Filed Pursuant to Rule 253(g)(2)
File No.: 024-10810
REGULATION A OFFERING CIRCULAR UNDER THE SECURITIES ACT OF 1933
OFFERING CIRCULAR DATED DECEMBER 2, 2020

EMERALD HEALTH PHARMACEUTICALS INC.
2,850,000 Shares of Common Stock
5910 Pacific Center Blvd, Ste 320
San Diego, California 92121
(800) 268-0719
www.emeraldpharma.life
Emerald Health Pharmaceuticals Inc., a Delaware corporation (the “Company”, “EHP”, “we”, or “our”), is offering up to 2,850,000 shares of our Common Stock, par value $0.0001 per share (the “Shares”) to be sold in this offering (the “Offering”). This Offering was originally qualified by the SEC on March 29, 2018.
Under our ongoing Offering, we have received aggregate investment commitments totaling approximately $49.1 million in gross proceeds through November 20, 2020. This includes completed sales of $43.6 million and investment commitments still in process of $5.5 million. Such investment commitments are awaiting completion of receipt or processing and there are no guarantees all funding commitments will ultimately be realized as gross proceeds. Effective October 30, 2020, the Company elected to terminate its prior financing round under the Offering, as we were nearing the maximum raise of $50 million from sales of qualified securities under our prior qualified offering circular.
We previously sold 2,539,836 shares of Common Stock under the Offering in March 2019 at $5.00 per share for gross proceeds of approximately $12.7 million. Following the qualification by the SEC of our Post-Qualification Amendment No. 3 to the Offering Circular on June 7, 2019, we began offering up to 6,216,803 shares of Common Stock at $6.00 per share. We are now adding and qualifying up to 2,850,000 shares of our Common Stock (the “Shares”) to be offered and sold in the Offering, at a purchase price of $6.00 per Share, for gross proceeds of up to $17.1 million (the “Maximum Amount”) pursuant to this Offering Circular (this “Offering Circular”). The Maximum Amount offered pursuant to this Offering Circular represents the value of the Shares available to be offered as of November 20, 2020 out of the maximum raise of $50 million allowed in a rolling 12-month period under a Regulation A, Tier 2 offering. During the period from November 20, 2019 through November 20, 2020, the Company has received a total of approximately $32.7 million in investment commitments under the Offering, of which $26.5 million represents completed sales and $5.5 million represents sales which are still in process, including $0.7 million for which funding has not yet been received.
We are selling our Shares on a “best efforts” basis through a Tier 2 offering pursuant to Regulation A (Regulation A+) under the Securities Act and we intend to sell the Shares either directly to investors or through registered broker-dealers who are paid commissions. The minimum investment amount from an investor is $3,000 for the purchase of 500 Shares; however, we expressly reserve the right to waive this minimum in the sole discretion of our management. See “Securities Being Offered” beginning on page 66 for a discussion of certain items required by Item 14 of Part II of Form 1-A. The Company has engaged Dalmore Group, LLC, a New York limited liability company and FINRA/SIPC registered broker-dealer ("Dalmore"), to provide broker-dealer services in connection with the Offering. This Offering will terminate on the earlier of (i) March 28, 2021, (ii) the date on which the Maximum Amount is sold, or (iii) when the Board of Directors of the Company elects to terminate the Offering (in each such case, the “Termination Date”). Investor funds will be deposited into an escrow account established for this Offering. We will hold closings upon the receipt of investors’ subscriptions and acceptance of such subscriptions by the Company. We may hold one or more additional closings for additional sales of Shares, until the earlier of: (i) the sale of the Maximum Amount or (ii) the Termination Date. There is no aggregate minimum requirement for the Offering to become effective; therefore, we reserve the right, subject to applicable securities laws, to apply the proceeds from the Offering towards our business strategy, including, without limitation, research and development expenses, offering expenses, working capital and general corporate purposes and, prior to our use of the proceeds, other uses, including capital preservation investments, as more specifically set forth in the “Use of Proceeds” section of this Offering Circular. We expect to continue the sale of the Shares subsequent to November 24, 2020, the date on which the Offering Statement of which this Offering Circular is a part (the “Offering Statement”) was qualified by the SEC.
Investing in the Shares involves a high degree of risk. These are speculative securities. You should purchase these securities only if you can afford a complete loss of your investment. See “Risk Factors” starting on page 5 for a discussion of certain risks that you should consider in connection with an investment in the Shares.
THE SEC DOES NOT PASS UPON THE MERITS OF OR GIVE ITS APPROVAL TO ANY SECURITIES OFFERED OR THE TERMS OF THE OFFERING, NOR DOES IT PASS UPON THE ACCURACY OR COMPLETENESS OF ANY OFFERING CIRCULAR OR OTHER SOLICITATION MATERIALS. THESE SECURITIES ARE OFFERED PURSUANT TO AN EXEMPTION FROM REGISTRATION WITH THE SEC; HOWEVER, THE SEC HAS NOT MADE AN INDEPENDENT DETERMINATION THAT THE SECURITIES OFFERED ARE EXEMPT FROM REGISTRATION.
| Price to Public | Underwriting Discount and Commissions (1) | Proceeds to Company (2) | Proceeds to Other | |||||||||||
| Per Share | $ | 6.00 | $ | 0.06 | $ | 5.94 | None | |||||||
| Total (3) | $ | 17,100,000 | $ | 171,000 | $ | 16,929,000 | None | |||||||
| (1) | We reserve the right to offer the Shares through broker-dealers who are registered with the Financial Industry Regulatory Authority (“FINRA”). The Company has engaged Dalmore Group, LLC, a New York member FINRA/SIPC (“Dalmore”), to perform administrative and compliance-related broker-dealer services in connection with this Offering, but not for underwriting services, pursuant to a Broker-Dealer Agreement between the Company and Dalmore whereby the Company paid Dalmore $28,000 in one-time setup fees and pays Dalmore a 1% commission on sales of Shares. The proceeds received in this Offering will be deposited into an escrow account (the “Escrowed Funds”) pursuant to our Escrow Services Agreement with Prime Trust, LLC. The Escrowed Funds will be distributed to the Company and the purchased Shares will be issued to the investors. The Company has no obligation to refund subscriptions. (See “Plan of Distribution and Selling Securityholders.”) |
| (2) | The amounts shown are before deducting estimated Offering costs to us of approximately $1.2 million, which include legal, accounting, printing, due diligence, marketing, consulting, selling and other costs incurred in the Offering. (See “Use of Proceeds” and “Plan of Distribution and Selling Securityholders.”) |
| (3) | The Shares are being offered pursuant to Regulation A of Section 3(b) of the Securities Act for Tier 2 offerings. The Shares are only issued to purchasers who satisfy the requirements set forth in Regulation A. We have the option in our sole discretion to waive the minimum investment. |
GENERALLY, NO SALE MAY BE MADE TO YOU IN THIS OFFERING IF THE AGGREGATE PURCHASE PRICE YOU PAY IS MORE THAN TEN PERCENT (10%) OF THE GREATER OF YOUR ANNUAL INCOME OR YOUR NET WORTH. DIFFERENT RULES APPLY TO ACCREDITED INVESTORS AND NON-NATURAL PERSONS. BEFORE MAKING ANY REPRESENTATION THAT YOUR INVESTMENT DOES NOT EXCEED APPLICABLE THRESHOLDS, WE ENCOURAGE YOU TO REVIEW RULE 251(D)(2)(I)(C) OF REGULATION A+. FOR GENERAL INFORMATION ON INVESTING, WE ENCOURAGE YOU TO REFER TO WWW.INVESTOR.GOV.
This Offering Circular contains all of the representations by us concerning this Offering, and no person shall make different or broader statements than those contained herein. Investors are cautioned not to rely upon any information not expressly set forth in this Offering Circular.
The securities underlying this Offering Circular may not be sold until qualified by the Securities and Exchange Commission. This Offering Circular is not an offer to sell, nor soliciting an offer to buy, any Shares in any state or other jurisdiction in which such sale is prohibited.
The Company is following the “Offering Circular” format of disclosure under Regulation A+.
The date of this Offering Circular is December 2, 2020
TABLE OF CONTENTS
i
IMPORTANT INFORMATION ABOUT THIS OFFERING CIRCULAR
We are offering to sell, and seeking offers to buy, our securities only in jurisdictions where such offers and sales are permitted. Please carefully read the information in this Offering Circular and any accompanying offering circular supplements, which we refer to collectively as the Offering Circular. You should rely only on the information contained in this Offering Circular. We have not authorized anyone to provide you with any information other than the information contained in this Offering Circular. The information contained in this Offering Circular is accurate only as of its date or as of the respective dates of any documents or other information incorporated herein by reference, regardless of the time of its delivery or of any sale or delivery of our securities. Neither the delivery of this Offering Circular nor any sale or delivery of our securities shall, under any circumstances, imply that there has been no change in our affairs since the date of this Offering Circular. This Offering Circular will be updated and made available for delivery to the extent required by the federal securities laws.
This Offering Circular is part of an offering statement that we filed with the SEC using a continuous offering process pursuant to Rule 251(d)(3)(i)(F) under the Securities Act. Periodically, we may provide an offering circular supplement that would add, update or change information contained in this Offering Circular. Any statement that we make in this Offering Circular will be modified or superseded by any inconsistent statement made by us in a subsequent offering circular supplement. The offering statement we filed with the SEC includes exhibits that provide more detailed descriptions of the matters discussed in this Offering Circular. You should read this Offering Circular and the related exhibits filed with the SEC and any offering circular supplement, together with additional information contained in our annual reports, semi-annual reports and other reports that we will file periodically with the SEC. The offering statement and all supplements and reports that we have filed or will file in the future can be read at the SEC website, www.sec.gov.
Unless otherwise indicated, data contained in this Offering Circular concerning the business of the Company are based on information from various public sources. Although we believe that these data are generally reliable, such information is inherently imprecise, and our estimates and expectations based on these data involve a number of assumptions and limitations. As a result, you are cautioned not to give undue weight to such data, estimates or expectations.
In this Offering Circular, unless the context indicates otherwise, references to the “Company,” “EHP,” “we,” “our,” and “us” refer to the activities of and the assets and liabilities of the business and operations of Emerald Health Pharmaceuticals Inc.
ii
CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING STATEMENTS
Some of the statements under “Summary,” “Risk Factors,” “Management’s Discussion and Analysis of Financial Condition and Results of Operations,” “Our Business” and elsewhere in this Offering Circular constitute forward-looking statements. Forward-looking statements relate to expectations, beliefs, projections, future plans and strategies, anticipated events or trends and similar matters that are not historical facts. In some cases, you can identify forward-looking statements by terms such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “potential,” “should,” “will” and “would” or the negatives of these terms or other comparable terminology.
You should not place undue reliance on forward looking statements. The cautionary statements set forth in this Offering Circular, including in “Risk Factors” and elsewhere, identify important factors which you should consider in evaluating our forward-looking statements. These factors include, among other things:
| ● | The success of our product candidates will require significant capital resources and years of clinical development efforts; | |
| ● | The results of clinical testing and trial activities of our products; | |
| ● | Our ability to obtain regulatory approval and market acceptance of, and reimbursement for our products; | |
| ● | Our ability to protect our intellectual property and to develop, maintain and enhance a strong brand; | |
| ● | Our ability to compete and succeed in a highly competitive and evolving industry; | |
| ● | Our limited operating history on which to judge our business prospects and management; | |
| ● | Our ability to raise capital and the availability of future financing; | |
| ● | Our ability to manage our research, development, expansion, growth and operating expenses; | |
| ● | The impact of the novel coronavirus (“COVID-19”) pandemic; and | |
| ● | Our reliance on third parties to conduct our research, preclinical studies, manufacturing and clinical trials. |
Although the forward-looking statements in this Offering Circular are based on our beliefs, assumptions and expectations, taking into account all information currently available to us, we cannot guarantee future transactions, results, performance, achievements or outcomes. No assurance can be made to any investor by anyone that the expectations reflected in our forward-looking statements will be attained, or that deviations from them will not be material and adverse. We undertake no obligation, other than as may be required by law, to re-issue this Offering Circular or otherwise make public statements updating our forward-looking statements.
iii
This summary highlights selected information contained elsewhere in this Offering Circular. This summary is not complete and does not contain all the information that you should consider before deciding whether to invest in our Common Stock. You should carefully read the entire Offering Circular, including the risks associated with an investment in the company discussed in the “Risk Factors” section of this Offering Circular, before making an investment decision. Some of the statements in this Offering Circular are forward-looking statements. See the section entitled “Cautionary Statement Regarding Forward-Looking Statements.”
Company Information
Emerald Health Pharmaceuticals Inc. (the Company, EHP, we, our, and us) was formed on March 2, 2017 under the laws of the State of Delaware, and is headquartered in San Diego, California. The Company was formed to acquire, discover, develop and commercialize drug candidates based on patented new chemical entities (NCEs) derived from cannabinoids (molecules found in cannabis).
In June 2017, pursuant to an Intellectual Property Transfer Agreement (IPTA) with Emerald Health Biotechnology España S.L.U. (EHBE), a limited liability company formed under the laws of Spain, formerly known as VivaCell Biotechnology España S.L. (VivaCell), the Company acquired three United States patents, two Japanese patents and one European patent and fourteen pending patent applications covering two series of molecules containing NCE derivatives of cannabidiol (CBD) and cannabigerol (CBG) for cash consideration of $112,000. Future payments are due upon completion of certain milestones and a 2.5% royalty will be paid to EHBE on all net revenues of any drug developed from the intellectual property acquired pursuant to the IPTA. The first milestone was achieved in 2019, resulting in payments to EHBE of approximately $440,000. The assets acquired under the IPTA were in the research stage. Concurrent with the entry into the IPTA, the Company entered into a Research Agreement with EHBE for an initial term of five years pursuant to which EHBE will perform certain functions to support the research efforts associated with our development of the acquired technology.
Our majority stockholder is Emerald Health Sciences Inc. (EHS). EHS is a private company formed to invest in companies operating within the cannabis/cannabinoid industry. As of the date of this Offering Circular, EHS owns approximately 51.3% of our outstanding Common Stock. Accordingly, EHS exerts and may continue to exert significant influence over us and any action requiring the approval of the holders of our Common Stock, including the election of directors and amendments to our organizational documents, such as increases in our authorized shares of Common Stock and approval of significant corporate transactions.
Our mailing address is Emerald Health Pharmaceuticals Inc., 5910 Pacific Center Blvd, Ste 320, San Diego, California 92121 and our telephone number is (800) 268-0719. Our website address is www.emeraldpharma.life. The information contained therein or accessible thereby shall not be deemed to be incorporated into this Offering Circular.
Our Business
We are a biotechnology/pharmaceutical company focused on developing drug product candidates currently containing novel, patented molecules chemically derived from two non-psychoactive cannabinoids, CBD and CBG, to treat diseases with unmet medical needs, primarily autoimmune, neurodegenerative, inflammatory and fibrotic diseases. We are currently developing two initial therapeutic product candidates that together target four initial disease indications, multiple sclerosis (MS), systemic sclerosis (SSc, a severe form of scleroderma), Parkinson’s disease (PD) and Huntington’s disease (HD). We believe treatments for these indications represent markets with underserved patient populations.
1
Our platform technology is a result of the unique convergence of biology, science and cannabinoids, and consists of a library of twenty-five novel, patented derivatives of CBD and CBG. The resulting molecules are new chemical entities (NCEs) covered by 19 issued international patents. In addition, we have 20 pending patent applications. We believe our cannabinoid-based technology platform represents an advancement to existing therapies because our NCEs are chemically modified from CBD and CBG to act on additional biological receptors and pathways in the body to specifically treat the diseases we are targeting, which CBD and CBG alone do not affect. We know of no other products on the market or product candidates in development that possess the same combined mechanism of action (MOA) as our novel molecules in our lead product candidates.
Our current product pipeline includes two initial product candidates from our library of NCEs, EHP-101 and EHP-102. EHP-101 is an oral formulation of a novel synthetic CBD derivative, known as VCE-004.8, and is our lead candidate, currently in Phase 2 clinical development. EHP-102 is a formulation of a novel synthetic CBG derivative, known as VCE-003.2, currently in preclinical development.
Based on our studies to date, we believe that these initial product candidates have the potential to treat several diseases with unmet medical need. We are currently targeting four distinct diseases, two for each of these initial product candidates. With EHP-101, we are initially targeting MS and SSc, and with EHP-102, we are initially targeting PD and HD. Other applications are also being investigated, with our two current product candidates, as well as different formulations and other molecules within our NCE portfolio.
In September 2019, we successfully completed a Phase 1 human clinical study in Australia to establish EHP-101’s safety, tolerability and pharmacokinetics (PK) in healthy volunteers. We have recently initiated a Phase 2a safety and efficacy study in SSc patients and we plan to initiate a Phase 2 study in MS patients. If such studies are successful, we plan to then advance the product candidates into additional efficacy studies thereafter.
We have completed preclinical proof of concept (POC) work for EHP-102. We are now in the manufacturing and formulation development stage and have begun initial clinical-enabling studies for HD and PD. If successful, we then plan to advance EHP-102 to Phase 1 human clinical studies.
We have been granted Orphan Drug Designation (ODD) from the Food and Drug Administration (FDA) in the United States (US) and from the European Medicines Agency (EMA) in the European Union (EU) for EHP-101 for the SSc indication and for EHP-102 for the HD indication. We have also received Fast Track designation by the FDA for EHP-101 for the SSc indication.
The starting material for the active pharmaceutical ingredient (API) in our product candidates are CBD and CBG, which may be classified by the United States Drug Enforcement Administration (DEA) as controlled substances in the United States depending on their origin and purity. In March 2019 we received a decision from the DEA that the API (VCE-004.8) in our lead product candidate (EHP-101) is not a controlled substance, based mainly on the fact that our molecule is (1) an NCE which is no longer CBD, (2) chemically derived from synthetic CBD as an NCE containing no remaining CBD or other controlled substances and (3) non-psychoactive. We have also received the same decision from the United Kingdom (UK) Home Office and Canada’s Controlled Substances Directorate. In general, the determination that VCE-004.8 is not a controlled substance reduces the costs and complexities otherwise associated with developing controlled substances. With this determination, manufacturing facilities do not require controlled substance certification for handling and dispensing the molecules and drug products. It also facilitates importation and simplifies the conduct of nonclinical and clinical studies, as contracted nonclinical research organizations and clinical sites have less administrative burden. Once we advance our second product candidate (EHP-102) further in development, we will request a similar decision from the DEA, and other countries, for this product candidate.
Intellectual Property
Our intellectual property is related to the two series of molecules in our portfolio, containing twenty-five different molecules. We have sought and intend to continue to seek additional appropriate patent protection for our product candidates, as well as other proprietary technologies and their uses by filing patent applications in the United States and selected other countries.
As of the date of this Offering Circular, we own a total of 19 issued patents and 20 pending patent applications, including composition of matter, use, and formulation patents. Our patent portfolio provides a relatively long window for development and commercialization. Our patents and patent applications will expire between 2030 and 2041 and may be eligible for patent term extension for delay caused by regulatory review, thereby further extending their patent terms. In addition, our patent portfolio includes use in many different indications, which we believe will allow us to develop products for additional patient populations in markets with unmet medical need.
2
Product Pipeline
Our current product pipeline consists of two initial product candidates, EHP-101 and EHP-102, which we are developing for four disease indications. We own all intellectual property and all global development and marketing rights with respect to our product pipeline.
Competition
The biotechnology and pharmaceutical industries are subject to rapid and intense technological change. We face, and will continue to face, competition in the development and marketing of our product candidates from biotechnology and pharmaceutical companies, research institutions, government agencies and academic institutions. Competition may also arise from, among other things:
| ● | other drug development technologies; | |
| ● | methods of preventing or reducing the incidence of disease, including vaccines; and | |
| ● | new small molecule or other classes of therapeutic agents. |
Developments by others may render our product candidates or technologies obsolete or noncompetitive.
Risks Related to Our Business
Our business and our ability to execute our business strategy are subject to a number of risks as more fully described in the section titled “Risk Factors” beginning on page 5. These risks include, among others:
| ● | The success of our product candidates will require significant capital resources and years of clinical development efforts; | |
| ● | The results of clinical testing and trial activities of our products; | |
| ● | Our ability to obtain regulatory approval and market acceptance of, and reimbursement for our products; | |
| ● | Our ability to protect our intellectual property and to develop, maintain and enhance a strong brand; | |
| ● | Our ability to compete and succeed in a highly competitive and evolving industry; | |
| ● | Our limited operating history on which to judge our business prospects and management; | |
| ● | Our ability to raise capital and the availability of future financing; | |
| ● | Our ability to manage our research, development, expansion, growth and operating expenses; | |
| ● | The impact of the COVID-19 pandemic; and | |
| ● | Our reliance on third parties to conduct our research, preclinical studies and expected clinical trials. |
Our financial statements have been prepared assuming we will continue as a going concern, which contemplates the realization of assets and the satisfaction of liabilities in the normal course of business. Since inception, we have funded operations with the proceeds from a revolving loan and advances of expenditures paid for on our behalf by our majority stockholder and the proceeds raised to date under this Offering, which total approximately $48.4 million as of November 20, 2020, inclusive of both completed sales and pending sales (received subscriptions) in process. We currently have a revolving loan agreement in place with our majority stockholder, however, we do not have an agreement in place to continue such funding and any further borrowing under such facility will require consent of our majority stockholder. Our future viability is largely dependent upon our ability to raise additional capital to finance our operations. Our management expects that future sources of funding may include sales of equity, obtaining loans, or other strategic transactions. Although our management continues to pursue these plans, there is no assurance that we will continue to be successful with this Offering or in obtaining sufficient financing on terms acceptable to us to continue to finance our operations, if at all. These circumstances raise substantial doubt on our ability to continue as a going concern, and our financial statements do not include any adjustments that might result from the outcome of these uncertainties.
3
REGULATION A+
We are offering our Common Stock pursuant to rules of the SEC mandated under the Jumpstart Our Business Startups Act of 2012 (the JOBS Act). These offering rules are often referred to as “Regulation A+.” We are relying upon “Tier 2” of Regulation A+, which allows us to offer and sell up to $50 million in a 12-month period. Pursuant to Rule 251(d)(3)(i)(F) under Regulation A+, the securities offered may be continuously offered and sold for up to three years following the initial qualification date of the offering statement under which they are being sold. The securities offered by our Offering Statement, originally qualified on March 29, 2018 (as the same has since been amended and supplemented) are being offered in a continuous offering process pursuant to Rule 251(d)(3)(i)(F). We are now adding and qualifying 2,850,000 additional Shares to the Offering Statement.
In accordance with the requirements of Tier 2 of Regulation A+, we are required to publicly file annual, semiannual, and current event reports with the SEC.
| Issuer: | Emerald Health Pharmaceuticals Inc., a Delaware corporation. | |
| Shares Offered: | A maximum of 2,850,000 Shares at an offering price of $6.00 per Share. | |
| Number of shares of Common Stock Outstanding and Issuable as of November 20, 2020(1) (2): | 19,968,042 shares of Common Stock. | |
| Number of shares of Common Stock to be Outstanding after the Offering (1) (2): | 22,818,042 shares of Common Stock if the 2,850,000 Shares are sold. | |
| Price per Share: | $6.00 | |
| Maximum Amount: | We are currently offering up to 2,850,000 Shares, at a price of $6.00 per Share, for total gross proceeds of $17,100,000. If the Maximum Amount of $17,100,000 is sold pursuant to this Offering Circular, a total of 11,457,342 shares of Common Stock will be issued under this Offering, assuming all investments in process are completed, for total gross proceeds since March 2019 of approximately $66.2 million. During the period from November 20, 2019 through November 20, 2020, we received investment commitments totaling approximately $32.7 million under the Offering, including $26.5 million in completed sales and $5.5 million representing pending sales still in process, including $0.7 million for which funding has not yet been received. | |
| Use of Proceeds: | If the Maximum Amount is sold, our net proceeds (after estimated offering expenses and selling commissions) will be approximately $15.7 million. We will use these net proceeds for research and development expenses (including clinical trials), working capital and general corporate purposes. In addition, a portion of the proceeds raised may be used in management’s discretion to repay, in whole or in part, the principal and/or the accrued interest on our loan to our majority stockholder and such other purposes, and prior to our use of the proceeds, invested in short-term, interest-bearing instruments as further described in the “Use of Proceeds” section of this Offering Circular. | |
| Risk Factors: | Investing in our Common Stock involves a high degree of risk. See “Risk Factors” starting on page 5. |
| (1) | In addition, there are 3,429,833 shares of Common Stock reserved for issuance under our 2018 Equity Incentive Plan as of November 20, 2020, with 2,857,500 shares of Common Stock issuable pursuant to outstanding awards of which 817,500 shares of Common Stock will be issuable upon exercise of outstanding awards at $6.00 per share, 275,000 shares of Common Stock will be issuable upon exercise of outstanding awards at $5.00 per share and 1,765,000 shares of Common Stock will be issuable upon exercise of outstanding awards at $2.50 per share. |
| (2) | Includes commitments we have received for an additional 913,410 shares of Common Stock for which processing or funding is not completed as of the date hereof. Such investment commitments are awaiting completion of receipt and processing and there are no guarantees all funding commitments will ultimately result in the issuance of Shares. |
4
An investment in our Common Stock involves a high degree of risk. You should carefully consider the risks described below, together with all of the other information included in this Offering Circular, before making an investment decision. If any of the following risks actually occurs, our business, financial condition or results of operations could suffer. In that case, the price of our shares of Common Stock could decline and you may lose all or part of your investment. See “Cautionary Statement Regarding Forward Looking Statements” above for a discussion of forward-looking statements and the significance of such statements in the context of this Offering Circular.
Risks Related to our Business and Industry
We are largely dependent on the success of our product candidates, EHP-101 and EHP-102, which are in clinical and preclinical development, respectively, and will require the effective execution of our business plan, significant capital resources and years of clinical development effort.
We currently have no products on the market. Our most advanced product candidate, EHP-101, completed a Phase 1 clinical trial in September 2019, and our second product candidate, EHP-102, is in preclinical development. Our business plan depends almost entirely on the successful preclinical and clinical development, regulatory approval and commercialization of EHP-101 and EHP-102, and substantial clinical development and regulatory approval efforts will be required before we are permitted to commence commercialization, if ever. It could be several years before we can complete a pivotal study for EHP-101 or EHP-102, if ever. The clinical trials and manufacturing and marketing of EHP-101 and EHP-102 will be subject to extensive and rigorous review and regulation by numerous government authorities in the United States (US), Australia, New Zealand, the European Union (EU), Canada, and other jurisdictions where we intend to perform studies and, if approved, market our product candidates. Before obtaining regulatory approvals for the commercial sale of any product candidate, we must demonstrate through preclinical testing and clinical trials that the product candidate is safe and effective for use in each target indication, and potentially in specific patient populations. This process can take many years and may include post-marketing studies and surveillance, which would require the expenditure of substantial resources beyond our existing funds. Of the large number of drugs in development for approval in the US and the EU, only a small percentage successfully complete the US Food and Drug Administration (FDA) regulatory approval process or are granted a marketing authorization by the European Medicines Agency (EMA) or the other competent authorities in the EU Member States, as applicable, and are commercialized. Accordingly, even if we are able to obtain the requisite financing to continue to fund our research, development and clinical programs, we cannot assure you that any of our product candidates will be successfully developed or commercialized.
Because the results of efficacy and preclinical studies are not necessarily predictive of future results, EHP-101 and EHP-102 may not have favorable results in our planned clinical trials.
Any positive results from efficacy in preclinical testing of EHP-101 and EHP-102 may not necessarily be predictive of the results from our current and planned clinical trials. In addition, our interpretation of clinical data or our conclusions based on our preclinical in vitro and in vivo models may prove inaccurate. Many companies in the pharmaceutical and biotechnology industries have suffered significant setbacks in clinical trials after achieving positive results in preclinical development, and we cannot be certain that we will not face similar setbacks. These setbacks have been caused by, among other things, preclinical findings while clinical trials were underway or safety or efficacy observations in clinical trials, including adverse events. Moreover, preclinical data can be susceptible to varying interpretations and analyses, and many companies that believed their product candidates performed satisfactorily in preclinical studies nonetheless failed to obtain FDA approval or a marketing authorization granted by the EMA. If we fail to produce positive results in our planned clinical trials of EHP-101 and EHP-102, the development timeline and regulatory approval and commercialization prospects for EHP-101 and EHP-102, and, correspondingly, our business and financial prospects, would be materially adversely affected.
5
Failures or delays in our planned clinical trials of EHP-101 or EHP-102 could result in increased costs to us and could delay, prevent or limit our ability to generate revenue and continue our business.
EHP-101 has completed a Phase 1 clinical study and is currently initiating a Phase 2 clinical study and EHP-102 is advancing through preclinical development. Successful completion of preclinical studies and clinical trials is a prerequisite to submitting a new drug application (NDA) to the FDA or a marketing authorization application (MAA) to the EMA, which are required for approval for commercialization. Clinical trials are expensive, difficult to design and implement, can take many years to complete and are uncertain as to outcome. A product candidate can unexpectedly fail at any stage of clinical development. The historic failure rate for product candidates is high due to many factors, including scientific feasibility, findings related to safety and efficacy, changing regulatory standards and standards of medical care and other variables. We do not know whether our clinical trials will begin or be completed on schedule, if at all, as the commencement and completion of clinical trials can be delayed or prevented for a number of reasons, including, among others:
| ● | delays in reaching or failing to reach agreement on acceptable terms with prospective clinical trial sites, the terms of which can be subject to extensive negotiation and may vary significantly among different clinical trial sites; | |
| ● | clinical sites or investigators deviating from trial protocol, failing to conduct the trial in accordance with applicable regulatory requirements, or dropping out of a trial or failure of third-party clinical trial managers to meet their contractual obligations or deadlines; | |
| ● | delays or inability in manufacturing or obtaining sufficient quantity or quality of a product candidate or other materials necessary to conduct clinical trials due to regulatory and manufacturing constraints; | |
| ● | delay or failure in reaching agreement with the FDA or a foreign regulatory authority on the design of a given trial, or in obtaining authorization to commence a trial; | |
| ● | difficulties obtaining institutional review board (IRB), Drug Enforcement Administration (DEA) or comparable foreign regulatory authority, or ethics committee approval to conduct a clinical trial; | |
| ● | challenges in recruiting and enrolling patients to participate in clinical trials, including the size and nature of the patient population, the proximity of patients to clinical trial sites, eligibility criteria for the clinical trial, the nature of the clinical trial protocol, the availability of approved effective treatments for the relevant indication and competition from other clinical trial programs for similar indications; | |
| ● | severe or unexpected toxicities or drug-related side effects in our preclinical studies or experienced by patients in our clinical trials or by individuals using drugs similar to our product candidates; | |
| ● | DEA or comparable foreign regulatory authority-related recordkeeping, reporting or security violations at a clinical trial site, leading the DEA, state authorities or comparable foreign regulatory authorities to suspend or revoke the site’s controlled substance registration and causing a delay or termination of planned or ongoing clinical trials; | |
| ● | regulatory concerns with cannabinoid products generally and the potential for abuse of those products; | |
| ● | difficulties retaining patients who have enrolled in a clinical trial who may withdraw due to lack of efficacy, side effects, personal issues or loss of interest and difficulties having subjects return for post-treatment follow-up; | |
| ● | ambiguous or negative interim results; or | |
| ● | lack of adequate funding to continue a clinical trial. |
6
In addition, a clinical trial may be suspended or terminated by us, the FDA, an Institutional Review Board (IRB), an ethics committee, a data safety monitoring board or other foreign regulatory authorities overseeing the clinical trial at issue due to a number of factors, including, among others:
| ● | failure to conduct the clinical trial in accordance with regulatory requirements or our clinical trial protocols; | |
| ● | inspection of the clinical trial operations or clinical trial sites by the FDA, the DEA, the EMA or other foreign regulatory authorities that reveals deficiencies or violations that require us to undertake corrective action, including the imposition of a clinical hold; | |
| ● | safety issues, including any issues that could be identified in our ongoing toxicology and mutagenicity studies; | |
| ● | adverse side effects or lack of effectiveness; and | |
| ● | changes in government regulations or administrative actions. |
If our clinical trials fail or are delayed for any of the above reasons, our development costs may increase, our approval process could be delayed and our ability to commercialize our product candidates could be materially harmed, which could have a material adverse effect on our business, financial condition or results of operations.
The regulatory approval processes of the FDA, the EMA and other comparable domestic and foreign regulatory authorities are lengthy, time-consuming and inherently unpredictable, and if we are ultimately unable to obtain timely regulatory approval for our product candidates, our business will be substantially harmed.
