Exhibit 99.1

NASDAQ: SLGL June, 2020

This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements other than statements of historical facts are forward-looking statements. In some cases, you can identify forward-looking statements by terms such as “may,” “will,” “should,” “expect,” “plan,” “anticipate,” “could,” “future,” “outlook,” “intend,” “target,” “project,” “contemplate,” “believe,” “estimate,” “predict,” “potential,” “continue,” or the negative of these terms or other similar expressions, although not all forward-looking statements contain these words. The forward-looking statements in this presentation relate to, among other things, statements regarding our anticipated NDA submission dates for EPSOLAY and TWYNEO, estimated timing for the approval and commercial launch of EPSOLAY and TWYNEO, and estimated sales of our product candidates. These statements are neither promises nor guarantees, but involve known and unknown risks, uncertainties, and other important factors that may cause our actual results, performance, or achievements to be materially different from any future results, performance, or achievements expressed or implied by the forward-looking statement, including but not limited to the following: risks relating to the timing of the submission of an NDA for EPSOLAY and an NDA for TWYNEO; the fact that we have and expect to continue to incur significant losses; our need for additional funding, which may not be available; our ability to complete the development of, and obtain marketing approval for, our product candidates; our ability to obtain and maintain regulatory approvals for our product candidates in our target markets and the possibility of adverse regulatory or legal actions relating to our product candidates even if regulatory approval is obtained; our ability to commercialize and launch our product candidates; our ability to obtain and maintain adequate protection of our intellectual property; our ability to manufacture our product candidates in commercial quantities, at an adequate quality or at an acceptable cost; our ability to establish adequate sales, marketing, and distribution channels; acceptance of our product candidates by healthcare professionals and patients; the possibility that we may face third-party claims of intellectual property infringement; the timing and results of clinical trials that we may conduct or that our competitors and others may conduct relating to our or their products; intense competition in our industry; potential product liability claims; potential adverse federal, state, and local government regulation in the United States, Europe, or Israel; the impact of pandemics, such as COVID-19 (coronavirus); and loss or retirement of key executives and research scientists. These and other important factors discussed in the Company's Annual Report on Form 20-F filed with the Securities and Exchange Commission (“SEC”) on March 24, 2020, and our other reports filed with the SEC could cause actual results to differ materially from those indicated by the forward-looking statements made in this presentation. Any such forward-looking statements represent management’s estimates as of the date of this presentation. While we may elect to update such forward-looking statements at some point in the future, unless required by applicable law, we disclaim any obligation to do so, even if subsequent events cause our views to change. Thus, one should not assume that our silence over time means that actual events are bearing out as expressed or implied in such forward-looking statements. These forward-looking statements should not be relied upon as representing our views as of any date subsequent to the date of this presentation. This presentation contains trademarks, trade names, and service marks of other companies, which are the property of their respective owners. We do not intend our use or display of other parties' trademarks, trade names, or service marks to imply, and such use or display should not be construed to imply, a relationship with, or endorsement or sponsorship of us by, these other parties. FORWARD-LOOKING STATEMENTS

EPSOLAY® & TWYNEO® COVID-19 We completed the clinical programs required for the submission of our NDAs for EPSOLAY and TWYNEO 1 We also met with the FDA (physically and through telecoms) for pre-NDA meetings 2 Exhibit batches for EPSOLAY were produced at full commercial scale and the next production step is the manufacture of the commercial/validation batches 3 Exhibit batches for TWYNEO were produced on a 200kg scale 4 Our CMOs (Contract Manufacturer Organizations) for EPSOLAY and TWYNEO are open despite COVID-19We therefore do not anticipate delays in the submission of the NDAs or the production of the commercial/validation batches for both EPSOLAY and TWYNEOThis is of course, a dynamic situation which we will be monitoring closely 5

Business Continuity Plan (BCP) and Disaster Recovery Plan (DRP) were in place ahead of the COVID-19 crisis, and our Information Technology (IT) infrastructure allows recovery in case of a disaster including secured remote access to serversPurchase orders were placed to increase our current inventoryCompany is following all restrictions published by the Israeli Ministry of Health (IMOH) SGT-210 Phase I proof-of-concept clinical study is ongoing subject to IMOH guidelines for COVID-19 All employees returned to work at our facilitiesCompany is taking all measures to ensure the well-being of our employees including frequent on-site cleaning and sanitary measures All business travel abroad was cancelled and replaced with telecoms and video conferences CURRENT MODUS OPERANDI COVID-19

Proprietary silica-based microencapsulation technology Positive Phase III results in acne vulgaris NDA submission expected in 2H/20Potential to be first FDA-approved acne treatment that contains fixed-dose combination of BPO and tretinoin Pending patent applications for tapinarof and roflumilast in various skin conditions EARLY STAGE Positive Phase III results in papulopustular rosaceaNDA submission expected in 2Q/20Potential to be the first single-active BPO approved by the FDA as a prescription drug product Ongoing Phase I proof-of-concept study for erlotinib gel in palmoplantar keratodermaResults expected next year SGT-210 Seven 50/50 gross profit-sharing collaborations with Perrigo$22.8 million in net revenues last year$3.4 million in net revenues in 1Q/20 GENERICS TWYNEO® TECHNOLOGY EPSOLAY® OUR DERMATOLOGY COMPANY OVERVIEW

