Exhibit 99.2
Financial results for the first half of 2017 24 August 2017

Forward-looking statements This presentation contains information pertaining to Zealand Pharma A/S (“Zealand"). Neither Zealand nor its management, directors, employees or representatives make any representation or warranty, express or implied, as to the accuracy or completeness of any of the information contained in this presentation or any other information transmitted or made available to the viewer or recipient hereof, whether communicated in written or oral form. This presentation does not constitute or form part of, and should not be construed as, an offer to sell or issue or the solicitation of an offer to buy or acquire Zealand securities, in any jurisdiction, or an inducement to enter into investment activity, nor shall there be any sale of Zealand securities in any state or jurisdiction in which such offer, solicitation or sale would be unlawful prior to registration or qualification under the securities laws of any such state or jurisdiction. No part of this presentation, nor the fact of its distribution, should form the basis of, or be relied on in connection with, any contract or commitment or investment decision whatsoever. This presentation contains forward-looking statements that reflect management's current views with respect to Zealand's product candidates' development, clinical and regulatory timelines and anticipated results, market opportunity, potential financial performance and other statements of future events or conditions. Although Zealand believes that the expectations reflected in such forward-looking statements are reasonable, no assurance can be given that such expectations will prove to have been correct. Accordingly, results could differ materially from those set out in the forward-looking statements as a result of various factors, many of which are beyond Zealand’s control. No reliance should be made on such forward-looking statements. Zealand does not intend to update the presentation, including the forward-looking statements contained therein, following distribution, beyond what is required by applicable law or applicable stock exchange regulations if and when circumstances arise that lead to changes compared to the date when these statements were provided.

Advanced pipeline focused on speciality gastrointestinal and metabolic diseases Type 2 diabetes drug* partnered with Sanofi launched in the U.S. and EU in 2017 New combination treatment addressing large market Key portfolio asset for Sanofi One Phase 3 program Two Phase 2 programs – One approaching Phase 3 Three Phase 1 programs Proven ability to advance new drug candidates Strong peptide research capability Multiple near-term value-driving catalysts Zealand’s attractive pipeline and anticipated growing revenues to serve as catalysts of value creation *Soliqua® 100/33 (U.S.) /Suliqua® (EU) – a combination product of basal insulin (Lantus®) and GLP-1 analog (Adlyxin®) and Adlyxin® (U.S.)/Lyxumia® (EU and RoW) – a once-daily GLP-1.

Highlights Financial highlights Business highlights Q2 Business highlights for the period after H1 financial results Nasdaq listing Financial guidance

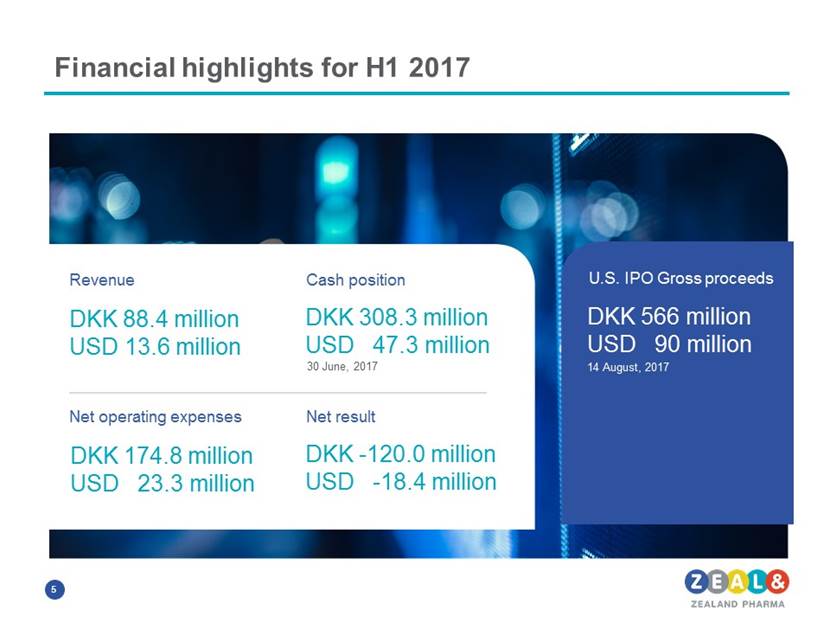
Financial highlights for H1 2017 Revenue DKK 88.4 million/ USD 13.6 million Cash position DKK 308.3 million USD 47.3 million 30 June, 2017 Net operating expenses DKK 174.8 million/ USD 23.3 million Net result DKK -120.0 million USD -18.4 million US IPO Gross proceeds DKK 566 million USD 90 million 14 August, 2017
Business highlights for Q2 2017 Glepaglutide Glepaglutide met primary endpoint in Phase 2 clinical trial for short bowel syndrome Dasiglucagon Positive results from two Phase 2a trials for use in a dual hormone pump system Orphan designation for congenital hyperinsulinism (CHI) obtained in the EU Helsinn Helsinn returns elsiglutide and all rights to develop products within cancer-supportive care

