Exhibit 99.1
The following presentation was made available to employees of Dova Pharmaceuticals, Inc.:
Notice to Investors
The tender offer for the outstanding shares of Dova Pharmaceuticals Inc. (“Dova”) common stock has not yet commenced. This presentation is being given to Dova’s employees for informational purposes only and such presentation is neither an offer to purchase nor a solicitation of an offer to sell any shares of Dova’s common stock, nor is it a substitute for the tender offer materials that Swedish Orphan Biovitrum AB (publ) (“Sobi”) and Dova will file with the United States Securities and Exchange Commission (the “SEC”) on Schedule TO. At the time any such tender offer is commenced, Sobi will file a Tender Offer Statement, containing an offer to purchase, a form of letter of transmittal and other related tender offer documents with the SEC, and Dova will file a Solicitation/Recommendation Statement relating to such tender offer with the SEC. DOVA’S STOCKHOLDERS ARE STRONGLY ADVISED TO READ THESE TENDER OFFER MATERIALS CAREFULLY AND IN THEIR ENTIRETY WHEN THEY BECOME AVAILABLE, AS THEY MAY BE AMENDED FROM TIME TO TIME, BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION ABOUT SUCH TENDER OFFER THAT DOVA’S STOCKHOLDERS SHOULD CONSIDER PRIOR TO MAKING ANY DECISIONS WITH RESPECT TO SUCH TENDER OFFER. Once filed, stockholders of Dova will be able to obtain a free copy of these documents at the website maintained by the SEC at www.sec.gov. Free copies of the offer to purchase, the related letter of transmittal and certain other offering documents will be made available by Sobi and when available may be obtained by directing a request to the Information Agent for the tender offer which will be named in the Schedule TO.
This presentation may contain “forward-looking statements.” Forward-looking statements include all statements that are not historical facts, including, among other things, plans, strategies, expectations for the future, statements regarding the expected timing of filings and approvals relating to the transaction, the expected timing of the completion of the transaction and the ability to complete the transaction or to satisfy the various closing conditions. Words such as “anticipate(s)”, “expect(s)”, “intend(s)”, “plan(s)”, “target(s)”, “project(s)”, “believe(s)”, “will”, “aim(s)”, “would”, “seek(s)”, “estimate(s)” and similar expressions are intended to identify such forward-looking statements.
Forward-looking statements are based on Sobi’s current expectations and beliefs, and Sobi can give no assurance that its expectations or beliefs will be attained. These forward-looking statements are not a guarantee of future performance and are subject to a number of known and unknown risks, uncertainties and other factors that could cause actual results or events to differ, possibly materially, from the expectations or estimates reflected in such forward-looking statements, including, among others. The forward-looking statements contained in this presentation speak only as of the date of this presentation, and Sobi does not undertake any obligation to revise or update any forward-looking statements to reflect new information, future events or circumstances after the date of the forward-looking statement. If one or more of these statements is updated or corrected, investors and others should not conclude that additional updates or corrections will be made.

30th September 2019 Introduction to Sobi
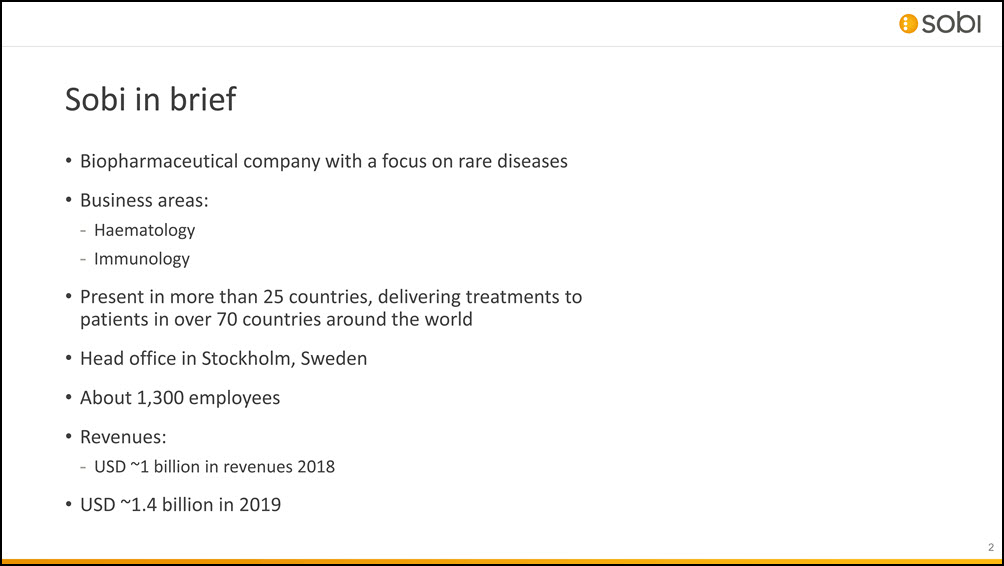
Sobi in brief 2 Biopharmaceutical company with a focus on rare diseasesBusiness areas:HaematologyImmunologyPresent in more than 25 countries, delivering treatments to patients in over 70 countries around the worldHead office in Stockholm, SwedenAbout 1,300 employeesRevenues:USD ~1 billion in revenues 2018USD ~1.4 billion in 2019