Aiming to provide effective and tolerable topical therapies to achieve local action THE SCIENCE BEHIND OUR PROPRIETARY TECHNOLOGY
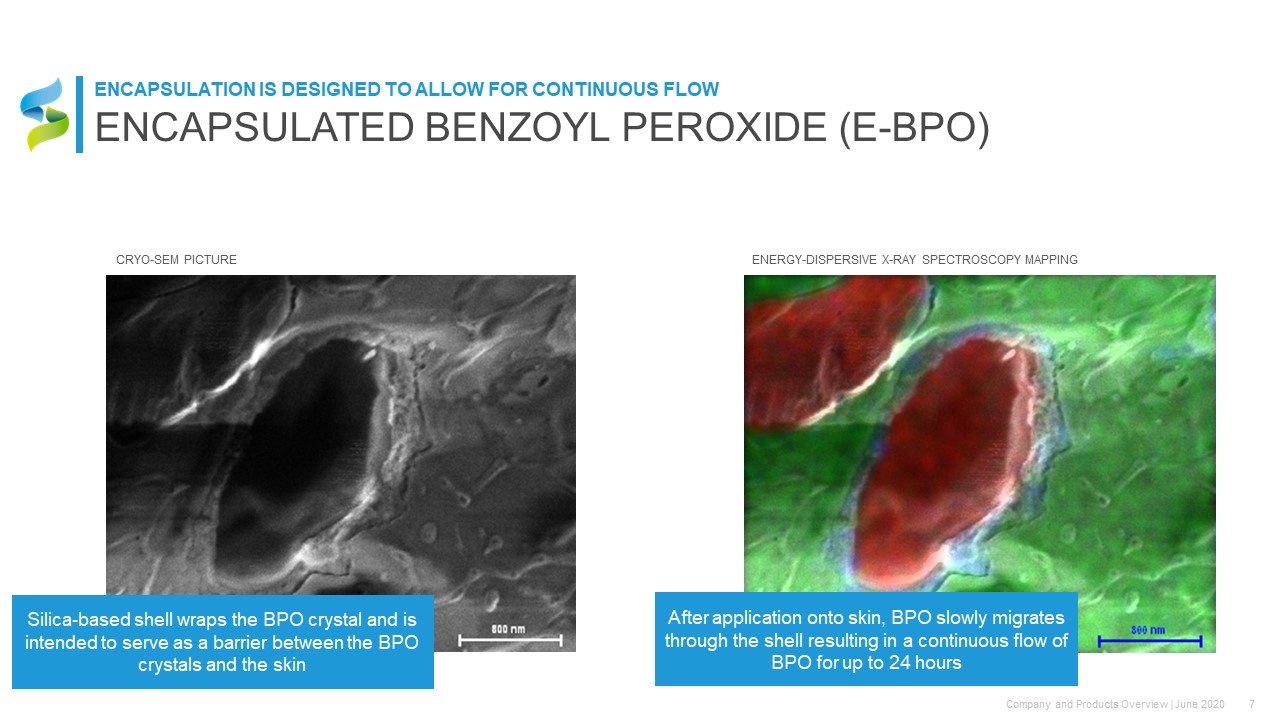
Silica-based shell wraps the BPO crystal and is intended to serve as a barrier between the BPO crystals and the skin CRYO-SEM PICTURE After application onto skin, BPO slowly migrates through the shell resulting in a continuous flow of BPO for up to 24 hours ENERGY-DISPERSIVE X-RAY SPECTROSCOPY MAPPING ENCAPSULATED BENZOYL PEROXIDE (E-BPO) ENCAPSULATION IS DESIGNED TO ALLOW FOR CONTINUOUS FLOW

Complete encapsulation allows stabilization of tretinoin in the presence of BPO SEM PICTURE Silica-based shell allows for slow delivery of tretinoin to the skin over time SEM PICTURE ENCAPSULATED TRETINOIN (E-TRETINOIN) ENCAPSULATION IS DESIGNED TO ENHANCE STABILITY

CHRONIC CONDITION WITH POOR ADHERENCE TO CURRENT TREATMENTS THE CHALLENGE Papulopustular RosaceaChronic, inflammatory condition that primarily affects the face and is often characterized by flushing, redness, inflamed bumps, and pustules How is it Treated?Topical antimicrobials (metronidazole, clindamycin)Topical anti-mite (ivermectin)Systemic antibiotics (minocycline, doxycycline) Current Treatment ShortfallsInsufficient efficacy resulting in poor adherenceSystemic side effectsContributing to antibiotic resistance UNMET NEED IN PAPULOPUSTULAR ROSACEA

