
Exhibit 99.1 BeiGene Mid-Year 2019 Clinical Data Wrap-Up June 20, 2019

Welcome Howard Liang, Ph.D., CFO and Chief Strategy Officer 2

Certain statements contained in this presentation and in the accompanying oral presentation, other than statements of fact that are independently verifiable at the date hereof, may constitute forward-looking statements. Disclosures Examples of such forward-looking statements include those regarding investigational drug candidates and clinical trials and the status and related results thereto, as well as those regarding continuing and further development and commercialization efforts and transactions with third parties. Such statements, based as they are on the current analysis and expectations of management, inherently involve numerous risks and uncertainties, known and unknown, many of which are beyond BeiGene’s control. Such risks include but are not limited to: the impact of general economic conditions, general conditions in the pharmaceutical industries, changes in the global and regional regulatory environments in the jurisdictions in which BeiGene does business, market volatility, fluctuations in costs and changes to the competitive environment. Consequently, actual future results may differ materially from the anticipated results expressed in the forward-looking statements. In the case of forward- looking statements regarding investigational drug candidates and continuing further development efforts, specific risks which could cause actual results to differ materially from BeiGene’s current analysis and expectations include: failure to demonstrate the safety, tolerability and efficacy of our drug candidates, final and quality controlled verification of data and the related analyses, the expense and uncertainty of obtaining regulatory approval, including from the FDA, NMPA (formerly CFDA/CDA) and EMA, the possibility of having to conduct additional clinical trials and BeiGene’s reliance on third parties to conduct drug development, manufacturing and other services. Further, even if regulatory approval is obtained, pharmaceutical products are generally subject to stringent on-going governmental regulation, challenges in gaining market acceptance and competition. These statements are also subject to a number of material risks and uncertainties that are described in BeiGene’s filings with the Securities and Exchange Commission (SEC). The reader should not place undue reliance on any forward-looking statements included in this presentation or in the accompanying oral presentation. These statements speak only as of the date made, and BeiGene is under no obligation and disavows any obligation to update or revise such statements as a result of any event, circumstances or otherwise, unless required by applicable legislation or regulation. Some of the clinical data in this presentation relating to BeiGene’s investigational drug candidates is from early phase, single-arm trials. When such data or data from later stage trials are presented in relation to other investigational or marketed drug products, the presentation and discussion are not based on head-to-head trials between BeiGene’s investigational drug candidates and other products. BeiGene is still conducting clinical trials and, as additional patients are enrolled and evaluated, data on BeiGene’s investigational drug candidates may change. This presentation and the accompanying oral presentation contains data and information obtained from third- party studies and internal company analysis of such data and information. BeiGene has not independently verified the data and information obtained from these sources. Forward-looking information obtained from these sources is subject to the same qualifications noted above. 3

Agenda Welcome – Howard Liang, Ph.D. Introduction – John V. Oyler Zanubrutinib – Jane Huang, M.D. • Pooled safety data summary • Phase 3 in WM: MYD88WT cohort • Phase 2 in R/R CLL/SLL • Phase 1 in WM cohort • Phase 1 in MCL cohort • Phase 2 in R/R MCL summary • Phase 1b Combination with Obinutuzumab in R/R FL and CLL Tislelizumab – Eric Hedrick, M.D. • Phase 2 in R/R cHL • Phase 1 NPC cohort Key Takeaways – Eric Hedrick, M.D. Q&A 4

Introduction John V. Oyler, Chairman, Co-Founder and CEO 5

Business Updates Regaining tislelizumab worldwide rights • Termination of collaboration agreement with Celgene ahead of pending BMS-Celgene merger • BeiGene to receive $150M payment from Celgene • Early resolution in the best interest of the asset and both companies • Minimal interruption in clinical execution – BeiGene has been leading 90% of the Phase 3 or potentially registration- enabling trials for tislelizumab • Full options available commercially, from going alone, to co-promotion, to out-licensing – we will seek to maximize the value of the asset Regulatory progress • Tislelizumab and zanubrutinib approvals in China expected in 2019 • Tislelizumab filed for urothelial carcinoma in China in May • ABRAXANE® filed for pancreatic cancer in China in May Clinical progress • Executing towards enrollment completion in 2019 for a large number of Phase 3 or potentially registration-enabling trials • Progress will be updated in quarterly earnings releases • Expect data readouts in 2019 / 2020 from a large number of key Phase 3 or potentially registration-enabling studies for zanubrutinib, tislelizumab and pamiparib 6

Tislelizumab’s Advanced Development Status Phase 3 (n=800) in 2L NSCLC Enrollment complete tislelizumab vs. docetaxel NSCLC Primary endpoint: OS Initiated in Nov. 2017 Enrollment expected to complete in 2019 Phase 3 (n=640) in 1L HCC Phase 2 (n=225) in 2L/3L HCC tislelizumab vs. sorafenib tislelizumab monotherapy Enrollment expected to complete in 2020-2021 Global HCC Primary endpoint: OS Primary endpoint: ORR by IRC Trials Initiated in Jan. 2018 Initiated in Apr. 2018, enrollment completed in 1Q:19 (China and Phase 3 (n=450) in 2L ESCC Phase 3 (n=480) in 1L advanced ESCC ROW) tislelizumab vs. single-agent chemo (paclitaxel, docetaxel, or irinotecan) tislelizumab or placebo + platinum- and fluoropyrimidine-based chemo ESCC Primary endpoint: OS Co-primary endpoints: PFS and OS Initiated in Jan. 2018 Initiated in Dec. 2018 Phase 3 (n=720) in 1L advanced GC R/R NK/T- Phase 2 (n=90) in 1L R/R Mature T- and NK- Neoplasms tislelizumab or placebo + platinum- and fluoropyrimidine-based chemo cell tislelizumab monotherapy GC Co-primary endpoints: PFS and OS lymphom Primary endpoints: ORR Initiated in Dec. 2018 as Initiated in Apr. 2018 Phase 3 (n=340) in 1L Stage IIIB or IV squamous NSCLC Phase 3 (n=320) in 1L Stage IIIB or IV non-squamous N SC LC Tislelizumab+ paclitaxel and carboplatin combo or nab-paclitaxel and Tislelizumab+ chemo (platinum-pemetrexed) vs. chemo carboplatin combo vs. paclitaxel and carboplatin combo NSCLC Primary endpoint: PFS 2,714 patients Primary endpoint: PFS Initiated in Jul. 2018 Initiated in Aug. 2018 currently enrolled Pivotal phase 2 (n=110) in 2L UC Pivotal phase 2 (n=70) in R/R cHL in these trials tislelizumab monotherapy China tislelizumab monotherapy UC Primary endpoint: ORR cHL Primary endpoint: ORR Trials 2,950 total Initiated in Jul. 2017, enrollment completed in 3Q:18, Initiated in Apr. 2017, enrollment completed in NDA accepted May 2019 4Q:17, NDA accepted in Aug 2018 patients dosed MSI-H or Phase 2 (n=60) in MSI-H or dMMR solid tumors across all Phase 3 (n=256) in 1L tislelizumab + chemo (gemcitabine dMMR tislelizumab monotherapy plus cisplatin) vs. placebo + chemo Primary endpoint: PFS tislelizumab solid Primary endpoint: ORR NPC Initiated in Apr. 2019 tumors Initiated in Sept. 2018 studies *Tislelizumab dosage 200mg every three weeks, Q3W. Global Ph3 trial in Stage III NSCLC is run by Celgene; global Ph2 in R/R/ NK/T-cell lymphoma and Ph2 trial in MSI-H or dMMR solid tumors in China are potentially registrational-enabling trials. ORR: Overall response rate; PFS: Progression-free survival; cCRT: concurrent chemoradiotherapy; IRC: Independent Review Committee; ITT: Intent-to-treat 7

