
Corporate Presentation Cantor Fitzgerald Global Healthcare Conference October 1, 2018

Forward-Looking Statements This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Words such as "may," "will," "expect," "plan," "anticipate" and similar expressions (as well as other words or expressions referencing future events or circumstances) are intended to identify forward-looking statements. Forward-looking statements contained in this presentation include statements about the progress, timing, clinical development and scope of clinical trials and the reporting of clinical data for the Company’s product candidates; the potential clinical benefit of the Company’s product candidates; and the timing and outcome of discussions with regulatory authorities. Each of these forward-looking statements involves risks and uncertainties. These statements are based on the Company’s current expectations and projections made by management and are not guarantees of future performance. Therefore, actual events, outcomes and results may differ materially from what is expressed or forecast in such forward-looking statements. Factors that may cause actual results to differ materially from these forward-looking statements are discussed in the Company’s filings with the U.S. Securities and Exchange Commission, including the "Risk Factors" sections contained therein. Except as otherwise required under federal securities laws, we do not have any intention or obligation to update or revise any forward-looking statements, whether as a result of new information, future events, changes in assumptions or otherwise. .

Ovid at a Glance: Goal to Build a Leading Company in Neurology Focus on serious unmet needs: >1 million patients and families living with devastating rare neurological disorders Broad pipeline: Multiple opportunities in rare conditions; novel compounds and mechanisms; first potential medicine to treat Angelman syndrome World-class partnership: Takeda Pharmaceuticals for OV935 in rare epilepsies 2H 2018 Events: Q3: OV101, positive Phase 2 STARS topline data Q4: OV101, final results for CGI-I, sleep and motor at AACAP Q4: OV935, topline data Phase 1b/2a in DEE multiple Q4: Other studies underway and being initiated
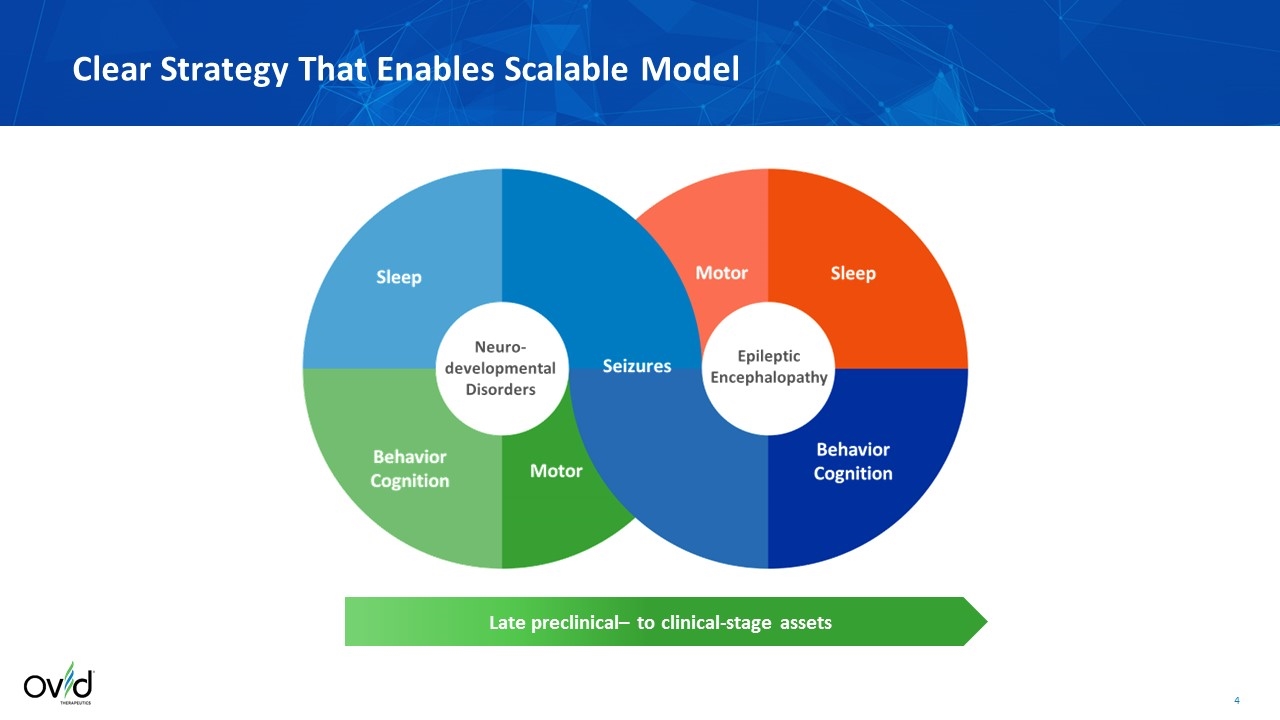
Late preclinical– to clinical-stage assets Clear Strategy That Enables Scalable Model
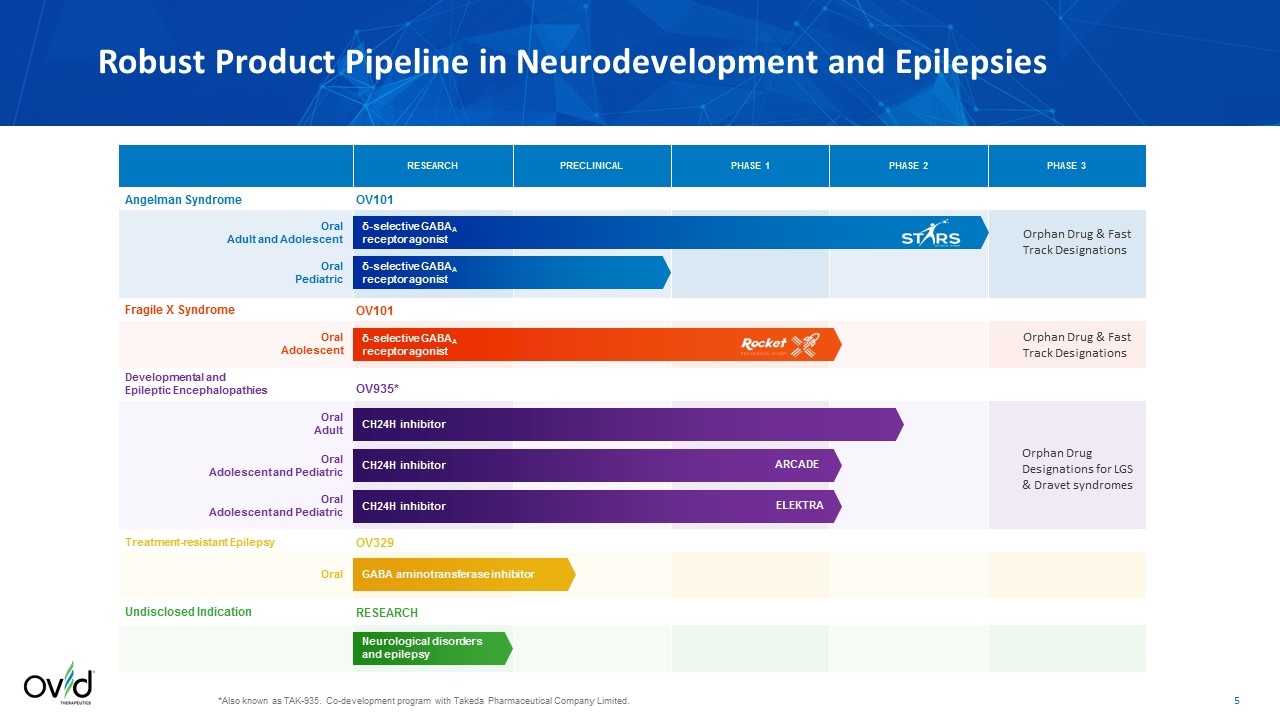
Robust Product Pipeline in Neurodevelopment and Epilepsies *Also known as TAK-935. Co-development program with Takeda Pharmaceutical Company Limited. δ-selective GABAA receptor agonist Angelman Syndrome RESEARCH PRECLINICAL PHASE 1 PHASE 2 PHASE 3 Fragile X Syndrome δ-selective GABAA receptor agonist δ-selective GABAA receptor agonist Oral Adolescent Oral Pediatric Oral Adult and Adolescent OV101 OV101 Developmental and Epileptic Encephalopathies CH24H inhibitor Oral Adult OV935* Undisclosed Indication Neurological disorders and epilepsy RESEARCH OV329 GABA aminotransferase inhibitor Oral Treatment-resistant Epilepsy CH24H inhibitor Oral Adolescent and Pediatric CH24H inhibitor Oral Adolescent and Pediatric ARCADE ELEKTRA Orphan Drug & Fast Track Designations Orphan Drug & Fast Track Designations Orphan Drug Designations for LGS & Dravet syndromes