Encapsulation was designed to allow the BPO to slowly migrate from the microcapsules to help reduce irritationNDA submission expected in 2Q/20Potential to be the first single-active BPO approved by the FDA as a prescription drug product Encapsulated Benzoyl Peroxide Cream, 5% SOL-GEL SOLUTION*EPSOLAY® * EPSOLAY is investigational. Safety and efficacy have not been established

Two Parallel, Multicenter, Double-Blinded, Randomized, Vehicle-Controlled Studies, 2:1 Ratio, QD EPSOLAY® PHASE III STUDIES

Inclusion Criteria How is it Treated? Investigator Global Assessment (IGA) Definition ≥18 years old; “Moderate” or “Severe” acne; ≥15 to ≤70 inflammatory lesions; ≤2 nodules Weeks 2, 4, 8, 12 (end of study) “Clear”: Skin clear of inflammatory papules or pustules“Almost Clear”: Very few small papules or pustules and very mild dull erythema is present“Mild”: Few small papules or pustules and mild dull or light pink erythema is present“Moderate”: Several to many small or larger papules or pustules and moderate light to bright red erythema is present“Severe”: Numerous small and/or larger papules or pustules and severe erythema that is bright red to deep red is present Proportion of patients with IGA “Clear” or “Almost Clear” relative to baseline at Week 12Absolute mean change in inflammatory lesion counts from baseline to Week 12 Primary Endpoints TWO CO-PRIMARY EFFICACY ENDPOINTS AT WEEK 12 PHASE III DESIGN
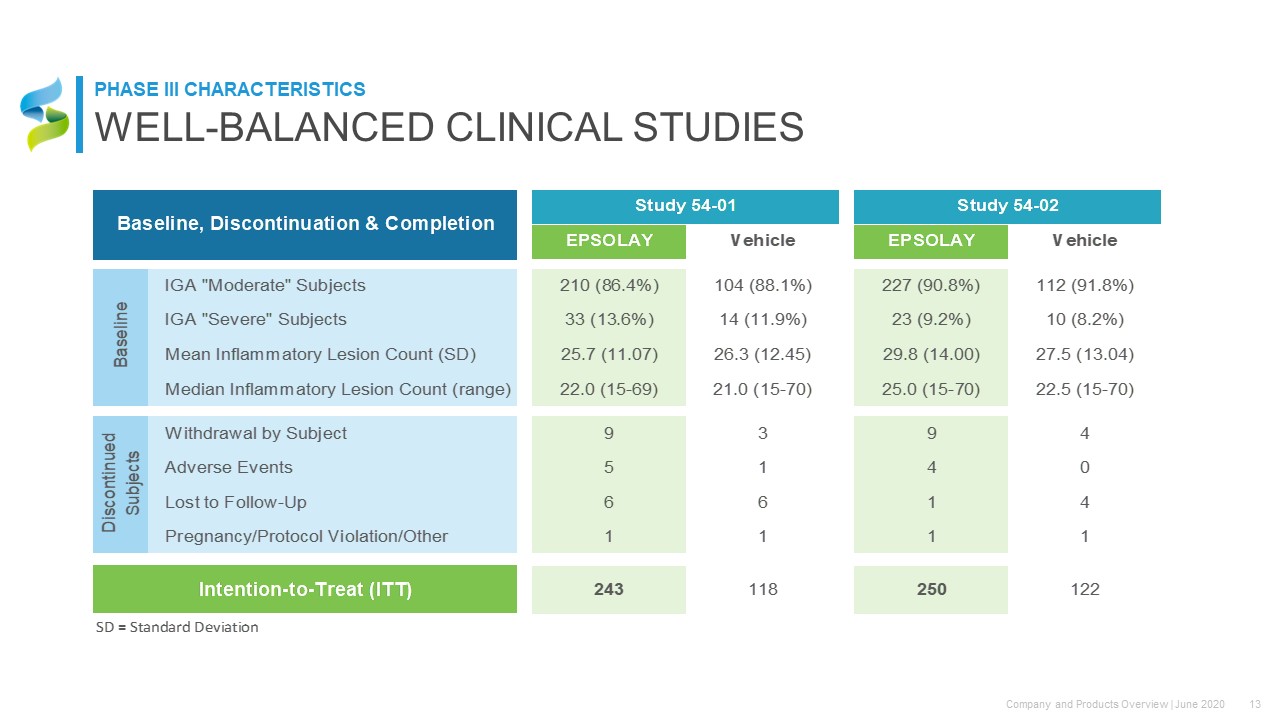
WELL-BALANCED CLINICAL STUDIES PHASE III CHARACTERISTICS

