019–2022 | ||
| Outside the Group | ||||
| Permanent representative of Elaia Partners | Amoeba | 2014–2018 | ||
| Permanent representative of Elaia Partners | Cytoo | 2009–2018 | ||
| Permanent representative of Elaia Partners | Median | 2008–2018 | ||
| Permanent representative of Elaia Partners | Nosopharm | 2016–2019 | ||
| Permanent representative of Elaia Partners | Fab’entech | 2014–2019 | ||
| Permanent representative of Elaia Partners | Exeliom | 2019 | ||
| Permanent representative of Elaia Partners | EnobraQ | 2017–2020 | ||
Number of shares held as of December 31, 2022: 0(1) | ||||
| (1) | As of the date of this report on corporate governance and to the best of the Company’s knowledge, Mr. Franck Lescure does not directly or indirectly hold any shares in the Company. The participation in the funds managed by Elaia Partners is described in chapter 8 of the management report. |
Ms. Leïla Nicolas, permanent representative of Go Capital, member of the Board of Directors (42 years old)
Leïla Nicolas started her career in 2004 as a product manager at Bayer Schering Pharma in the field of multiple sclerosis and oncology. She then joined the Biotechnology and Life Sciences sector as strategic marketing manager of a start-up in Alsace (Polyplus-transfection). Leïla Nicolas concluded several licensing agreements and acquired a strong expertise in intellectual property. Leïla Nicolas was then involved in the creation of a company that develops and sells environmental health analysis kits before joining Go Capital in late 2013.
The company Go Capital was appointed as a member of the former Supervisory Board by the Ordinary and Extraordinary General Meeting of Shareholders of December 22, 2017 for a term of six years. The Ordinary and Extraordinary General Meeting of Shareholders of December 24, 2020 decided to reduce the term of office of members of the Supervisory Board from six to four years and noted, as a result, that the term of office of Go Capital as a member of the Supervisory Board will expire at the end of the General Meeting of Shareholders called in 2021 to approve the financial statements for the fiscal year ended December 31, 2020. The Ordinary General Meeting of Shareholders of June 22, 2021 renewed Go Capital’s term of office of member of the Supervisory Board for a period of four years, i.e. until the end of the General Meeting of Shareholders called in 2025 to approve the financial statements for the fiscal year ended December 31, 2024. Ms. Leila Nicolas has been the permanent representative of Go Capital on the Company’s Supervisory Board since its appointment on December 22, 2017.
48
On May 19, 2022, the Ordinary General Meeting of the Company’s Shareholders resolved to adopt a method of management and administration with a Board of Directors and general management. The meeting therefore dismissed all the members of its Supervisory Board, including the company Go Capital.
In addition, the same meeting of May 19, 2022 appointed the company Go Capital as a director of the Company for a term of four (4) years, i.e. until the end of the General Meeting of Shareholders to be held in 2026 to approve the financial statements for the fiscal year ended December 31, 2025.
Ms. Leila Nicolas has been the permanent representative of Elaia Partners on the Board of Directors of the Company since its appointment on May 19, 2022.
Other offices and positions held as of December 31, 2022
Positions | Companies | Start dates | ||
| Member of the Strategy Committee | HTC Assistance | 2021 | ||
| Member of the Strategy Committee | Kalsiom | 2020 | ||
| Director | Acticor Biotech | 2020 | ||
| Director | VitaDx | 2017 | ||
| Director | Coave Therapeutics (formerly Horama) | 2016 | ||
| Member of the Strategy Committee | Tricares | 2015 | ||
| Member of the Strategy Committee | I-SEP | 2015 |
Other offices and positions held over the last five years
Positions | Companies | Dates | ||
| Within the Company | ||||
| Member of the Supervisory Board | Pherecydes Pharma | 2017–2022 | ||
| Outside the Group | ||||
| Member of the Strategy Committee | Atlanthera | 2014–2022 | ||
| Member of the Strategy Committee | Biosency | 2018–2022 | ||
| Director | Graftys | 2019–2020 | ||
| Member of the Strategy Committee | Carlina | 2014–2020 | ||
| Member of the Strategy Committee | Kemiwatt | 2014–2018 | ||
| Member of the Strategy Committee | Surfact’Green | 2016–2018 | ||
| Director | VitamFero | 2014–2018 | ||
| Member of the Strategy Committee | Biosency | 2018–2021 | ||
| Member of the Strategy Committee | Celenys | 2014–2017 | ||
Number of shares held as of December 31, 2022: 0(1) | ||||
| (1) | As of the date of this report on corporate governance and to the best of the Company’s knowledge, Ms. Leïla Nicolas does not directly or indirectly hold any shares in the Company. The participation in the funds managed by Go Capital is described in chapter 8 of the management report. |
49
Mr. Guy Rigaud, member of the Board of Directors (75 years old)
Guy Rigaud has 30 years of experience in private equity in more than 300 young regional companies (more than half of them in the technology sector). Founder and President of the Management Board of Rhône Alpes Création from 1990 to 2012, Guy Rigaud has participated in five IPOs on Euronext Paris and two on Nasdaq (in the context of industrial disposals). Guy Rigaud was a member of the Board of Directors of April Group (insurance), a company listed on Euronext Paris, for 12 years. Since 2012, Guy Rigaud is the founder and associate director of a seed capital fund created with four “family offices.”
Mr. Guy Rigaud was appointed as non-voting member of the former Supervisory Board by the Ordinary and Extraordinary General Meeting of Shareholders of December 22, 2017 for a term of six years. The Ordinary and Extraordinary General Meeting of Shareholders of December 24, 2020 noted the resignation of Mr. Guy Rigaud from his position as non-voting member and resolved to appoint him as a member of the former Supervisory Board for a term of four years, i.e., until the end of the General Meeting of Shareholders called in 2024 to approve the financial statements for the fiscal year ended December 31, 2023.
On May 19, 2022, the Ordinary General Meeting of the Company’s Shareholders resolved to adopt a method of management and administration with a Board of Directors and general management. The meeting therefore dismissed all the members of its Supervisory Board, including Mr. Guy Rigaud.
In addition, the same meeting of May 19, 2022 appointed Mr. Guy Rigaud as a director of the Company for a term of four (4) years, i.e. until the end of the General Meeting of Shareholders to be held in 2026 to approve the financial statements for the fiscal year ended December 31, 2025.
Other offices and positions held as of December 31, 2022
Positions | Companies | Start dates | ||
| None | ||||
| Other offices and positions held over the last five years | ||||
Positions | Companies | Dates | ||
| Within the Company | ||||
| Member of the Supervisory Board | Pherecydes Pharma | 2017–2022 | ||
| Outside the Group | ||||
| Permanent representative of Eurekap | Kalray | |||
| Member of the Board of Directors | Kreaxi | |||
| Member of the Strategy Committee | LX Repair | |||
| Member of the Strategy Committee | GlycoBar | |||
Number of shares held as of December 31, 2022: 3,997 | ||||
Mr. Robert Sebbag, member of the Board of Directors (72 years old)
Robert Sebbag, M.D. is attached to Public Hospitals of Paris (Hôpitaux de Paris) and works at the Pitié Salpêtrière hospital in the department of infectious and tropical diseases. He is also a member of the board and founding member of Action Contre la Faim and member of the board of Fondation Mérieux.
50
From 2006 to 2016, he was Vice President Access to Medicines at Sanofi. He previously held several positions at Rhône Poulenc Santé, Fondation Elf, and Aventis-Pasteur. He is a former member of the Executive Committee of the CEO round table of the Gate Foundation and a member of the board of LEEM (Les Entreprises du médicament).
Mr. Robert Sebbag was appointed as a member of the former Supervisory Board by the Ordinary and Extraordinary General Meeting of Shareholders of December 24, 2020 for a term of four years, i.e. until the end of the General Meeting of Shareholders called in 2024 to approve the financial statements for the fiscal year ended December 31, 2023.
On May 19, 2022, the Ordinary General Meeting of the Company’s Shareholders resolved to adopt a method of management and administration with a Board of Directors and general management. The meeting therefore dismissed all the members of its Supervisory Board, including Mr. Robert Sebbag.
In addition, the same meeting of May 19, 2022 appointed Mr. Robert Sebbag as a director of the Company for a term of four (4) years, i.e. until the end of the General Meeting of Shareholders to be held in 2026 to approve the financial statements for the fiscal year ended December 31, 2025.
Other offices and positions held as of December 31, 2022
Positions | Companies | Start dates | ||
| Fondation Merieux | ||||
| Action contre la faim | ||||
| Provepharm | ||||
Other offices and positions held over the last five years
Positions | Companies | Dates | ||
| Within the Company | ||||
| Member of the Supervisory Board | Pherecydes Pharma | 2020–2022 | ||
| Outside the Group | ||||
| Positive Planet | ||||
| Number of shares held as of December 31, 2022: 0 | ||||
Mr. Eric Leire, member of the Board of Directors (65 years old)
Eric Leire, M.D. is currently CEO of Genflow Biosciences. He is also a board member of publicly traded and private biotechnology companies (Immunethep, BSIM Therapeutics and Inhatarget Therapeutics). Dr. Leire has served as CEO of several private and publicly traded U.S. biotechnology companies. He has listed several biotech companies on various stock exchanges (OMX.Nasdaq; OTC.QB and Nasdaq). He was a research associate at the Harvard AIDS Institute and Partner at Biofund Venture Capital. He has an M.D. from the University of Grenoble and a MBA from HEC and Kellogg School of Management, Northwestern University. He is also the inventor of numerous patents in the pharmaceutical field.
51
Mr. Eric Leire was appointed as a member of the former Supervisory Board by the Ordinary and Extraordinary General Meeting of Shareholders of December 24, 2020 for a term of four years, i.e. until the end of the General Meeting of Shareholders called in 2024 to approve the financial statements for the fiscal year ended December 31, 2023.
On May 19, 2022, the Ordinary General Meeting of the Company’s Shareholders resolved to adopt a method of management and administration with a Board of Directors and general management. The meeting therefore dismissed all the members of its Supervisory Board, including Mr. Eric Leire.
In addition, the same meeting of May 19, 2022 appointed Mr. Eric Leire as a director of the Company for a term of four (4) years, i.e. until the end of the General Meeting of Shareholders to be held in 2026 to approve the financial statements for the fiscal year ended December 31, 2025.
Other offices and positions held as of December 31, 2022
Positions | Companies | Start dates | ||
| CEO | Genflow Biosciences | 2010 | ||
| President | Immunethep | 2018 | ||
| Non-executive director | BSIM | 2018 | ||
| Non-executive director | Inhatarget | 2020 |
Other offices and positions held over the last five years
Positions | Companies | Dates | ||
| Within the Company | ||||
| Member of the Supervisory Board | Pherecydes Pharma | 2020–2022 | ||
| Outside the Group | ||||
| None | ||||
| Number of shares held as of December 31, 2022: 0 | ||||
| 14.1.3 | Censor |
Pursuant to Article 17 of the articles of association, the Board of Directors may appoint non-voting members. These non-voting members, the number of which may not exceed three (3), form an advisory board. They are freely chosen because of their skills and expertise.
