
CORPORATE OVERVIEW August 2024 ®

2 Forward-looking statements This presentation may contain “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995 relating to our business, operations, and financial conditions, including but not limited to express or implied statements regarding the current beliefs, expectations and assumptions regarding the future of our business, future plans and strategies, , including statements regarding the estimated market for our product candidates, if approved, our development plans, our preclinical and clinical results and other future conditions, including our cash runway, and the safety, efficacy, and regulatory and clinical design or progress, potential regulatory submissions, approvals and timing thereof of any of our product candidates. Any forward-looking statements in this presentation are based on management’s current expectations and beliefs and are subject to a number of risks, uncertainties and important factors that may cause actual events or results to differ materially from those expressed or implied by any forward-looking statements contained in this presentation, including, without limitation, risks relating to: (i) the success and timing of our ongoing clinical trials, (ii) the success and timing of our product development activities and initiating clinical trials, (iii) the success and timing of our collaboration partners’ product development activities, (iv) the timing of and our ability to obtain and maintain regulatory approval of any of our product candidates, (v) our plans to research, discover and develop additional product candidates, (vi) our ability to enter into collaborations for the development of new product candidates, (vii) our ability to establish manufacturing capabilities, and our collaboration partners’ abilities to manufacture our product candidates and scale production, (viii) our ability to meet any specific milestones set forth herein, and (ix) the potential addressable market sizes for product candidates. New risks and uncertainties may emerge from time to time, and it is not possible to predict all risks and uncertainties. Except as required by applicable law, we do not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise. Although we believe the expectations reflected in such forward-looking statements are reasonable, we can give no assurance that such expectations will prove to be correct. Accordingly, readers are cautioned not to place undue reliance on these forward-looking statements. For further information regarding the risks, uncertainties and other factors that may cause differences between our expectations and actual results, you should review the “Risk Factors” section of our Annual Report on Form 10-K for the year ended December 31, 2023 filed with the Securities and Exchange Commission (“SEC”) and our other filings with the SEC. Certain information contained in this presentation relates to or is based on studies, publications, surveys and other data obtained from third-party sources and our own internal estimates and research. While we believe these third-party sources to be reliable as of the date of this presentation, we have not independently verified, and make no representation as to the adequacy, fairness, accuracy or completeness of, any information obtained from third-party sources. In addition, all of the market data included in this presentation involves a number of assumptions and limitations, and there can be no guarantee as to the accuracy or reliability of such assumptions. Finally, while we believe our own internal research is reliable, such research has not been verified by any independent source.

3 4 >$7B 5 2 2027 Praxis is positioned to bring more innovation to patients Assets in late stage in 2024 Commercial opportunity across the portfolio Readouts within the next eighteen months Discovery platforms to optimize drug development Cash runway into

4 GENETICS Focus on therapeutic targets identified through human genetics TRANSLATIONAL TOOLS Translational tools validate potential of target and product candidate and can provide early proof of biology EFFICIENT & RIGOROUS Efficient, rigorous clinical development paths to proof- of-concept in humans applying an agile way of working PATIENT-GUIDED Patient-guided development strategies to deliver on what patients actually need Four pillars guide how we develop medicines
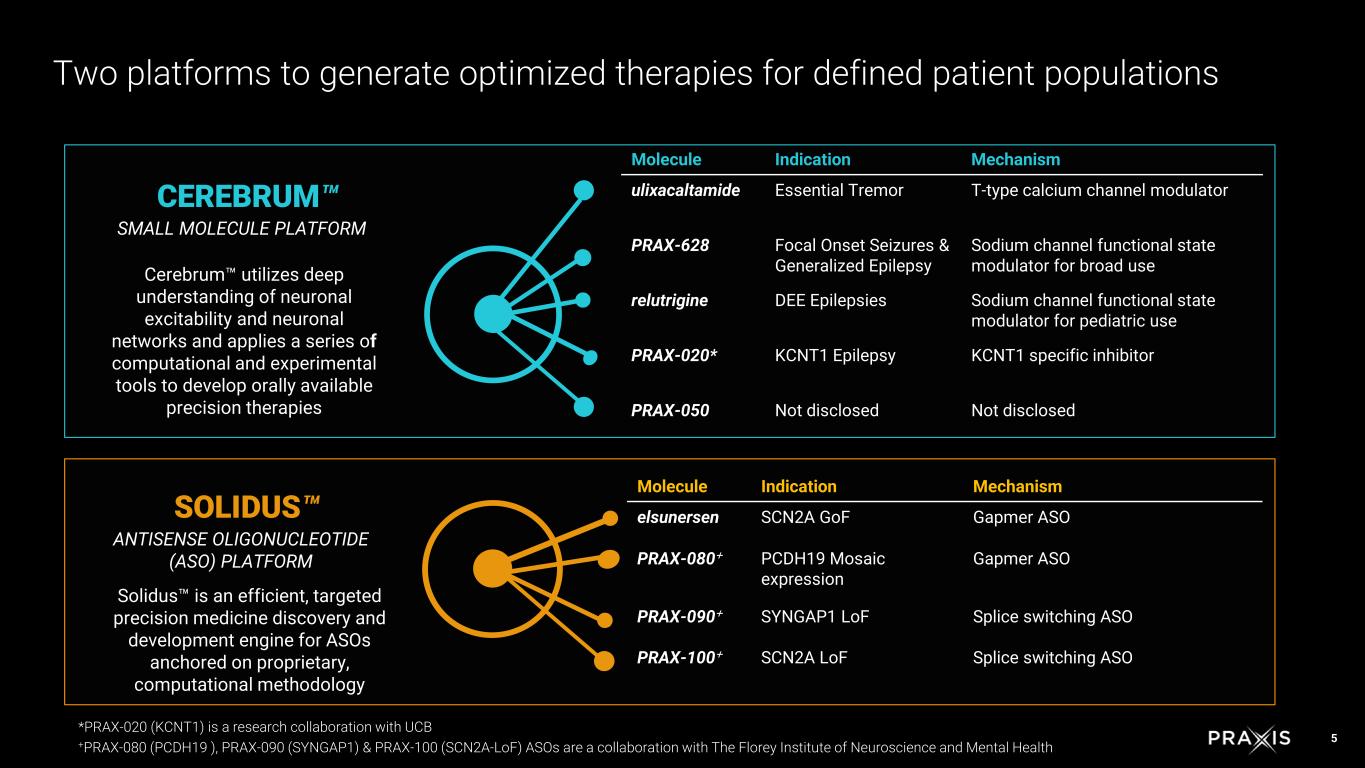
5 CEREBRUM SMALL MOLECULE PLATFORM Cerebrum utilizes deep understanding of neuronal excitability and neuronal networks and applies a series of computational and experimental tools to develop orally available precision therapies SOLIDUS ANTISENSE OLIGONUCLEOTIDE (ASO) PLATFORM Solidus is an efficient, targeted precision medicine discovery and development engine for ASOs anchored on proprietary, computational methodology Two platforms to generate optimized therapies for defined patient populations Molecule Indication Mechanism ulixacaltamide Essential Tremor T-type calcium channel modulator PRAX-628 Focal Onset Seizures & Generalized Epilepsy Sodium channel functional state modulator for broad use relutrigine DEE Epilepsies Sodium channel functional state modulator for pediatric use PRAX-020* KCNT1 Epilepsy KCNT1 specific inhibitor PRAX-050 Not disclosed Not disclosed Molecule Indication Mechanism elsunersen SCN2A GoF Gapmer ASO PRAX-080+ PCDH19 Mosaic expression Gapmer ASO PRAX-090+ SYNGAP1 LoF Splice switching ASO PRAX-100+ SCN2A LoF Splice switching ASO *PRAX-020 (KCNT1) is a research collaboration with UCB +PRAX-080 (PCDH19 ), PRAX-090 (SYNGAP1) & PRAX-100 (SCN2A-LoF) ASOs are a collaboration with The Florey Institute of Neuroscience and Mental Health

