Free signup for more
- Track your favorite companies
- Receive email alerts for new filings
- Personalized dashboard of news and more
- Access all data and search results
Filing tables
Filing exhibits
IMCR similar filings
- 8 May 24 Immunocore reports first quarter financial results and provides a business update
- 5 Apr 24 Other Events
- 28 Feb 24 Immunocore reports fourth quarter and full year 2023 financial results and provides a business update
- 2 Feb 24 Immunocore Prices Upsized Convertible Senior Notes Offering
- 29 Jan 24 Immunocore Announces Proposed Convertible Senior Notes Offering
- 5 Jan 24 Immunocore announces strategic priorities and pipeline expansion ahead of 42nd Annual J.P. Morgan Healthcare Conference presentation
Filing view
External links
Exhibit 99.2

Transformative immunomodulating medicines for patients 4Q 2023 & FY 2023 Financial Results & Business Update

This presentation contains “forward-looking statements” within the meaning of the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. Words such as “may”, “will”, “believe”, “expect”, “plan”, “anticipate” and similar expressions (as well as other words or expressions referencing future events or circumstances) are intended to identify forward-looking statements. All statements, other than statements of historical facts, included in this presentation are forward-looking statements. These statements include, but are not limited to, Immunocore’s capabilities across oncology, autoimmune and infectious disease therapeutic areas and its ability to grow, maximize and further develop the KIMMTRAK platform, and advance the clinical and pre-clinical programs, including the PRAME franchise, PIWIL 1 target other programs; the estimated market size and patient population for KIMMTRAK and Immunocore’s other product candidates; the three potential growth areas of KIMMTRAK including HLA-A02+ melanoma, metastatic cutaneous melanoma and adjuvant uveal melanoma; the outlook for 2024 and growth drivers for the commercial performance of KIMMTRAK including the momentum of KIMMTRAK in the United States, planned launches in additional countries, expanded access to KIMMTRAK in the United States and globally, early patient identification and indication expansion; expected submission of investigational new drug applications or clinical trial applications; the potential regulatory approval, expected clinical benefits and availability of Immunocore’s product candidates; the ability to enter into pricing agreements and to translate such pricing agreement into a successful launch; the accrual assumptions regarding the outcome of price negotiations in France; the potential benefits and advantages KIMMTRAK and Immunocore’s