Zanubrutinib Jane Huang, M.D., CMO, Hematology

Zanubrutinib Pooled Safety Abstract PS1159 24th European Hematology Association Congress Jane Huang, M.D., CMO, Hematology 9
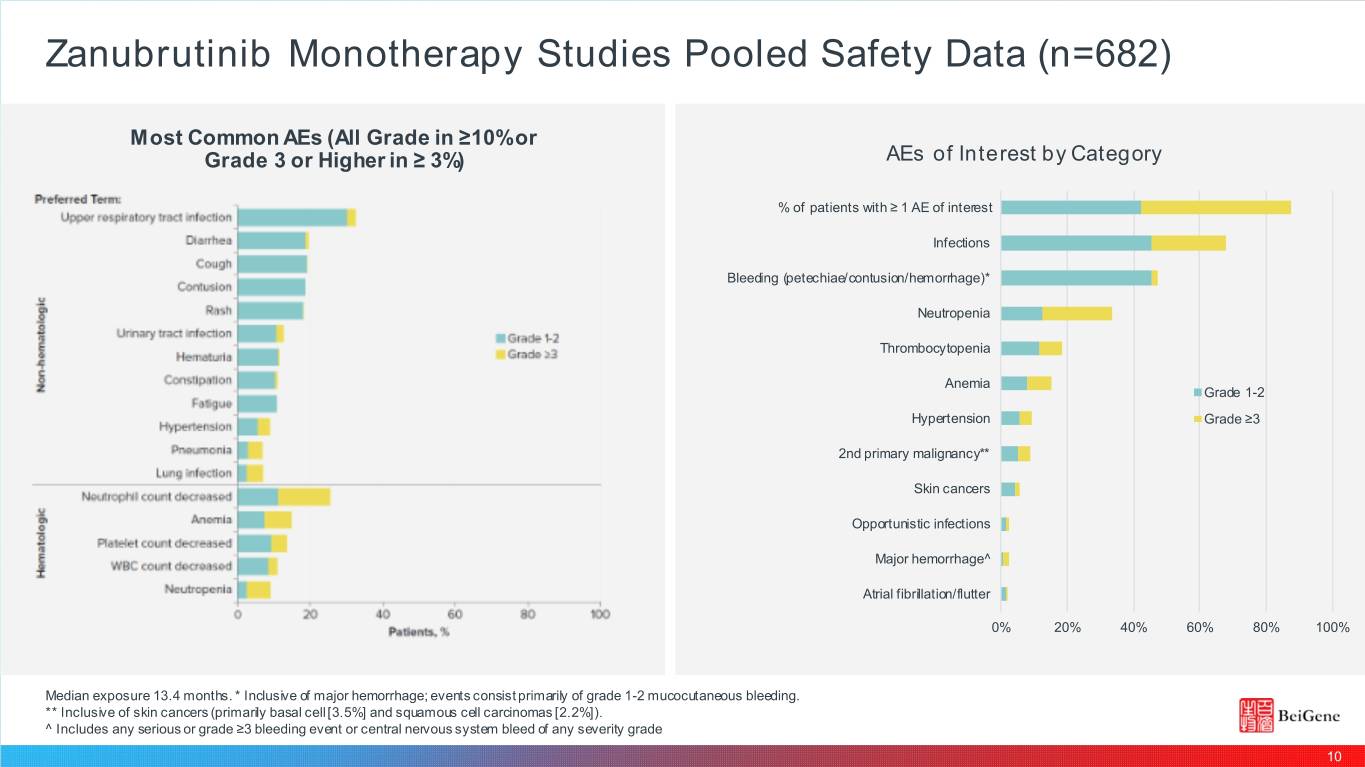
Zanubrutinib Monotherapy Studies Pooled Safety Data (n=682) M ost Common AEs (All Grade in ≥10% or Grade 3 or Higher in ≥ 3%) AEs of Interest by Category % of patients with ≥ 1 AE of interest Infections Bleeding (petechiae/contusion/hemorrhage)* Neutropenia Thrombocytopenia Anemia Grade 1-2 Hypertension Grade ≥3 2nd primary malignancy** Skin cancers Opportunistic infections Major hemorrhage^ Atrial fibrillation/flutter 0% 20% 40% 60% 80% 100% Median exposure 13.4 months. * Inclusive of major hemorrhage; events consist primarily of grade 1-2 mucocutaneous bleeding. ** Inclusive of skin cancers (primarily basal cell [3.5%] and squamous cell carcinomas [2.2%]). ^ Includes any serious or grade ≥3 bleeding event or central nervous system bleed of any severity grade 10

Pooled Safety Data* Zanubrutinib Zanubrutinib Acalabrutinib6 Ibrutinib Background EHA 20181 EHA 20192 Rate n 476 682 612 7565; 1,124b; 2,0904-2,1523 1,605c Major hemorrhage % 2% 2.5% 2.8% 4%5 (Gr≥3) (2%) (2.1%) (2.0%) 3%5 [events/100 pt. yrs.] [2.07] - [3.03] [1.93] Atrial fibrillation % ~2% 1.9% 2.9% 9%c (Gr≥3) (0.2%) (0.6%) (1.0%) (4.1%)c [events/100 pt. yrs.] [1.56] - [3.34] [0.844] Diarrhea ~15% 19.4% 40% 39%c (Gr≥3) (1%) (0.9%) (2.1%) (3%)c Median exposure, mo 7.0 (0.02-36.05) 13.4 (6.1-19.6) 18.5 (0.03- 14.8moca (25th-75th percentile) 37.4)# (range)# * Pooled safety data from separate trials and sources. Limitations regarding cross-trial comparisons apply. Sources: 1 Tam et al, EHA 2018; 2 Tam et al, EHA 2019; 3 Caron, F Blood Advances 1:12 2019; 4 Leong, D Blood 128:1 2016; 5 O’Brien S Clin Lymphoma Myeloma & Leukemia 18:10 2018; 6 Byrd et al, ASH 2017; a Median treatment duration; b Data from label out of 1,124 patients; c Data from label out of 1,605 patients 11

Zanubrutinib Phase 3 in WM: MYD88WT Cohort Abstract PF487 24th European Hematology Association Congress Jane Huang, M.D., CMO, Hematology 12

WT Trial Design: Phase 3 in WM, MYD88 Cohort Data cut: Feb 28, 2019 Ar m A Cohort 1: R/R or TN* WM with MYD88L265P Mutation Zanubrutinib 160 mg bid until PD MYD88MUT WM patients R (n=201) 1:1 Ar m B Ibrutinib Stratification factors: 420mg qd until PD • CXCR4 mutational status (CXCR4WHIM vs CXCR4WT or missing) • No. of prior lines of therapy (0 vs 1-3 vs > 3) Cohort 2: WM with wild type MYD88; present in ~10% of enrolled patients Ar m C MYD88WT WM patients Zanubrutinib (n = 26 WT + 2 unknown) 160 mg bid until PD bid, twice daily ; PD, progressive disease; qd, once daily; R/R, relapsed/refractory; TN, treatment-naïve; *TN must be unsuitablefor standard chemoimmunotherapy. EUDRACT 2016-002980-33; NCT03053440 13

Patient and Disease Characteristics Data cut: Feb 28, 2019 Characteristic Total (n=26) Age, years, median (range) 71.5 (39-87) Gender, n (%) Male 14 (53.8) Female 12 (46.2) ECOG performance status, n (%) 0 9 (34.6) 1 14 (53.8) 2 3 (11.5) Prior treatment status Treatment-naïve, n (%) 5 (19.2) Relapsed/refractory (R/R), n (%) 21 (80.8) Number of prior therapies for R/R patients, median (range) 1 (1-5) Extramedullary Disease present at baseline 13 (50.0) Genotype, n (%) MYD88WT/CXCR4WT 23 (88.5) MYD88WT/CXCR4WHIM 1 (3.8) MYD88WT/CXCR4 unknown 2 (7.7) ECOG, Eastern Cooperative Oncology Group. 14

Best Response Data cut: Feb 28, 2019 Total Best response, n (%) (n=26) Overall response rate (ORR) 21 (80.8) Major response rate (MRR, PR or better) 14 (53.8) VGPR 6 (23.1)a,b PR 8 (30.8)b MR 7 (26.9) SD 4 (15.4) PD 1 (3.8) Time to MR, med (range), mo Months 2.9 (1.9 -7.4) Study follow-up time, med (range), mo Months 12.2 (2.3 - 21.7) aOne patient achieved IgM complete response (normalized IgM and negative immunofixation since Cycle 11, with bulky extramedullary disease improving). bIncluding pts confirmed by next-generation sequencing of no other activating MYD88 mutations: 3 of 6 VGPR (including IgM CR); 3 of 8 PR. CR, complete response; MR, minor response; PD, progressive disease; PR, partial response; SD, stable disease; VGPR, very good partial response. 15