OV101 Potential First-in-Class Therapeutic for Angelman & Fragile X Syndromes

OV101: Significant Opportunity in Two Serious Rare Disorders Angelman syndrome Fragile X syndrome Well defined Genetic Disorders No approved therapies in US, EU, or ROW; treatment is limited to supportive care Lifelong disorders that present early in childhood Disruption of behavior, anxiety and cognitive functions Most common inherited form of intellectual disability Prevalence**: ~1/3,600-4,000 males ~1/4,000-6,000 females Global disruption in behavior, motor, sleep and cognitive functions Prevalence in general population*: 1 in 12,000 to 20,000 people . Sources: *NORD, NIH website, Genetics Home Reference and NCBI . ** National Fragile X Foundation
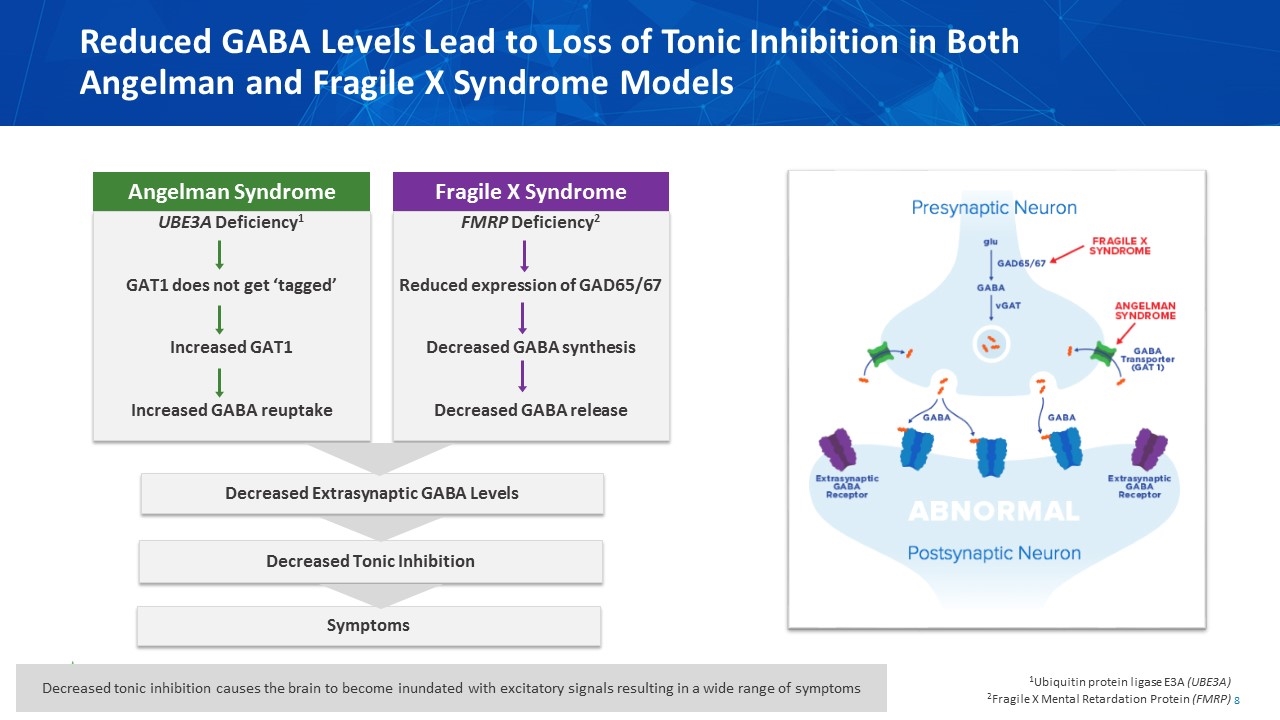
Reduced GABA Levels Lead to Loss of Tonic Inhibition in Both Angelman and Fragile X Syndrome Models FMRP Deficiency2 Reduced expression of GAD65/67 Decreased GABA synthesis Decreased GABA release UBE3A Deficiency1 GAT1 does not get ‘tagged’ Increased GAT1 Increased GABA reuptake Decreased Extrasynaptic GABA Levels Decreased Tonic Inhibition Symptoms 1Ubiquitin protein ligase E3A (UBE3A) 2Fragile X Mental Retardation Protein (FMRP) Fragile X Syndrome Angelman Syndrome Decreased tonic inhibition causes the brain to become inundated with excitatory signals resulting in a wide range of symptoms
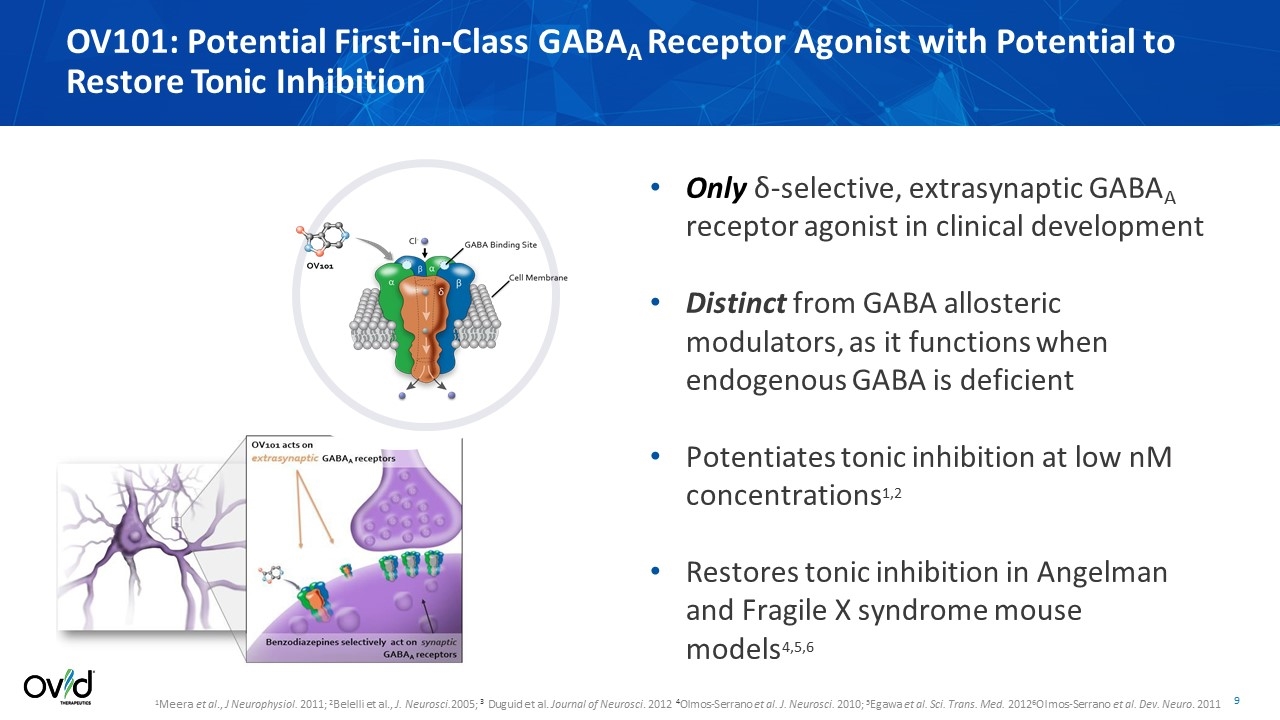
1Meera et al., J Neurophysiol. 2011; 2Belelli et al., J. Neurosci.2005; 3 Duguid et al. Journal of Neurosci. 2012 4Olmos-Serrano et al. J. Neurosci. 2010; 5Egawa et al. Sci. Trans. Med. 20126Olmos-Serrano et al. Dev. Neuro. 2011 Only δ-selective, extrasynaptic GABAA receptor agonist in clinical development Distinct from GABA allosteric modulators, as it functions when endogenous GABA is deficient Potentiates tonic inhibition at low nM concentrations1,2 Restores tonic inhibition in Angelman and Fragile X syndrome mouse models4,5,6 OV101: Potential First-in-Class GABAA Receptor Agonist with Potential to Restore Tonic Inhibition