They are appointed for a term of four (4) years expiring at the end of the Ordinary General Meeting of Shareholders called to approve the financial statements for the previous fiscal year and held in the year during which the term of office of said non-voting member expires.
The board of non-voting members examines the questions that the Board of Directors or its President submits to it for review. The non-voting members attend the meetings of the Board of Directors and take part in the deliberations in an advisory capacity only, but their absence does not affect the validity of the deliberations.
They are called to the meetings of the Board of Directors under the same conditions as the voting members. The Board of Directors may compensate the non-voting members by deducting their compensation from the total compensation allocated by the General Meeting of Shareholders to the members of the Board of Directors.
52
As of December 31, 2022, the Company has no non-voting members of the Board of Directors.
| 14.1.4 | Change in the composition of the former Management Board, the former Supervisory Board, and the Board of Directors since January 1, 2022 |
Change in the composition of the former Supervisory Board since January 1, 2022
On May 19, 2022, the Ordinary General Meeting of the Company’s Shareholders resolved to adopt a method of management and administration with a Board of Directors and general management and, as a result, dismissed all the members of its Supervisory Board.
Change in the composition of the former Management Board since January 1, 2022
On May 19, 2022, the Ordinary General Meeting of the Company’s Shareholders resolved to adopt a method of management and administration with a Board of Directors and general management and, as a result, dismissed all the members of its Management Board.
Change in the composition of the Board of Directors since January 1, 2022
The Ordinary General Meeting of the Company’s Shareholders held on May 19, 2022 established the Board of Directors by appointing as directors:
| • | Mr. Didier Hoch; |
| • | Ms. Maryvonne Hiance; |
| • | Mr. Guy Rigaud; |
| • | Mr. Robert Sebbag; |
| • | Mr. Eric Leire; |
| • | The company Go Capital; and |
| • | The company Elaia Partners. |
On May 19, 2022, the Board of Directors of the Company unanimously appointed Mr. Didier Hoch as President of the Board of Directors for the duration of his term of office as a member of the Board of Directors.
Since May 19, 2022, the Board of Directors of the Company has not changed.
Change in the composition of non-voting members of the Board of Directors since January 1, 2022
None.
53
| 14.2 | DELEGATIONSOFAUTHORITYFORCAPITALINCREASESDURINGTHEFISCALYEARENDED DECEMBER 31, 2022 |
The table below summarizes the delegations of authority granted to the Board of Directors by the ordinary and extraordinary general meeting of the shareholders of the Company held on May 19, 2022 (hereinafter, the “CGM”) and by the Extraordinary General Meeting of Shareholders of the Company held on December 15, 2022 (hereinafter, the “EGM”). Please note that the minutes of the EGM and the CGM will soon be available on the Company’s website in the “Shareholders” section (www.pherecydes-pharma.com/).
Delegations of authority granted to the the Extraordinary General Meeting of Shareholders | Maximum nominal amount of capital increase | Expiration of the delegation of authority | Use of delegations of authority granted by the Board of Directors/ Number of shares issued | Price determination methods | ||||
1. Issue of ordinary shares or other securities giving access to the share capital of the Company with preferential subscription rights for Shareholders (fifteenth resolution of the CGM of May 19, 2022). | €4,000,000 | Delegation of authority expired following the implementation of a new delegation of authority by the EGM of December 15, 2022. | None. | The Board of Directors has full powers, including the right to sub-delegate such powers in accordance with the law, to determine, under the conditions laid down by the law and the articles of association, the dates, terms and conditions of any issues, as well as the form and characteristics of the shares or securities giving access to the share capital or debt securities to be issued, with or without premium. |
54
Delegations of authority granted to the the Extraordinary General Meeting of Shareholders | Maximum nominal amount of capital increase | Expiration of the delegation of authority | Use of delegations of authority granted by the Board of Directors/ Number of shares issued | Price determination methods | ||||
2. Issue of ordinary shares or other securities giving access to the share capital of the Company, without the preferential subscription rights of shareholders, by way of a public offering other than those referred to in Article L.411-2-1° of the French Monetary and Financial Code (sixteenth resolution of the CGM of May 19, 2022)(1). | €4,000,000 | Delegation of authority expired following the implementation of a new delegation of authority by the EGM of December 15, 2022. | €215,943/215,943 shares (September 21, 2022). | The amount to be received by the Company for each of the shares issued or to be issued under this delegation of authority, after taking into account, in the event of the issue of autonomous share subscription or allocation warrants, the issue price of said warrants, will be set by the Board of Directors and must be at least equal to the average weighted by the Company share price volumes on the Euronext Growth Paris market over the last five (5) trading sessions preceding the pricing of the issue, potentially reduced by a maximum discount of 25%. |
55
Delegations of authority granted to the the Extraordinary General Meeting of Shareholders | Maximum nominal amount of capital increase | Expiration of the delegation of authority | Use of delegations of authority granted by the Board of Directors/ Number of shares issued | Price determination methods | ||||
3. Issue of ordinary shares or other securities giving access to the share capital of the Company, without the preferential subscription rights of shareholders, up to a maximum of 20% of the share capital per year, in the context of public offerings aimed exclusively at a limited circle of investors acting on their own behalf or at the qualified investors referred to in Article L.411-2-1° of the French Monetary and Financial Code (seventeenth resolution of the CGM of May 19, 2022)(2). | €4,000,000 | Delegation of authority expired following the implementation of a new delegation of authority by the EGM of December 15, 2022. | €345,406/345,406 shares (September 21, 2022). | The amount received or to be received by the Company for each of the shares issued or to be issued under this delegation of authority, after taking into account, in the event of the issue of autonomous share subscription or allocation warrants, the issue price of said warrants, will be set by the Board of Directors and must be at least equal to the average weighted by the Company share price volumes on the Euronext Growth Paris market over the last five (5) trading sessions preceding the pricing of the issue, potentially reduced by a maximum discount of 25%. |
56
Delegations of authority granted to the the Extraordinary General Meeting of Shareholders | Maximum nominal amount of capital increase | Expiration of the delegation of authority | Use of delegations of authority granted by the Board of Directors/ Number of shares issued | Price determination methods | ||||
4. Issue of ordinary shares or other securities giving access to the share capital of the Company to designated categories of investors (eighteenth resolution of the CGM of May 19, 2022)(3). | €4,000,000 | Delegation of authority expired following the implementation of a new delegation of authority by the EGM of December 15, 2022. | €759,481/759,481 shares (September 21, 2022). | The subscription price of such securities and their dividend entitlement date will be determined by the board of directors, it being specified that the amount received or to be received by the Company for each of the shares issued under this delegation of authority will be at least equal to the average weighted by the Company share price volumes on the Euronext Growth Paris market over the last five (5) trading sessions preceding the pricing of the issue. This average may be corrected to take into account differences in the dividend entitlement date and may be reduced by a maximum discount of 25%. |
57
Delegations of authority granted to the the Extraordinary General Meeting of Shareholders | Maximum nominal amount of the capital increase | Expiration of the delegation of authority | Use of delegations of authority granted by the Board of Directors/ Number of shares issued | Price determination methods | ||||
5. Free allocation of new or existing shares of the company (twentieth resolution of the CGM of May 19, 2022)(4). | Free share allocations carried out under this authorization may not give entitlement to a total number of shares exceeding 10% of the number of shares comprising the share capital calculated at the date of allocation. | July 19, 2025. | 58,523 shares (May 19, 2022). | Free. |
58
Delegations of authority granted to the the Extraordinary General Meeting of Shareholders | Maximum nominal amount of the capital increase | Expiration of the delegation of authority | Use of delegations of authority granted by the Board of Directors/ Number of shares issued | Price determination methods | ||||
6. Allocation of Company share subscription or purchase options (twenty-first resolution of the CGM of May 19, 2022)(5). | Share subscription and purchase options granted under this authorization may not give entitlement to a total number of shares exceeding 10% of the number of shares comprising the share capital calculated at the date of allocation. | July 19, 2025. | None. | The exercise price of the options will be set by the Board of Directors on the day the options are granted and may not be less than (a) in the case of subscription options, the lower between 80% of the average of the prices quoted on the five trading sessions preceding the day on which the options are granted and the subscription price of the most recent capital increase preceding the day on which the options are granted, and (b) in the case of purchase options, the value indicated in (a) above, or the average purchase price of the shares referred to in Article L.225-179 of the French Commercial Code. |
59
Delegations of authority granted to the the Extraordinary General Meeting of Shareholders | Maximum nominal amount of the capital increase | Expiration of the delegation of authority | Use of delegations of authority granted by the Board of Directors/ Number of shares issued | Price determination methods | ||||
7. Issue of share subscription warrants, without preferential subscription rights, to a category of persons (twenty-second resolution of the CGM of May 19, 2022). | The exercise of the warrants issued under this resolution may not give rise to a number of ordinary shares representing more than 15% of the number of shares comprising the share capital calculated at the date of allocation. | November 19, 2023. | None. | The Board of Directors shall have full powers, with the option to sub-delegate such powers under legal and regulatory conditions, to determine the subscription and exercise conditions of the share subscription warrants, and in particular the time frames and dates of exercise of the share subscription warrants, the number of shares to which each share subscription warrant entitles the holder, the terms and conditions for paying up the shares subscribed for by exercising the share subscription warrants, as well as their dividend entitlement date, which may be retroactive. |
60
Delegations of authority granted to the the Extraordinary General Meeting of Shareholders | Maximum nominal amount of capital increase | Expiration of the delegation of authority | Use of delegations of authority granted by the Board of Directors/ Number of shares issued | Price determination methods | ||||
8. Issue of ordinary shares or other securities giving access to the share capital of the Company with preferential subscription rights for shareholders (second resolution of the EGM of December 15, 2022). | €4,000,000 | February 15, 2025 | None. | The Board of Directors has full powers, including the right to sub-delegate such powers in accordance with the law, to determine, under the conditions laid down by the law and the articles of association, the amount of the capital increase and, more generally, the amount of the issue in the event of the issue of securities, the issue price and the amount of the premium which may, where applicable, be requested on issue. |
61
Delegations of authority granted to the the Extraordinary General Meeting of Shareholders | Maximum nominal amount of capital increase | Expiration of the delegation of authority | Use of delegations of authority granted by the Board of Directors/ Number of shares issued | Price determination methods | ||||
9. Issue of ordinary shares or other securities giving access to the share capital of the Company, without the preferential subscription rights of shareholders, by way of a public offering, excluding those referred to in Article L.411-2-1° of the French Monetary and Financial Code (third resolution of the EGM of December 15, 2022). | €4,000,000 | February 15, 2025 | None. | The issue price of the shares that may be issued pursuant to this delegation of authority will be set by the Board of Directors and must be at least equal to the average weighted by the Company share price volumes on the Euronext Growth Paris market over the last five (5) trading sessions preceding the pricing of the issue, potentially reduced by a maximum discount of 25%. |
62
Delegations of authority granted to the the Extraordinary General Meeting of Shareholders | Maximum nominal amount of capital increase | Expiration of the delegation of authority | Use of delegations of authority granted by the Board of Directors/ Number of shares issued | Price determination methods | ||||
10. Issue of ordinary shares or other securities giving access to the share capital of the Company, without the preferential subscription rights of shareholders, by way of a public offering referred to in Article L.411-2 1° of the French Monetary and Financial Code (fourth resolution of the EGM of December 15, 2022). | €4,000,000 | February 15, 2025 | None. | The issue price of the shares that may be issued pursuant to this delegation of authority will be set by the Board of Directors and must be at least equal to the average weighted by the Company share price volumes on the Euronext Growth Paris market over the last five (5) trading sessions preceding the pricing of the issue, potentially reduced by a maximum discount of 25%. |
63
Delegations of authority granted to the the Extraordinary General Meeting of Shareholders | Maximum nominal amount of capital increase | Expiration of the delegation of authority | Use of delegations of authority granted by the Board of Directors/ Number of shares issued | Price determination methods | ||||
11. Issue of ordinary shares, without preferential subscription rights, to categories of persons meeting specific characteristics (fifth resolution of the EGM of December 15, 2022)(6). | €4,000,000 | June 15, 2024 | €717,702/717,702 shares February 17, 2023) | The issue price of the shares that may be issued pursuant to this delegation of authority will be set by the Board of Directors and must be at least equal to the average weighted by the Company share price volumes on the Euronext Growth Paris market over the last five (5) trading sessions preceding the pricing of the issue, potentially reduced by a maximum discount of 25%. |
| (1) | The preferential subscription rights of shareholders to the securities covered by this resolution are waived without the designation of specific beneficiaries; |
| (2) | The preferential subscription rights of shareholders to the securities covered by this resolution are waived by way of an offer to qualified investors or to a restricted circle of investors referred to in Article L.411-2-1° of the French Monetary and Financial Code; |
| (3) | The preferential subscription rights of shareholders to the securities covered by this resolution are waived in favor of designated categories of investors; |
| (4) | Free share allocations carried out under this authorization may not give entitlement to a total number of shares exceeding 10% of the number of shares comprising the share capital calculated at the date of allocation; |
| (5) | Share subscription and purchase options granted under this authorization may not give entitlement to a total number of shares exceeding 10% of the number of shares comprising the share capital calculated at the date of allocation; |
| (6) | The preferential subscription rights of shareholders to the securities covered by this resolution are waived in favor of categories of persons meeting specific characteristics. |
64
| 14.3 | AGREEMENTSENTEREDINTOWITHCORPORATEOFFICERS |
| 14.3.1 | Related-party agreements |
The related-party agreements referred to in Articles L.225-38 et seq. of the French Commercial Code are described in chapter 11 of the management report, which contains a list of all agreements authorized by the Board of Directors under the provisions of Articles L.225-38 et seq. of the French Commercial Code.
