Zealand Pharma expands its peptide platform with acquisition of Encycle Therapeutics October 22, 2019

Forward-looking statements This presentation contains information pertaining to Zealand Pharma A/S (“Zealand"). Neither Zealand nor its management, directors, employees or representatives make any representation or warranty, express or implied, as to the accuracy or completeness of any of the information contained in this presentation or any other information transmitted or made available to the viewer or recipient hereof, whether communicated in written or oral form. This presentation does not constitute or form part of, and should not be construed as, an offer to sell or issue or the solic itation of an offer to buy or acquire Zealand securities, in any jurisdiction, or an inducement to enter into investment activity, nor shal l there be any sale of Zealand securities in any state or jurisdiction in which such offer, solicitation or sale would be unlawful prior to registration or qualification under the securities laws of any such state or jurisdiction. No part of this presentation, nor the fact of i ts distribution, should form the basis of, or be relied on in connection with, any contract or commitment or investment decision whatsoever. This presentation contains forward-looking statements that reflect management's current views with respect to Zealand's product candidates' development, clinical and regulatory timelines and anticipated results, market opportunity, potential financial performance and other statements of future events or conditions. Although Zealand believes that the expectations reflected in such forward-looking statements are reasonable, no assurance can be given that such expectations will prove to have been correct. Accordingly, results could differ materially from those set out in the forward-looking statements as a result of various factors, many of which are beyond Zealand’s control. No reliance should be made on such forward-looking statements. Zealand does not intend to update the presentation, including the forward-looking statements contained therein, following distribution, beyond what is required by applicable law or applicable stock exchange regulations if and when circumstances ar ise that lead to changes compared to the date when these statements were provided. 1

Strengthening our leadership in peptide therapeutics and novel treatments for gastrointestinal diseases Strategic pipeline expansion Growing the Zealand Pharma peptide platform technology with a unique oral Leveraging R&D expertise Utilizing Zealand Pharma’s world-leading capabilities in peptide development to realize full therapeutic potential Clinically validated mode of action Lead pre-clinical candidate to target integrin 47, which is involved inflammatory bowel disease where a significant unmet need for oral therapy remains The target is clinically validated by vedolizumab, an approved, infusion-only 47 integrin inhibitor in 2

Financially compelling strategic transaction 100% Contingent Consideration No upfront payment Key Figures Future potential earn-outs of up to US $80 million contingent on successful achievement of development, regulatory and commercial milestones • Of which, US $10 million would be payable by successful completion of Phase 2 Payable in cash and/or ZEAL equity at Zealand’s discretion Mid-single digit royalty On global net sales from the lead asset 3 No change to Zealand’s financial guidance on net expenses for 2019 Contingent value rights linked to lead asset only Zealand acquires only outstanding shares and intellectual property of Encycle Therapeutics

