patents under the 2012 agreement will remain in force until the expiration of the last 2012 HB4 patent in 2033, unless terminated earlier in accordance with its terms.
Our patents or patent applications generally relate to compositions of matter for DNA and protein sequences, plants, plant parts and enzymes, and methods of improving plants and bacteria. We continue to file new patent applications. The main countries in which we seek patent protection are the United States, Brazil, Argentina, Australia, India, China, Mexico and certain other countries in South America and Europe.
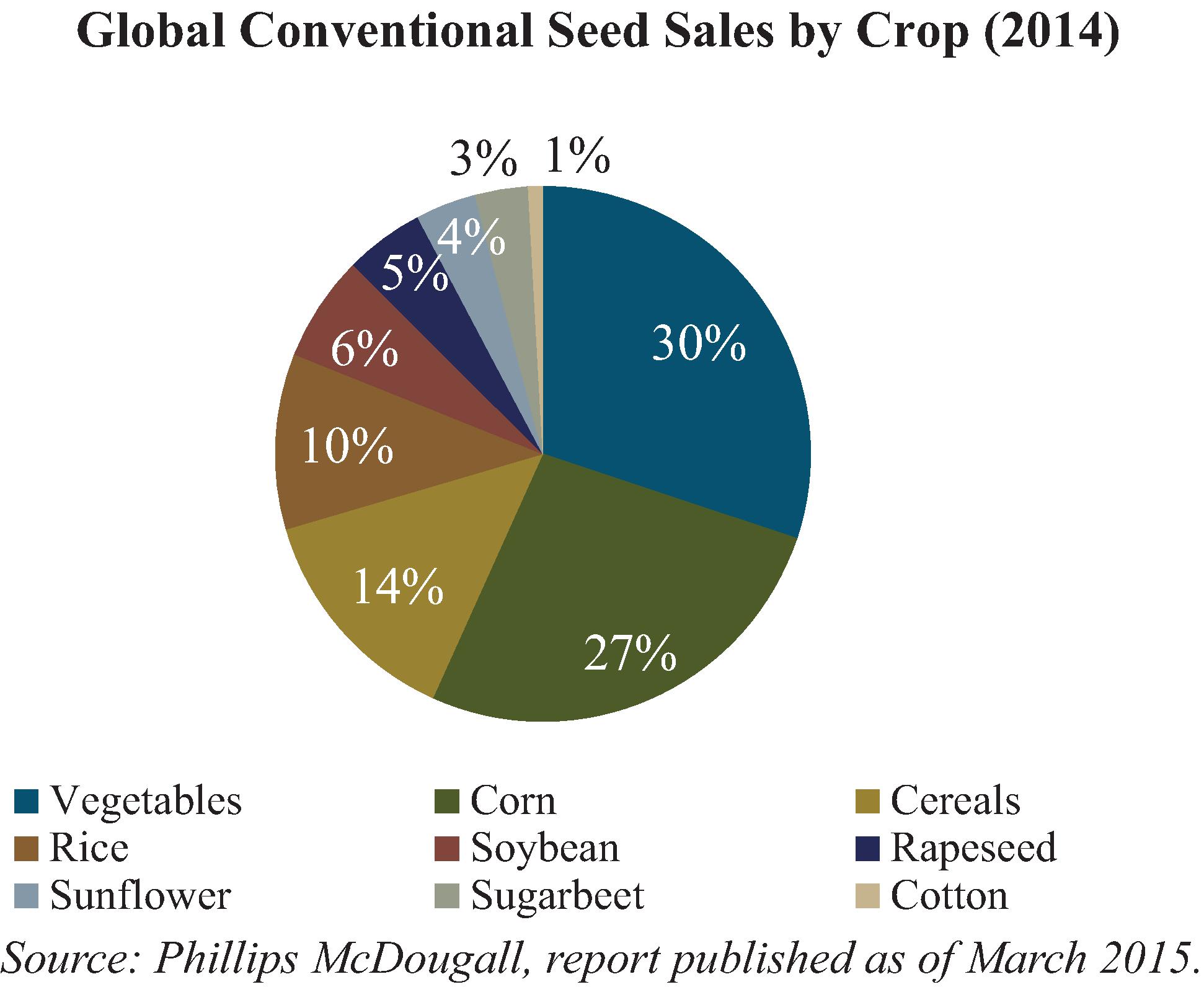
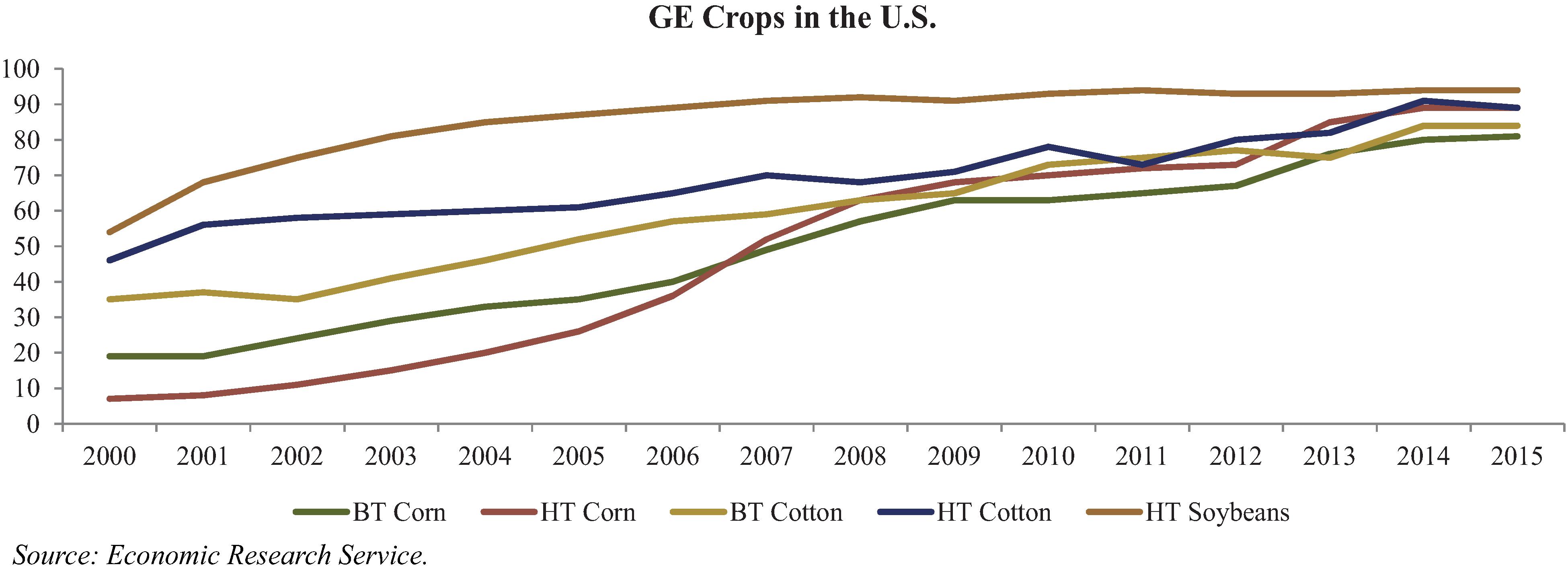
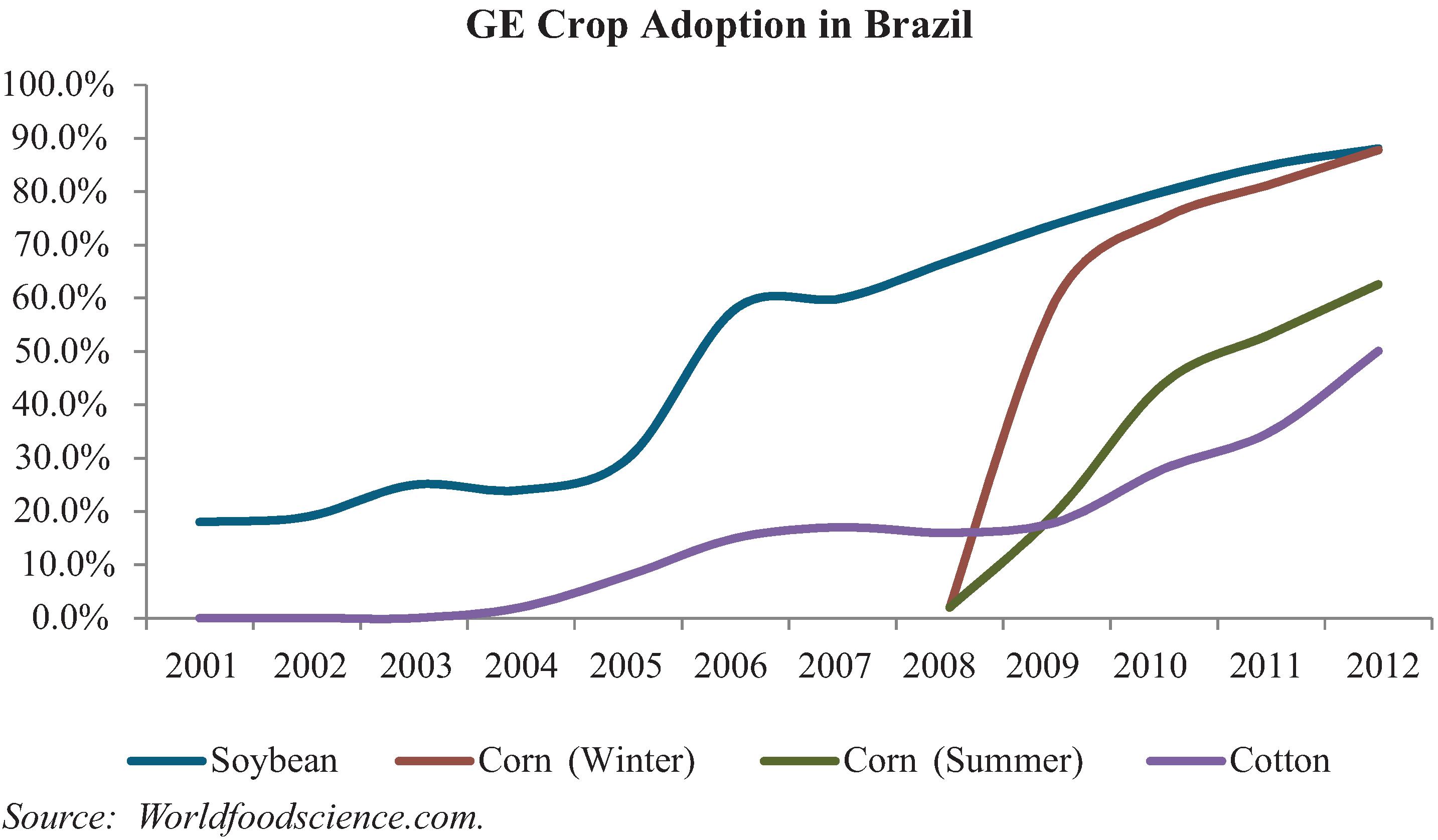
To date, we have identified and sought patent protection in our capacity as either title holder or exclusive licensee for over 101 plant genes linked to traits such as improved yield, drought tolerance, increased performance in high saline environments, NUE, WUE and TREF technologies, delayed senescence, herbicide resistance, reduced lignin technology and molecular farming technology for the production of chymosin in plants. These genes are currently protected through our own 24 patents and 17 patent applications. We have also licensed 60 patents linked to traits such as NUE, WUE and TREF technologies, delayed senescence, reduced lignin technology and herbicide resistance for use in our products.