Adverse Events Overview Data cut: Feb 28, 2019 Treatment Emergent Adverse Event n (%) 2 patients discontinued due to AEs Patients w ith ≥1 AE grade ≥3 12 (46.2) • Grade 4 subdural hemorrhage Patients w ith ≥1 serious AE 8 (30.8) • Grade 3 diarrhea a AE leading to treatment discontinuation 2 (7.7) Major hemorrhage occurred in 2 Fatal AE 0 patients • Gastric ulcer hemorrhage AE of interest (BTK inhibitor class) • Periorbital hematoma, subdural Bleeding/petechiae/bruis ing of any grade 9 (34.6) hematoma, and subdural Most commonly grade 1 contusion 4 (15.4) hemorrhage; treatment was Diarrhea 5 (19.2) permanently discontinued per Hypertension 5 (19.2) protocol Grade 3 or 4 cytopenia 4 (15.4) No fatal treatment emergent AEs or Grade 3 or 4 infections 3 (11.5) atrial fibrillation/flutter events have Second malignancyb 3 (11.5) been reported Major hemorrhagec 2 (7.7) Atrial fibrillation/flutter 0 Common adverse events (> 15% of patients) include, all grades (Gr≥3): hypertension 19.2%(11.5%), diarrhea 19.2%(7.7%), pneumonia 15.4%(3.8%), upper respiratory tract infection 15.4%, muscle spasm 15.4%, contusion 15.4%, constipation 15.4% AE, adv erse event; SAE, serious adverse event. aGrade 4 subdural hemorrhage (related) and grade 3 diarrhea (related). bBasal cell carcinoma (n=2) and Queyrat erythroplasia (n=1). cDefined as any-grade ≥3 hemorrhage or any-grade CNS hemorrhage: gastric ulcer hemorrhage; 1 patient had periorbital hematoma, subdural hematoma, and subdural hemorrhage. 16
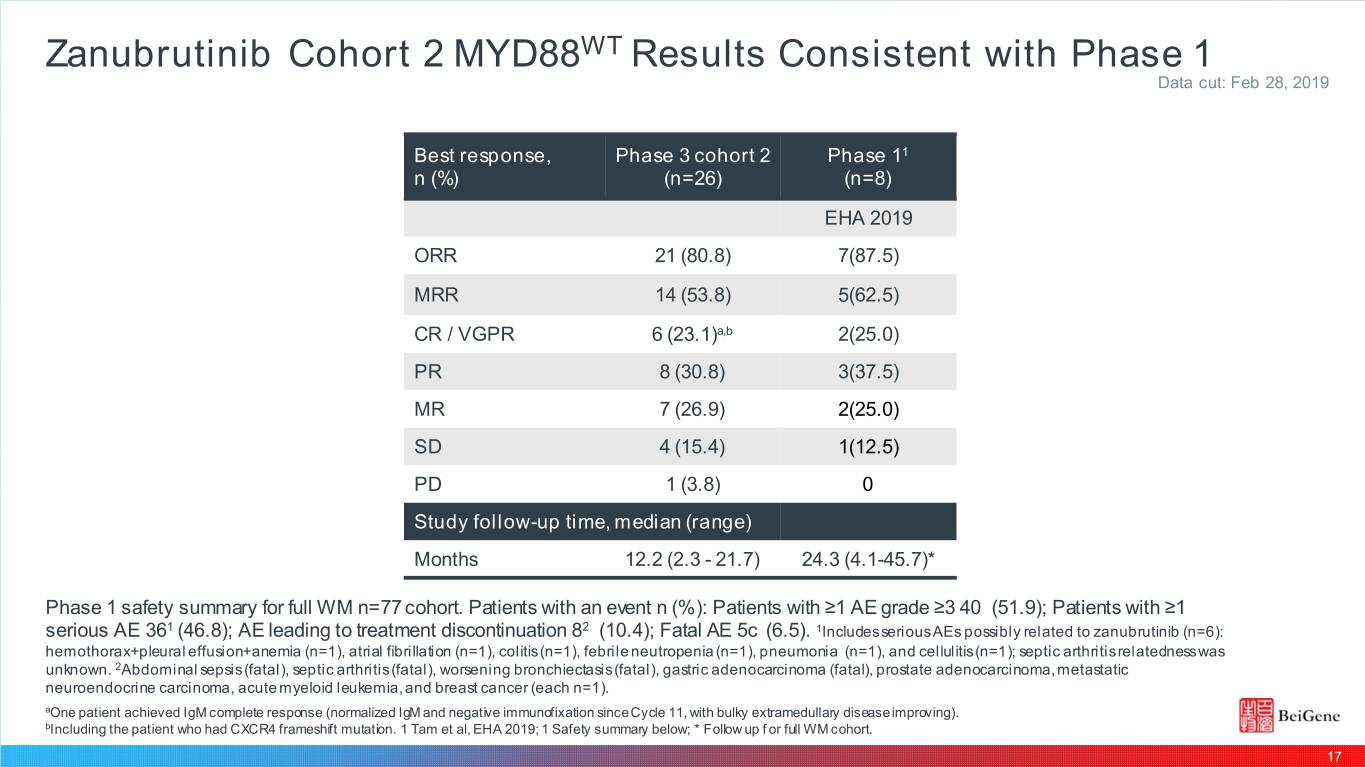
Zanubrutinib Cohort 2 MYD88WT Results Consistent with Phase 1 Data cut: Feb 28, 2019 Be st re sponse , Phase 3 cohort 2 Phase 11 n (%) (n=26) (n=8) EHA 2019 ORR 21 (80.8) 7(87.5) MRR 14 (53.8) 5(62.5) CR / VGPR 6 (23.1)a,b 2(25.0) PR 8 (30.8) 3(37.5) MR 7 (26.9) 2(25.0) SD 4 (15.4) 1(12.5) PD 1 (3.8) 0 Study follow-up time, median (range) Months 12.2 (2.3 - 21.7) 24.3 (4.1-45.7)* Phase 1 safety summary for full WM n=77 cohort. Patients with an event n (%): Patients with ≥1 AE grade ≥3 40 (51.9); Patients with ≥1 serious AE 361 (46.8); AE leading to treatment discontinuation 82 (10.4); Fatal AE 5c (6.5). 1Includes serious AEs possibly related to zanubrutinib (n=6): hemothorax+pleural effusion+anemia (n=1), atrial fibrillation (n=1), colitis (n=1), febrile neutropenia (n=1), pneumonia (n=1), and cellulitis (n=1); septic arthritis relatedness was unknown. 2Abdominal sepsis (fatal), septic arthritis (fatal), worsening bronchiectasis (fatal), gastric adenocarcinoma (fatal), prostate adenocarcinoma, metastatic neuroendocrine carcinoma, acute myeloid leukemia, and breast cancer (each n=1). aOne patient achieved IgM complete response (normalized IgM and negative immunofixation since Cycle 11, with bulky extramedullary disease improving). bIncluding the patient who had CXCR4 frameshift mutation. 1 Tam et al, EHA 2019; 1 Safety summary below; * Follow up f or full WM cohort. 17

Zanubrutinib Phase 2 in R/R CLL/SLL in China (ICML) Abstract 015 15th International Conference on Malignant Lymphoma Jane Huang, M.D., CMO, Hematology 18

Trial Design: Phase 2 in R/R CLL/SLL in China R/R CLL/SLL Key Criteria Zanubrutinib 160 mg bid Inclusion Criteria until progression, intolerable toxicity, • ≥18 years old or end of study • At least one treatment indication • Measurable lesion Exclusion Criteria • Richter syndrome Objectives • Insufficient organ function • Primary: IRC-assessed ORR • Secondary: PFS, DOR, TTR, safety • Exploratory: Biomarkers Response assessment • iwCLL 2008 criteria for CLL (with modification for PRL (Cheson, Hallek 2012)) • CT-based assessment according to Lugano Classification for SLL1 bid, twice a day ; CLL, chronic lymphocytic leukemia; DOR, duration of response; IRC, independent review committee; PK, pharmacokinetics; SLL, small lymphocytic lymphoma; TTR, time to response. 1. Cheson BD et al. J Clin Oncol. 2014;32(27):3059-3067. 19

Patient and Disease Characteristics Data cut: Dec 14, 2018 Baseline Characteristics n = 91 Baseline Characteristics n = 91 Median age (range), years 61.0 (35 - 87) Splenomegaly, n (%) 56 (61.5) Male, n (%) 52 (57.1) Hepatomegaly, n (%) 11 (12.1) Late stage a, n (%) 63 (69.2) Absolute lymphocyte count, n (%) 9 57 (62.6) Prior therapy, n (%) <25 ×10 /L Alkylator (including bendamustine) 68 (74.7) 25 – 100 ×109/L 26 (28.6) Purine analog 52 (57.1) >100 × 109/L 8 (8.8) Anti-CD20 antibody 54 (59.3) Refractory to last therapy, n (%) 72 (79.1) Genetic Characteristics n = 91 ECOG PS 0/1, n (%) 88 (96.7) TP53 mutation and/or 17p deletion, n (%) 22 (24.2) Bulky disease, n (%) LDi ≥5 cm 40 (44.4) IGHV unmutated, n (%) 51 (56.0) Beta-2 microglobulin >3.5 mg/L, n (%) 68 (74.7) Cytogenetic abnormalities, n (%) 17p deletion 17 (18.7) 11q deletion 20 (22.0) 13q deletion 41 (45.1) Trisomy 12 21 (23.1) a Percentages are based on number of CLL patients with Binet C and SLL patients with stage III and IV. ECOG PS, Eastern Cooperative Oncology Group performance status. 20