No other agreements have been concluded between the Company and its corporate officers other than those referred to in this chapter.
| 14.3.2 | Agreements between a corporate officer of the Company or a shareholder holding more than 10% and a company in which Pherecydes Pharma holds, directly or indirectly, more than half of the share capital |
None.
CHAPTER 15—OBSERVATIONS OF THE BOARD OF DIRECTORS
At its meeting of March 30, 2023, the Board of Directors of the Company audited the annual financial statements for the fiscal year ended December 31, 2022.
During this meeting, the Company’s Statutory Auditor presented a summary of their audit on the said statements, which resulted in a report without observations. In addition, the Audit Committee, composed of two members of the Board of Directors, one of whom is independent, presented its conclusions, which corroborate those of the statutory auditor.
In this context and after having deliberated, the Board of Directors unanimously indicated that it had no particular observations to make, either with regard to the Management Board report on the management of the Company or with regard to the annual financial statements for the fiscal year ended December 31, 2022.
Nantes, France,
March 30, 2023
The Board of Directors
65
ANNUAL FINANCIAL STATEMENTS FOR THE FISCAL YEAR ENDED DECEMBER 31, 2022
67
PHERECYDES PHARMA
Table of contents
Balance sheet – Assets | 1 | |||
Balance sheet – Liabilities | 2 | |||
Income statement | 3 | |||
Income statement (continued) | 4 | |||
Explanatory notes | 5 |
| CPA AUDIT |
PHERECYDES PHARMA Financial situation as of 12/31/2022
Balance sheet – Assets
| From 01/01/2022 to 12/31/2022 | To 12/31/2021 | |||||||||||||||
Balance sheet – Assets | Gross | Depr. Amort. Prov. | Net | Net | ||||||||||||
Uncalled capital | ||||||||||||||||
Fixed assets | ||||||||||||||||
Intangible assets | ||||||||||||||||
Preliminary and formation expenses | ||||||||||||||||
Development costs | 5,554,789 | 277,739 | 5,277,049 | |||||||||||||
Concessions, patents and similar rights | 123,633 | 92,550 | 31,083 | 37,253 | ||||||||||||
Goodwill | ||||||||||||||||
Other intangible assets | 5,027,058 | 1,263,418 | 3,763,640 | 6,209,991 | ||||||||||||
Advances and prepayments on intangible assets | ||||||||||||||||
Property, plant and equipment | ||||||||||||||||
Land | ||||||||||||||||
Buildings | ||||||||||||||||
Industrial fixtures, fittings, plant machinery & equipment | 721,995 | 586,901 | 135,094 | 143,476 | ||||||||||||
Other tangible fixed assets | 839,799 | 415,709 | 424,090 | 519,551 | ||||||||||||
Assets under construction | ||||||||||||||||
Advances and prepayments | 113,538 | 113,538 | ||||||||||||||
Financial fixed assets | ||||||||||||||||
Shares in associated companies | ||||||||||||||||
Other equity investments | ||||||||||||||||
Loans to group companies | ||||||||||||||||
Long-term investments | ||||||||||||||||
Other long-term securities | 50,930 | 25,661 | 25,269 | 105,673 | ||||||||||||
Loans | ||||||||||||||||
Other financial fixed assets | 124,556 | 124,556 | 137,040 | |||||||||||||
FIXED ASSETS | 12,556,297 | 2,775,515 | 9,780,782 | 7,152,984 | ||||||||||||
Current assets | ||||||||||||||||
Inventories of products and work in progress | ||||||||||||||||
Raw materials, supplies and other consumables | ||||||||||||||||
Work in progress: | ||||||||||||||||
– For production | ||||||||||||||||
– For services | ||||||||||||||||
Intermediate and finished goods | ||||||||||||||||
Inventories of goods purchased for resale | ||||||||||||||||
Advances and prepayments paid on orders | ||||||||||||||||
Receivables | ||||||||||||||||
Trade accounts receivable | 126,604 | 126,604 | ||||||||||||||
Other | 2,241,186 | 2,241,186 | 1,552,905 | |||||||||||||
Capital called up and unpaid | ||||||||||||||||
Marketable securities | ||||||||||||||||
Company own shares | ||||||||||||||||
Other securities | ||||||||||||||||
Liquid debts | ||||||||||||||||
Cash and cash equivalents | 1,035,127 | 1,035,127 | 5,357,080 | |||||||||||||
Prepaid expenses | 32,412 | 32,412 | 21,904 | |||||||||||||
CURRENT ASSETS | 3,435,329 | 3,435,329 | 6,931,889 | |||||||||||||
Debt issuance costs to be spread over several periods | ||||||||||||||||
Premiums arising from bond redemptions | ||||||||||||||||
Unrealized exchange loss | ||||||||||||||||
|
|
|
|
|
|
|
| |||||||||
TOTAL ASSETS | 15,991,626 | 2,775,515 | 13,216,110 | 14,084,872 | ||||||||||||
|
|
|
|
|
|
|
| |||||||||
| CPA AUDIT | Page 1 |
PHERECYDES PHARMA Financial situation as of 12/31/2022
Balance sheet – Liabilities
| From 01/01/2022 | From 01/01/2021 | |||||||
Balance sheet – Liabilities | To 12/31/2022 | To 12/31/2021 | ||||||
Shareholders’ equity | ||||||||
Equity capital (of which paid up) 7,221,477 | 7,221,477 | 5,852,308 | ||||||
Premiums arising from share issues, mergers, assets brought into business | 8,903,830 | 7,117,723 | ||||||
Revaluation reserves | ||||||||
Equity method evaluation difference | ||||||||
- Legal | ||||||||
- Statutory or contractual Reserves: | ||||||||
- Regulated | ||||||||
- Other | 9,475,980 | 9,483,148 | ||||||
Profits/losses brought forward | -13224788 | -10035731 | ||||||
Result for the year (profit or loss) | -4,854,342 | -3189057 | ||||||
Investment grants | ||||||||
Regulated provisions | ||||||||
SHAREHOLDERS’ EQUITY | 7,522,158 | 9,228,391 | ||||||
Other liable equity capital | ||||||||
Proceeds from issues of equity securities | ||||||||
Conditional advances | 531,882 | 766,667 | ||||||
Other | ||||||||
OTHER LIABLE EQUITY CAPITAL | 531,882 | 766,667 | ||||||
Provisions for contingencies and charges | ||||||||
Provisions for: | ||||||||
- Contingencies | ||||||||
- Charges | ||||||||
PROVISIONS FOR CONTINGENCIES AND CHARGES | ||||||||
Loans and debt | ||||||||
Convertible debenture loans | 9,484 | 23,558 | ||||||
Other debenture loans | ||||||||
- Loans and debt: From credit institutions: | 360,000 | 400,000 | ||||||
- From various lenders | 2,168,780 | 2,000,000 | ||||||
Advances and prepayments received for orders in progress | ||||||||
Financial borrowings and debt: | ||||||||
- Trade accounts payable | 1,589,993 | 784,409 | ||||||
- Tax and payroll | 791,278 | 881,847 | ||||||
- Fixed assets | ||||||||
Other debts | 2,535 | |||||||
Liquid debts | ||||||||
Deferred income | 240,000 | |||||||
LOANS AND DEBT | 5,162,071 | 4,089,814 | ||||||
Unrealized exchange profit | ||||||||
|
|
|
| |||||
TOTAL LIABILITIES | 13,216,110 | 14,084,872 | ||||||
|
|
|
| |||||
| CPA AUDIT | Page 2 |
PHERECYDES PHARMA Financial situation as of 12/31/2022
Income statement
| From 01/01/2022 to 12/31/2022 |
|
| As of 12/31/2021 | |||||||||||||
Income statement | France | Exportation | Total | Total | ||||||||||||
Operating income | ||||||||||||||||
Sales of goods | ||||||||||||||||
Production sold: | ||||||||||||||||
- For production | ||||||||||||||||
- For services | ||||||||||||||||
Net revenue | ||||||||||||||||
Production: | ||||||||||||||||
- Inventoried | ||||||||||||||||
- Capitalized | 3,155,303 | 2,142,291 | ||||||||||||||
Operating grants received | ||||||||||||||||
Reversals of amortization, depreciation, impairment and provisions, transfers of expenses | 5,140 | 58,025 | ||||||||||||||
Other income | 6,792 | 65 | ||||||||||||||
OPERATING INCOME | 3,167,235 | 2,200,380 | ||||||||||||||
Operating expenses | ||||||||||||||||
Purchases of goods (including customs duties) | ||||||||||||||||
Change in inventory (goods) | ||||||||||||||||
Purchases of raw materials, supplies and other consumables | ||||||||||||||||
Change in inventory (raw materials, supplies and other consumables) | ||||||||||||||||
Other external expenses and purchases* | 5,960,963 | 3,386,273 | ||||||||||||||
Taxes and similar levies | 79,361 | 27,678 | ||||||||||||||
Wages and salaries | 2,330,672 | 2,015,555 | ||||||||||||||
Social security contributions | 990,671 | 893,130 | ||||||||||||||
Charges for: | ||||||||||||||||
- Depreciation or amortization on fixed assets | 444,423 | 158,731 | ||||||||||||||
- Impairment of fixed assets | ||||||||||||||||
- Impairment of current assets | ||||||||||||||||
- Provisions for contingencies and charges | ||||||||||||||||
Other charges | 51,188 | 39,708 | ||||||||||||||
OPERATING EXPENSES | 9,857,278 | 6,521,075 | ||||||||||||||
* Including: | ||||||||||||||||
- Equipment leasing royalties | 100,566 | 58,217 | ||||||||||||||
- Property leasing royalties | ||||||||||||||||
OPERATING RESULTS | -6,690,043 | -4,320,694 | ||||||||||||||
Profit allocated or loss transferred | ||||||||||||||||
Loss incurred or profit transferred | ||||||||||||||||
Financial income | ||||||||||||||||
Financial income from investments | ||||||||||||||||
Income from other securities and fixed asset receivables | ||||||||||||||||
Other interest and similar income | 921 | 39 | ||||||||||||||
Reversals of provisions, impairment, and transfers of expenses | ||||||||||||||||
Positive exchange rate differences | 86 | |||||||||||||||