other product candidates will provide for patients; expectations regarding the design, progress, timing, enrollment, scope, expansion, and results of Immunocore’s existing and planned clinical trials, those of Immunocore’s collaboration partners or the combined clinical trials with Immunocore’s collaboration partners; the timing and sufficiency of clinical trial outcomes to support potential approval of any of Immunocore’s product candidates or those of, or combined with, its collaboration partners; Immunocore’s goals to develop and commercialize product candidates based on its KIMMTRAK platform alone or with collaboration partners; Immunocore’s vision for the clinical benefit of its ImmTAAI platform; and Immunocore’s expectations regarding the use of its cash and cash equivalents, including the net proceeds from the convertible note financing. Any forward- looking statements are based on management’s current expectations and beliefs of future events and are subject to a number of risks and uncertainties that could cause actual events or results to differ materially and adversely from those set forth in or implied by such forward-looking statements, many of which are beyond Immunocore’s control. These risks and uncertainties include, but are not limited to, the impact of worsening macroeconomic conditions on Immunocore’s business, financial position, strategy and anticipated milestones, including Immunocore’s ability to conduct ongoing and planned clinical trials; Immunocore’s ability to obtain a clinical supply of current or future product candidates or commercial supply of KIMMTRAK or any future approved products, including as a result of health epidemics or pandemic, war in Ukraine, the conflict between Hamas and Israel, or global geopolitical tension; Immunocore’s ability to obtain and maintain regulatory approval of its product candidates, including KIMMTRAK; Immunocore’s ability and plans in continuing to establish and expand a commercial infrastructure and to successfully launch, market and sell KIMMTRAK and any future approved products; Immunocore’s ability to successfully expand the approved indications for KIMMTRAK or obtain marketing approval for KIMMTRAK in additional geographies in the future; the delay of any current or planned clinical trials, whether due to patient enrollment delays or otherwise; Immunocore’s ability to successfully demonstrate the safety and efficacy of its product candidates and gain approval of its product candidates on a timely basis, if at all; competition with respect to market opportunities; unexpected safety or efficacy data observed during preclinical studies or clinical trials; actions of regulatory agencies, which may affect