P<0.001 P<0.001 Change from Baseline in Inflammatory Lesion Count Success in IGA Week 12Success in IGA (ITT)�� Week 12Inflammatory Lesion Count Change from Baseline (ITT) Study 54-01 Study 54-02 SUCCESS IN PRIMARY ENDPOINTS PHASE III RESULTS P<0.001 P<0.001 Study 54-01 Study 54-02
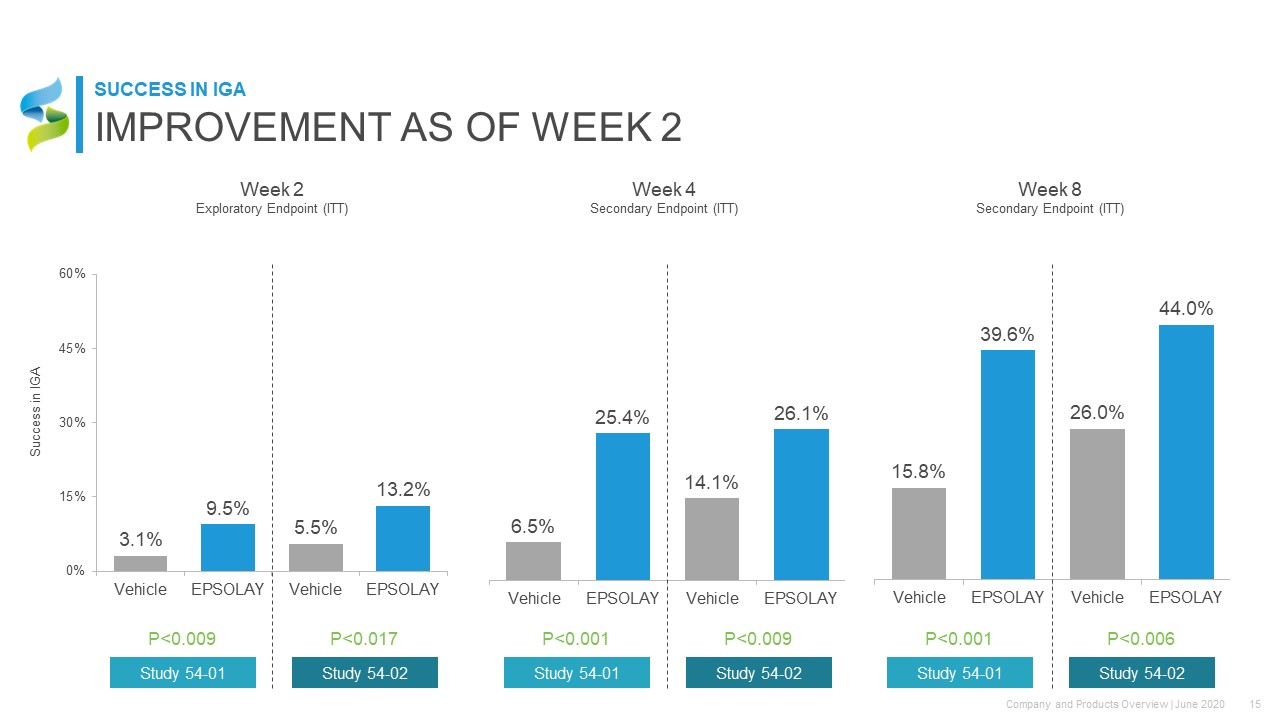
Success in IGA Week 2Exploratory Endpoint (ITT) Week 8Secondary Endpoint (ITT) P<0.009 P<0.017 Study 54-01 Study 54-02 P<0.001 P<0.009 Study 54-01 Study 54-02 P<0.001 P<0.006 Study 54-01 Study 54-02 Week 4Secondary Endpoint (ITT) IMPROVEMENT AS OF WEEK 2 SUCCESS IN IGA
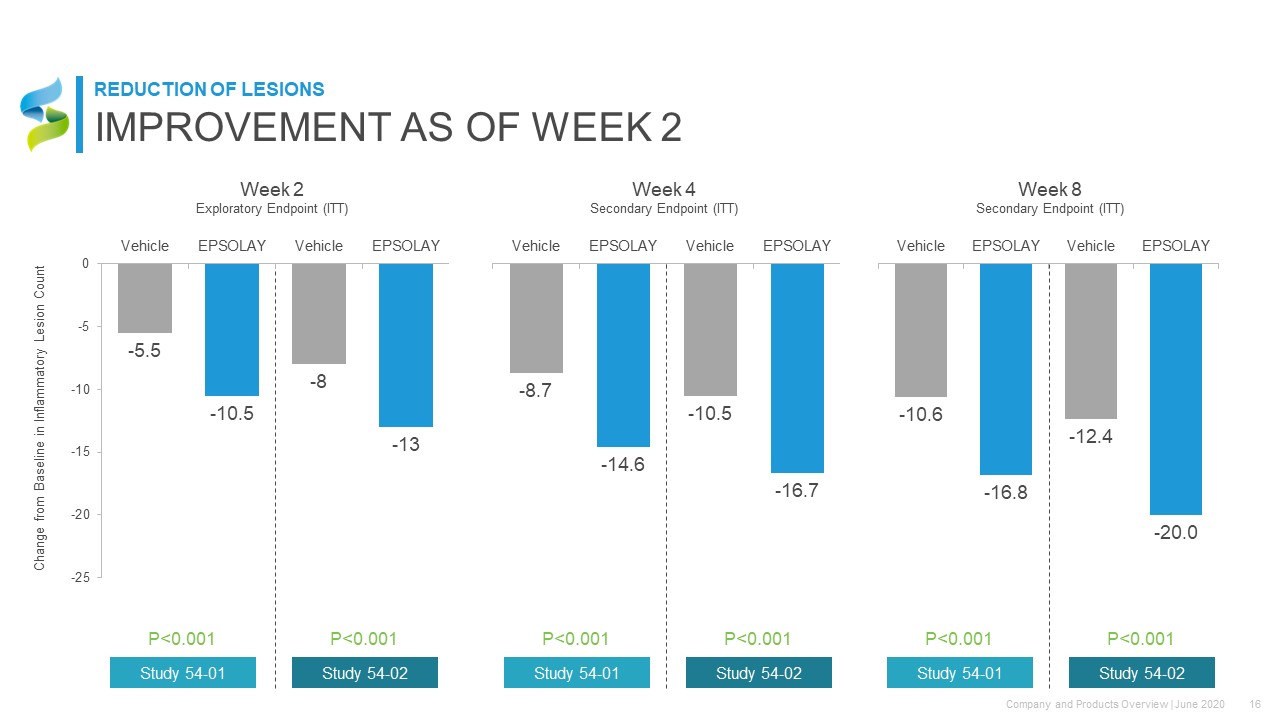
Week 2Exploratory Endpoint (ITT) Week 4Secondary Endpoint (ITT) Week 8Secondary Endpoint (ITT) Change from Baseline in Inflammatory Lesion Count P<0.001 P<0.001 Study 54-01 Study 54-02 P<0.001 P<0.001 Study 54-01 Study 54-02 P<0.001 P<0.001 Study 54-01 Study 54-02 IMPROVEMENT AS OF WEEK 2 REDUCTION OF LESIONS