OV101 in Angelman: Phase 2 Randomized, Double Blind, Placebo Controlled Trial Orphan Drug and Fast Track designations First-ever double-blind, placebo-controlled clinical trial in Angelman syndrome . Source: Ovid press release August 6, 2018. STARS Topline Phase 2 Data. Primary Endpoint: Safety and tolerability of OV101 vs. placebo from baseline to week 12 Exploratory Endpoints: Efficacy measures of OV101 vs. placebo for overall clinical change from baseline to week 12 Global function, behavior, sleep and motor EEG, metabolomics and quality of life Trial Design: n=88 (randomized) Adults and adolescents (age 13-49 years) 12 sites in US, 1 site in Israel Evaluated QD (15mg) and BID (15mg evening; 10mg morning) doses

STARS Topline Data: Key Take-aways as of August 6, 2018 . Source: Ovid press release August 6, 2018. STARS Topline Phase 2 Data. First positive clinical data for Angelman syndrome in >50 years Younger patients receiving single daily dose showed a trend of greater response Data consistent with hypothesis that younger patients have greater treatment effect compared to older ages Effect appears to persist over time from 6 to 12 weeks Prespecified Analysis Other Takeaways Primary Endpoint: Achieved primary endpoint of safety and tolerability; Overall favorable risk profile and well tolerated First Prespecified Endpoint: Global Function: statistically significant improvement demonstrated in CGI-I in 15mg QD and combined OV101 treatment groups Other Prespecified Endpoints: No statistically significant improvements as measured using ABC, sleep diary (parent/caregiver), ADAMS, mPOMA-G