the initiation, timing and progress of clinical trials or future regulatory approval; Immunocore’s need for and ability to obtain additional funding, on favorable terms or at all, including as a result of worsening macroeconomic conditions, including changes inflation and interest rates and unfavorable general market conditions, and the impacts thereon of the war in Ukraine, the conflict between Hamas and Israel, and global geopolitical tension; Immunocore’s ability to obtain, maintain and enforce intellectual property protection for KIMMTRAK or any product candidates it is developing; and the success of Immunocore’s current and future collaborations, partnerships or licensing arrangements. These and other risks and uncertainties are described in greater detail in the section titled "Risk Factors" in Immunocore’s filings with the Securities and Exchange Commission, including Immunocore’s most recent Annual Report on Form 10- K for the year ended December 31, 2023 filed with the Securities and Exchange Commission on February 28, 2024, as well as discussions of potential risks, uncertainties, and other important factors in Immunocore’s subsequent filings with the Securities and Exchange Commission. All forward looking statements contained in this presentation speak only as of the date on which they were made and should not be relied upon as representing its views as of any subsequent date. Except to the extent required by law, Immunocore undertakes no obligation to update such statements to reflect events that occur or circumstances that exist after the date on which they were made. Certain information contained in this presentation relates to or is based on studies, publications, surveys, and other data obtained from third party sources and Immunocore’s own internal estimates and research. While Immunocore believes these third party sources to be reliable as of the date of this presentation, it has not independently verified, and makes no representation as to the adequacy, fairness, accuracy, or completeness of, any information obtained from third party sources. KIMMTRAK™ is a trademark owned or licensed to Immunocore. 2 Forward Looking Statements

Agenda Overview & 2023 highlights Bahija Jallal, CEO Maximizing potential of KIMMTRAK® Ralph Torbay, Head of Commercial Advancing clinical portfolio & innovating for sustainable growth David Berman, Head of R&D Delivering on our promise Bahija Jallal, CEO Q&A Session 3 4Q23 & FY23 financial results Brian Di Donato, CFO & Head of Strategy

4 We want to radically improve outcomes for patients with cancer, infectious diseases, and autoimmune diseases, by pioneering and delivering transformative immunomodulating medicines