Best Response Data cut: Dec 14, 2018 Response by IRC n = 91 ORR including PR-L, n (%) 77 (84.6) BOR, n (%) Complete response (CR) 3 (3.3) Partial response (PR) 54 (59.3) Partial response with lymphocytosis (PR-L) 20 (22.0) Stable disease (SD) 4 (4.4) Progressive disease (PD) 4 (4.4) Not evaluable a 3 (3.3) Discontinued prior to first post-baseline assessment 3 (3.3) The ORR was 91.2% (83.4, 96.1) and the PR or higher rate was 72.5% (62.2, 81.4) as assessed by investigators High concordance rate for overall response assessments was 91.2% between IRC and investigator assessments Median study follow-up 15.08 months aMissing anatomy imaging for 2 patients, and without evidence of response maintenance for at least 2 months for 1 patient, separately. BOR, best overall response. 21
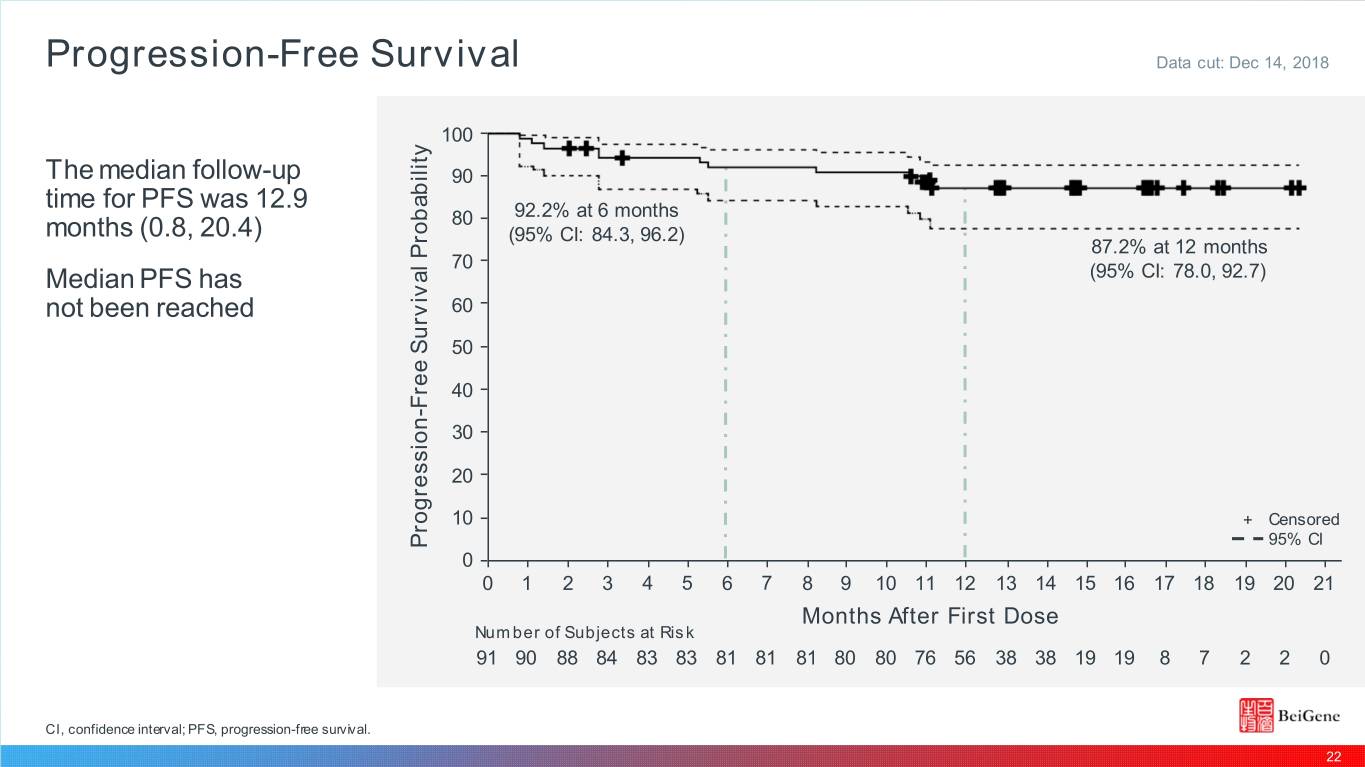
Progression-Free Survival Data cut: Dec 14, 2018 100 The median follow-up 90 time for PFS was 12.9 80 92.2% at 6 months months (0.8, 20.4) (95% CI: 84.3, 96.2) 87.2% at 12 months 70 Median PFS has (95% CI: 78.0, 92.7) not been reached 60 50 40 30 20 10 + Censored Progression - Free Survival Probability 95% CI 0 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 Months After First Dose Number of Subjects at Risk 91 90 88 84 83 83 81 81 81 80 80 76 56 38 38 19 19 8 7 2 2 0 CI, confidence interval; PFS, progression-free survival. 22

TEAE Regardless of Causality Data cut: Dec 14, 2018 Serious AEs were reported TEAE with Frequency ≥10% in 33% patients and grade ≥3 Neutrophil count decreased AEs were reported in 76%. Upper respiratory tract infection Purpura Platelet count decreased There were 8 patients reported Haematuria AEs leading to treatment Anaemia Hypokalaemia discontinuation Cough Hyperglycaemia Three patient-reported AEs White blood cell count decreased leading to death, all within 30 Carbon dioxide increased Diarrhoea days of last dose Lung infection • Lung infection / cardiac failure / Urinary tract infection respiratory (unlikely related) R ash Urobilinogen urine increased • Cardiopulmonary failure (unlikely Alanine aminotransferase… related) Hypoalbuminaemia Hyperuricaemia ≥ Grade 3 All grades • MODS (not related) in the setting Aspartate aminotransferase… of disease progression Pyre xia • These were determined unlikely or 0 10 20 30 40 50 60 70 80 90 100 unrelated to zanubrutinib treatment Patients, % ALT, alanine aminotransferase; AST, aspartate aminotransferase; TEAE, treatment-emergent adverse event; MODS, multiple organ dysfunction syndrome 23

Ibrutinib in Chinese Patients with R/R CLL Ibrutinib Trial PCI-32765CLL3002* Arms Ibrutinib Rituximab Investigator Investigator n 107 53 mFU months 17.8 ORR% (w/o PR-L) 45% 5.6% ORR% (including lymphocytosis) 57% 5.6% CR 1.9% 0 PR 43% 5.6% PR-L 11% 0 AEs, n (%); Ibrutinib (n=86) All Grade (%) Grade 3 or 4 (%) Rituximab (n=42) All Grade (%) Grade 3 or 4 (%): Diarrhea 25 (29.1), 2 (2.3), 3 (7.1), 0; Nasopharyngitis 14 (16.3), 1 (1.2), 1 (2.4), 0; Pneumonia 22 (25.6), 17 (19.8), 7 (16.7), 4 (9.5); Upper respiratory tract infection 20 (23.3), 6 (7.0), 4 (9.5), 1 (2.4); Rash 20 (23.3), 1 (1.2), 4 (9 .5), 0; Cough 19 (22.1), 1 (1.2), 2 (4.8), 0; Thrombocytopenia 15 (17.4), 5 (5.8), 1 (2.4), 0; Leukocytosis 12 (14.0), 12 (14.0), 0, 0; Fatigue 13 (15.1), 0, 3 (7.1), 0; Musculoskeletal Pain 13 (15.1), 0, 0, 0; Lymphocyte count increased 10 (11.6), 8 (9.3), 0, 0; Lactate dehydrogenase increased 9 (10.5), 2 (2.3), 1 (2.4), 0; Vertigo 9 (10.5), 0, 0, 0; Neutrophil count decreased (62.8), (37.2), (54.8), (33.3); Platelet count decreased (65.1), (15.1), (45.2), (9.5); Hemoglobin decreased (46.5), (1.2), (26.2), (0). Source: *Ibrutinib Chinese label 24