6 Program Pre clin Ph 1 Ph 2 Ph 3 Upcoming Catalyst Ulixacaltamide Essential Tremor ESSENTIAL3 Study1 placebo controlled Q4 2024 interim, 2H 2024 topline results ESSENTIAL3 Study 2 randomized withdrawal PRAX-628 Focal Onset Seizures & Generalized Epilepsy EMPOWER observational study Q3 2024 begin enrollment RADIANT open label 2H 2024 begin enrollment, 1H 2025 topline results POWER1 Phase 2/3 Q4 2024 begin enrollment, 2H 2025 topline results POWER2 Phase 2/3 1H 2025 begin enrollment Relutrigine DEEs EMBOLD SCN2A and SCN8A DEEs Q3 2024 topline results PRAX-020 KCNT1 Elsunersen SCN2A GoF DEE Phase 1/2 Enrolling Registrational Harmonize global protocol, 2H 2024 initiate PRAX-080 PCDH19 PRAX-090 SYNGAP1 PRAX-100 SCN2A LoF Four clinical stage assets and multitude of early-stage programs *PRAX-020 (KCNT1) is a research collaboration with UCB +PRAX-080 (PCDH19 ), PRAX-090 (SYNGAP1) & PRAX-100 (SCN2A-LoF) ASOs are a collaboration with The Florey Institute of Neuroscience and Mental Health CEREBRUM SMALL MOLECULE PLATFORM SOLIDUS ASO PLATFORM

7 CEREBRUM SMALL MOLECULE PLATFORM

8 Ulixacaltamide Milestones Q4 2024: Interim analysis of Essential3 Study 1 2H 2024: Topline results for Essential 3 2025: NDA filling

9 Essential tremor market is significantly underserved and ready for disruption $4bn+ US ET Market 1. GHOSH (2016) (P.231, C.1, PH.1, L.1-2), 2. Elble RJ. Curr Neurol Neurosci Rep. 2013 Jun;13(6):353. 3. Putzke JD, et al. J Neurol Neurosurg Psychiatry. 2006 Nov;77(11):1235-7. 4. Vetterick, C., Lyons, K.E., Matthews, L.G. et al. The Hidden Burden of Disease and Treatment Experiences of Patients with Essential Tremor: A Retrospective Claims Data Analysis. Adv Ther (2022). https://doi.org/10.1007/s12325-022-02318-8 • Essential Tremor (ET) is the most prevalent movement disorder • People with ET experience significant disturbance of their daily activities • Hallmark feature of ET is action tremor that primarily affects the hands2,3 • Almost all ET patients suffer from at least one comorbid condition (e.g., depression, anxiety, sleep disorders, cognitive dysfunction)4 Vast majority of patients are left without any treatment option • <30% of patients are eligible to receive propranolol due to other medications/health conditions • Of those who start propranolol >50% discontinue after only 1 month • Of those who start propranolol <20% still receive propranolol after 2 years ~2M ET patients seeking treatment 7M US Prevalence1