Business highlights for the period thereafter Dasiglucagon First patients dosed in Phase 3 trial for severe hypoglycemia in diabetes Orphan designation for congenital hyperinsulinism (CHI) obtained in the U.S. Boehringer Ingelheim First patients dosed in Phase 1 trial in amylin program partnered with BI First patients dosed in Phase 1 trial in GLP1/glucagon program partnered with BI Soliqua® 100/33 Soliqua® 100/33 had 62% commercial access as of 1 July, 2017 US IPO 2017 Capital increase of DKK 566 million/USD 90 million through the Nasdaq Global Select Market listing in the U.S.

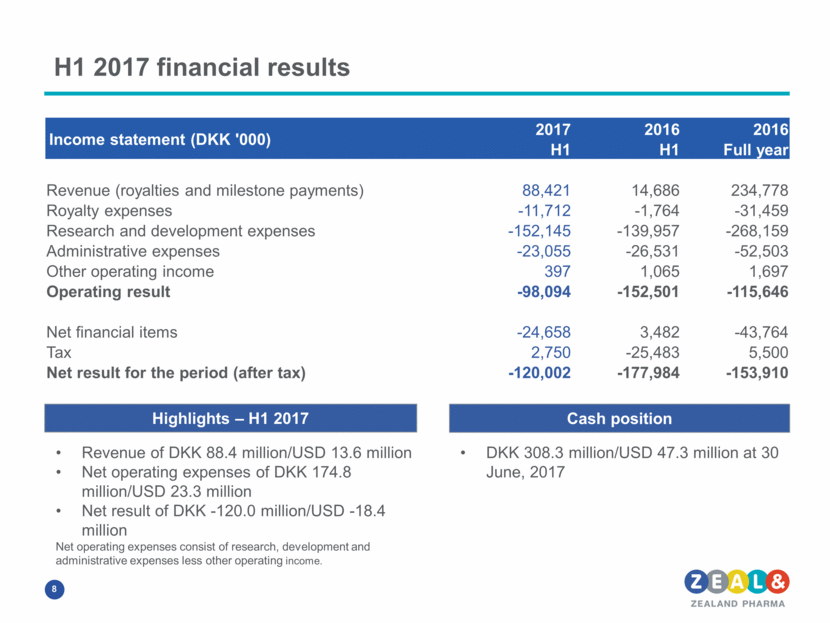
H1 2017 financial results Income statement (DKK '000) 2017 2016 2016 H1 H1 Full year Revenue (royalties and milestone payments) 88,421 14,686 234,778 Royalty expenses -11,712 -1,764 -31,459 Research and development expenses -152,145 -139,957 -268,159 Administrative expenses -23,055 -26,531 -52,503 Other operating income 397 1,065 1,697 Operating result -98,094 -152,501 -115,646 Net financial items -24,658 3,482 -43,764 Tax 2,750 -25,483 5,500 Net result for the period (after tax) -120,002 -177,984 -153,910 Cash position Highlights – H1 2017 DKK 308.3 million/USD 47.3 million at 30 June, 2017 Revenue of DKK 88.4 million/USD 13.6 million Net operating expenses of DKK 174.8 million/USD 23.3 million Net result of DKK -120.0 million/USD -18.4 million Net operating expenses consist of research, development and administrative expenses less other operating income.
Listing on the Nasdaq Global Select Market in the U.S. 5,031,250 shares issued as ADS’s, trading on Nasdaq Global Select Market in New York A total of 30,718,652 shares 16.4% are issued as ADS’s 83.6% on the Copenhagen stock exchange Total gross proceeds amount to DKK 566 million / USD 90 million