Net income from disposals of marketable securities | ||||||||||||||||
FINANCIAL INCOME | 1,007 | 39 | ||||||||||||||
Financial expenses | ||||||||||||||||
Financial allowances for amortization, depreciation, impairment and provisions | 11,833 | 13,828 | ||||||||||||||
Interest and similar expenses | 52,661 | 32,558 | ||||||||||||||
Negative exchange rate differences | 17 | |||||||||||||||
Net expenses from disposals of marketable securities | ||||||||||||||||
FINANCIAL EXPENSES | 64,511 | 46,386 | ||||||||||||||
FINANCIAL RESULTS | -63,504 | -46,346 | ||||||||||||||
CURRENT RESULTS BEFORE TAXES | -6,753,546 | -4,367,040 | ||||||||||||||
| CPA AUDIT | Page 3 |
PHERECYDES PHARMA Financial situation as of 12/31/2022
Income statement (continued)
Income statement (continued) | As of 12/31/2022 | As of 12/31/2021 | ||||||
Extraordinary income | ||||||||
On management operations | 240,000 | |||||||
On capital transactions | 355,974 | 345,700 | ||||||
Reversals of provisions, impairment, and transfers of expenses | ||||||||
EXTRAORDINARY INCOME | 595,974 | 345,700 | ||||||
Extraordinary expenses | ||||||||
On management operations | 15,000 | |||||||
On capital transactions | 84,849 | 27,692 | ||||||
Extraordinary allowances for amortization, depreciation, impairment and provisions | 113,538 | |||||||
EXTRAORDINARY EXPENSES | 84,849 | 156,230 | ||||||
EXTRAORDINARY RESULTS | 511,125 | 189,470 | ||||||
Employee shareholding | ||||||||
Income tax | -1,388,080 | -988,514 | ||||||
|
|
|
| |||||
TOTAL INCOME | 3,764,216 | 2,546,119 | ||||||
|
|
|
| |||||
TOTAL EXPENSES | 8,618,558 | 5,735,176 | ||||||
|
|
|
| |||||
PROFIT OR LOSS (Total income - Total expenses) | -4,854,342 | -3,189,057 | ||||||
|
|
|
| |||||
| CPA AUDIT | Page 4 |
PHERECYDES PHARMA Financial situation as of 12/31/2022
Explanatory notes
The balance sheet before distribution for the period ended December 31, 2022, which totals €13,216,110, and the income statement for the fiscal year, which shows a profit of –€4,854,342, presented in the form of a list.
This current financial situation covers a period of 12 months, from 01/01/2022 to 12/31/2022. To facilitate comparative reading:
| • | The balance sheet as of December 31, 2022 has been compared to December 31, 2021. |
| • | The income statement as of December 31, 2022 has been compared to December 31, 2021. |
The following notes and tables are an integral part of the financial situation.
| CPA AUDIT | Page 5 |
PHERECYDES PHARMA Financial situation as of 12/31/2022
TABLE OF CONTENTS
TYPE OF ACTIVITY
BASIS FOR THE PREPARATION OF THE ANNUAL FINANCIAL STATEMENTS AND ACCOUNTING PRINCIPLES
| • | Accounting principles and general conventions |
| • | Going concern principles |
| • | Judgments and estimates of the Management of the Company |
| • | Financial statements reporting currency |
SIGNIFICANT EVENTS AND INFORMATION NECESSARY FOR A FAIR PRESENTATION OF RESULTS
| • | Significant events during the reporting period |
| • | Significant events after the reporting period |
ACCOUNTING METHODS AND PRINCIPLES
NOTES TO THE ANNUAL FINANCIAL STATEMENTS
| • | Statement of fixed assets |
| • | Statement of amortization and depreciation |
| • | Analysis of maturities and receivables |
| • | Accrued income and receivables |
| • | Share capital and changes in equity |
| • | Potential share capital |
| • | Conditional advances |
| • | Statement of provisions |
| • | Analysis and maturities of liabilities |
| • | Accrued expenses and credit notes to be issued |
| • | Deferred income and expenses |
| • | Capitalized production |
| • | Other external expenses and purchases |
| • | Non-recurring income from operations |
| • | Corporate tax and loss carryforwards |
OTHER ADDITIONAL INFORMATION
| • | Average workforce |
| • | Off-balance sheet commitments |
| • | Related parties |
| CPA AUDIT | Page 6 |
PHERECYDES PHARMA Financial situation as of 12/31/2022
| 1. | Type of activity |
Pherecydes Pharma conducts research and development activity in the medical field. This includes:
| • | developing expertise; |
| • | obtaining patents and licenses in the field of biology, medicine and, more generally, in the field of life sciences on its own behalf or on behalf of third parties with a view to the marketing and distribution of its products; |
| • | any service activity related to the life sciences field. |
| 2. | Basis for the preparation of the annual financial statements and accounting principles |
ACCOUNTING PRINCIPLES AND GENERAL CONVENTIONS
The annual financial statements have been prepared in accordance with generally accepted accounting principles in France and in compliance with the provisions of ANC regulation number 2014-03, the French Commercial Code and French Decree no. 83-1020 of November 29, 1983.
These financial statements were approved by the board of directors on March 30, 2023.
The general accounting conventions have been applied with regard to the principle of prudence, in accordance with the basic assumptions:
| • | going concern (as defined in Article L.123-20 of the French Commercial Code); |
| • | consistency of accounting methods from one fiscal year to the next; |
| • | intangibility of the opening balance sheet, |
| • | independence of fiscal years; |
and in accordance with the general rules governing the preparation and presentation of annual financial statements.
The basic method used to value items entered in the accounts is the historical cost method.
GOING CONCERN PRINCIPLE
The company’s cash balance on December 31, 2022 amounts to €1,035,127.
The going concern principle of accounting has been adopted in view of the following elements:
| • | The Company’s historical deficit position is explained by the innovative nature of the products under development, which involves a research and development phase lasting several years. |
| • | The outlook for the Company’s growth over the next twelve months, on which the budget is based, are considered reasonable in the light of the Company’s recent development. |
| • | On September 22, 2022, Pherecydes Pharma successfully carried out a capital increase of €3.1 million, of which €2.6 million from institutional and historical investors and €0.5 million from individuals, via the PrimaryBid platform. |
| • | On February 15, 2023, Pherecydes Pharma and ERYTECH Pharma (“ERYTECH”; Nasdaq & Euronext: ERYP), a clinical-stage biopharmaceutical company developing innovative therapies by encapsulating therapeutic drug substances inside red blood cells, announced their proposed strategic combination intended to create a global player in extended phagotherapy and accelerate the development of a portfolio of drug candidates targeting pathogenic bacteria and potential other indications of high unmet medical needs. |
The strategic combination would build on the complementary expertise and capabilities of both companies to accelerate the development of extended phagotherapies for antimicrobial resistance, in particular via the PhagoDAIR phase II study conducted by PHERECYDES, as well as other anti-infective fields and therapeutic areas with high unmet medical needs.
The cash runway of the new entity resulting from the merger would extend into the third quarter of 2024, with a consolidated cash position of approximately €41 million as of December 31, 2022, and would enable funding of existing and novel programs through multiple clinical milestones.
This transaction would pave the way for the expansion of PHERECYDES development programs in antimicrobial resistance and complete the strategic review process announced by ERYTECH on several occasions since November 2021.
The transaction expected to close at the end of the second quarter of 2023.
The transaction, supported by the key shareholders of PHERECYDES and ERYTECH, will result in former PHERECYDES shareholders holding approximately 49% of the combined entity.
ERYTECH and PHERECYDES intend to merge their operations and relocate all teams to ERYTECH’s premises in Lyon, France, where they will benefit from being located within a major European center for infectious diseases.
| • | On February 17, 2023, Pherecydes Pharma successfully completed a €1.5 million capital increase |
| • | At the same time, and to meet its cash needs to finance its activities for the next twelve months, the Company is seeking additional financing solutions of various kinds, including equity financing and simple or bonded debt, from third parties or its shareholders, as well as grants and/or repayable advances specifically related to the Company’s research programs. |
| CPA AUDIT | Page 7 |
PHERECYDES PHARMA Financial situation as of 12/31/2022
JUDGMENTS AND ESTIMATES OF THE MANAGEMENT OF THE COMPANY
The preparation of financial statements requires management to make judgments, estimates and assumptions that have an impact on the amounts of assets and liabilities, as well as on the income statement items for the period. These estimates consider economic data that may change over time and include contingencies.