ONSET OF ACTION AS OF WEEK 2 Subject 116-009 || 41 years old | Female | White | Not Hispanic or Latino* * Individual results vary BASELINE “Severe”; 31 inflamed lesions WEEK 2 “Clear”; No inflamed lesions WEEK 4 “Clear”; No inflamed lesions WEEK 12 “Almost Clear”; 1 inflamed lesion WEEK 8 “Clear”; No inflamed lesions

Phase III Studies Followed by 40 Weeks Long-Term Safety Study Extension Percentage of Subjects IMPROVEMENT IN IGA* LONG-TERM SAFETY STUDY * This study was not designed for efficacy; however, efficacy was evaluated. Interpret results with caution

Success in IGA * Sol-Gel did not conduct a head-to-head comparison trial or study. The results described above are for illustrative purposes only and should not be construed as conclusions to be drawn as if we conducted a head-to-head comparison trial or study IMPROVEMENT OVER TIME SIDE-BY-SIDE WITH HISTORICAL RESULTS*

Success in IGA Inflammatory Lesion Percent Change from Baseline 10-week study EPSOLAY® 16-week studyPer os 12-week study 12-week study 12-week study * Sol-Gel did not conduct a head-to-head comparison trial or study. The results described above are for illustrative purposes only and should not be construed as conclusions to be drawn as if we conducted a head-to-head comparison trial or study Baseline Characteristics of Active Arm IGA Severe 33 23 82 113 26 65 0 52 48 51 71 Moderate 210 227 369 346 172 418 557 67 77 444 443 Mild 0 0 0 0 0 0 0 8 17 0 0 Inflammatory Lesions 25.7 29.8 31.0 33.3 21.6 21.7 18.3 19.5 20.5 28.5 30.0 12-week study Difference from Vehicle PRIMARY ENDPOINTS SIDE-BY-SIDE WITH HISTORICAL RESULTS*

TREATMENT-EMERGENT ADVERSE EVENTS PRIMARILY MILD-TO-MODERATE

% of Subjects Week 12 % of Subjects DRYNESS SCALING ITCHING BURNING/STINGING None Mild Moderate Severe Baseline Study 54-01 LOCAL SKIN IRRITATIONS FEWER AT WEEK 12 THAN AT BASELINE

DRYNESS SCALING ITCHING BURNING/STINGING Study 54-02 % of Subjects Week 12 % of Subjects Baseline LOCAL SKIN IRRITATIONS COMPARABLE TO VEHICLE None Mild Moderate Severe

MULTIFACTORIAL DISEASE REQUIRING POWERFUL COMBINATION TREATMENTS THE CHALLENGE Acne VulgarisA multifactorial disease of the pilosebaceous unit, involving abnormalities in sebum production, follicular epithelial desquamation, bacterial proliferation, and inflammation How is it Treated?Topical BPO, retinoids (such as tretinoin, adapalene), antibiotics, and their combinationsOral Isotretinoin and antibiotics Current Treatment ShortfallsInsufficient efficacy negatively affects self-esteemSystemic side effectsContributes to antibiotic resistance UNMET NEED IN ACNE VULGARIS