Financial guidance for 2017 unchanged DKK USD Revenue from milestone payments* 100 million** 15 million** Royalty revenue from Sanofi collaboration* No guidance Net operating expenses 390-410 million 60-63 million Operating loss before royalty income and royalty expenses 290-310 million 45-48 million *Zealand does not guide on royalty revenue, as Sanofi has not provided guidance. ** USD 10 million hereof was received in January 2017

Portfolio update Soliqua® 100/33 update Pipeline Glepaglutide Dasiglucagon Orphan designation of dasiglucagon Boehringer Ingelheim Summary Q&A

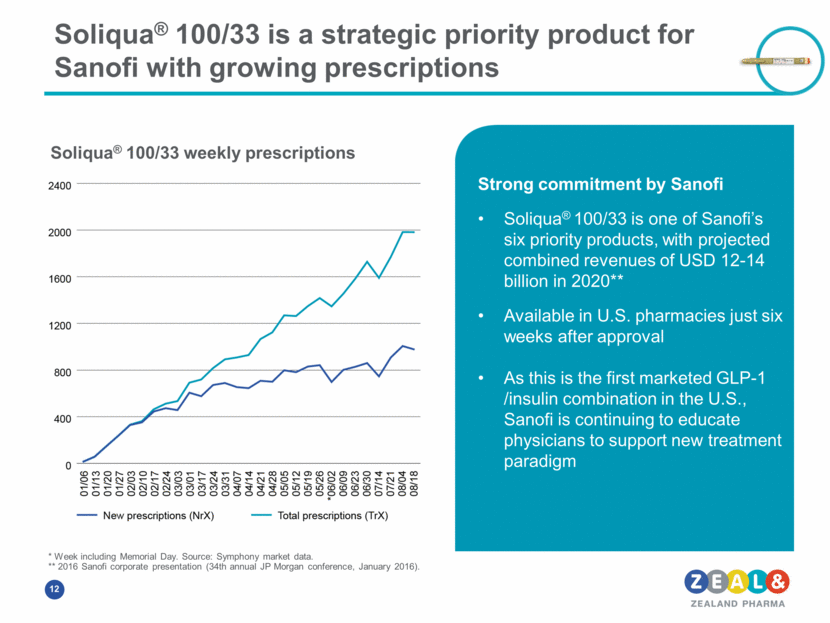
Soliqua® 100/33 is a strategic priority product for Sanofi with growing prescriptions Strong commitment by Sanofi Soliqua® 100/33 is one of Sanofi’s six priority products, with projected combined revenues of USD 12-14 billion in 2020** Available in U.S. pharmacies just six weeks after approval As this is the first marketed GLP-1 /insulin combination in the U.S., Sanofi is continuing to educate physicians to support new treatment paradigm * Week including Memorial Day. Source: Symphony market data. ** 2016 Sanofi corporate presentation (34th annual JP Morgan conference, January 2016). Soliqua® 100/33 weekly prescriptions
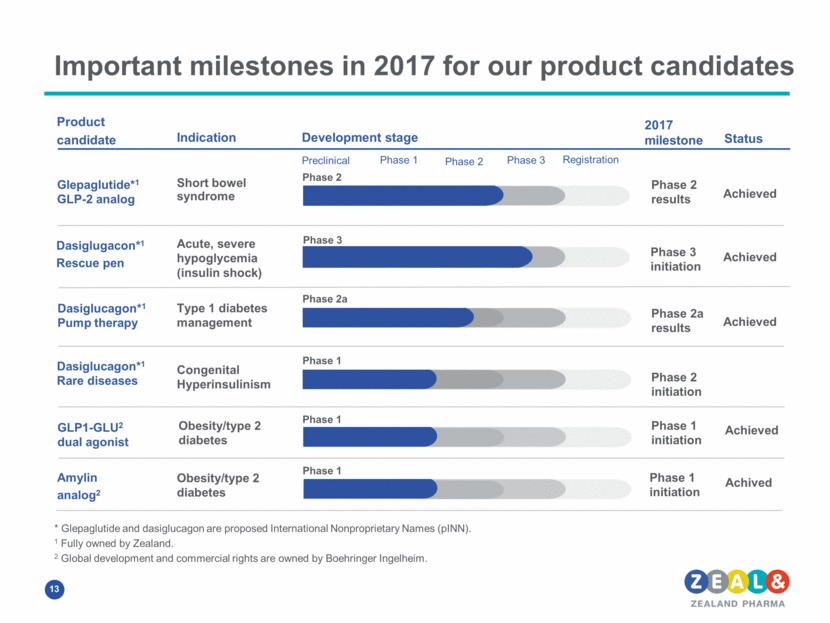
Important milestones in 2017 for our product candidates Product candidate Indication Development stage Preclinical Phase 1 Phase 2 Phase 3 Registration 2017 milestone Glepaglutide*1 GLP-2 analog Dasiglugacon*1 Rescue pen Amylin analog2 GLP1-GLU2 dual agonist Short bowel syndrome Acute, severe hypoglycemia (insulin shock) Type 1 diabetes management Obesity/type 2 diabetes Obesity/type 2 diabetes Phase 2 Phase 2a Phase 1 Phase 1 Phase 2 results Phase 3 initiation Phase 2a results Phase 1 initiation Phase 1 initiation * Glepaglutide and dasiglucagon are proposed International Nonproprietary Names (pINN). 1 Fully owned by Zealand. 2 Global development and commercial rights are owned by Boehringer Ingelheim. Phase 3 Congenital Hyperinsulinism Phase 1 Phase 2 initiation Dasiglucagon*1 Pump therapy Dasiglucagon*1 Rare diseases Achieved Status Achieved Achieved Achieved Achived
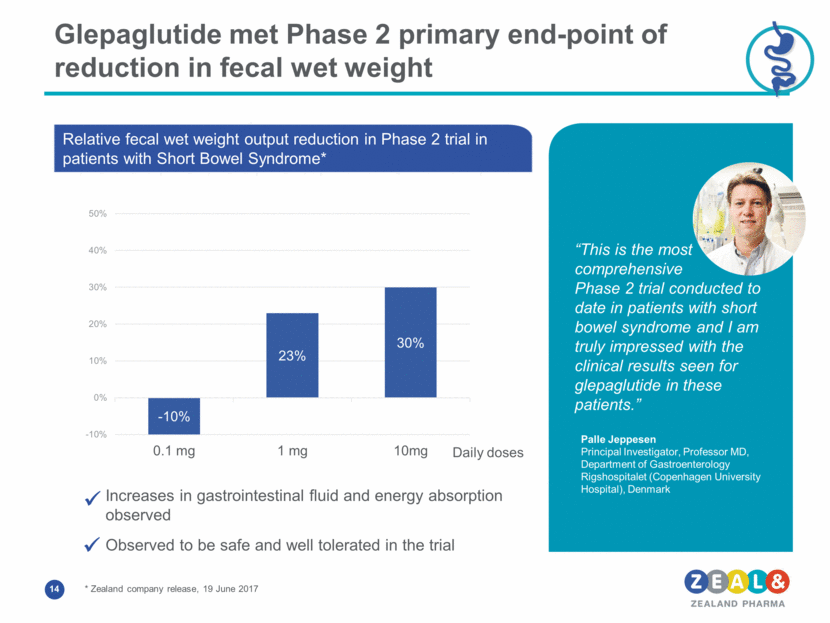