The estimates and underlying assumptions are based on past experience and other factors considered reasonable in light of current and anticipated circumstances and the economic environment. They thus serve as a basis for exercising the judgment required to determine the book value of assets and liabilities that cannot be obtained directly from other sources. Actual values may differ from estimated values.
The estimates and underlying assumptions are reviewed on an ongoing basis. The principal estimates are related to:
| • | The life of fixed assets; |
| • | The value of capitalized development costs; |
| • | The value of capitalized production; |
| • | The Research Tax Credit. |
FINANCIAL STATEMENTS REPORTING CURRENCY
The financial statements, as well as all the notes and tables presented in these explanatory notes, are expressed in euros.
| 3. | Significant events and information necessary for a fair presentation of results |
| 3.1. | Significant events during the reporting period from January 1 to December 31, 2022 |
| • | Obtaining a €2.1 million grant for the PhagECOLI project conducted in partnership with the CEA |
In January 2022, the company announced that the PhagECOLI project was selected for the “Emerging infectious diseases and new RBC threats”
Call for Expressions of Interest. Within this framework, the partnership will receive grants and repayable advances for an amount of €2.1 million over the three years of the project’s implementation.
| • | Obtaining CPP approval to initiate the PhagoDAIR program |
In January 2022, the company received CPP approval for the PhagoDAIR study. With this green light, the Company has all the regulatory approvals to initiate this phase I/II study.
| • | Capital increases |
The company carried out four capital increases during the first half of 2022:
| • | By allocating 7,168 free shares on February 4, 2022 with a nominal value of €1, i.e. a capital increase of €7,168. |
| • | By exercising a start-up stock option (BSPCE) in April 2022 for a total of 10,116 shares with a nominal value of €1, which resulted in a capital increase of €10,116 and a total share issue premium of €10,470.06 |
| • | By exercising a start-up stock option in July 2022 for a total of 5,396 shares with a nominal value of €1, which resulted in a capital increase of €5,396 and a total share issue premium of €5,584.86 |
| • | By exercising a start-up stock option in August 2022 for a total of 25,659 shares with a nominal value of €1, which resulted in a capital increase of €25,659 and a total share issue premium of €26,557.06 |
| • | By fundraising in September 2022 for a total of 1,320,830 shares with a nominal value of €1, which resulted in a capital increase of €1,320,830 and a total share issue premium of €1,743,495. |
| CPA AUDIT | Page 8 |
PHERECYDES PHARMA Financial situation as of 12/31/2022
| • | Conclusion of a grant and repayable advance contract |
In February 2022, BPIFRANCE entered into a grant and repayable advance agreement with Pherecydes Pharma to finance the PhagECOLI program for an amount of €1,687,800. This financing will be 60% in the form of a grant and 40% in the form of a repayable advance, to be paid after each milestone and conditional on Pherecydes Pharma providing a certain number of elements.
The advance is repayable quarterly starting on December 31, 2026, unless the program is unsuccessful.
As of December 31, 2022, €169 thousand have been received in repayable advances, and €253 thousand in grants.
| 3.2. | Significant events after the reporting period |
| • | Capital increase |
On February 22, 2023, management decided to carry out a capital increase through the issue of 717,702 new shares with a nominal value of €1 each and a total share issue premium of €782,295.18 (i.e., a share issue premium of €1.09 per ordinary share.)
| • | Strategic combination with Erytech |
On February 15, 2023, Pherecydes Pharma and ERYTECH Pharma (“ERYTECH”; Nasdaq & Euronext: ERYP), a clinical-stage biopharmaceutical company developing innovative therapies by encapsulating therapeutic drug substances inside red blood cells, announced their proposed strategic combination intended to create a global player in extended phagotherapy and accelerate the development of a portfolio of drug candidates targeting pathogenic bacteria and potential other indications of high unmet medical needs.
| 4. | Accounting methods and principles |
| 4.1. | RESEARCH AND DEVELOPMENT COSTS |
The capitalized development costs relate to clearly individualized subjects with a high probability of technical success and commercial profitability or economic viability. Pherecydes Pharma has considered as eligible for capitalization the development in-progress costs of products for which the regulatory authorities have validated the Company’s good manufacturing practices or have given the Company authorization to proceed with human clinical trials and/or Early Access Approval (AAC).
As of December 31, 2022, the Company has capitalized research and development expenses for a gross value of €5,554,789. See notes 5.12 and 5.14.
| CPA AUDIT | Page 9 |
PHERECYDES PHARMA Financial situation as of 12/31/2022
| 4.2. | PROPERTY, PLANT AND EQUIPMENT AND INTANGIBLE ASSETS |
Intangible assets
Intangible assets are recorded at their initial acquisition cost minus accumulated amortization and impairment losses.
All intangible assets have a finite life and are amortized over their estimated useful lives using the straight-line method on the following bases:
| • | Patents: 20 years |
However, an impairment test is performed whenever there is an indication of impairment loss.
In the income statement, depreciation, amortization, and impairment charges on intangible assets are recorded under “Depreciation or amortization charges on fixed assets” or “Provisions on fixed assets.”
Property, plant, and equipment
Property, plant, and equipment are valued at acquisition or production cost, taking into account the costs required to bring the assets into use and after deducting trade discounts, rebates and cash discounts obtained, minus accumulated depreciation.
All property, plant and equipment are depreciated over their useful lives using the straight-line method on the following bases:
| Industrial machinery | 6 years | |
| Work and installations | from 3 to 10 years | |
| Office and IT equipment | from 2 to 5 years | |
| Furniture | 5 years |
In the income statement, depreciation charges on property, plant and equipment are recorded under “Depreciation or amortization charges on fixed assets.”
| 4.3. | FINANCIAL ASSETS |
The gross value is the purchase cost excluding incidental expenses. When the inventory value is lower than the gross value, an impairment is recorded for the difference.
| 4.4. | RECEIVABLES AND FINANCIAL BORROWINGS AND DEBT |
Receivables and payables are valued at their nominal value. An impairment is recorded when the inventory value is lower than the book value.
| 4.5. | CASH AND CASH EQUIVALENTS |
Cash and cash equivalents include those in banks and in hand. Cash and cash equivalents are valued at their nominal value.
| 4.6. | OTHER LIABLE EQUITY CAPITAL |
The Company benefits from three conditional advances granted by BPI France. Repayment of this advance is conditional on the success of the subsidized project.
The balance of the conditional advances is reduced by the repayments made according to the contractual schedule.
For easier reading, these advances, which appeared on the “Other” line of the “Other liable equity capital” item, have been repositioned on the “Conditional advances” line of this same item in the company’s balance sheet on December 31, 2022.
| 4.7. | OTHER LOANS AND FINANCIAL DEBT |
For easier reading, the loans taken out with BPI, which appeared on the “Loans and debt from credit institutions” line have been repositioned on the “Other loans and financial debt” line in the company’s balance sheet on December 31, 2022.
| CPA AUDIT | Page 10 |
PHERECYDES PHARMA Financial situation as of 12/31/2022
| 4.8. | GRANTS |
The grants received are investment grants insofar as they finance long-term activities.
When these grants are definitively acquired by the company (i.e., there is no risk of them being challenged or needing to be repaid, in particular through conditions precedent), they are recognized, in proportion to the progress of the subsidized study:
| • | either as extraordinary income, if the financed expenses have been recorded as expenses (recognized for the share of the expenses incurred); |
| • | or as shareholders’ equity if the financed expenses have been capitalized (recognized over the same period and at the same rate as the fixed asset). |
Where applicable, they are recognized as advances and prepayments received under liabilities for financial borrowings and debt.
| 4.9. | EXCEPTIONAL RESULTS |
Income and charges which, by their nature or occurrence, are not part of the Company’s regular activities, are recognized as extraordinary results.
| 4.10. | PENSION LIABILITIES |
The only pension liability borne by the Company is that of retirement bonuses for employees. Legislation requires a bonus to be paid to employees when they retire based on their length of service and their salary at retirement age.
The Company has decided not to make any provision in its financial statements for retirement bonuses or pension supplements for its employees. The corresponding expense is recorded in the period during which the bonus is actually paid. The liabilities are presented as off-balance sheet commitments. See note 5.16.
| 5. | NOTES TO THE ANNUAL FINANCIAL STATEMENTS |
| 5.1. | Statement of fixed assets |
Framework A | Gross value at beginning of fiscal year | Increases | ||||||||||
Intangible assets | Revaluation for the fiscal year | Acquisitions, receivables, transfers | ||||||||||
Research and development costs | 5,554,789 | |||||||||||
Other intangible asset items concessions patents | 7,597,042 | 3,108,438 | ||||||||||
|
|
|
| |||||||||
TOTAL | 7,597,042 | 8,663,227 | ||||||||||
|
|
|
| |||||||||
Property, plant, and equipment | ||||||||||||
Land | ||||||||||||
Buildings on own land Buildings on land owned by third parties Building general facilities, fixtures, and fittings | ||||||||||||
Industrial fixtures, fittings, plant machinery & equipment | 672,286 | 49,709 | ||||||||||
Other general facilities, fixtures, and fittings Transportation equipment Office and IT equipment, furniture | 596,504 | |||||||||||
Reusable packaging materials and other | 236,333 | 6,963 | ||||||||||
Tangible assets in progress | ||||||||||||
Advances and prepayments | 113,538 | |||||||||||
|
| |||||||||||
TOTAL | 1,618,660 | 56,671 | ||||||||||
|
|
|
| |||||||||
Financial fixed assets | ||||||||||||
Shares in associated companies Other equity investments Other long-term securities | 119,501 | |||||||||||
Loans and other financial fixed investments | 137,040 | 3,421 | ||||||||||
|
|
|
| |||||||||
TOTAL | 256,541 | 3,421 | ||||||||||
|
|
|
| |||||||||
OVERALL TOTAL | 9,472,244 | 8,723,319 | ||||||||||
|
|
|
| |||||||||
| CPA AUDIT | Page 11 |
PHERECYDES PHARMA Financial situation as of 12/31/2022
Framework B | Decreases | Gross value of fixed assets at end of fiscal year | Statutory reval. or valu. using the equity method | |||||||||||||
| Transfer | Disposal | Original value of fixed assets at end of fiscal year | ||||||||||||||
Research and development costs | 5,554,789 | |||||||||||||||
Other intangible asset items | 5,554,789 | 5,150,690 | ||||||||||||||
|
|
|
| |||||||||||||
TOTAL | 5,554,789 | 10,705,479 | ||||||||||||||
|
|
|
| |||||||||||||
Land | ||||||||||||||||
Buildings on own land | ||||||||||||||||
Buildings on land owned by third parties Building general facilities, fixtures, and fittings Industrial fixtures, fittings, plant machinery & equipment | 721,995 | |||||||||||||||
Other general facilities, fixtures, and fittings | 596,504 | |||||||||||||||
Transportation equipment Office and IT equipment, furniture | 243,295 | |||||||||||||||
Reusable packaging materials and other | ||||||||||||||||
Tangible assets in progress | ||||||||||||||||
Advances and prepayments | 113,538 | |||||||||||||||
|
| |||||||||||||||
TOTAL | 1,675,332 | |||||||||||||||
|
| |||||||||||||||
Shares in associated companies | ||||||||||||||||
Other equity investments | ||||||||||||||||
Other long-term securities | 68,571 | 50,930 | ||||||||||||||
Loans and other financial fixed investments | 15,904 | 124,556 | ||||||||||||||
|
|
|
| |||||||||||||
TOTAL | 84,475 | 175,486 | ||||||||||||||
|
|
|
|
|
|
|
| |||||||||
OVERALL TOTAL | 5,554,789 | 84,475 | 12,556,297 | |||||||||||||
|
|
|
|
|
| |||||||||||
| CPA AUDIT | Page 12 |
PHERECYDES PHARMA Financial situation as of 12/31/2022
The gross value of research and development costs amounts to €10,705 thousand, of which €5,151 thousand in assets under construction and €5,555 thousand in intangible assets made available during the anti-Staphylococcus aureus program.