Encapsulated Benzoyl Peroxide 3% &Encapsulated Tretinoin 0.1%, Cream SOL-GEL SOLUTION*TWYNEO® * TWYNEO is investigational. Safety and efficacy have not been established Encapsulation was designed to stabilize tretinoin and to enable both tretinoin and BPO to slowly migrate from their microcapsules to help reduce irritationNDA submission expected in 2H/20Potential to be first FDA-approved acne treatment that contains fixed-dose combination of BPO and tretinoin

Two Parallel, Multicenter, Double-Blinded, Randomized, Vehicle-Controlled Studies, 2:1 Ratio, QD TWYNEO® PHASE III STUDIES

Inclusion Criteria Visits Investigator Global Assessment (IGA) Definition ≥9 tears old; “Moderate” or “Severe” acne; ≥20 to ≤100 inflammatory lesions; ≥30 to ≤150 non-inflammatory lesions; ≤2 cysts/nodules Weeks 2, 4, 8, 12 (end of study) “Clear”: Normal, clear skin with no evidence of acne vulgaris“Almost Clear”: Rare non-inflammatory lesions present, with rare non-inflamed papules (papules must be resolving and may be hyperpigmented, though not pink-red) “Mild”: Some non-inflammatory lesions are present, with few inflammatory lesions (papules/pustules only; no nodulo-cystic lesions)“Moderate”: Multiple Non-inflammatory lesions and, inflammatory lesions are evident (several to many comedones and papules/pustules, and there may or may not be one small nodulo-cystic lesion)“Severe”: Inflammatory lesions are more apparent, many comedones and papules/pustules, there may or may not be a few nodulo-cystic lesions Proportion of subjects with an assessment of "Clear" or "Almost Clear" and with at least a 2-grade improvement in IGA from baseline at Week 12Absolute change in inflammatory lesion counts from baseline at Week 12Absolute change in non-inflammatory lesion counts from baseline at Week 12 Primary Endpoints THREE CO-PRIMARY EFFICACY ENDPOINTS AT WEEK 12 PHASE III DESIGN
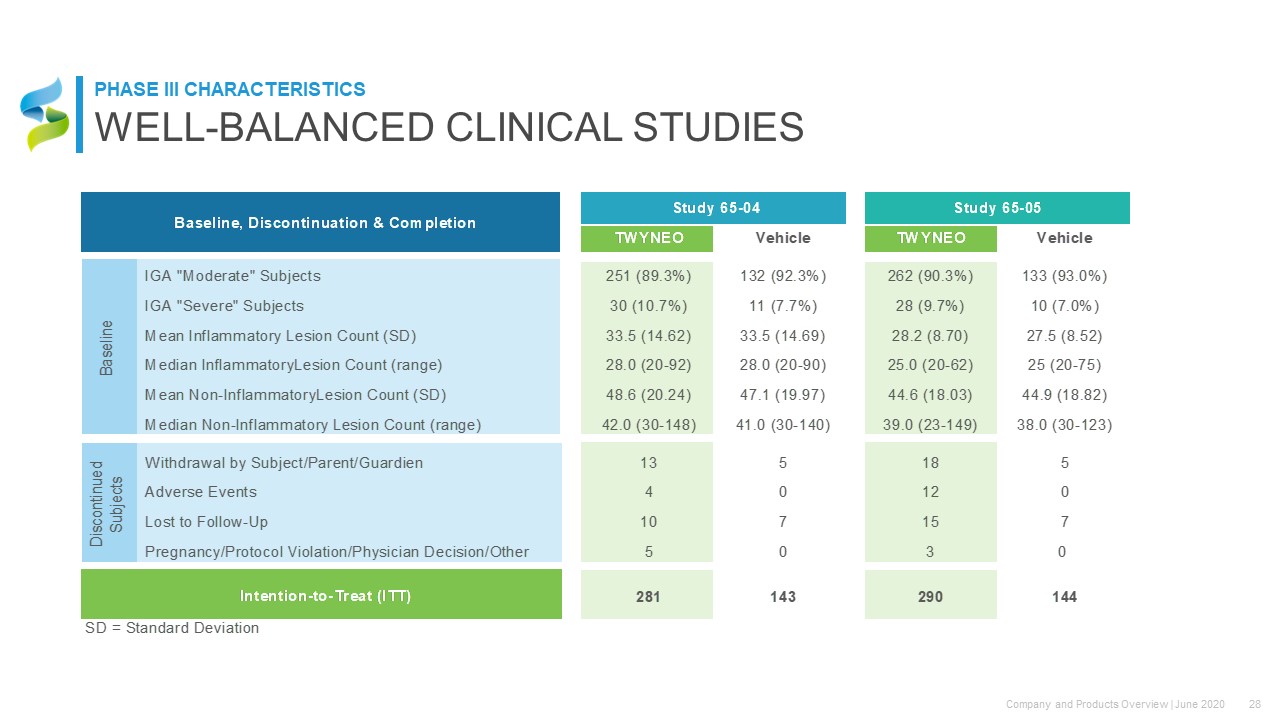
WELL-BALANCED CLINICAL STUDIES PHASE III CHARACTERISTICS