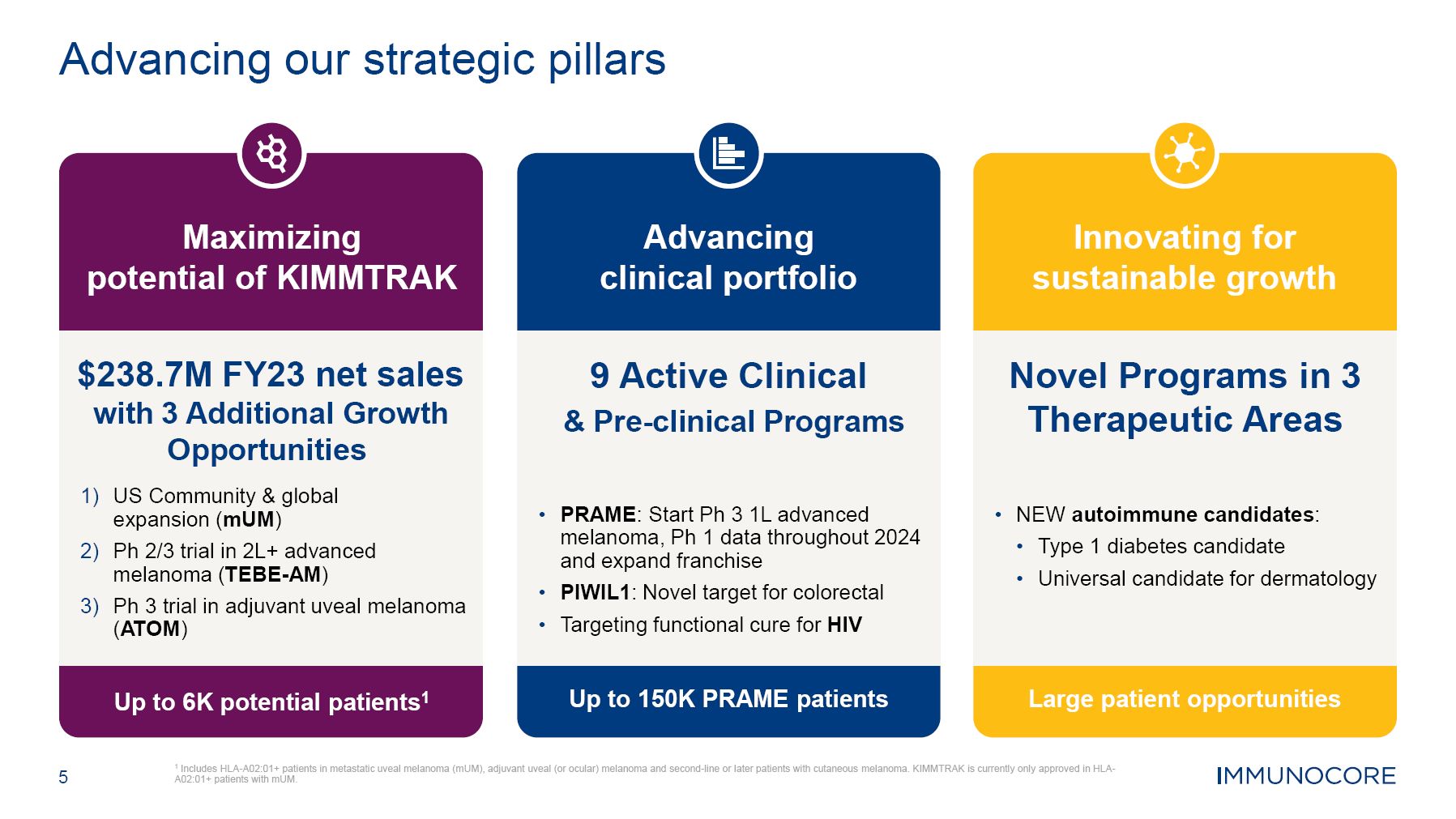
$238.7M FY23 net sales with 3 Additional Growth Opportunities US Community & global expansion (mUM) Ph 2/3 trial in 2L+ advanced melanoma (TEBE-AM) Ph 3 trial in adjuvant uveal melanoma (ATOM) Maximizing potential of KIMMTRAK Up to 6K potential patients1 9 Active Clinical & Pre-clinical Programs PRAME: Start Ph 3 1L advanced melanoma, Ph 1 data throughout 2024 and expand franchise PIWIL1: Novel target for colorectal Targeting functional cure for HIV Advancing clinical portfolio Up to 150K PRAME patients Novel Programs in 3 Therapeutic Areas NEW autoimmune candidates: Type 1 diabetes candidate Universal candidate for dermatology Innovating for sustainable growth Large patient opportunities 5 1 Includes HLA-A02:01+ patients in metastatic uveal melanoma (mUM), adjuvant uveal (or ocular) melanoma and second-line or later patients with cutaneous melanoma. KIMMTRAK is currently only approved in HLA- A02:01+ patients with mUM. Advancing our strategic pillars

Delivering across our three strategic pillars Ralph Torbay Head of Commercial Maximizing potential of KIMMTRAK Innovating for sustainable growth Advancing clinical portfolio 6
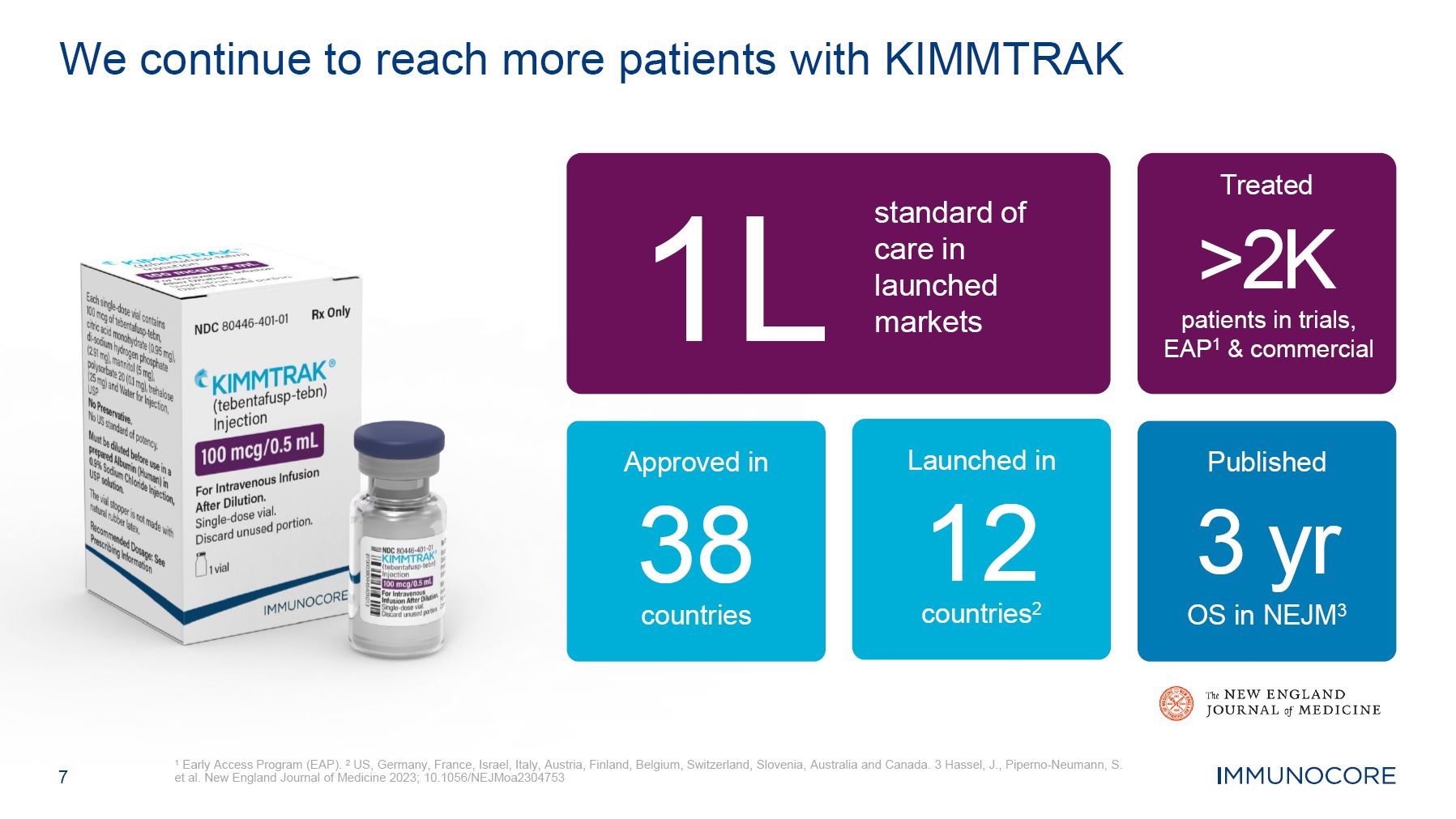
7 1 Early Access Program (EAP). 2 US, Germany, France, Israel, Italy, Austria, Finland, Belgium, Switzerland, Slovenia, Australia and Canada. 3 Hassel, J., Piperno-Neumann, S. et al. New England Journal of Medicine 2023; 10.1056/NEJMoa2304753 We continue to reach more patients with KIMMTRAK 1L standard of care in launched markets Approved in 38 countries Launched in 12 countries2 Treated >2K patients in trials, EAP1 & commercial Published 3 yr OS in NEJM3
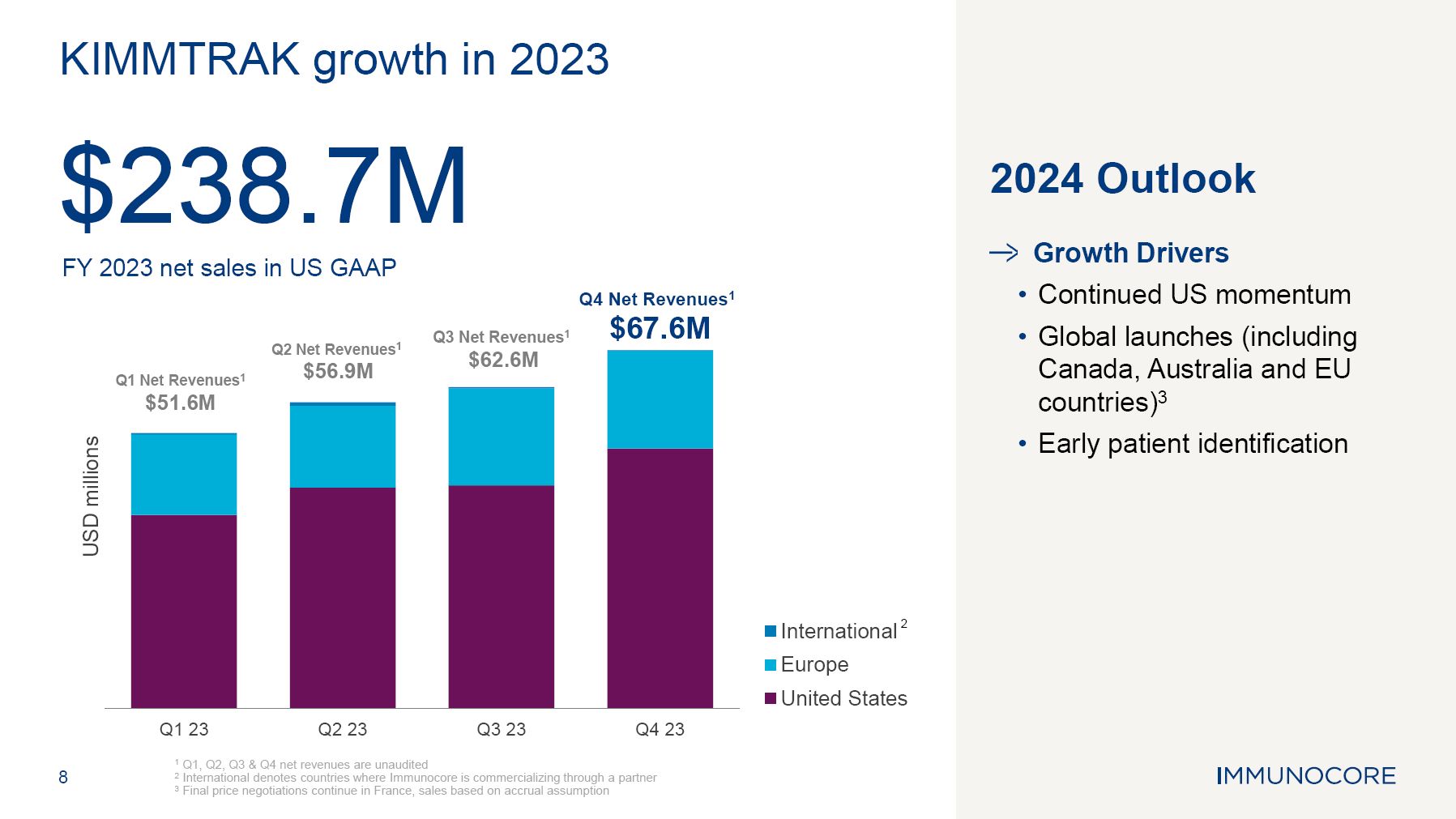
USD millions Q1 Net Revenues1 $51.6M Q2 Net Revenues1 $56.9M Q3 Net Revenues1 $62.6M Q4 Net Revenues1 $67.6M 8 Q1 23 Q2 23 Q3 23 Q4 23 1 Q1, Q2, Q3 & Q4 net revenues are unaudited 2 International denotes countries where Immunocore is commercializing through a partner 3 Final price negotiations continue in France, sales based on accrual assumption KIMMTRAK growth in 2023 $238.7M FY 2023 net sales in US GAAP 2024 Outlook Growth Drivers Continued US momentum Global launches (including Canada, Australia and EU countries)3 Early patient identification International 2 Europe United States
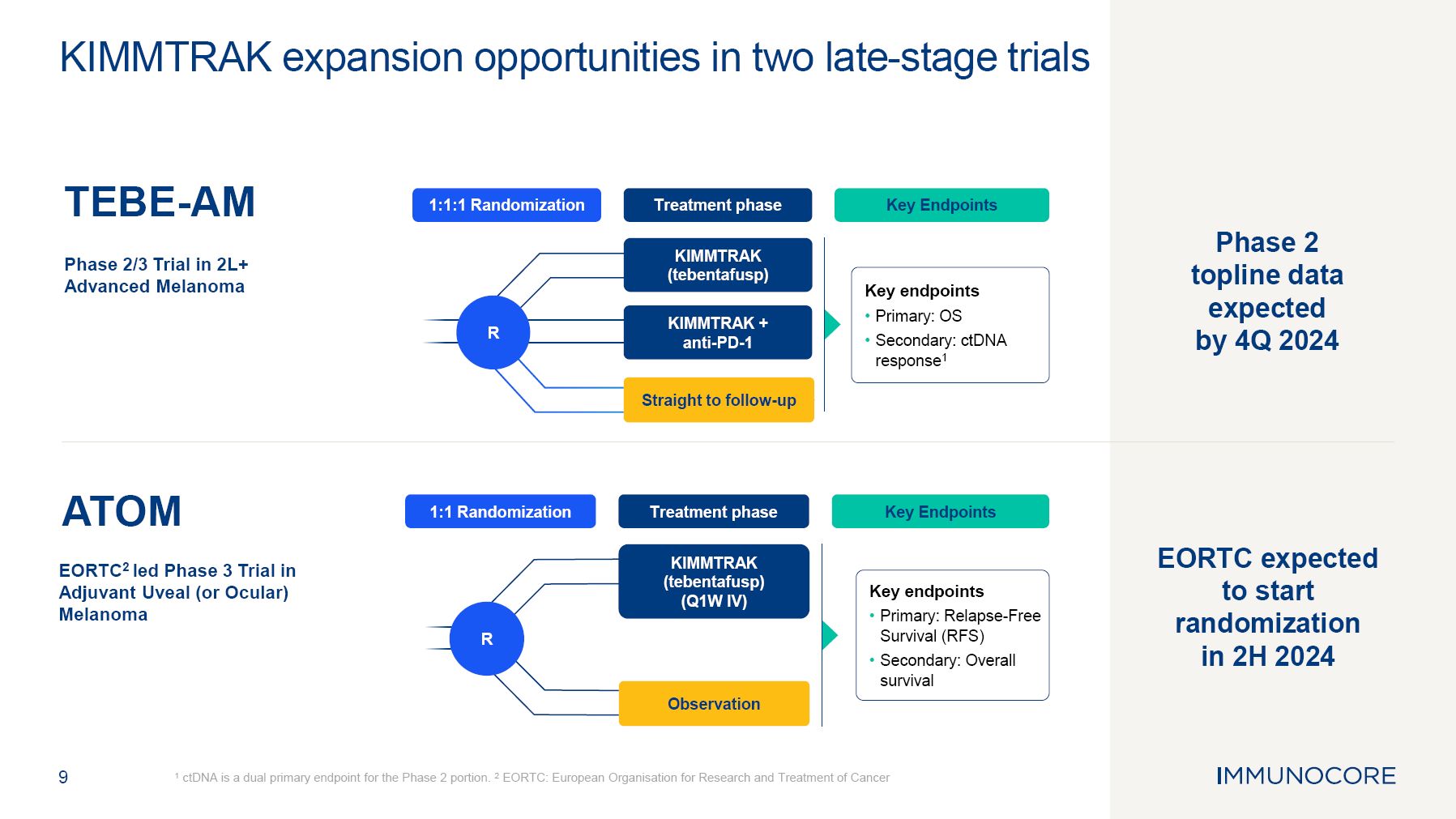
9 1 ctDNA is a dual primary endpoint for the Phase 2 portion. 2 EORTC: European Organisation for Research and Treatment of Cancer KIMMTRAK expansion opportunities in two late-stage trials Phase 2 topline data expected by 4Q 2024 Key endpoints Primary: Relapse-Free Survival (RFS) Secondary: Overall survival Treatment phase 1:1 Randomization KIMMTRAK (tebentafusp) (Q1W IV) Observation R Key Endpoints EORTC expected to start randomization in 2H 2024 Treatment phase 1:1:1 Randomization KIMMTRAK (tebentafusp) KIMMTRAK + anti-PD-1 R Key Endpoints Key endpoints Primary: OS Secondary: ctDNA response1 Straight to follow-up TEBE-AM Phase 2/3 Trial in 2L+ Advanced Melanoma ATOM EORTC2 led Phase 3 Trial in Adjuvant Uveal (or Ocular) Melanoma