10 Speaking Dressing Using Keys Hygiene Pouring Working Writing Drinking from a glass Feeding with a spoon Carrying food trays, plates or similar items Overall disability with most affected task Each point reduction provides benefit to a patient • Improvement based on regaining function • ADL assessment performed by a physician • Aligned with FDA as primary endpoint for Essential3 studies Modified Activities of Daily Living 11 (mADL11) as Primary Endpoint TETRAS = TRG Essential Tremor Rating Assessment Scale; ADL = Activities of Daily Living Each item is individually scored, up to a total of 33 0 = Slightly abnormal. Tremor is present but does not interfere with __. 1 = Mildly abnormal. Spills a little. 2 = Moderately abnormal. Spills a lot or changes strategy to complete task. 3 = Severely abnormal. Cannot drink from a glass or uses straw or sippy cup. 11 items from the well-established TETRAS ADL scale

11 • Alignment with FDA on dose and primary endpoint • Phase 3 program design structured around patient needs • Robust recruitment strategy Essential1 Phase 2b study set foundation for the Essential3 Phase 3 program Sets up a clear path to registration • Strong efficacy signal with robust endpoint (mADL11) • Early clinical benefit in 8-Week Study • Long-term, durable benefit • Well-tolerated with a differentiated safety profile • Tested Phase 3 design concepts Validated the clinical hypothesis p = 0.042 ULIXACALTAMIDE (n=78) PLACEBO (n=38) Improvement in mADL11 in 8 weeks -2.69 -0.88 Im pr ov em en t Results from Essential1 study, p value is nominal

12 Using Essential1 to define clinical meaningfulness in essential tremor MSD=Meaningful Score Difference, PGI-C = Patient Global Impression of Change 8 6 4 2 0 -2 -4 -6 -8 -10 -12 -14 -16 -18 -20 • Patient response on mADL11 endpoint was well-correlated to PGI-C response • Aligned with recently issued guidance from Clinical Outcomes Assessment for novel endpoints ESSENTIAL1 ANCHOR-BASED ASSESSMENT PGI-C:mADL11
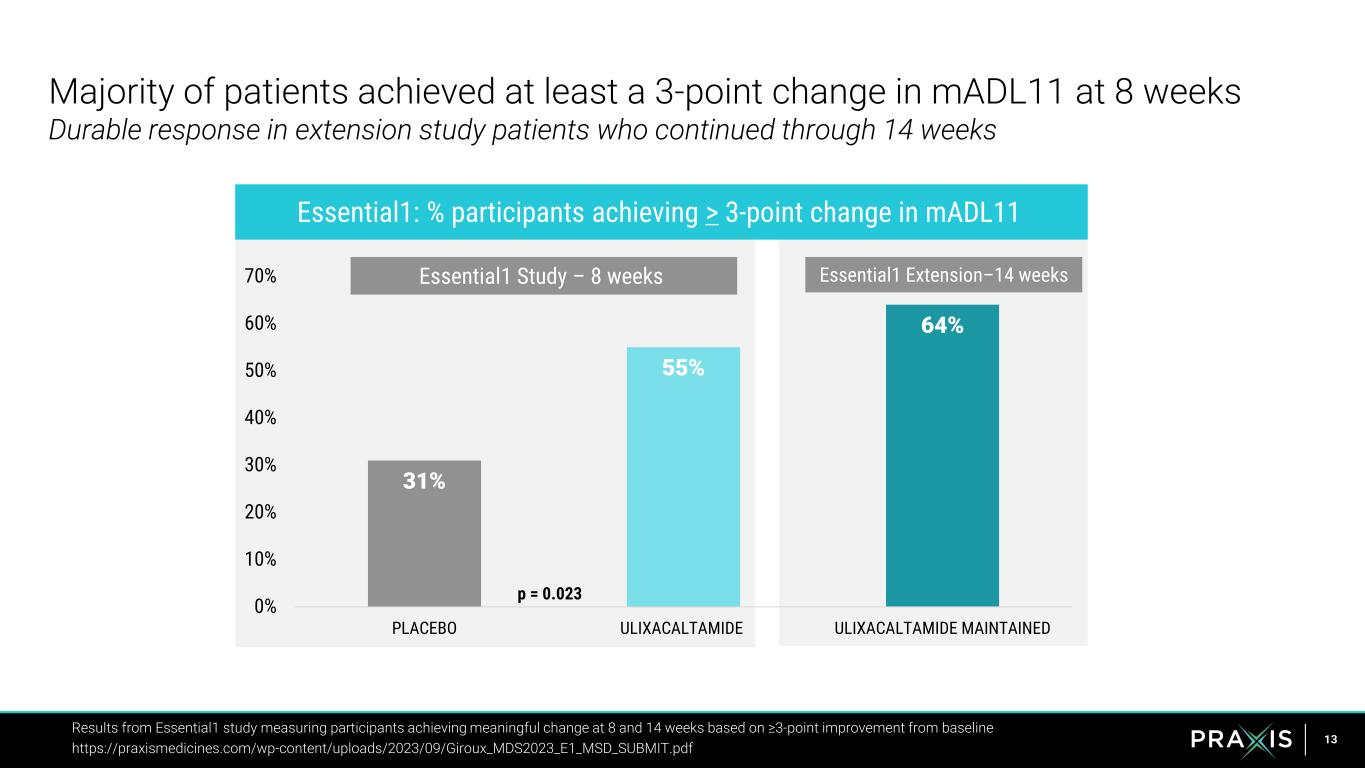
13 31% 55% 64% 0% 10% 20% 30% 40% 50% 60% 70% PLACEBO ULIXACALTAMIDE ULIXACALTAMIDE MAINTAINED Majority of patients achieved at least a 3-point change in mADL11 at 8 weeks Durable response in extension study patients who continued through 14 weeks Results from Essential1 study measuring participants achieving meaningful change at 8 and 14 weeks based on ≥3-point improvement from baseline https://praxismedicines.com/wp-content/uploads/2023/09/Giroux_MDS2023_E1_MSD_SUBMIT.pdf Essential1 Study – 8 weeks Essential1: % participants achieving > 3-point change in mADL11 Essential1 Extension–14 weeks p = 0.023
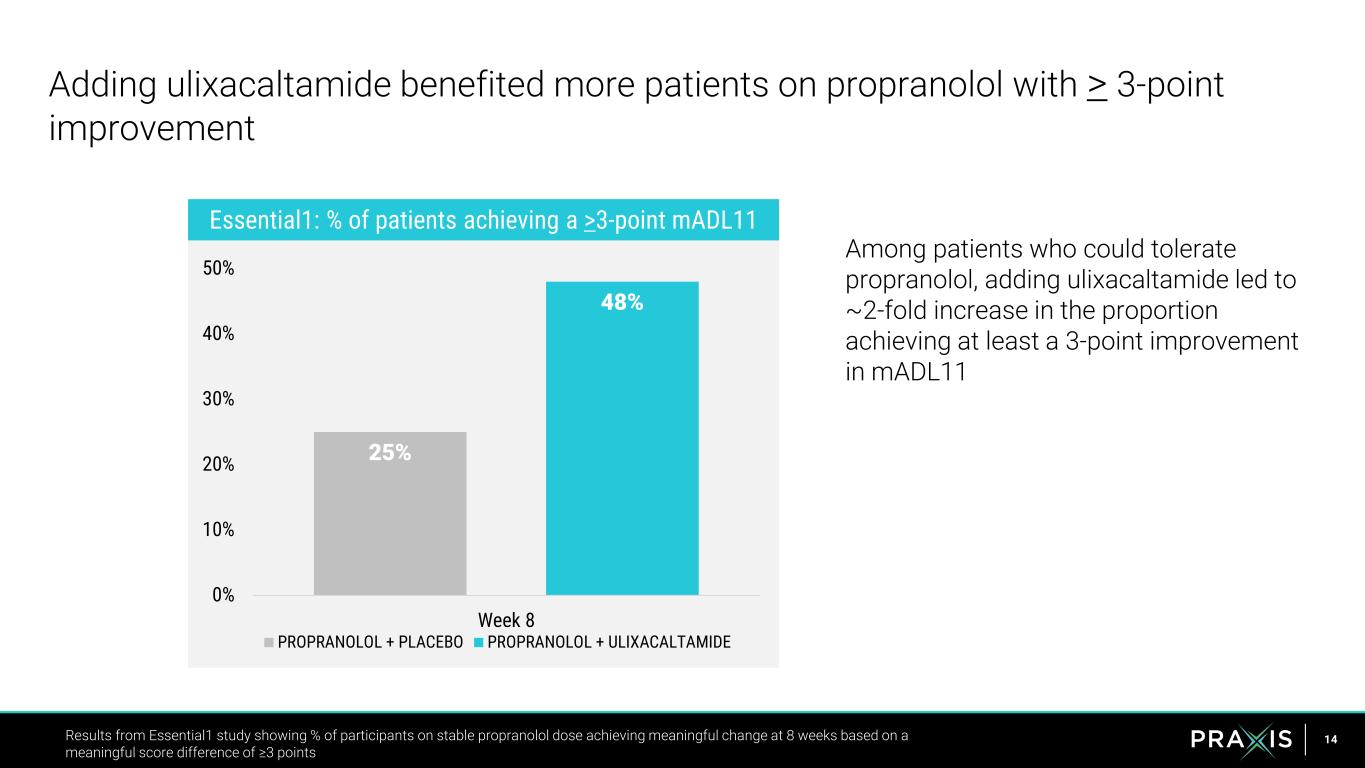
14 25% 48% 0% 10% 20% 30% 40% 50% Week 8 PROPRANOLOL + PLACEBO PROPRANOLOL + ULIXACALTAMIDE Adding ulixacaltamide benefited more patients on propranolol with > 3-point improvement Results from Essential1 study showing % of participants on stable propranolol dose achieving meaningful change at 8 weeks based on a meaningful score difference of ≥3 points Essential1: % of patients achieving a >3-point mADL11 Among patients who could tolerate propranolol, adding ulixacaltamide led to ~2-fold increase in the proportion achieving at least a 3-point improvement in mADL11