As of December 31, 2022, the gross costs recorded as assets under construction amounted to €5,151 thousand. They mainly correspond to development costs for various assets:
| • | PSEUDO for total gross amount of €3,778 thousand |
| • | PHAGOBURN for total gross amount of €1,263 thousand |
Other intangible assets correspond to patents. The Company has four patent families (two for anti-Pseudomonas aeruginosa phages, one for anti-Staphylococcus aureus phages and one for anti-E. coli phages). These patents have been granted in some jurisdictions and are pending in others.
| CPA AUDIT | Page 13 |
PHERECYDES PHARMA Financial situation as of 12/31/2022
| 5.2. | Statement of amortization and depreciation and impairment |
| Framework A | ||||||||||||||||
SITUATIONS AND MOVEMENTS DURING THE FISCAL YEAR | ||||||||||||||||
DEPRECIABLE OR AMORTIZABLE ASSETS | Amount at the beginning of the fiscal year | Increase | Decrease | Amount at the end of the fiscal year | ||||||||||||
Intangible assets | ||||||||||||||||
Research and development costs | 277,739 | 277,739 | ||||||||||||||
Software | 86,380 | 6,170 | 92,550 | |||||||||||||
R&D cost provision | 1,263,418 | 1,263,418 | ||||||||||||||
Assets under construction | 113,537 | 113,537.6 | ||||||||||||||
|
|
|
|
|
|
|
| |||||||||
TOTAL | 1,463,335 | 283,909 | 1,747,245 | |||||||||||||
|
|
|
|
|
|
|
| |||||||||
Property, plant, and equipment | ||||||||||||||||
Land | ||||||||||||||||
Buildings on own land | ||||||||||||||||
Buildings on land owned by third parties | ||||||||||||||||
Building general facilities, fixtures, and fittings | ||||||||||||||||
Industrial fixtures, fittings, plant machinery & equip. | 528,810 | 58,090 | 586,901 | |||||||||||||
Other general facilities, fixtures, and fittings | 182,733 | 58,414 | 241,147 | |||||||||||||
Transportation equipment | ||||||||||||||||
Office and IT equipment, furniture | 130,553 | 44,009 | 174,562 | |||||||||||||
Reusable packaging materials and other | ||||||||||||||||
|
|
|
|
|
|
|
| |||||||||
TOTAL | 842,096 | 160,514 | 1,002,609 | |||||||||||||
|
|
|
|
|
|
|
| |||||||||
OVERALL TOTAL | 2,305,431 | 444,423 | 2,749,854 | |||||||||||||
|
|
|
|
|
|
|
| |||||||||
The impairment of €1,263 thousand corresponds to expenses related to the Phagoburn project, which stopped in 2018. As the analyses performed in 2018 showed negative results, the phages were not usable and therefore the project could not, in its current state, generate future revenue. As a result, the total amount of capitalized costs on this project had been impaired in 2018, as a matter of prudence, pending the possible resumption of the project.
| CPA AUDIT | Page 14 |
PHERECYDES PHARMA Financial situation as of 12/31/2022
| 5.3. | Analysis of maturities and receivables |
Receivables(a) | Gross amount | Asset liquidity | ||||||||||
| Maturities of less than 1 year | Maturities of more than 1 year | |||||||||||
Non-current assets | ||||||||||||
Loans to group companies | ||||||||||||
Loans (1)(2) | ||||||||||||
Other financial fixed assets | 124,556 | 124,556 | ||||||||||
Current assets | ||||||||||||
Doubtful or disputed receivables | ||||||||||||
Other trade accounts receivable | 126,604 | 126,604 | ||||||||||
Receivables on loaned securities | ||||||||||||
Personnel and related expenses | 2,171 | 2,171 | ||||||||||
Social security and related liabilities | 3,717 | 3,717 | ||||||||||
Income tax | 1,388,080 | 1,388,080 | ||||||||||
Value added tax | 345,305 | 345,305 | ||||||||||
Other taxes and similar levies | ||||||||||||
Other | ||||||||||||
Trade accounts payable to group companies and associates (2) | ||||||||||||
Other receivables (including receivables from repurchase agreements) | 479,698 | 479,698 | ||||||||||
Prepaid expenses | 32,412 | 32,412 | ||||||||||
|
|
|
|
|
| |||||||
TOTAL | 2,502,543 | 2,377,978 | 124,556 | |||||||||
|
|
|
|
|
| |||||||
| (1) | Loans granted during the year |
(1) Repayments received during the year
(2) Loans and advances granted to individuals
| 5.4. | Accrued income and receivables |
(Article R.123-189 of the French Commercial Code)
ACCRUED INCOME INCLUDED IN THE | Fiscal year ended | Fiscal year ended | ||||||
| 12/31/2022 | 12/31/2021 | |||||||
Loans to group companies | ||||||||
Other long-term securities | ||||||||
Loans | ||||||||
Other financial fixed assets | ||||||||
Trade accounts receivable | ||||||||
Other accounts receivable | 3,717 | 3,063 | ||||||
Marketable securities | ||||||||
Cash and cash equivalents | ||||||||
|
|
|
| |||||
TOTAL | 3,717 | 3,063 | ||||||
|
|
|
| |||||
| CPA AUDIT | Page 15 |
PHERECYDES PHARMA Financial situation as of 12/31/2022
| 5.5. | Share capital and changes in equity |
On December 31, 2022, the share capital amounted to €7,221,477.
It is divided into 7,221,477 shares with a nominal value of €1 each
The change in share capital over the period is as follows:
| Quantity | Value | |||||||||||||||||||||||
Shareholders | Ordinary shares | P1 shares | Q2 Share subscription warrants (BSA T2) | Total shares | Security value | Share capital amount | ||||||||||||||||||
12/31/2021 | 5,852,308 | — | — | 5,852,308 | € | 1.00 | € | 5,852,308 | ||||||||||||||||
12/31/2022 | 7,221,477 | — | — | 7,221,477 | € | 1.00 | € | 7,221,477 | ||||||||||||||||
Change | 1,369,169 | — | — | 1,369,169 | € | 1.00 | € | 1,369,169 | ||||||||||||||||
The table of changes in shareholders’ equity is as follows:
in euros | 12/31/2021 | Allocation of income | Exercise of start-up stock options | Free share allocation | Capital increase (fundraising) | 12/31/2022 | ||||||||||||||||||
Share capital | 5,852,308 | 41,171 | 7,168 | 1,320,830 | 7,221,477 | |||||||||||||||||||
Premiums arising from share issues, mergers, assets brought into business | 7,117,723 | 42,612 | 1,743,495 | 8,903,830 | ||||||||||||||||||||
Statutory reserves | — | |||||||||||||||||||||||
Other reserves | 9,483,148 | -7,168 | 9,475,980 | |||||||||||||||||||||
Profits/losses brought forward | -10,035,731 | -3,189,057 | -13,224,788 | |||||||||||||||||||||
Result for the fiscal year | -3,189,057 | 3,189,057 | -4,854,342 | |||||||||||||||||||||
Shareholders’ equity | 9,228,391 | — | 83,783 | — | 3,064,325 | 7,522,158 | ||||||||||||||||||
| 5.6. | Potential share capital |
| CPA AUDIT | Page 16 |
PHERECYDES PHARMA Financial situation as of 12/31/2022
The table below details the total number of shares that may be issued by type of financial instrument on December 31, 2022:
Potential share capital | Number | Parity | Maximum number of shares to be issued | |||||||||
2017 start-up stock option | — | 1 for 1 | — | |||||||||
2018 start-up stock option | 4,200 | 1 for 1 | 4,200 | |||||||||
BSPCE 2019 | 29,538 | 1 for 1 | 29,538 | |||||||||
2020 start-up stock option | 144,200 | 1 for 1 | 144,200 | |||||||||
2021 start-up stock option | 437,977 | 1 for 1 | 437,977 | |||||||||
2022 start-up stock option | — | 1 for 1 | — | |||||||||
Number of potential shares | 615,915 | |||||||||||
Number of existing shares | 7,221,477 | |||||||||||
Overall dilution | 7.85867 | % | ||||||||||
The impact of the overall dilution on the situation of the associates following the exercise of all the instruments detailed above would be:
| Effect of dilution on earnings per share | Situation of the shareholder before | Situation of the shareholder after | ||||||
For €1 of earnings per share | 1.000000 | 0.92145736 | ||||||
The characteristics of the capital instruments issued are summarized below.