We are not permitted to market our product candidates in the US or the EU until we receive approval of an NDA from the FDA or an MAA from the EMA, or in any foreign countries until we receive the requisite approval from such countries. Prior to submitting an NDA to the FDA or an MAA to the EMA for approval of our product candidates we will need to complete our preclinical studies and initiate and complete multiple clinical trials. Successfully completing our clinical program and obtaining approval of an NDA or MAA is a complex, lengthy, expensive and uncertain process, and the FDA or EMA may delay, limit or deny approval of our product candidates for many reasons, including, among others, because:
| ● | we may not be able to demonstrate that our product candidates are safe and effective in treating patients to the satisfaction of the FDA, EMA or other applicable foreign regulatory agencies; | |
| ● | the results of our clinical trials may not meet the level of statistical or clinical significance required by the FDA, EMA or other applicable foreign regulatory agencies for marketing approval; | |
| ● | the FDA, EMA or other applicable foreign regulatory agencies may disagree with the number, design, size, conduct or implementation of our clinical trials; | |
| ● | the FDA, EMA or other applicable foreign regulatory agencies may require that we conduct additional clinical trials; | |
| ● | the FDA, EMA or other applicable foreign regulatory authorities may not approve the formulation, labeling or specifications of our product candidates; | |
| ● | the contract research organizations (CROs) and other contractors that we may retain to conduct our clinical trials may take actions outside of our control that materially adversely impact our clinical trials; | |
| ● | the FDA, EMA or other applicable foreign regulatory agencies may find the data from preclinical studies and clinical trials insufficient to demonstrate that EHP-101 or EHP-102 are safe and effective for their proposed indications; |
7
| ● | the FDA, EMA or other applicable foreign regulatory agencies may disagree with our interpretation of data from our preclinical studies and clinical trials; | |
| ● | the FDA, EMA or other applicable foreign regulatory agencies may not accept data generated at our clinical trial sites or may disagree with us over whether to accept efficacy results from clinical trial sites outside the US, outside the EU, or outside a specific country, as applicable, where the standard of care is potentially different from that in the US or in the EU, as applicable; | |
| ● | if and when our NDAs or MAAs or other applications for regulatory approval are submitted to the FDA, EMA, or other applicable foreign regulatory agencies, as applicable, the regulatory authorities may have difficulties scheduling the necessary review meetings in a timely manner, may recommend against approval of our application or may recommend or require, as a condition of approval, additional preclinical studies or clinical trials, limitations on approved labeling or distribution and use restrictions; | |
| ● | the FDA may require development of a Risk Evaluation and Mitigation Strategy (REMS), which would use risk minimization strategies to ensure that the benefits of certain prescription drugs outweigh their risks, as a condition of approval or post-approval, and the EMA or other applicable foreign regulatory agencies may grant only conditional marketing authorization or impose specific obligations as a condition for marketing authorization, or may require us to conduct post-authorization safety studies; | |
| ● | the FDA, DEA, EMA or other applicable foreign regulatory agencies may not approve the manufacturing processes or facilities of third-party manufacturers with which we contract, or DEA or other applicable foreign regulatory agency quotas may limit the quantities of controlled substances available to our manufacturers; or | |
| ● | the FDA, EMA or other applicable foreign regulatory agencies may change their approval policies or adopt new regulations. |
Any of these factors, many of which are beyond our control, could increase development costs, jeopardize our ability to obtain regulatory approval for and successfully market EHP-101 or EHP-102 and generate product revenue. Moreover, because our business is almost entirely dependent upon these two candidates, any such setback in our pursuit of regulatory approval would have a material adverse effect on our business and prospects.
We have conducted a Phase 1 clinical trial for EHP-101 outside the US, and we are initiating a Phase 2 clinical trial for EHP-101 inside and outside the US, and we may choose to conduct additional clinical trials for EHP-101 and EHP-102 outside the US, and the FDA may not accept data from such trials.
We completed a Phase 1 clinical trial for EHP-101 in Australia and we are initiating a Phase 2 clinical trial for EHP-101 in Australia, New Zealand and the US, and we may choose to conduct additional clinical trials for EHP-101 and EHP-102 in countries outside the US, including Australia and New Zealand, subject to applicable regulatory approval. We plan to submit NDAs for EHP-101 and EHP-102 to the FDA upon completion of all requisite clinical trials. Although the FDA may accept data from clinical trials conducted outside the US, acceptance of such study data by the FDA is subject to certain conditions. For example, the clinical trial must be conducted in accordance with Good Clinical Practice (GCP) requirements and the FDA must be able to validate the data from the clinical trial through an onsite inspection if it deems such inspection necessary. There can be no assurance the FDA will accept data from clinical trials conducted outside of the US. If the FDA does not accept any such data, it would likely result in the need for additional clinical trials, which would be costly and time-consuming and delay aspects of our development plan.
8
Even if EHP-101 or EHP-102 receive regulatory approval, they may still face future development and regulatory difficulties.
If we obtain regulatory approval for EHP-101 or EHP-102, such approval would be subject to extensive ongoing requirements by the FDA, EMA and other foreign regulatory authorities, and potentially the DEA, including requirements related to the manufacture, quality control, further development, labeling, packaging, storage, distribution, safety surveillance, import, export, advertising, promotion, recordkeeping and reporting of safety and other post-market information. The safety profile of any product will continue to be closely monitored by the FDA, EMA and other comparable foreign regulatory authorities. If the FDA, EMA, DEA or any other comparable foreign regulatory authority become aware of new safety information after approval of any of our product candidates, these regulatory authorities may require labeling changes or establishment of a Risk Evaluation and Mitigation Strategy (REMS), impose significant restrictions on a product’s indicated uses or marketing, initiate a change in the drug’s controlled substance schedule, impose ongoing requirements for potentially costly post-approval studies or post-market surveillance, impose a recall or seek to withdraw marketing approval altogether.
In addition, manufacturers of therapeutic products and their facilities are subject to continual review and periodic inspections by the FDA, the EMA and other comparable foreign regulatory authorities for compliance with current Good Manufacturing Practices (cGMPs). Further, manufacturers of controlled substances must obtain and maintain necessary DEA and state registrations and registrations with applicable foreign regulatory authorities and must establish and maintain processes to ensure compliance with DEA and state requirements and requirements of applicable foreign regulatory authorities governing, among other things, the storage, handling, security, recordkeeping and reporting for controlled substances. If we or a regulatory agency discover previously unknown problems with a product, such as adverse events of unanticipated severity or frequency, or problems with the facility where the product is manufactured, a regulatory agency may impose restrictions on that product, the manufacturing facility or us, including requiring recall or withdrawal of the product from the market or suspension of manufacturing. If we, our product candidates or the manufacturing facilities for our product candidates fail to comply with applicable regulatory requirements, a regulatory agency may, among other things, impose penalties or require us to undertake certain actions, each of which could be costly and time-consuming.
The occurrence of any event or penalty described above may inhibit our ability to commercialize our product candidates and may otherwise have a material adverse effect on our business, financial condition and results of operations.
Even if EHP-101 and EHP-102 advance through preclinical studies and clinical trials, we may experience difficulties in managing our growth and expanding our operations.
We have limited resources to carry out objectives for our current and future preclinical studies and clinical trials. Clinical trials are a time-consuming, expensive and uncertain process. In addition, while we have experienced management and expect to contract out many of the activities related to conducting these programs, we are a small company with only fifteen full-time employees, three part-time employees and several consultants and therefore have limited internal resources both to conduct preclinical studies and clinical trials and to monitor third-party providers. As our product candidates advance through preclinical studies and clinical trials, we will need to expand our development, regulatory and manufacturing operations, either by expanding our internal capabilities or contracting with other organizations to provide these capabilities for us. In the future, we expect to have to manage additional relationships with collaborators or partners, suppliers and other organizations. Our ability to manage our operations and future growth will require us to continue to improve our operational, financial and management controls, reporting systems and procedures.
EHP-101 and EHP-102 may be subject to controlled substance laws and regulations; failure to receive necessary approvals may delay the launch of our products and failure to comply with these laws and regulations may adversely affect the results of our business operations.
Under the Controlled Substances Act (CSA), both CBD and CBG, derived from certain parts of the cannabis plant, fall into drug code 7350 and are considered controlled substances that are illegal under the CSA. In 2017, the DEA clarified its position on materials or products that would be considered to fall within the 7350 drug code. The DEA’s position is now that materials or products that consist solely of parts of the cannabis plant excluded from the CSA definition of marijuana are excluded from the 7350 (marijuana) or 7360 (marijuana) drug codes.
EHP-101 and EHP-102 are NCEs, which are not parts of the cannabis plant. They are synthetically manufactured derivatives of synthetically manufactured CBD and CBG. Even though our NCEs are not part of the cannabis plant, and therefore should not fall into either the 7350 or 7360 drug code, certain of these synthetically manufactured derivatives (though not all) may still be considered controlled substances under the CSA because they are derived from CBD and CBG molecules.
9
We have sought a decision from the US DEA regarding the controlled substance status of the API in our lead product candidate (EHP-101) and in March 2019 we received a decision from the DEA that the API (VCE-004.8) in our lead product candidate (EHP-101) is not a controlled substance, based partly on the fact that our molecule is chemically derived from synthetic CBD as an NCE containing no remaining CBD or other controlled substances. We have also received the same decision from the United Kingdom (UK) Home Office and Canada’s Controlled Substances Directorate. With this determination manufacturing facilities do not require controlled substance certification for handling and dispensing the molecules and drug products. It also facilitates importation and simplifies the conduct of nonclinical and clinical studies, as contracted nonclinical research organizations and clinical sites have less administrative burden. Once we advance our second product candidate (EHP-102) further in development, we will request a similar decision from the DEA, and other countries, for this product candidate.
The determination of the controlled substance status by the DEA in the US has not yet been sought by us for EHP-102. The scheduling process may also take one or more years beyond FDA approval, thereby significantly delaying product launch of EHP-102. However, if considered controlled substances, the DEA must issue a temporary order scheduling the drug within 90 days after FDA approves the drug and DEA receives a scientific and medical evaluation and scheduling recommendation from the Department of Health and Human Services (HHS). Furthermore, if any foreign regulatory authority determines that EHP-101 or the FDA, DEA, or any foreign regulatory authority determines EHP-102 may have potential for abuse, it may require us to generate more clinical data than that which is currently anticipated, which could increase the cost and/or delay the launch of EHP-101 or EHP-102 in those countries.
When pharmaceutical products are deemed controlled substances, they are subject to a high degree of regulation under the CSA, which establishes, among other things, certain registration, manufacturing quotas, security, recordkeeping, reporting, import, export and other requirements administered by the DEA. The DEA classifies controlled substances into five schedules: Schedule I, II, III, IV or V substances. Schedule I substances by definition have a high potential for abuse, have no currently “accepted medical use” in the US, lack accepted safety for use under medical supervision, and may not be prescribed, marketed or sold in the US. Pharmaceutical products approved for use in the US may be listed as Schedule II, III, IV or V, with Schedule II substances considered to present the highest potential for abuse or dependence and Schedule V substances the lowest relative risk of abuse among such substances. Schedule I and II drugs are subject to the strictest controls under the CSA, including manufacturing and procurement quotas, security requirements and criteria for importation. In addition, dispensing of Schedule II drugs is further restricted. For example, they may not be refilled without a new prescription.
Cannabis and certain of the natural molecules found in the cannabis plant are currently Schedule I controlled substances. Products approved for medical use in the US that contain cannabis or cannabis extracts must therefore be placed in Schedules II - V, since approval by the FDA satisfies the “accepted medical use” requirement.
If any of our molecules are considered to be controlled substances because they were derived from cannabinoid molecules, to conduct preclinical studies and clinical trials in the US prior to approval, each of our research sites may be required to submit a research protocol to the DEA and obtain and maintain DEA researcher registration that will allow those sites to handle and dispense the product candidates and to obtain the product from our manufacturer. If the DEA delays or denies the grant of a research registration to one or more research sites, the preclinical studies or clinical trials could be significantly delayed, and we could lose and be required to replace clinical trial sites, resulting in additional costs.
In the event any of our drug candidates are considered to be controlled substances, if they are scheduled as Schedule II or III, we will need to identify wholesale distributors with the appropriate DEA registrations and authority to distribute the products to pharmacies and other healthcare providers, and these distributors would need to obtain Schedule II or III distribution registrations. The failure to obtain, a delay in obtaining, or the loss of any of those registrations could result in increased costs to us. Furthermore, state and federal enforcement actions, regulatory requirements, and legislation intended to reduce prescription drug abuse, such as the requirement that physicians consult a state prescription drug monitoring program, may make physicians less willing to prescribe, and pharmacies to dispense, Schedule II products. Further, if any of our molecules are deemed to be a Schedule II drug, the DEA must establish an annual aggregate quota for the amount that may be manufactured or produced in the US based on the DEA’s estimate of the quantity needed to meet legitimate medical, scientific, research and industrial needs. This limited aggregate amount that the DEA allows to be produced in the US each year is allocated among individual companies, which, in turn, must annually apply to the DEA for individual manufacturing and procurement quotas. The quotas apply equally to the manufacturing of the Active Pharmaceutical Ingredients (APIs) and production of dosage forms. The DEA may adjust aggregate production quotas a few times per year, and individual manufacturing or procurement quotas from time to time during the year, although the DEA has substantial discretion in whether or not to make such adjustments for individual companies. A failure by us to obtain adequate quota could have a material adverse effect on our business, financial condition, results of operations and cash flows.
10
There is material uncertainty as to how the Department of Justice will enforce laws and regulations related to marijuana under the CSA and evaluate marijuana cases for prosecution in light of various states legalizing marijuana for medicinal and/or recreational purposes. Such uncertainty creates risk in the Company’s business as it relates to the research, development, marketing and sale of those of its products derived from CBD and CBG which have not already been determined to not be a controlled substance.
If the Department of Justice exercises its prosecutorial discretion and prosecutes companies researching, developing, marketing or selling products containing controlled substances, and it is determined that any of our product candidates contain controlled substances, the results of our business operations may be adversely affected. If we or any party which we must indemnify is prosecuted as a result, it may distract management’s attention from our primary business and result in significant litigation costs. The recent DOJ actions underscore the uncertainty and often swift changing nature of the regulatory environment surrounding the CBD and CBG drug platforms.
Individual states have also established controlled substance laws and regulations. Though state-controlled substance laws often mirror federal law, because the states are separate jurisdictions, they may separately schedule our product candidates as well. While some states automatically schedule a drug based on federal action, other states schedule drugs through rulemaking or a legislative action. State scheduling may delay commercial sale of any product for which we obtain federal regulatory approval and adverse scheduling could have a material adverse effect on the commercial attractiveness of such product. We or our partners must also obtain separate state registrations, permits or licenses in order to be able to obtain, handle, and distribute controlled substances for clinical trials or commercial sale, and failure to meet applicable regulatory requirements could lead to enforcement and sanctions by the states in addition to those from the DEA or otherwise arising under federal law.
As with the federal controlled substance regulations, because EHP-102 is synthetically manufactured from synthetic CBG, and since under certain circumstances CBG is a controlled substance under the CSA, failure to receive regulatory approvals, or the risk of facing prosecution at either the federal or state level may hinder our operations and delay the launch of this product. We may face delays in our preclinical studies and clinical trials with EHP-102 in the US prior to approval. Under either DEA or state regulatory guidelines, each of our research sites may be required to submit a research protocol and obtain and maintain researcher registrations that would allow those sites to handle and dispense our product candidate and to obtain the product from our manufacturer. If the DEA or state regulatory body delays or denies the grant of a research registration to one or more research sites, the preclinical studies or clinical trials could be significantly delayed, and we could lose and be required to replace clinical trial sites, resulting in additional costs.
The manufacture, packaging and distribution of EHP-101 and EHP-102 is currently carried out in Europe, the US and China by large-scale contract manufacturers. We have conducted a Phase 1 clinical trial for EHP-101 in Australia and are initiating a multi-national Phase 2 clinical trial in Australia, New Zealand, and the US. In addition, we may decide to develop, manufacture or commercialize our product candidates in additional countries. As a result, we may also be subject to controlled substance laws and regulations from the Therapeutic Goods Administration in Australia and from other regulatory agencies in other countries where we develop, manufacture or commercialize EHP-101 or EHP-102 in the future. We plan to submit NDAs for EHP-101 and EHP-102 to the FDA upon completion of all requisite clinical trials and may require additional DEA approvals at such time as well.
If any of our product candidates are determined to be controlled substances, political and social pressures and adverse publicity could lead to delays in approval of, and increased expenses for, our product candidates. These pressures could also limit or restrict the introduction and marketing of our product candidates. Adverse publicity from cannabis misuse or adverse side effects from cannabis or other cannabinoid science products may adversely affect the commercial success or market penetration achievable by our product candidates.
11
Product shipment delays could have a material adverse effect on our business, results of operations and financial condition.
The shipment, import and export of EHP-101 and EHP-102 may require import and export licenses. In the US, the FDA, US Customs and Border Protection, and the DEA (for EHP-102); in Europe, where EHP-101and EHP-102 are currently manufactured, the EMA and the European Commission; in Australia, where we conduct clinical trials, the Australian Customs and Board Protection Service and the Therapeutic Goods Administration; and in other countries, similar regulatory authorities, regulate the import and export of pharmaceutical products that contain controlled substances. Specifically, the import and export processes require the issuance of import and export licenses by the relevant controlled substance authority in both the importing and exporting country. We may not be granted, or if granted, maintain, such licenses from the authorities in certain countries.
We have been granted orphan drug status by the FDA and EMA for EHP-101 for the treatment of SSc, and we have been granted orphan drug status by the FDA and EMA for EHP-102 for the treatment of HD, but we may be unable to maintain the benefits associated orphan drug status, including market exclusivity, which may cause our revenue, if any, to be reduced.
Regulatory authorities in some jurisdictions, including the US and EU, may designate drugs for relatively small patient populations as orphan drugs. The FDA may grant orphan drug designation to drugs intended to treat a rare disease or condition that affects fewer than 200,000 individuals annually in the US, or, if the disease or condition affects more than 200,000 individuals annually in the US, if there is no reasonable expectation that the cost of developing and making the drug would be recovered from sales in the US. In the EU, the EMA’s Committee for Orphan Medicinal Products grants orphan drug designation to promote the development of products that are intended for the diagnosis, prevention or treatment of life-threatening or chronically debilitating conditions affecting not more than five in 10,000 persons in the EU. Additionally, designation is granted for products intended for the diagnosis, prevention or treatment of a life-threatening, seriously debilitating or serious and chronic condition and when, without incentives, it is unlikely that sales of the drug in the EU would be sufficient to justify the necessary investment in developing the drug.
In the US, orphan drug designation entitles a party to financial incentives, such as opportunities for grant funding towards clinical trial costs, tax credits for certain research and user fee waivers under certain circumstances. In addition, if a product receives the first FDA approval for the drug and indication for which it has orphan drug designation, the product is entitled to seven years of market exclusivity, which means the FDA may not approve any other application for the same drug for the same indication for a period of seven years, except in limited circumstances, such as a showing of clinical superiority over the product with orphan drug exclusivity. Orphan drug exclusivity does not prevent the FDA from approving a different drug for the same disease or condition, or the same drug for a different disease or condition. In the EU, orphan drug designation also entitles a party to financial incentives such as reduction of fees or fee waivers and ten years of market exclusivity following drug approval. This period may be reduced to six years if the orphan drug designation criteria are no longer met, including where it is shown that the product is sufficiently profitable so that market exclusivity is no longer justified.
As a result, even though EHP-101 has received orphan drug designation in SSc in the US and in Europe, and EHP-102 has received orphan drug designation in HD in the US and EU, the FDA can still approve other drugs that have a different active ingredient for use in treating the same indications. If EHP-101 receives orphan drug exclusivity in the EU, the EMA could also, in defined circumstances, approve a competing product. Furthermore, the FDA can waive orphan drug exclusivity if we are unable to manufacture sufficient supply of EHP-101 or if the FDA finds that a subsequent applicant for SSc demonstrates clinical superiority to EHP-101. In addition, the European Commission could reduce the term of exclusivity if EHP-101 is sufficiently profitable.
We have received orphan drug designation for EHP-101 in SSc and for EHP-102 in HD from the FDA and the EMA, but exclusive marketing rights in the US may be limited if we seek approval for an indication broader than the orphan designated indication and may be lost if the FDA or EMA later determines that the request for designation was materially defective or if the manufacturer is unable to assure sufficient quantities of the product to meet the needs of patients with the rare disease or condition.
12
Even if we are able to commercialize EHP-101 or EHP-102, the products may not receive coverage and adequate reimbursement from third-party payors, which could harm our business.
The availability of reimbursement by governmental and private payors is essential for most patients to be able to afford expensive treatments. Sales of our product candidates, if approved, will depend substantially on the extent to which the costs of these product candidates will be paid by health maintenance, managed care, pharmacy benefit and similar healthcare management organizations, or reimbursed by government health administration authorities, private health coverage insurers and other third-party payors. If reimbursement is not available, or is available only to limited levels, we may not be able to successfully commercialize EHP-101 or EHP-102. Even if coverage is provided, the approved reimbursement amount may not be high enough to allow us to establish or maintain pricing sufficient to realize a sufficient return on our investment.
There is significant uncertainty related to the insurance coverage and reimbursement of newly approved products. In the US, the principal decisions about reimbursement for new medicines are typically made by the Centers for Medicare & Medicaid Services (CMS), an agency within the HHS, as CMS decides whether and to what extent a new medicine will be covered and reimbursed under Medicare. Private payors tend to follow CMS to a substantial degree.
Outside the US, particularly in EU Member States, the pricing of prescription drugs is subject to governmental control. In these countries, pricing negotiations or the successful completion of Health Technology Assessment (HTA) procedures with governmental authorities can take considerable time after receipt of marketing authorization for a product. In addition, there can be considerable pressure by governments and other stakeholders on prices and reimbursement levels, including as part of cost containment measures. Certain countries allow companies to fix their own prices for medicines but monitor and control company profits. Political, economic and regulatory developments may further complicate pricing negotiations, and pricing negotiations may continue after reimbursement has been obtained. Reference pricing used by various EU Member States and parallel distribution, or arbitrage between low-priced and high-priced EU member states, can further reduce prices. In some countries, we or our collaborators may be required to conduct a clinical trial or other studies that compare the cost-effectiveness of our product candidates to other available therapies in order to obtain or maintain reimbursement or pricing approval. Publication of discounts by third-party payors or authorities may lead to further pressure on the prices or reimbursement levels within the country of publication and other countries. If reimbursement of any product candidate approved for marketing is unavailable or limited in scope or amount, or if pricing is set at unsatisfactory levels, our business, financial condition, results of operations or prospects could be adversely affected.
Our relationships with customers and third-party payors will be subject to applicable anti-kickback, fraud and abuse and other healthcare laws and regulations, which could expose us to criminal sanctions, civil penalties, contractual damages, reputational harm and diminished profits and future earnings.
Healthcare providers, physicians and third-party payors play a primary role in the recommendation and prescription of any product candidates for which we obtain marketing approval. Our future arrangements with third-party payors and customers may expose us to broadly applicable fraud and abuse and other healthcare laws and regulations that may constrain the business or financial arrangements and relationships through which we market, sell and distribute our products for which we obtain marketing approval. As a pharmaceutical company, even though we do not and will not control referrals of healthcare services or bill directly to Medicare, Medicaid or other third-party payors, certain federal and state healthcare laws and regulations pertaining to fraud and abuse and patients’ rights are and will be applicable to our business.
Efforts to ensure that our business arrangements with third parties will comply with applicable healthcare laws and regulations will involve substantial costs. It is possible that governmental authorities will conclude that our business practices may not comply with current or future statutes, regulations or case law involving applicable fraud and abuse or other healthcare laws and regulations. If our operations are found to be in violation of any of these laws or any other governmental regulations that may apply to us, we may be subject to significant civil, criminal and administrative penalties, damages, fines, imprisonment, exclusion from government funded healthcare programs, such as Medicare and Medicaid, and the curtailment or restructuring of our operations. If any physicians or other healthcare providers or entities with whom we expect to do business are found to not be in compliance with applicable laws, they may be subject to criminal, civil or administrative sanctions, including exclusions from government funded healthcare programs.
13
Our employees may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements, which could subject us to significant liability and harm our reputation.
We are exposed to the risk of employee fraud or other misconduct. Misconduct by employees could include intentional failures to comply with DEA, FDA or EMA regulations or similar regulations of other foreign regulatory authorities or to provide accurate information to the DEA, FDA, EMA or other foreign regulatory authorities. In addition, misconduct by employees could include intentional failures to comply with certain manufacturing standards, to comply with US federal and state healthcare fraud and abuse laws and regulations and similar laws and regulations established and enforced by comparable foreign regulatory authorities, to report financial information or data accurately or to disclose unauthorized activities to us. Employee misconduct could also involve the improper use of information obtained in the course of clinical trials, which could result in regulatory sanctions and serious harm to our reputation. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a significant impact on our business and results of operations, including the imposition of significant fines or other sanctions.
If we are unable to develop sales, marketing and distribution capabilities or enter into agreements with third parties to perform these functions on acceptable terms, we may be unable to generate revenue.
We do not currently have any sales, marketing or distribution capabilities. If EHP-101 or EHP-102 is approved, we will need to develop internal sales, marketing and distribution capabilities to commercialize such products, which would be expensive and time-consuming, or enter into collaborations with third parties to perform these services. If we decide to market our products directly, we will need to commit significant financial and managerial resources to develop a marketing and sales force with technical expertise and supporting distribution, administration and compliance capabilities. If we rely on third parties with such capabilities to market our products or decide to co-promote products with collaborators, we will need to establish and maintain marketing and distribution arrangements with third parties, and there can be no assurance that we will be able to enter into such arrangements on acceptable terms or at all. In entering into third-party marketing or distribution arrangements, any revenue we receive will depend upon the efforts of the third parties and there can be no assurance that such third parties will establish adequate sales and distribution capabilities or be successful in gaining market acceptance of any approved product. If we are not successful in commercializing any product approved in the future, either on our own or through third parties, our business, financial condition and results of operations could be materially adversely affected.
Our product candidates, if approved, may be unable to achieve broad market acceptance and, consequently, limit our ability to generate revenue and profits from new products.
Even when product development is successful and regulatory approval has been obtained, our ability to generate significant revenue and profits depends on the acceptance of our products by physicians and patients. The market acceptance of any product depends on a number of factors, including but not limited to awareness of a product’s availability and benefits, the indication statement and warnings approved by regulatory authorities in the product label, continued demonstration of efficacy and safety in commercial use, perceptions by members of the health care community, including physicians, about the safety and effectiveness of our drugs, physicians’ willingness to prescribe the product, reimbursement from third-party payors such as government healthcare systems and insurance companies, the price of the product, pharmacological benefit and cost-effectiveness of our products relative to competing products; the nature of any post-approval risk management plans mandated by regulatory authorities, competition, and the effectiveness of marketing and distribution efforts. Any factors preventing or limiting the market acceptance of our product candidates could have a material adverse effect on our business, results of operations and financial condition.
14
If we receive regulatory approvals, we intend to market EHP-101 and EHP-102 in multiple jurisdictions where we have limited or no operating experience and may be subject to increased business and economic risks that could affect our financial results.
If we receive regulatory approvals, we plan to market EHP-101 and EHP-102 in jurisdictions where we have limited or no experience in marketing, developing and distributing our products. Certain markets have substantial legal and regulatory complexities that we may not have experience navigating. We are subject to a variety of risks inherent in doing business internationally, including risks related to the legal and regulatory environment in non-US jurisdictions, including with respect to privacy and data security, trade control laws and unexpected changes in laws, regulatory requirements and enforcement, as well as risks related to fluctuations in currency exchange rates and political, social and economic instability in foreign countries. If we are unable to manage our international operations successfully, our financial results could be adversely affected. In addition, controlled substance legislation may differ in other jurisdictions and could restrict our ability to market our products internationally. Most countries are parties to the Single Convention on Narcotic Drugs 1961, which governs international trade and domestic control of narcotic substances, including cannabis extracts. Countries may interpret and implement their treaty obligations in a way that creates a legal obstacle to us obtaining marketing approval for EHP-101 or EHP-102 in those countries. These countries may not be willing or able to amend or otherwise modify their laws and regulations to permit EHP-101 or EHP-102 to be marketed, or achieving such amendments to the laws and regulations may take a prolonged period of time. We would be unable to market EHP-101 or EHP-102 in countries with such obstacles in the near future or perhaps at all without modification to laws and regulations.
Any inability to attract and retain qualified key management and technical personnel would impair our ability to implement our business plan.
Our success largely depends on the continued service of key management and other specialized personnel. The loss of one or more members of our management team or other key employees or consultants could delay our research and development programs and materially harm our business, financial condition, results of operations and prospects. The relationships that our team has cultivated within the life sciences industry makes us particularly dependent upon their continued employment or services with us. Because our management team is not obligated to provide us with continued service, they could terminate their employment or services with us at any time without penalty, subject to providing any required advance notice. We do not maintain key person life insurance policies for any members of our management team. Our future success and growth will depend in large part on our continued ability to attract and retain other highly qualified scientific, technical and management personnel and consultants, as well as personnel and consultants with expertise in clinical testing, manufacturing, governmental regulation and commercialization. We face competition for personnel and consultants from other companies, universities, public and private research institutions, government entities and other organizations.
We face substantial competition, which may result in others discovering, developing or commercializing products before or more successfully than we do.
The development and commercialization of drugs is highly competitive. We compete with a variety of multinational pharmaceutical companies and specialized biotechnology companies, as well as products and processes being developed at universities and other research institutions. Our competitors have developed, are developing or will develop product candidates and processes competitive with our product candidates. Competitive therapeutic treatments include those that have already been approved and accepted by the medical community and any new treatments that may enter the market. We believe that a significant number of products are currently available, under development, and may become commercially available in the future, for the treatment of indications for which we may try to develop product candidates. If either of our product candidates, EHP-101 and EHP-102, is approved for the indications we are currently pursuing, it will compete with a range of therapeutic treatments that are either in development or currently marketed.
While our molecules are NCEs and, therefore, are not technically cannabis molecules, we are aware of multiple companies that are working in the cannabis therapeutic area, including pharmaceutical companies such as GW Pharmaceuticals plc (GW), which markets Sativex, a botanical cannabinoid for the treatment of spasticity due to multiple sclerosis and Epidiolex, a botanical cannabinoid for the treatment of two rare childhood seizure disorders.
More established companies may have a competitive advantage over us due to their greater size, cash flows and institutional experience. Compared to us, many of our competitors may have significantly greater financial, technical and human resources. As a result of these factors, our competitors may have an advantage in marketing their approved products and may obtain regulatory approval of their product candidates before we are able to, which may limit our ability to develop or commercialize our product candidates. Our competitors may also develop drugs that are safer, more effective, more widely used and less expensive than ours, and may also be more successful than us in manufacturing and marketing their products. These advantages could materially impact our ability to develop and, if approved, commercialize EHP-101 or EHP-102 successfully.
15
Our product candidates may compete with non-synthetic cannabinoid drugs, including therapies such as GW’s Sativex and Epidiolex. Our product candidates may also compete with medical and recreational marijuana, in markets where the recreational and/or medical use of marijuana is legal. There is support in the US for further legalization of marijuana. In markets where recreational and/or medical marijuana is not legal, our product candidates may compete with marijuana purchased in the illegal drug market. We cannot assess the extent to which patients may utilize marijuana obtained illegally for the treatment of the indications for which we are developing EHP-101 and EHP-102.
Mergers and acquisitions in the pharmaceutical and biotechnology industries may result in even more resources being concentrated among a smaller number of our competitors. Smaller and other early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. These companies compete with us in recruiting and retaining qualified scientific, management and commercial personnel, establishing clinical trial sites and subject registration for clinical trials, as well as in acquiring technologies complementary to, or necessary for, our programs.
Product liability lawsuits against us could cause us to incur substantial liabilities.
Our use of EHP-101 and EHP-102 in clinical trials and the sale of EHP-101 and EHP-102, if approved, exposes us to the risk of product liability claims. Product liability claims might be brought against us by patients, healthcare providers or others selling or otherwise coming into contact with EHP-101 or EHP-102. For example, we may be sued if any product we develop allegedly causes injury or is found to be otherwise unsuitable during product testing, manufacturing, marketing or sale. Any such product liability claims may include allegations of defects in manufacturing, defects in design, a failure to warn of dangers inherent in the product, including as a result of interactions with alcohol or other drugs, negligence, strict liability, and a breach of warranties. Claims could also be asserted under state consumer protection acts. If we become subject to product liability claims and cannot successfully defend ourselves against them, we could incur substantial liabilities. In addition, regardless of merit or eventual outcome, product liability claims may result in, among other things:
| ● | withdrawal of patients from our expected clinical trials; | |
| ● | substantial monetary awards to patients or other claimants; | |
| ● | decreased demand for EHP-101 or EHP-102 following marketing approval, if obtained; | |
| ● | damage to our reputation and exposure to adverse publicity; | |
| ● | increased FDA warnings on product labels or increased warnings imposed by the European Commission or other applicable regulatory authorities; | |
| ● | litigation costs; | |
| ● | distraction of management’s attention from our primary business: | |
| ● | loss of revenue; and | |
| ● | the inability to successfully commercialize EHP-101 or EHP-102, if approved. |
16
Business or economic disruptions or global health concerns could seriously harm our development efforts and increase our costs and expenses.
Broad-based business or economic disruptions could adversely affect our ongoing or planned research and development. For example, in December 2019 an outbreak of a novel strain of coronavirus originated in Wuhan, China, and has since spread to a number of other countries, including the US. To date, the COVID-19 outbreak has already resulted in extended shutdowns of certain businesses in the Wuhan region and has had ripple effects to businesses around the world. The outbreak has resulted in and may continue to have additional or more extensive travel restrictions, closures, disruptions of businesses or facilities in China and other affected regions around the world, and lead to social, economic, political and/or labor/workforce instability in the affected areas, which may impact our, our suppliers’ and/or our customers’ operations. The impact of the COVID-19 health emergency has also affected the regulatory agencies’ ability to monitor and perform routine regulatory reviews and inspections, which may prolong regulatory processes.