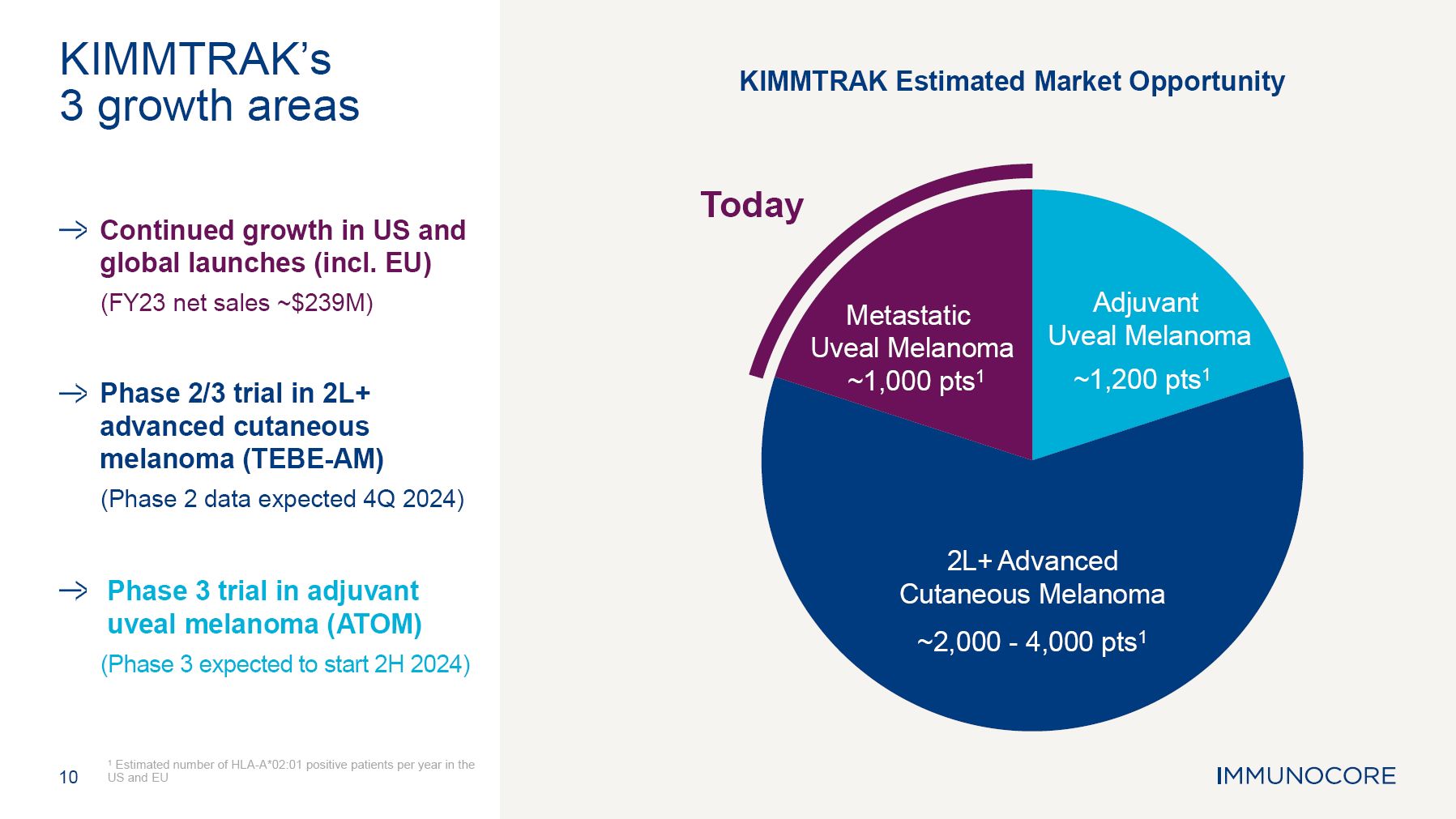
10 1 Estimated number of HLA-A*02:01 positive patients per year in the US and EU KIMMTRAK’s 3 growth areas Continued growth in US and global launches (incl. EU) (FY23 net sales ~$239M) Phase 2/3 trial in 2L+ advanced cutaneous melanoma (TEBE-AM) (Phase 2 data expected 4Q 2024) Phase 3 trial in adjuvant uveal melanoma (ATOM) (Phase 3 expected to start 2H 2024) Today Adjuvant Uveal Melanoma ~1,200 pts1 2L+ Advanced Cutaneous Melanoma ~2,000 - 4,000 pts1 Metastatic Uveal Melanoma ~1,000 pts1 KIMMTRAK Estimated Market Opportunity

Delivering across our three strategic pillars David Berman Head of Research & Development Innovating for sustainable growth Maximizing potential of KIMMTRAK Advancing clinical portfolio 11
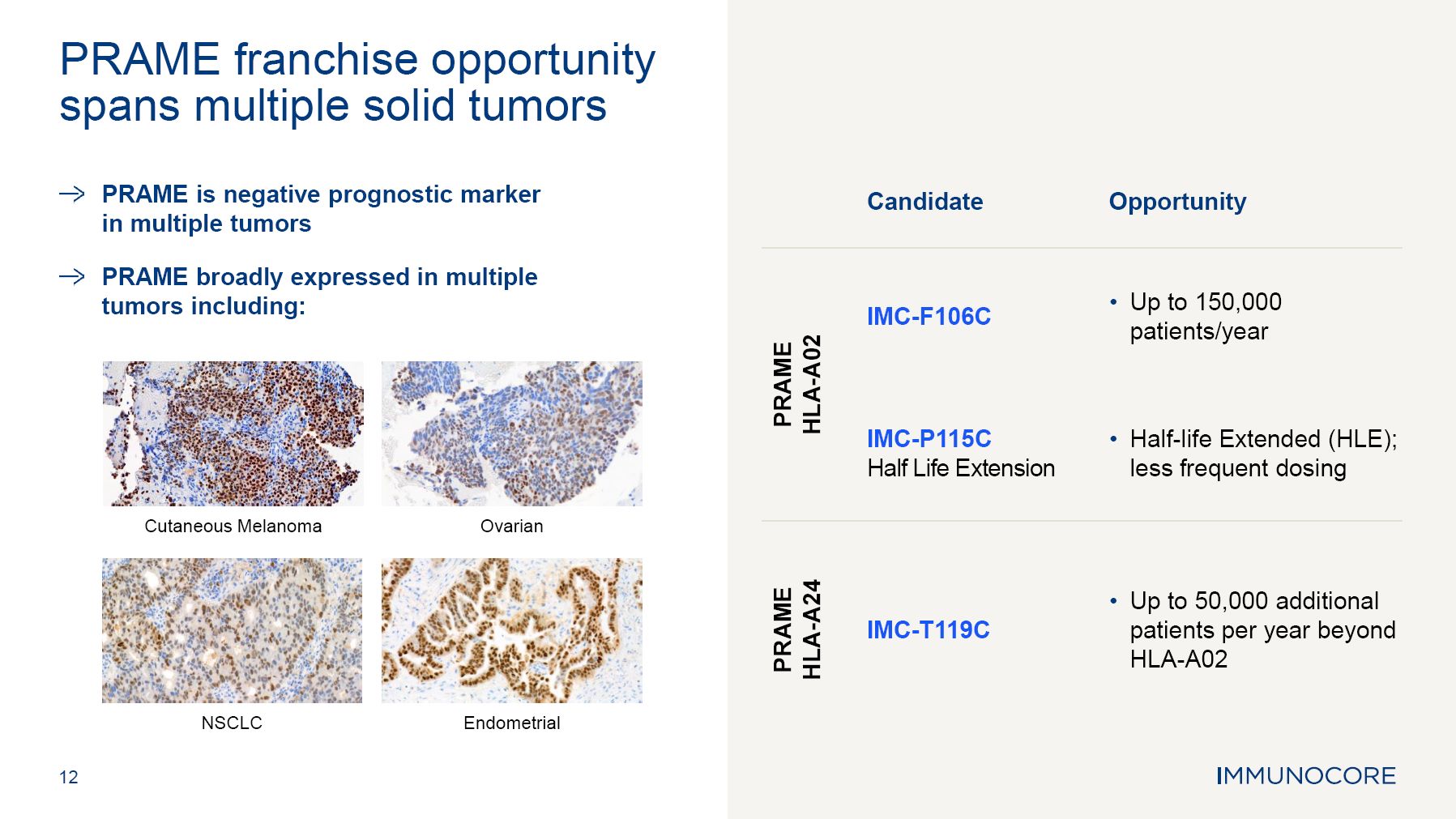
PRAME is negative prognostic marker in multiple tumors PRAME broadly expressed in multiple tumors including: 12 PRAME franchise opportunity spans multiple solid tumors Cutaneous Melanoma NSCLC Ovarian Endometrial Candidate Opportunity IMC-F106C Up to 150,000 patients/year PRAME HLA-A02 IMC-P115C Half Life Extension Half-life Extended (HLE); less frequent dosing PRAME HLA-A24 IMC-T119C Up to 50,000 additional patients per year beyond HLA-A02
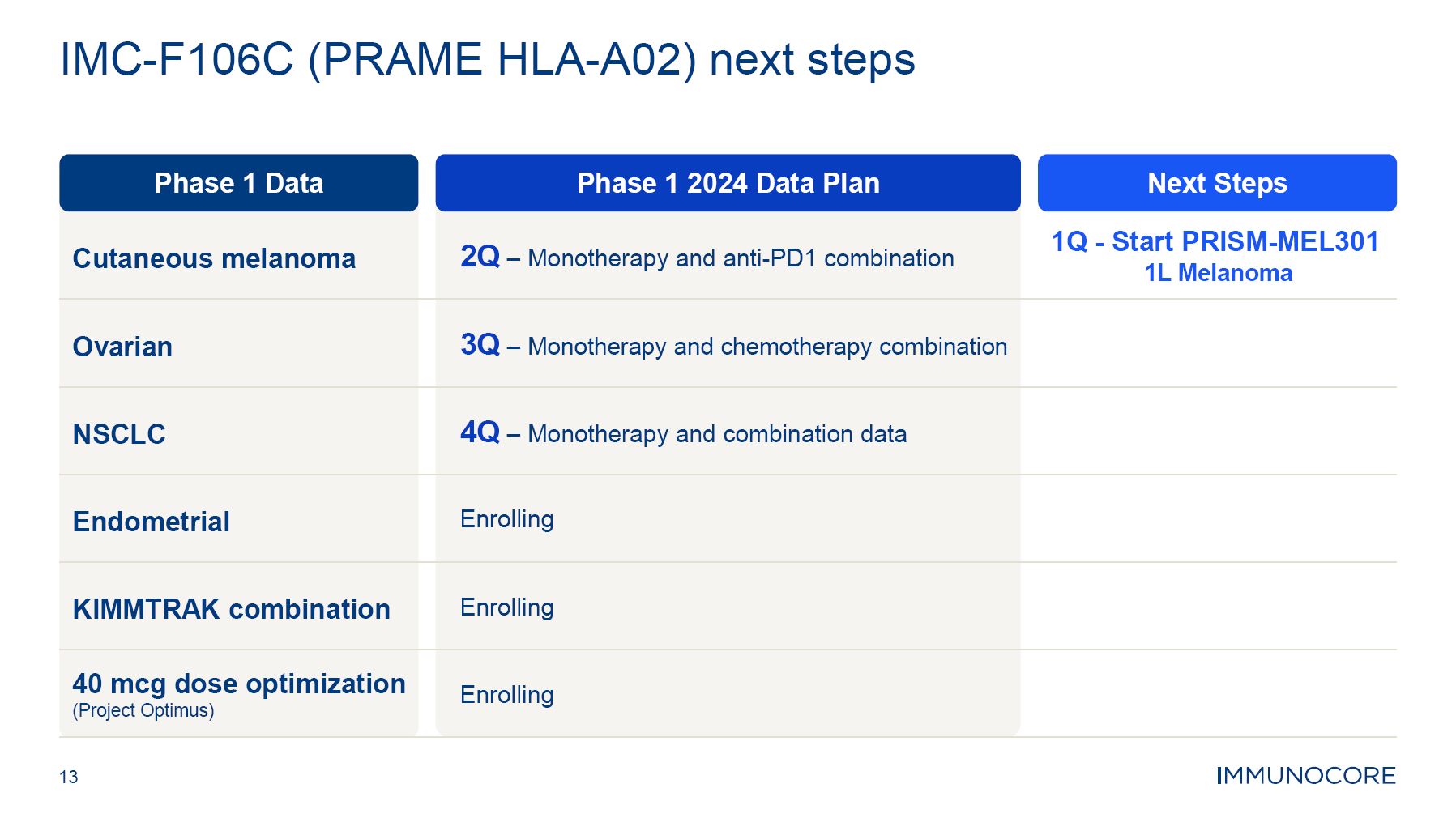
13 IMC-F106C (PRAME HLA-A02) next steps Phase 1 2024 Data Plan Next Steps Phase 1 Data Cutaneous melanoma 2Q – Monotherapy and anti-PD1 combination 1Q - Start PRISM-MEL301 1L Melanoma Ovarian 3Q – Monotherapy and chemotherapy combination NSCLC 4Q – Monotherapy and combination data Endometrial Enrolling KIMMTRAK combination Enrolling 40 mcg dose optimization (Project Optimus) Enrolling
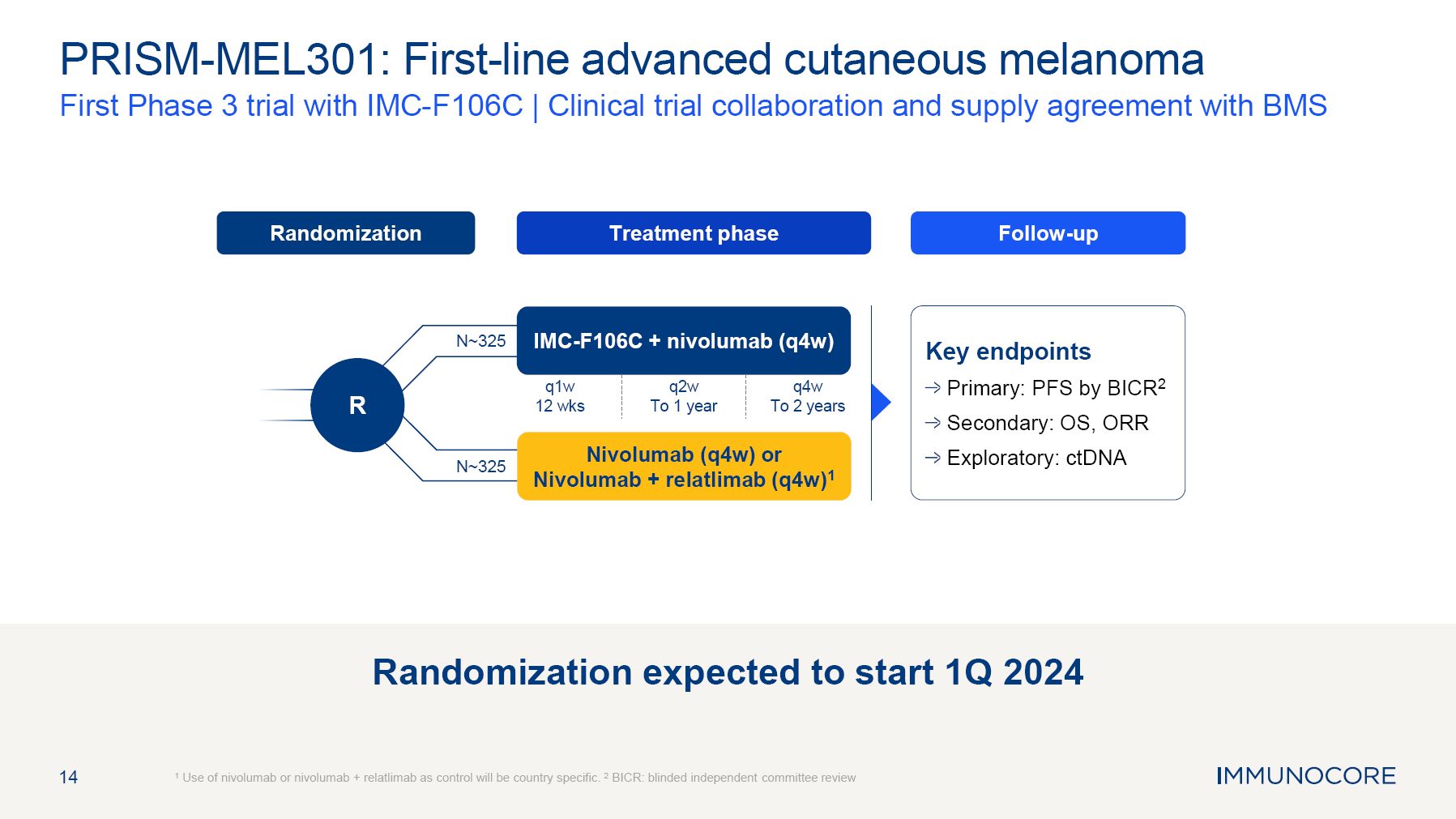
14 PRISM-MEL301: First-line advanced cutaneous melanoma First Phase 3 trial with IMC-F106C | Clinical trial collaboration and supply agreement with BMS 1 Use of nivolumab or nivolumab + relatlimab as control will be country specific. 2 BICR: blinded independent committee review Treatment phase Follow-up Randomization R Key endpoints Primary: PFS by BICR2 Secondary: OS, ORR Exploratory: ctDNA IMC-F106C + nivolumab (q4w) Nivolumab (q4w) or Nivolumab + relatlimab (q4w)1 N~325 N~325 q1w 12 wks q2w To 1 year q4w To 2 years Randomization expected to start 1Q 2024
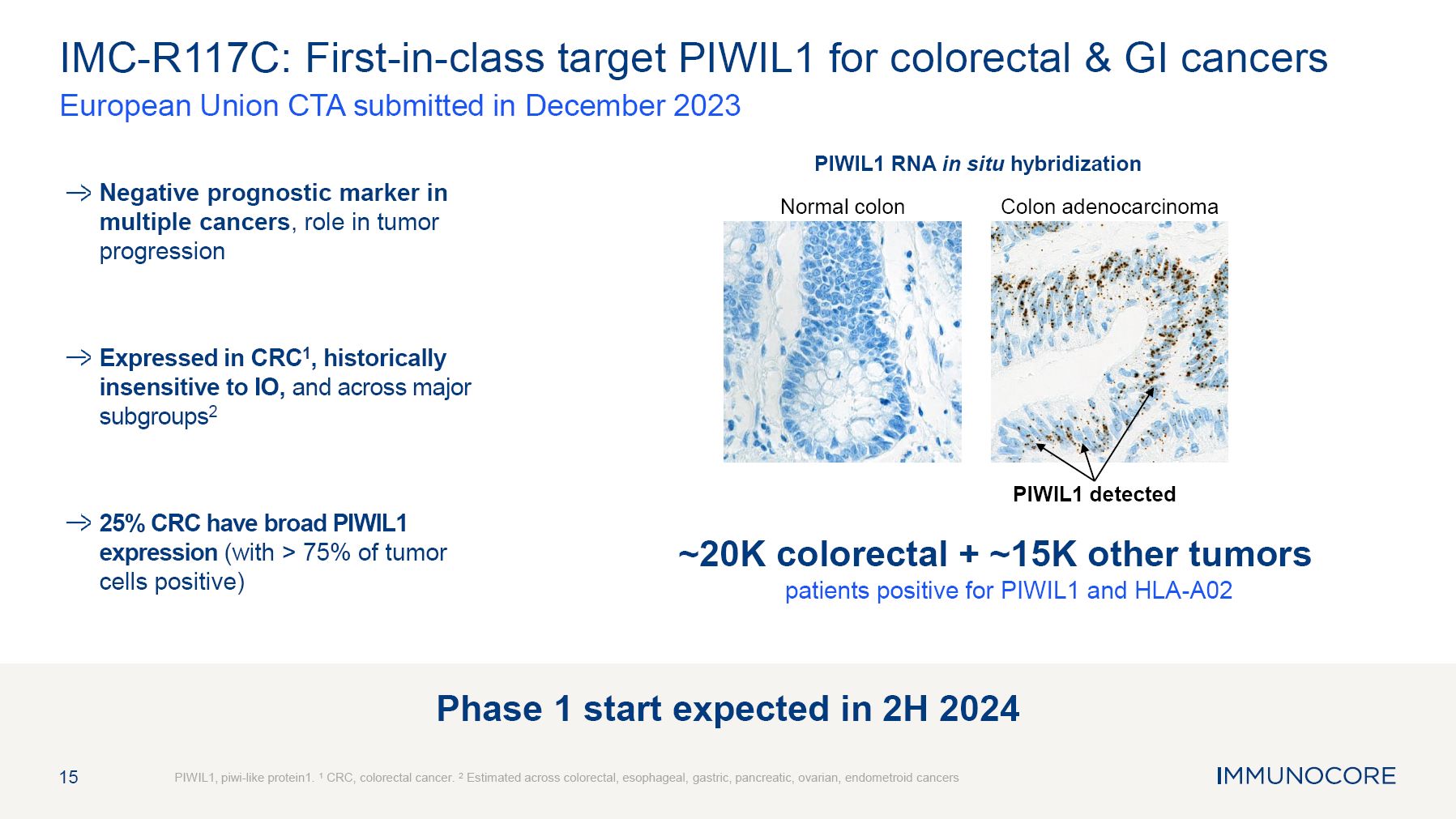