Strategic Direction Become a leader in rarediseases Diversify revenues inhematology Expand geographicfootprint Strengthenlate-stage pipeline 3
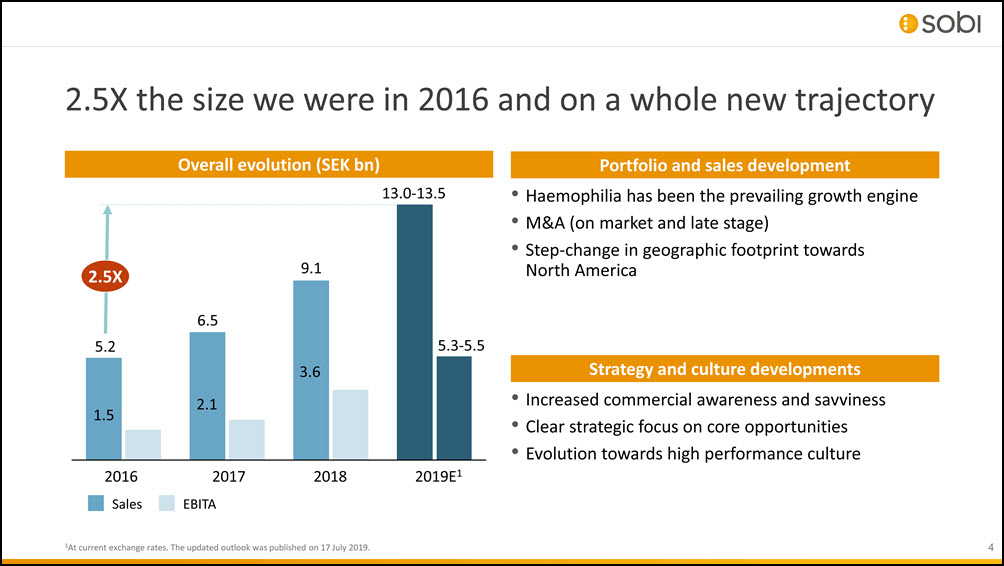
2.5X the size we were in 2016 and on a whole new trajectory 1At current exchange rates. The updated outlook was published on 17 July 2019. Overall evolution (SEK bn) 2018 6.5 2019E1 5.2 1.5 2.1 9.1 3.6 13.0-13.5 5.3-5.5 2.5X 2016Sales 2017EBITA Haemophilia has been the prevailing growth engineM&A (on market and late stage)Step-change in geographic footprint towards North America Portfolio and sales development Increased commercial awareness and savvinessClear strategic focus on core opportunitiesEvolution towards high performance culture Strategy and culture developments 4

Short Term Mid-term Haematology BIVV001 MEDI8897 Sobi’s portfolio is well positioned for growth Immunology 6

Fundamentally attractive rare disease marketStrong and well-positioned portfolio for growthContinue to pursue M&A Ample opportunity to take Sobi to the next level in the raredisease space 7

We currently have leading on- market products for improving the lives of people with haemophilia Current TPO market is ~USD 2bn and estimated to grow at ~5 per cent per year Current launch in US in ITP and near-term launch in Europe Doptelet is expected to be first in class in the CIT indication Interim results suggest that BIVV001 has the possibility enable patients to live an active life with very few limitations We have brought the first innovation in 20 years in a very conservative market Sobi will bring another game changer to Haemophilia market with BIVV001 mid-term Significant room to grow in a morecompetitive environment Doptelet will create a strong cadence of launches across multiple geographies & indications BIVV001 8 Haematology will be a dynamically growing franchise in the yearsto come

Fully integrated functions in the US 125 total employees, including North American team of 300 total employees Sales force team supporting Synagis, Gamifantand Kineret 9 Medical affairs across indications Strong support functions for commercialisation Sales reps focused on haematology in the US R&D, focused on developing Doptelet in furtherindications Supporting other critical functions

What happens next? 10 The next few weeks will be business as usualWe will get to know the teamsWe expect to close the transaction mid-November 2019Our plan will be to keep the focus on ITP launch and CIT approvalDova will become a key part of the Sobi family

rarestrength