Global epidemics and pandemics, such as the coronavirus, could also negatively affect the hospitals and clinical sites in which we conduct any of our clinical trials, which could have a material adverse effect on our business and our results of operation and financial condition. We cannot presently predict the scope and severity of any potential business shutdowns or disruptions, but if we or any of the third parties with whom we engage, including the suppliers, clinical trial sites, regulators and other third parties with whom we conduct business, were to experience shutdowns or other business disruptions, our ability to conduct our business in the manner and on the timelines presently planned could be materially and negatively impacted.
Risks Related to Our Intellectual Property
If we are unable to protect our intellectual property rights or if our intellectual property rights are inadequate for our technology and product candidates, our competitive position could be harmed.
Our commercial success will depend in large part on our ability to obtain and maintain patent and other intellectual property protection in the US and other countries with respect to our proprietary technology and products. We rely on trade secret, patent, copyright and trademark laws, and confidentiality and other agreements with employees and third parties, all of which offer only limited protection. We seek to protect our proprietary position by filing and prosecuting patent applications in the US and abroad related to our technologies and products that are important to our business.
The patent positions of biotechnology and pharmaceutical companies generally are highly uncertain, involve complex legal and factual questions and have in recent years been the subject of much litigation. As a result, the issuance, scope, validity, enforceability and commercial value of our patents are highly uncertain. The steps we have taken to protect our proprietary rights may not be adequate to preclude misappropriation of our proprietary information or infringement of our intellectual property rights, both inside and outside the US. Our pending applications cannot be enforced against third parties practicing the technology claimed in such applications unless and until patents issue from such applications. Further, the examination process may require us to narrow the claims for our pending patent application, which may limit the scope of patent protection that may be obtained if these applications issue. We do not know whether the pending patent applications for any of our product candidates will result in the issuance of any patents that protect our technology or products, or if any of our issued patents will effectively prevent others from commercializing competitive technologies and products. The rights already granted under any of our currently issued patents and those that may be granted under future issued patents may not provide us with the proprietary protection or competitive advantages we are seeking. If we are unable to obtain and maintain patent protection for our technology and products, or if the scope of the patent protection obtained is not sufficient, our competitors could develop and commercialize technology and products similar or superior to ours, and our ability to successfully commercialize our technology and products may be adversely affected. It is also possible that we will fail to identify patentable aspects of inventions made in the course of our development and commercialization activities before it is too late to obtain patent protection on them.
Because the issuance of a patent is not conclusive as to its inventorship, scope, validity or enforceability, our issued patents may be challenged in the courts or patent offices in the US and abroad. Any granted patents may be subjected to further post-grant proceedings that could limit their scope or enforceability. Claims that are amended during post-grant proceedings may not be broad enough to provide meaningful protection, which could limit our ability to stop others from using or commercializing similar or identical technology and products, or limit the duration of the patent protection for our technology and products. Publications of discoveries in the scientific literature often lag behind the actual discoveries, and patent applications in the US and other jurisdictions are typically not published until 18 months after filing. Therefore, we cannot be certain that we were the first to make the inventions claimed in our owned patents or pending patent application, or that we were the first to file for patent protection of such inventions.
17
Protecting against the unauthorized use of our patented technology, trademarks and other intellectual property rights is expensive, difficult and may in some cases not be possible. In some cases, it may be difficult or impossible to detect third-party infringement or misappropriation of our intellectual property rights, even in relation to issued patent claims, and proving any such infringement may be even more difficult.
Obtaining and maintaining our patent protection depends on compliance with various procedural, document submission, fee payment and other requirements imposed by governmental patent agencies, and our patent protection could be reduced or eliminated for non-compliance with these requirements.
The US Patent and Trademark Office (USPTO) and various foreign national or international patent agencies require compliance with a number of procedural, documentary, fee payment and other similar provisions during the patent application process. Periodic maintenance fees on any issued patent are due to be paid to the USPTO and various foreign national or international patent agencies in several stages over the lifetime of the patent. While an inadvertent lapse can in certain cases be cured by payment of a late fee or by other means in accordance with the applicable rules, there are situations in which noncompliance can result in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. Non-compliance events that could result in abandonment or lapse of patent rights include, but are not limited to, failure to timely file national and regional stage patent applications or continuing applications thereof, based on our international patent applications, failure to respond to official actions within prescribed time limits, non-payment of fees and failure to properly legalize and submit formal documents. If we fail to maintain the patents and patent applications covering our product candidates, our competitors might be able to enter the market, which would have a material adverse effect on our business.
We may become subject to claims by third parties asserting that we or our employees have misappropriated their intellectual property, or claiming ownership of what we regard as our own intellectual property.
Our commercial success depends upon our ability to develop, manufacture, market and sell our product candidates, and to use our related proprietary technologies without violating the intellectual property rights of others. We may become party to, or threatened with, future adversarial proceedings or litigation regarding intellectual property rights with respect to our product candidates, including interference or derivation proceedings before the USPTO. Third parties may assert infringement or post grant invalidation claims against us based on existing patents or patents that may be granted in the future. If we are found to infringe a third party’s intellectual property rights, we could be required to obtain a license from such third party to continue commercializing our product candidates. However, we may not be able to obtain any required license on commercially reasonable terms or at all. Under certain circumstances, we could be forced, including by court order, to cease commercializing the applicable product candidate. In addition, in any such proceeding or litigation, we could be found liable for monetary damages. A finding of infringement could prevent us from commercializing our product candidates or force us to cease some of our business operations, which could materially harm our business. Any claims by third parties that we have misappropriated their confidential information or trade secrets could have a similar negative impact on our business.
While our preclinical studies and expected clinical trials are ongoing, we believe that the use of EHP-101 and EHP-102 in these preclinical studies and expected clinical trials falls within the scope of the exemptions provided by 35 USC. Section 271(e)(1) in the US, which exempts from patent infringement liability activities reasonably related to the development and submission of information to the FDA. As EHP-101 and EHP-102 progress toward commercialization, the possibility of a patent infringement claim against us increases. We attempt to ensure that our product candidates and the methods we employ to manufacture them, as well as the methods for their uses we intend to promote, do not infringe other parties’ patents and other proprietary rights. There can be no assurance they do not, however, and competitors or other parties may assert that we infringe their proprietary rights in any event.
18
We may become involved in lawsuits to protect or enforce our intellectual property, which could be expensive, time consuming and unsuccessful and have a material adverse effect on the success of our business.
Competitors may infringe our patents or misappropriate or otherwise violate our intellectual property rights. To counter infringement or unauthorized use, litigation may be necessary in the future to enforce or defend our intellectual property rights, to protect our trade secrets or to determine the validity and scope of our own intellectual property rights or the proprietary rights of others. Also, third parties may initiate legal proceedings against us to challenge the validity or scope of intellectual property rights we own. These proceedings can be expensive and time consuming. Many of our current and potential competitors have the ability to dedicate substantially greater resources to defend their intellectual property rights than we can. Accordingly, despite our efforts, we may not be able to prevent third parties from infringing upon or misappropriating our intellectual property. Litigation could result in substantial costs and diversion of management resources, which could harm our business and financial results. In addition, in an infringement proceeding, a court may decide that a patent owned by us is invalid or unenforceable, or may refuse to stop the other party from using the technology at issue on the grounds that our patents do not cover the technology in question. An adverse result in any litigation proceeding could put one or more of our patents at risk of being invalidated, held unenforceable or interpreted narrowly. Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation. There could also be public announcements of the results of hearings, motions or other interim proceedings or developments.
We may not be able to protect our intellectual property rights throughout the world.
Filing, prosecuting and defending patents on all of our product candidates throughout the world would be prohibitively expensive. Therefore, we have filed applications and/or obtained patents only in key markets such as the US, EU, Japan, Canada and selected other countries. Competitors may use our technologies in jurisdictions where we have not obtained patent protection to develop their own products and, further, may be able to export otherwise infringing products to territories where we have patent protection but where enforcement is not as strong as that in the US. Recent US case law indicates that patent enforcement may not provide enough protection against resale of lower priced drugs in the US made in extraterritorial jurisdictions. These products may compete with our products in jurisdictions where we do not have any issued patents and our patent claims or other intellectual property rights may not be effective or sufficient to prevent them from so competing.
Many companies have encountered significant problems in protecting and defending intellectual property rights in certain foreign jurisdictions. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents and other intellectual property protection, particularly those relating to pharmaceuticals, which could make it difficult for us to stop the infringement of our patents or marketing of competing products in violation of our proprietary rights generally. As a result, proceedings to enforce our patent rights in certain foreign jurisdictions could result in substantial cost and divert our efforts and attention from other aspects of our business and could be unsuccessful.
Patent terms may be inadequate to protect our competitive position on our product candidates for an adequate amount of time.
Given the amount of time required for the development, testing and regulatory review of new product candidates, patents protecting such candidates might expire before or shortly after such candidates are commercialized. We expect to seek extensions of patent terms in the US and, if available, in other countries where we are prosecuting patents. In the US, the Drug Price Competition and Patent Term Restoration Act of 1984 permits a patent term extension of up to five years beyond the normal expiration of the patent, which is limited to the active ingredient and approved indication (or any additional indications approved during the period of extension). However, the applicable authorities, including the FDA and the USPTO, and any equivalent regulatory authorities in other countries, may not agree with our assessment of whether such extensions are available, and may refuse to grant extensions to our patents, or may grant more limited extensions than we request. If this occurs, our competitors may be able to take advantage of our investment in development and clinical trials by referencing our clinical and preclinical data and launch their product earlier than might otherwise be the case.
19
Risks Related to Our Company
We have a very limited operating history on which to judge our business prospects and management.
The Company was incorporated on March 2, 2017 and only commenced operations thereafter. Accordingly, we have a very limited operating history upon which to base an evaluation of our business and prospects. Operating results for future periods are subject to numerous uncertainties and we cannot assure you that the Company will achieve or sustain profitability. The Company’s prospects must be considered in light of the risks encountered by companies in the early stage of development, particularly companies in new and rapidly evolving markets. Future operating results will depend upon many factors, including our success in attracting and retaining motivated and qualified personnel, our ability to establish short term credit lines or obtain financing from other sources, such as the contemplated Offering, our ability to develop and market new products, control costs, and general economic conditions. We cannot assure you that the Company will successfully address any of these risks.
Our financial situation creates doubt whether we will continue as a going concern.
Since inception, the Company has not generated revenues, has incurred losses and has an accumulated deficit of $33.1 million as of June 30, 2020. Further, we expect to incur a net loss for the fiscal year ending December 31, 2020 and thereafter, primarily as a result of increased operating expenses related to the expected clinical trials. There can be no assurances that we will be able to achieve a level of revenues adequate to generate sufficient cash flow from operations or obtain funding from this Offering or additional financing through private placements, public offerings and/or bank financing necessary to support our working capital requirements. To the extent that funds generated from any private placements, public offerings and/or bank financing are insufficient, we will have to raise additional working capital. No assurance can be given that additional financing will be available, or if available, will be on acceptable terms. These conditions raise substantial doubt about our ability to continue as a going concern. If adequate working capital is not available, we may be forced to discontinue operations, which would cause investors to lose their entire investment. Our auditors have indicated that these conditions raise substantial doubt about the Company’s ability to continue as a going concern.
We will need but may be unable to obtain additional funding on satisfactory terms, which could dilute our stockholders or impose burdensome financial restrictions on our business.
We have relied upon our majority stockholder and our current Regulation A+ offering to finance our operations to date, and in the future, we plan to rely on additional sources to fund all of the cash requirements of our activities. However, there can be no assurance that our current funding sources or activities will continue to finance our operations or that we will be able to generate any significant cash from our operating activities in the future. Our majority stockholder has financed our operations through a revolving loan agreement, under which we have the ability to continue borrowing although such continued funding is not guaranteed. The loan may be repaid by us or, at the option of our majority stockholder, converted by our majority stockholder into shares of the Company at $2.00 per share, which, if converted would significantly dilute stockholders purchasing Shares in this Offering. Total aggregate advances under the loan as of June 30, 2020 amounted to $11.3 million, of which $5.0 million has been repaid and $2.5 million has been converted into 1,250,000 shares of Common Stock. As of June 30, 2020, $3.75 million of principal and $0.4 million of interest payable remains outstanding under the loan. If you purchase Shares in this Offering, your ownership interest in our Common Stock may be diluted if our majority stockholder elects to convert the additional amounts outstanding under the loan in the future.
Future financings may not be available on a timely basis, in sufficient amounts or on terms acceptable to us, if at all. Any debt financing or other financing of securities senior to the Common Stock will likely include financial and other covenants that will restrict our flexibility. Any failure to comply with these covenants would have a material adverse effect on our business, prospects, financial condition and results of operations because we could lose our existing sources of funding and impair our ability to secure new sources of funding. However, there can be no assurance that the Company will be able to generate any investor interest in its securities. If we do not obtain additional financing, our business will never commence, in which case you would likely lose the entirety of your investment in us.
We will incur increased costs as a result of our public reporting obligations, and our management team will be required to devote substantial time to new compliance initiatives.
We are currently subject to the periodic reporting requirements of Regulation A for qualified issuers. Particularly after we are no longer an “emerging growth company,” we will continue to incur significant legal, accounting and other expenses that we did not incur as a private company. Our management and other personnel would need to devote a substantial amount of time to comply with our reporting obligations. Moreover, these reporting obligations will increase our legal and financial compliance costs and will make some activities more time-consuming and costly.
20
Failure to develop our internal controls over financial reporting as we grow could have an adverse impact on us.
As our Company matures we will need to continue to develop and improve our current internal control systems and procedures to manage our growth. We are required to establish and maintain appropriate internal controls over financial reporting. Failure to establish appropriate controls, or any failure of those controls once established, could adversely impact our public disclosures regarding our business, financial condition or results of operations. In addition, management’s assessment of internal controls over financial reporting may identify weaknesses and conditions that need to be addressed in our internal controls over financial reporting or other matters that may raise concerns for investors. Any actual or perceived weaknesses and conditions that need to be addressed in our internal control over financial reporting, disclosure of management’s assessment of our internal controls over financial reporting or disclosure of our public accounting firm’s attestation to or report on management’s assessment of our internal controls over financial reporting may have an adverse impact on the price of our Common Stock.
Our internal computer systems, or those of our third-party CROs or other contractors or consultants, may fail or suffer security breaches, which could result in a material disruption of our product candidates’ development programs.
Despite the implementation of security measures, our internal computer systems and those of our third-party CROs and other contractors and consultants are vulnerable to damage from computer viruses, unauthorized access, natural disasters, terrorism, war and telecommunication and electrical failures. While we have not experienced any such system failure, accident, or security breach to date, if such an event were to occur and cause interruptions in our operations, it could result in a material disruption of our programs. For example, the loss of clinical trial data for our product candidates could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data. To the extent that any disruption or security breach results in a loss of or damage to our data or applications or other data or applications relating to our technology or product candidates, or inappropriate disclosure of confidential or proprietary information, we could incur liabilities and the further development of our product candidates could be delayed. The sharing of important clinical data can raise significant Health Insurance Portability and Accountability Act (HIPAA) privacy and security compliance issues. Also, General Data Protection Regulation (GDPR) severely restricts the sharing of the personal health information related to EU residents or transferring that data outside of the EU. HIPAA and GDPR compliance can increase costs and liabilities as we collect and analyze data.
Unpredictable events, such as the COVID-19 outbreak, and associated business disruptions, including delayed clinical trials and laboratory resources, could harm our financial condition, affect our operations, increase our costs and expenses, and impact our ability to raise capital.
Our operations could be subject to unpredictable events, such as earthquakes, power shortages, telecommunications failures, water shortages, floods, hurricanes, typhoons, fires, extreme weather conditions, medical epidemics such as the COVID-19 outbreak, and other natural or manmade disasters or business interruptions, for which we may not be insured. We do not carry insurance for all categories of risk that our business may encounter. The occurrence of any of these business disruptions could seriously harm our operations and financial condition, delay our product development and regulatory approvals of clinical trials, and increase our costs and expenses. Additionally, COVID-19 has caused significant disruptions to the global financial markets, which could impact our ability to raise additional capital. The ultimate impact on us and any delays in our research and development is unknown, but our operations and financial condition could suffer in the event of any of these types of unpredictable events. Further, any significant uninsured liability may require us to pay substantial amounts, which would adversely affect our business, results of operations, financial condition and cash flows.
21
Risks Related to Our Financial Position and Need for Capital
Even if this Offering is successful, we will need to raise additional funding, which may not be available on acceptable terms, or at all. Failure to obtain this necessary capital when needed may force us to delay, limit or terminate our product development efforts or other operations.
The proceeds from the offering of Shares pursuant to this Offering Circular will be up to $17,100,000 before deducting offering expenses payable by us. We expect that if the maximum sale of shares is achieved, the net proceeds from this Offering will be sufficient to fund our current operations at least through the end of 2021. However, (a) we may not achieve the maximum sale of shares, and/or (b) our operating plan may change as a result of many factors currently unknown to us, and we may need to seek additional funds sooner than planned, through public or private equity or debt financings, government or other third-party funding, marketing and distribution arrangements and other collaborations, strategic alliances and licensing arrangements or a combination of these approaches. In any event, we will require additional capital to obtain regulatory approval for, and to commercialize, our product candidates. Raising funds in the current economic environment may present additional challenges. It is not certain that we have accounted for all costs and expenses of future development and regulatory compliance. Even if we believe we have sufficient funds for our current or future operating plans, we may seek additional capital if market conditions are favorable or if we have specific strategic considerations.
Any additional fundraising efforts may divert our management from their day-to-day activities, which may adversely affect our ability to develop and commercialize our product candidates. In addition, we cannot guarantee that future financing will be available in sufficient amounts or on terms acceptable to us, if at all. Moreover, the terms of any financing may adversely affect the holdings or the rights of our stockholders and the issuance of additional securities, whether equity or debt, by us, or the possibility of such issuance, may cause the market price of our shares to decline. The sale of additional equity or convertible securities may dilute our existing stockholders. The incurrence of indebtedness would result in increased fixed payment obligations and we may be required to agree to certain restrictive covenants, such as limitations on our ability to incur additional debt, limitations on our ability to acquire, sell or license intellectual property rights and other operating restrictions that could adversely impact our ability to conduct our business. We could also be required to seek funds through arrangements with collaborative partners or otherwise at an earlier stage than otherwise would be desirable and we may be required to relinquish rights to some of our technologies or product candidate or otherwise agree to terms unfavorable to us, any of which may have a material adverse effect on our business, operating results and prospects.
If we are unable to obtain funding on a timely basis, we may be required to significantly curtail, delay or discontinue one or more of our research or development programs or the commercialization of any product candidate or be unable to expand our operations or otherwise capitalize on our business opportunities, as desired, which could materially affect our business, financial condition and results of operations.
If you purchase our Shares in this Offering, you will incur immediate and substantial dilution in the book value of your Shares.
You will suffer immediate and substantial dilution in the net tangible book value of the Shares you purchase in this Offering. Assuming an offering price of $6.00 per share and all 2,850,000 Shares are sold for gross proceeds of $17,100,000, purchasers of Shares in this Offering will experience immediate dilution of $4.40 per share in net tangible book value of the Shares. In addition, investors purchasing Shares in this Offering will contribute up to 25% of the total amount invested by stockholders since inception but will only own 12.5% of the shares of Common Stock outstanding.
In addition, our majority stockholder has financed our operations through a revolving loan agreement. We have the ability to continue borrowing under the loan although such continued borrowing is not guaranteed. The outstanding loan balance and accrued interest may be repaid by us or, at the option of our majority stockholder, converted by our majority stockholder into shares of the Company at $2.00 per share. As of June 30, 2020, $11.3 million has been advanced to us under the loan, of which $5.0 million has been repaid and $2.5 million has been converted into 1,250,000 shares of Common Stock. As of June 30, 2020, $3.75 million of principal and $0.4 million of interest payable remains outstanding under the loan. If you purchase Shares in this Offering, your ownership interest in our Common Stock may be diluted if our majority stockholder elects to convert the additional amounts outstanding under the loan in the future.
No minimum capitalization.
We do not have a minimum capitalization and we may use the proceeds from this Offering immediately following our acceptance of the corresponding subscription agreements. There can be no assurance we will sell the Maximum Amount in this Offering. As of November 20, 2020, we have received investment commitments totaling approximately $49.1 million, including both completed and pending sales, under this Offering, however there is no assurance that we will raise sufficient capital solely from this Offering to implement our business plan, potentially resulting in greater operating losses unless we are able to raise the required capital from alternative sources. There is no assurance that alternative capital, if needed, would be available on terms acceptable to us, or at all.
22
Risks Related to Our Common Stock
We have a significant stockholder, which may limit your ability to influence corporate matters and may give rise to conflicts of interest.
Our majority stockholder is EHS, a private company focused on building companies advancing the development of cannabis and cannabinoids. As of November 20, 2020, EHS owned approximately 51.3% of our outstanding Common Stock. Following this Offering, assuming all of the Shares offered hereby are sold, we anticipate that EHS will own approximately 45% of our outstanding Common Stock. In addition, EHS may elect to convert all or any portion of the outstanding balance under our revolving loan agreement (currently a principal balance of $3.75 million) into shares of our Common Stock at a conversion price of $2.00 per share. Accordingly, EHS exerts and may continue to exert significant influence over us and any action requiring the approval of the holders of our Common Stock, including the election of directors and amendments to our organizational documents, such as increases in our authorized shares of Common Stock and approval of significant corporate transactions. This concentration of ownership may prevent or discourage unsolicited acquisition proposals or offers for our Common Stock that you may feel are in your best interest as one of our stockholders. Furthermore, the interests of EHS may not always coincide with your interests or the interests of other stockholders and EHS may act in a manner that advances its best interests and not necessarily those of other stockholders, including seeking a premium value for its Common Stock, which might affect the prevailing market price for our Common Stock.
We engage in transactions with related parties and such transactions present possible conflicts of interest that could have an adverse effect on us.
We have entered into transactions with related parties. The details of certain of these transactions are set forth in “Interest of Management and Others in Certain Transactions.” One such transaction is our loan received from EHS, our majority stockholder, which has an outstanding principal balance of $3.75 million as of June 30, 2020.
Related party transactions create the possibility of conflicts of interest with regard to our management, including that:
| ● | we may enter into contracts between us, on the one hand, and related parties, on the other, that are not the result of arm’s-length transactions; | |
| ● | our executive officers and directors that hold positions of responsibility with related parties may be aware of certain business opportunities that are appropriate for presentation to us as well as to such other related parties and may present such business opportunities to such other parties; and | |
| ● | our executive officers and directors that hold positions of responsibility with related parties may have significant duties with, and spend significant time serving, other entities and may have conflicts of interest in allocating time. |
Such conflicts could cause an individual in our management to seek to advance his or her economic interests or the economic interests of certain related parties above ours. Further, the appearance of conflicts of interest created by related party transactions could impair the confidence of our investors. Notwithstanding this, it is possible that a conflict of interest could have a material adverse effect on our liquidity, results of operations and financial condition.
23
Our executive officers, directors, major stockholder and their respective affiliates will continue to exercise significant control over our Company after this Offering, which will limit your ability to influence corporate matters and could delay or prevent a change in corporate control.
Immediately following the completion of this Offering, and disregarding any Shares that they purchase in this Offering, if any, our executive officers, directors, and majority stockholder will own, in the aggregate, approximately 49% of our outstanding Common Stock, assuming we issue the number of shares of Common Stock as set forth on the cover page of this Offering Circular. Please see “Security Ownership of Management & Certain Security Holders” on page 63 for more information. As a result, these stockholders may be able to influence our management and affairs and control the outcome of matters submitted to our stockholders for approval, including the election of directors and any sale, merger, consolidation, or sale of all or substantially all of our assets. These stockholders acquired their shares of Common Stock for substantially less than the price of the Shares being acquired in this Offering, and these stockholders may have interests, with respect to their Common Stock, that are different from those of investors in this Offering and the concentration of voting power among one or more of these stockholders may have an adverse effect on the price of our Common Stock. In addition, this concentration of ownership might adversely affect the market price of our Common Stock by:
| ● | delaying, deferring or preventing a change of control of the Company; | |
| ● | impeding a merger, consolidation, takeover or other business combination involving the Company; or | |
| ● | discouraging a potential acquirer from making a tender offer or otherwise attempting to obtain control of the Company. |
Conflicts of Interest
The Company may be subject to various potential conflicts of interest because of the fact that some of its officers and directors may be engaged in a range of business activities. Two of the non-employee directors of the Company are also directors of EHS. In addition, the Company’s executive officers and directors may devote time to their outside business interests, so long as such activities do not materially or adversely interfere with their duties to the Company. In some cases, the Company’s executive officers and directors may have fiduciary obligations associated with these business interests that interfere with their ability to devote time to the Company’s business and affairs and that could adversely affect the Company’s operations. These business interests could require significant time and attention of the Company’s executive officers and directors.
We have broad discretion in how we use the proceeds of this Offering and may not use these proceeds effectively, which could affect our results of operations and cause our Common Stock price to decline.
We will have considerable discretion in the application of the net proceeds of this Offering. We intend to use the net proceeds from this Offering primarily to fund our business strategy, including without limitation, new and ongoing research and development expenses, offering expenses, working capital and other general corporate purposes, and, prior to our use of the proceeds, other uses, including short-term, interest-bearing and capital preservation investments, as more specifically set forth in “Use of Proceeds.” As a result, investors will be relying upon management’s judgment with only limited information about our specific intentions for the use of the balance of the net proceeds of this Offering. We may use the net proceeds for purposes that do not yield a significant return or any return at all for our stockholders. In addition, pending their use, we may invest the net proceeds from this Offering in a manner that does not produce income or that loses value.
There is no existing market for our Common Stock, and you cannot be certain that an active trading market or a specific share price will be established.
Prior to this Offering, there has been no public market for shares of our Common Stock. We cannot predict the extent to which investor interest in our Company will lead to the development of a trading market or how liquid that market might become. The Offering price for the Shares has been arbitrarily determined by the Company and may not be indicative of the price that will prevail in any trading market following this Offering, if any. The market price for our Common Stock may decline below the Offering price, and our stock price is likely to be volatile.
We will use our best efforts to list our Common Stock for trading on a securities exchange however it is uncertain when our Common Stock will be listed on an exchange for trading, if ever.
There is currently no public market for our Common Stock and there can be no assurance that one will ever develop. Our Board of Directors, in its sole discretion, may choose to take actions necessary to list our Common Stock on a national securities exchange, but is not obligated to do so. As a result, our Common Stock sold in this Offering may not be listed on a securities exchange for an extended period of time, if at all. If our Common Stock is not listed on an exchange it may be difficult to sell or trade in shares of our Common Stock.
24
If our stock price fluctuates after the Offering, you could lose a significant part of your investment.
The market price of our Common Stock could be subject to wide fluctuations in response to, among other things, the risk factors described in this section of this Offering Circular, and other factors beyond our control, such as fluctuations in the valuation of companies perceived by investors to be comparable to us. Furthermore, the stock markets have experienced price and volume fluctuations that have affected and continue to affect the market prices of equity securities of many companies. These fluctuations often have been unrelated or disproportionate to the operating performance of those companies. These broad market and industry fluctuations, as well as general economic, political, and market conditions, such as recessions, interest rate changes or international currency fluctuations, may negatively affect the market price of our Common Stock. In the past, many companies that have experienced volatility in the market price of their stock have been subject to securities class action litigation. We may be the target of this type of litigation in the future. Securities litigation against us could result in substantial costs and divert our management’s attention from other business concerns, which could seriously harm our business.
Limitations of director liability and indemnification of directors, officers and employees.
Our Certificate of Incorporation, as amended (Certificate of Incorporation) limits the liability of directors to the maximum extent permitted by Delaware law. Delaware law provides that directors of a corporation will not be personally liable for monetary damages for breach of their fiduciary duties as directors, except for liability for any:
| ● | breach of their duty of loyalty to us or our stockholders; | |
| ● | act or omission not in good faith or that involves intentional misconduct or a knowing violation of law; | |
| ● | unlawful payments of dividends or unlawful stock repurchases or redemptions as provided in Section 174 of the Delaware General Corporation Law; or | |
| ● | transactions for which the directors derived an improper personal benefit. |
These limitations of liability do not apply to liabilities arising under the federal or state securities laws and do not affect the availability of equitable remedies such as injunctive relief or rescission. Our corporate bylaws provide that we will indemnify our directors, officers and employees to the fullest extent permitted by law. Our bylaws also provide that we are obligated to advance expenses incurred by a director or officer in advance of the final disposition of any action or proceeding. We believe that these bylaw provisions are necessary to attract and retain qualified persons as directors and officers. We have entered into, and are authorized to enter into, indemnification agreements with our current and future officers and directors. The limitation of liability in our Certificate of Incorporation, bylaws and the indemnification agreements we have entered into with our officers and directors may discourage stockholders from bringing a lawsuit against directors for breach of their fiduciary duties. They may also reduce the likelihood of derivative litigation against directors and officers, even though an action, if successful, might provide a benefit to us and our stockholders. Our results of operations and financial condition may be harmed to the extent we pay the costs of settlement and damage awards against directors and officers pursuant to these indemnification provisions.
After the completion of this Offering, we may be at an increased risk of securities class action litigation.
Historically, securities class action litigation has often been brought against a company following a decline in the market price of its securities. This risk is especially relevant for us because biotechnology and pharmaceutical companies have experienced significant stock price volatility in recent years. If we were to be sued, it could result in substantial costs and a diversion of management’s attention and resources, which could harm our business.
We do not intend to pay dividends on our Common Stock and, consequently, your ability to achieve a return on your investment will depend on appreciation in the price of our Common Stock.
We have never declared or paid any cash dividend on our Common Stock and do not currently intend to do so in the foreseeable future. We currently anticipate that we will retain future earnings for the development, operation and expansion of our business and do not anticipate declaring or paying any cash dividends in the foreseeable future. Therefore, the success of an investment in the Shares will depend upon any future appreciation in their value. There is no guarantee that the Shares will appreciate in value or even maintain the price at which you purchased them.
25
We may terminate this Offering at any time during the Offering Period.
We reserve the right to terminate this Offering at any time, regardless of the number of Shares sold. In the event that we terminate this Offering at any time prior to the sale of all of the Shares offered hereby, whatever amount of capital that we have raised at that time will have already been utilized by the Company and no funds will be returned to subscribers.
Risks Related to Our Dependence on Third Parties
We will rely on third parties to conduct our research, preclinical studies and current and expected clinical trials. If these third parties do not successfully carry out their contractual duties or meet expected deadlines, we may not be able to obtain regulatory approval for or commercialize our product candidates.
We rely on CROs, clinical data management organizations and consultants to design and/or conduct, supervise and monitor our research, preclinical studies and clinical trials. We and our CROs are required to comply with various regulations which are enforced by the FDA and other regulatory agencies, including GCP and Good Laboratory Practices (GLP), and guidelines of the Competent Authorities of Member States of the EEA and comparable foreign regulatory authorities to ensure that the health, safety and rights of animals and patients are protected in preclinical studies, clinical development and clinical trials, and that trial data integrity is assured. Regulatory authorities ensure compliance with these requirements through periodic inspections of trial sponsors, principal investigators and trial sites. Our reliance on third parties that we do not control does not relieve us of these responsibilities and requirements. If we or any of our CROs fail to comply with applicable requirements, the clinical data generated in our clinical trials may be deemed unreliable and the FDA, the EMA or other comparable foreign regulatory authorities may require us to perform additional clinical trials before approving our marketing applications. We cannot assure you that upon inspection by a given regulatory authority, such regulatory authority will determine that any of our clinical trials comply with such requirements. In addition, our clinical trials must be conducted with products produced under cGMP requirements, which mandate, among other things, the methods, facilities and controls used in manufacturing, processing and packaging of a drug product to ensure its safety and identity. Failure to comply with these regulations may require us to repeat preclinical studies and clinical trials, which would delay the regulatory approval process.
Our CROs are not our employees, and except for remedies available to us under our agreements with such CROs, we cannot control whether they devote sufficient time and resources to our ongoing research, preclinical and clinical programs. If CROs do not successfully carry out their contractual duties or obligations or meet expected deadlines or if the quality or accuracy of the clinical data they obtain is compromised due to the failure to adhere to our clinical protocols, regulatory requirements or for other reasons, our clinical trials may be extended, delayed or terminated and we may not be able to obtain regulatory approval for or successfully commercialize our product candidates. As a result, our operations and the commercial prospects for our product candidates would be harmed, our costs could increase and our ability to generate revenue could be delayed or reduced. If their facilities are unable to operate because of an accident or incident, even for a short period of time, some or all of our research and development programs may be harmed or delayed, and our operations and financial condition could suffer.
Because we have relied on third parties, our internal capacity to perform these functions is limited. In addition, the use of third-party service providers requires us to disclose our proprietary information to these parties, which could increase the risk that this information will be misappropriated. We currently have a small number of employees, which limits the internal resources we have available to identify and monitor our third-party providers. To the extent we are unable to identify and successfully manage the performance of third-party service providers in the future, our business may be adversely affected. Though we carefully manage our relationships with our CROs, there can be no assurance that we will not encounter similar challenges or delays in the future or that these delays or challenges will not have a material adverse impact on our business, financial condition and prospects.
26
We rely on third-party manufacturers and suppliers to produce preclinical and clinical supplies and intend to rely on third-party manufacturers for commercial supplies, of APIs and final dosage forms for EHP-101 and EHP-102, if approved.