Glepaglutide met Phase 2 primary end-point of reduction in fecal wet weight Increases in gastrointestinal fluid and energy absorption observed Observed to be safe and well tolerated in the trial “This is the most comprehensive Phase 2 trial conducted to date in patients with short bowel syndrome and I am truly impressed with the clinical results seen for glepaglutide in these patients.” Palle Jeppesen Principal Investigator, Professor MD, Department of Gastroenterology Rigshospitalet (Copenhagen University Hospital), Denmark Relative fecal wet weight output reduction in Phase 2 trial in patients with Short Bowel Syndrome* * Zealand company release, 19 June 2017 Daily doses - 10% 23% 30% -10% 0% 10% 20% 30% 40% 50% 0.1 mg 1 mg 10mg Fecal wet weight output reduction
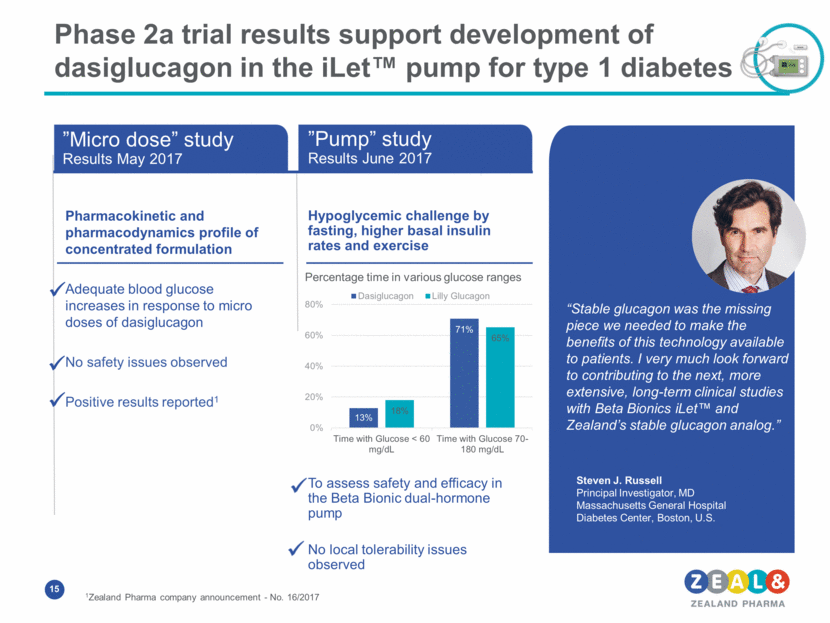
Phase 2a trial results support development of dasiglucagon in the iLet™ pump for type 1 diabetes Pharmacokinetic and pharmacodynamics profile of concentrated formulation Adequate blood glucose increases in response to micro doses of dasiglucagon No safety issues observed Positive results reported1 Hypoglycemic challenge by fasting, higher basal insulin rates and exercise To assess safety and efficacy in the Beta Bionic dual-hormone pump No local tolerability issues observed ”Micro dose” study Results May 2017 ”Pump” study Results June 2017 Steven J. Russell Principal Investigator, MD Massachusetts General Hospital Diabetes Center, Boston, U.S. “Stable glucagon was the missing piece we needed to make the benefits of this technology available to patients. I very much look forward to contributing to the next, more extensive, long-term clinical studies with Beta Bionics iLet™ and Zealand’s stable glucagon analog.” Percentage time in various glucose ranges 1Zealand Pharma company announcement - No. 16/2017 13% 71% 18% 65% 0% 20% 40% 60% 80% Time with Glucose < 60 mg/dL Time with Glucose 70- 180 mg/dL Dasiglucagon Lilly Glucagon
Orphan designation of dasiglucagon for congenital hyperinsulinism (CHI) obtained in the EU and the U.S. Disease Congenital hyperinsulinism (CHI) is a rare condition associated with high levels of insulin in the blood, leading to severe hypoglycemia in newborns and children Repeated episodes of low blood sugar is associated with the risk of serious complications such as brain damage, seizures and coma Prevalence Approximately 1/25,000 – 1/50,000 newborns CHI is more common in certain populations, affecting up to 1 in 2,500 newborns Current treatment Current medical treatment opportunities are insufficient and many CHI children undergo major pancreatic surgery

