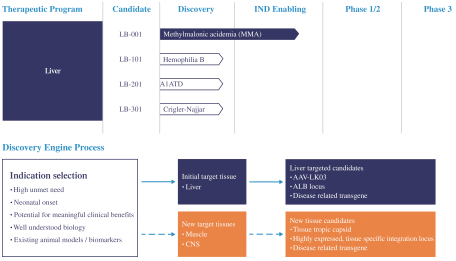
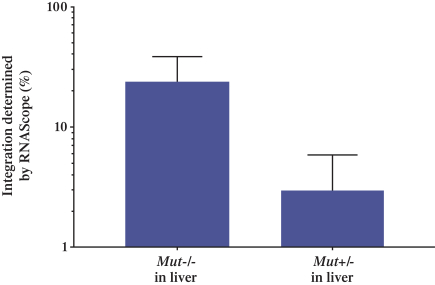
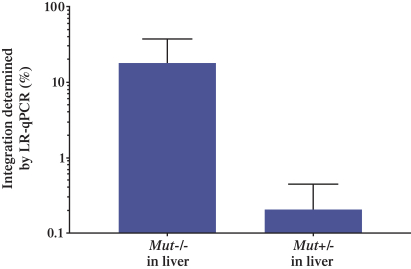
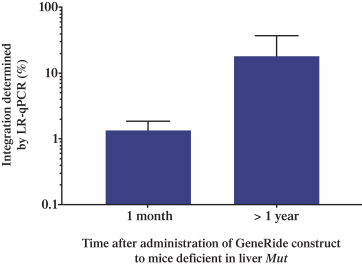
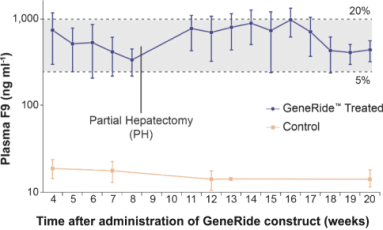
patent applications may not result in issued patents which protect our business, in whole or in part, or which effectively prevent others from commercializing competitive products. Changes in either the patent laws or their interpretation in the United States and other countries may diminish our ability to protect our inventions, obtain, maintain, and enforce our intellectual property rights and, more generally, could affect the value of our intellectual property or narrow the scope of our owned and licensed patents. With respect to bothin-licensed and owned intellectual property, we cannot predict whether the patent applications we and our licensors are currently pursuing will issue as patents in any particular jurisdiction or whether the claims of any issued patents will provide sufficient protection from competitors. In addition, the laws of foreign countries may not protect our rights to the same extent or in the same manner as the laws of the United States.
The patent prosecution process is expensive, time-consuming, and complex. We may not be able to file, prosecute, maintain, enforce, or license all necessary or desirable patent applications at a reasonable cost or in a timely manner. It is also possible that we, or any future partners, collaborators or licensees, may fail to identify patentable aspects of inventions made in the course of development and commercialization activities in time to obtain patent protection. Therefore, we may miss potential opportunities to strengthen our patent position. Additionally, although we enter into agreements containingnon-disclosure and confidentiality obligations with parties who have access to confidential or patentable aspects of our research and development output, such as our employees, collaborators, contract manufacturers, consultants, and other third parties, any of these parties may breach the agreements and disclose such output before a patent application is filed, thereby jeopardizing our ability to seek patent protection.
It is possible that defects of form in the preparation or filing of our patents or patent applications may exist, or may arise in the future, for example with respect to proper priority claims, inventorship, claim scope or patent term adjustments. If we or our partners, collaborators, licensees, or licensors, whether current or future, fail to establish, maintain, or protect such patents and other intellectual property rights, such rights may be reduced or eliminated. If our partners, collaborators, licensees, or licensors are not fully cooperative or disagree with us as to the prosecution, maintenance, or enforcement of any patent rights, such patent rights could be compromised. If there are material defects in the form, preparation, prosecution, or enforcement of our patents or patent applications, such patents may be invalid and/or unenforceable, and such applications may never result in valid, enforceable patents. Any of these outcomes could impair our ability to prevent competition from third parties, which may have an adverse impact on our business.
Pending patent applications cannot be enforced against third parties practicing the technology claimed in such applications unless and until a patent issues from such applications. Assuming the other requirements for patentability are met, currently, the first to file a patent application is generally entitled to the patent. However, prior to March 16, 2013, in the United States, the first to invent was entitled to the patent. Publications of discoveries in the scientific literature often lag behind the actual discoveries, and patent applications in the United States and other jurisdictions are typically not published until 18 months after filing, or in some cases, not at all. Therefore, we cannot be certain that we were the first to make the inventions that will be claimed in our future patents or future patent applications, or that we will be the first to file for patent protection of such inventions. Similarly, we cannot be certain that parties from whom we do or may license or purchase patent rights were the first to make relevant claimed inventions, or were the first to file for patent protection for them. If third parties have filed patent applications on inventions claimed in our patents or applications on or before March 15, 2013, an interference proceeding in the United States can be initiated by such third parties to determine who was the first to invent any of the subject matter covered by the patent claims of our applications. If third parties have filed such applications after March 15, 2013, a derivation proceeding in the United States can be initiated by such third parties to determine whether our invention was derived from theirs.
The patent position of biotechnology and pharmaceutical companies generally is highly uncertain, involves complex legal and factual questions, and has been the subject of much litigation in recent years. No consistent policy regarding the breadth of claims allowed in biotechnology and pharmaceutical patents has emerged to date in the United States or in many foreign jurisdictions. As a result, the issuance, scope, validity, enforceability, and
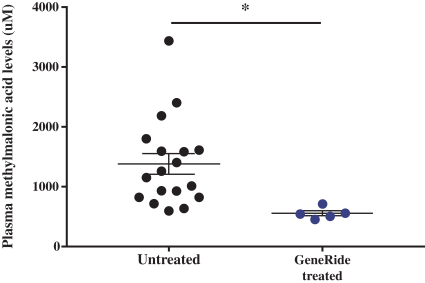
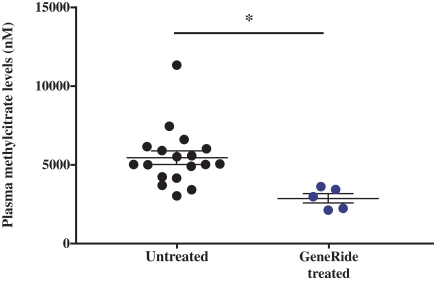
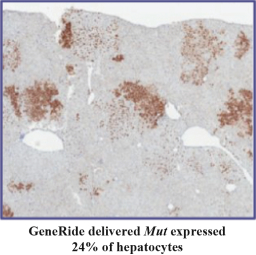
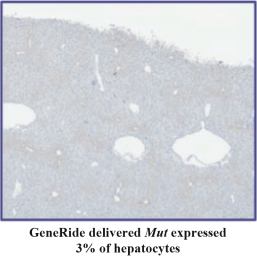
76