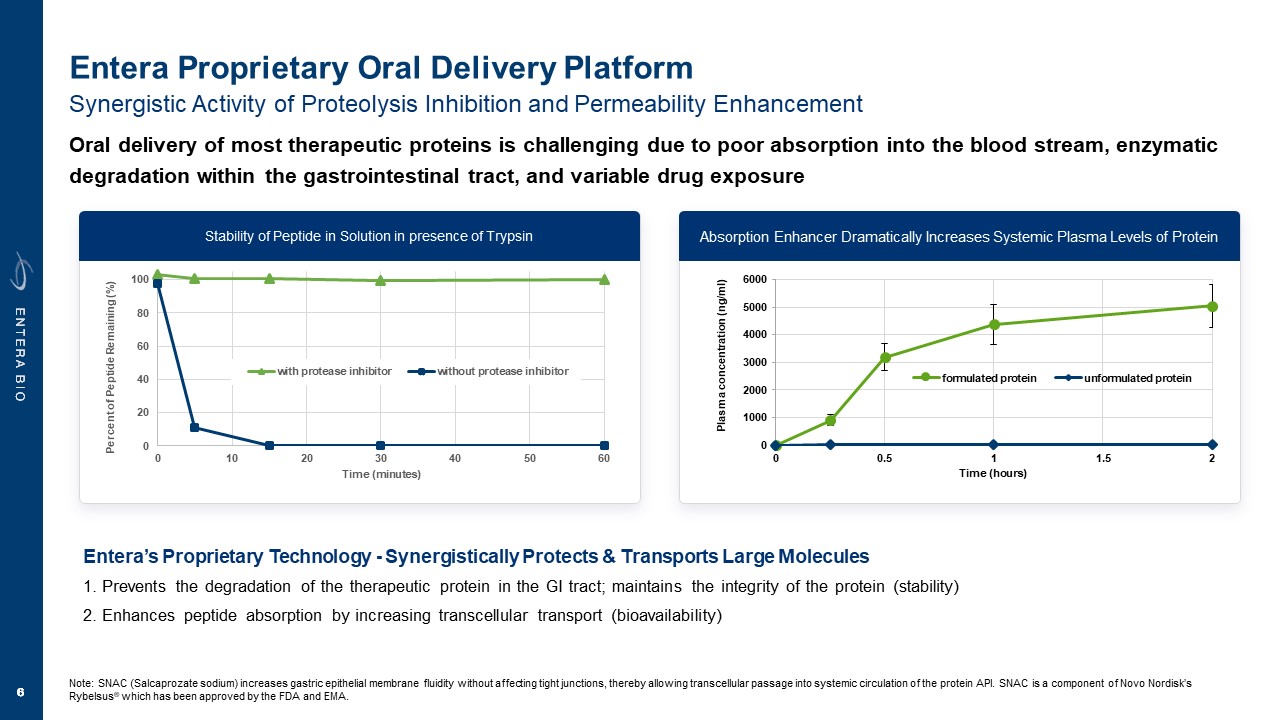
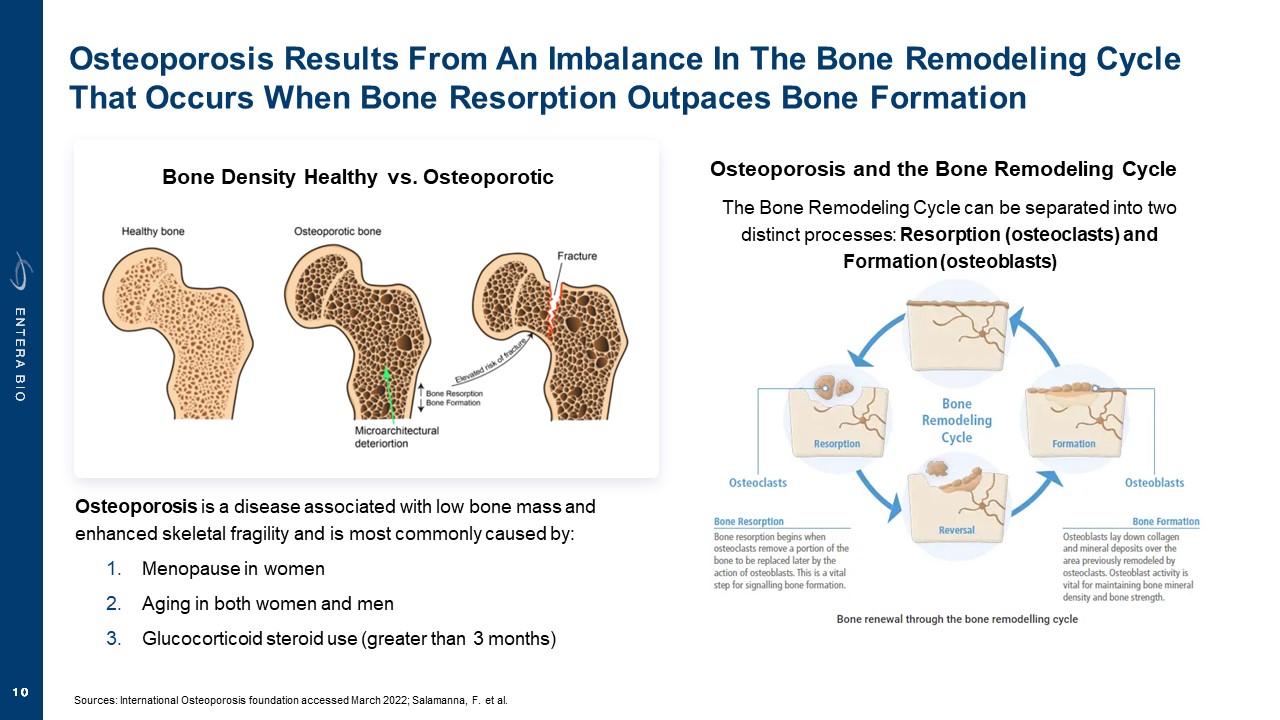
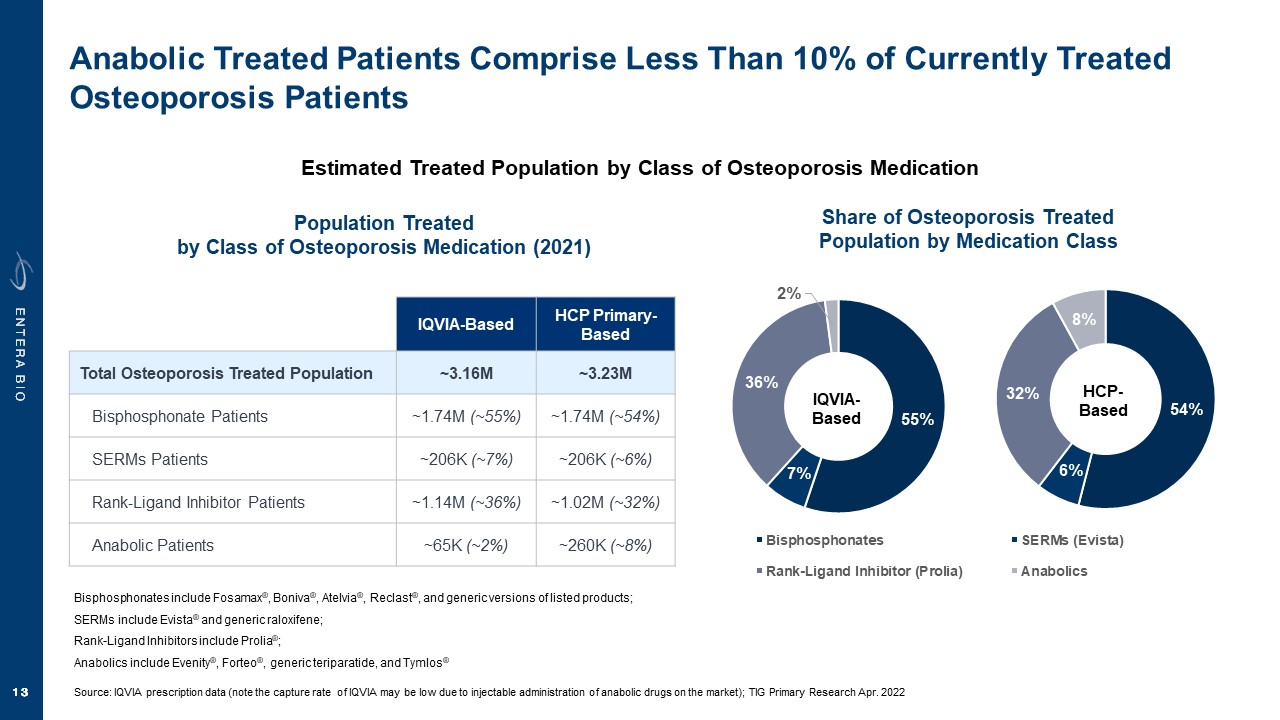
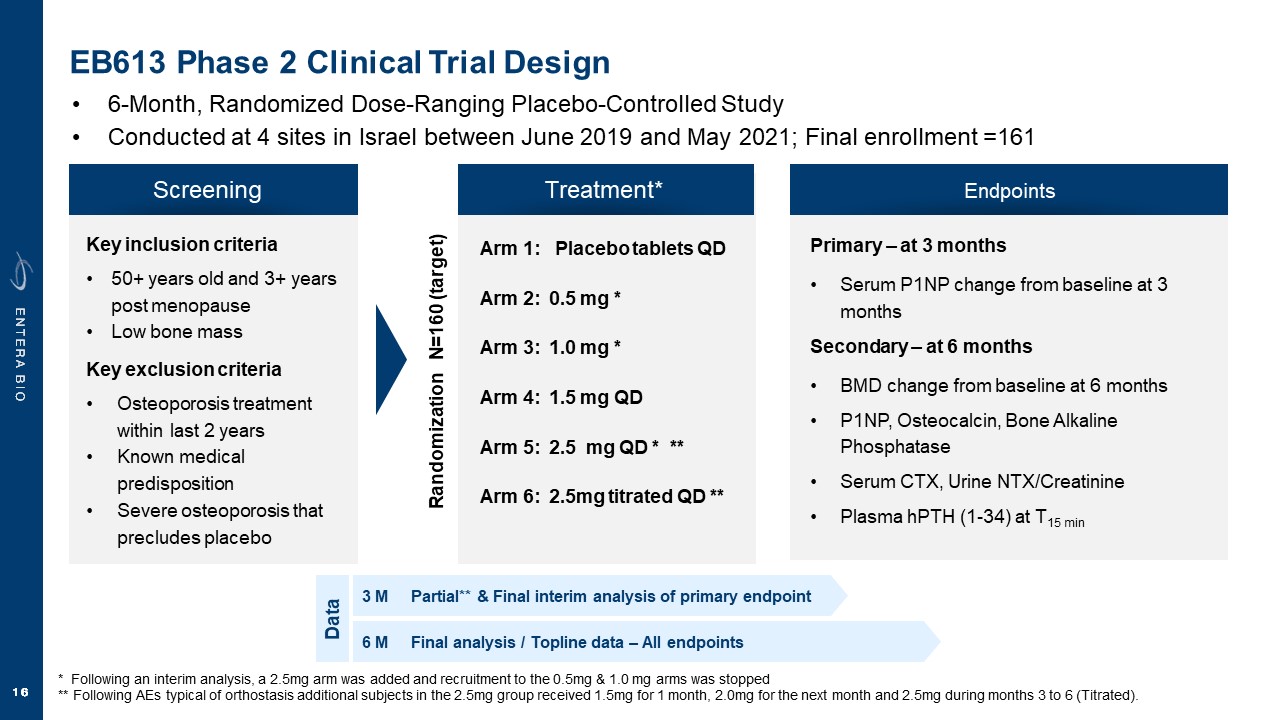
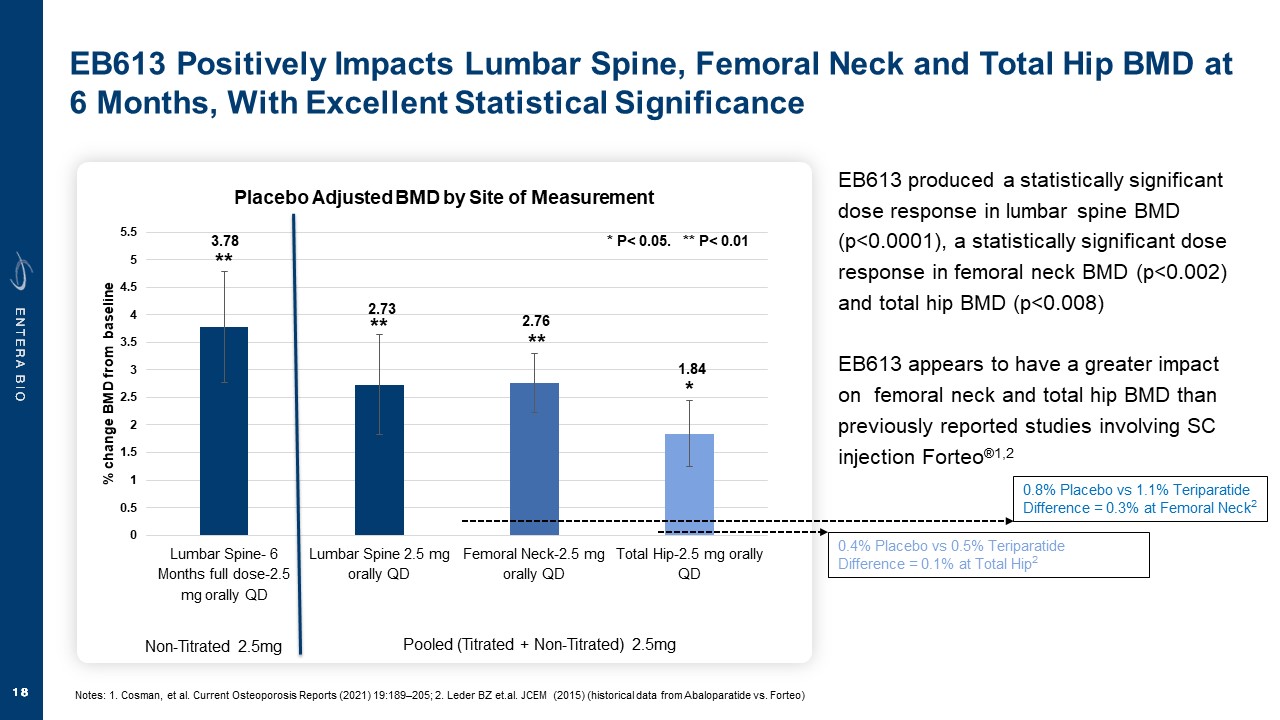
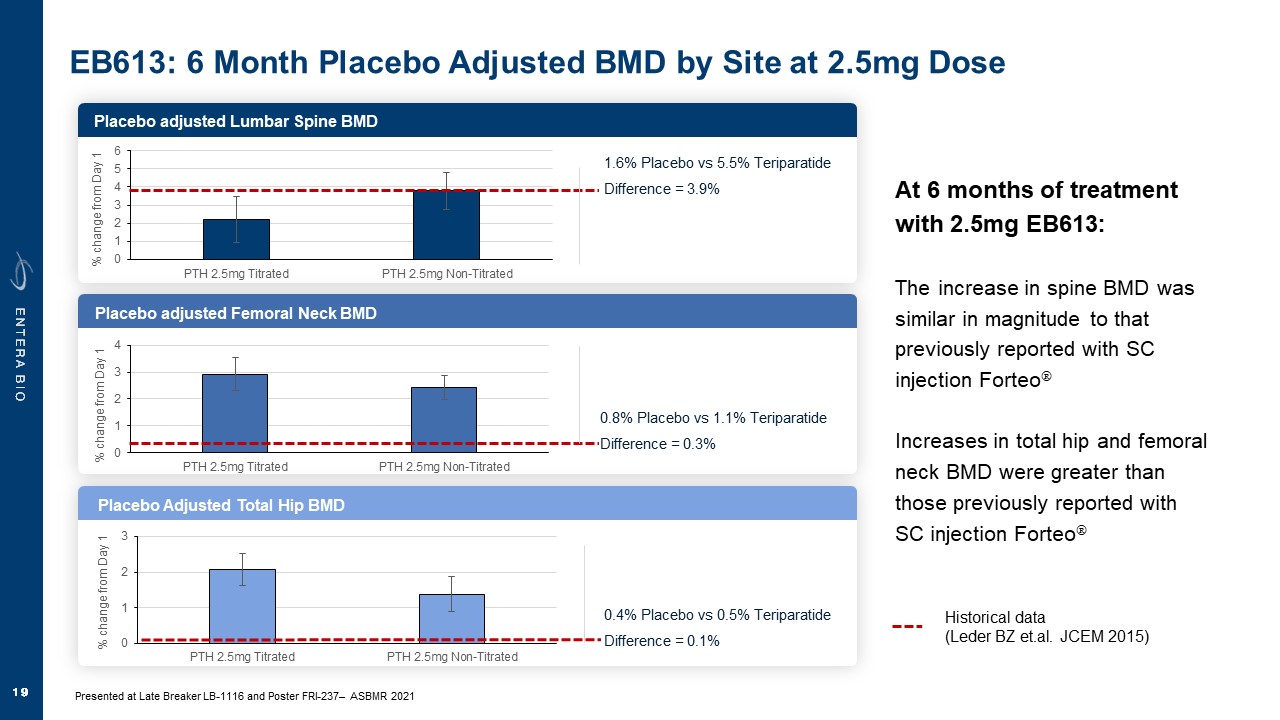
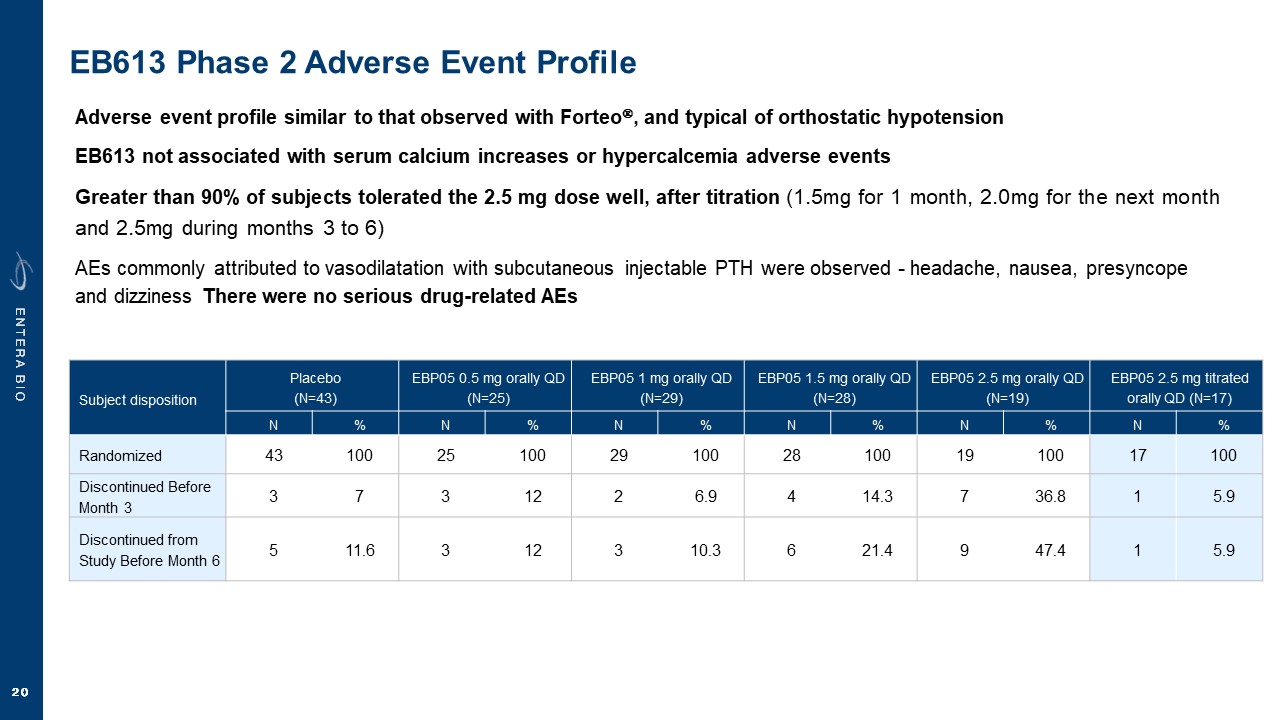
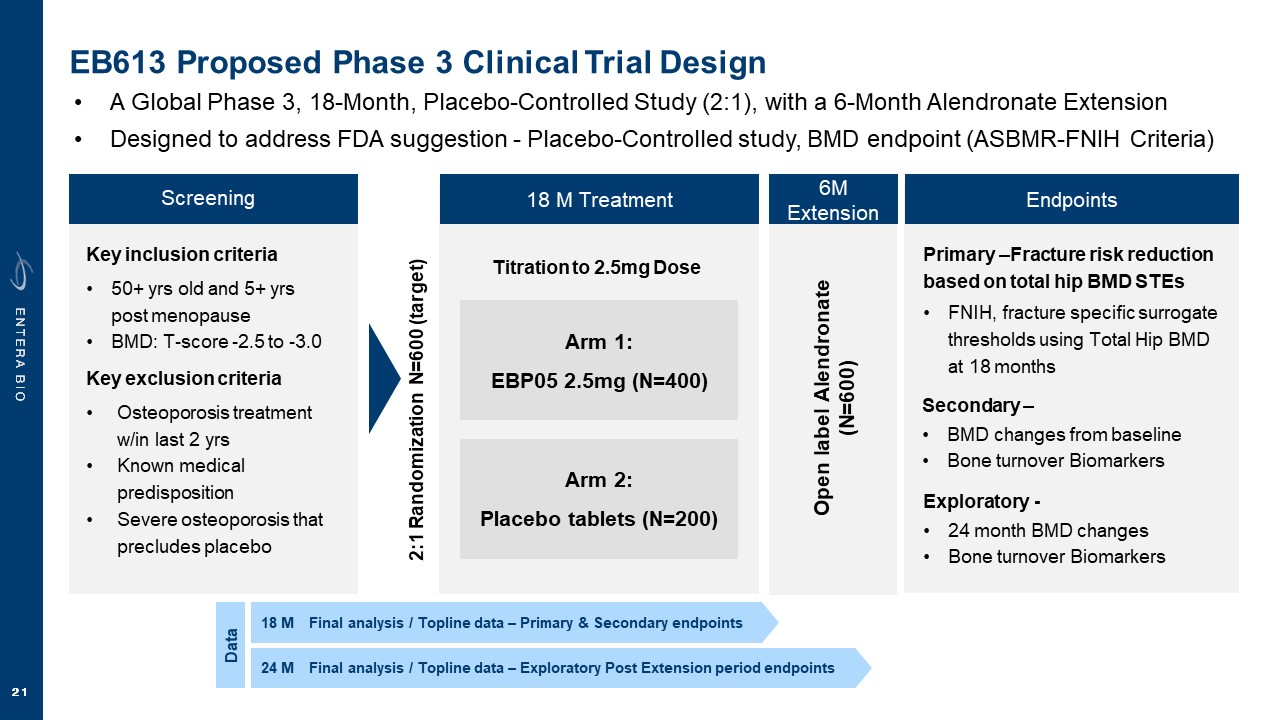
Disclaimer Various statements in this presentation are “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995. All statements (other than statements of historical facts) in this presentation regarding our prospects, plans, financial position, business strategy and expected financial and operational results may constitute forward-looking statements. Words such as, but