| • | Start-up stock options: |
The number of start-up stock options on December 31, 2022 is 615,450, or:
| • | 0 x 2017 start-up stock options |
| • | 4,200 x 2018 start-up stock options |
| • | 29,538 x 2019 start-up stock options |
| • | 144,200 x 2020 start-up stock options |
| • | 437,977 x 2021 start-up stock options |
| • | 0 x 2022 start-up stock options |
| 5.7. | Conditional advances |
On December 31, 2022, conditional advances amounted to €531,882, with the following characteristics:
| • | PHAGOGRAM advance received from BPI France |
| • | Amount received: €117,882 |
| • | Balance of the remaining advance receivable: €0 |
| • | PHAGOSCLIN advance received from BPI France |
| • | Amount received: €700,000 |
| • | Amount to be repaid: €414,000 |
| • | The Company was granted a €240,000 debt write-off in the first half of 2022. |
| • | PHAGECOLI advance received from BPI France |
| • | Amount received: €168,780 |
| • | Balance of the remaining advance receivable: €506,340 – pending completion of milestones 1 and 2 |
Estimated repayment schedule:
Project | < 1 year | 1 to 5 years | 5 years | |||||||||
Phagogram | 117,882 | |||||||||||
Phagosclin | 69,000 | 345,000 | ||||||||||
PhagECOLI | 168,780 | |||||||||||
|
|
|
|
|
| |||||||
Total | 69,000 | 631,662 | — | |||||||||
|
|
|
|
|
| |||||||
| CPA AUDIT | Page 17 |
PHERECYDES PHARMA Financial situation as of 12/31/2022
| 5.8. | Statement of provisions and impairments |
BREAKDOWN OF PROVISIONS | Amount at the beginning of the fiscal year | Increase in charges for the fiscal year | Decreases reversed at the end of the fiscal year | Amount at the end of the fiscal year | ||||||||||||
Regulated | ||||||||||||||||
Provision for the reconstitution of mining deposits | ||||||||||||||||
Provisions for capital expenditure | ||||||||||||||||
Provisions for price increases | ||||||||||||||||
Accelerated depreciation or amortization | ||||||||||||||||
Tax Prov. for foreign operations before 01/01/1992 | ||||||||||||||||
Tax Prov. for foreign operations after 01/01/1992 | ||||||||||||||||
Provisions for start-up loans | ||||||||||||||||
Other regulated provisions | ||||||||||||||||
|
|
|
|
|
|
|
| |||||||||
TOTAL | ||||||||||||||||
|
|
|
|
|
|
|
| |||||||||
Contingencies and charges | ||||||||||||||||
Provisions for disputes | ||||||||||||||||
Provisions for guarantees given to customers | ||||||||||||||||
Provisions for losses on futures contracts | ||||||||||||||||
Provisions for fines and penalties | ||||||||||||||||
Provisions for foreign exchange losses | ||||||||||||||||
Provisions for pensions and benefit obligations | ||||||||||||||||
Provisions for taxes | ||||||||||||||||
Provisions for replacement of fixed assets | ||||||||||||||||
Provisions for major repairs and maintenance | ||||||||||||||||
Provisions for social security charges on paid leave | ||||||||||||||||
Other provisions for contingencies and charges | ||||||||||||||||
|
|
|
|
|
|
|
| |||||||||
TOTAL | ||||||||||||||||
|
|
|
|
|
|
|
| |||||||||
| CPA AUDIT | Page 18 |
PHERECYDES PHARMA Financial situation as of 12/31/2022
BREAKDOWN OF IMPAIRMENT LOSSES | Amount at the beginning of the fiscal year | Increase in charges for the fiscal year | Decreases reversed at the end of the fiscal year | Amount at the end of the fiscal year | ||||||||||||
Impairment | ||||||||||||||||
Intangible assets | 1,376,956 | 1,376,956 | ||||||||||||||
Property, plant, and equipment | ||||||||||||||||
Fixed assets – investments in associates | ||||||||||||||||
Fixed assets – investments in subsidiaries and affiliates | ||||||||||||||||
Financial fixed assets | 13,828 | 61,280 | 49,447 | 25,661 | ||||||||||||
On inventories and work in progress | ||||||||||||||||
On trade accounts receivables | ||||||||||||||||
Other provisions for impairment losses | ||||||||||||||||
|
|
|
|
|
|
|
| |||||||||
TOTAL | 1,390,783 | 61,280 | 49,447 | 1,402,616 | ||||||||||||
|
|
|
|
|
|
|
| |||||||||
OVERALL TOTAL | 1,390,783 | 61,280 | 49,447 | 1,402,616 | ||||||||||||
|
|
|
|
|
|
|
| |||||||||
Of which charges and reversals: – operational | ||||||||||||||||
Of which charges and reversals: – financial | 11,833 | |||||||||||||||
Of which charges and reversals: – extraordinary | ||||||||||||||||
Investments in associates: Total impairment loss |
| |||||||||||||||
| CPA AUDIT | Page 19 |
PHERECYDES PHARMA Financial situation as of 12/31/2022
| 5.9. | Analysis and maturities of liabilities |
Financial borrowings and debt (b) | Gross amount | Degree of liability repayment | ||||||||||||||
| Maturities of less than 1 year | Maturities | |||||||||||||||
| Of more than 1 year | Of more than 5 years | |||||||||||||||
Convertible debenture loans (1) | 9,484 | 9,484 | ||||||||||||||
Other debenture loans (1) | ||||||||||||||||
Loans and debt from credit institutions (1) | ||||||||||||||||
– less than 1 year at inception | ||||||||||||||||
– more than 1 year at inception | 360,000 | 360,000 | ||||||||||||||
Other loans and financial debt (1)(2) | 2,700,662 | 2,700,662 | ||||||||||||||
Trade accounts payable | 1,191,929 | 1,191,929 | ||||||||||||||
Personnel and related expenses | 356,314 | 356,314 | ||||||||||||||
Social security and related liabilities | 390,482 | 390,482 | ||||||||||||||
Income tax | ||||||||||||||||
Value added tax | ||||||||||||||||
Secured bonds | ||||||||||||||||
Other taxes and similar | 44,482 | 44,482 | ||||||||||||||
Trade accounts payable – fixed assets | ||||||||||||||||
Trade accounts payable to group companies and associates (2) | ||||||||||||||||
Other payables (including payables on repurchase agreements) | 2,535 | 2,535 | ||||||||||||||
Payables on borrowed securities | ||||||||||||||||
Deferred income | 240,000 | 240,000 | ||||||||||||||
|
|
|
| |||||||||||||
TOTAL | 5,295,888 | 5,295,888 | ||||||||||||||
|
|
|
| |||||||||||||
| (1) | Loans taken out during the year |
| (1) | Loans repaid during the year |
| (2) | Loans and debt contracted with individual associates |
Trade accounts payables consist mainly of €376,135 in invoices not yet received and €1,191,929 in supplier invoices.
| CPA AUDIT | Page 20 |
PHERECYDES PHARMA Financial situation as of 12/31/2022
| 5.10. | Accrued expenses and credit notes to be issued |
(Article R.123-189 of the French Commercial Code)
ACCRUED CHARGES INCLUDED IN THE FOLLOWING BALANCE SHEET ITEMS | Fiscal year ended | Fiscal year ended | ||||||
| 12/31/2022 | 12/31/2021 | |||||||
Convertible debenture loans | ||||||||
Other debenture loans | ||||||||
Loans and debt from credit institutions | ||||||||
Other loans and financial debt | ||||||||
Trade accounts payable | 376,135 | 498,760 | ||||||
Debts Tax and payroll | 529,808 | 477,889 | ||||||
Trade accounts payable – fixed assets | ||||||||
Other debts | ||||||||
|
|
|
| |||||
TOTAL | 905,943 | 976,649 | ||||||
|
|
|
| |||||
| 5.11. | Deferred income and expenses |
DEFERRED INCOME | Fiscal year ended | Fiscal year ended | ||||||
| 12/31/2022 | 12/31/2021 | |||||||
Operating income | 240,000 | |||||||
Financial income | ||||||||
Extraordinary income | ||||||||
|
| |||||||
TOTAL | 240,000 | |||||||
|
| |||||||
PREPAID EXPENSES | Fiscal year ended | Fiscal year ended | ||||||
| 12/31/2022 | 12/31/2021 | |||||||
Operating expenses | 32,412 | 21,904 | ||||||
Financial expenses | ||||||||
Extraordinary expenses | ||||||||
|
|
|
| |||||
TOTAL | 32,412 | 21,904 | ||||||
|
|
|
| |||||
| 5.12. | Capitalized production |
Capitalized production for the period amounts to €3,155,303 and corresponds to development costs capitalized during the period.
| CPA AUDIT | Page 21 |
PHERECYDES PHARMA Financial situation as of 12/31/2022
| 5.13. | Other external expenses and purchases |
As of December 31, 2022, other external expenses and purchases are broken down as follows:
Other external expenses and purchases | 12/31/2022 | 12/31/2021 | ||||||
Sub-contracting | 2,356,723 | 1,045,683 | ||||||
IT consulting | 7,765 | 23,315 | ||||||
Electricity | 25,372 | 36,903 | ||||||
Purchase of small equipment and consumables | 891,758 | 437,572 | ||||||
Managed services | 6,912 | 47,158 | ||||||
Equipment leasing | 100,566 | 58,217 | ||||||
Rentals | 271,608 | 259,294 | ||||||
Rental charges | 164,438 | 138,606 | ||||||
Maintenance and repairs | 55,452 | 43,022 | ||||||
Insurance | 43,846 | 31,438 | ||||||
Documentation | 53,896 | 5,518 | ||||||
Temporary staff | 3,500 | 0 | ||||||
Legal costs and fees | 1,744,124 | 1,164,965 | ||||||
Advertising, catalogs and printed materials | 1,275 | 3,000 | ||||||
Transportation of goods | 116,414 | 36,987 | ||||||
Travel and entertainment expenses | 75,257 | 26,433 | ||||||
Telephone and internet | 17,813 | 10,339 | ||||||
Bank services | 6,590 | 5,683 | ||||||
Dues and other contributions | 17,654 | 12,141 | ||||||
|
|
|
| |||||
Total | 5,960,963 | 3,386,273 | ||||||
|
|
|
| |||||
| 5.14. | Non-recurring income from operations |
Extraordinary income relates to the share of investment grants recognized in the income statement in the amount of €355,974 and to the debt write-off on the advance received from BPI France for the PHAGOSCLIN project in the amount of €240 thousand.
Grant income recognized in fiscal year 2022 amounted to €355 thousand: €102 thousand for PHAGOGRAM and €253 thousand for PHAGECOLI.
For the record:
| • | PHAGOGRAM grant received from BPI France |
| • | Total amount of the grant: euros253,763 |
| • | Funds received as of December 31, 2022: euros253,763 |
| • | PHAGECOLI grant received from BPI France |
| • | Total amount of the grant: €1,012,680 |
| • | Funds received as of December 31, 2022: euros253,170 |
| 5.15. | Corporate tax and loss carryforwards |
As the Company does not generate taxable income, it does not incur any tax expenses. The amount recognized in the income statement for corporate income tax is composed of the Research Tax Credit (RTC).
The Company’s research and development work generated an RTC of €989 thousand for 2021, for which reimbursement was requested and received in September 2022.
| CPA AUDIT | Page 22 |
PHERECYDES PHARMA Financial situation as of 12/31/2022
For 2022, the research tax credit is estimated at €1,388 thousand.
The calculation bases are as follows:
Base | June 22 | |||
Personnel | 1,838,202 | |||
Charges | 64,260 | |||
Patents | 268,138 | |||
R&D sub-contracting | 2,416,694 | |||
Grant | 942,602 | |||
| 5.16. | Additional information |
| • | Average workforce |
| Paid staff | Staff seconded to the company | |||||||
Employees | 27 | |||||||
|
| |||||||
TOTAL | 27 | |||||||
|
| |||||||
| • | Off-balance sheet commitments |
Retirement bonuses
As of December 31, 2021, the estimated amount of the retirement bonus obligation was €100,366.
As of December 31, 2022, considering the company’s data and the actuarial assumptions used, mainly a gross discount rate of 0.98%, the total retirement bonus liability amounts to €67,403.