15 Essential3: An innovative Phase 3 program that optimizes all aspects of study conduct Single Recruiting Expert Review for Eligibility Screening Blinded Study Randomization Study 1: Placebo-controlled Parallel Group Study Ulixacaltamide Placebo Study 2: Randomized Withdrawal Study Ulixacaltamide Randomization 60 mg Placebo Long-term Safety Study Ulixacaltamide CT.gov NCT06087276 8 weeks 4 weeks 12 weeks

16 Essential3 Program is well powered Study Study 1 – Parallel Design Study 2 – Randomized Withdrawal Participants 400 200 Primary endpoint and power mADL11 Change from Baseline to Week 12 between ulixacaltamide and placebo 90% power to detect difference Difference in maintenance of response rate during the 4- week RW period between ulixacaltamide and placebo 90% power to detect difference Stratification Intention tremor status, family history, and propranolol use Main Secondary endpoints o TETRAS-ADL o CGI o PGI CGI = Clinical Global Impression (Severity); PGI = Patient Global Impression (Severity and Change)

17 Path to success De-risked Trial design based on key learnings from Essential1 Regulatory alignment based on successful End-of-Phase 2 meeting Efficient Focused execution Single protocol: Optimized screening, enrollment, analysis Streamlined Design Decentralized study to expand reach and reduce study burden to participants Patient-driven Approach mADL11 as a clinically meaningful primary endpoint NDA Readiness Clear path to filing in 2025 Interim Analysis Increases optionality, including potential for sample size re-estimation