Other Zanubrutinib Studies Jane Huang, M.D., CMO, Hematology 25

Updated WM Cohort Safety and Efficacy from Global Phase 1 (EHA) Data cut: Sep 16, 2018 Be st re sponse , All Efficacy TN Patients (n=24) R/R Patients (n=49) n (%) Evaluable (n=73) ORR 67 (92) 23 (96) 44 (90) CR 1(1) 0(0) 1(2) VGPR 30 (41) 7 (29) 23 (47) PR 29 (40) 14 (58) 15 (31) MR 7 (10) 2 (8) 5 (10) Study follow-up, 23.9 (4.4-45.7) 12.3 (5.9-28.0) 24.8 (4.4-45.7) median (range), mo Adverse Event Overview n (%) Event Patients with ≥1 AE Grade ≥3 40 (51.9) Patients with ≥1 serious AE 36a (46.8) AE leading to treatment discontinuation 8b (10.4) Fatal AE 5c (6.5) AE of special interest n(%): Petechiae/purpura/contusion 34 (44.2), Diarrhea 13 (16.9), Hypertension 9 (11.7), Major hemorrhage 2 (2.6), Atrial fibrillation/flutter 4 (5.2); Major hemorrhage defined as any grade ≥3 hemorrhage or any-grade central nervous system hemorrhage, gastrointestinal hemorrhage (n=1), grade 3 hemorrhagic cystitis (n=1). aIncludes serious AEs possibly related to zanubrutinib (n=6): hemothorax+pleural effusion+anemia (n=1), atrial fibrillation (n=1), colitis (n=1), febrile neutropenia (n=1), pneumonia (n=1), and cellulitis (n=1); septic arthritis relatedness was unknown. bAbdominal sepsis (fatal), septic arthritis (fatal), worsening bronchiectasis (fatal), gastric adenocarcinoma (fatal), prostate adenocarcinoma, metastatic neuroendocrine carcinoma, acute myeloid leukemia, and breast cancer (each n=1). cSeptic arthritis (patient also reported disease progression), worsening bronchiectasis, abdominal sepsis, gastric adenocarcinoma, and scedosporium infection (each n=1). 26
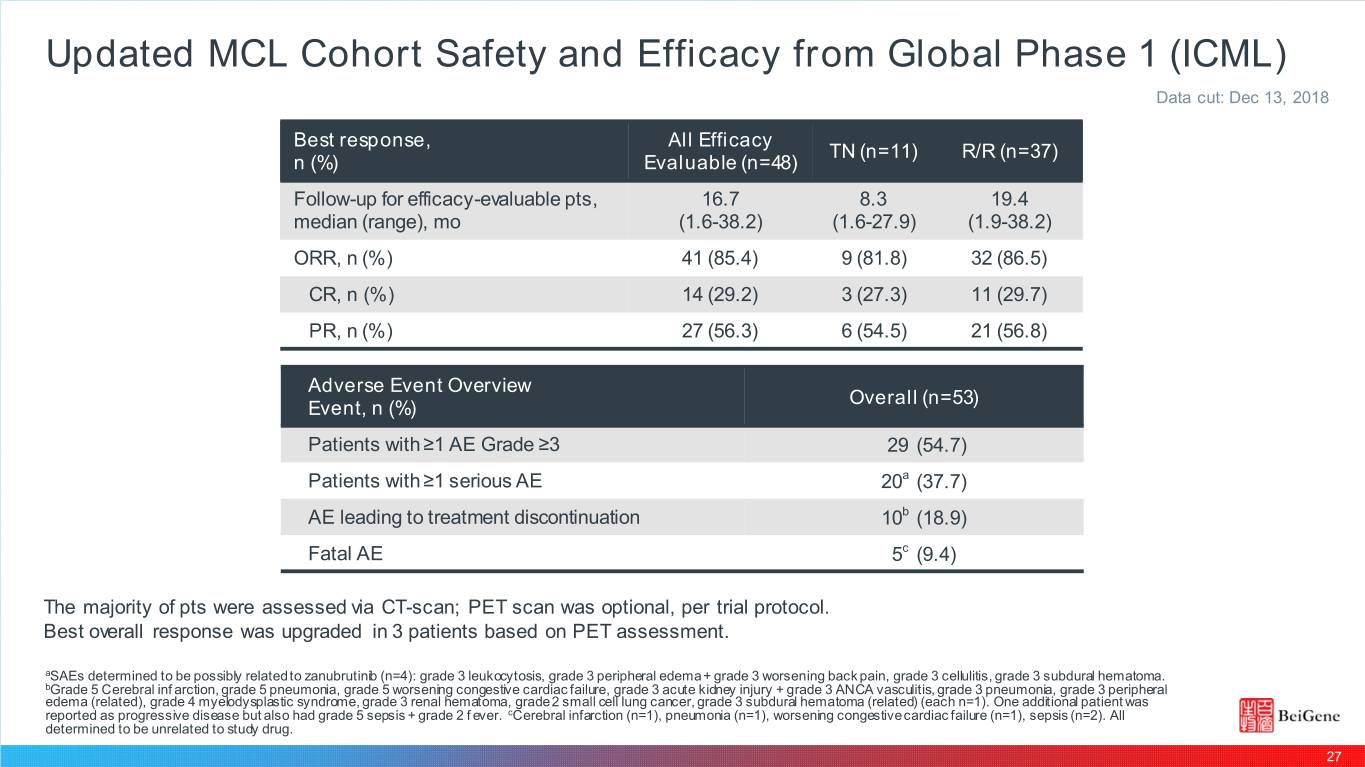
Updated MCL Cohort Safety and Efficacy from Global Phase 1 (ICML) Data cut: Dec 13, 2018 Be st re sponse , All Efficacy TN (n=11) R/R (n=37) n (%) Evaluable (n=48) Follow-up for efficacy-evaluable pts, 16.7 8.3 19.4 median (range), mo (1.6-38.2) (1.6-27.9) (1.9-38.2) ORR, n (%) 41 (85.4) 9 (81.8) 32 (86.5) CR, n (%) 14 (29.2) 3 (27.3) 11 (29.7) PR, n (%) 27 (56.3) 6 (54.5) 21 (56.8) Adverse Event Overview Overall (n=53) Event, n (%) Patients with ≥1 AE Grade ≥3 29 (54.7) Patients with ≥1 serious AE 20a (37.7) AE leading to treatment discontinuation 10b (18.9) Fatal AE 5c (9.4) The majority of pts were assessed via CT-scan; PET scan was optional, per trial protocol. Best overall response was upgraded in 3 patients based on PET assessment. aSAEs determined to be possibly related to zanubrutinib (n=4): grade 3 leukocytosis, grade 3 peripheral edema + grade 3 worsening back pain, grade 3 cellulitis, grade 3 subdural hematoma. bGrade 5 Cerebral inf arction, grade 5 pneumonia, grade 5 worsening congestive cardiac failure, grade 3 acute kidney injury + grade 3 ANCA vasculitis, grade 3 pneumonia, grade 3 peripheral edema (related), grade 4 myelodysplastic syndrome, grade 3 renal hematoma, grade 2 small cell lung cancer, grade 3 subdural hematoma (related) (each n=1). One additional patient was reported as progressive disease but also had grade 5 sepsis + grade 2 f ever. cCerebral infarction (n=1), pneumonia (n=1), worsening congestive cardiac failure (n=1), sepsis (n=2). All determined to be unrelated to study drug. 27

Updated Data from Zanubrutinib Phase 2 China Pivotal Study in R/R MCL (ICML) Data cut: Feb 15, 2019 Best response, n (%) Data cutoff Mar 2018 Data cutoff Feb n = 85a 15, 2019 n = 86 INV IRC INV ORR 72 (84.7) 71 (83.5) 72 (83.7) CR 62 (72.9) 50 (58.8) 67 (77.9) PR 10 (11.8) 21 (24.7) 5 (5.8) SD 1 (1.2) 2 (2.4) 1 (1.2) PD 8 (9.4) 6 (7.1) 8 (9.3) Discontinued prior to 4 (4.7) 5 (5.9) 5 (5.8) first assessment No evidence of disease - 1 (1.2) - Note: Only 4 patients were at risk at the last event time. Median follow-up, mo 9 9 18.4 Summary of TEAEs Regardless of Causality, n (%): Grade ≥3 TEAEs, 36 (41.9); Serious TEAEs, 21 (24.4); TEAEs leading to study drug discontinuation, 8 (9.3); TEAEs leading to death1 5 (5.8)2 {Death3 2 (2.3), Pneumonia 1 (1.2), Cerebral hemorrhage 1 (1.2), Traffic accident 1 (1.2)}. 1Death w ithin 30 days of last dose of zanubrutinib. 2Four events related, 1 event unrelated (traffic accident). 3One subject discontinued treatment due to disease progression prior to death. aThe ef f icacy report was based on modified safety population which excluded patient 20612006 who had local pathological diagnosis of MCL only but did not have confirmation of MCL by central rev iew. 28