Presentation of STARS Phase 2 Results American Academy of Child & Adolescent Psychiatry (AACAP) Poster Presentation Accepted – Thursday, Oct 25, 2018: Title: Topline Results from a Phase 2 Adult and Adolescent Angelman Syndrome Clinical Trial: A Randomized, Double-Blind, Safety and Efficacy Study of Gaboxadol (OV101) Plan to host investor conference call and webcast during AACAP Exploratory and Post-Hoc Analysis: Expect to provide final results on CGI-I, sleep and motor
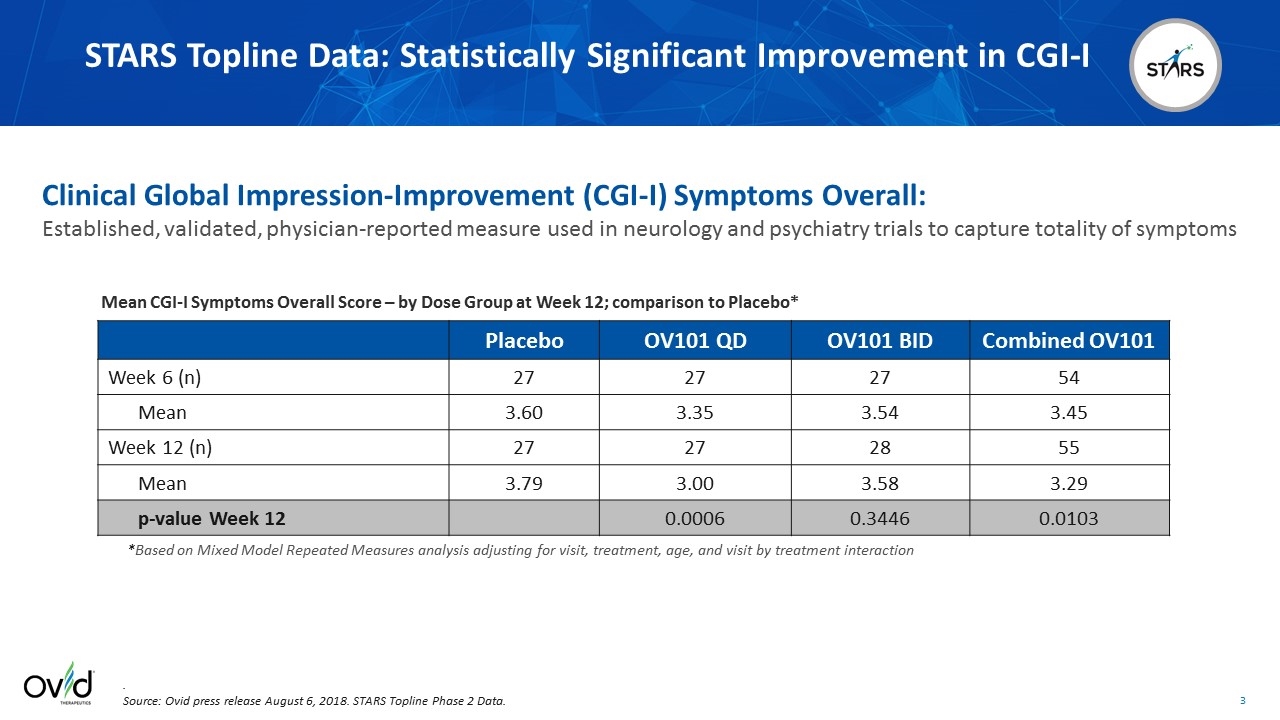
Placebo OV101 QD OV101 BID Combined OV101 Week 6 (n) 27 27 27 54 Mean 3.60 3.35 3.54 3.45 Week 12 (n) 27 27 28 55 Mean 3.79 3.00 3.58 3.29 p-value Week 12 0.0006 0.3446 0.0103 *Based on Mixed Model Repeated Measures analysis adjusting for visit, treatment, age, and visit by treatment interaction Mean CGI-I Symptoms Overall Score – by Dose Group at Week 12; comparison to Placebo* STARS Topline Data: Statistically Significant Improvement in CGI-I . Source: Ovid press release August 6, 2018. STARS Topline Phase 2 Data. Clinical Global Impression-Improvement (CGI-I) Symptoms Overall: Established, validated, physician-reported measure used in neurology and psychiatry trials to capture totality of symptoms
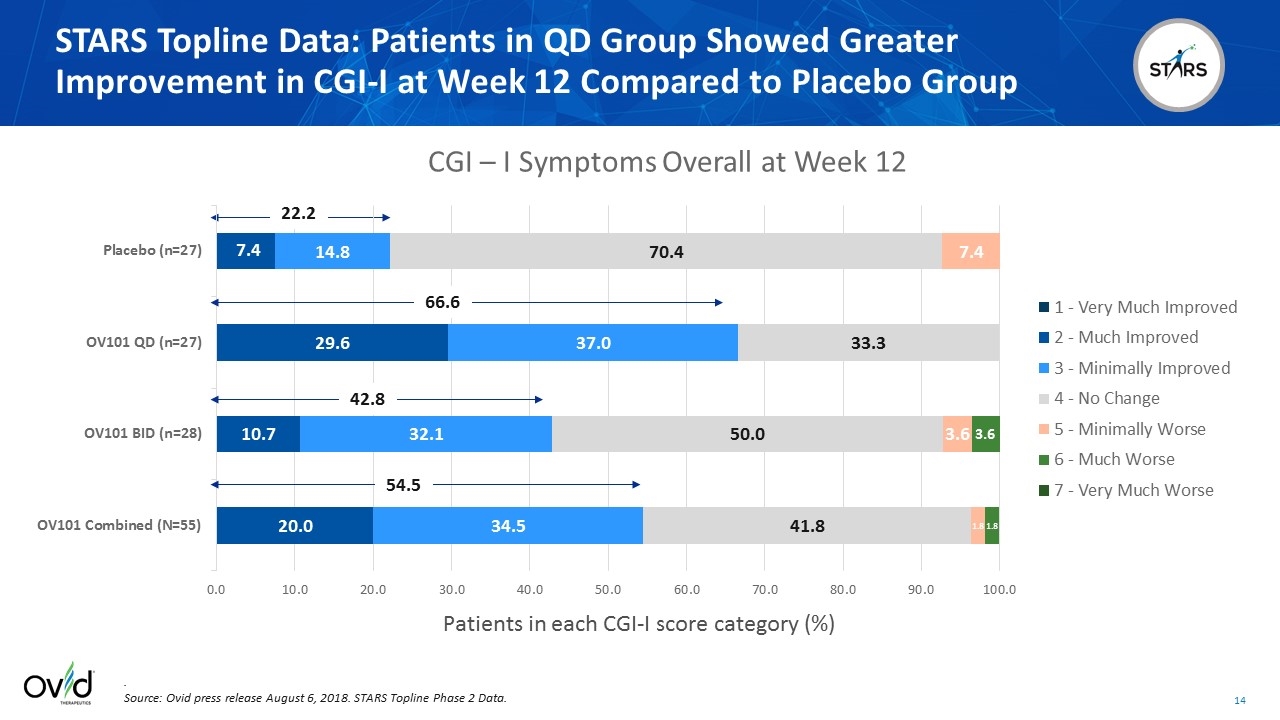
Patients in each CGI-I score category (%) STARS Topline Data: Patients in QD Group Showed Greater Improvement in CGI-I at Week 12 Compared to Placebo Group . Source: Ovid press release August 6, 2018. STARS Topline Phase 2 Data. 22.2 66.6 42.8

OV101’s Development in Angelman Syndrome Medical Conference Final results for CGI, sleep and motor at AACAP Oct 2018 Additional Trials Plan to initiate Angelman syndrome open-label extension study (ELARA) in Q4 2018 Regulatory Dialogue Expect to meet with the FDA by the end of the year; will provide update on next steps for OV101’s development after agency’s official feedback Next steps