18 PRAX-628 Milestones Q3 2024: Begin enrolling EMPOWER observational study 2H 2024: Begin enrolling RADIANT open label study Q4 2024: Begin enrolling POWER1 registrational study 1H 2025: Begin enrolling POWER2 registrational study 1H 2025: Topline results for RADIANT 2H 2025: Topline results for POWER1

19 The Praxis epilepsy portfolio targets significant unmet need in both the common and rare epilepsy markets >$3B+ US Common and Developmental Epilepsy Market Opportunity 3.5M US Prevalence for Common Epilepsy 14K+ US Prevalence for Rare Epilepsies covered by Praxis’ Portfolio1 1 SCN2A Gof, SCN2A LoF, SYNGAP1, PCDH19, SCN8A, KCNT1 2 PRAX-020 (KCNT1) is a research collaboration with UCB 3 PRAX-080 (PCDH19 ), PRAX-090 (SYNGAP1) & PRAX-100 (SCN2A-LoF) ASOs are a collaboration with The Florey Institute of Neuroscience and Mental Health 4 Poke G, Stanley J, Scheffer IE, Sadleir LG. Epidemiology of Developmental and Epileptic Encephalopathy and of Intellectual Disability and Epilepsy in Children Praxis Epilepsy Portfolio PRAX-628 PRAX-0202 PRAX-0803 PRAX-0903 PRAX-1003 elsunersen relutrigine >100k Developmental Epilepsies with high seizure burden using sodium channel blockers4

20 Differentiated Profile Goal: Preferential action against neuronal hyperexcitability PRAX-628: Precision medicine therapeutic for focal onset seizures and generalized epilepsy Next generation, functionally selective small molecule targeting the hyperexcitable states of sodium channels in the brain, with the potential to address limitations of current treatments • Ideal safety/tolerability profile • Achieves brain penetration • Rapidly achieves therapeutic concentrations without titration • Favorable half-life and PK profile • Optimized efficacy STIMULATION LEVEL epileptic range IDEAL DRUG healthy neuron non preferring drug NE UR AL A CT IV IT Y Untreated neuron

21 0.1 1 10 100 0 25 50 75 100 Dose (mg/kg) Pe rc en t p ro te ct ed PRAX-628 Cenobamate Lamotrigine XEN1101 Carbamazepine NBI-921352 PRAX-628 shows a differentiated pre-clinical profile Protective index (PI) measured as tolerability / efficacy (TD50 / ED50); MES = maximal electroshock seizure 1. https://praxismedicines.com/wp-content/uploads/2023/12/Anderson_AES2023_Predictive-Validity_Poster_Final.pdf; 2. Praxis data on file 3. https://praxismedicines.com/wp-content/uploads/2023/12/Kahlig_AES2023_628-In-Vivo_Poster_Final.pdf PRAX-628 has differentiated potency in the MES model3 Preclinical decision tree optimized for focal seizure drug discovery1 Progress Development Abandon Progress DevelopmentAbandon PI >2 PI >2 PI >2 PI < 2 PI < 2 PI < 2 Audiogenic MES (mouse) 6-Hz, 32mA (mouse) PRAX-628 Results2
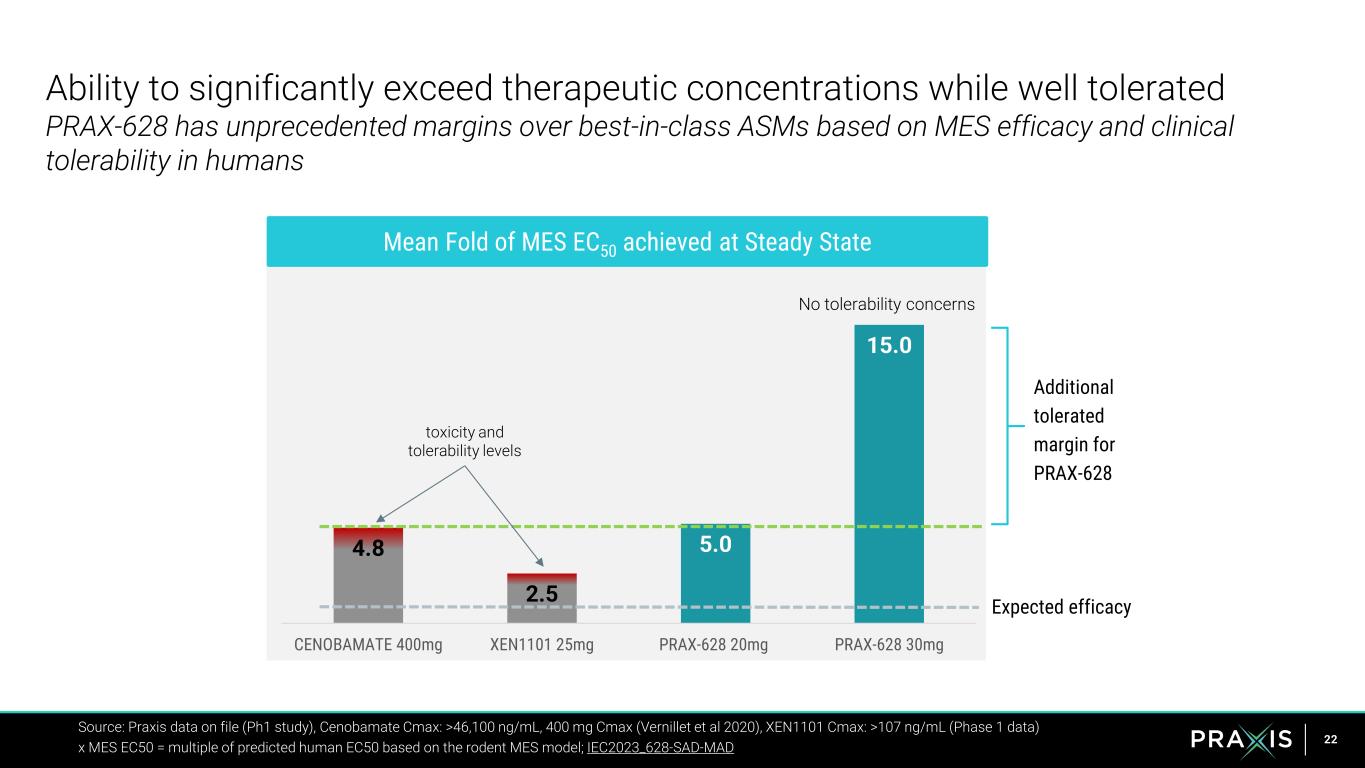
22 Source: Praxis data on file (Ph1 study), Cenobamate Cmax: >46,100 ng/mL, 400 mg Cmax (Vernillet et al 2020), XEN1101 Cmax: >107 ng/mL (Phase 1 data) x MES EC50 = multiple of predicted human EC50 based on the rodent MES model; IEC2023_628-SAD-MAD 4.8 2.5 5.0 15.0 CENOBAMATE 400mg XEN1101 25mg PRAX-628 20mg PRAX-628 30mg Mean Fold of MES EC50 achieved at Steady State Expected efficacy Additional tolerated margin for PRAX-628 toxicity and tolerability levels No tolerability concerns Ability to significantly exceed therapeutic concentrations while well tolerated PRAX-628 has unprecedented margins over best-in-class ASMs based on MES efficacy and clinical tolerability in humans