Updated Data from Zanubrutinib in Combination with Obinutuzumab Remains Consistent in More Patients TN CLL/SLL R/R CLL/SLL R/R FL (n = 20) (n = 25) (n = 36) Follow-up median (range), mo 28.8 (13.9 - 34.8) 28.9 (7.9 – 36.9) 20.1 (2.3-37.2) Best response, n (%) ORR 20 (100.0) 23 (92.0) 26 (72.2) CR* 6 (30.0) 7 (28.0) 14 (38.9) PR 14 (70.0) 16 (64.0) 12 (33.3) SD 0 2 (8.0) 6 (16.7) PD 0 0 4 (11.1) ORR for Del(17p) or p53 6 (100) 8 (80) n/a Safety summary, n (%) for CLL/SLL (n = 45) FL (n=36) : Patients w ith any AE 45 (100.0) 35 (97.2); Patients w ith any treatment related AE 43 (95.6) 30 (83.3); Patients w ith ≥1 grade ≥3 AE 33 (73.3) 19 (52.8); Patients w ith AEs leading to treatment discontinuation 4 (8.9)a 3 (8.3)b. Patients w ith AE leading to death 1 (2.2%), squamous cell carcinoma in patient w ith a history of squamous cell carcinoma. aCLL/SLL: patient with a history of squamous cell carcinoma discontinued due to squamous cell carcinoma, disseminated cryptococcal infection, pneumonia, and neoplasm. bR/R FL: lethargy, ascites, and back pain. *3 out 6 tested in PB were MRD negative at <10-4. FL, f ollicular lymphoma; SD, stable disease. Source: Tam et al, ICML 2019 29

Tislelizumab Eric Hedrick, M.D., Chief Advisor

Tislelizumab Pivotal Phase 2 in R/R cHL (EHA) Abstract PF469 24th European Hematology Association Congress Eric Hedrick, M.D., Chief Advisor 31
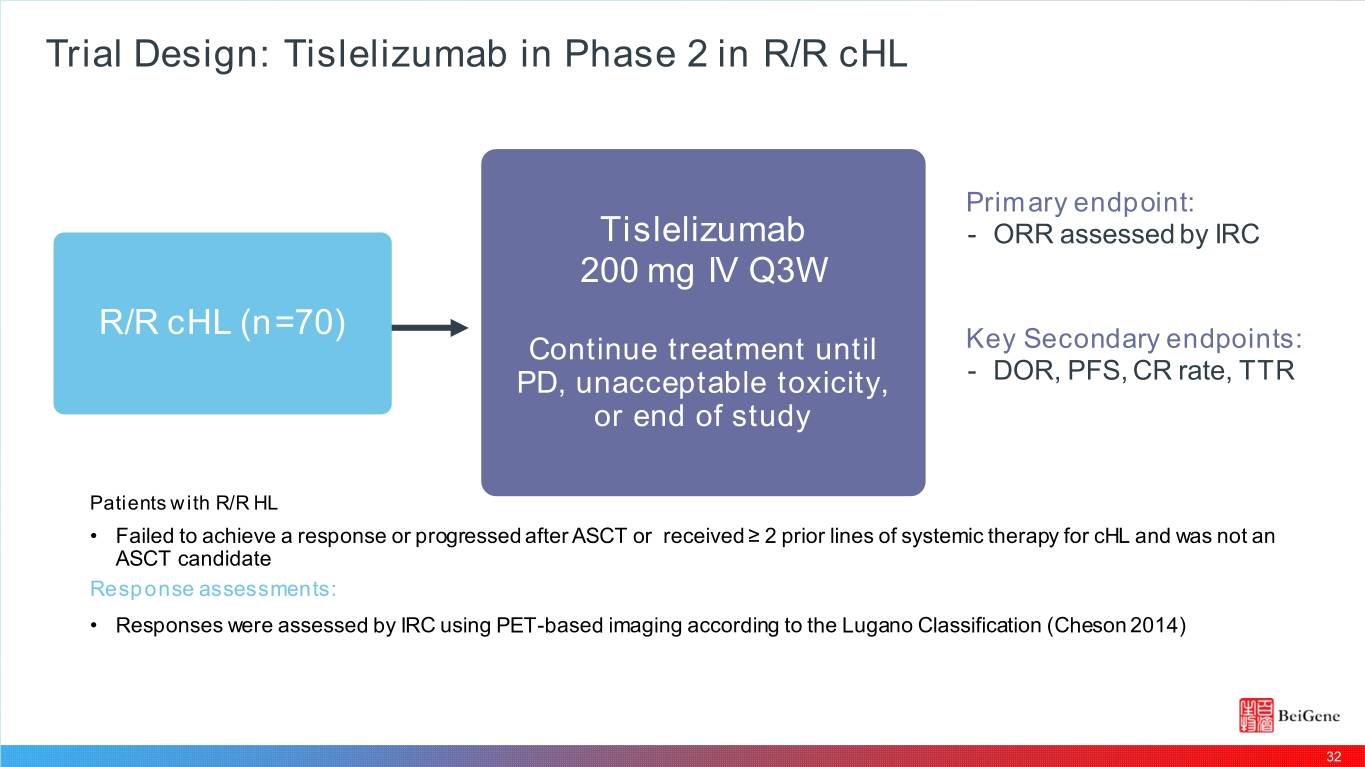
Trial Design: Tislelizumab in Phase 2 in R/R cHL Primary endpoint: Tislelizumab - ORR assessed by IRC 200 mg IV Q3W R/R cHL (n=70) Continue treatment until Key Secondary endpoints: PD, unacceptable toxicity, - DOR, PFS, CR rate, TTR or end of study Patients with R/R HL • Failed to achieve a response or progressed after ASCT or received ≥ 2 prior lines of systemic therapy for cHL and was not an ASCT candidate Response assessments: • Responses were assessed by IRC using PET-based imaging according to the Lugano Classification (Cheson 2014) 32

Patient and Disease Characteristics Data cut: Nov 26, 2018 Baseline Characteristics Total (n=70) Age (years), median (range) 32.5 (18, 69) Age group <65 / 65-74 years, n (%) 66 (94.3) / 4 (5.7) Sex, male / female, n (%) 40 (57.1) / 30 (42.9) Time since first diagnosis of cHL (months), median (range) 25.33 (4.6, 262.3) Stage IV at study entry, n (%) 42 (60.0) Bulky disease*, n (%) 8 (11.4) Bone marrow involvement, n (%) 22 (31.4) B-symptom(s), n (%) 26 (37.1) Ineligible for prior ASCT†, n (%) Failure to achieve an objective response to salvage chemotherapy 53 (75.7) Inadequate stem cell collection or unable to collect stem cells 2 (2.9) Co-morbidities 2 (2.9) Prior lines of systemic therapy, median (range) 3 (2-11) Type of prior therapy, n (%) Chemotherapy 70 (100.0) Radiotherapy 21 (30.0) ASCT 13 (18.6) Immunotherapy‡ 15 (21.4) Brentuximab vedotin 4 (5.7) *Mediastinal mass ratio of 0.33 or size of any single node/nodal mass ≥10 cm in diameter. †All receiv ed ≥ 2 prior regimens. ‡Immunotherapy included brentuximab vedotin, rituximab, cytokine-induced killer cell transfusion, thalidomide, and lenalidomide. 33
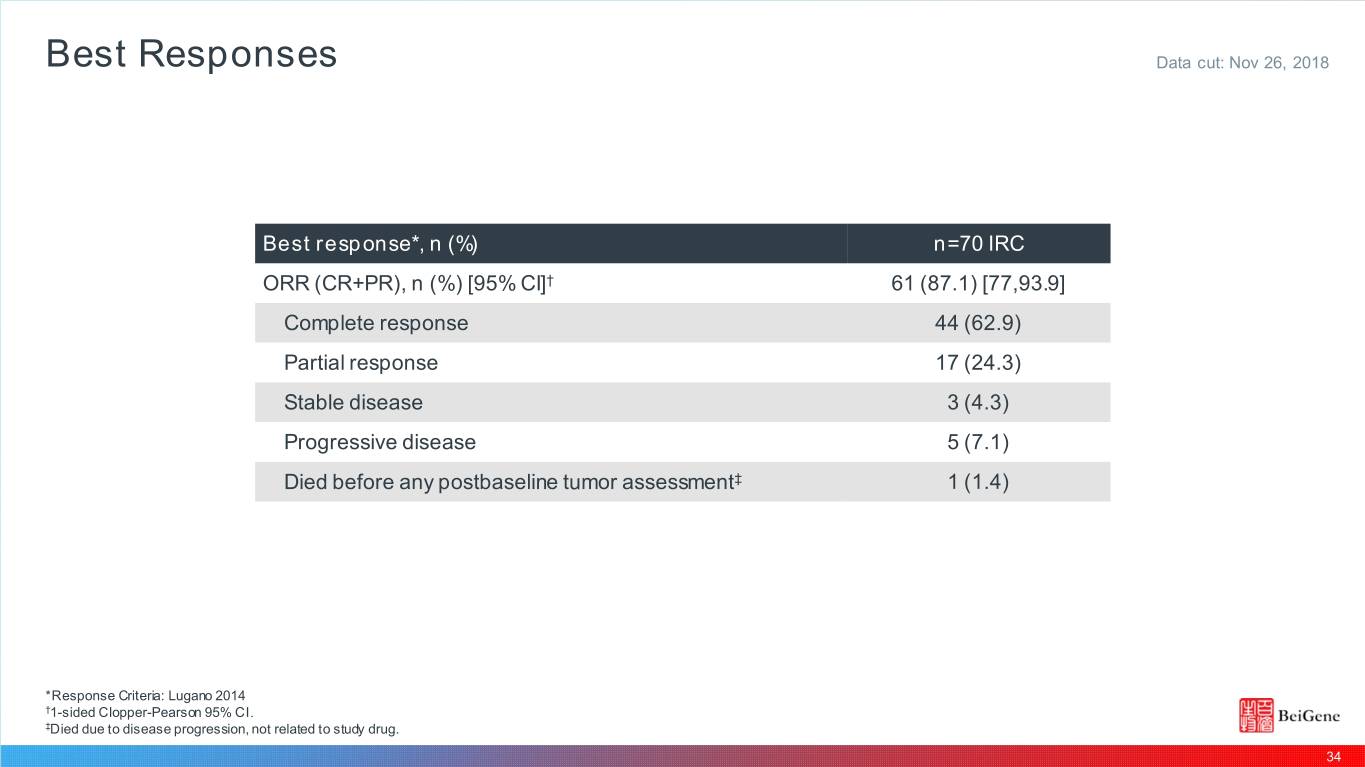
Best Responses Data cut: Nov 26, 2018 Be st re sponse*, n ( %) n=70 IRC ORR (CR+PR), n (%) [95% CI]† 61 (87.1) [77,93.9] Complete response 44 (62.9) Partial response 17 (24.3) Stable disease 3 (4.3) Progressive disease 5 (7.1) Died before any postbaseline tumor assessment‡ 1 (1.4) *Response Criteria: Lugano 2014 †1-sided Clopper-Pearson 95% CI. ‡Died due to disease progression, not related to study drug. 34