Negative prognostic marker in multiple cancers, role in tumor progression Expressed in CRC1, historically insensitive to IO, and across major subgroups2 25% CRC have broad PIWIL1 expression (with > 75% of tumor cells positive) 15 IMC-R117C: First-in-class target PIWIL1 for colorectal & GI cancers European Union CTA submitted in December 2023 PIWIL1, piwi-like protein1. 1 CRC, colorectal cancer. 2 Estimated across colorectal, esophageal, gastric, pancreatic, ovarian, endometroid cancers PIWIL1 RNA in situ hybridization Normal colon Colon adenocarcinoma PIWIL1 detected ~20K colorectal + ~15K other tumors patients positive for PIWIL1 and HLA-A02 Phase 1 start expected in 2H 2024
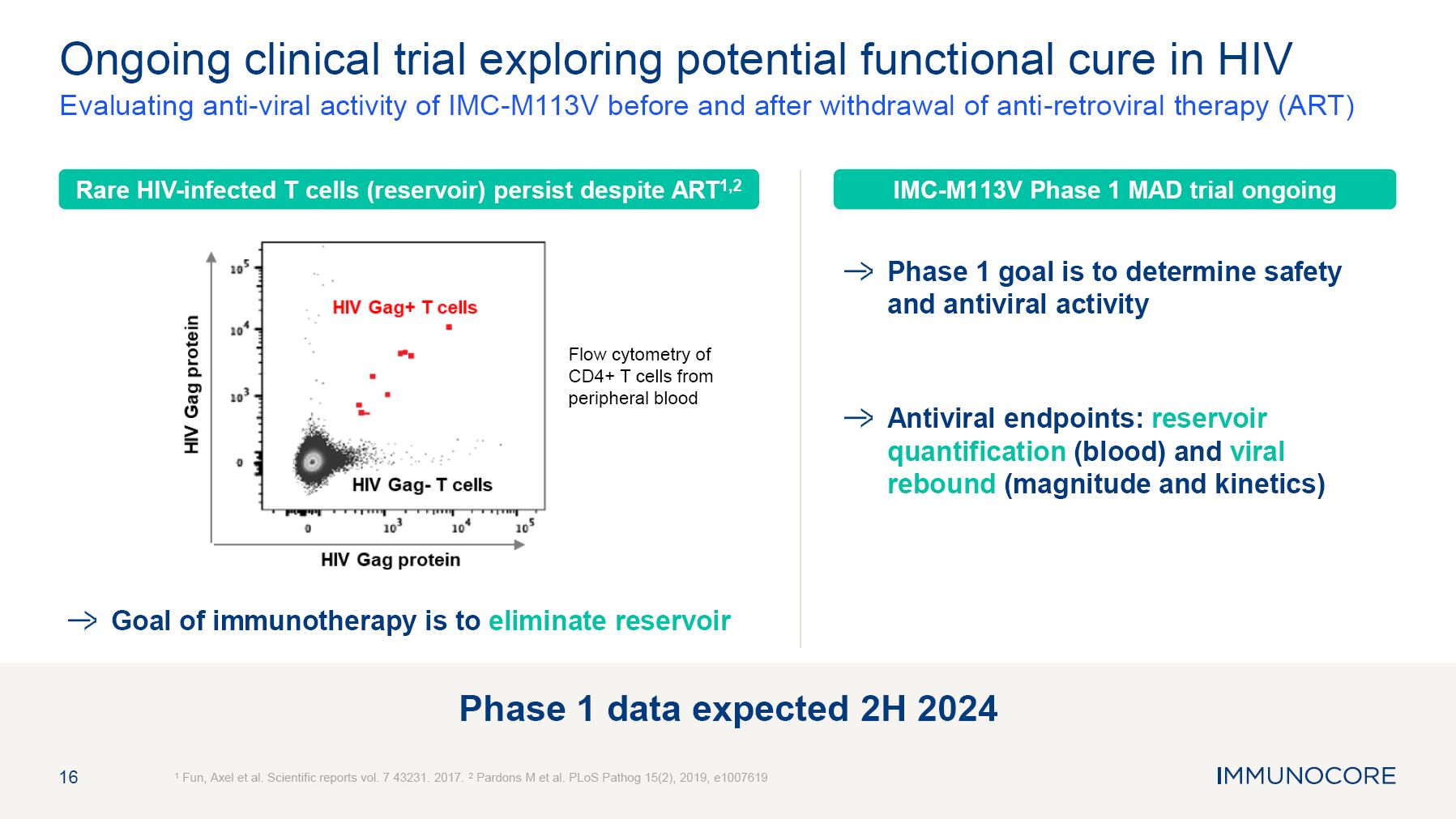
16 Ongoing clinical trial exploring potential functional cure in HIV Evaluating anti-viral activity of IMC-M113V before and after withdrawal of anti-retroviral therapy (ART) 1 Fun, Axel et al. Scientific reports vol. 7 43231. 2017. 2 Pardons M et al. PLoS Pathog 15(2), 2019, e1007619 Phase 1 goal is to determine safety and antiviral activity Antiviral endpoints: reservoir quantification (blood) and viral rebound (magnitude and kinetics) Flow cytometry of CD4+ T cells from peripheral blood IMC-M113V Phase 1 MAD trial ongoing Rare HIV-infected T cells (reservoir) persist despite ART1,2 Goal of immunotherapy is to eliminate reservoir Phase 1 data expected 2H 2024

Delivering across our three strategic pillars Advancing clinical portfolio Maximizing potential of KIMMTRAK Innovating for sustainable growth David Berman Head of Research & Development 17
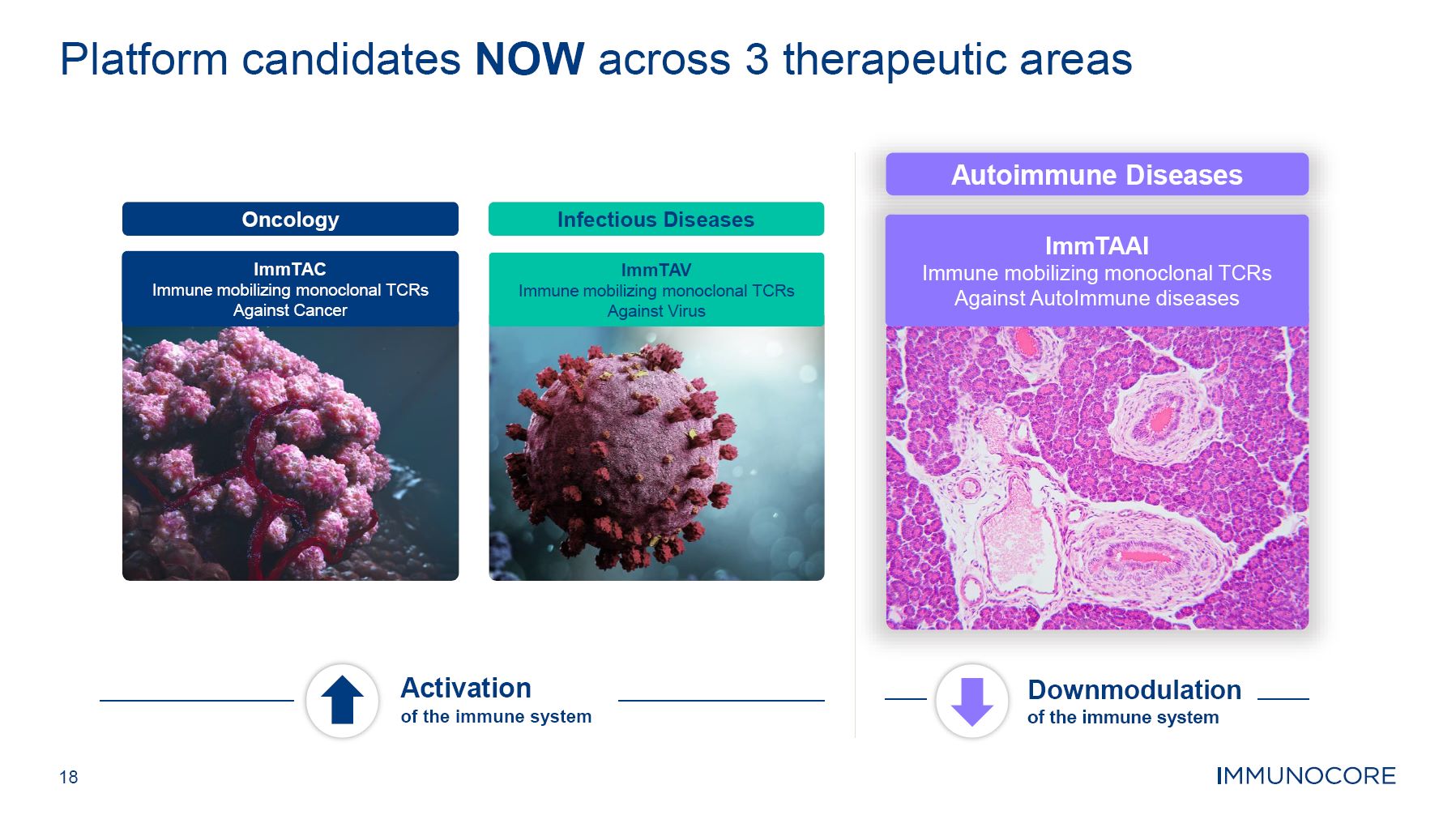
18 Platform candidates NOW across 3 therapeutic areas Oncology ImmTAC Immune mobilizing monoclonal TCRs Against Cancer Infectious Diseases ImmTAV Immune mobilizing monoclonal TCRs Against Virus Autoimmune Diseases ImmTAAI Immune mobilizing monoclonal TCRs Against AutoImmune diseases Activation of the immune system Downmodulation of the immune system
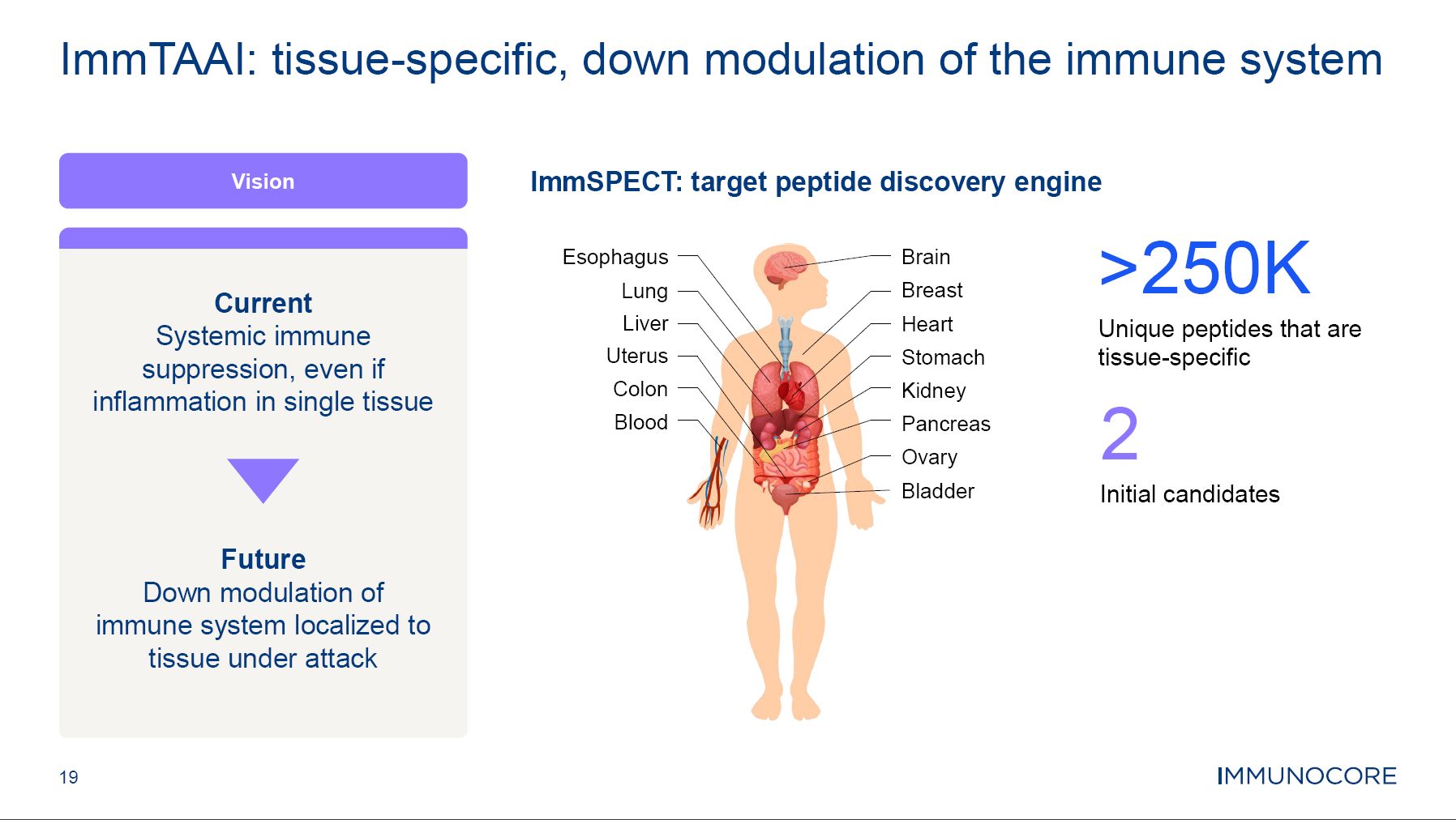
19 ImmTAAI: tissue-specific, down modulation of the immune system Vision Current Systemic immune suppression, even if inflammation in single tissue Future Down modulation of immune system localized to tissue under attack ImmSPECT: target peptide discovery engine >250K Unique peptides that are tissue-specific 2 Initial candidates Brain Breast Heart Stomach Kidney Pancreas Ovary Bladder Esophagus Lung Liver Uterus Colon Blood
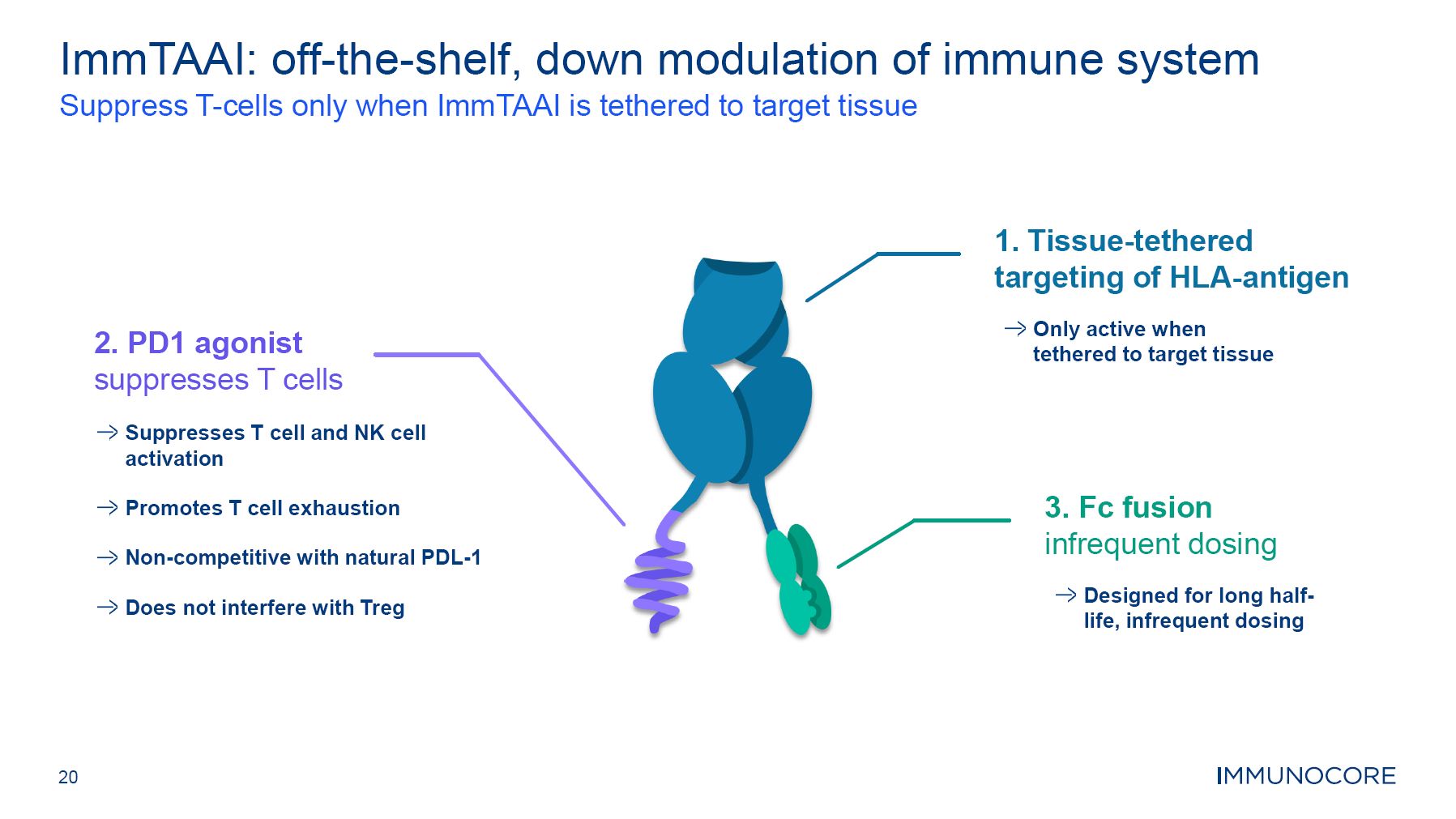
20 ImmTAAI: off-the-shelf, down modulation of immune system Suppress T-cells only when ImmTAAI is tethered to target tissue 2. PD1 agonist suppresses T cells 3. Fc fusion infrequent dosing Designed for long half- life, infrequent dosing Suppresses T cell and NK cell activation Promotes T cell exhaustion Non-competitive with natural PDL-1 Does not interfere with Treg 1. Tissue-tethered targeting of HLA-antigen Only active when tethered to target tissue