23 - 5 10 15 20 25 0 1 2 3 4 5 6 7 8 QE EG C OM PO SI TI VE C HA NG E HOURS POST DOSE 0 5 23 0 4 8 12 16 20 24 Cenobamate XEN1101 PRAX-628 • Composite endpoint from qEEG showed separation of drug versus placebo in the SAD and MAD cohorts • Difference between PRAX-628 and placebo significant for all doses at first point measured • Effect consistent with known PK profile • PRAX-628 achieves nearly complete coverage on Day 1 400 mg 25 mg 30 mg PRAX-628 composite qEEG change Hours of day 1 drug coverage Phase 1 study demonstrated brain activity and rapid achievement of therapeutic concentrations

24 • Patients must demonstrate PPR response during screening and baseline to be evaluable • Response Assessment • Partial: Reduction, other than to zero, in the number of generalized PPR events at any assessment period vs baseline • Complete: Reduction to zero in the number of generalized PPR events at any assessment period vs baseline • Safety monitored and PK samples collected during observation period The Phase 2 PRAX-628 Photo Paroxysmal Response (PPR) study demonstrated proof of concept; de-risks advancing to studies in focal and generalized epilepsy https://investors.praxismedicines.com/static-files/edc15000-7fcb-4d6d-b1de-1819898214a8 15 mg 45 mg Part A Part B Assessment Screening Baseline 24h Dose Categorical Response Response Rate 15 mg None 0% (0/5) Partial 20% (1/5) Complete 80% (4/5) 45 mg None 0% (0/3) Complete 100% (3/3) Evaluable Response 100% (8/8) Study Results • 100% response in treated patients • PRAX-628 achieved between 3-13x multiples of MES EC50 exposure • Safety was consistent with prior dose escalation study and AEs were mild

25 PRAX-628 presents an ideal precision ASM profile https://praxismedicines.com/wp-content/uploads/2023/12/Kahlig_AES2023_628-In-Vivo_Poster_Final.pdf https://praxismedicines.com/wp-content/uploads/2023/09/IEC2023_628-SAD-MAD.pdf Ability to significantly exceed therapeutic concentrations while well tolerated Rapidly achieves therapeutic concentrations after once-daily dose Significantly more potent than competitive molecules in highly translatable pre-clinical models Ideal Treatment Proof of concept achieved in epilepsy patients Initiate three efficacy trials in the ENERGY program

26 PRAX-628 ENERGY program to demonstrate efficacy and bring an improved therapy to focal and generalized epilepsy patients • Registry to help epilepsy patients track their seizures • In partnership with The Epilepsy Study Consortium POWER1 designed to maximize opportunity to demonstrate efficacy POWER2 additionally designed to evaluate broader dosing Evaluate efficacy, safety and extensive PK in broader epilepsy patients EMPOWER POWER1 POWER2 RADIANT 2H 2024 1H 20261H 2025 2H 2025Objective

27 EMPOWER Observational Study to better understand patient journey In partnership with The Epilepsy Study Consortium (TESC) PA TI EN T PA TH W AY Multi-channel engagement of epilepsy patients to EMPOWER Patient consented and provides core medical data Patient is given a seizure diary/epilepsy tracker, trained using TESC guided materials Subset of patients contacted directly by a nurse navigator to help define their epilepsy/seizures Patients who may be eligible for the interventional part of the POWER program will receive direct follow up and/or be directed into the POWER trials