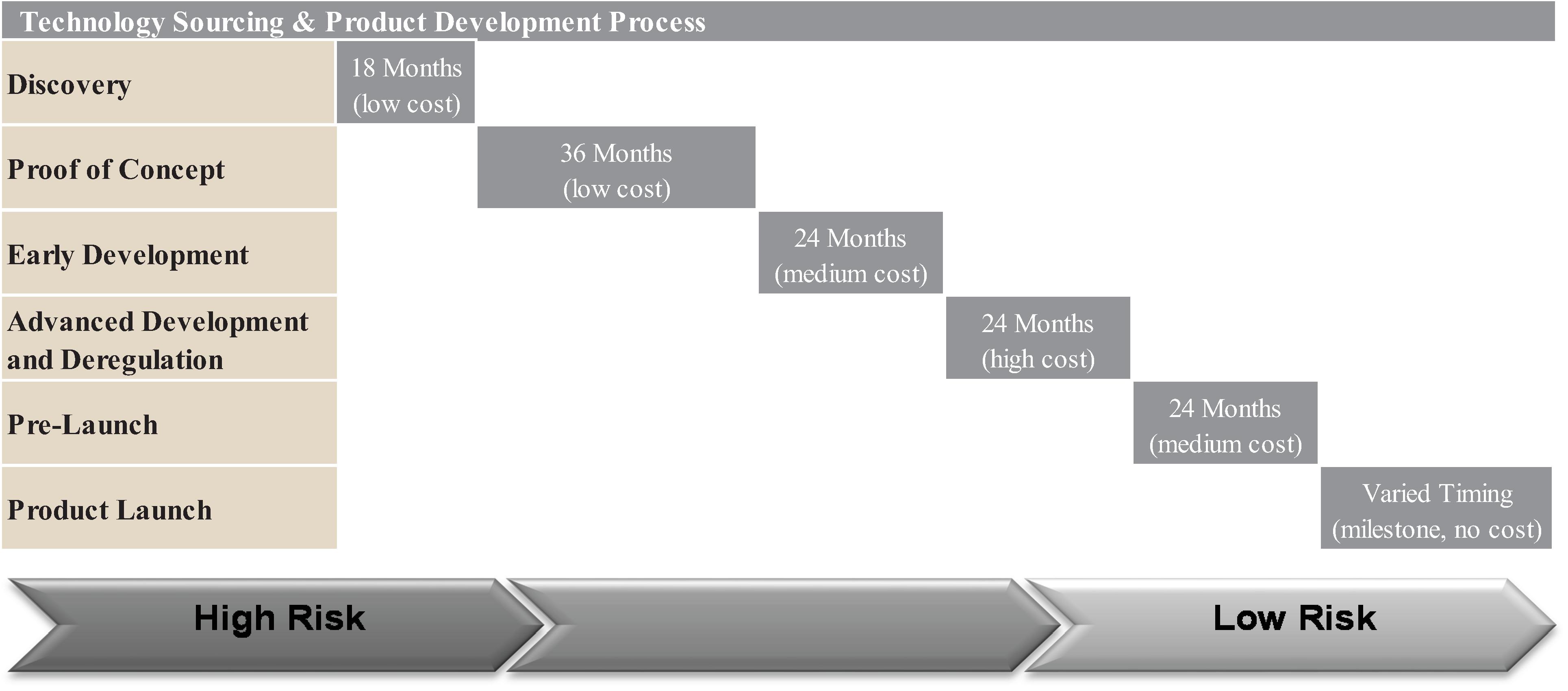
The discovery phase of our research and development process is based largely on collaborations with governmental agencies and scientific institutions, such as CONICET, the University of Illinois, the University of Victoria, the National University of the Litoral and various research universities throughout the world. See “—Research & Development Process—Discovery.” After we determine that we have discovered a new trait, trait composition, industrial enzyme, or a production methodology, we file a PCT patent application under the Patent Cooperation Treaty, or PCT. The PCT application allows an applicant to file one single application to seek protection for an invention in 148 countries throughout the world.
Within 18 months of the PCT filing, we file national applications in the countries in which we would like to seek protection. The main PCT countries in which we file in addition to the United States include Brazil, Australia, India, China, Mexico and certain other countries in South America and Europe. For non-PCT countries such as Argentina, Uruguay and Paraguay, a national filing is made at the same time as the PCT filing or 12 months after the PCT filing.