We rely on third parties to supply the materials for, and manufacture, our research and development, and preclinical and clinical trial APIs. We do not own manufacturing facilities or supply sources for such components and materials. There can be no assurance that our supply of research and development, preclinical and clinical development drugs and other materials will not be limited, interrupted, restricted in certain geographic regions or of satisfactory quality or continue to be available at acceptable prices. In particular, any replacement of our API manufacturer could require significant effort and expertise because there may be a limited number of qualified manufacturers.
The manufacturing process for our product candidates is subject to review by the FDA, EMA, and other foreign regulatory authorities and potentially the DEA. Suppliers and manufacturers must meet applicable manufacturing requirements and undergo rigorous facility and process validation tests required by regulatory authorities in order to comply with regulatory standards such as cGMP. In addition, our manufacturers must ensure consistency among batches, including preclinical, clinical and, if approved, commercial batches. Demonstrating such consistency may require typical manufacturing controls as well as clinical data. Our manufacturers must also ensure that our batches conform to release specifications. Further, if one or more of our products are considered to be or contain a controlled substance, manufacturers of controlled substances must obtain and maintain necessary DEA and state registrations and registrations with applicable foreign regulatory authorities, and must establish and maintain processes to ensure compliance with DEA and state requirements and requirements of applicable foreign regulatory authorities governing, among other things, the storage, handling, security, recordkeeping and reporting for controlled substances. In the event that any of our suppliers or manufacturers fails to comply with such requirements or to perform its obligations to us in relation to quality, timing or otherwise, or if our supply of components or other materials becomes limited or interrupted for other reasons, we may be forced to manufacture the materials ourselves, for which we currently do not have the capabilities or resources, or enter into an agreement with another third party, which we may not be able to do on reasonable terms, if at all. In some cases, the technical skills or technology required to manufacture our product candidates may be unique or proprietary to the original manufacturer and we may have difficulty, or there may be contractual restrictions prohibiting us from, transferring such skills or technology to another third party and a feasible alternative may not exist. These factors would increase our reliance on such manufacturer or require us to obtain a license from such manufacturer in order to have another third party manufacture our product candidates. If we are required to change manufacturers for any reason, we will be required to verify that the new manufacturer maintains facilities and procedures that comply with quality standards and with all applicable regulations and guidelines. The delays associated with the verification of a new manufacturer could negatively affect our ability to develop product candidates in a timely manner or within budget.
We expect to continue to rely on third-party manufacturers if we receive regulatory approval for any product candidate. To the extent that we have existing, or enter into future, manufacturing arrangements with third parties, we will depend on these third parties to perform their obligations in a timely manner consistent with contractual and regulatory requirements, including those related to quality control and assurance. If we are unable to obtain or maintain third-party manufacturing for product candidates, or to do so on commercially reasonable terms, we may not be able to develop and commercialize our product candidates successfully. Our or a third party’s failure to execute on our manufacturing requirements could adversely affect our business in a number of ways, including:
| ● | an inability to initiate or continue preclinical studies or expected clinical trials of product candidates under development; | |
| ● | delay in submitting regulatory applications, or receiving regulatory approvals, for product candidates; | |
| ● | loss of the cooperation of a collaborator; | |
| ● | subjecting our product candidates to additional inspections by regulatory authorities; and | |
| ● | in the event of approval to market and commercialize a product candidate, an inability to meet commercial demands for our products. |
Our third-party manufacturers also may use hazardous materials, including chemicals and compounds that could be dangerous to human health and safety or the environment, and their operations may also produce hazardous waste products. In the event of contamination or injury, our third-party manufacturers could be held liable for damages or be penalized with fines in an amount exceeding their resources, which could result in our clinical trials or regulatory approvals being delayed or suspended.
27
As of November 20, 2020, an aggregate of 19,054,632 shares of our Common Stock were outstanding. There are also an additional 913,410 shares of Common Stock issuable pertaining to commitments we have received under the Offering for which processing or funding is not completed as of the date hereof. In addition, there are 3,429,833 shares of Common Stock reserved for issuance under our 2018 Equity Incentive Plan as of November 20, 2020, with 2,857,500 shares of Common Stock issuable pursuant to outstanding awards, of which 817,500 shares of Common Stock will be issuable upon exercise of outstanding awards at $6.00 per share, 275,000 shares of Common Stock will be issuable upon exercise of outstanding awards at $5.00 per share and 1,765,000 shares of Common Stock will be issuable upon exercise of outstanding awards at $2.50 per share. Future awards could be issued at per share prices above or below the Offering Price.
If you purchase Shares in this Offering, your ownership interest in our Common Stock will be diluted immediately, to the extent of the difference between the price to the public charged for each share in this Offering and the net tangible book value per share of our Common Stock after this Offering.
We currently have a revolving convertible loan agreement with our majority stockholder. The loan may be converted by our majority stockholder into Common Stock at $2.00 per share (the Conversion). Total aggregate advances under the loan as of June 30, 2020 amounted to $11.3 million, of which $5.0 million has been repaid and $2.5 million has been converted into 1,250,000 shares of Common Stock. As of June 30, 2020, $3.75 million of principal and $0.4 million of interest payable remains outstanding under the loan. If you purchase Shares in this Offering, your ownership interest in our Common Stock may be diluted if our majority stockholder elects to convert the additional amounts outstanding under the loan in the future.
Our net tangible book value as of June 30, 2020 was $(1.8) million or $(0.11) per share based on 15,925,711 outstanding shares of Common Stock as of June 30, 2020. Our pro forma net tangible book value as of June 30, 2020 includes net proceeds from the sales and pending sales of 4,037,331 shares of Common Stock under the Offering from July 1, 2020 through November 20, 2020 of approximately, $22.6 million. Net tangible book value per share equals the amount of our total tangible assets less total liabilities, divided by the total number of shares of our Common Stock outstanding, all as of the date specified.
If the Maximum Amount of $17,100,000 is raised in this Offering, after deducting approximately $1.4 million in Offering expenses, including broker-dealer fees and commissions payable by us, our pro forma as adjusted net tangible book value at June 30, 2020 would be approximately $20.8 million or $1.04 per share and our pro forma net tangible book value after completion of the Offering would be approximately $36.5 million or $1.60 per share. This amount represents an immediate increase in pro forma net tangible book value of $0.56 per share to our existing stockholders at June 30, 2020, and an immediate dilution in pro forma net tangible book value of approximately $4.40 per share to investors purchasing Shares in this Offering.
The following table illustrates the per share dilution to investors discussed above, assuming the sale of, respectively, 100%, 75%, 50% and 25% of the Shares offered for sale in this Offering (after deducting our estimated offering expenses of $1.4 million) as of June 30, 2020:
| Funding Level | $ | 17,100,000 | $ | 12,825,000 | $ | 8,550,000 | $ | 4,275,000 | ||||||||
| Offering Price | $ | 6.00 | $ | 6.00 | $ | 6.00 | $ | 6.00 | ||||||||
| Pro forma net tangible book value per Share at June 30, 2020(1) | $ | 1.04 | $ | 1.04 | $ | 1.04 | $ | 1.04 | ||||||||
| Increase per Share attributable to investors in this Offering | $ | 0.56 | $ | 0.42 | $ | 0.27 | $ | 0.11 | ||||||||
| Proforma net tangible book value per Share after Offering | $ | 1.60 | $ | 1.46 | $ | 1.31 | $ | 1.15 | ||||||||
| Dilution to investors after the Offering | $ | 4.40 | $ | 4.54 | $ | 4.69 | $ | 4.85 | ||||||||
| Increase per Share attributable to investors in this Offering after Conversion and Option Exercise | $ | 0.81 | $ | 0.70 | $ | 0.59 | $ | 0.47 | ||||||||
| Pro forma net tangible book value per Share after the Offering and Conversion and Option Exercise | $ | 1.85 | $ | 1.74 | $ | 1.63 | $ | 1.51 | ||||||||
| Dilution to investors after the Offering, Conversion and Option Exercise(2) | $ | 4.15 | $ | 4.26 | $ | 4.37 | $ | 4.49 |
| (1) | Includes 4,037,331 additional shares for which subscription commitments have been received between July 1, 2020 and November 20, 2020. |
| (2) | Includes the exercise of 2,857,500 outstanding options under the 2018 Equity Incentive Plan. |
28
The following tables set forth, assuming the sale of, respectively, 100%, 75%, 50% and 25% of the shares offered for sale in this Offering the total number of shares previously sold to existing stockholders as of November 20, 2020, the total consideration paid for the foregoing and the respective percentages applicable to such purchased shares and consideration paid based on an average price of $0.22 per share paid by our founding stockholders, $5.62 paid by all other stockholders and $6.00 per share paid by new investors in the Offering. The tables do not include the effects of Conversion or any exercise of outstanding awards under the 2018 Equity Incentive Plan.
| Shares Purchased | Total Consideration | |||||||||||||||
| Number | Percentage | Amount | Percentage | |||||||||||||
| Assuming 100% of Shares Sold: | ||||||||||||||||
| Founding Stockholders | 11,175,000 | 48.97 | % | $ | 2,500,993 | 3.62 | % | |||||||||
| All Other Stockholders | 8,793,042 | 38.54 | % | $ | 49,432,716 | 71.61 | % | |||||||||
| New Investors in this Offering | 2,850,000 | 12.49 | % | $ | 17,100,000 | 24.77 | % | |||||||||
| Total | 22,818,042 | 100.00 | % | $ | 69,033,709 | 100.00 | % | |||||||||
| Shares Purchased | Total Consideration | |||||||||||||||
| Number | Percentage | Amount | Percentage | |||||||||||||
| Assuming 75% of Shares Sold: | ||||||||||||||||
| Founding Stockholders | 11,175,000 | 50.55 | % | $ | 2,500,993 | 3.86 | % | |||||||||
| All Other Stockholders | 8,793,042 | 39.78 | % | $ | 49,432,716 | 76.33 | % | |||||||||
| New Investors in this Offering | 2,137,500 | 9.67 | % | $ | 12,825,000 | 19.80 | % | |||||||||
| Total | 22,105,542 | 100.00 | % | $ | 64,758,709 | 100.00 | % | |||||||||
| Shares Purchased | Total Consideration | |||||||||||||||
| Number | Percentage | Amount | Percentage | |||||||||||||
| Assuming 50% of Shares Sold: | ||||||||||||||||
| Founding Stockholders | 11,175,000 | 52.24 | % | $ | 2,500,993 | 4.13 | % | |||||||||
| All Other Stockholders | 8,793,042 | 41.10 | % | $ | 49,432,716 | 81.73 | % | |||||||||
| New Investors in this Offering | 1,425,000 | 6.66 | % | $ | 8,550,000 | 14.14 | % | |||||||||
| Total | 21,393,042 | 100.00 | % | $ | 60,483,709 | 100.00 | % | |||||||||
| Shares Purchased | Total Consideration | |||||||||||||||
| Number | Percentage | Amount | Percentage | |||||||||||||
| Assuming 25% of Shares Sold: | ||||||||||||||||
| Founding Stockholders | 11,175,000 | 54.03 | % | $ | 2,500,993 | 4.45 | % | |||||||||
| All Other Stockholders | 8,793,042 | 42.52 | % | $ | 49,432,716 | 87.94 | % | |||||||||
| New Investors in this Offering | 712,500 | 3.45 | % | $ | 4,275,000 | 7.61 | % | |||||||||
| Total | 20,680,542 | 100.00 | % | $ | 56,208,709 | 100.00 | % | |||||||||
29
PLAN OF DISTRIBUTION & SELLING SECURITYHOLDERS
The Shares are being offered by us on a “best-efforts” basis. There is no aggregate minimum to be raised in order for the Offering to become effective and therefore the Offering will be conducted on a “rolling basis.” This means we are entitled to begin applying “dollar one” of the proceeds from the Offering towards our business strategy, including, without limitation, research and development expenses, offering expenses, working capital, general corporate purposes, and, prior to our use of the proceeds, other uses, including short-term, interest-bearing investments, as more specifically set forth in the “Use of Proceeds” starting on page 31.
The Shares may be offered through broker-dealers who are registered with FINRA. The proceeds received in this Offering will be deposited into an escrow account (the “Escrowed Funds”) pursuant to our Escrow Services Agreement with Prime Trust, LLC dated July 26, 2019. The Escrowed Funds will be distributed to the Company and the purchased Shares will be issued to the investors upon closing. The proceeds of this Offering may be deposited directly into the Company’s operating account for immediate use by it, with no obligation to refund subscriptions. The Company has engaged Dalmore, a broker-dealer registered with the SEC and a member of FINRA, to perform administrative, compliance and related broker-dealer services in connection with this Offering, including the review of investor information, including KYC (Know Your Customer) data, AML (Anti-Money Laundering) and other compliance checks, and review of subscription agreements and investor information.
Generally speaking, Rule 3a4-1 provides an exemption from the broker-dealer registration requirements of the Exchange Act for persons associated with an issuer that participate in an offering of the issuer’s securities. None of our officers or directors are subject to any statutory disqualification, as that term is defined in Section 3(a)(39) of the Exchange Act. None of our officers or directors will be compensated in connection with his participation in the offering by the payment of commissions or other remuneration based either directly or indirectly on transactions in our securities. None of our officers or directors are, or have been within the past 12 months, a broker or dealer, and none of them are, or have been within the past 12 months, an associated person of a broker or dealer. At the end of the offering, our officers or directors will continue to primarily perform substantial duties for the Company or on its behalf otherwise than in connection with transactions in securities. Our officers or directors will not participate in selling an offering of securities for any issuer more than once every 12 months other than in reliance on Exchange Act Rule 3a4-1(a)(4)(i) or (iii) except that for securities issued pursuant to rule 415 under the Securities Act, the 12 months shall begin with the last sale of any security included within one rule 415 registration.
Selling Securityholders
No securities are being sold for the account of securityholders; all net proceeds of this Offering will go to the Company.
30
If the Maximum Amount is sold in the Offering pursuant to this Offering Circular, the maximum gross proceeds from the sale of our Shares will be $17,100,000. The net proceeds from the Offering are expected to be approximately $15.7 million if the Maximum Amount is sold, after the payment of Offering costs, including broker-dealer fees and selling commissions, legal and accounting costs, filing fees, marketing and selling expenses. The estimate of the budget for Offering costs is an estimate only and the actual Offering costs may differ. We expect from time to time to evaluate the acquisition of businesses, intellectual property, products and technologies for which a portion of the net proceeds may be used, although we currently are not planning or negotiating any such transactions. The following table represents management’s best estimate of the uses of the net proceeds received from the sale of the Shares assuming the sale of, respectively, 100%, 75%, 50% and 25% of Shares offered for sale in this Offering.
Percentage of Offering Sold
| 100% | 75% | 50% | 25% | |||||||||||||
| Clinical studies | $ | 10,994,200 | $ | 8,031,625 | $ | 5,069,050 | $ | 2,106,475 | ||||||||
| Research and development | $ | 1,256,480 | $ | 917,900 | $ | 579,320 | $ | 240,740 | ||||||||
| General and administrative | $ | 3,455,320 | $ | 2,524,225 | $ | 1,593,130 | $ | 662,035 | ||||||||
| TOTAL | $ | 15,706,000 | $ | 11,473,750 | $ | 7,241,500 | $ | 3,009,250 |
We are a pre-revenue clinical-stage biotechnology/pharmaceutical company and began operations in March 2017. Our plan of operations for the next few years includes advancing the development of two initial therapeutic product candidates, EHP-101 and EHP-102, that together target four initial indications: MS, SSc, PD and HD. We have successfully completed a Phase 1 human clinical study in Australia to establish EHP-101 safety and pharmacokinetics (PK) in healthy volunteers. We have initiated a Phase 2a safety and efficacy study in SSc patients and we plan to commence a Phase 2 study in MS patients later this year. If such studies are successful, the product candidates will then advance into additional efficacy studies thereafter. We have completed preclinical proof of concept (POC) work for EHP-102. We are now in the manufacturing and formulation development stage and, if successfully completed, expect to begin clinical-enabling studies for HD and PD and then advance to Phase 1 human clinical studies.
The amounts set forth above are our current estimates for such development, and we cannot be certain that actual costs will not vary from these estimates. Our management has significant flexibility and broad discretion in applying the net proceeds received in this Offering. We cannot assure you that our assumptions, expected costs and expenses and estimates will prove to be accurate or that unforeseen events, problems or delays will not occur that would require us to seek additional debt and/or equity funding, which may not be available on favorable terms, or at all. See “Risk Factors” starting on page 5.
The Company intends to use a portion of the proceeds raised in this Offering to fund the compensation payable to its executive officers and directors as described under “Compensation of Directors and Executive Officers” below. In addition, a portion of the proceeds raised may be used to pay, in whole or in part, the principal and/or the accrued interest on our loan with EHS.
This expected use of the net proceeds from this Offering represents our intentions based upon our current financial condition, results of operations, business plans and conditions. As of the date of this Offering Circular, we cannot predict with certainty all of the particular uses for the net proceeds to be received upon the closing of this Offering or the amounts that we will actually spend on the uses set forth above. The amounts and timing of our actual expenditures may vary significantly depending on numerous factors. As a result, our management will retain broad discretion over the allocation of the net proceeds from this Offering.
Although our business does not presently generate any cash, we believe that if we raise the Maximum Amount in this Offering, that we will have sufficient capital to finance our operations for at least through the end of 2021. However, if we do not sell the Maximum Amount or if our operating and development costs are higher than expected, we will need to obtain additional financing prior to that time. Further, we expect that during and/or after such period, we will be required to raise additional funds to finance our operations until such time that we can conduct profitable revenue-generating activities.
Pending our use of the net proceeds from this Offering, we intend to invest the net proceeds in a variety of capital preservation investments, including, without limitation, short-term, investment grade, interest bearing instruments and United States government securities. We may also use a portion of the net proceeds for the investment in strategic partnerships and possibly the acquisition of complementary businesses, products or technologies, although we have no present commitments or agreements for any specific acquisitions or investments.
31
Overview
We are a biotechnology/pharmaceutical company focused on developing drug product candidates currently containing novel, patented molecules chemically derived from two non-psychoactive cannabinoids, cannabidiol (CBD) and cannabigerol (CBG) to treat diseases with unmet medical needs, primarily autoimmune, neurodegenerative, inflammatory and fibrotic diseases. We are currently developing two initial therapeutic product candidates that together target four initial indications, multiple sclerosis (MS), systemic sclerosis (SSc, a severe form of scleroderma), Parkinson’s disease (PD) and Huntington’s disease (HD). We believe treatments for these indications represent markets with underserved patient populations.
Our platform technology is a result of the unique convergence of biology, science and cannabinoids, and consists of a library of twenty-five novel, patented derivatives of CBD and CBG. The resulting molecules are new chemical entities (NCEs) covered by seventeen issued international patents. In addition, we have twenty-one pending patent applications. We believe our cannabinoid-based technology platform represents an advancement to existing therapies because our NCEs are derived from CBD and CBG and chemically modified to act on additional biological receptors and pathways in the body to specifically treat the diseases we are targeting, which CBD and CBG alone do not affect.
Our current product pipeline includes two initial product candidates from our library of NCEs, EHP-101 and EHP-102. EHP-101 is an oral formulation of a novel synthetic CBD derivative, known as VCE-004.8, and is our lead candidate, currently in Phase 2 clinical development; EHP-102 is a formulation of a novel synthetic CBG derivative, known as VCE-003.2, currently in preclinical development.
Based on our studies to date, we believe that these initial product candidates have the potential to treat several diseases with unmet medical need. We are currently targeting four distinct diseases, two for each of these initial product candidates. With EHP-101, we are initially targeting MS and SSc, and with EHP-102, we are initially targeting PD and HD. Other applications are also being investigated, with our two current product candidates, different formulations and other molecules within our NCE portfolio.
We have been granted Orphan Drug Designation (ODD) from the FDA in the US and from the EMA in the EU for EHP-101 for the SSc indication and for EHP-102 for the HD indication. We have also received Fast Track designation by the FDA for EHP-101 for the SSc indication.
The starting material for the active pharmaceutical ingredient (API) in our product candidates are CBD and CBG, which may be classified by the DEA as controlled substances in the US depending on their origin and purity. In March 2019 we received a decision from the DEA that the API (VCE-004.8) in our lead product candidate (EHP-101) is not a controlled substance, based mainly on the facts that our molecule is (1) an NCE which is no longer CBD, (2) chemically derived from synthetic CBD containing no remaining CBD or other controlled substances and (3) non-psychoactive. We have also received the same decision from the UK Home Office and Canada’s Controlled Substances Directorate. In general, the determination that VCE-004.8 is not a controlled substance reduces the costs and complexities otherwise associated with developing controlled substances. With this determination, manufacturing facilities do not require controlled substance certification for handling and dispensing the molecules and drug products. It also facilitates importation and simplifies the conduct of nonclinical and clinical studies, as contracted nonclinical research organizations and clinical sites have less administrative burden. Once we advance our second product candidate (EHP-102) further in development, we will request a similar decision from the DEA, and other countries, for this product candidate.
32
Background and Pathology
The Endocannabinoid System
Based on current scientific knowledge, we believe the body’s endocannabinoid system (ECS) promotes homeostasis (biological balance) in our cells, tissues and organs, which can help reduce inflammation and support brain, immune, and nervous system function and overall health and wellness.
The ECS is presently thought to include:
| ● | main receptor sites on cells called cannabinoid type-1 (CB1) and cannabinoid type-2 (CB2) receptors; | |
| ● | compounds known as endocannabinoids, such as anandamide and 2-arachydonoil glycerol, which are produced in the body from dietary fats, that bind to and/or affect, CB1 and CB2 receptors; and | |
| ● | enzymes that impact the production and metabolism of these endocannabinoids. |
In general, receptors within the body send vital information to cells, organs, and the nervous system, and are critical to maintaining optimal health and a stable internal environment, or homeostasis, despite fluctuations in the external environment. In the human body, the ECS is believed to have more cellular receptor sites than any other receptor system. The ECS is comprised of receptor sites, mainly CB1 and CB2 receptors, that can be described as “lock-and-key” mechanisms, which are activated (unlocked) by specific molecules (the keys) produced in the body (endocannabinoids), or from plants (phytocannabinoids) and cannabinoid-like compounds, both natural and synthetic.
Endocannabinoids and their receptors are found throughout the body: in the brain, organs, connective tissues, glands, and immune cells. In each tissue, the ECS performs different functions, but the goal is always the same: homeostasis.
We believe the role of the ECS in maintaining homeostasis could make it a promising target in medicine. The widespread distribution of cannabinoid receptors suggests that the ECS is important in maintaining overall bodily function and health, including the brain and peripheral organs. This, along with the ability of our novel CBD and CBG derivative molecules to affect additional receptors and pathways within the body, provides us with the potential for unique therapeutic advantages over current therapies for the diseases we are targeting.
Only CB1receptors in the brain are responsible for the psychoactive effects of cannabis. As with the natural molecules, CBD and CBG, our synthetic derivative molecules do not interact with CB1 receptors and, therefore, have no potential for psychoactive effects. In our studies conducted to date, this lack of psychoactive effects has been verified.
Our Unique Technological Advantage
Our patented molecules are NCEs derived from CBD and CBG. Based on the scientific and anecdotal information currently available, we believe that CBD and CBG have a good safety profile and may provide positive health and therapeutic effects, primarily through interactions with the ECS. Our strategy in the creation of our cannabinoid derivatives is to improve upon these health and therapeutic benefits and maintain the good safety profile by chemically modifying the CBD and CBG molecules so some interact with certain ECS receptors more efficiently, as well as additional well-known receptors and physiologic pathways outside the ECS related to specific, life-threatening diseases. Using this strategy, we currently have rationally designed 25 molecules with possible capabilities to treat diseases based on their MOA. We know of no other products on the market or product candidates in development that possess the same combined multi-target MOA as our novel molecules in our lead product candidates. For additional information on our patent families, see “Intellectual Property” below.
33
Biologic Receptors and Physiologic Pathways Involved in Our Initial Product Candidates
We believe that one of the competitive advantages of our technology is the combined effect that the API (VCE-004.8) in our lead product candidate (EHP-101) has on various biologic receptors and physiologic/biochemical pathways not displayed by other molecules (including cannabinoids). Our synthetic cannabinoid derivatives are designed to improve the bioactivities of their natural precursors and provide opportunities to advance their development into pharmaceutical products. Some examples of these receptors and pathways are:
Cannabinoid Receptor Type-2 (CB2)
Cannabinoids are a complex group of molecules that comprise phytocannabinoids (from plants), endocannabinoids (naturally occurring within the body) and synthetic cannabinoids (made synthetically). Cannabinoids were initially identified by their ability to affect the classical endocannabinoid receptors CB1 and CB2. Tetrahydrocannabinol (THC), the primary psychoactive component of cannabis, produces many of its psychoactive effects by engaging CB1 cannabinoid receptors. CB2 receptors have been the subject of considerable attention, primarily due to their promising therapeutic potential for treating various pathologies while avoiding the adverse psychotropic effects that can accompany CB1 receptor-based therapies. We believe the multi-target activity of cannabinoids (specifically CBD and CBG) accounts for their ability to modulate several key processes including neuroprotection, inflammation, immunomodulation and vascular responses. In studies conducted to date, our CBD-derived technology has shown it can enhance the CB2 receptor modulation activity of CBD and, therefore, can potentially increase therapeutic benefits.
Peroxisome Proliferator-Activated Receptors
Peroxisome Proliferator-Activated Receptors (PPARs) play essential roles in the regulation of cellular differentiation, development, and metabolism (carbohydrate, lipid, protein). Three types of PPARs have been identified, alpha (α), gamma (γ), and beta/delta (β/δ). PPARγ is a nuclear receptor originally implicated in the regulation of cell growth, lipid metabolism and blood sugar regulation. However, PPARγ is broadly expressed and has been recognized to play a key role in inflammatory and connective tissue balance. PPARγ activators have been shown to prevent inflammation, dermal fibrosis and loss of fatty tissue. PPARγ is activated by some endocannabinoids and related signaling lipids, as well as by certain natural and synthetic cannabinoids. In studies conducted to date, our CBD- and CBG-derived technology has shown modulation of PPARγ activity, providing the potential for immunomodulatory activity and neuroprotection and the promotion of oligodendrocyte progenitor cell differentiation and enhancement of their antioxidant defenses.
34
Hypoxia-Inducible Factor Pathway
Hypoxia-Inducible Factor (HIF)-1 is a protein complex that plays an integral role in the body’s response to low oxygen concentrations, or hypoxia. HIF-1 is among the primary genes involved in the homeostatic process and has two subunits, HIF-1α and HIF-1β. HIF operates in all mammalian cell types and responds to changes in oxygen, providing cells with a master regulator that coordinates changes in gene transcription. Hypoxia preconditioning induced by mild hypoxia can be beneficial in a wide number of disorders including neurologic and inflammatory diseases. Cellular adaptation to severe or mild hypoxia begins immediately with the activation of the HIF pathway, and regulates a plethora of genes involved in many biological processes, including red blood cell production, angiogenesis, neuroprotection, myelination, vascular tone and immunity. HIF-1α activation may play a role in the inflammatory and the remitting phases of MS. In addition, there is evidence suggesting that activation of the HIF pathway may be also linked to neuroprotection and myelination. Thus, the erythropoietin (EPO) gene is HIF-dependent and EPO is neuroprotective in different animal models of MS. In addition, HIF-1α activates several blood vessel-forming genes, including vascular endothelial growth factor (VEGF-A) and fibroblast growth factor-2 (FGF-2), which are mainly produced by vascular endothelial cells. The vascular endothelial cells produce factors that help maintain brain homeostasis within the context of the neurovascular unit. In general, HIF-1α activates many genes whose products exert neuroprotective activities and also HIF-1α activation is implicated in the modulation of the immune system. In studies conducted to date, our CBD-derived technology has shown activation of the HIF pathway, providing the potential for neuroprotection and vascular protection.
Others
The API (VCE-003.2) in our second product candidate (EHP-102) has been shown to affect PPARγ, like EHP-101. Unlike EHP-101, however, it does not affect CB2 receptors or the HIF pathway, but has been shown to activate a transcription factor involved in nerve cell differentiation (the chicken ovalbumin upstream promoter transcription factor-interacting protein, Ctip2). It also reduces the expression of cyclooxygenase-2 (COX-2) in nerve cells (involved in inflammation and pain) and is an activator of the extracellular signal-regulated kinases (ERK) pathway (a member of the mitogen-activated protein kinases [MAPK] pathway) in hippocampal neuronal progenitor cells.
Our Initial Product Candidates
We call our initial product candidates EHP-101 and EHP-102. EHP-101 is a formulation of one of our CBD derivatives (VCE-004.8) for oral administration and EHP-102 is a formulation of one of our CBG derivatives (VCE-003.2) currently being developed for oral administration. Based on the combination of biologic receptors and physiologic/chemical pathways affected by our product candidates, we believe our cannabinoid-based technology could be suitable medications for neurodegenerative, neurological, autoimmune, inflammatory, metabolic and fibrotic disorders. We believe that EHP-101 and EHP-102 have the potential to be disease modifying, while most other compounds approved for many of these diseases are limited to targeting the symptoms of the disease.
Here is a summary of our two initial product candidates:
EHP-101 (VCE-004.8)
Overview
Our lead product candidate, EHP-101, is an oral formulation of an NCE called VCE 004.8, derived from CBD that affects some of the known validated biologic receptors and physiologic pathways involved in MS and SSc. Thus, our first two chosen indications for EHP-101 are MS and SSc.
We believe that the PPARγ and CB2activators have strong potential to directly affect the disease in MS and SSc. EHP-101 is a formulated product containing VCE-004.8, a ligand agonist of PPARγ and CB2, that can directly bind and activate both receptors as demonstrated by in vitro binding and transcriptional assays. We believe the combination of activities toward both PPARγ and CB2-dependent signaling pathways could represent an important advancement in the development of anti-inflammatory and antifibrotic therapies for MS and SSc.
35
In addition to PPARγ and CB2, VCE-004.8 also activates the HIF pathway, which also has potential benefits in MS and SSc. Studies have indicated that HIF-1α activation may play a role in inflammatory and remitting phases of MS. For instance, HIF-1α activates many genes whose products exert neuroprotective activities. HIF-1α activation is also implicated in the modulation of the immune system. In addition, there is strong evidence suggesting that activation of the HIF pathway may be linked to neuroprotection and myelination. HIF-1α also activates several genes involved in vascular physiology, including VEGF-A and FGF-2, which are mainly produced by vascular endothelial cells. The vascular endothelial cells produce factors that maintain brain homeostasis.
Formulation and Pharmacokinetics
EHP-101 is formulated as a combination of corn oil and long-chain mono-, di, and triglycerides with the API known as VCE-004.8.
To date, the pharmacokinetic (PK) profile of EHP-101 has been studied in a number of mice, rat and dog studies. In addition, PK has also now been evaluated in the Phase 1 human study on EHP-101. PK studies evaluate the absorption, distribution, metabolism, and excretion (ADME) from the body and measures, among other things, the concentration of the substance in plasma.
Toxicology
To date, we have completed extensive preclinical animal toxicology studies on EHP-101 that supported a human clinical Phase 1 study, which was completed in September 2019. Additional studies have been completed or are ongoing to support Phase 2 studies worldwide. In addition, using a CB1 ligand agonist assay, we have found that EHP-101 has no CB1 activity resulting in no psychotropic effects.
Manufacturing and Supply for EHP-101
A cGMP process has been developed to manufacture EHP-101 API and drug product through our contract manufacturers. Our contract manufacturers (CMOs) of the API have produced several multi-kilogram scale bulk batches for use in our preclinical and clinical studies. We have transferred the manufacturing of the API (VCE-004.8) to a CMO with large scale and commercial capabilities. EHP-101 API is a synthetic molecule, produced from synthetic CBD, and we believe there are readily available supplies of all raw materials needed for the manufacture of EHP-101. We do not own or operate manufacturing facilities for the production of EHP-101. We expect to depend on third-party suppliers and manufacturing organizations for all of our clinical trial quantities of raw materials, drug substance, and finished product.
Our Completed Clinical Trials
Our first-in-human Phase 1 study was conducted between September 2018 and September 2019, studying 104 subjects at 12 different dose levels. A total of 48 subjects (6 subjects per cohort) received a single dose of either 0.91 mg, 3 mg, 9 mg, 20 mg, 25 mg (fasted and fed), 50 mg, 100 mg or 185 mg of EHP-101. Another 32 subjects (8 subjects per cohort) received daily repeated doses for 7 days of either 20 mg once per day (QD), 25 mg twice per day (BID), 51.9 mg QD, or 50 mg BID of EHP-101. Twenty-four (24) subjects received placebo (2 subjects at each tested dose level in the total of 12 cohorts studied).
The most common side effects reported in some subjects included mild-to-moderate headache, paresthesia (a feeling of pins and needles in the limbs), mild blurred vision as well as mild abdominal pain at the highest dose only. Similar side effects were observed with placebo treatments and no maximum tolerated dose was reached. There were no clinically significant abnormalities in vital signs, physical examination, echocardiograms, clinical laboratory parameters, or ophthalmological assessments.
36
The half-life of a 25 mg single dose was approximately 2 hours during fasted state and 7 hours during fed state. A mean increase of 1.5-fold in the maximum plasma concentration (Cmax) and area under the curve (AUC, which represents the cumulative plasma concentration over time) was observed post administration with food. No drug accumulation in blood was observed with QD dosing and minimal accumulation was observed with BID dosing for 7 days.
The Cmax for a predicted anticipated therapeutic dose (ATD) was reached with a 20 mg single dose and the targeted drug exposure based on AUC was approached with a 50 mg single dose and 25 mg BID multiple dosing for 7 days, thus helping the selection of the dosing and treatment regimen for Phase 2 studies.
