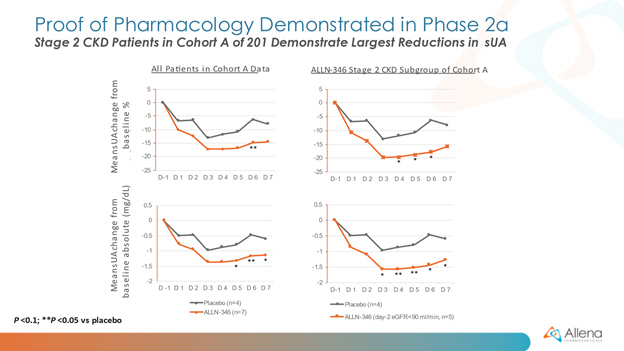
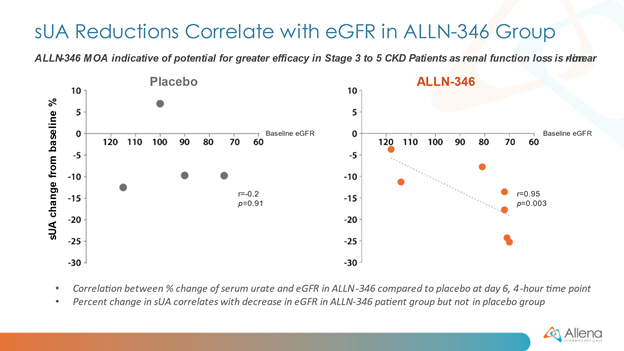
David S. Goldfarb, M.D., Professor of Medicine and Physiology and Clinical Chief of Nephrology at NYU Langone Medical Center, added, “Hyperuricemia and the resulting clinical manifestations of gout represent a progressive and challenging metabolic derangement for CKD patients, especially as kidney function decreases. ALLN-346 acts by a novel gut-restricted mechanism, which is conceptually similar to currently available agents used to treat excesses of phosphorus and potassium via the GI tract in CKD patients. I am encouraged by this initial data in hyperuricemic patients and look forward to seeing additional data from the Phase 2a program in patients with more advanced CKD, since these patients are especially susceptible to a higher risk of morbidity and complications associated with gout and worsening CKD.”
Webinar Today to Review ALLN-346 Study 201 Data with Key Opinion Leaders
Allena will be hosting a KOL webinar at 8:00 am ET today to review today’s ALLN-346 data announcement with Key Opinion Leaders, Robert Terkeltaub, M.D. of the University of California, San Diego and David S. Goldfarb, M.D. of NYU Langone Medical Center.
A listen-in only webcast of the conference call with corresponding slides can be accessed at this link and also via Allena’s website, within the Investors & News/Events & Presentations section. A replay of the webcast can also be accessed at this same link.
Reloxaliase Program Update
Reloxaliase is an investigational, first-in-class, non-absorbed, orally administered enzyme for the treatment of enteric hyperoxaluria (EH), a metabolic disorder characterized by high levels of urinary oxalate (UOx) that can lead to kidney stone disease and chronic kidney disease, and for which there are no currently approved therapies. The Company is currently studying reloxaliase in the pivotal Phase 3 URIROX-2 trial, a multicenter, global, randomized, double-blind, placebo-controlled study designed to evaluate the safety and efficacy of reloxaliase in approximately 200 patients with enteric hyperoxaluria over a minimum treatment period of two years. Previously reported results of the URIROX-1 study are expected to be published during the first half of 2022.
URIROX-2 is being conducted at 122 sites in 16 countries. Through year-end 2021, 78 subjects have been enrolled and will comprise the cohort for the first planned interim analysis, targeted for late Q1 2022. Since initiation of the trial in Q4 2018, approximately 530 patient-months of biochemical, clinical, radiographic, and safety data have been accumulated for reloxaliase and placebo-treated patients, which represents the most comprehensive data set from a randomized clinical trial of EH patients.
As a result of the ongoing pandemic, the Company’s cash constraints and other corporate considerations, the Company is currently evaluating potential modifications to the URIROX-2 trial design, including potentially reducing the sample size and/or shortening the patient treatment period. The Company plans to engage with the FDA during Q1 2022 to consider such potential modifications and the impact they may have, if any, upon the status of URIROX-2 as a Phase 3 registrational trial and the Company’s accelerated approval strategy.