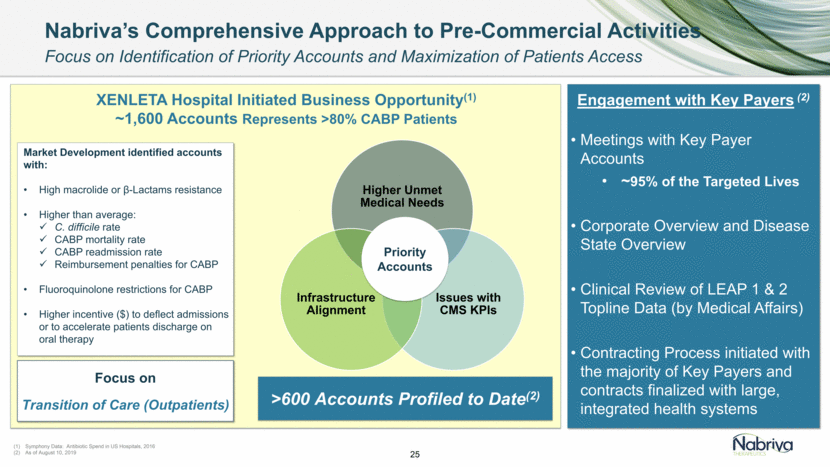
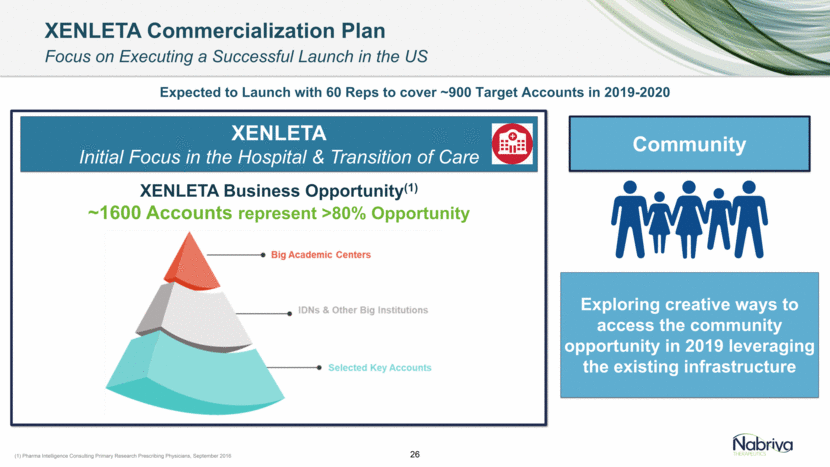
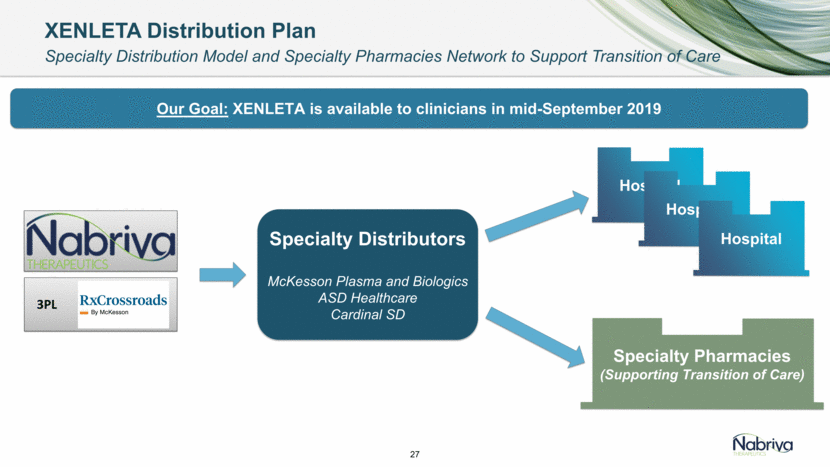
Safe Harbor and Disclaimer This presentation contains forward-looking statements about future expectations, plans and prospects for Nabriva Therapeutics, including but not limited to statements about launch and commercialization of XENLETA for the treatment of CABP, the development of CONTEPO for cUTI, the clinical utility of XENLETA for CABP and of CONTEPO for cUTI, plans for and timing of the review of regulatory filings for CONTEPO, efforts to bring XENLETA and CONTEPO to market, the market opportunity for and the potential market acceptance of XENLETA for CABP and CONTEPO for cUTI, plans for distributing and timing of the availability of XENLETA, the development of XENLETA and CONTEPO for additional indications, the development of additional formulations of XENLETA and CONTEPO, plans to pursue research and development of other product candidates, the sufficiency of Nabriva Therapeutics’ existing cash resources and other statements containing the words “anticipate,” “believe,” “estimate,” “expect,” “intend,” “may,” “plan,” “predict,” “project,” “target,” “potential,” “likely,” “will,” “would,” “could,” “should,” “continue,” and similar expressions, constitute forward-looking statements within the meaning of The Private Securities Litigation Reform Act of 1995. Actual results may differ materially from those indicated by such forward-looking statements as a result of various important factors, including: Nabriva Therapeutics’ ability to successfully implement its commercialization plans for XENLETA and whether market demand for XENLETA is consistent with its expectations, Nabriva Therapeutics’ ability to build and maintain a sales force and prepare for commercial launch of XENLETA on the timeline expected, or at all, the content and timing of decisions made by the U.S. Food and Drug Administration and other regulatory authorities, Nabriva Therapeutics’ reliance on third-party manufacturers for the commercial supply of XENLETA and third-party distributors to make XENLETA available to hospitals and the medical community and the ability of such third parties to comply with regulatory requirements, the uncertainties inherent in the initiation and conduct of clinical trials, availability and timing of data from clinical trials, whether results of early clinical trials or studies in different disease indications will be indicative of the results of ongoing or future trials, uncertainties associated with regulatory review of clinical trials and applications for marketing approvals, the availability or commercial potential of CONTEPO for the treatment of cUTI or of XENLETA for the treatment of CABP, the ability to retain and hire key personnel, the sufficiency of cash resources and need for additional financing and such other important factors as are set forth in Nabriva Therapeutics’ annual and quarterly reports and other filings on file with the U.S. Securities and Exchange Commission. In addition, the forward-looking statements included in this presentation represent Nabriva Therapeutics’ views as of the date of this presentation. Nabriva Therapeutics anticipates that subsequent events and developments will cause its views to change. However, while Nabriva Therapeutics may elect to update these forward-looking statements at some point in the future, it specifically disclaims any obligation to do so. These forward-looking statements should not be relied upon as representing Nabriva Therapeutics’ views as of any date subsequent to the date of this presentation. 2