OV101 in Fragile X Syndrome: Phase 2 Clinical Trials Underway Orphan Drug and Fast Track designations Primary Objective: Assess safety and tolerability of OV101 over 12 weeks of treatment across different daily dosing regimens Patient Population: Adult and adolescent males with diagnosis of FXS (n=30) Ages: 13-22 years Primary objective: Non-drug study to assess suitability of scales to measure behavior, sleep, and functioning in individuals with Fragile X syndrome
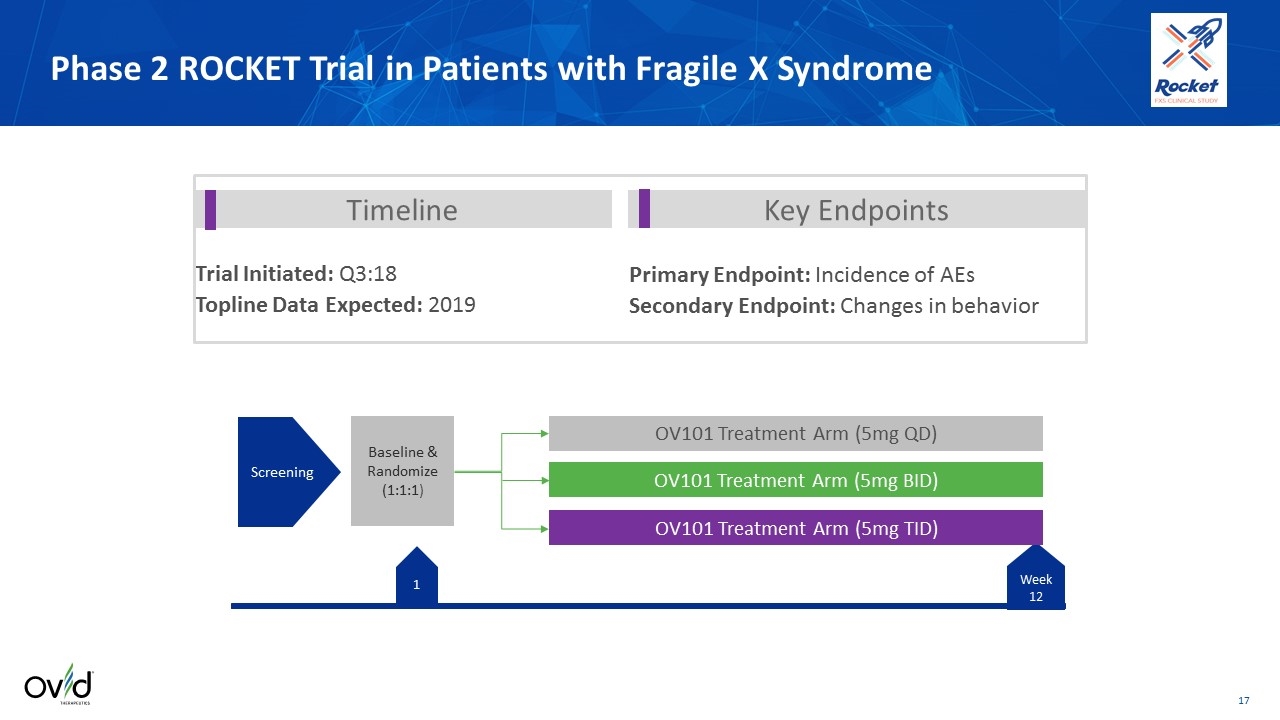
1 -21 OV101 Treatment Arm (5mg QD) OV101 Treatment Arm (5mg TID) OV101 Treatment Arm (5mg BID) Screening Baseline & Randomize (1:1:1) Phase 2 ROCKET Trial in Patients with Fragile X Syndrome Week 12 Timeline Key Endpoints Primary Endpoint: Incidence of AEs Secondary Endpoint: Changes in behavior Trial Initiated: Q3:18 Topline Data Expected: 2019