28 • Measuring seizure frequency, seizure freedom, safety and pharmacokinetics • Will allow the evaluation of PRAX-628 in a broader population, including generalized epilepsy • Initiate in 2H 2024 with topline results in 1H 2025 RADIANT Phase 2 open label study to evaluate safety and efficacy in focal onset or generalized epileptic seizures QD = Once daily Screening / Observation 4 weeks 30 mg PRAX-628 QD 8 weeks n ~ 50 Safety Follow-up 2 weeks post dose
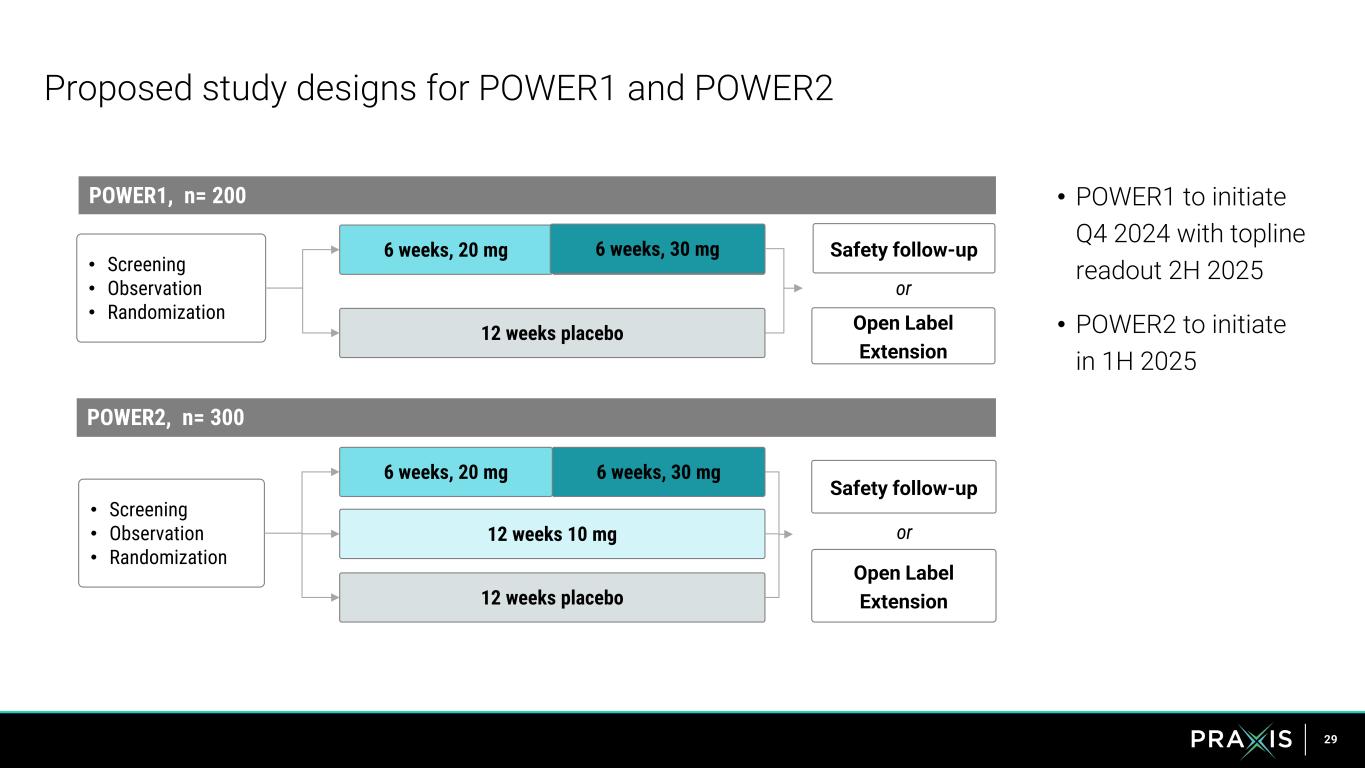
29 Proposed study designs for POWER1 and POWER2 6 weeks, 30mg 12 weeks placebo • Screening • Observation • Randomization POWER1, n= 200 Safety follow-up • POWER1 to initiate Q4 2024 with topline readout 2H 2025 • POWER2 to initiate in 1H 2025 6 weeks, 20 mg POWER2, n= 300 • Screening • Observation • Randomization 6 weeks, 30 mg 12 weeks 10 mg 6 weeks, 20 mg 12 weeks placebo 6 s, 30 g Safety follow-up Open Label Extension or Open Label Extension or

30 Relutrigine (PRAX-562) Milestones Q3 2024: Topline results in Phase 2 EMBOLD Study

31 Preclinical and emerging clinical data demonstrate relutrigine has the potential to be a first- and best-in-class small molecule for DEEs Superior selectivity for disease-state NaV channel hyperexcitability Convenient auto-titration regimen with stable PK Unprecedented therapeutic window with potential for superior safety and efficacy RELUTRIGINE SCN2A, SCN8A FORMULATED FOR PEDIATRIC USE SMALL MOLECULE FUNCTIONAL STATE MODULATOR

32 Relutrigine Phase 1 summary Source: Praxis data on file; https://investors.praxismedicines.com/news-releases/news-release-details/praxis-precision-medicines-provides-corporate-update-and-5 * Co-administration of supra-therapeutic doses of relutrigine and oxcarbazepine led to additive sodium blocking effects, including resulting in SAEs All TEAEs mild to moderate as stand-alone therapy*, with headache & dizziness most common TEAEs Significant changes observed between placebo and relutrigine on qEEG biomarkers Relutrigine has been generally well tolerated in over 130 healthy volunteers No MTD at exposures multiple fold above therapeutic range indicates potential for superior therapeutic index