Success in IGA Week 12Success in IGA (ITT) P<0.001 Study 65-04 P<0.017 Study 65-05 SUCCESS IN IGA PHASE III RESULTS
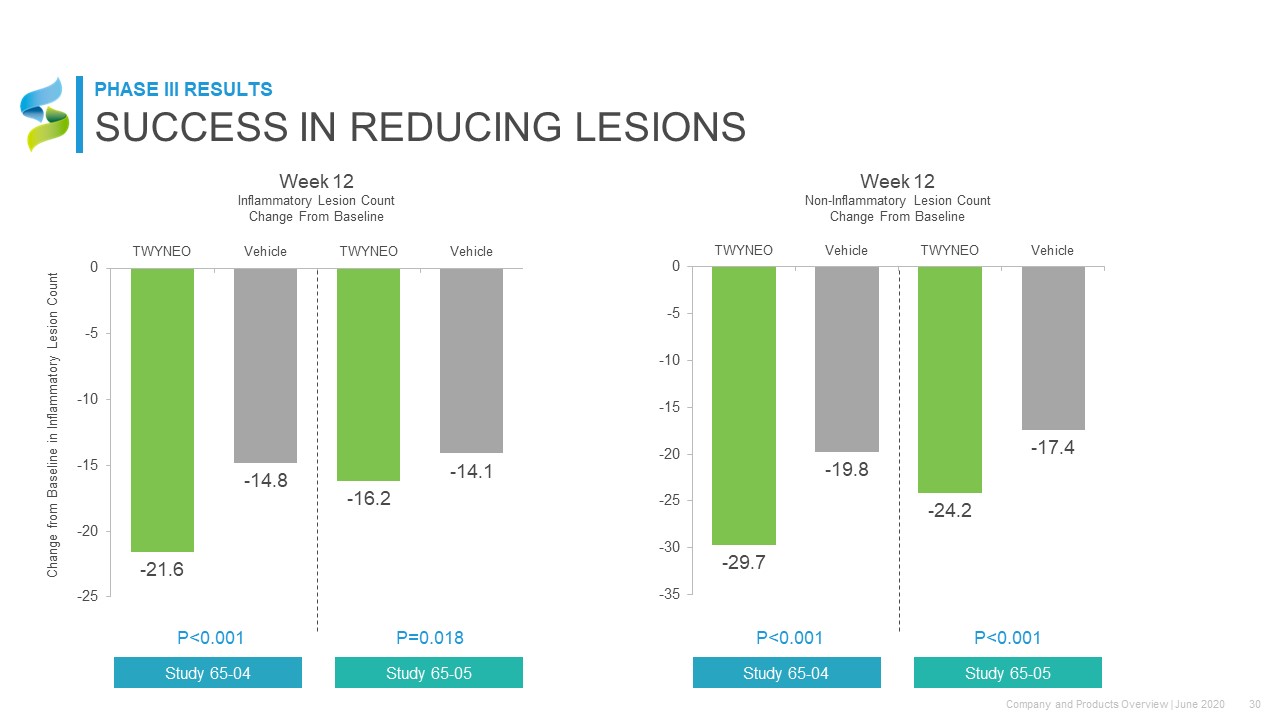
Week 12Inflammatory Lesion Count Change From Baseline Change from Baseline in Inflammatory Lesion Count P<0.001 Study 65-04 P=0.018 Study 65-05 P<0.001 Study 65-04 P<0.001 Study 65-05 Week 12Non-Inflammatory Lesion Count Change From Baseline SUCCESS IN REDUCING LESIONS PHASE III RESULTS

IMPROVEMENT IN SEVERE PATIENT Subject 507-003 || 18 years old | Female | White | Not Hispanic or Latino* * Individual results vary BASELINE “Severe”; 29 inflamed lesions31 non-inflamed lesions; 1 nodule WEEK 12 “Moderate”; 9 inflamed lesions5 non-inflamed lesions; No nodules

Trials with Highest Difference in IGA Between the Active Arm and the Vehicle Arm * Sol-Gel did not conduct a head-to-head comparison trial or study. The results described above are for illustrative purposes only and should not be construed as conclusions to be drawn as if we conducted a head-to-head comparison trial or study Success in IGA Normalized to Vehicle TWYNEO® SUCCESS IN IGA SIDE-BY-SIDE WITH HISTORICAL RESULTS*

Moderate Subjects at Baseline Only Success in IGA Normalized to Vehicle SUCCESS IN IGA SIDE-BY-SIDE WITH HISTORICAL RESULTS* Trials with Highest Difference in IGA Between the Active Arm and the Vehicle Arm TWYNEO® * Sol-Gel did not conduct a head-to-head comparison trial or study. The results described above are for illustrative purposes only and should not be construed as conclusions to be drawn as if we conducted a head-to-head comparison trial or study