Preliminary drug-related biomarker analysis in plasma on Day 7 supports the mechanism of action of EHP-101 related to the effects on the HIF pathway, PPARγ and CB2 receptors. Some proteins were upregulated (increased concentrations post-dosing) related to vascular endothelial cell function (HIF pathway activation), lipid metabolism and control of inflammation whereas other proteins related to CB2and PPARγ activation (inflammation and immunomodulation) were downregulated (decreased concentrations post-dosing). Further quantitative analyses by other techniques are ongoing to assess multiple specific proteins.
Our Planned Clinical Trials
We have begun a Phase 2a clinical study of EHP-101 in SSc patients in Australia, New Zealand and the United States. The GLP preclinical studies required to advance into Phase 2 human studies in MS, which are more than 3-month treatment duration, and into longer SSc human studies, have been completed. We designed the completed Phase 1 study so that this single study allows us to proceed into Phase 2 for both MS and SSc. Prior to initiating Phase 1, we sought advice from the FDA on (1) our strategy of pursuing one Phase 1 study in support of subsequent Phase 2 studies for both the MS and SSc indications, (2) the design of the planned Phase 1 study, with the aim of designing the study so that the subsequent Phase 2 studies could be completed wholly or partially in the United States, and (3) the preliminary planned design of our Phase 2 clinical studies. Prior to initiating Phase 2a in SSc, we obtained regulatory approvals, including an Investigational New Drug (IND) clearance, Fast Track designation, and Orphan Drug Designation from the US FDA.
EHP-101 Indication 1: Multiple Sclerosis
MS is a chronic autoimmune disease of the central nervous system (CNS) that affects over 2.3 million patients worldwide, and despite being the most common human primary demyelinating disease of the CNS, there is presently no cure for MS. Myelin provides insulation for nerve fibers and is essential to maintain nerve conduction velocity. The hallmarks of MS include neuroinflammation, the loss of myelin (i.e., demyelination) and nerve cell damage. Disease progression is thought to be composed of two underlying processes: myelin destruction, or demyelination, with failure to remyelinate and progressive nerve cell damage with little capacity for recovery. Exacerbated innate and adaptive immune responses contribute to the pathophysiology of the disease and the majority of current therapies for MS are directed towards modulation of the immune response. However, therapies aimed to remyelinate nerve cells are needed.
Cannabinoids such as CBD that do not bind and activate CB1 and therefore do not produce psychotropic effects are considered of special interest as therapeutic agents in CNS diseases. In the CNS, there is evidence that CB2 receptors regulate neurotoxicity in certain cells of the CNS, called microglia. Cannabinoids also bind and activate the nuclear receptor superfamily of PPARs. Three forms of PPARs have been identified (PPARγ, PPARα and PPARβ/δ) and within these receptors, PPARγ can be activated weakly by cannabinoids such as CBD. Due, in part, to their PPARγ-activating properties, we believe these cannabinoids may exert anti-inflammatory activities, thus showing a therapeutic potential for the treatment of inflammatory diseases. PPARγ has been detected in certain nerve cells and participates in mechanisms that control activation of inflammatory response including modulation of cytokines and chemokine expression, neuronal dysfunction, and neurodegeneration. Neuroinflammation is an integral component to disorders such as Alzheimer’s disease, PD, stroke and MS. PPARγ activators have been shown to reduce the incidence and severity of disease in experimental models of MS, such as experimental autoimmune encephalomyelitis (EAE), and a small clinical trial suggested that PPARγ could be a pharmacological target for the management of MS.
37
EHP-101 has been shown to bind to CB2and PPARγ receptors, providing anti-inflammatory activity in the CNS as well as other positive biologic effects. Microglial cells specifically express CB2receptors in EAE tests, and activated microglial cells expressed 10-fold more CB2receptor than microglia in the resting state, suggesting that this receptor plays an important role in microglial cell function in the CNS during autoimmune-induced inflammation. In addition, PPARγ is expressed in different CNS cell types and its activation by ligand agonists provides immunomodulatory activity and neuroprotection and promotes oligodendrocyte progenitor cell differentiation and enhances their antioxidant defenses.
HIF operates in all mammalian cell types and responds to changes in oxygen, providing cells with a master regulator that coordinates changes in gene transcription. Hypoxia preconditioning induced by mild hypoxia can be beneficial in a wide number of disorders including neurologic and inflammatory diseases. Our preliminary studies indicate that EHP-101 stabilizes HIF-1α and activates the HIF pathway in different CNS cells as demonstrated by in vivo protein expression and functional assays. The results of these assays show that EHP-101 up-regulates the expression of neuroprotective genes such as erythropoietin and VEGF-A.
In vivo experiments in two mouse models of MS, EAE and Theiler virus-induced encephalopathy (TMEV) using VCE-004.8, the API in EHP-101, have resulted in the prevention of demyelination, nerve cell damage and immune cell infiltration. VCE-004.8 also down-regulated the expression of several genes including chemokines, cytokines and adhesion molecules, which are closely associated with MS pathophysiology. In addition, EHP-101, the oral formulation of VCE-004.8, resulted in the reduction of MS symptoms in both mouse models.
EHP-101 has also been shown to induce statistically significant remyelination of the brain in two different cuprizone (CPZ) mouse models compared to placebo. To evaluate the effect of EHP-101 on myelin damage in a cuprizone-induced demyelination murine model, brain coronal sections from animals after 6 weeks of CPZ 0.2% diet and 2 weeks of EHP-101 treatment were evaluated. In this model, EHP-101 treatment began after CPZ diet removal to directly evaluate the product effects on remyelination. Spontaneous recovery from demyelination was insignificant after 1 and 2 weeks but remyelination was significantly accelerated by EHP-101 treatment. Interestingly, EHP-101 enhanced remyelination in both white and gray matter of the brain, the corpus callosum and the cortex.
Furthermore, the potential of oral administration of EHP-101 to promote remyelination was evaluated in a cuprizone/rapamycin (C/R) mouse model (“augmented cuprizone model”), a more aggressive demyelination model since the concomitant administration of rapamycin with cuprizone for 12 weeks reduces spontaneous myelin production by blocking differentiation of oligodendrocyte progenitor cells. In the study, oral administration of EHP-101 once daily at 5, 10, and 20 mg/kg for 6 weeks induced statistically significant, dose-dependent remyelination of demyelinated axons in the white matter (corpus callosum) at 10 mg/kg (p < 0.005) and 20 mg/kg (p < 0.001) versus vehicle-treated controls as shown by the enhanced density of myelinated axons.
Summary of Mechanisms of EHP-101 in MS
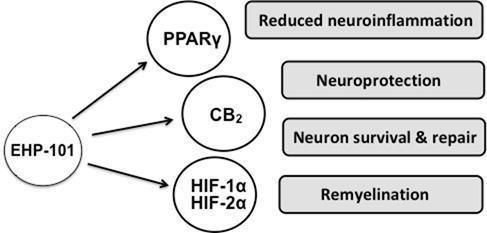 | EHP-101 is a multifunctional drug acting at different molecular targets involved in the pathophysiology of MS. EHP-101 has potent anti-inflammatory and neuroprotective activity through effects on PPARγ and CB2. In addition, EHP-101 activates the HIF pathway and mediates the expression of growth factors that play a role in CNS activity, homeostasis, and remyelination. |
In summary, we believe that EHP-101 is a promising product candidate for MS treatment, by ameliorating neuroinflammation through PPARγ/CB2receptors and by inducting neuroprotection and remyelination through activation of the HIF pathway. These activities are summarized in the diagram above.
38
EHP-101: Systemic Sclerosis (SSc)
Our other indication for EHP-101 is SSc, a rare, heterogeneous severe and life-threatening form of scleroderma that involves three main hallmarks: fibroblast dysfunction leading to increased deposition of extracellular matrix proteins, small vessel damage resulting in tissue hypoxia and an immune response with autoantibody production. SSc is characterized by progressive thickening and fibrosis of the skin secondary to excessive collagen accumulation, that can be limited to skin areas below, but not above, the elbows and knees, with or without involvement of the face (limited cutaneous SSc) or wider skin areas (skin on the arms, above and below the elbows, frequently on the legs, above and below the knees, with or without involvement of the face as well as on the torso) (diffuse cutaneous SSc, or dcSSc), both with internal organ involvement (e.g., lung, kidney, heart, stomach, bowels).
SSc is initiated by microvascular injury and inflammation followed by fibroblast activation, a key event in fibrosis development. Activated fibroblasts are responsible for the excessive collagen synthesis and transforming growth factor beta (TGFβ) production. TGFβ signaling plays a critical role in the regulation of cell growth, differentiation, and development in a wide range of biological systems. Excessive TGFβ signaling is the hallmark of SSc and different strategies aimed to disrupt this signaling pathway have been proposed for the treatment of SSc and related fibrotic diseases. Different studies prove that PPARγ and CB2 receptors are potential therapeutic targets for the disease because of their involvement in the inhibition of inflammation and fibrosis progression.
Recent evidence indicates that genetic and pharmacological manipulation of the endocannabinoid system modulates the fibrotic response. Thus, CB1and CB2receptors have shown different patterns in experimental models of dermal fibrosis. While CB1 activation is detrimental for the disease, CB2 activation has shown protection in mice from experimental dermal fibrosis. As stated previously, EHP-101 has been shown to effect CB2 but not CB1 receptors.
SSc is a rare disease with no cure, with a combined prevalence of more than 200,000 patients in the US, Europe, Australia and Japan. We have been granted orphan drug designation (ODD) by the FDA in the US and the EMA in the EU. SSc is a condition that is both chronically debilitating and life-threatening. The debilitating nature of the disease manifests itself through the development of painful lesions, disfigurement and the loss of ability to function normally. Currently there are no treatments for SSc that could stop the pathological fibrotic transformation in tissues of patients with SSc. Despite recent progress in the understanding of SSc pathophysiology, the current therapeutic recommendations focus on the management of organ specific morbidity and no single therapeutic agent has been proven to be efficacious as a universal disease-modifying agent that provides benefit to SSc patients regardless of which organs are affected by the disease.
SSc is life-threatening because of its effects on the vital organs: heart, lungs, kidneys and the digestive tract. The life-threatening acute onset of severe hypertension and renal failure occurs in approximately 66% of patients with SSc within the first 12 months of diagnosis. Up to 80% of SSc patients develop interstitial lung disease. Despite advances in early recognition of the disease and availability of efficient treatments for some of its organ complications, the mortality of SSc continues to be high. Although the median survival rate is 21.9 years, it varies significantly among different population groups. Additionally, the side effects of immunosuppressive treatments, which SSc patients typically receive as a standard of care, often result in life-threatening complications requiring intensive care unit admissions. Furthermore, the sites of SSc activity, such as skin, lung and liver, are susceptible to malignant transformation. The incidences of skin, lung and liver cancers are 3 to 4-fold higher in SSc patients compared to general population.
As described previously, EHP-101 behaves as a dual activator of PPARγ and CB2 receptors and, therefore, inhibits collagen synthesis. Moreover, EHP-101 has been shown to inhibit the TGFβ-associated differentiation of cells (called myofibroblasts) that are responsible for fibrosis.
The anti-fibrotic efficacy in vivo was investigated in a mouse model of scleroderma induced by bleomycin (BLM) in 64 mice (8 total groups with 8 mice in each group, using doses of 10 mg/kg and 20 mg/kg). EHP-101 reduced dermal thickness, blood vessel collagen accumulation and prevented other negative cell activities in the skin. In addition, it prevented the expression of several key genes associated with fibrosis, qualifying this synthetic cannabinoid NCE as a promising compound for the management of systemic sclerosis/scleroderma and, potentially, other fibrotic diseases.
39
As expected, histological examination of the skin after BLM administration resulted in increased dermal thickness and loss of the subcutaneous fat layer. Oral treatment with EHP-101 demonstrated a positive effect by reducing the progression of dermal thickness, skin fibrosis, and perivascular collagen deposition. The fact that pre-treatment with the CB2antagonist AM630 or the PPARγ antagonist T007907 partially negated the effect of EHP-101 indicates that the anti-fibrotic response was dependent on the dual PPARγ and CB2activation.
HIF activators have not been investigated extensively in fibrotic diseases such as SSc, however, based on our preclinical studies, we believe that the potential of this class of compounds to induce vascular protection is expected from other experimental models.
Summary of Mechanisms of EHP-101 in SSc
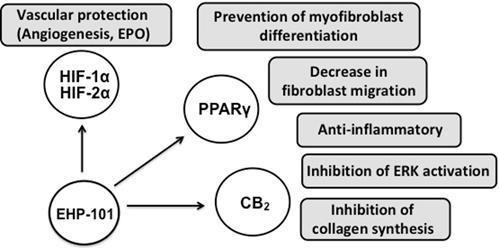 | EHP-101 is a multifunctional drug acting at different molecular targets that are the hallmark of SSc. EHP-101 has potent anti-inflammatory and anti-fibrotic activities by targeting PPARγ and CB2. EHP-101 also inhibits fibroblast to myofibroblast differentiation and collagen synthesis. In addition, EHP-101 activates the HIF pathway and mediates the expression of growth factors that can help vascular remodeling that is impaired in the disease. |
EHP-102 (VCE-003.2)
Overview
Our second product candidate, EHP-102, is an oral formulation of VCE-003.2, an NCE derived from CBG that affects some of the accepted biologic receptors and pathways involved in various neurodegenerative diseases. Based on preclinical studies to date, we are developing EHP-102 initially for two indications, HD and PD.
VCE-003.2 is a CBG aminoquinone derivative acting as a ligand activator of PPARγ measured by binding and transcriptional assays. Preclinical studies have shown that VCE-003.2 is neuroprotective and anti-inflammatory in animal models of PD and HD, as measured by proinflammatory cytokines and behavioral score, respectively. In addition, VCE-003.2 also reduced mutant huntingtin (different spelling than the disease) protein aggregates (altered huntingtin protein is associated with HD) detected by confocal microscopy techniques.
In addition to PPARγ, EHP-102 has been shown to activate a transcription factor involved in nerve cell differentiation (the chicken ovalbumin upstream promoter transcription factor-interacting protein, Ctip2). EHP-102 also reduces the expression of cyclooxygenase-2 (COX-2) in nerve cells (involved in inflammation and pain). Furthermore, EHP-102 is an activator of the extracellular signal-regulated kinases (ERK) pathway (a member of the mitogen-activated protein kinases [MAPK] pathway) in hippocampal neuronal progenitor cells. This pathway influences nerve cell survival. In summary, EHP-102 exerts potent anti-inflammatory activity in the CNS and enhances neurogenesis (the regeneration of nerve tissue) and can, therefore, provide benefits in neurodegenerative diseases such as HD and PD.
40
Formulation and Pharmacokinetics
We are currently in the manufacturing and formulation development stage for EHP-102. Upon finalization of manufacturing process development and formulation prototype selection, we plan to initiate the PK and nonclinical studies required for human clinical development.
Safety, Toxicology and Clinical Trials
We have begun the basic non-clinical studies required to advance to human studies. Once the manufacturing and formulation development activities are completed for EHP-102, we plan to initiate an animal safety and toxicology program in support of initiating human clinical development.
Manufacturing and Supply for EHP-102
An industrial scalable process is being developed to manufacture the EHP-102 API (VCE-003.2) and drug product through our contract manufacturers. We do not own or operate manufacturing facilities for the production of EHP-102. We expect to depend on third-party suppliers and manufacturing organizations for all of our preclinical and clinical trial quantities of raw materials, drug substance and drug product.
Our Planned Clinical Trials
Since Phase 1 human studies are not expected to begin within the next year, we have not yet begun to develop the full clinical development plan for EHP-102.
EHP-102 Indication 1: Huntington’s Disease
HD is an autosomal dominant genetic disorder causing the mutation of a protein called the huntingtin protein which causes progressive degeneration of nerve cells in the brain, specifically, cells of the basal ganglia. This devastating and disabling disease affects middle-aged people with typical onset between the ages of 30 and 50. The genetic defect that causes HD is an abnormal repeat of certain DNA sequences on chromosome number 4. With each successive generation, the number of repeats increases. There is a 50% chance that the disease will be passed to offspring.
HD is a rare disease with approximately 30,000 patients annually in the United States. We have been granted ODD by the FDA in the United States and by the EMA in Europe.
One of the hallmark signs and symptoms of this disease is involuntary movements and tics. These involuntary movements and tics begin distally and move proximally as the disease progresses. The involuntary movements and tics are not specific and may involve the hands, feet, and face, with the most prominent movements taking place in the muscles of the back. The involuntary movements lead to the appearance that a patient is inebriated. Swallowing becomes difficult, and patients are at risk for choking. Reduced movement and rigidity are common. Patients may also suffer from flexion contractures and become bed bound. Finally, the patient is completely dependent on others for care. The risk of choking increases and the involuntary movements may become more severe or completely disappear. At this point the patients can no longer walk. Cognitive symptoms may include feelings of low self-esteem, guilt, anxiety, apathy, irritability, aggression, dementia and psychosis with paranoia and auditory hallucinations. The cause of death is usually from secondary causes of the disease such as choking and infection.
There is no known curative treatment for HD. Treatment is mostly directed at symptomatic relief with suppression of the involuntary movements and tics. Dopamine-depleting agents, dopamine activators, benzodiazepines, glutamate antagonists, acetylcholinesterase inhibitors, dopamine antagonists, anti-seizure medications, cannabinoids, lithium, along with deep brain stimulation and fetal cell transplantation are being used to treat HD.
41
The molecular mechanisms of HD pathophysiology are unclear. The current model of disease progression includes development of mitochondrial dysfunction in the huntingtin protein. PPARγ is believed to play a key role in neurodegenerative diseases as it regulates neural progenitor cell proliferation and differentiation. Studies have demonstrated that there are significant defects in the PPARγ signaling pathway in mutant huntingtin expressing cells as compared to wild-type huntingtin protein cells. PPARγ activators improve mitochondrial function in cells expressing mutant huntingtin. The activation of the PPARγ signaling pathway can help mitochondrial function, a pivotal process in the pathogenesis of HD. Therefore, the PPARγ pathway could be a rational therapeutic target in the treatment of HD.
Preclinical in vitro studies have shown that VCE-003.2 preserves the ability to activate PPARγ and exerts a prosurvival action in progenitor cells during neuronal differentiation. In addition to EHP-102’s effect on PPARγ, our studies indicate effects on other receptor targets involved in neurodegeneration. For example, EHP-102 reduced mutant huntingtin aggregates in striatal cells as noted above. The neuroprotective profile of EHP-102 was also analyzed using three in vivo models of striatal neurodegeneration, which mimic HD in humans. EHP-102 inhibited the up-regulation of proinflammatory markers and improved antioxidant defenses in the brain of the test animals.
To assess the pathophysiological relevance of the neuroprotective action of EHP-102 in vivo, we employed three mouse models of the disease.
In summary, these studies suggest that EHP-102 displays neuroprotective and anti-inflammatory activities in different mouse models of HD. For example, in these models EHP-102 has:
| ● | prevented neural damage and neuroinflammation; | |
| ● | alleviated motor symptomatology; | |
| ● | improved motor symptomatology in mice expressing a mutated form of huntingtin protein in the brain; | |
| ● | inhibited the up-regulation of proinflammatory markers such as COX-2, TNF-α, and IL-6; | |
| ● | improved oxidative stress markers; and | |
| ● | promoted neurogenesis in the striatum. |
These data suggest that EHP-102 could have potential for the treatment of HD and other neurodegenerative diseases with neuroinflammatory traits.
EHP-102 Indication 2: Parkinson’s Disease
PD is a long-term degenerative disorder of the central nervous system that mainly affects the motor system. It is a disease where damaged neurons do not produce sufficient dopamine (dopamine helps transmit impulses from the brain to the muscles). Over 10 million people suffer from PD worldwide. The symptoms generally present slowly over time. Early in the disease, the most obvious symptoms are shaking, rigidity, slowness of movement, and difficulty with walking. Thinking and behavioral problems may also occur. Dementia becomes common in the advanced stages of the disease. Depression and anxiety are also common, occurring in more than a third of people with PD. Other symptoms may include sensory, sleep, and emotional problems. The main motor symptoms are collectively called “Parkinsonism,” or a “Parkinsonian syndrome.”
Inflammation is a key pathogenic event in PD, so anti-inflammatory strategies are being investigated to limit neuronal deterioration in this disease. Certain cannabinoids have been shown to have anti-inflammatory and neuroprotective properties. In addition, epidemiological data support that the regular use of non-steroidal anti-inflammatory drugs (e.g., ibuprofen) reduces the risk of developing PD. In light of this, different anti-inflammatory agents have been investigated, at preclinical and clinical levels, with variable success in affecting the symptoms of PD.
42
Cannabinoids have been investigated for the reduction of inflammatory events in PD due mainly to selective CB2 receptor activation. Activating these receptors elicited frequent positive responses, predominantly by recruiting microglial cells and infiltrated macrophages to the CNS areas lesioned in PD, as well as on the anti-inflammatory and neuroprotective effects derived from the selective activation of these receptors. In addition, another possible rationale for the therapeutic potential in PD may be the possibility of targeting PPARγ receptors with certain cannabinoids. Such conclusion is based on: (i) the relevant role played by these nuclear receptors in the control of inflammation in numerous pathological conditions (ii) the well-described PPARγ-mediated anti-inflammatory activity of certain cannabinoids in different models of central and peripheral inflammation, and (iii) the effects of non-cannabinoid PPARγ activators (e.g., thiazolidinediones) in PD. The three different PPAR isotypes (α, β/δ and γ) are expressed in all cell types in the brain and all of them, by functioning in an integrated manner as a complex system – the so-called PPAR triad, have been reported to have neuroprotective properties.
Given the activity of EHP-102 on the PPARγ receptor, which is an important factor involved in the control of inflammation, we evaluated its anti-inflammatory/neuroprotective properties in a typical in vivo inflammatory model of PD, lipopolysaccharide (LPS)-lesioned mice. Positive results were obtained in all measurements assessed, both qualitative and quantitative. Unlike EHP-101, EHP-102 has no activity on CB2, however, in this study proinflammatory mediators such as tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), and inducible nitric oxide synthase (iNOS) were strongly reduced by the treatment with EHP-102. Thus, in this model and others, therefore, EHP-102 reduced inflammatory marker expression and prevented dopaminergic neuronal loss (i.e., the loss of nerve cells that produce dopamine, which is the main pathologic feature of PD). It also improved clinical symptoms and recovered movement parameters (motor coordination and activity) in mice injected with 3-NP and treated with our drug candidate. These data lead us to believe EHP-102 could be a potential treatment for PD.
Intellectual Property
We believe it is important to our success that we:
| ● | obtain and maintain patent and other legal protections for the proprietary molecules, technology, inventions and improvements we consider important to our business; | |
| ● | prosecute our patent applications and defend our issued patents; | |
| ● | preserve the confidentiality of our trade secrets; and | |
| ● | operate without infringing the patents and proprietary rights of third parties. |
We have sought and intend to continue to seek appropriate patent protection for our product candidates, as well as other proprietary technologies and their uses by filing additional patent applications in the United States and selected other countries.
As of the date of this Offering Circular, we owned a total of 19 granted patents: three United States patents, three Japanese patents, two European patents, two Mexican patents, two patents in the Russian Federation, two Australian patents, two Israeli patents, two Indian patents and one patent in China. In addition, we have 20 pending patent applications. These patents and patent applications will expire between 2030 through 2041 and could be eligible for patent term extension for delay caused by regulatory review, thereby further extending their patent terms. Our patent portfolio includes use in many different indications, which we believe could provide us with patent protection for our developed products for additional patient populations in markets with unmet medical need.
43
Our patent plan is focused on providing patent protection for our NCEs derived from cannabinoids, their formulation and therapeutic applications. The following is a summary of our seven patent families:
| Family Number | Patent Publication/Application Number | Status | Expiry | Title | Description | |||||
| 3.1 | US8772349 EP2551255B1* JP05575324B2 WO 2011/117429 *Validated in DE, ES, GB, FR, IT, & NL | Granted Granted Granted Expired | 2030 | Cannabinoid Quinone Derivatives | Cannabinoid quinone derivatives to be used as medicaments, particularly as PPARγ activators for treating diseases which etiology is based on an impaired PPARγ function and can benefit from PPARγ activation | |||||
| 3.2 | US9802880 AU2015222384 CA2937275A1 CN106061937A EP2913321A1 JP6619349 KR2016126006 MX2016010952 WO2015128200A1 BRPI1619891A2 IN201647028497A RU2684913 IL247149 HK17103324.2 | Granted Granted Pending Pending Pending Granted Pending Granted Expired Pending Granted Granted Granted Pending | 2035 | Cannabigerol Derivatives | CBG derivatives to be used as medicaments in therapy particularly for treating PPARγ-related diseases due to their high PPARγ activatory effect | |||||
3.3
| WO PCT/EP2019/084764 | Pending | 2038 | Cannabigerol Quinone Acid and Salts | CBG quinone acid and its salts, and new methods of synthesis | |||||
| 4.1 | US9701618 AU2014390738 CA2945867A1 CN106232570A EP3131874A1* JP06167248B2 KR2016146765A IN201617038938A BRPI1623902A2 MX2016013151A WO2015158381A1 RU2667504 IL248030 HK17104665.7 *Validated in BE, CH, DE, DK, ES, FR, GB, IE, IT, LI, & NL | Granted Granted Pending Granted Granted Granted Pending Granted Pending Granted Expired Granted Granted Pending | 2034 | Cannabidiol Quinone Derivatives | CBD quinone derivatives to be used as medicaments in therapy, particularly or treating diseases and conditions responsive to PPARγ modulation due to their high PPARγ activatory effect | |||||
| 4.2 | WO2018177516 AU2017406103 CA3058352 EP3600274A1 IL269623 JP2019553369 US16498768 | Expired Pending Pending Pending Pending Pending Allowed | 2037 | Cannabidiol Derivatives as inhibitors of the HIF prolyl hydroxylases activity | CBD quinone derivatives to be used as medicaments in therapy, particularly for treating diseases and conditions responsive to HIF-1 activation | |||||
| 4.3 | PCT/US2020/017035 | Pending
| 2040 | Formulations of Cannabidiol Derivatives | Formulations of CBD derivatives to be used as Modulators of Cannabinoid Receptor Type 2 (CB2) | |||||
| 4.4 | US63/020584 US63/020584 | Pending Pending | 2041 2041 | Compositions of Cannabidiol Derivatives and their use as modulators of Cannabinoid Receptor Type 2 (CB2) Cannabidiol Quinone Derivatives | Composition and Method for the treatment and preventions of cardiac fibrosis Composition and Method for the Treatment and Prevention of Cardiac, Pulmonary, Dermal, and Renal Fibrosis |
44
Government Regulation and Product Approval
Government regulation and product approval is required in order for a new drug to enter the market. We are currently in the clinical development stage (Phase 2) for EHP-101 and in the preclinical development stage for EHP-102. Since we plan to conduct our Phase 2 clinical studies in Australia, New Zealand and the US, approvals to conduct clinical studies in those countries are required from the Health Research Ethics Committee (HREC) and TGA in Australia, the Director General of Health and an Ethics Committee in New Zealand, and the FDA (IND clearance) and a central IRB (WIRB-Copernicus) in the US. To date, all these approvals have been granted, except for the Ethics Committee in New Zealand, which is pending.
FDA Approval Process
In the US, pharmaceutical products are subject to extensive regulation by the FDA. The Federal Food, Drug, and Cosmetic Act, or the FDC Act, and other federal and state statutes and regulations, govern, among other things, the research, development, testing, manufacture, storage, recordkeeping, approval, labeling, promotion and marketing, distribution, post-approval monitoring and reporting, sampling, and import and export of pharmaceutical products. Failure to comply with applicable US requirements may subject a company to a variety of administrative or judicial sanctions, such as imposition of clinical holds, FDA refusal to approve pending NDAs, warning letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, refusals of government contracts, restitution, disgorgement, civil penalties and criminal prosecution.
Pharmaceutical product development in the US typically involves pre-clinical laboratory and animal tests and the submission to the FDA of an IND, which must become effective before clinical testing may commence. For commercial approval, the sponsor must submit adequate tests by all methods reasonably applicable to show that the drug is safe for use under the conditions prescribed, recommended or suggested in the proposed labeling. The sponsor must also submit substantial evidence, generally consisting of adequate, well-controlled clinical trials to establish that the drug will have the effect it purports or is represented to have under the conditions of use prescribed, recommended or suggested in the proposed labeling. In certain cases, the FDA may determine that a drug is effective based on one clinical study plus confirmatory evidence. Satisfaction of FDA pre-market approval requirements typically takes many years and the actual time required may vary substantially based upon the type, complexity and novelty of the product or disease.
Pre-clinical tests include laboratory evaluation of product chemistry, formulation and toxicity, as well as animal trials to assess the characteristics and potential safety and efficacy of the product. The conduct of the pre-clinical tests must comply with federal regulations and requirements, including the FDA’s good laboratory practices regulations and the US Department of Agriculture’s (USDA’s) regulations implementing the Animal Welfare Act. The results of pre-clinical testing are submitted to the FDA as part of an IND along with other information, including information about product chemistry, manufacturing and controls, and a proposed clinical trial protocol. Long-term pre-clinical tests, such as animal tests of reproductive toxicity and carcinogenicity, may continue after the IND is submitted.
A 30-day waiting period after the submission of each IND is required prior to the commencement of clinical testing in humans. If the FDA has not imposed a clinical hold on the IND or otherwise commented or questioned the IND within this 30-day period, the clinical trial proposed in the IND may begin.
Clinical trials involve the administration of the investigational new drug to healthy volunteers or patients under the supervision of a qualified investigator. Clinical trials must be conducted: (i) in compliance with federal regulations, (ii) in compliance with Good Clinical Practice (GCP), an international standard meant to protect the rights and health of patients and to define the roles of clinical trial sponsors, administrators and monitors, and (iii) under protocols detailing the objectives of the trial, the parameters to be used in monitoring safety and the effectiveness criteria to be evaluated. Each protocol involving testing on US patients and subsequent protocol amendments must be submitted to the FDA as part of the IND.
45
The FDA may order the temporary, or permanent, discontinuation of a clinical trial at any time or impose other sanctions if it believes that the clinical trial either is not being conducted in accordance with FDA requirements or presents an unacceptable risk to the clinical trial patients. The trial protocol and informed consent information for patients in clinical trials must also be submitted to an institutional review board, or IRB, for approval. An IRB may also require the clinical trial at the site to be halted, either temporarily or permanently, for failure to comply with the IRB’s requirements or may impose other conditions.
Clinical trials to support NDAs for marketing approval are typically conducted in three sequential phases, but the phases may overlap. In general in Phase 1, the initial introduction of the drug into healthy human subjects or patients, the drug is tested to assess metabolism, pharmacokinetics, pharmacological actions, side effects associated with increasing doses and, if possible, early evidence on effectiveness. Phase 2 usually involves trials in a limited patient population to determine the effectiveness of the drug for a particular indication, dosage tolerance and optimum dosage, and to identify common adverse effects and safety risks. If a compound demonstrates evidence of effectiveness and an acceptable safety profile in Phase 2 evaluations, Phase 3 trials are undertaken to obtain the additional information about clinical efficacy and safety in a larger number of patients, typically at geographically dispersed clinical trial sites, to permit the FDA to evaluate the overall benefit-risk relationship of the drug and to provide adequate information for the labeling of the drug. In most cases, the FDA requires two adequate and well-controlled Phase 3 clinical trials to demonstrate the efficacy of the drug. The FDA may, however, determine that a drug is effective based on one clinical study plus confirmatory evidence. Only a small percentage of investigational drugs complete all three phases and obtain marketing approval. In some cases, the FDA may require post-market studies, known as Phase 4 studies, to be conducted as a condition of approval in order to gather additional information on the drug’s effect in various populations and any side effects associated with long-term use. Depending on the risks posed by the drugs, other post-market requirements may be imposed.
After completion of the required clinical testing, an NDA is prepared and submitted to the FDA. The FDA approval of the NDA is required before marketing of the product may begin in the US The NDA must include the results of all pre-clinical, clinical, and other testing and a compilation of data relating to the product’s pharmacology, chemistry, manufacture, and controls. The cost of preparing and submitting an NDA is substantial.
The FDA has 60 days from its receipt of an NDA to determine whether the application will be accepted for filing based on the agency’s threshold determination that it is sufficiently complete to permit substantive review. Once the submission is accepted for filing, the FDA begins an in-depth review. Under the statute and implementing regulations, the FDA has 180 days (the initial review cycle) from the date of filing to issue either an approval letter or a complete response letter, unless the review period is adjusted by mutual agreement between the FDA and the applicant or as a result of the applicant submitting a major amendment. In practice, the performance goals established pursuant to the Prescription Drug User Fee Act have effectively extended the initial review cycle beyond 180 days. The FDA’s current performance goals call for the FDA to complete review of 90 percent of standard (non-priority) NDAs within 10 months of receipt and within six months for priority NDAs, but two additional months are added to standard and priority NDAs for a new molecular entity (NME).
The FDA may also refer applications for novel drug products, or drug products that present difficult questions of safety or efficacy, to an advisory committee, which is typically a panel that includes clinicians and other experts, for review, evaluation and a recommendation as to whether the application should be approved. The FDA is not bound by the recommendation of an advisory committee, but it generally follows such recommendations. Before approving an NDA, the FDA will typically inspect one or more clinical sites to assure compliance with GCP. Additionally, the FDA will inspect the facility or the facilities at which the drug is manufactured. The FDA will not approve the product unless compliance with cGMP is satisfactory and the NDA contains data that provide substantial evidence that the drug is safe and effective in the indication studied.