not limited to, “anticipate,” “believe,” “can,” “could,” “expect,” “estimate,” “design,” “goal,” “intend,” “may,” “might,” “objective,” “plan,” “predict,” “project,” “target,” “likely,” “should,” “will,” and “would,” or the negative of these terms and similar expressions or words, identify forward-looking statements. Forward-looking statements are based upon current expectations that involve risks, changes in circumstances, assumptions and uncertainties. Forward-looking statements should not be read as a guarantee of future performance or results and may not be accurate indications of when such performance or results will be achieved. Important factors that could cause actual results to differ materially from those reflected in Entera’s forward-looking statements include, among others: changes in the interpretation of clinical data; results of our clinical trials; the FDA’s interpretation and review of our results from and analysis of our clinical trials; unexpected changes in our ongoing and planned preclinical development and clinical trials, the timing of and our ability to make regulatory filings and obtain and maintain regulatory approvals for our product candidates; the potential disruption and delay of manufacturing supply chains; loss of available workforce resources, either by Entera or its collaboration and laboratory partners; impacts to research and development or clinical activities that Entera is contractually obligated to provide, such as those pursuant to Entera’s agreement with Amgen; overall regulatory timelines; the size and growth of the potential markets for our product candidates; the scope, progress and costs of developing Entera’s product candidates; Entera’s reliance on third parties to conduct its clinical trials; Entera’s expectations regarding licensing, business transactions and strategic collaborations; Entera’s operation as a development stage company with limited operating history; Entera’s ability to continue as a going concern absent access to sources of liquidity; Entera’s ability to obtain and maintain regulatory approval for any of its product candidates; Entera’s ability to comply with Nasdaq’s minimum listing standards and other matters related to compliance with the requirements of being a public company in the United States; Entera’s intellectual property position and its ability to protect its intellectual property; and other factors that are described in the “Cautionary Statements Regarding Forward-Looking Statements,” “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” sections of Entera’s most recent Annual Report on Form 10-K filed with the SEC, as well as the company’s subsequently filed Quarterly Reports on Form 10-Q and Current Reports on Form 8-K. There can be no assurance that the actual results or developments anticipated by Entera will be realized or, even if substantially realized, that they will have the expected consequences to, or effects on, Entera. Therefore, no assurance can be given that the outcomes stated or implied in such forward-looking statements and estimates will be achieved. Entera cautions investors not to rely on the forward-looking statements Entera makes in this presentation. The information in this presentation is provided only as of the date of this presentation, and Entera undertakes no obligation, to update or revise publicly any forward-looking statements, whether as a result of new information, future events or otherwise, except to the extent required by law.