21 IMC-S118AI (PPIxPD1) for type 1 diabetes Pancreas-tethered ImmTAAI (HLA-A02) protects against killing by autoreactive T cells 1 Quantity measured as area under the curve. 2 Current Diabetes Reports (2023) 23:277–291 (~700K in US & ~700K in EU5) ImmTAAI binds specifically to pre-pro-insulin (PPI) peptide on pancreatic β-cells Potent protection of β-cells from killing by autoreactive T cells 60 80 100 120 140 160 10 100 0.001 0.01 0.1 1 [ImmTAAI] nM β cell number1 Immune system attacks and kills the beta cells responsible for controlling glucose levels through the release of insulin ~1.4M HLA-A2+ type 1 diabetes patients (US + EU5)2 Autoreactive killing Protection PPI ImmTAAI Non-tethered ImmTAAI β-cells alone β-cells + T cells β-cell marker

ImmTAAI binds specifically to APC in skin 22 Universal (non-HLA restricted) candidate for dermatology Antigen presenting cell (APC) tethered ImmTAAI inhibits T cell activation Potent inhibition of cytokine release Non-tethered ImmTAAI Tethered ImmTAAI ImmTAAI (nM) IL-2 release (%stim control) Potential dermatological diseases: atopic dermatitis, psoriasis, and other skin diseases

4Q23 & FY23 financial results Brian Di Donato Chief Financial Officer & Head of Strategy 23
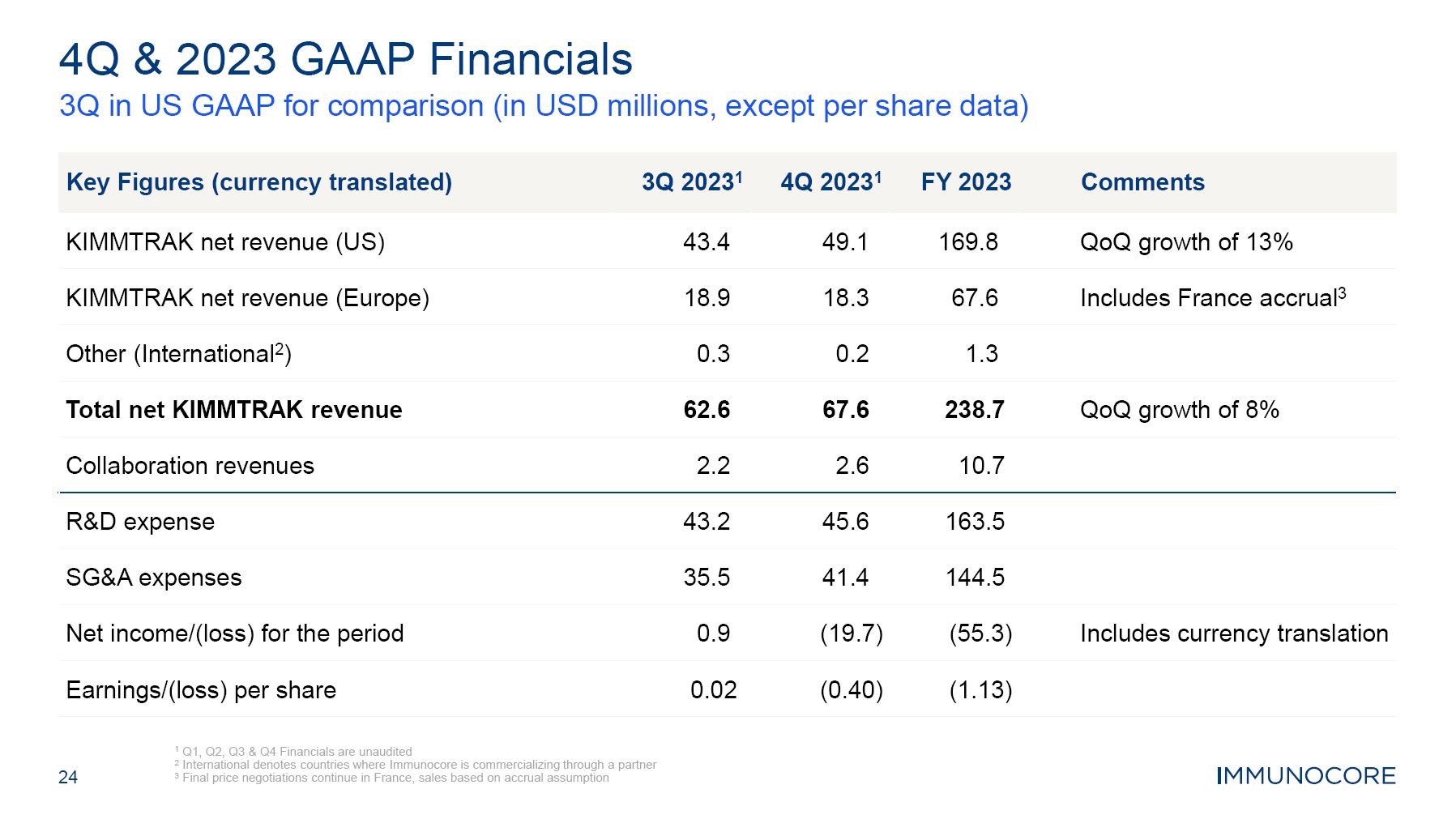
24 4Q & 2023 GAAP Financials 3Q in US GAAP for comparison (in USD millions, except per share data) 1 Q1, Q2, Q3 & Q4 Financials are unaudited 2 International denotes countries where Immunocore is commercializing through a partner 3 Final price negotiations continue in France, sales based on accrual assumption Key Figures (currency translated) 3Q 20231 4Q 20231 FY 2023 Comments KIMMTRAK net revenue (US) 43.4 49.1 169.8 QoQ growth of 13% KIMMTRAK net revenue (Europe) 18.9 18.3 67.6 Includes France accrual3 Other (International2) 0.3 0.2 1.3 Total net KIMMTRAK revenue 62.6 67.6 238.7 QoQ growth of 8% Collaboration revenues 2.2 2.6 10.7 R&D expense 43.2 45.6 163.5 SG&A expenses 35.5 41.4 144.5 Net income/(loss) for the period 0.9 (19.7) (55.3) Includes currency translation Earnings/(loss) per share 0.02 (0.40) (1.13)
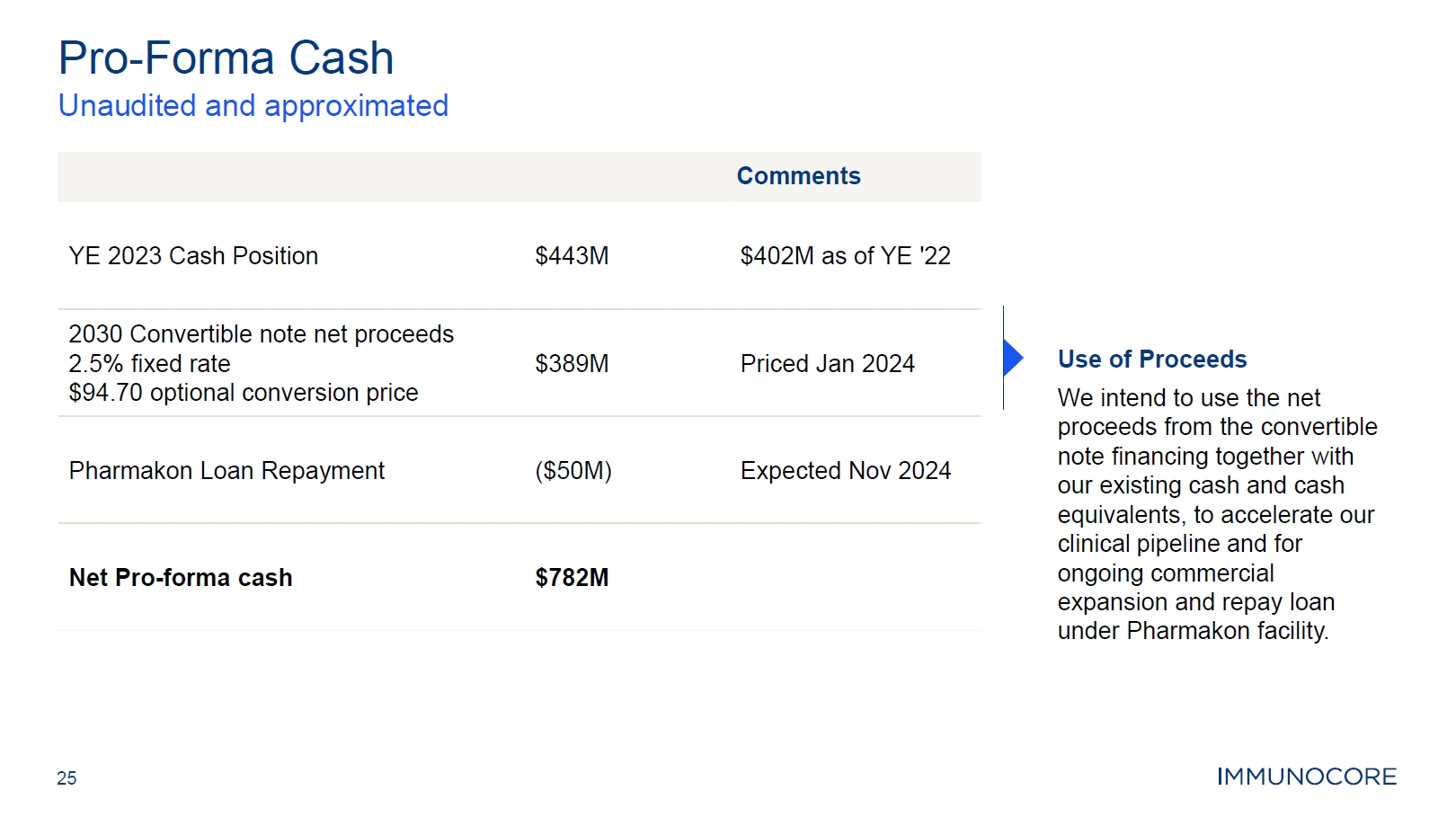
25 Pro-Forma Cash Unaudited and approximated Use of Proceeds We intend to use the net proceeds from the convertible note financing together with our existing cash and cash equivalents, to accelerate our clinical pipeline and for ongoing commercial expansion and repay loan under Pharmakon facility. Comments YE 2023 Cash Position $443M $402M as of YE '22 2030 Convertible note net proceeds 2.5% fixed rate $94.70 optional conversion price $389M Priced Jan 2024 Pharmakon Loan Repayment $50M Expected Nov 2024 Net Pro-forma cash $782M