Additionally, we seek additional protection of our seed and germplasm intellectual property through PVP certificates, which preserve a variety owner’s exclusive rights to sell, reproduce, import, and export a plant variety and its seed. The duration of PVP protection varies among jurisdictions, and is 20 years from the time of issue in the United States and 20 and 15 years from the time of issue in Argentina and Brazil, respectively. In addition, in Argentina, we have received, as owner and/or as licensee, registrations with the RNC for 18 wheat varieties, two corn varieties, three sorghum varieties, two sunflower varieties and 14 soybean varieties, all of which we are marketing in Argentina. We are currently seeking registration with the RNC for 17 soybean varieties, one wheat variety, two amaranth varieties and 22 alfalfa varieties. We have also received the registration for one soybean variety in South Africa.
We seek to protect our non-patent intellectual property such as know-how and regulatory data through contracts and confidentiality mechanisms. Know-how generated by the activities of our companies is protected by specific services agreements or employment agreements. Employment agreements include undertakings regarding confidentiality and assignment of inventions and discoveries. Our regulatory data is protected by standard confidentiality and data protection mechanisms. We have 29 trademarks and 16 trademark applications in Argentina, Brazil, United States and Uruguay, including Bioceres, HB4, SPC, and Trigall Genetics.
We will continue to file and prosecute patent, PVP certificate and trademark applications in the United States and foreign jurisdictions, as well as maintain trade secrets as is consistent with our business plan in an ongoing effort to protect our intellectual property.
Government Regulation
Our business is subject to regulation related to agriculture, health and the environment. To operate, we must obtain various permits and licenses from government authorities and municipalities in our active jurisdictions, and we must maintain our compliance with the terms of those permits, licenses and other government standards as necessary. These laws and regulations, particularly in relation to biotechnology, are not fully settled, but continue to evolve in order to keep pace with technological advances.