33 Relutrigine Phase 2 EMBOLD study topline data expected in Q3 2024 + Participants receive either 0.5 mg/kg/day relutrigine QD for 16 weeks or 0.5 mg/kg/day relutrigine QD for 12 weeks & matching placebo QD for 4 weeks. Participants in the relutrigine/placebo arm will receive placebo for 4 consecutive weeks during the 16-week treatment period, with timing of placebo administration blinded for both participants and investigator. Dose adjustment is permitted to a max of 1.0 mg/kg/day and a min of 0.25 mg/kg/day. Safety Follow- Up SAFETY FOLLOW- UP PERIOD (4 WEEKS) DOUBLE-BLIND TREATMENT PERIOD (16 WEEKS) Relutrigine 1:1 Randomization N=~20 (SCN2A/SCN8A) OLE TREATMENT PERIOD (48 WEEKS) Relutrigine 0.5 mg/kg/day Placebo for 4 weeks/relutrigine for 12 weeks+ 0.5 mg/kg/day PRIMARY ENDPOINT: Incidence and severity of treatment-emergent adverse events (TEAEs) KEY SECONDARY: Change from baseline in monthly (28 day) motor seizure frequency
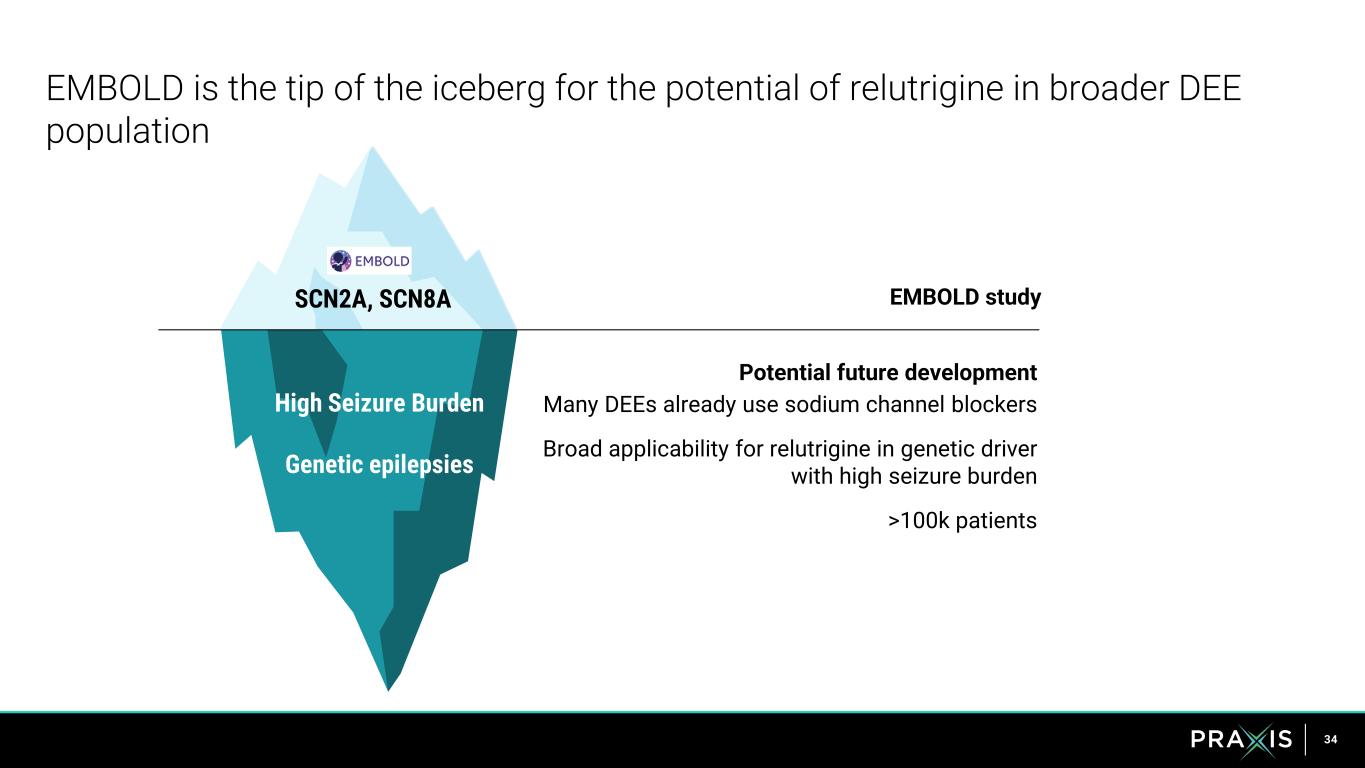
34 EMBOLD is the tip of the iceberg for the potential of relutrigine in broader DEE population SCN2A, SCN8A EMBOLD study Potential future development Many DEEs already use sodium channel blockers Broad applicability for relutrigine in genetic driver with high seizure burden >100k patients High Seizure Burden Genetic epilepsies

35 Elsunersen (PRAX-222) SOLIDUS ASO PLATFORM

36 Elsunersen specifically designed for SCN2A GoF patients DISEASE OVERVIEW • Epilepsy and developmental impairment before the age of 16 years occurs in 1 in 340 children • The majority of these cases result from sporadic de novo genetic variation • In both de novo and familial forms of epilepsy some 900 genes have been identified • Many of these genes also represent opportunities for intervention in other neurological disorders • Patients with SCN2A-DEE have a debilitating and ultimately fatal trajectory • Affects approximately 1,500 patients in the US RESEARCH APPROACH Modifying gene expression to address both gain and loss of function disorders is achieved through various ASO modalities to either silence harmful genes or enhance expression of reduced function or missing genes

37 Significant reduction in seizures observed for SCN2A patients Data from Part 1 of EMBRAVE Study, https://praxismedicines.com/wp-content/uploads/2023/12/Frizzo_AES2023_EMBRAVE_Poster_Final.pdf -39% -43% Im pr ov em en t Mean Median 52% 48% Mean Median Im pr ov em en t • No TEAEs or SAEs considered related to study drug • All TEAEs recovered/resolved Overall % reduction in seizures from 28-day baseline (n=4) Overall relative % increase in seizure-free days from 28-day baseline (n=4)

®