After the FDA evaluates the NDA and the manufacturing facilities, it issues either an approval letter or a complete response letter. A complete response letter generally outlines the deficiencies in the submission and may require substantial additional testing, or information, in order for the FDA to reconsider the application. If, or when, those deficiencies have been addressed to the FDA’s satisfaction in a resubmission of the NDA, the FDA will issue an approval letter. The FDA has committed to reviewing 90 percent of resubmissions within two to six months depending on the type of information included.
46
An approval letter authorizes commercial marketing of the drug with specific prescribing information for specific indications. As a condition of NDA approval, the FDA may require a risk evaluation and mitigation strategy, or REMS, to help ensure that the benefits of the drug outweigh the potential risks. REMS can include medication guides, communication plans for health care professionals, and elements to assure safe use, or ETASU. ETASU can include, but are not limited to, special training or certification for prescribing or dispensing, dispensing only under certain circumstances, special monitoring, and the use of patient registries. The requirement for a REMS can materially affect the potential market and profitability of the drug. Moreover, product approval may require substantial post-approval testing and surveillance to monitor the drug’s safety or efficacy. Once granted, product approvals may be withdrawn if compliance with regulatory standards is not maintained or problems are identified following initial marketing.
Disclosure of Clinical Trial Information
Sponsors of clinical trials of certain FDA-regulated products, including prescription drugs, are required to register and disclose certain clinical trial information on a public website maintained by the US National Institutes of Health. Information related to the product, patient population, phase of investigation, study sites and investigator, and other aspects of the clinical trial is made public as part of the registration. Sponsors are also obligated to disclose the results of these trials after completion. Disclosure of the results of these trials can be delayed for up to two years if the sponsor certifies that it is seeking approval of an unapproved product or that it will file an application for approval of a new indication for an approved product within one year. Competitors may use this publicly available information to gain knowledge regarding the design and progress of our development programs.
Fast Track Designation, Accelerated Approval, and Breakthrough Therapy Designation
The FDA is required to facilitate the development, and expedite the review, of drugs that are intended for the treatment of a serious or life-threatening disease or condition for which there is no effective treatment and which demonstrate the potential to address unmet medical needs for the condition. Under the Fast Track Program, the sponsor of a new drug candidate may request that the FDA designate the drug candidate for a specific indication as a Fast Track drug concurrent with, or after, the filing of the IND for the drug candidate. The FDA must determine if the drug candidate qualifies for Fast Track designation within 60 days of receipt of the sponsor’s request.
Under the FDA’s Accelerated Approval regulations, the FDA may approve a drug for a serious or life-threatening illness that provides meaningful therapeutic benefit to patients over existing treatments based upon a surrogate endpoint that is reasonably likely to predict clinical benefit, or on a clinical endpoint that can be measured earlier than irreversible morbidity or mortality, that is reasonably likely to predict an effect on irreversible morbidity or mortality or other clinical benefit, taking into account the severity, rarity or prevalence of the condition and the availability or lack of alternative treatments.
In clinical trials, a surrogate endpoint is a measurement of laboratory or clinical signs of a disease or condition that substitutes for a direct measurement of how a patient feels, functions or survives. Surrogate endpoints can often be measured more easily or more rapidly than clinical endpoints. A drug candidate approved on this basis is subject to rigorous post-marketing compliance requirements, including the completion of Phase 4 or post-approval clinical trials to confirm the effect on the clinical endpoint. Failure to conduct required post-approval studies, or confirm a clinical benefit during post-marketing studies, will allow the FDA to withdraw the drug from the market on an expedited basis. Unless otherwise informed by the FDA, for an accelerated approval product an applicant must submit to the FDA for consideration during the preapproval review period copies of all promotional materials, including promotional labeling as well as advertisements, intended for dissemination or publication within 120 days following marketing approval. After 120 days following marketing approval, unless otherwise informed by the FDA, the applicant must submit promotional materials at least 30 days prior to the intended time of initial dissemination of the labeling or initial publication of the advertisement.
In addition to other benefits such as the ability to use surrogate endpoints and engage in more frequent interactions with the FDA, the FDA may initiate review of sections of a Fast Track drug’s NDA before the application is complete. This rolling review is available if the applicant provides, and the FDA approves, a schedule for the submission of the remaining information and the applicant pays applicable user fees. However, the FDA’s time period goal for reviewing an application does not begin until the last section of the NDA is submitted. Additionally, the Fast Track designation may be withdrawn by the FDA if the FDA believes that the designation is no longer supported by data emerging in the clinical trial process.
47
Finally, as part of FDA’s expedited programs to facilitate drug development and product approval for serious or life-threatening conditions, Breakthrough Therapy Designation (BTD) requires preliminary clinical evidence that demonstrates the drug may have substantial improvement on at least one clinically significant endpoint over available therapy. A BTD conveys all of the FTD features plus more intensive FDA guidance on an efficient drug development program, an organizational commitment involving senior FDA managers, and eligibility for rolling review and priority review (the NDA review clock will begin earlier).
Orphan Drugs
Under the Orphan Drug Act, the FDA may grant orphan drug designation to drugs intended to treat a rare disease or condition - generally a disease or condition that affects fewer than 200,000 individuals in the US (or affects more than 200,000 in the US and for which there is no reasonable expectation that the cost of developing and making available in the US a drug for such disease or condition will be recovered from sales in the US of such drug). Orphan drug designation must be requested before submitting an NDA. After the FDA grants orphan drug designation, the generic identity of the drug and its potential orphan use are disclosed publicly by the FDA. Orphan drug designation does not convey any advantage in, or shorten the duration of, the regulatory review and approval process. The first NDA applicant to receive FDA approval for a particular active ingredient to treat a particular disease with FDA orphan drug designation is entitled to a seven-year exclusive marketing period in the US for that product, for that indication. During the seven-year exclusivity period, the FDA may not approve any other applications to market the same drug for the same disease, except in limited circumstances, such as a showing of clinical superiority to the product with orphan drug exclusivity. If the FDA designates an orphan drug based on a finding of clinical superiority, the FDA must provide a written notification to the sponsor that states the basis for orphan designation, including “any plausible hypothesis” relied upon by the FDA. The FDA must also publish a summary of its clinical superiority findings upon granting orphan drug exclusivity based on clinical superiority. Orphan drug exclusivity does not prevent the FDA from approving a different drug for the same disease or condition, or the same drug for a different disease or condition. Among the other benefits of orphan drug designation are tax credits for certain research and a waiver of the NDA application user fee.
Controlled Substances Laws
The federal Controlled Substances Act of 1970, or CSA and its implementing regulations establish a “closed system” of distribution for controlled substances. The CSA imposes registration, security, recordkeeping and reporting, storage, manufacturing, distribution, labeling, importation, exportation, disposal and other requirements under the oversight of the DEA. The DEA is the federal agency responsible for regulating controlled substances, and requires those individuals or entities that manufacture, import, export, distribute, research, or dispense controlled substances to comply with the regulatory requirements to prevent the diversion of controlled substances to illicit channels of commerce.
The DEA categorizes controlled substances into one of five schedules — Schedule I, II, III, IV, or V — with varying qualifications for listing in each schedule. Schedule I substances by definition have a high potential for abuse, have no currently “accepted medical use” in treatment in the United States and lack accepted safety for use under medical supervision. They may be used only in federally approved research programs and may not be marketed or sold for dispensing to patients in the United States. Pharmaceutical products having a currently accepted medical use may be listed as Schedule II, III, IV or V substances, with Schedule II substances presenting the highest potential for abuse and physical or psychological dependence, and Schedule V substances presenting the lowest relative potential for abuse and dependence. The regulatory requirements are more restrictive for Schedule II substances than Schedule III-V substances. For example, all Schedule II drug prescriptions must be signed by a physician, physically presented to a pharmacist in most situations, and cannot be refilled. While cannabis and THC are Schedule I controlled substances, products approved for medical use in the United States that contain cannabis, THC or cannabis/THC extracts must be placed in Schedules II-V, since approval by the FDA satisfies the “acceptable medical use” requirement.
48
Facilities that research, manufacture, distribute, import or export any controlled substance must register annually with the DEA. The DEA registration is specific to the particular location, activity(ies) and controlled substances utilized. For example, separate registrations are required for importation and manufacturing activities, and each registration authorizes which schedules of controlled substances the registrant may handle. However, certain coincident activities are permitted without obtaining a separate DEA registration, such as distribution of controlled substances by the manufacturer that produces them.
The DEA inspects all manufacturing facilities to review security, record keeping, reporting and compliance with other DEA regulatory requirements prior to issuing a controlled substance registration. The specific security requirements vary by the type of business activity and the schedule and quantity of controlled substances handled. The most stringent requirements apply to manufacturers of Schedule I and Schedule II substances. Required security measures commonly include background checks on employees and physical control of controlled substances through storage in approved vaults, safes and cages, and through use of alarm systems and surveillance cameras. An application for a manufacturing registration as a bulk manufacturer (not a dosage form manufacturer or a repacker/relabeler) for a Schedule I or II substance must be published in the Federal Register, and is open for 30 days to permit interested persons to submit comments, objections, or requests for a hearing. A copy of the notice of the Federal Register publication is forwarded by the DEA to all those registered, or applicants for registration, as bulk manufacturers of that substance. Once registered, manufacturing facilities must maintain records documenting the manufacture, receipt and distribution of all controlled substances. Manufacturers must submit periodic reports to the DEA of the distribution of Schedule I and II controlled substances, Schedule III narcotic substances, and other designated substances. Registrants must also report any controlled substance thefts or significant losses and must adhere to certain requirements to dispose of controlled substances. As with applications for registration as a bulk manufacturer, an application for an importer registration for a Schedule I or II substance must also be published in the Federal Register, which remains open for 30 days for comments. Imports of Schedule I and II controlled substances for commercial purposes are generally restricted to substances not already available from a domestic supplier or where there is not adequate competition among domestic suppliers. In addition to an importer or exporter registration, importers and exporters must obtain a permit for every import or export of a Schedule I and II substance, Schedule III, IV and V narcotic, specially designated Schedule III non-narcotics, or Schedule IV or V narcotics controlled in Schedule I or II by the Convention on Psychotropic Substances and submit import or export declarations for Schedule III, IV and V non-narcotics. In some cases, Schedule III non-narcotic substances may be subject to the import/export permit requirement, if necessary, to ensure that the US complies with its obligations under international drug control treaties.
For drugs manufactured in the United States, the DEA establishes annually an aggregate quota for the amount of substances within Schedules I and II that may be manufactured or produced in the United States based on the DEA’s estimate of the quantity needed to meet legitimate medical, scientific, research and industrial needs. This limited aggregate amount of cannabis that the DEA allows to be produced in the United States each year is allocated among individual companies, which, in turn, must annually apply to the DEA for individual manufacturing and procurement quotas. The quotas apply equally to the manufacturing of the API and production of dosage forms. The DEA may adjust aggregate production quotas a few times per year, and individual manufacturing or procurement quotas from time to time during the year, although the DEA has substantial discretion in whether or not to make such adjustments for individual companies.
The states also maintain separate controlled substance laws and regulations, including licensing, recordkeeping, security, distribution, and dispensing requirements. State authorities, including Boards of Pharmacy, regulate use of controlled substances in each state. Failure to maintain compliance with applicable requirements, particularly as manifested in the loss or diversion of controlled substances, can result in enforcement action that could have a material adverse effect on our business, operations and financial condition. The DEA may seek civil penalties, refuse to renew necessary registrations, or initiate proceedings to revoke those registrations. In certain circumstances, violations could lead to criminal prosecution.
We currently manufacture the API and formulations, as well as perform most of the preclinical testing for EHP-101 and EHP-102 in Europe, China and Canada. We have completed a Phase 1 trial for EHP-101 in Australia and have begun study site initiation activities for a Phase 2a study in Australia, New Zealand and the US. We may decide to develop, manufacture or commercialize our product candidates in the United States or additional countries in the future. As a result, we may be subject to controlled substance laws and regulations from regulatory agencies in countries where we develop, manufacture or commercialize EHP-101 and EHP-102 in the future.
49
While the starting material for the active pharmaceutical ingredient (API) in our product candidates are CBD and CBG, which may be classified by the United States Drug Enforcement Administration (DEA) as controlled substances in the United States depending on their origin and purity, in March 2019 we received a decision from the DEA that the API (VCE-004.8) in our lead product candidate (EHP-101) is not a controlled substance, based partly on the fact that our molecule is chemically derived from synthetic CBD as an NCE containing no remaining CBD or other controlled substances. We have also received the same decision from the UK Home Office and Canada’s Controlled Substances Directorate. VCE-004.8 not being a controlled substance eliminates costs and complexities associated with developing controlled substances, by facilitating the manufacturing and import of the product to the US and simplifying the conduct of nonclinical studies and the selection of US clinical sites to conduct the clinical studies being planned in MS and SSc patients. Once we advance our second product candidate (EHP-102) further in development, we will request a similar decision from the DEA and other countries for this product candidate.
Employees
As the date of this Offering Circular, we employed fifteen (15) full-time employees, one (1) part-time employee and three (3) temporary part-time employees.
Legal Proceedings
We are not currently a party to any legal proceedings, the adverse outcome of which, individually or in the aggregate, we believe will have a material adverse effect on our business, financial condition or operating results.
The Company does not own any real estate. The Company currently leases office space at 5910 Pacific Center Boulevard in San Diego, California. We believe our leased office space is suitable and adequate for the Company’s current business operations.
50
MANAGEMENT’S DISCUSSION & ANALYSIS OF FINANCIAL CONDITION & RESULTS OF OPERATIONS
You should read the following discussion and analysis of our financial condition and results of our operations together with our financial statements and the notes thereto appearing elsewhere in this Offering Circular. This discussion contains forward-looking statements reflecting our current expectations, whose actual outcomes involve risks and uncertainties. Actual results and the timing of events may differ materially from those stated in or implied by these forward-looking statements due to a number of factors, including those discussed in the sections entitled “Risk Factors” starting on page 5, “Cautionary Statement Regarding Forward-Looking Statements” starting on page iii, and elsewhere in this Offering Circular. Please see the notes to our Financial Statements for information about our Significant Accounting Policies.
Results of Operations for the Years Ended December 31, 2019 and 2018 and the Six Months Ended June 30, 2020 and June 30, 2019
Revenues
The Company is a pre-revenue development stage biotechnology company focused on the development of product candidates to treat diseases with unmet medical needs. We have no products approved for commercial sale and have not generated any revenues from product sales since our inception in March 2017.
Research and Development Expenses
Research and development expenses consist primarily of expenses associated with preclinical development and clinical trials, payments to third-party contract research organizations, or CROs, contract manufacturing organizations, or CMOs, contractor laboratories and independent contractors, and personnel related expenses, such as salaries, benefits, travel and other related expenses, including stock-based compensation. To date, our research and development expenses have related primarily to the development of, and clinical trials for, our lead product candidate EHP-101.
Our research and development expenses were $9.9 million for the year ended December 31, 2019, compared to $5.7 million for the year ended December 31, 2018. This increase was primarily related to an increase in clinical expense of approximately $1.2 million (net of an increase in incentive and other tax receivables of $1.0 million) related to a Phase 1 clinical study that commenced in September 2018 and was completed in August 2019; and an increase in nonclinical development, manufacturing, and regulatory expenses of approximately $1.1 million compared to the prior year. In addition, personnel related expenses for the year ended December 31, 2019 increased by approximately $1.5 million in part due to an increase in headcount and increased stock-based compensation compared to the prior year. We also expensed approximately $0.4 million during the year ended December 31, 2019, for milestone payments due to Emerald Health Biotechnology España S.L.U. (EHBE) related to the completion of the Phase 1 clinical study.
Our research and development expenses were approximately $3.3 million for the six months ended June 30, 2020, compared to approximately $5.1 million for the six months ended June 30, 2019. This decrease was primarily related to an overall reduction in expenditures related to advancing our lead product candidate EHP-101, including a reduction in clinical expenses and related contract manufacturing costs, regulatory expenses and non-clinical studies, of approximately $2.3 million related to a Phase 1 clinical study that commenced in September 2018 and was completed in August 2019. This decrease in research and development expenses was offset by an increase in stock-based compensation and personnel-related expenses of approximately $0.5 million compared to the prior period.
We expect research and development expenses to continue to increase in 2020 as compared to 2019 as we advance our clinical trials and continue our preclinical development. These expenditures are subject to numerous uncertainties regarding timing and cost to completion. Completion of our preclinical development and clinical trials may take several years or more and the length of time generally varies according to the type, complexity, novelty and intended use of a product candidate.
General and Administrative Expenses
General and administrative expenses consist primarily of legal and patent fees, professional service fees, facility and office expenses, and personnel related expenses, such as salaries, benefits, travel and other related expenses, including stock-based compensation.
51
Our general and administrative expenses were $4.2 million for the year ended December 31, 2019, compared to $2.0 million for the year ended December 31, 2018. This increase was primarily related to an increase in personnel related expenses, including stock-based compensation expense of approximately $1.9 million compared to the prior year and an increase in legal and professional fees and general corporate expenses of approximately $0.3 million.
Our general and administrative expenses were approximately $1.8 million for the six months ended June 30, 2020, compared to approximately $2.1 million for six months ended June 30, 2019. This decrease was primarily related to a reduction in stock-based compensation expense of approximately $0.4 million compared to the prior period, offset by an increase in personnel-related expenses of approximately $0.1 million compared to the prior period.
Other (Income)/Expense
Other (income)/expense consists of related party interest expense, related party interest income, and foreign currency losses.
Since inception, we have received advances from EHS to fund our operations, under a revolving loan agreement (the Related Party Loan). During the year ended December 31, 2019, we recognized $2.4 million in interest expense on our Related Party Loan, compared to $0.7 million in interest expense for the year ended December 31, 2018. The increase is due primarily to a non-cash charge of $1.4 million for accretion of a beneficial conversion feature on the Related Party Loan. The remaining increase is due to the additional borrowings during the year ended December 31, 2019. During the six months ended June 30, 2020, we recognized approximately $0.3 million in interest expense on the Related Party Loan, compared to approximately $2.0 million in interest expense for the six months ended June 30, 2019. The decrease is due primarily to a non-cash charge of approximately $1.4 million recorded during the six months ended June 30, 2019 related to accretion of a beneficial conversion feature on the Related Party Loan. The remaining decrease is due to a lower principal balance on the Related Party Loan, during the six months ended June 30, 2020, as compared to the six months ended June 30, 2019.
We recognized a foreign currency loss of $33,282 for the year ended December 31, 2019, compared to a loss of $54,473 for the year ended December 31, 2018. During the six months ended June 30, 2020 we recognized a foreign currency loss of $20,729 compared to a foreign currency loss of $21,839 for the six months ended June 30, 2019. Foreign currency losses are due primarily to the timing of fluctuations in the exchange rates between the US Dollar and other foreign currencies, related to contracts and other transactions which are denominated in currencies other than the US Dollar.
Net Loss
Our net loss was $16.4 million for the year ended December 31, 2019, compared to $8.4 million for the year ended December 31, 2018. Our net loss was approximately $5.4 million for the six months ended June 30, 2020, compared to approximately $9.2 million for the six months ended June 30, 2019.
Liquidity and Capital Resources
To date, we have generated no cash from operations and negative cash flows from operating activities. Our operations have been funded with capital raised from the Offering, along with the proceeds from the Related Party Loan with our majority stockholder, EHS. We have the ability to continue borrowing under the Related Party Loan, but there is no guarantee of continued funding under the loan agreement. The Related Party Loan may be repaid by us or, at the option of EHS, converted by EHS into shares of the Company at $2.00 per share. Amounts advanced under the Related Party Loan and accrued interest are due upon demand.
The Offering was qualified by the SEC in March 2018. We initially offered a maximum of 10,000,000 shares of Common Stock on a “best efforts” basis, at a price of $5.00 per share. In July 2019, following qualification by the SEC of our Post-Qualification Offering Circular Amendment No. 3 as of June 7, 2019, we began offering shares of Common Stock at a price of $6.00 per share. As of November 20, 2020, since the commencement of the Offering in March 2019, we have received commitments for the sale of a total of 8.5 million shares of Common Stock pursuant to the Offering for gross proceeds of approximately $48.4 million, inclusive of both completed sales and pending sales (received subscriptions) in process. In addition, we have also received commitments totaling $0.7 million for which funding has not yet been received.
In addition, during the six months ended June 30, 2019, we also sold 65,700 shares of Common Stock for gross proceeds of $328,500 in an exempt offshore offering under Regulation S under the Securities Act.
52
Our future expenditures and capital requirements will depend on numerous factors, including the success of our Offering and the progress of our research and development efforts.
Our business does not presently generate any cash. We believe that if we raise the $17.1 million (the Maximum Amount) allowed under our Offering pursuant to this Offering Circular, we will have sufficient capital to finance our operations for at least through the end of 2021, however, if we do not sell the Maximum Amount or if our operating and development costs are higher than expected, we will need to obtain additional financing prior to that time. We do not have any track record for self-underwritten Regulation A+ offerings, and there can be no assurance we will raise the Maximum Amount. Further, we expect that during and/or after such period, we will be required to raise additional funds to finance our operations until such time that we can conduct profitable revenue-generating activities. However, no assurances can be made that we will be successful obtaining additional equity or debt financing, or that ultimately, we will achieve profitable operations and positive cash flow.
On April 22, 2020, as a result of the COVID-19 global pandemic, we received loan proceeds of $292,152 (the PPP Loan) from Silicon Valley Bank pursuant to the Paycheck Protection Program (the PPP) established as part of the Coronavirus Aid, Relief and Economic Security Act (the CARES Act). The PPP Loan, which is evidenced by a Note dated April 21, 2020, matures on April 21, 2022 and bears interest at a rate of 1% per annum, payable monthly commencing on November 21, 2020. The Note may be prepaid at any time prior to maturity with no prepayment penalties. The principal and interest accrued under the PPP Loan are forgivable after eight weeks as long as the loan proceeds are used for eligible purposes, including payroll, benefits, rent and utilities. The amount of loan forgiveness will be reduced if the Company terminates employees or reduces salaries during the eight-week period. We intend to use the proceeds of the PPP Loan for purposes consistent with the PPP. While we believe that our use of the PPP Loan proceeds will meet the conditions for forgiveness, no assurances can be made that we will not take actions that could cause the Company to be ineligible for forgiveness of the PPP Loan, in whole or in part.
Credit Facilities
In September 2017, the Company and EHS entered the Related Party Loan, which was amended in January 2018 and November 2019. Borrowings under the Related Party Loan may be drawn down from time to time, and may be repaid by us in cash, or at the option of EHS, converted into shares of the Company at $2.00 per share or at a price to be equally agreed to between EHS and the Company. In November 2019, the Related Party Loan was further amended to reduce the interest rate from 12% to 10%, compounded semiannually. The Related Party Loan is payable upon demand and has no expiration date.
In April 2019, we received written notice of demand (Notice) from EHS for payment of all accrued interest on the Related Party Loan as of March 31, 2019, which resulted in a cash payment of $1,044,901 to EHS. Also in April 2019, we received a second Notice from EHS that called for the following, upon qualification by the SEC of our Form 1-A Post-Qualification Offering Circular Amendment on Form 1-A (the Post-Qualification Offering Circular Amendment): (1) repayment of $2,000,000 of the unpaid principal balance under the loan, and (2) the conversion of an additional $2,500,000 of the unpaid principal balance under the loan at a conversion price of $2.00 per share. Our Post-Qualification Offering Circular Amendment was qualified by the SEC on June 7, 2019 and the repayment of the $2,000,000 of unpaid principal was transacted as a cashless discharge and offset between the Related Party Loan and the Related Party Note Receivable (as defined below). Concurrently, 1,250,000 shares of EHP common stock were issued to EHS at a conversion price of $2.00 per share, further reducing the principal balance of the Related Party Loan by $2,500,000.
In November 2019, we received written notice of demand from EHS for payment of $3,000,000 of unpaid principal balance and $178,933 of accrued interest on the Related Party Loan, which was transacted as a cashless discharge and offset between the Related Party Loan and the remaining unpaid principal and accrued interest balances under the Related Party Note Receivable (as defined below) as of November 15, 2019.
In April 2020 and June 2020, we received written notice of demand from EHS for payments of $150,000 and $200,000, respectively, of accrued interest on the Related Party Loan. On October 6, 2020, we received written notice of demand from EHS for payment of all accrued interest due under the Related Party Loan as of that date, which amounted to $472,524.
53
In May 2019, our Board of Directors authorized a funding arrangement with EHS (the Related Party Note Receivable), which was amended in August 2019 and September 2019 to extend the repayment dates, whereby we may advance funds to EHS in the form of interest bearing (12%) short term notes, up to an aggregate principal amount of $6,000,000 under a Promissory Note between EHS and EHP (the Promissory Note). Advances under the Promissory Note were originally due for repayment with accrued and unpaid interest three months from the date of the advance. A total of $5,000,000 was advanced and $178,933 accrued as interest receivable under the Related Party Note Receivable, all of which was offset through cashless discharges against the unpaid principal and accrued interest payable balances, respectively, under the existing Related Party Loan with EHS.
As of June 30, 2020, we had an outstanding balance of approximately $3.75 million under the Related Party Loan plus accrued interest of approximately $0.4 million. We have the ability to continue borrowing under this loan agreement; however, there is no guarantee of continued funding.
In January 2020, our Australian subsidiary, EHP Australia, entered into a loan agreement with Rocking Horse Nominees Pty Ltd (Rocking Horse), whereby Rocking Horse advanced $AU1.2 million (approximately $0.8 million USD) to EHP Australia. The loan is secured by the tax incentive refund anticipated to be received during 2020 for eligible spending incurred under the Australian research and development tax incentive program during 2019. The loan had an upfront establishment fee of 1.2% and accrued interest at 1.25% per month compounded. Approximately $53,000 in interest expense was incurred during the six months ended June 30, 2020, and the loan and all outstanding interest were repaid to Rocking Horse as of June 30, 2020.
Going Concern
Our financial statements have been prepared on a going concern basis, which contemplates the realization of assets and the satisfaction of liabilities in the normal course of business. Our ability to continue as a going concern is contingent upon its ability to raise additional capital as required. During the period from March 2, 2017 (inception) through June 30, 2020, we have incurred cumulative net losses of approximately $33.1 million. Currently, we intend to finance our operations through equity and debt financings.
We continually evaluate our plan of operations to determine the manner in which we can most effectively utilize our limited cash resources. The timing of completion of any aspect of our plan of operations is highly dependent upon the availability of cash to implement that aspect of the plan and other factors beyond our control. There is no assurance that we will successfully obtain the required capital or revenues, or, if obtained, that the amounts will be sufficient to fund our ongoing operations.
These circumstances raise substantial doubt on our ability to continue as a going concern. Our financial statements do not include any adjustments relating to the recoverability and classification of recorded asset amounts or classification of liabilities that might result from this uncertainty.
Capital Expenditures
We do not have any contractual obligations for ongoing capital expenditures at this time.
Contractual Obligations, Commitments and Contingencies
We may be required to make future payments to Emerald Health Biotechnology España S.L.U. (EHBE), formerly VivaCell Biotechnology España S.L. (VivaCell) based on the achievement of milestones set forth in the intellectual property transfer agreement. These milestone payments are based on the achievement of development or regulatory milestones, including completion of various phases of clinical trials, filing of product license applications and approval of product licenses from the United States FDA or a foreign regulatory agency. The aggregate amount of additional milestone payments that we could be required to pay under our agreement with EHBE is 2.7 million Euros, or approximately $3.0 million USD per product, based upon the exchange rate at June 30, 2020. These amounts assume that all remaining milestones associated with the milestone payments are met. In the event that product license approval for any of the related products is obtained, we are required to make royalty payments of 2.5% of net revenues from commercial sales of the related products.
Milestone payments of approximately $440,000 have been paid to EHBE in accordance with the Intellectual Property Transfer Agreement (IPTA). The milestone liability was accrued in 2019. Approximately $300,000 of that liability was paid to EHBE during the six months ended June 30, 2020 and the remaining liability was paid subsequent to June 30, 2020. Because future milestones are contingent, we are not in a position to reasonably estimate how much, if any, additional milestone payments will ultimately be paid, or when. Many of the remaining milestone events are related to progress in clinical trials which will take several years to achieve.
54
On May 1, 2018, we entered into a two-year non-cancelable building lease for our corporate headquarters in San Diego, California. Effective August 15, 2019, the lease was amended to include additional space at the existing premises and to extend the term of the original lease through August 31, 2022. Under the lease, we currently pay a combined base rent of $21,238 per month through August 2021, after which time the base rent will increase by approximately 3% per year. As of June 30, 2020, our remaining obligations under this operating lease were $ 126,199 in 2020, $257,312 in 2021 and $174,818 in 2022.
In July 2020, we entered into an agreement to sublease a portion of our existing non-cancelable building lease to a tenant, effective August 1, 2020, and continuing through August 31, 2022. The Company expects to incur a loss of approximately $55,000 as a result of the transaction.
Off-Balance Sheet Arrangements
We did not have during the periods presented, and we do not currently have, any off-balance sheet arrangements.
Plan of Operations
As noted above, the continuation of our current plan of operations requires us to raise significant additional capital. If we are successful in raising capital through the sale of Shares offered for sale in this Offering Circular, we believe that the Company will have sufficient cash resources to fund its plan of operations at least through the end of 2021. If we are unable to do so, we may have to curtail and possibly cease some operations.
We are a pre-revenue development stage biotechnology company and began operations in March 2017. Our plan of operations for the next few years includes developing two initial therapeutic product opportunities, EHP-101 and EHP-102, that together target four initial indications: multiple sclerosis (MS), systemic scleroderma (SSc), Parkinson’s disease (PD) and Huntington’s disease (HD). We have successfully completed a Phase 1 human clinical study in Australia to establish EHP-101 safety and pharmacokinetics (PK) in healthy volunteers. We have initiated a Phase 2a safety and efficacy study in SSc patients and we plan to commence a Phase 2 study in MS patients later this year. If such studies are successful, the product candidates will then advance into additional efficacy studies thereafter. We have completed preclinical proof of concept (POC) work for EHP-102. POC is defined herein as the demonstration of positive benefits in animal models for the intended human therapeutic use We are now in the manufacturing and formulation development stage and, if successfully completed, expect to begin clinical-enabling studies for HD and PD and then advance to Phase 1 human clinical studies. Our expenses have increased significantly as we continue to advance our human clinical trials.
We continually evaluate our plan of operations to determine the manner in which we can most effectively utilize our limited cash resources. The timing of completion of any aspect of our plan of operations is highly dependent upon the availability of cash to implement that aspect of the plan and other factors beyond our control. There is no assurance that we will successfully obtain the required capital or revenues, or, if obtained, that the amounts will be sufficient to fund our ongoing operations.
These circumstances raise substantial doubt on our ability to continue as a going concern. Our financial statements do not include any adjustments relating to the recoverability and classification of recorded asset amounts, or amounts and classification of liabilities that might result from this uncertainty.
Quantitative and Qualitative Disclosures about Market Risk
In the ordinary course of our business, we are not exposed to market risk of the sort that may arise from changes in interest rates or foreign currency exchange rates, or that may otherwise arise from transactions in derivatives.
Contingencies
Certain conditions may exist as of the date the financial statements are issued, which may result in a loss to the Company, but which will only be resolved when one or more future events occur or fail to occur. The Company’s management, in consultation with its legal counsel as appropriate, assesses such contingent liabilities, and such assessment inherently involves an exercise of judgment. In assessing loss contingencies related to legal proceedings that are pending against the Company or unasserted claims that may result in such proceedings, the Company, in consultation with legal counsel, evaluates the perceived merits of any legal proceedings or unasserted claims, as well as the perceived merits of the amount of relief sought or expected to be sought therein. If the assessment of a contingency indicates it is probable that a material loss has been incurred and the amount of the liability can be estimated, then the estimated liability would be accrued in the Company’s financial statements. If the assessment indicates a potentially material loss contingency is not probable, but is reasonably possible, or is probable, but cannot be estimated, then the nature of the contingent liability, together with an estimate of the range of possible loss, if determinable and material, would be disclosed. Loss contingencies considered remote are generally not disclosed unless they involve guarantees, in which case the guarantees would be disclosed. We are not aware of any matters which result in a loss contingency.
55
Relaxed Ongoing Reporting Requirements
Regulation A+ provides that a filer can take advantage of an extended transition period for complying with new or revised accounting standards. We have elected to avail ourselves of this exemption and, therefore, we will not be subject to the same adoption period for new or revised accounting standards as public companies.
Upon the completion of this Offering, we may elect to become a public reporting company under the Securities Exchange Act of 1934, as amended (the Exchange Act). If we elect to do so, we will be required to publicly report on an ongoing basis as an “emerging growth company” (as defined in the Jumpstart Our Business Startups Act of 2012, which we refer to as the JOBS Act) under the reporting rules set forth under the Exchange Act. As defined in the JOBS Act, an emerging growth company is defined as a company with less than $1 billion in revenue during its last fiscal year. An emerging growth company may take advantage of specified reduced reporting and other burdens that are otherwise applicable generally to public companies.