OV935 New Approach to Potentially Treat Rare Developmental and Epileptic Encephalopathies
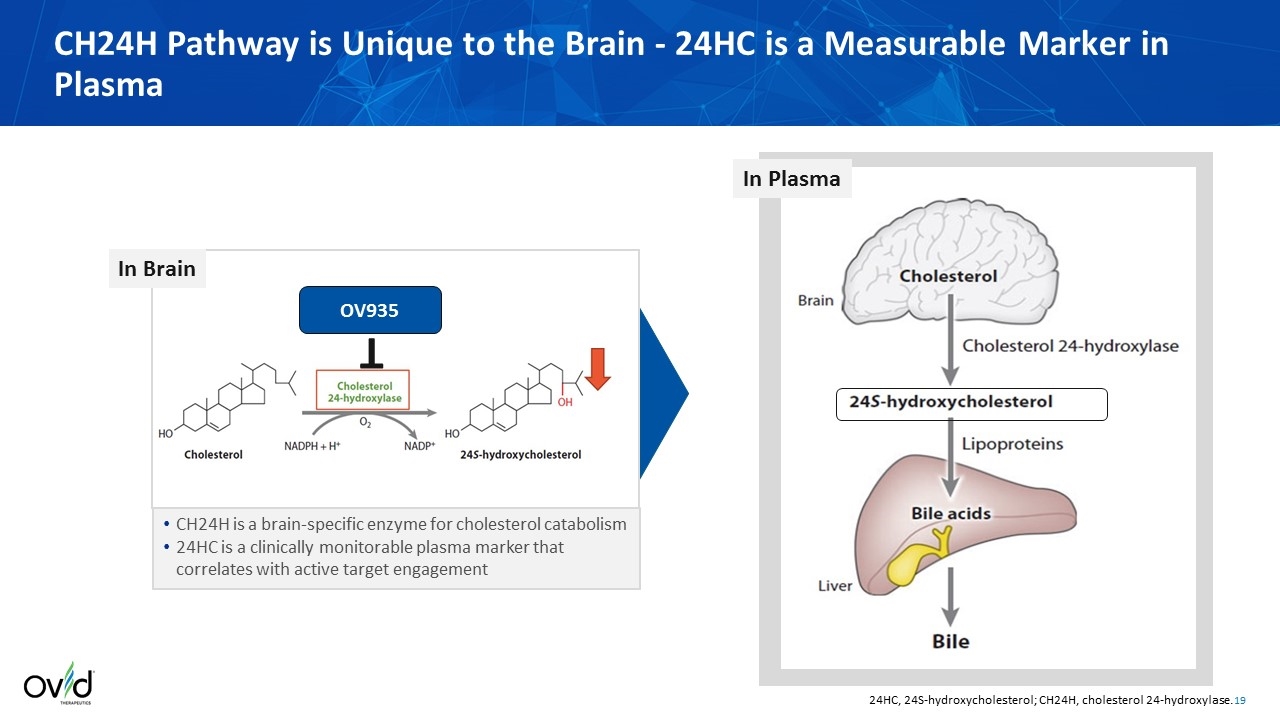
CH24H is a brain-specific enzyme for cholesterol catabolism 24HC is a clinically monitorable plasma marker that correlates with active target engagement CH24H Pathway is Unique to the Brain - 24HC is a Measurable Marker in Plasma OV935 T 24HC, 24S-hydroxycholesterol; CH24H, cholesterol 24‑hydroxylase. In Plasma In Brain
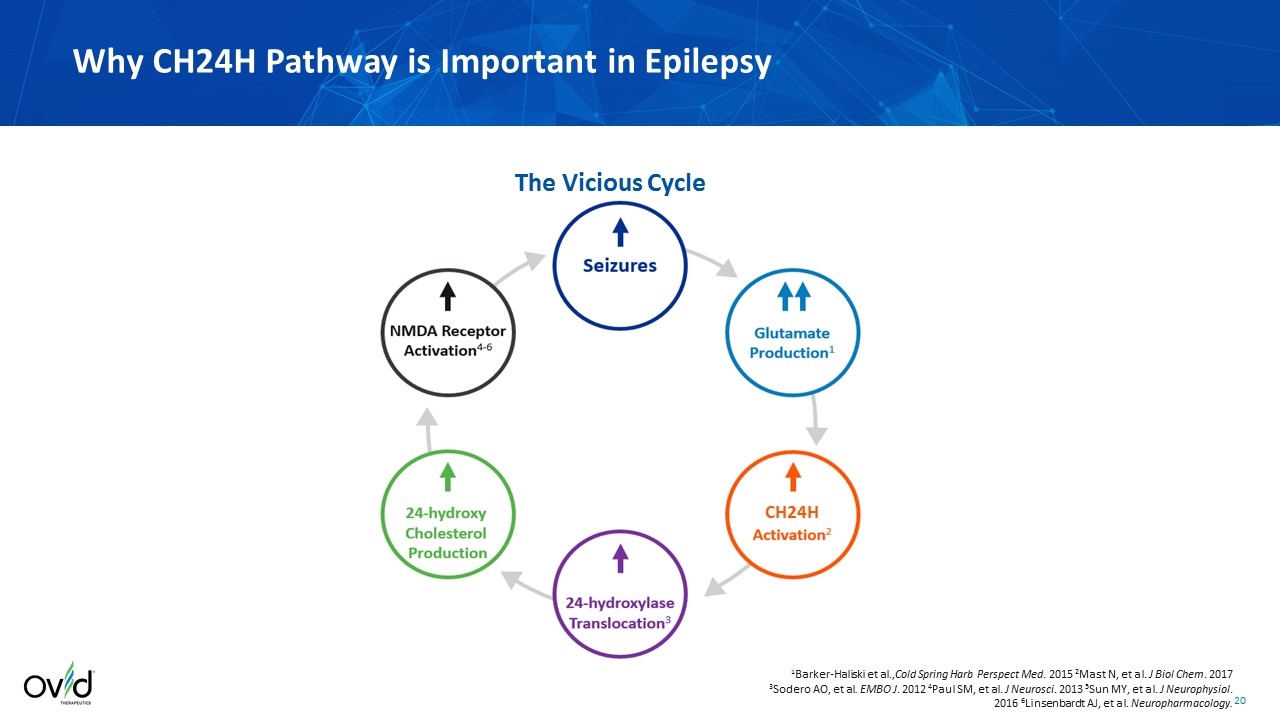
Why CH24H Pathway is Important in Epilepsy 1Barker-Haliski et al.,Cold Spring Harb Perspect Med. 2015 2Mast N, et al. J Biol Chem. 2017 3Sodero AO, et al. EMBO J. 2012 4Paul SM, et al. J Neurosci. 2013 5Sun MY, et al. J Neurophysiol. 2016 6Linsenbardt AJ, et al. Neuropharmacology. The Vicious Cycle CH24H
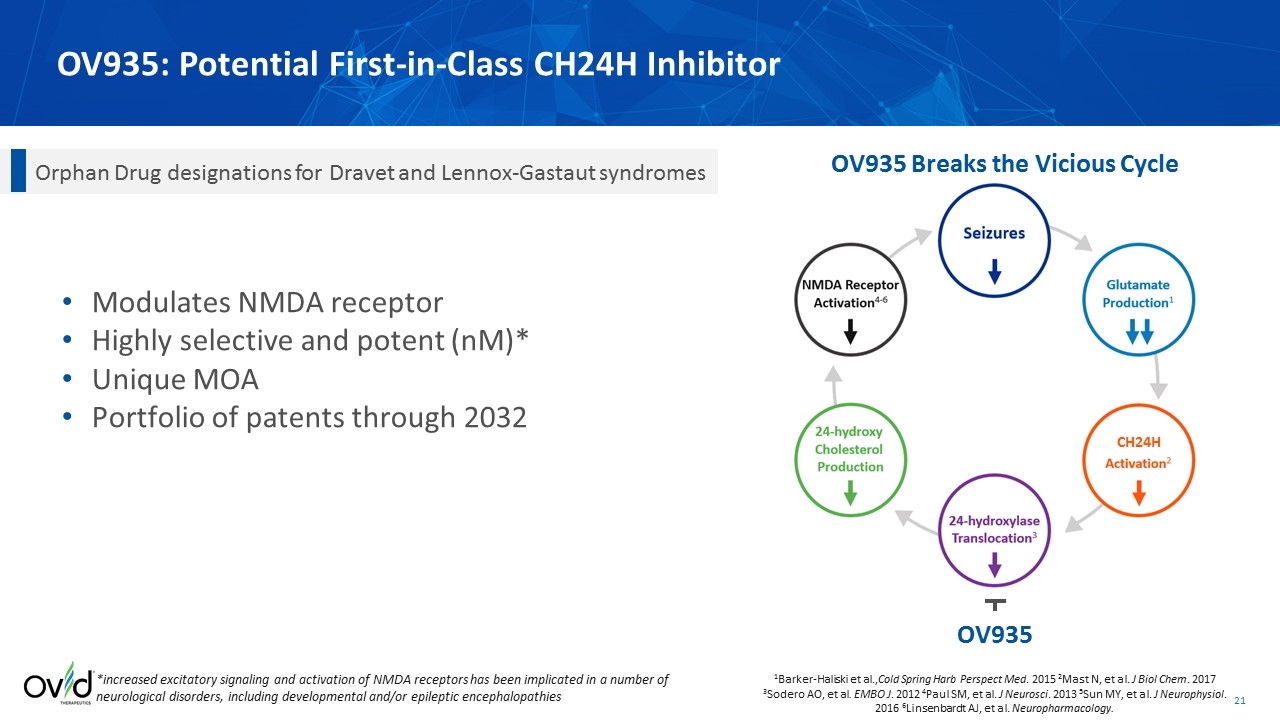
OV935: Potential First-in-Class CH24H Inhibitor OV935 1Barker-Haliski et al.,Cold Spring Harb Perspect Med. 2015 2Mast N, et al. J Biol Chem. 2017 3Sodero AO, et al. EMBO J. 2012 4Paul SM, et al. J Neurosci. 2013 5Sun MY, et al. J Neurophysiol. 2016 6Linsenbardt AJ, et al. Neuropharmacology. OV935 Breaks the Vicious Cycle Modulates NMDA receptor Highly selective and potent (nM)* Unique MOA Portfolio of patents through 2032 Orphan Drug designations for Dravet and Lennox-Gastaut syndromes *increased excitatory signaling and activation of NMDA receptors has been implicated in a number of neurological disorders, including developmental and/or epileptic encephalopathies CH24H

Refractory seizures do not respond to medicines that exert anti-seizure effect through same target or MOA as marketed AEDs Approximately 100,000- 200,000 US Population with Rare Seizure Disorders* Illustrative purposes only 1 out of 3 PATIENTS Small number achieve seizure freedom with addition of 1 or more AEDs 1 out of 2 PATIENTS ALL EPILEPSY PATIENTS Current marketed AEDs act through four main mechanisms: 1. Modulation of voltage-gated ion channels 2. Enhancement of GABA-mediated inhibition 3. Interactions with elements of the synaptic release machinery 4. Blockage of ionotropic glutamate receptors Seizure free with one AED Refractory seizures despite available medications Novel Mechanism of Actions Needed to Treat Refractory Seizures Source: *Orphanet