TREATMENT-EMERGENT ADVERSE EVENTS PRIMARILY MILD-TO-MODERATE

DRYNESS SCALING ITCHING BURNING None Mild Moderate Severe ERYTHEMA PIGMENTATION STINGING % of Subjects % of Subjects % of Subjects Study 65-04 Week 12 Baseline Week 2 �� LOCAL SKIN REACTIONS MILD AND IMPROVED OVER TIME

DRYNESS SCALING ITCHING BURNING ERYTHEMA PIGMENTATION STINGING % of Subjects % of Subjects % of Subjects Study 65-05 Week 12 Baseline Week 2 LOCAL SKIN REACTIONS MILD AND IMPROVED OVER TIME None Mild Moderate Severe

EPSOLAY is protected until 2032 by granted patents and until 2040 by pending patentTWYNEO is protected until 2038 by granted patents and until 2040 by pending patent23 pending patent applications for erlotinib, tapinarof and roflumilast in various skin conditions (as of June 16, 2020) BROAD LONG-TERM INTELLECUAL PROPERTYESTATE

COMMERCIALIZATION& FINANCIALS

Source: IQVIA; Year 2019 PAPULOPUSTULAR ROSACEA US MARKET 2019 (IN $US) Oral vsTopical Topical Generic vsTopical Branded Branded Topicals are Important Segment

Branded Topical Combinations are Important SegmentTretinoin is the Most Prescribed Topical Retinoid ACNE VULGARIS US MARKET 2019 (IN $US) Adapalene vsTretinoin Oral vsTopical Topical Generic vsTopical Branded Combinations vs Single Active Source: IQVIA; Year 2019

Sources: NaviSync LLC (Morristown, NJ), Sol-Gel Managed Market Access for Acne and Rosacea, July 2019 NaviSync LLC (Morristown, NJ), Twyneo Payer Market Research Topline Summary, February 2020 TWYNEO® EPSOLAY® “All respondents recognized the product as a unique molecule for rosacea”“Near unanimous recognition as additional option for rosacea”“If priced and rebated similarly to the covered products, coverage seems likely” “Unique MOA will qualify it for formulary addition, price will determine its position”“If you price it like Epiduo, it will be managed like Epiduo”“If similarly priced with better tolerability, it would become preferred brand” EPSOLAY & TWYNEO ARE COMPELLING ENOUGH TO DRIVE PAYOR COVERAGE

Efficiently Reaching 80% Dermatology TRx in Acne and Papulopustular Rosacea 45-62Sales Reps 3,300Dermatology Offices 6,500Dermatologists 80%Potential Market Value 6,000NPs/PAs Source: Syneos Health (Morrisville, NC), Sol-Gel Market Analysis, June 2019 LEAN COMMERCIALIZATION APPROACH

Seven collaborations with Perrigo with 50/50 gross profit sharingIn February 2019, Perrigo launched acyclovir cream, 5%, developed in collaboration with Sol-Gel. As of today this is the only generic product on the market other than an authorized generic. This product generated $22.8 million in net revenues in 2019 and 3.4 million in net revenues in 1Q/20In January 2018, Perrigo received tentative approval from the FDA for ivermectin cream, 1%, developed in collaboration with Sol-Gel. Perrigo was second to file LUCRATIVE GENERIC PIPELINE

Gross proceeds of $86.3 million raised in IPO on February 5, 2018Gross proceeds of $11.5 and $23 million raised in public follow-on offerings on August 12, 2019 and February 13, 2020, respectivelyAdditional $5 million investment by controlling shareholder in April 202022,972,335 Ordinary Shares as of April 13, 2020$3.4 million net revenues from generic products in Q1/2020 $66.2 million in cash and investments as of March 31, 2020Cash resources will enable funding of operational and capital expenditure requirements into mid-2021Sol-Gel does not plan to raise additional dilutive capital to fund pre-commercialization activities STRONG FINANCIAL PROFILE

LOOKING FORWARD

PALMOPLANTAR KERATODERMA SGT-210 WHAT’S AHEAD Palmoplantar keratoderma (PPK) is a group of skin conditions characterized by thickening of the skin on the palms of the hands and soles of the feetPhase I proof-of-concept study for erlotinib gel in PPK is ongoing

2019 2020 2021 NDA submissionfor EPSOLAY Phase I results for SGT-210 Potential FDA approvaland launch of EPSOLAY Potential FDA approvaland launch of TWYNEO Revenues from generics DONE Phase III results for EPSOLAY DONE Granted patent forTWYNEO until 2038 DONE Phase I studyfor SGT-210 IN PROGRESS NDA submissionfor TWYNEO Phase III results for TWYNEO DONE RECENT MILESTONES& NEXT STEPS

NASDAQ: SLGL