For so long as we remain an “emerging growth company,” we may take advantage of certain exemptions from various reporting requirements that are applicable to other Exchange Act reporting companies that are not “emerging growth companies,” including but not limited to:
| ● | not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act; | |
| ● | taking advantage of extensions of time to comply with certain new or revised financial accounting standards; | |
| ● | being permitted to comply with reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements; and | |
| ● | being exempt from the requirement to hold a non-binding advisory vote on executive compensation and stockholder approval of any golden parachute payments not previously approved. |
If we are required to publicly report under the Exchange Act as an “emerging growth company”, we expect to take advantage of these reporting exemptions until we are no longer an emerging growth company. We would remain an “emerging growth company” for up to five years, though if the market value of our Common Stock that is held by non-affiliates exceeds $700 million, we would cease to be an “emerging growth company.”
We have commenced reporting under the Regulation A+ reporting requirements. If we elect not to become a public reporting company under the Exchange Act, we will be required to continue to publicly report on an ongoing basis under the reporting rules set forth in Regulation A+ for Tier 2 issuers. The ongoing reporting requirements under Regulation A+ are more relaxed than for “emerging growth companies” under the Exchange Act. The differences include, but are not limited to, being required to file only annual and semi-annual reports, rather than annual and quarterly reports. Annual reports are due within 120 calendar days after the end of the issuer’s fiscal year, and semi-annual reports are due within 90 calendar days after the end of the first six months of the issuer’s fiscal year.
Trend Information
Because we are still in the startup phase and have only recently commenced our research and product development, we are unable to identify any recent trends in revenue or expenses. Thus, except as set forth below, we are unable to identify any known trends, uncertainties, demands, commitments or events involving our business that are reasonably likely to have a material effect on our revenues, income from continuing operations, profitability, liquidity or capital resources, or that would cause the reported financial information in this Offering Circular to not necessarily be indicative of future operating results or financial condition.
Unpredictable events, such as the COVID-19 outbreak, and associated business disruptions, including delayed clinical trials and laboratory resources, could harm our financial condition, affect our operations, increase our costs and expenses, and impact our ability to raise capital. Our operations may be subject to unpredictable events, such as earthquakes, power shortages, telecommunications failures, water shortages, floods, hurricanes, typhoons, fires, extreme weather conditions, medical epidemics or pandemics such as the COVID-19 outbreak, and other natural or manmade disasters or business interruptions, for which we may not be insured. We do not carry insurance for all categories of risk that our business may encounter. The occurrence of any of these business disruptions could seriously harm our operations and financial condition, delay our product development and regulatory approvals of clinical trials, and increase our costs and expenses. Additionally, COVID-19 has caused significant disruptions to the global financial markets, which could impact our ability to raise additional capital. The ultimate impact on us and any delays in our research and development is unknown, but our operations and financial condition could suffer as a result of any of these types of unpredictable events. Further, any significant uninsured liability may require us to pay substantial amounts, which would adversely affect our business, results of operations, financial condition and cash flows.
56
DIRECTORS, EXECUTIVE OFFICERS AND SIGNIFICANT EMPLOYEES
The table below sets forth our directors and executive officers as of the date of this Offering Circular.
| Name | Position | Age | Term of Office | Approximate hours per week for part-time employees | |||||||
| Officers and Significant Employees: | |||||||||||
| James DeMesa | President and Chief Executive Officer | 62 | (1) | ||||||||
| Lisa Sanford | Chief Financial Officer | 54 | (2) | ||||||||
| Alain Rolland | Chief Operating Officer | 60 | (3) | ||||||||
| Joachim Schupp | Chief Medical Officer | 67 | (4) | ||||||||
| Eduardo Muñoz | Chief Scientific Officer | 61 | Since June 2017 | 25 hours per week (5) | |||||||
| Nancy Coulson | Senior Vice President Regulatory and Quality Affairs | 76 | (6) | 20 hours per week | |||||||
| Directors: | |||||||||||
| James L. Heppell | Director & Chairman | 65 | (7) | ||||||||
| James DeMesa | Director | 62 | Since December 2019 | ||||||||
| Gaetano A. Morello | Director | 59 | Since March 2017 | ||||||||
| Punit S. Dhillon | Director | 40 | Since March 2017 | ||||||||
| (1) | James DeMesa has served as Chief Executive Officer since March 2017. He was appointed as President and as a director in November 2019, subsequent to the resignation of Avtar Dhillon as President in August 2019 and as Chairman in November 2019. |
| (2) | Lisa Sanford was appointed Interim Chief Financial Officer as of October 1, 2018 and was appointed Chief Financial Officer as of April 1, 2019. From July 20, 2018 through September 30, 2018 she served as Vice President of Finance. |
| (3) | Alain Rolland was appointed Chief Development Officer as of May 1, 2018 and was appointed Chief Operating Officer as of September 1, 2019. From February 1, 2018 through April 20, 2018 he served as Vice President of Product Development and from April 21, 2018 through August 31, 2019 he served as Chief Development Officer. |
| (4) | Joachim Schupp was appointed Chief Medical Officer as of January 1, 2019. From August 1, 2018 through December 31, 2018 he served as Senior Vice President of Medical Affairs. |
| (5) | Dr. Muñoz is a consultant who may be deemed a significant employee and acts as our Chief Scientific Officer pursuant to a consulting agreement with the University of Córdoba where Dr. Muñoz is employed in the Department of Cellular Biology, Physiology and Immunology. |
| (6) | Nancy Coulson was engaged as a senior regulatory advisor, averaging approximately 80 hours per month from March 2017 through January 2020. Effective February 1, 2020, Ms. Coulson was hired as a part-time employee serving as Senior Vice President of Regulatory and Quality Affairs. |
| (7) | James L. Heppell has served as a Director since 2017 and was appointed Chairman as of November 15, 2019, upon the resignation of Avtar Dhillon as Executive Chairman. |
There is no arrangement or understanding between the persons described above and any other person pursuant to which the person was selected to his or her office or position.
57
Executive Officers, Directors and Significant Employees
James M. DeMesa, MD, MBA, President, Chief Executive Officer and Director. Dr. DeMesa has over 30 years of experience in biotech and pharmaceutical leadership, product development, and clinical and regulatory management. He has completed partnerships and collaborations with pharmaceutical, biotech, and medical device companies and has raised more than $200 million to advance product development to clinical stage, regulatory approval, and commercialization. He is a former practicing physician and CEO of two public biotech companies: Migenix, from 2001 to 2008 and GenSci Regeneration Sciences, from 1996 to 2001 (now part of Integra LifeSciences). Dr. DeMesa also currently serves as Director for two biotech companies: OncoSec Medical Incorporated and Induce Biologics. Prior to his CEO roles, Dr. DeMesa was Vice President, Medical and Regulatory Affairs at Biodynamics International (now part of RTI Surgical) and Bentley Pharmaceuticals (now part of Teva Pharmaceuticals). Dr. DeMesa received a bachelor’s degree in Chemistry, and MD, and MBA degrees from the University of South Florida and did his medical residency at the University of North Carolina.
Eduardo Muñoz, MD, PhD, Chief Scientific Officer. Dr. Muñoz is a Professor of Immunology in the Department of Cell Biology, Physiology and Immunology of the University of Córdoba (Spain) since 1992 and Director of the Inflammation and Cancer Research Group at the Institute Maimonides for Biomedical Research of Córdoba since 2012. Dr. Muñoz has more than 30 years of experience in biomedical research and is the author of more than 250 articles, patents, and book chapters with more than 9,000 citations. He is experienced in the mechanism of actions of cannabinoids and endocannabinoids as well as the development of cannabinoid-based new chemical entities. Dr. Muñoz belongs to the editorial board of several scientific journals and is a co-founder of three biotech companies, Emerald Health Biotechnology España, S.L.U. (Spain), Glactone Pharma AB (Sweden) and InnnoHealth Group (now part of Evonik Industries AG). He received a PhD in Medicine and Surgery at the University of Córdoba and was an associate researcher at Tufts University in Boston, and at the Institute Pasteur in Paris.
Lisa Sanford, Chief Financial Officer. Ms. Sanford has 30 years of diversified experience in finance and accounting in the life sciences, biotechnology, and pharmaceutical industries. From April 2000 through July 2018, Ms. Sanford managed her own consulting business, providing finance and accounting services for both public and private companies. Ms. Sanford also served as an audit senior manager at Ernst & Young LLP, where she worked for twelve years and was involved in multiple IPOs and business combinations. She received her bachelor’s degree in Accounting from Lehigh University and is a Certified Public Accountant.
Alain Rolland, PharmD, PhD, Chief Operating Officer and Executive Vice President. Dr. Rolland has over 30 years of international leadership experience in pharmaceutical and biotech companies. He has focused on the discovery and development of biologics and small molecules in a variety of therapeutic areas including immuno-oncology, cardiovascular and hematological disorders, dermatology, and infection disease vaccines. Prior to joining EHP, Dr. Rolland was a co-founder and served as CEO, President and Director of CHIME BioTherapeutics. He was previously Executive Vice President and Chief Scientific Officer at HUYA Bioscience International, Executive Vice President, Product Development at Vical, and Senior Vice President, Preclinical R&D, Head of the Woodlands Center of Valentis. Dr. Rolland has published over 90 scientific articles and book chapters and is editor of three scientific books. He is a member of several scientific societies, the founding Editor-in-Chief of Current Pharmaceutical Biotechnology, and an editorial board member of several journals. He has been honored by the American Association of Pharmaceutical Scientists as an “AAPS Fellow” for professional excellence and outstanding contributions to the pharmaceutical sciences. Dr. Rolland earned his doctorate degree in Pharmacy (Pharm.D.) and in Pharmaceutical Sciences (Ph.D.) from Rennes University, France.
Joachim P.H. Schupp, MD, Dr. med, Chief Medical Officer. Dr. Schupp has over 30 years of international pharmaceutical industry experience in all phases of drug development and several therapeutic areas. He directed multiple Phase 1 – IV clinical studies and led multiple international cross-functional project teams at Ciba-Geigy and Novartis Pharmaceuticals in Switzerland, which resulted in worldwide approval of several New Drug Applications (NDAs), Biologics License Applications (BLAs) and supplemental applications for small molecule drugs, biologics and devices currently on the market. Dr. Schupp also served as Vice President, Clinical & Regulatory Affairs at HUYA Bioscience International, Chief Medical Officer at Imprimis/Transdel Pharmaceuticals, Inc., Vice President, Clinical Development at Apricus Biosciences, Inc., Vice President, Medical Affairs at Adventrx Pharmaceuticals, Inc. and Vice President, Clinical Data Services at ProSanos, Inc. Prior to joining EHP, Dr. Schupp managed his own consulting business (MEQVal), providing services as a medical monitor and drug safety physician. Dr. Schupp received his MD and doctorate (Dr. med.) from Freie Universität Berlin in Germany and practiced medicine in Germany, South Africa, UK and Switzerland.
58
Nancy Coulson, Senior Vice President Regulatory and Quality Affairs. Ms. Coulson has over 30 years of experience in providing strategic counsel for regulatory, clinical, and quality affairs. As a senior advisor for medical device and pharmaceutical companies, she manages US and international regulatory documents, briefing packages, and global regulatory dossiers across multiple product categories. Ms. Coulson has also completed several successful pre-approval inspections for new drug and device manufacturing facilities. Most recently, she was Worldwide Director, Regulatory Affairs at Cordis, a Johnson & Johnson company, where she provided strategic direction on global regulatory submissions. She also held scientific and senior regulatory positions at Bristol-Myers Squibb, Bausch & Lomb, GenSci, and Migenix. She received a Bachelor of Science in Chemistry from LeMoyne College and an MBA from Chapman University.
James L. Heppell, BSc, LLB, Chairman. Mr. Heppell has been involved with the Emerald Health Group since 2014. From 2003 to 2014, Mr. Heppell was Co-Founder, President and Director of BC Advantage Funds (VCC) Ltd., a venture fund focused on investing in and building technology, life sciences and clean technology companies. Mr. Heppell was founder and CEO of the Advantage Life Sciences I Fund, which won the Canadian Venture Capital Deal of the Year Award in 2006 for having the highest realized return of all venture capital funds in Canada. Earlier in his career, he practiced corporate securities law with Fasken Martineau DuMoulin and later served as President and CEO of Catalyst Corporate Finance Lawyers, a boutique corporate finance law firm representing life science and tech companies. He is a past member of the Securities Policy Advisory Committee to the British Columbia Securities Commission and is a Past-Chairman of the Securities Section of the Canadian Bar Association. Over the years, Mr. Heppell has written a number of articles, co-edited the Annotated British Columbia Securities Act and coordinated and taught numerous courses on corporate finance and corporate governance issues. He earned a BSc in microbiology and a law degree from the University of British Columbia.
Gaetano A. Morello, ND, Director. Dr. Morello is an accomplished clinician with direct, first-hand experience in the clinical and medical application of cannabinoids. Dr. Morello has practiced at the Complex Chronic Disease Program (CCDP) at Woman’s Hospital in Vancouver, Canada since 2013 and is also a study investigator at the CCDP Clinical Cannabis Trial. He has also served on the Quality Assurance Committee for the College of Naturopathic Physicians of British Columbia since 2010 as well as other health and medical panels. He has authored Cleanse, The Healing Power, of the Endocannabinoid System, Ultimate Inside Out Approach, Whole Body Cleansing, Stress and Anxiety, A Powerful Antioxidant, and was a contributing author to A Textbook of Natural Medicine, and numerous journal publications. He has made more than 500 medical presentations in the US, Canada, Australia, Germany, and Italy in the last decade. Dr. Morello has a BSc in Cell Biology/ Nutrition from the University of British Columbia and a Doctorate in Naturopathic Medicine from Bastyr University.
Punit S. Dhillon, Director. Mr. Dhillon was appointed CEO and Chair of Emerald Bioscience, Inc. (OTCQB: EMBI) in August 2020. He is the co-founder and former President & CEO of OncoSec Medical Incorporated (NASDAQ: ONCS), a leading biopharmaceutical company developing cancer immunotherapies for the treatment of solid tumors, where he served as an Executive until March 2018 and a Director until February 2020. Mr. Dhillon serves as a Director and Audit Committee Chair for Emerald Health Therapeutics, and also serves as Director for Arch Therapeutics Inc. (OTCQB: ARTH). Prior to OncoSec, from 2003-2011, he served as Vice President of Finance and Operations at Inovio Pharmaceuticals, Inc. (NASDAQ: INO). Collectively, he has led and assisted in raising over $500 million through financings, M&A deals and several licensing transactions with large pharma. His management experience spans corporate finance, M&A integration, in-licensing and out-licensing of key intellectual property, strategy implementation, corporate transactions and collaborations with leading universities and global disease specific opinion leaders. Mr. Dhillon also co-founded and is the Director of the Young Entrepreneur Leadership Launchpad (YELL), a registered Canadian charity that partners with schools to support entrepreneurial learning. Mr. Dhillon holds a BA (Honours) in Political Science and a minor in Business Administration from Simon Fraser University.
59
Board Leadership Structure and Risk Oversight
The Board oversees our business and considers the risks associated with our business strategy and decisions. The Board currently implements its risk oversight function as a whole. Each of the Board committees, when established, will also provide risk oversight in respect of its areas of concentration and reports material risks to the Board for further consideration.
Term of Office
Officers hold office until his or her successor is elected and qualified. Directors are appointed to serve for one year until the meeting of the Board following the annual meeting of stockholders and until their successors have been elected and qualified.
Director Independence
We use the definition of “independence” of The NASDAQ Stock Market to make this determination. NASDAQ Listing Rule 5605(a)(2) provides that an “independent director” is a person other than an officer or employee of the company or any other individual having a relationship which, in the opinion of the Company’s Board, would interfere with the exercise of independent judgment in carrying out the responsibilities of a director. The NASDAQ listing rules provide that a director cannot be considered independent if:
| ● | the director is, or at any time during the past three years was, an employee of the company; |
| ● | the director or a family member of the director accepted any compensation from the company in excess of $120,000 during any period of twelve consecutive months within the three years preceding the independence determination (subject to certain exemptions, including, among other things, compensation for board or board committee service); |
| ● | the director or a family member of the director is a partner in, controlling stockholder of, or an executive officer of an entity to which the company made, or from which the company received, payments in the current or any of the past three fiscal years that exceed 5% of the recipient’s consolidated gross revenue for that year or $200,000, whichever is greater (subject to certain exemptions; |
| ● | the director or a family member of the director is employed as an executive officer of an entity where, at any time during the past three years, any of the executive officers of the company served on the compensation committee of such other entity; or |
| ● | the director or a family member of the director is a current partner of the company’s outside auditor, or at any time during the past three years was a partner or employee of the company’s outside auditor, and who worked on the company’s audit. |
Under such definitions, we have three independent directors. However, our Common Stock is not currently quoted or listed on any national exchange or interdealer quotation system with a requirement that a majority of our Board be independent and, therefore, the Company is not subject to any director independence requirements.
Certain Relationships
Two of our non-employee directors, James Heppell and Gaetano Morello, also serve on the Board of Directors of our majority stockholder, EHS.
Punit Dhillon, who serves on our Board of Directors, is the nephew of Dr. Avtar Dhillon, former President and Executive Chairman. Currently, Dr. Avtar Dhillon serves as a Board Observer and provides corporate finance and strategic business advisory services to the Company.
60
Involvement in Certain Legal Proceedings
To our knowledge, except as described below none of our current directors or executive officers has, during the past ten years:
| ● | been convicted in a criminal proceeding or been subject to a pending criminal proceeding (excluding traffic violations and other minor offenses); |
| ● | had any bankruptcy petition filed by or against the business or property of the person, or of any partnership, corporation or business association of which he or she was a general partner or executive officer, either at the time of the bankruptcy filing or within two years prior to that time; |
| ● | been subject to any order, judgment, or decree, not subsequently reversed, suspended or vacated, of any court of competent jurisdiction or federal or state authority, permanently or temporarily enjoining, barring, suspending or otherwise limiting, his involvement in any type of business, securities, futures, commodities, investment, banking, savings and loan, or insurance activities, or to be associated with persons engaged in any such activity; |
| ● | been found by a court of competent jurisdiction in a civil action or by the SEC or the Commodity Futures Trading Commission to have violated a federal or state securities or commodities law, and the judgment has not been reversed, suspended, or vacated; |
| ● | been the subject of, or a party to, any federal or state judicial or administrative order, judgment, decree, or finding, not subsequently reversed, suspended or vacated (not including any settlement of a civil proceeding among private litigants), relating to an alleged violation of any federal or state securities or commodities law or regulation, any law or regulation respecting financial institutions or insurance companies including, but not limited to, a temporary or permanent injunction, order of disgorgement or restitution, civil money penalty or temporary or permanent cease-and-desist order, or removal or prohibition order, or any law or regulation prohibiting mail or wire fraud or fraud in connection with any business entity; or |
| ● | been the subject of, or a party to, any sanction or order, not subsequently reversed, suspended or vacated, of any self-regulatory organization (as defined in Section 3(a)(26) of the Securities Exchange Act of 1934, as amended (the Exchange Act)), any registered entity (as defined in Section 1(a)(29) of the Commodity Exchange Act), or any equivalent exchange, association, entity or organization that has disciplinary authority over its members or persons associated with a member.
|
Gaetano Morello, one of our non-employee directors, was appointed as Chief Executive Officer of Emerald Health Bioceuticals Inc. (“EHB”) on February 7, 2020. EHB is a natural health consumer products company that is also majority-owned by EHS, our majority stockholder. On October 20, 2020, EHB filed a Chapter 7 bankruptcy petition in the U.S. Bankruptcy Court for the Southern District of California, in part due to the material adverse impact of the COVID-19 pandemic on EHB’s business.
Except as set forth above and in our discussion below in “Interest Of Management And Others In Certain Transactions,” none of our directors or executive officers has been involved in any transactions with us or any of our directors, executive officers, affiliates or associates which are required to be disclosed pursuant to the rules and regulations of the SEC.
We are not currently a party to any legal proceedings, the adverse outcome of which, individually or in the aggregate, we believe will have a material adverse effect on our business, financial condition or operating results.
61
COMPENSATION OF DIRECTORS AND EXECUTIVE OFFICERS
The following table represents information regarding the total compensation for the three highest paid executive officers or directors of the Company during the last completed fiscal year ended December 31, 2019:
| Cash Compensation | Other Compensation | Total Compensation | ||||||||||||
| Name | Capacity in which compensation was received | ($) | ($)(4) | ($) | ||||||||||
| James M. DeMesa (1) | President & Chief Executive Officer | $ | 351,250 | $ | 140,000 | $ | 491,250 | |||||||
| Alain Rolland (2) | Chief Operating Officer | $ | 306,250 | $ | 67,123 | $ | 373,373 | |||||||
| Joachim Schupp (3) | Chief Medical Officer | $ | 287,500 | $ | - | $ | 287,500 | |||||||
| All directors as a group (4 persons) | Director | $ | 123,875 | $ | - | $ | 123,875 | |||||||
| (1) | Dr. DeMesa’s annual salary was increased to $375,000 as of April 1, 2019. He is eligible to earn a bonus of up to 50% of his annual salary, subject to Board approval and achievement of certain milestones and metrics. In October 2020, Dr. DeMesa’s annual salary was increased to $470,000. |
| (2) | Dr. Rolland’s salary was increased to $325,000 as of April 1, 2019. He is eligible to earn a bonus of up to 40% of his annual salary, subject to Board approval and achievement of certain milestones and metrics. In October 2020, Dr. Rolland’s annual salary was increased to $390,000. |
| (3) | Dr. Schupp’s salary was increased to $300,000 as of April 1, 2019. He is eligible to earn a bonus of up to 40% of his annual salary, subject to Board approval and achievement of certain milestones and metrics. In October 2020, Dr. Schupp’s annual salary was increased to $350,000. |
| (4) | Other compensation consists of bonuses paid during 2019. |
Director Compensation
In April 2019, the Board of Directors approved a compensation plan which provides for our non-employee directors to begin receiving compensation for their services effective April 1, 2019. Annual Board compensation consist of cash retainers for various responsibilities and option grants in an amount to be determined as a percentage of the outstanding shares of the Company.
In January 2018, two of our directors, Gaetano A. Morello and Punit S. Dhillon, were each granted 100,000 options to purchase our Common Stock at $5.00 per share. In December 2018, the three non-employee directors were granted options to purchase a total of 300,000 shares of our Common Stock at $2.50 per share. Additional grants to purchase a total of 75,000 shares at $6.00 were made to such directors in August 2020.
Employment Agreements
We have employment agreements with our four executive officers, James DeMesa, Alain Rolland, Joachim Schupp and Lisa Sanford. The initial term of each employment agreement is for a period of three years to be extended automatically for successive one-year periods unless terminated earlier by either party upon written notice at least ninety days prior to the end of that period. The Company may terminate the executive officer’s employment, for cause, as defined in the agreement, at any time, without any advance notice. Further, subject to the terms of the agreement, the executive officer may terminate employment with us, at any time for any reason or no reason at all, upon six weeks’ advance written notice. Subject to the notice provisions described in the agreement, the executive officer may terminate employment with us for good cause as defined in the agreement. Subject to the agreement provisions, in situations where the Company terminates the executive officer’s employment without cause, or the term of the agreement ends without the Company offering to extend the agreement on the same terms, or the executive officer resigns for good cause, then the executive officer will be, under certain conditions, entitled to severance compensation from the Company equal to six months of executive officer’s then current base salary.
62
SECURITY OWNERSHIP OF MANAGEMENT & CERTAIN SECURITYHOLDERS
The following table shows the beneficial ownership of our Common Stock as of November 20, 2020 held by: (i) each person known to us to be the beneficial owner of more than 10% of any class of our voting securities; (ii) each director who is the beneficial owner of more than 10% of any class of our voting securities; (iii) each executive officer who is the beneficial owner of more than 10% of any class of our voting securities; and (iv) all directors and executive officers as a group. As of November 20, 2020, there were 19,054,632 shares of our Common Stock outstanding. There were additional 913,310 shares for which commitments have been received under the Offering, but processing or funding is not completed as of the date hereof. Such investment commitments are awaiting completion of receipt and processing and there are no guarantees all funding commitments will ultimately result in the issuance of Shares.
Beneficial ownership is determined in accordance with the rules of the SEC, and generally includes voting power and/or investment power with respect to the securities held. Shares of Common Stock subject to the conversion of a security, or subject to options and warrants currently exercisable or which may become exercisable within 60 days of the date of this Offering Circular, are deemed outstanding and beneficially owned by the person holding such options or warrants for purposes of computing the number of shares and percentage beneficially owned by such person, but are not deemed outstanding for purposes of computing the percentage beneficially owned by any other person. The persons or entities named have sole voting and investment power with respect to all shares of Common Stock shown as beneficially owned by them.
The percentages below are based on fully diluted shares of our Common Stock as of November 20, 2020 and include the 913,310 additional shares issuable upon completion of pending subscription agreements. Unless otherwise indicated, the business address of each person listed is c/o Emerald Health Pharmaceuticals Inc., 5910 Pacific Center Blvd, Ste 320, San Diego, CA 92121.
| Title of Class | Name and Address of Beneficial Owner: | Amount and Nature of Beneficial Ownership | Amount and Nature of Beneficial Ownership Acquirable by Exercise of Option or Conversion of Security | Percent of Class | ||||||
| Common Stock | All directors and named executive officers as a group (9 persons) | 382,000 shares owned | 1,111,996 shares acquirable | 7.1 | % | |||||
| Greater than 10% Beneficial Owners: | ||||||||||
| Common Stock | Emerald Health Sciences Inc. 200-375 Water Street, Vancouver, BC, V6B OM9 | 10,250,000 shares owned | 1,894,358 shares acquirable | 55.6 | % | |||||
63
INterest of Management and others in CERTAIN TRANSACTIONS
Transactions with Related Persons
Except as described below and except for employment arrangements which are described above under “Compensation of Directors and Executive Officers,” there has not been, nor is there currently proposed, any transaction in which we are or were a participant, the amount involved exceeds the lesser of $120,000 or 1% of the average of our total assets at year end for the last two completed fiscal years, and any of our directors, executive officers, holders of more than 10% of our Common Stock or any immediate family member of any of the foregoing had or will have a direct or indirect material interest.
In June 2017, we entered into the IPTA and a Research Agreement with EHBE. During the year ended December 31, 2018, EHBE became a wholly owned subsidiary of Emerald Health Research Inc. (EHR), which is a wholly owned subsidiary of our majority stockholder, EHS. EHP has no ownership or voting rights related to EHR or EHBE.
In September 2017, we entered into a revolving loan agreement (the Related Party Loan) with EHS, our majority stockholder, which was amended in January 2018 and November 2019. Borrowings under the Related Party Loan may be drawn down from time to time, and may be repaid by us in cash, or at the option of EHS, converted into shares of the Company at $2.00 per share or at a price to be equally agreed to between EHS and the Company. In November 2019, the Related Party Loan was further amended to reduce the interest rate from 12% to 10%, compounded semiannually. The Related Party Loan is payable upon demand and has no expiration date.
In April 2019, we received written notice of demand (Notice) from EHS for payment of all accrued interest on the Related Party Loan as of March 31, 2019, which resulted in a cash payment of $1,044,901 to EHS. Also in April 2019, we received a second Notice from EHS that called for the following, upon qualification by the SEC of our Form 1-A Post-Qualification Offering Circular Amendment on Form 1-A filed in May 2019 (the Post-Qualification Offering Circular Amendment): (1) repayment of $2,000,000 of the unpaid principal balance under the loan, and (2) the conversion of an additional $2,500,000 of the unpaid principal balance under the loan at a conversion price of $2.00 per share. Our Post-Qualification Offering Circular Amendment was qualified by the SEC on June 7, 2019 and the repayment of the $2,000,000 of unpaid principal was transacted as a cashless discharge and offset between the Related Party Loan and the Related Party Note Receivable (as defined below). Concurrently, 1,250,000 shares of EHP common stock were issued to EHS at a conversion price of $2.00 per share, further reducing the principal balance of the Related Party Loan by $2,500,000. In April 2020 and June 2020, we received written notice of demand from EHS for payments of $150,000 and $200,000, respectively, of accrued interest on the Related Party Loan. On October 6, 2020, we received written notice of demand from EHS for payment of all accrued interest due under the Related Party Loan as of that date, which amounted to $472,524.
In November 2019, we received written notice of demand from EHS for payment of $3,000,000 of unpaid principal balance and $178,933 of accrued interest on the Related Party Loan, which was transacted as a cashless discharge and offset between the Related Party Loan and the remaining unpaid principal and accrued interest balances under the Related Party Note Receivable (as defined below) as of November 15, 2019. As of June 30, 2020, we had an outstanding balance of approximately $3.75 million under the Related Party Loan, plus accrued interest of approximately $0.4 million. We have the ability to continue borrowing under this loan agreement; however, there is no guarantee of continued funding. A portion of the proceeds raised in this Offering may be used to pay, in whole or in part, the principal and accrued interest outstanding under the Related Party Loan.
In May 2019, our Board of Directors authorized an investment note arrangement with EHS (the Related Party Note Receivable), which was amended in August 2019 and September 2019 to extend the repayment dates, whereby we may advance funds to EHS in the form of interest bearing (12%) short term notes, up to an aggregate principal amount of $6,000,000 under a Promissory Note between EHS and EHP (the Promissory Note). Advances under the Promissory Note were originally due for repayment with accrued and unpaid interest three months from the date of the advance. A total of $5,000,000 was advanced and $178,933 accrued as interest receivable under the Related Party Note Receivable, all of which was offset through cashless discharges against the unpaid principal and accrued interest payable balances, respectively, under the existing Related Party Loan with EHS. As of June 30, 2020, there were no outstanding receivable balances remaining under the Related Party Note Receivable.
64
In June 2019, the Company entered into an Independent Contractor Agreement (the Independent Contractor Agreement) effective April 1, 2019, with EHS, pursuant to which EHS agreed to provide such services as are mutually agreed upon by the Company and EHS, which services may include, without limitation, corporate advisory services. The Independent Contractor Agreement had an initial term of ten years, and specifies compensation which is mutually agreed upon by the Company’s chief executive officer and EHS’ chief executive officer. In November 2019, our Board of Directors approved the termination of the Independent Contractor Agreement, which termination was effective December 31, 2019.
In November 2019, the Company and EHS entered into a Board Observer Agreement, whereby the Company granted to EHS the right to designate an observer on our Board of Directors for so long as EHS maintains ownership of any securities of the Company. Dr. Avtar Dhillon was appointed as the initial Board Observer pursuant to the Board Observer Agreement.
In December 2019, the Company entered into an Independent Contractor Services Agreement with Dr. Avtar Dhillon, pursuant to which Dr. Dhillon will provide ongoing corporate finance and strategic business advisory services to the Company. In exchange for his services, upon the Company completing a material financing. Dr. Dhillon will receive a monthly fee of $10,000, accruing from the effective date of the agreement. The Independent Contractor Services Agreement has an initial term of one year and will renew automatically thereafter unless terminated earlier by either party. Either party may terminate the agreement for cause upon written notice to the other party, or without cause upon 30 days’ prior written notice to the other party.
Punit Dhillon, who serves on our Board of Directors, is the nephew of Dr. Avtar Dhillon, former President and Executive Chairman. Currently, Dr. Avtar Dhillon serves as a Board Observer and provides corporate finance and strategic business advisory services to the Company.
Two of our non-employee directors on our Board of Directors are also the Board of Directors of our majority stockholder, EHS.
We have also entered into indemnification agreements with each of our directors and executive officers. In general, these indemnification agreements require the Company to indemnify a director to the fullest extent permitted by law against liabilities that may arise by reason of his or her service for the Company.
Review, Approval and Ratification of Related Party Transactions
The Audit Committee of the Board of Directors (established in March 2019) reviews and approves all related party transactions.
65
The following is a summary of the rights of our capital stock as provided in our Certificate of Incorporation, and bylaws. For more detailed information, please see our Certificate of Incorporation and bylaws which have been filed as exhibits to the Offering Statement of which this Offering Circular is a part.
General
The Company is authorized to issue one class of stock. The total number of shares of stock which the Company is authorized to issue is One Hundred Million (100,000,000) shares of capital stock, consisting of One Hundred Million (100,000,000) shares of Common Stock. As of November 20, 2020, the Company had 19,054,632 shares of Common Stock outstanding. There were additional 913,310 shares for which commitments have been received under the Offering but processing or funding is not completed as of the date hereof. Such investment commitments are awaiting completion of receipt and processing and there are no guarantees all funding commitments will ultimately result in the issuance of Shares.
In addition, as of November 20, 2020, 3,429,833 shares of Common Stock were reserved for issuance under our 2018 Equity Incentive Plan, of which 2,857,500 shares of our Common Stock will be issuable upon exercise of outstanding grants.
Common Stock Voting
The holders of the Common Stock are entitled to one vote for each share held on all matters to be voted on by the Company’s stockholders. There shall be no cumulative voting.
Dividends
The holders of shares of Common Stock are entitled to dividends when and as declared by the Board from funds legally available therefor if, as and when determined by the Board of Directors of the Company in their sole discretion, subject to provisions of law, and any provision of the Company’s Certificate of Incorporation, as amended from time to time. There are no preemptive, conversion or redemption privileges, nor sinking fund provisions with respect to the Common Stock.
Liquidation
In the event of any voluntary or involuntary liquidation, dissolution or winding up of our affairs, the holders of our Common Stock will be entitled to share ratably in the net assets legally available for distribution to stockholders after the payment of or provision for all of our debts and other liabilities.
Fully Paid and Non-assessable
All outstanding shares of Common Stock are, and the Common Stock to be outstanding upon completion of this Offering will be, duly authorized, validly issued, fully paid and non-assessable.