Focus of Broad OV935 Development Program: Treatment Refractory Seizures Developmental and Epileptic Encephalopathies (DEE): Significant unmet needs with few available therapies Dravet 80% have mutation in SCN1A gene US prevalence: ~7000-9,000 Lennox-Gastaut Some patients have SCN2A mutations <10% controlled by existing medications CDKL5 (CDD) Mutation of cyclin-dependent kinase-like 5 gene on X chromosome US prevalence: increasing with genetic testing Few treatmen t options US prevalence: ~30,000 No approved medicines Dup15q Caused by duplications of chromosome 15q11.2-q131.1 US incidence at birth: ~1:30,000 No approved medicines
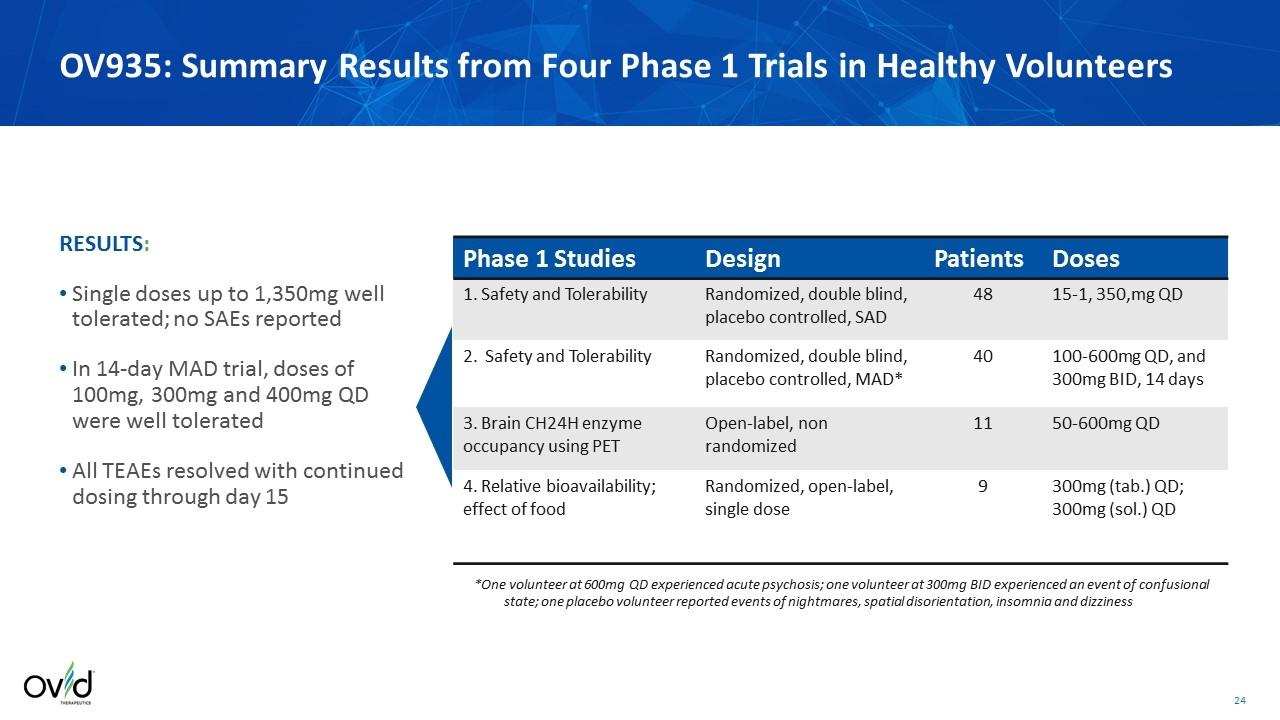
Phase 1 Studies Design Patients Doses 1. Safety and Tolerability Randomized, double blind, placebo controlled, SAD 48 15-1, 350,mg QD 2. Safety and Tolerability Randomized, double blind, placebo controlled, MAD* 40 100-600mg QD, and 300mg BID, 14 days 3. Brain CH24H enzyme occupancy using PET Open-label, non randomized 11 50-600mg QD 4. Relative bioavailability; effect of food Randomized, open-label, single dose 9 300mg (tab.) QD; 300mg (sol.) QD RESULTS: Single doses up to 1,350mg well tolerated; no SAEs reported In 14-day MAD trial, doses of 100mg, 300mg and 400mg QD were well tolerated All TEAEs resolved with continued dosing through day 15 *One volunteer at 600mg QD experienced acute psychosis; one volunteer at 300mg BID experienced an event of confusional state; one placebo volunteer reported events of nightmares, spatial disorientation, insomnia and dizziness OV935: Summary Results from Four Phase 1 Trials in Healthy Volunteers
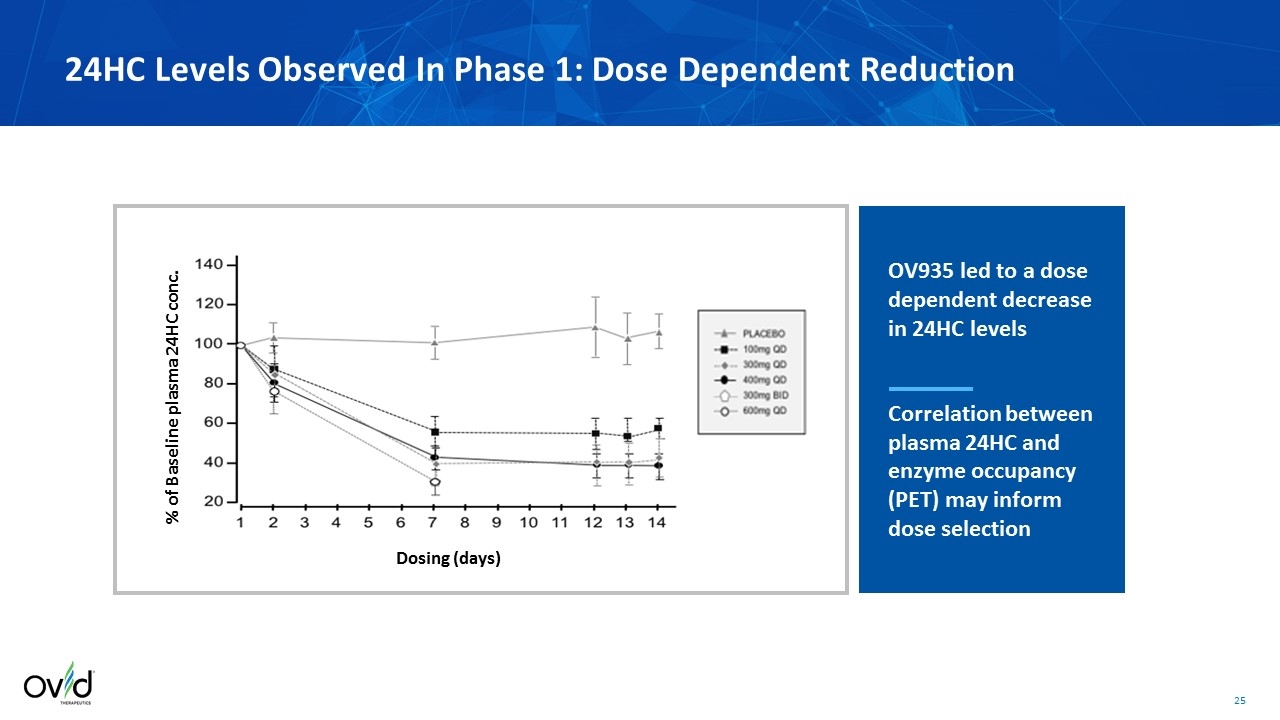
24HC Levels Observed In Phase 1: Dose Dependent Reduction Dosing (days) % of Baseline plasma 24HC conc. OV935 led to a dose dependent decrease in 24HC levels Correlation between plasma 24HC and enzyme occupancy (PET) may inform dose selection
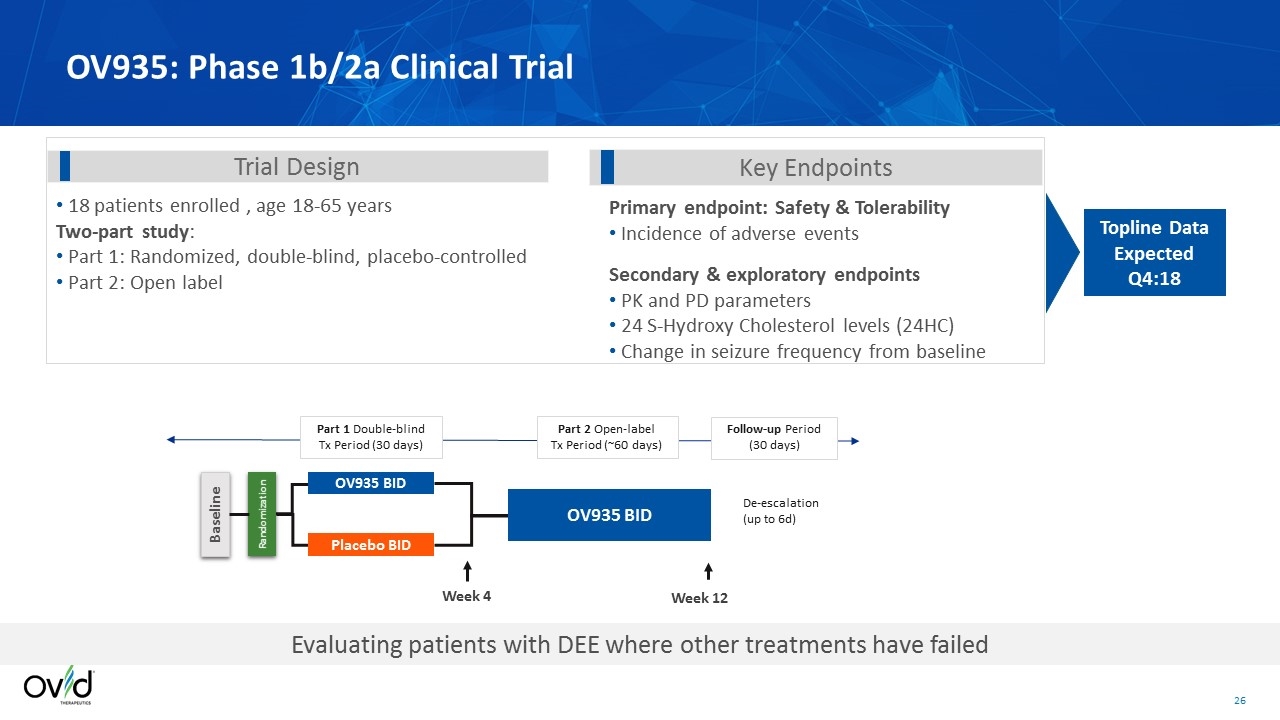
OV935: Phase 1b/2a Clinical Trial Week 12 Week 4 Randomization OV935 BID Placebo BID Baseline OV935 BID Topline Data Expected Q4:18 18 patients enrolled , age 18-65 years Two-part study: Part 1: Randomized, double-blind, placebo-controlled Part 2: Open label Primary endpoint: Safety & Tolerability Incidence of adverse events Secondary & exploratory endpoints PK and PD parameters 24 S-Hydroxy Cholesterol levels (24HC) Change in seizure frequency from baseline Evaluating patients with DEE where other treatments have failed Trial Design Key Endpoints Part 1 Double-blind Tx Period (30 days) Part 2 Open-label Tx Period (~60 days) Follow-up Period (30 days) De-escalation (up to 6d)