Summary of Adverse Events Data cut: Nov 26, 2018 Consistent type and frequency of immune-related AEs TEAE, n (%) n=70 Grade ≥3 TEAE 21 (30) TEAEs in ≥10% of Patients or Grade ≥3 TEAEs Serious TEAE 12* (17.1) in ≥2 Patients Regardless of Causality † TEAE leading to treatment discontinuation 4 (5.7) Pyrexia TEAE leading to death 0 (0.0) Hypothyroidism Immune-related (ir) TEAEs (by aggregate category) Weight increased Upper respiratory tract infection ≥1 irTEAE 27 (38.6) Cough Thyroid disorder 16 (22.9) Pru ritus Pneumonitis 5 (7.1) White blood cell count decreased Skin adverse reactions 6 (8.6) Alanine aminotransferase increased Myositis / rhabdomyolysis / Neutrophil count decreased ‡ 1 (1.4) Gr 1-2 cardiomyopathy Rash Diarrhea Gr ≥3 Nephritis and renal dysfunction 1 (1.4) Anemia Other immune-related reactions (lipase 1(1.4) Pneumonitis increased) 0% 20% 40% 60% Patients (%) *SAEs in all 11 patients determined to be possibly related to tislelizumab. †Pneumonitis (n = 2), f ocal segmental glomerulosclerosis (n = 1), organizing pneumonia (n = 1). ‡Blood creatine phosphokinase increased. TEAE, treatment-emergent adverse events by individual preferred term. 35

Progression-Free Survival – 91% of CRs Were Ongoing Data cut: Nov 26, 2018 40 of 44 CR’s and 10 of 17 PRs ongoing Figure out these numbers. 1.0 95.7% + Censored 82.1% 0.8 - Fr ee 0.6 0.4 Progression Survival Probability* 0.2 0.0 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 Time (months) At Risk 70 70 70 66 63 61 53 48 47 47 28 28 28 28 12 12 12 0 The majority of patients achieved a response by the first response assessment 36
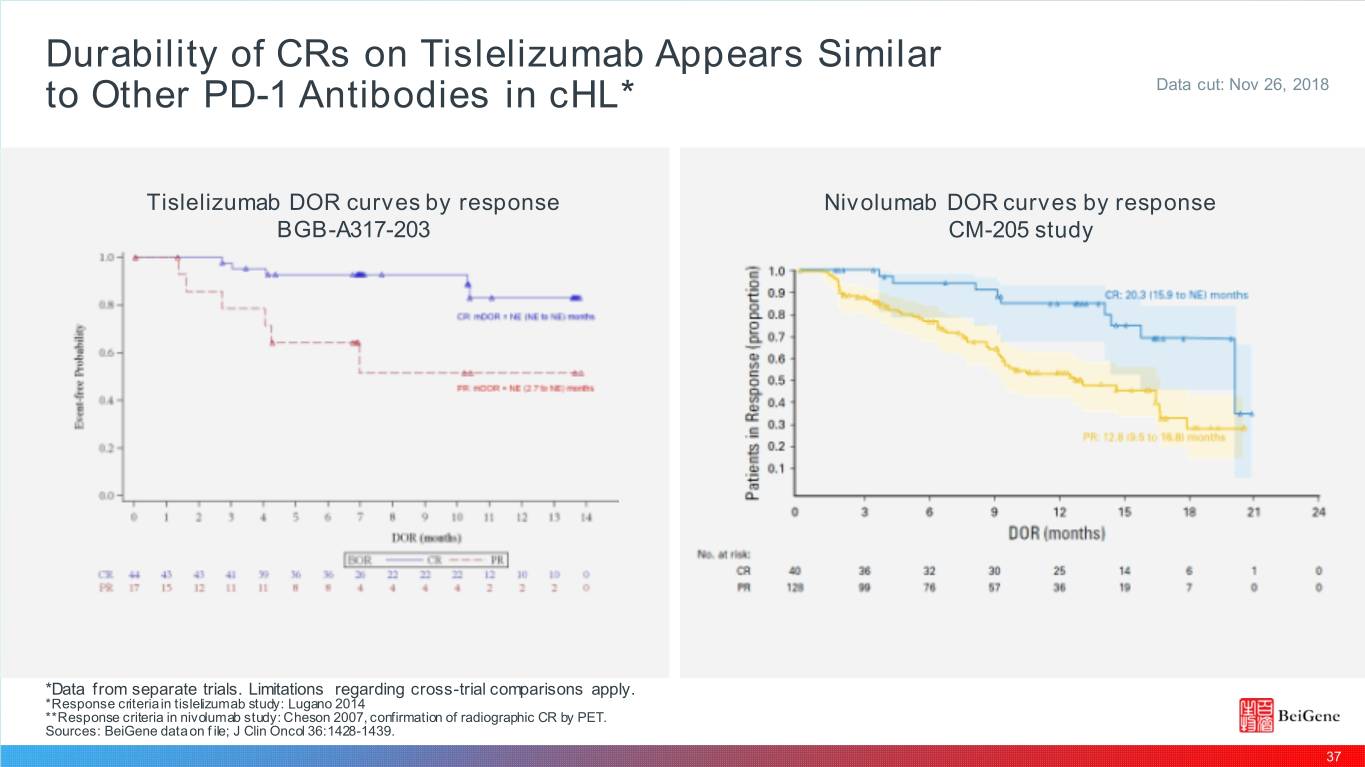
Durability of CRs on Tislelizumab Appears Similar to Other PD-1 Antibodies in cHL* Data cut: Nov 26, 2018 Tislelizumab DOR curves by response Nivolumab DOR curves by response BGB-A317-203 CM-205 study *Data from separate trials. Limitations regarding cross-trial comparisons apply. *Response criteria in tislelizumab study: Lugano 2014 **Response criteria in nivolumab study: Cheson 2007, confirmation of radiographic CR by PET. Sources: BeiGene data on f ile; J Clin Oncol 36:1428-1439. 37

Tislelizumab in NPC (ASCO) Abstract 2556 American Society of Clinical Oncology Eric Hedrick, M.D., Chief Advisor 38