The following assumptions were used:
| • | Change in compensation: 1.5% |
| • | Turn-over: average |
| • | Mortality: INSEE 2019 |
| • | Retirement age: 62 years |
See note 4.10
Donations
Donations of batches of bacteriophage cocktails and placebo products with an estimated value of €380 thousand have been pledged on the condition that a future clinical trial agreement is entered into.
| • | Related parties |
There were no significant transactions with related parties during the year that were not concluded under normal market conditions.
| CPA AUDIT | Page 23 |
STATUTORY AUDITORS REPORT ON THE ANNUAL FINANCIAL STATEMENTS FOR THE FISCAL YEAR
ENDED DECEMBER 31, 2022
69
PHERECYDES PHARMA
Statutory Auditors report on the
annual financial statements
(Fiscal year ended December 31, 2022)

Statutory Auditors report on the annual
financial statements
(Fiscal year ended December 31, 2022)
To the General Meeting of Shareholders
PHERECYDES PHARMA
22, Boulevard Benoni Goullin
44200 Nantes, France
Opinion
In compliance with the assignment entrusted to us by your articles of association, we have audited the annual financial statements of the company PHERECYDES PHARMA for the fiscal year ended December 31, 2022, as attached to this report.
We hereby certify that the annual financial statements are consistent with French accounting rules and principles and give a true and fair view of the operating results for the fiscal year just ended and of the financial position and assets and liabilities of the company at the end of that fiscal year.
Basis of opinion
Audit framework
We carried out our audit in accordance with the professional standards applicable in France. We believe that the information we have obtained is sufficient and appropriate to provide a basis for our opinion.
Our responsibilities under these standards are outlined in the “Responsibilities of the Statutory Auditor in relation to the audit of the annual financial statements” section of this report.
Independence
We have carried out our audit in accordance with the rules of independence provided for in the French Commercial Code and in the Code of Ethics for Statutory Auditors for the period from January 1, 2022 to the issue date of our report.
Observations
Without calling into question the opinion expressed above, we would like to draw your attention to note 2 to the annual financial statements, which sets out the information underlying the maintenance of operating principle.
PricewaterhouseCoopers Audit, S.A.S., 63, rue de Villiers 92208 Neuilly-sur-Seine, France
Cedex Telephone: +33 (0)1 56 57 58 59, www.pwc.fr
Accounting firm registered with the Paris – Ile de France association of chartered accountants. Statutory auditing firm, member of the Versailles and Centre regional associations of statutory auditors. Simplified joint stock company (Société par Actions Simplifiée) with capital of €2,510,460. Registered office: 63 rue de Villiers 92200 Neuilly-sur-Seine, France. Nanterre Trade and Companies Register 672 006 483. VAT no. FR 76 672 006 483. Siret 672 006 483 00362. APE Code 6920 Z. Offices: Bordeaux, Grenoble, Lille, Lyon, Marseille, Metz, Nantes, Neuilly-Sur-Seine, Nice, Poitiers, Rennes, Rouen, Strasbourg, Toulouse.
PHERECYDES PHARMA
Statutory Auditors report on the annual financial statements
for the fiscal year ended December 31, 2022 – Page 2
Justification of assessments
Pursuant to the provisions of Articles L.823-9 and R.823-7 of the French Commercial Code relating to the justification of our assessments, we hereby inform you that the most significant assessments that we have made, in our professional opinion, concerned the appropriateness of the accounting principles applied:
| • | As part of our assessment of the accounting principles applied by your company, we examined the methods used to capitalize development costs and ensured that the notes to the financial statements entitled “Research and development costs,” “Statement of fixed assets,” “Fixed assets” and “Depreciation, amortization and impairment” provide appropriate information. |
These assessments were made in the context of our audit of the annual financial statements taken as a whole and the formation of our opinion expressed above. We do not express our opinion on any individual item in these annual financial statements.
Specific verifications
In accordance with professional standards applicable in France, we also performed the specific verifications required by law and regulations.
Information provided in the management report and in other documents on the financial situation and the annual financial statements sent to the shareholders
We have no observations to make with regard to the fair presentation of the information provided in the management report of the Board of Directors and in the other documents addressed to shareholders relating to the financial position and the annual financial statements, nor do we have any observations to make with regard to the conformity of such documents with the annual financial statements.
We hereby attest to the fair presentation of the information relating to the payment periods mentioned in Article D.441-6 of the French Commercial Code, and to its conformity with the annual financial statements.
Report on corporate governance
We hereby certify that the report of the Board of Directors on corporate governance contains the information required by Article L.225-37-4 of the French Commercial Code.
Other information
Pursuant to the law, we have ensured that other information relating to the identity of shareholders or voting right holders have been communicated in the management report.
Responsibilities of management and those charged with corporate governance in relation to the annual financial statements
It is the responsibility of management to prepare the annual financial statements in such a way that they give a true and fair view in accordance with French accounting rules and principles, and to implement any internal control procedures it deems necessary to ensure that the annual financial statements are free from material misstatement, whether due to fraud or error.
PHERECYDES PHARMA
Statutory Auditors report on the annual financial statements
for the fiscal year ended December 31, 2022 – Page 3
When preparing the annual financial statements, it is the responsibility of management to evaluate the company’s ability to continue as a going concern, to disclose in such financial statements, where appropriate, the necessary information on the going concern and to apply the going concern accounting policy, unless there are plans for the company to be liquidated or to cease its activities.
The annual financial statements have been approved by the Board of Directors.
Responsibilities of the Statutory Auditor in relation to the audit of the annual financial statements
It is our responsibility to prepare a report on the annual financial statements. Our objective is to obtain reasonable assurance that the annual financial statements as a whole are free of material misstatement. Reasonable assurance is a high level of assurance but does not guarantee that an audit carried out in accordance with professional standards will systematically detect any material misstatement. Misstatements may arise from fraud or error and are considered material when one can reasonably expect that they may, individually or together, influence the economic decisions that users of the financial statements make based on them.
As specified in Article L.823-10-1 of the French Commercial Code, our role in certifying the financial statements is not to guarantee the viability or quality of the management of your company.
As part of an audit carried out in accordance with the professional standards applicable in France, the Statutory Auditor exercises their professional judgment throughout the audit. In addition:
| • | they identify and assess the risks of material misstatement in the annual financial statements, whether due to fraud or error; define and implement audit procedures to address such risks; and collect information that they believe is sufficient and appropriate to provide a basis for their opinion. The risk of not detecting material misstatement resulting from fraud is higher than the risk of not detecting material misstatement resulting from error, as fraud may involve collusion, falsification, deliberate omissions, misrepresentation or the circumvention of internal control; |
| • | they become aware of the internal control relevant to the audit in order to define audit procedures that are appropriate to the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the internal control; |
| • | they assess the appropriateness of the accounting methods used and the reasonableness of the accounting estimates made by management, as well as the related information provided in the annual financial statements; |
| • | they assess the appropriateness of management’s application of the going concern accounting policy and, based on the information collected, whether there is any material uncertainty related to events or circumstances likely to affect the company’s ability to continue as a going concern. This assessment is based on the information collected up to the date of the report, bearing in mind that circumstances or subsequent events may call into question the company’s ability to continue as a going concern. If they conclude that a material uncertainty exists, they draw the attention of the readers of their report to the information provided in the annual financial statements concerning this uncertainty or, if this information is not provided or is not relevant, they issue a qualified opinion or refuse to certify; |
PHERECYDES PHARMA
Statutory Auditors report on the annual financial statements
for the fiscal year ended December 31, 2022 – Page 4
| • | they assess the overall presentation of the annual financial statements and whether the annual financial statements reflect the underlying transactions and events in such a way that gives a true and fair view. |
Signed in Neuilly-sur-Seine, France, April 26, 2023
Statutory Auditor
PricewaterhouseCoopers Audit
Cédric Mazille
STATUTORY AUDITORS SPECIAL REPORT ON RELATED-PARTY AGREEMENTS
71
PHERECYDES PHARMA
Statutory Auditors special report on related-party agreements
(General Meeting of Shareholders to approve
the financial statements for the fiscal year ended
December 31, 2022)

Statutory Auditors special report on related- party agreements
(General Meeting of Shareholders to approve the financial statements for the fiscal year ended December 31, 2022)
To the General Meeting of Shareholders of the company,
PHERECYDES PHARMA
22, Boulevard Benoni Goullin
44200 Nantes, France
In our capacity as Statutory Auditors of your company, we hereby present our report on related-party agreements.
Our responsibility is to inform you, based on the information provided to us, of the characteristics, the essential terms and conditions and the reasons justifying the company’s interest in the agreements of which we have been informed or of which we may have become aware in the course of our assignment, without commenting on their usefulness or appropriateness or on the existence of other agreements. It is your responsibility, under the terms of Article R.225-58 of the French Commercial Code, to assess the benefits resulting from entering into these agreements prior to their approval.
In addition, it is our responsibility, where applicable, to provide you with the information required by Article R.225-58 of the French Commercial Code relating to the execution, during the past fiscal year, of agreements already approved by the General Meeting of Shareholders.
We have performed the procedures we’ve deemed necessary to comply with the professional standards issued by the French Institute of Statutory Auditors relating to this assignment.
AGREEMENTS SUBMITTED TO THE APPROVAL OF THE GENERAL MEETING OF SHAREHOLDERS
We hereby inform you that we have not been advised of any agreements authorized and entered into during the past fiscal year that should be submitted to the approval of the General Meeting of Shareholders pursuant to the provisions of Article L.225-86 of the French Commercial Code.
PricewaterhouseCoopers Audit, S.A.S., 63, rue de Villiers 92208 Neuilly-sur-Seine, France Cedex Telephone: +33 (0)1 56 57 58 59, www.pwc.fr
Accounting firm registered with the Paris – Ile de France association of chartered accountants. Statutory auditing firm, member of the Versailles and Centre regional associations of statutory auditors. Simplified joint stock company (Société par Actions Simplifiée) with capital of €2,510,460. Registered office: 63 rue de Villiers 92200 Neuilly-sur-Seine, France. Nanterre Trade and Companies Register 672 006 483. VAT no. FR 76 672 006 483. Siret 672 006 483 00362. APE Code 6920 Z. Offices: Bordeaux, Grenoble, Lille, Lyon, Marseille, Metz, Nantes, Neuilly-Sur-Seine, Nice, Poitiers, Rennes, Rouen, Strasbourg, Toulouse.
PHERECYDES PHARMA
Statutory Auditors special report on related-party agreements
(General Meeting of Shareholders to approve the financial statements for the fiscal year ended December 31, 2022) – Page 2
AGREEMENTS ALREADY APPROVED BY THE GENERAL MEETING OF SHAREHOLDERS
We hereby inform you that we have not been advised of any agreement previously approved by the General Meeting of Shareholders whose execution has continued during the past fiscal year.
Signed in Neuilly-sur-Seine, France, April 26, 2023
Statutory Auditor
PricewaterhouseCoopers Audit

Cédric Mazille