Delivering on our promise 26 Bahija Jallal Chief Executive Officer

27 Looking ahead Commercial milestones Clinical milestones KIMMTRAK Continued global growth driving by the United States and additional launches 2024 KIMMTRAK Expansion Topline data from Ph 2 2L+ advanced cutaneous melanoma (TEBE-AM) 4Q 2024 First patient randomized in Ph 3 registrational adjuvant uveal melanoma trial (ATOM); led by EORTC 2H 2024 PRAME Franchise First patient randomized in Ph 3 registrational 1L advanced cutaneous melanoma (PRISM-MEL301) 1Q 2024 Cutaneous melanoma data from Phase 1 PRAME trial 2Q 2024 Serous ovarian data from Phase 1 PRAME trial 3Q 2024 NSCLC data from Phase 1 PRAME trial 4Q 2024 IND/CTA for PRAME-HLE trial Mid-2024 IND/CTA for PRAME-A24 trial 4Q 2024 PIWIL1 First patient dosed in PIWIL1 Phase 1 trial 2H 2024 Infectious Diseases Data from Ph 1 HIV MAD/POC trial 2H 2024 Enroll Ph 1 HBV MAD (now including HCC) trial 2024 Autoimmune Diseases Initiating CMC manufacturing for autoimmune candidates 2024
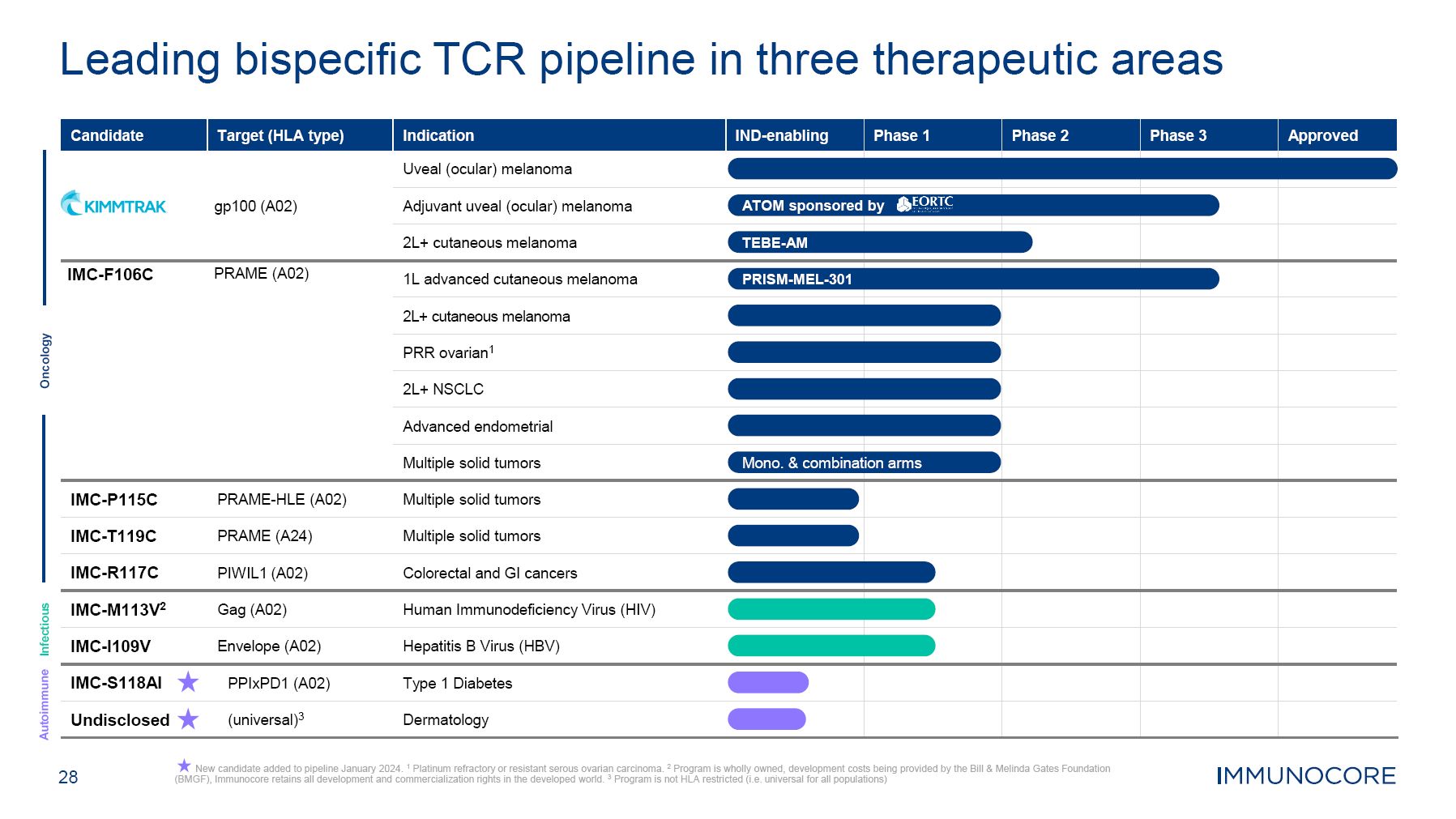
28 New candidate added to pipeline January 2024. 1 Platinum refractory or resistant serous ovarian carcinoma. 2 Program is wholly owned, development costs being provided by the Bill & Melinda Gates Foundation (BMGF), Immunocore retains all development and commercialization rights in the developed world. 3 Program is not HLA restricted (i.e. universal for all populations) Leading bispecific TCR pipeline in three therapeutic areas Oncology Autoimmune Infectious Candidate Target (HLA type) gp100 (A02) Indication Uveal (ocular) melanoma IND-enabling Phase 1 Phase 2 Phase 3 Approved Adjuvant uveal (ocular) melanoma ATOM sponsored by 2L+ cutaneous melanoma TEBE-AM IMC-F106C PRAME (A02) 1L advanced cutaneous melanoma PRISM-MEL-301 2L+ cutaneous melanoma PRR ovarian1 2L+ NSCLC Advanced endometrial Multiple solid tumors Mono. & combinati on arms IMC-P115C PRAME-HLE (A02) Multiple solid tumors IMC-T119C PRAME (A24) Multiple solid tumors IMC-R117C PIWIL1 (A02) Colorectal and GI cancers IMC-M113V2 Gag (A02) Human Immunodeficiency Virus (HIV) IMC-I109V Envelope (A02) Hepatitis B Virus (HBV) IMC-S118AI PPIxPD1 (A02) Type 1 Diabetes Undisclosed (universal)3 Dermatology

29 Bahija Jallal PhD Chief Executive Officer Ralph Torbay Head of Commercial Brian Di Donato Chief Financial Officer and Head of Strategy Mohammed Dar MD SVP, Clinical Development and Chief Medical Officer David Berman MD, PhD Head of Research and Development John Goll SVP, Finance and Chief Accounting Officer Q&A Session

Thank you 30