Boehringer Ingelheim programs – two product candidates on obesity/type 2 diabetes Dasiglucagon First patients dosed in Phase 1 trial in GLP1/glucagon program on obesity/diabetes First patients dosed in Phase 1 trial in amylin program on obesity/diabetes GLP-1/glucagon dual-acting agonist Long-acting amylin analog Initiation of Phase 1 trial triggered a milestone payment of EUR 4 million EUR 283 million outstanding in milestone payments Mid single-digit to low double-digit percentage royalties on global sales EUR 365 million outstanding in milestone payments High single-digit to low double-digit percentage royalties on global sales Zealand has two collaborations with Boehringer Ingelheim who funds all research, development and commercialization activities

Positive clinical results from three Phase 2 studies Summary from H1 2017 and the period thereafter Growing revenue Attractive later stage pipeline Strong cash position

Q&A - Upcoming events September 11-13, 2017 Morgan Stanley Global Healthcare Conference, New York September 19, 2017 InvestorDagen, Copenhagen September 27, 2017 ABGSC Small & Mid Cap efterårsseminar, Copenhagen S2:00pm – 3:00pm14:00 – 15:00 eight one media November 15, 2017 Jefferies Healthcare Conference, London November 16, 2017 BryanGarnier Healthcare Conference, Paris December 6-7, 2017 Citi Global Healthcare Conference, New York Upcoming events in 2017 Morgan Stanley Global Healthcare Conference, New York 11-13 September ABGSC Small and Midcap seminar, Copenhagen 27 September Jefferies Healthcare Conference, London 15 November BryanGarnier Healthcare Conference, Paris 16 November Citi Global Healthcare Conference, New York 6-7 December