Tislelizumab Nasopharyngeal Carcinoma Cohort from Global Phase 1 NPC (n=21) Preliminary Antitumor Activity Median age, years (min, max) 48 (35, 61) Male 17 (81) • A total of 21 patients were evaluable for antitumor Sex Female 4 (19) activity, defined as any patient who had measurable Prior anticancer radiotherapy 21 (100) disease at baseline and at least one postbaseline tumor assessment 0 1 (5) 1 6 (29) Prior anticancer therapy regimens • A total of nine patients (n=8, PD-L1+; n=1, PD-L1-) 2 4 (19) achieved a confirmed PR, nine patients (n=6, PD-L1+; ≥3 10 (48) n=2, PD-L1-; n=1, unknown) achieved confirmed SD 0 8 (38) ECOG status 1 13 (62) • Confirmed ORR was 43% (95% confidence interval Poorly differentiated 2 (10) [CI]: 21.8-66.0) Histologic grade Undifferentiated 16 (76) • CBR and DCR were 62% (95% CI: 38.4-81.9) Unknow n 3 (14) and 86% (95% CI: 63.7-97.0), respectively Locally advanced 3 (14) Tumor stage • Median duration of response was 8.3 months (95% CI: Metastatic 18 (86) 3.9, not reached); follow-up time for responders was PD-L1 positive (PD-L1+)* 16 (76) 4.8 months (95% CI: 2.1-11.1) PD-L1 status PD-L1 negative (PD-L1-)* 4 (19) Unknow n 1 (5) Data presented as n (%) except for age. *PD-L1-positive status defined as ≥10% of tumor cells with PD-L1 membrane staining, as retrospectively assessed by central lab; †PD-L1-negative status defined as <10% of tumor cells with PD- L1 membrane staining, as retrospectively assessed by central lab. NPC, nasopharyngeal carcinoma; PD-L1, programmed cell death ligand-1. 39
Key Takeaways Eric Hedrick, M.D., Chief Advisor 40
Zanubrutinib Key Takeaways • Results from non-randomized MYD88WT cohort of Phase 3 trial in WM confirmed activity of zanubrutinib in this difficult- to-treat population, with ORR and VGPR rates consistent with results from MYD88WT patients treated in Phase 1 • First presentation of pivotal Phase 2 data in Chinese patients with relapsed or refractory CLL demonstrated efficacy consistent with the experience with zanubrutinib in Western CLL patients • Updated integrated safety analysis of data from 682 patients with a median exposure of 13.4 months continues to show low rates of atrial fibrillation, serious bleeding, and diarrhea • Updated pivotal China R/R MCL data demonstrated high CR rate • Zanubrutinib / GAZYVA combination appears highly active in FL and supports ongoing pivotal Phase 2 trial 41

Tislelizumab Key Takeaways • Follow-up from the Phase 2 study of tislelizumab in Chinese patients with classical Hodgkin’s lymphoma confirms a high complete response rate and encouraging durability of complete responses • ASCO data show encouraging activity of tislelizumab in nasopharyngeal cancer and supports ongoing Phase 3 • Tislelizumab is under CDE review for approval in cHL and urothelial cancer in China, and the broad Phase 3 program is maturing, with initial read-out in HCC and completion of enrollment to multiple trials in 2019 42
Q&A 43

Thank You 44
Backup 45

PD-1 Inhibitor Data in R/R cHL* Pembrolizumab and Nivolumab Pembrolizumab1 Nivolumab2 Company Mer c k BMS n 210 243 A SCT-ineligible OR A SCT-failure Eligibility A SCT-failure Prior brentuximab vedotina Prior brentuximab vedotina Prior Lines, med (range) 4 (1-12) 4 (2-15) Prior therapy A SCT 129 (61%) 243 (100%) Brentuximab Vedotin 150 (71%) 180 (74%) Follow -up (med) 15.9 months 18 months Response Criteria Cheson 2007 Lugano 2014 Cheson 2007 ORR 71% 73% 69% CR 25% 31% 16% PR 47% 42% 53% SD 12% 8% 19% * Data from separate trials. Limitations regarding cross-trial comparisons apply. Sources: 1 Blood 2017 130:4085; 2 JCO 2018 a Prior brentuximab vedotin required for 2 of 3 study cohorts 46

Reported PD-1 Inhibitor Data in R/R cHL* Sintilimaband Camrelizumab Sintilimab1 Camrelizumab2 Company Innovent Hengrui n 96 66 Eligibility > 2 prior lines of therapya > 2 prior lines of therapya Prior Lines, med (range) 3 (1-13) 3 (2-10) Prior therapy A SCT 18 (19%) 9 (14%) Brentuximab Vedotin NR 5 (8%) Follow -up (med) 14 >6 Response Criteria Cheson 2007 Lugano 2014 ORR 85% 85% CR 29% 30% PR 56% 54% SD 13% 12% * Data from separate trials. Limitations regarding cross-trial comparisons apply. Sources: 1 ASCO 2019 (Abstract 7533); 2 CSCO 2018 a ineligibility for ASCT was not required 47
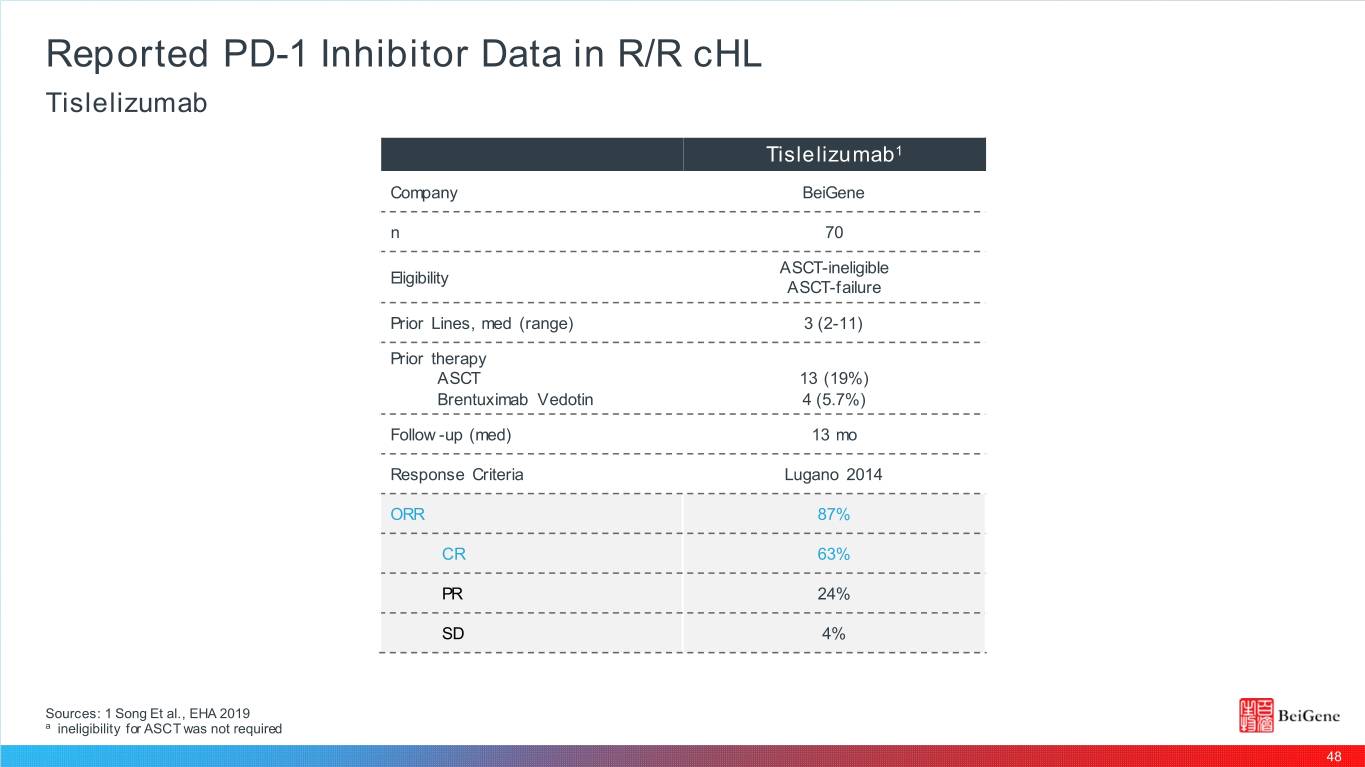
Reported PD-1 Inhibitor Data in R/R cHL Tislelizumab Tislelizumab1 Company BeiGene n 70 A SCT-ineligible Eligibility A SCT-failure Prior Lines, med (range) 3 (2-11) Prior therapy A SCT 13 (19%) Brentuximab Vedotin 4 (5.7%) Follow -up (med) 13 mo Response Criteria Lugano 2014 ORR 87% CR 63% PR 24% SD 4% Sources: 1 Song Et al., EHA 2019 a ineligibility for ASCT was not required 48