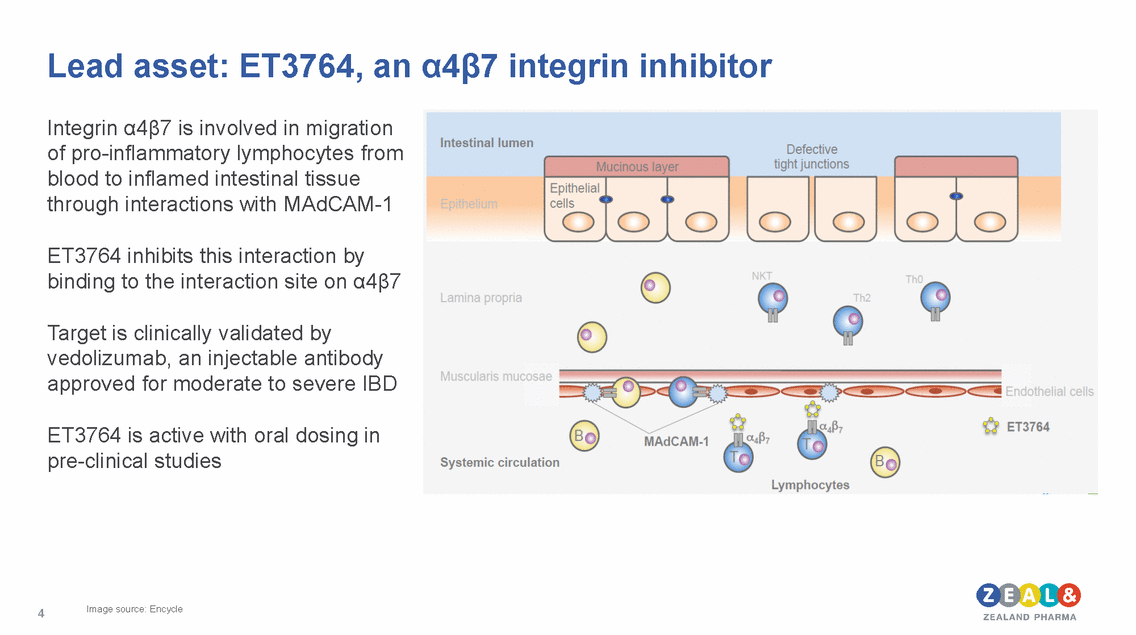
Lead asset: ET3764, an 47 integrin inhibitor Integrin 47 is involved in migration of pro-inflammatory lymphocytes from blood to inflamed intestinal tissue through interactions with MAdCAM-1 ET3764 inhibits this interaction by binding to the interaction site on 47 Target is clinically validated by vedolizumab, an injectable antibody approved for moderate to severe IBD ET3764 is active with oral dosing in pre-clinical studies Image source: Encycle 4
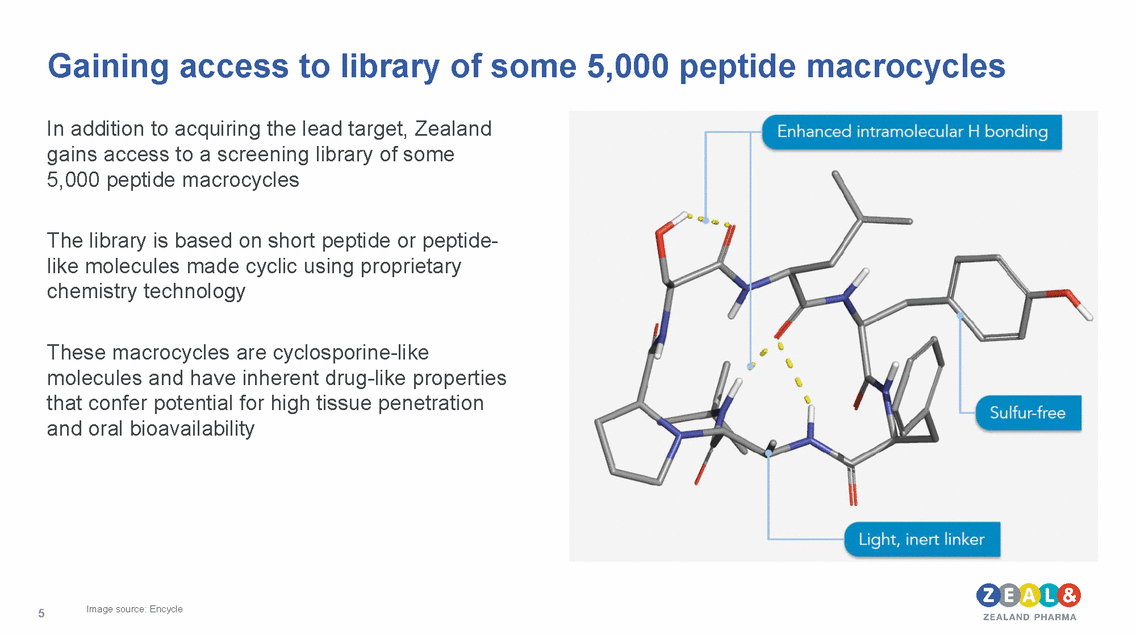
Gaining access to library of some 5,000 peptide macrocycles In addition to acquiring the lead target, Zealand gains access to a screening library of some 5,000 peptide macrocycles The library is based on short peptide or peptide-like molecules made cyclic using proprietary chemistry technology These macrocycles are cyclosporine-like molecules and have inherent drug-like properties that confer potential for and oral bioavailability high tissue penetration Image source: Encycle 5
On a clear path toward achieving Zealand Pharma’s strategic objectives Strategic pipeline expansion of the Zealand Pharma peptide platform a unique oral peptide technology with Leveraging Zealand Pharma’s R&D expertise in peptide development Making a financially compelling strategic transaction • • • No upfront payment $10m in earn-outs could be payable through successful completion of Phase 2 No impact to financial guidance on net operating expenses for fiscal year 2019 6

Q&A Upcoming Investor Events Bryan Garnier 7th Annual European Healthcare November 12, 2019, Paris Conference Zealand Pharma Interim Report for Q3 2019 November 14, 2019, Copenhagen Jefferies 2019 Healthcare Conference November 20, 2019, London Økonomisk Ugebrev November 27, 2019, Life Science Investor Conference Copenhagen