OV935 Phase 1b/2a Trial: Study Population Eligibility Criteria Treatment resistant seizures Failed at least two prior treatment regimens Formal IQ <70 Epilepsies not caused by brain trauma Currently taking 1-4 AEDS General Characteristics: All-comers Patients enrolled not well controlled on current seizure medications/regimen Diverse group of seizure types Small study (number and duration) As of April 1, 2018 AAN poster: (n=15 of 18)*: Baseline seizure frequency (median 31.11 per month) Young (mean age 29.6) *Source: M. Asgharnejad, AAN 2018 Poster Presentation:

OV935: Rare Developmental and Epileptic Encephalopathies (DEE) ARCADE ELEKTRA ENDYMION Multi-center, long-term open label extension (OLE) study of patients with DEE who participated in a previous OV935 clinical study Phase 2, multicenter, open-label, pilot study in approximately 30 pediatric patients, aged 2 to 17 years old, with CDKL5 deficiency disorder (CDD) or Duplication 15q (Dup15q) syndrome Phase 2, multicenter, randomized, double-blind, placebo controlled, study in 126 pediatric patients, aged 2 to 17 years old, with Dravet syndrome or Lennox-Gastaut syndrome. First patient randomized in September Trials initiated Q3 2018 Broad OV935 Development Program

Financial Highlights 2017: ~ $75 million IPO 2015: ~$75 million Series B Financing 2014: ~$5 million Series A Financing Cash Position as of June 30, 2018: $62.6 million in cash, cash equivalents and short-term investments ~24.6 million shares of common stock outstanding ~$155M raised in equity financings to date with broad institutional investor support

*Also known as TAK-935. Co-development program with Takeda Pharmaceutical Company Limited pursuant to a license and collaboration agreement ** Preclinical data suggest potential for use of OV329 in a variety of treatment-resistant epilepsies 2018 Key Pipeline Milestones: A Year of Execution and Data Readouts Angelman Syndrome OV101 First Half Second Half Fragile X Syndrome OV101 DEE & Rare Epilepsies OV935* Treatment-resistant Epilepsies OV329 Program Completed enrollment in Phase 2 STARS trial Five poster presentations at AAN, including patient baseline demographics for STARS FDA Fast Track Designation Economic and clinical burden of Fragile X syndrome presentation at AAN Six posters at AAN, including patient baseline demographics of Phase 1b/2a trial New preclinical data - oral presentation at EILAT First presentation of data at EILAT XIV** Phase 2 STARS top-line data Present STARS results at AACAP Initiate ELARA open-label extension study in Q4:18 Initiated Phase 2 ROCKET trial in adolescents and young adults Initiated accompanying SKY ROCKET non-drug study in Q3:18 Initiated ENDYMION open-label extension Initiated studies in younger patients with DEE (ELEKTRA) and CDD and Dup15 syndromes (ARCADE) (Q3:18) Completed enrollment in Phase 1b/2a adult trial Top-line Phase 1b/2a data (Q4:18) Presentation at additional medical conference

Ovid Therapeutics Executing on a clear vision, strategy and plan Driving development of potentially first medicine to treat Angelman syndrome Major opportunities in several rare disorders of the brain Robust clinical pipeline with two different molecules Programs based on novel mechanisms of action We call this BoldMedicineTM