Changes in Authorized Number
The number of authorized shares of Common Stock may be increased or decreased subject to the Company’s legal commitments at any time and from time to time to issue them, by the affirmative vote of the holders of a majority of the stock of the Company entitled to vote.
66
Delaware Anti-Takeover Statute
We may become subject to Section 203 of the Delaware General Corporation Law, which prohibits persons deemed to be “interested stockholders” from engaging in a “business combination” with a publicly held Delaware corporation for three years following the date these persons become interested stockholders unless the business combination is, or the transaction in which the person became an interested stockholder was, approved in a prescribed manner or another prescribed exception applies. Generally, an “interested stockholder” is a person who, together with affiliates and associates, owns, or within three years prior to the determination of interested stockholder status did own, 15% or more of a corporation’s voting stock. Generally, a “business combination” includes a merger, asset or stock sale, or other transaction resulting in a financial benefit to the interested stockholder. The existence of this provision may have an anti-takeover effect with respect to transactions not approved in advance by the Board of Directors. A Delaware corporation may “opt out” of these provisions with an express provision in its original certificate of incorporation or an express provision in its certificate of incorporation or bylaws resulting from a stockholders’ amendment approved by at least a majority of the outstanding voting shares. We have not opted out of these provisions. As a result, mergers or other takeover or change in control attempts of us may be discouraged or prevented.
Equity Incentive Plan
Compensation of Directors and Executive Officers
Each of the executive officers and directors listed above is eligible to receive equity compensation at the discretion of our board. In January 2018, the Company granted options to purchase 295,000 shares of Common Stock at $5.00 per share with one-third of the options vesting on the first, second and third anniversary of the date of grant. In December 2018, the Company granted options to purchase 1,780,000 shares of Common Stock at $2.50 per share with 800,000 (Directors’) options vesting monthly over twelve months. The remaining 980,000 options vest over three years, with 25% vesting upon grant and 25% vesting on each of the first, second and third anniversary of the date of grant. In August 2020, the Company granted additional options to purchase 782,500 shares of Common Stock at $6.00 per share, with 75,000 directors’ options vesting monthly over twelve months. The remaining 707,500 options vest over three years, with 25% vesting upon grant and 25% vesting on each of the first, second and third anniversary of the grant date.
Upon completion of this Offering, our executive officers and directors will be eligible to receive equity awards under our equity incentive plans at any time at the discretion of our Board of Directors.
2018 Plan
We adopted the 2018 Equity Incentive Plan, as amended December 13, 2018 and August 12, 2020 (the 2018 Plan), on January 29, 2018. The 2018 Plan provides for the grant of incentive stock options, non-qualified stock options, stock appreciation rights, restricted stock, restricted stock units, dividend equivalent rights and other stock and cash-based awards. Shares issued under the 2018 Plan will be shares of our Common Stock. Incentive stock options may be granted only to our employees and employees of any parent or subsidiary corporation. All other awards may be granted to our employees, directors or consultants and to employees, directors or consultants of any affiliated entity.
Share Reserve
Pursuant to the amendment adopted on August 12, 2020, the number of shares of our Common Stock authorized to be issued under the 2018 Plan is equal to 18% of the number of issued and outstanding shares of Common Stock of the Company as of the applicable date of issuance. As of November 20, 2020, 3,429,833 shares of our Common Stock were reserved for issuance pursuant to awards under the 2018 Plan. In general, shares subject to awards granted under the 2018 Plan that are not issued or that are returned to us, for example, because the award is forfeited, the shares are retained by us in satisfaction of amounts owed with respect to an award or the shares are surrendered in payment of an exercise or purchase price or tax withholding, will again become available for awards under the 2018 Plan.
Administration
Our Board of Directors or a committee of our Board of Directors will administer the 2018 Plan. The administrator has the power to determine when awards will be granted, which employees, directors or consultants will receive awards, the terms of the awards, including the number of shares subject to each award and the vesting schedule of the awards, and to interpret the terms of the 2018 Plan and the award agreements. The administrator also has the authority to reduce the exercise prices of outstanding stock options and the base appreciation amount of any stock appreciation right if the exercise price or base appreciation amount exceeds the fair market value of the underlying shares, and to cancel such options and stock appreciation rights in exchange for new awards, in each case without stockholder approval.
Stock Options
The 2018 Plan allows for the grant of incentive stock options that qualify under Section 422 of the Code and non-qualified stock options. The exercise price of all options granted under the 2018 Plan must at least be equal to the fair market value of our Common Stock on the date of grant. The term of an option may not exceed 10 years, except that with respect to any employee who owns more than 10% of the voting power of all classes of our outstanding stock or any parent or subsidiary corporation as of the grant date, the term must not exceed five years, and the exercise price must equal at least 110% of the fair market value on the grant date.
67
After the continuous service of an option recipient terminates, the recipient’s options may be exercised, to the extent vested, for the period of time specified in the option agreement. However, an option may not be exercised later than the expiration of its term.
Stock Appreciation Rights
The 2018 Plan allows for the grant of stock appreciation rights. Stock appreciation rights allow the recipient to receive the appreciation in the fair market value of our Common Stock between the date of grant and the exercise date. The administrator will determine the terms of stock appreciation rights, including when such rights become exercisable and whether to pay the increased appreciation in cash or with shares of our Common Stock, or a combination thereof, except that the base appreciation amount used to determine the cash or shares to be issued pursuant to the exercise of a stock appreciation right will be no less than 100% of the fair market value per share on the date of grant. After the continuous service of a recipient of a stock appreciation right terminates, the recipient’s stock appreciation right may be exercised, to the extent vested, only to the extent provided in the stock appreciation right agreement.
Restricted Stock Awards
The 2018 Plan allows for the grant of restricted stock. Restricted stock awards are shares of our Common Stock that vest in accordance with terms and conditions established by the administrator. The administrator will determine the number of shares of restricted stock granted to any employee, director or consultant. The administrator may impose whatever conditions on vesting that it determines to be appropriate. For example, the administrator may set restrictions based on the achievement of specific performance goals or on the continuation of service or employment. Shares of restricted stock that do not vest are subject to repurchase or forfeiture.
Restricted Stock Units
The 2018 Plan allows for the grant of restricted stock units. Restricted stock units are awards that will result in payment to a recipient at the end of a specified period only if the vesting criteria established by the administrator are achieved or the award otherwise vests. The administrator may impose whatever conditions to vesting, or restrictions and conditions to payment, that it determines to be appropriate. The administrator may set restrictions based on the achievement of specific performance goals or on the continuation of service or employment. The administrator may specify in an award agreement that earned restricted stock units may be settled in shares of our Common Stock, other securities, cash or a combination thereof.
Other Awards
The 2018 Plan also allows for the grant of cash or stock-based awards that may or may not be subject to restrictions.
Terms of Awards
The administrator of the 2018 Plan determines the provisions, terms and conditions of each award, including vesting schedules, forfeiture provisions, form of payment (cash, shares, or other consideration) upon settlement of the award, payment contingencies and satisfaction of any performance criteria.
Performance Criteria
The 2018 Plan includes the following performance criteria that may be considered, individually or in combination, by the administrator: (i) increase in share price; (ii) earnings per share; (iii) total stockholder return, (iv) return on equity, (v) return on assets, (vi) return on investment; (vii) net operating income, (viii) cash flow, (ix) revenue; (x) economic value added, (xi) personal management objectives; or (xii) other measures of performance selected by the administrator.
68
Transferability of Awards
The 2018 Plan allows for the transfer of awards under the 2018 Plan only (i) by will, (ii) by the laws of descent and distribution and (iii) for awards other than incentive stock options, to the extent and in the manner authorized by the administrator. Only the recipient of an incentive stock option may exercise such award during his or her lifetime.
Certain Adjustments
In the event of certain changes in our capitalization, to prevent enlargement of the benefits or potential benefits available under the 2018 Plan, the administrator will make adjustments to one or more of the number of shares that are covered by outstanding awards, the exercise or purchase price of outstanding awards, the numerical share limits contained in the 2018 Plan and any other terms that the administrator determines require adjustment.
Changes in Control
The 2018 Plan provides that in the event of a corporate transaction, as such term is defined in the 2018 Plan, each outstanding award, to the extent not assumed or replaced, will automatically vest and become exercisable or be released from restrictions on transfer or forfeiture rights. To the extent outstanding awards are assumed or replaced in the event of a corporate transaction, each award will automatically vest and become exercisable or be released from restrictions on transfer or forfeiture rights if the holder’s employment is terminated without cause or for good reason (as such terms are defined in the 2018 Plan) within 12 months after the corporate transaction. In the event of a change in control, each award will automatically vest and become exercisable or be released from restrictions on transfer or forfeiture rights if the holder’s employment is terminated without cause or for good reason (as such terms are defined in the 2018 Plan) within 12 months after the change in control.
Plan Amendments and Termination
The 2018 Plan will automatically terminate 10 years following the date it became effective, unless we terminate it sooner. In addition, our Board of Directors has the authority to amend, suspend or terminate the 2018 Plan, subject to stockholder approval in the event such approval is required by law provided such action does not adversely affect the rights under any outstanding award.
Transfer Agent and Registrar
The transfer agent and registrar for our Common Stock is Computershare Trust Company, N.A. which agent is registered pursuant to Section 17A(c) of the Exchange Act.
Penny Stock Regulation
The SEC has adopted regulations which generally define “penny stock” to be any equity security that has a market price of less than $5.00 per share or an exercise price of less than $5.00 per share. Such securities are subject to rules that impose additional sales practice requirements on broker-dealers who sell them. For transactions covered by these rules, the broker-dealer must make a special suitability determination for the purchaser of such securities and have received the purchaser’s written consent to the transaction prior to the purchase. Additionally, for any transaction involving a penny stock, unless exempt, the rules require the delivery, prior to the transaction, of a disclosure schedule prepared by the SEC relating to the penny stock market. The broker-dealer also must disclose the commissions payable to both the broker-dealer and the registered representative, current quotations for the securities and, if the broker-dealer is the sole market-maker, the broker-dealer must disclose this fact and the broker-dealer’s presumed control over the market. Finally, among other requirements, monthly statements must be sent disclosing recent price information for the penny stock held in the account and information on the limited market in penny stocks. As our Common Stock immediately following this Offering may be subject to such penny stock rules, purchasers in this Offering will in all likelihood find it more difficult to sell their Common Stock shares in the secondary market.
69
The Company, which currently has approximately 8,300 stockholders, is an alternative reporting company under Regulation A+, Tier 2 of the Securities Act. There is no public trading market for the Common Stock shares of the Company. The Company may, as an alternative reporting company, qualify its Common Stock shares for quotation on the NASDAQ or OTCBB (the Over the Counter Bulletin Board) or other secondary market for which the Company’s Common Stock may then qualify, upon raising a minimum of $5 million and progressing further in its clinical trials, in the discretion of the Company’s Board of Directors. As of the date of this Offering Circular, the Board of Directors has not taken any action to list the Company’s Common Stock on the NASDAQ, OTCBB or any other market or exchange. (See Risk Factors starting on page 5.)
We plan to retain any earnings for the foreseeable future for our operations. We have never paid any dividends on our Common Stock and do not anticipate paying any cash dividends in the foreseeable future. Any future determination to pay cash dividends will be at the sole discretion of our Board and will depend on our financial condition, operating results, capital requirements and such other factors as our Board deems relevant.
WHERE YOU CAN FIND MORE INFORMATION
We have filed with the SEC a Regulation A+ Offering Statement on Form 1-A under the Securities Act with respect to the shares of Common Stock offered hereby. This Offering Circular, which constitutes a part of the Offering Statement, does not contain all of the information set forth in the Offering Statement or the exhibits and schedules filed therewith. For further information about us and the Common Stock offered hereby, we refer you to the Offering Statement and the exhibits and schedules filed therewith. Statements contained in this Offering Circular regarding the contents of any contract or other document that is filed as an exhibit to the Offering Statement are not necessarily complete, and each such statement is qualified in all respects by reference to the full text of such contract or other document filed as an exhibit to the Offering Statement. The SEC maintains an Internet website that contains reports and other information about issuers, including us, that file electronically with the SEC. The address of this site is www.sec.gov.
70
EMERALD HEALTH PHARMACEUTICALS INC.
As of and for the Years Ended December 31, 2019 and 2018
As of and for the Six Months Ended June 30, 2020 and 2019
F-1
To the Board of Directors of
Emerald Health Pharmaceuticals Inc.
5910 Pacific Center Blvd, Suite 300
San Diego, CA 92121
We have audited the accompanying consolidated financial statements of Emerald Health Pharmaceuticals Inc. and its subsidiaries (the "Company"), which comprise the consolidated balance sheets as of December 31, 2019 and 2018, and the related consolidated statements of operations and comprehensive loss, stockholders' deficit, and cash flows for the years then ended, and the related notes to the consolidated financial statements.
Management’s Responsibility for the Consolidated Financial Statements
Management is responsible for the preparation and fair presentation of these consolidated financial statements in accordance with accounting principles generally accepted in the United States of America; this includes the design, implementation, and maintenance of internal control relevant to the preparation and fair presentation of consolidated financial statements that are free from material misstatement, whether due to fraud or error.
Auditors’ Responsibility
Our responsibility is to express an opinion on these consolidated financial statements based on our audits. We conducted our audits in accordance with auditing standards generally accepted in the United States of America. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free from material misstatement.
An audit involves performing procedures to obtain audit evidence about the amounts and disclosures in the consolidated financial statements. The procedures selected depend on the auditor’s judgment, including the assessment of the risks of material misstatement of the consolidated financial statements, whether due to fraud or error. In making those risk assessments, the auditor considers internal control relevant to the Company's preparation and fair presentation of the consolidated financial statements in order to design audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company's internal control. Accordingly, we express no such opinion. An audit also includes evaluating the appropriateness of accounting policies used and the reasonableness of significant accounting estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements.
We believe that the audit evidence we have obtained is sufficient and appropriate to provide a basis for our audit opinion.
Opinion
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of Emerald Health Pharmaceuticals Inc. and its subsidiaries as of December 31, 2019 and 2018, and the results of their operations and their cash flows for the years then ended in accordance with accounting principles generally accepted in the United States of America.
Emphasis of Matter Regarding Going Concern
The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 1 to the consolidated financial statements, the Company has suffered recurring losses from operations and cash outflows from operations since inception and has an accumulated deficit and negative working capital and has stated that substantial doubt exists about its ability to continue as a going concern. Management’s evaluation of the events and conditions and management’s plans regarding these matters are also described in Note 1. The consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty. Our opinion is not modified with respect to this matter.
/s/ DELOITTE & TOUCHE LLP
San Diego, California
April 28, 2020
F-2
Emerald Health Pharmaceuticals Inc.
Consolidated Balance Sheets
| December 31, | ||||||||
| 2019 | 2018 | |||||||
| Assets | ||||||||
| Current assets: | ||||||||
| Cash and cash equivalents | $ | 983,261 | $ | 137,706 | ||||
| Deferred stock issuance costs | - | 306,560 | ||||||
| Incentive and other tax receivables | 1,420,107 | 416,106 | ||||||
| Other current assets | 548,280 | 292,211 | ||||||
| Total current assets | 2,951,648 | 1,152,583 | ||||||
| Property plant and equipment, net | 52,458 | 81,003 | ||||||
| Total assets | $ | 3,004,106 | $ | 1,233,586 | ||||
| Liabilities and stockholders’ deficit | ||||||||
| Current liabilities: | ||||||||
| Accounts payable | $ | 1,945,549 | $ | 707,815 | ||||
| Accrued expenses | 1,977,627 | 990,273 | ||||||
| Accrued interest payable | 505,289 | 700,647 | ||||||
| Related party loan | 3,750,701 | 9,576,321 | ||||||
| Total current liabilities | 8,179,166 | 11,975,056 | ||||||
| Commitments and contingencies (Note 6) | ||||||||
| Stockholders’ deficit: | ||||||||
| Common stock, $0.0001 par value; 100,000,000 shares authorized, 14,492,085 shares issued and 14,417,085 shares outstanding at December 31, 2019; 100,000,000 shares authorized, 10,000,000 shares issued and 9,925,000 shares outstanding at December 31, 2018 | 1,449 | 1,000 | ||||||
| Additional paid-in-capital | 22,546,309 | 574,522 | ||||||
| Accumulated other comprehensive loss | (38,724 | ) | (1,808 | ) | ||||
| Accumulated deficit | (27,684,086 | ) | (11,315,176 | ) | ||||
| Treasury stock, at cost (common stock: 75,000 at December 31, 2019 and December 31, 2018) | (8 | ) | (8 | ) | ||||
| Total stockholders’ deficit | (5,175,060 | ) | (10,741,470 | ) | ||||
| Total liabilities and stockholders’ deficit | $ | 3,004,106 | $ | 1,233,586 | ||||
See accompanying Notes to Consolidated Financial Statements.
F-3
Emerald Health Pharmaceuticals Inc.
Consolidated Statements of Operations and Comprehensive Loss
| Years Ended December 31, | ||||||||
| 2019 | 2018 | |||||||
| Revenue | $ | - | $ | - | ||||
| Operating expenses: | ||||||||
| Research and development | 9,930,943 | 5,672,893 | ||||||
| General and administrative | 4,194,301 | 2,004,572 | ||||||
| Total operating expenses | 14,125,244 | 7,677,465 | ||||||
| Operating loss | (14,125,244 | ) | (7,677,465 | ) | ||||
| Other (income)/expenses: | ||||||||
| Related party interest income | (178,933 | ) | - | |||||
| Related party interest expense | 2,389,317 | 667,155 | ||||||
| Foreign exchange loss | 33,282 | 54,473 | ||||||
| Net loss | (16,368,910 | ) | (8,399,093 | ) | ||||
| Other comprehensive income (loss): | ||||||||
| Foreign currency translation adjustments | (36,916 | ) | (1,163 | ) | ||||
| Comprehensive loss | $ | (16,405,826 | ) | $ | (8,400,256 | ) | ||
| Net loss per share, basic and diluted | $ | (1.27 | ) | $ | (0.84 | ) | ||
| Weighted-average common shares outstanding, basic and diluted | 12,936,182 | 9,987,466 | ||||||
See accompanying Notes to Consolidated Financial Statements.
F-4
Emerald Health Pharmaceuticals Inc.
Consolidated Statements of Stockholders’ Deficit
| Common Stock Outstanding | Additional Paid in | Note Receivable from | Accumulated Other Comprehensive | Accumulated | Treasury Stock | Total Stockholders' | ||||||||||||||||||||||||||||||
| Shares | Amount | Capital | Stockholder | Loss | Deficit | Shares | Amount | Deficit | ||||||||||||||||||||||||||||
| Balance at December 31, 2017 | 10,000,000 | $ | 1,000 | $ | - | $ | - | $ | (645 | ) | $ | (2,916,083 | ) | - | $ | - | $ | (2,915,728 | ) | |||||||||||||||||
| Repurchase of common stock from founder | (75,000 | ) | 75,000 | (8 | ) | (8 | ) | |||||||||||||||||||||||||||||
| Stock-based compensation expense | 574,522 | 574,522 | ||||||||||||||||||||||||||||||||||
| Net loss and comprehensive loss | (1,163 | ) | (8,399,093 | ) | (8,400,256 | ) | ||||||||||||||||||||||||||||||
| Balance at December 31, 2018 | 9,925,000 | $ | 1,000 | $ | 574,522 | $ | - | $ | (1,808 | ) | $ | (11,315,176 | ) | 75,000 | $ | (8 | ) | $ | (10,741,470 | ) | ||||||||||||||||
| Issuance of common stock under Regulation A offering, net of issuance costs | 3,176,385 | 317 | 15,738,585 | 15,738,902 | ||||||||||||||||||||||||||||||||
| Issuance of common stock under Regulation S offering, net of issuance costs | 65,700 | 7 | 276,717 | 276,724 | ||||||||||||||||||||||||||||||||
| Issuance of common stock upon conversion of related party loan | 1,250,000 | 125 | 2,499,875 | 2,500,000 | ||||||||||||||||||||||||||||||||
| Accretion of beneficial conversion feature on related party loan | 1,360,840 | 1,360,840 | ||||||||||||||||||||||||||||||||||
| Funds advanced under note receivable from stockholder | (5,000,000 | ) | (5,000,000 | ) | ||||||||||||||||||||||||||||||||
| Discharge between related party loan and note receivable from stockholder | 5,000,000 | 5,000,000 | ||||||||||||||||||||||||||||||||||
| Stock-based compensation expense | 2,095,770 | 2,095,770 | ||||||||||||||||||||||||||||||||||
| Net loss and comprehensive loss | (36,916 | ) | (16,368,910 | ) | (16,405,826 | ) | ||||||||||||||||||||||||||||||
| Balance at December 31, 2019 | 14,417,085 | $ | 1,449 | $ | 22,546,309 | $ | - | $ | (38,724 | ) | $ | (27,684,086 | ) | 75,000 | $ | (8 | ) | $ | (5,175,060 | ) | ||||||||||||||||
See accompanying Notes to Consolidated Financial Statement.
F-5
Emerald Health Pharmaceuticals Inc.
Consolidated Statements of Cash Flows
| Years Ended December 31, | ||||||||
| 2019 | 2018 | |||||||
| Operating activities | ||||||||
| Net loss | $ | (16,368,910 | ) | $ | (8,399,093 | ) | ||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||
| Depreciation | 28,545 | 16,310 | ||||||
| Stock-based compensation | 2,095,770 | 574,522 | ||||||
| Accretion of beneficial conversion feature on related party loan | 1,360,840 | - | ||||||
| Changes in operating assets and liabilities: | ||||||||
| Incentive and other tax receivables | (1,004,001 | ) | (416,106 | ) | ||||
| Other current assets | (256,069 | ) | (277,851 | ) | ||||
| Accounts payable | 1,262,919 | (254,230 | ) | |||||
| Accrued expenses | 978,482 | 234,976 | ||||||
| Accrued interest payable | (195,358 | ) | 666,490 | |||||
| Net cash used in operating activities | (12,097,782 | ) | (7,854,982 | ) | ||||
| Investing activities | ||||||||
| Purchases of property and equipment | - | (97,313 | ) | |||||
| Net cash used in investing activities | - | (97,313 | ) | |||||
| Financing activities | ||||||||
| Issuance of common stock | 16,846,973 | - | ||||||
| Repurchase of common stock from founder | - | (8 | ) | |||||
| Funds received under related party loan | 1,674,380 | 8,311,675 | ||||||
| Funds advanced under note receivable from stockholder | (5,000,000 | ) | - | |||||
| Stock issuance costs | (541,100 | ) | (273,292 | ) | ||||
| Net cash provided by financing activities | 12,980,253 | 8,038,375 | ||||||
| Effect of exchange rate changes on cash | (36,916 | ) | (1,163 | ) | ||||
| Net increase in cash and cash equivalents | 845,555 | 84,917 | ||||||
| Cash and cash equivalents at beginning of period | 137,706 | 52,789 | ||||||
| Cash and cash equivalents at end of period | $ | 983,261 | $ | 137,706 | ||||
| Supplemental disclosure of cash flow information: | ||||||||
| Interest paid to related party | $ | 1,044,901 | $ | - | ||||
| Non-cash investing and financing activities: | ||||||||
| Conversion of related party loan to common stock | $ | 2,500,000 | $ | - | ||||
| Discharge between related party loan and note receivable from stockholder | $ | 5,000,000 | $ | - | ||||
| Deferred stock issuance costs in accounts payable and accrued expenses | $ | 16,955 | $ | 33,268 | ||||
See accompanying Notes to Consolidated Financial Statements.
F-6
Emerald Health Pharmaceuticals Inc.
Notes to Consolidated Financial Statements
1. Description of Business and Going Concern
Emerald Health Pharmaceuticals Inc. (EHP, or the Company) was incorporated in the state of Delaware in March 2017. The Company is a biotechnology/pharmaceutical company, formed to acquire, discover, develop and commercialize drug product candidates containing novel, patented molecules chemically derived from non-psychoactive cannabinoids (molecules found in cannabis). EHP is focused on developing product candidates to treat diseases with unmet medical needs primarily in inflammatory, autoimmune, metabolic, neurodegenerative and fibrotic diseases. The Company is currently developing two initial product candidates that together target four initial diseases, multiple sclerosis (MS), scleroderma/systemic sclerosis (SSc), Parkinson’s disease (PD) and Huntington’s disease (HD).
The Company acquired certain intellectual property from Emerald Health Biotechnology España, S.L.U. (EHBE), formerly known as VivaCell Biotechnology España S.L. (VivaCell). During the year ended December 31, 2018, EHBE became a wholly owned subsidiary of Emerald Health Research Inc. (EHR) which is a wholly owned subsidiary of Emerald Health Sciences Inc. (EHS). EHS is also the majority stockholder of EHP. EHP has no ownership or voting rights related to EHBE. See Note 7.
The Company is subject to risks common to other life science companies in the development stage including, but not limited to, uncertainty of product development and commercialization, lack of marketing and sales history, development by its competitors of new technological innovations, dependence on key personnel, market acceptance of products, product liability, protection of proprietary technology, ability to raise additional financing, and compliance with FDA and other government regulations. If the Company does not successfully commercialize any product candidates, it will be unable to generate recurring product revenue or achieve profitability.
The Company’s financial statements have been prepared assuming the Company will continue as a going concern, which contemplates the realization of assets and the satisfaction of liabilities in the normal course of business. The Company has experienced losses and recurring cash outflows from operations since inception and has an accumulated deficit of $27,684,086 and negative working capital as of December 31, 2019. The Company has funded operations with capital raised from an ongoing Tier 2 offering (the Offering) pursuant to Regulation A (Regulation A) under the Securities Act of 1933, as amended (the Securities Act), as well as an exempt offshore offering under Regulation S under the Securities Act, along with the proceeds from a revolving loan with its majority stockholder. Amounts advanced under the loan and accrued interest are due upon demand. See Note 3.
The future viability of the Company is largely dependent upon its ability to raise additional capital to finance its operations. Management expects that future sources of funding may include sales of equity, obtaining loans, or other strategic transactions. Although management continues to pursue these plans, there is no assurance that the Company will be successful in obtaining sufficient financing on terms acceptable to the Company to fund continuing operations, if at all. These circumstances raise substantial doubt about the Company’s ability to continue as a going concern. The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
The Company is undertaking a “best efforts” offering of its common stock to raise additional capital. See Note 4.
2. Significant Accounting Policies
Basis of Presentation
The accompanying consolidated financial statements have been prepared in conformity with accounting principles generally accepted in the United States (GAAP).
F-7
Consolidation
The consolidated financial statements include the accounts of the Company and its wholly owned subsidiaries, Emerald Health Pharmaceuticals Australia Pty Ltd. (EHP Australia) and Emerald Health Pharmaceuticals, España Sociedad Limitada (EHP España). EHP Australia’s functional currency, the Australian dollar, is also its reporting currency, and its financial statements are translated to US dollars prior to consolidation. EHP España’s functional currency, the Euro, is also its reporting currency, and its financial statements are translated to US dollars prior to consolidation. All intercompany accounts and transactions have been eliminated in consolidation.
Use of Estimates
The preparation of financial statements in conformity with generally accepted accounting principles requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities at the date of the financial statements and the reported amounts of revenue and expenses during the reporting period. Actual results could differ from those estimates.
Cash and Cash Equivalents
The Company considers all highly liquid investments with an original maturity of three months or less from the date of purchase to be cash equivalents. As of December 31, 2019, the Company’s cash deposits are held in an FDIC-insured financial institution.
Incentive and Tax Receivables
The Company’s subsidiary, EHP Australia, is incorporated in Australia and is eligible to participate in an Australian research and development tax incentive program. As part of this program, EHP Australia is eligible to receive a cash refund from the Australian Taxation Office (ATO) for a percentage (currently 43.5%) of the research and development costs incurred by EHP Australia. The cash refund is available to eligible companies with an annual aggregate revenue of less than $AU20.0 million (Australian Dollars) during the reimbursable period. As of December 31, 2019 and 2018, the Company’s estimate of the amount of cash refunds expected to be received for eligible spending as part of this incentive program was $1.3 million and $0.4 million, respectively, which amounts are included in incentive and other tax receivables. In July 2019, the Company received $0.4 million as a cash refund from the ATO for eligible spending incurred during the year ended December 31, 2018.
In addition, EHP Australia incurs Goods and Services Tax (GST) on services provided by Australian vendors. As an Australian entity, EHP Australia is entitled to a refund of the GST paid. The Company’s estimate of the amount of cash refund expected to be received related to GST incurred as of December 31, 2019 and 2018, was $78,198 and $26,041, respectively, which amounts are included in incentive and other tax receivables.
Property and Equipment
Property and equipment generally consist of computer equipment and software and office furniture and are recorded at cost and depreciated over the estimated useful lives of the assets (generally three to five years) using the straight-line method. Leasehold improvements are stated at cost and are amortized on a straight-line basis over the lesser of the remaining term of the related lease or the estimated useful lives of the assets. Repairs and maintenance costs are charged to expense as incurred and improvements and betterments are capitalized. When assets are retired or otherwise disposed of, the cost and accumulated depreciation are removed from the consolidated balance sheets and any resulting gain or loss is reflected in the consolidated statements of operations in the period realized.
F-8
Impairment of Long-lived Assets
The Company reviews property and equipment for impairment on an annual basis and whenever events or changes in circumstances indicate that the carrying amount of such assets may not be recoverable. An impairment loss would be recognized when estimated future undiscounted cash flows relating to the asset or asset group are less than its carrying amount. An impairment loss is measured as the amount by which the carrying amount of an asset or asset group exceeds its fair value. While the Company’s current and historical operating losses and negative cash flows are possible indicators of impairment, management believes that future cash flows to be generated by these assets support the carrying value of its long-lived assets and, accordingly, did not recognize any impairment losses during the years ended December 31, 2019 and 2018.
Research and Development
Research and development costs are charged to expense as incurred and consist primarily of contract research fees, contract manufacturing costs, consultant fees, preclinical and clinical studies and study related costs, compensation and related benefits, and non-cash stock-based compensation. At the end of each reporting period, the Company compares the payments made to its vendors, clinical research organizations and consultants to the estimated progress towards completion of the related project. Factors that the Company considers in preparing these estimates include the number of patients enrolled in studies, milestones achieved, and other criteria related to the efforts of its vendors. These estimates will be subject to change as additional information becomes available. Depending on the timing of payments to vendors and estimated services provided, the Company will record net prepaid or accrued expenses related to these costs. Research and development expenses are recorded net of expected refunds of eligible research and development costs paid pursuant to the Australian research and development tax incentive program and GST incurred on services provided by Australian vendors.
Income Taxes
The Company has incurred net operating losses from inception through December 31, 2019. Therefore, no United States federal, state, or foreign income taxes are expected to be paid for 2019 or 2018 and no amounts payable have been recorded as of December 31, 2019 and 2018.
Due to the Company’s history of losses since inception, there is not enough evidence at this time to support the conclusion that it will generate future income of a sufficient amount and nature to utilize the benefits of the Company’s net deferred tax assets. Accordingly, the Company fully reduced its net deferred tax assets by a valuation allowance, since it has been determined that it is more likely than not that all of the deferred tax assets will not be realized.
The Tax Reform Act of 1986 contains provisions which limit the ability to utilize the net operating loss carryforwards in the case of certain events including significant changes in ownership interests. If the Company’s net operating loss carryforwards are limited, and the Company has taxable income which exceeds the permissible yearly net operating loss carryforwards, the Company would incur a federal income tax liability even though net operating loss carryforwards would be available in future years.
F-9
Fair Value Measurements
The Company does not have any financial assets and liabilities reported at fair value on a recurring basis. The carrying amounts of the Company’s financial instruments including cash and cash equivalents, deferred stock issuance costs, incentive and tax receivables, other current assets, property plant and equipment, net, accounts payable and accrued expenses approximate fair value due to the short-term nature of those instruments. The Company’s related party loan and the associated accrued interest payable is carried at amortized cost. Due to the related party nature of these advances with the controlling shareholder, management has concluded that its fair value is not reasonably determinable (see Note 3).
The Company determines fair value based upon the exit price that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants, as determined by either the principal market or the most advantageous market. Inputs used in the valuation techniques to derive fair values are classified based on a three-level hierarchy. These levels are:
Level 1—Quoted prices (unadjusted) in active markets that are accessible at the measurement date for assets or liabilities. The fair value hierarchy gives the highest priority to Level 1 inputs.
Level 2—Observable prices that are based on inputs not quoted on active markets but corroborated by market data.
Level 3—Unobservable inputs are used when little or no market data is available. The fair value hierarchy gives the lowest priority to Level 3 inputs.
Business Segments
The Company operates within the United States, Europe, and Australia, in one business segment, which is dedicated to research of drug candidates based on patented synthetic new chemical entities (NCEs) derived from non-psychoactive cannabinoid molecules.
Stock-Based Compensation
The Company accounts for stock option awards in accordance with Financial Accounting Standards Board Accounting Standards Codification (ASC) Topic No. 718, Compensation-Stock Compensation. Under FASB ASC Topic No. 718, compensation expense related to stock-based payments is recorded over the requisite service period based on the grant date fair value of the awards. Compensation previously recorded for unvested stock options that are forfeited is reversed upon forfeiture. The Company uses the Black-Scholes option pricing model for determining the estimated fair value for stock-based awards. The Black-Scholes model requires the use of assumptions which determine the fair value of stock-based awards, including the option’s expected term and the price volatility of the underlying stock. See Note 5.
Net Loss per Share
The Company computes basic net loss per common share by dividing the applicable net loss by the weighted average number of common shares outstanding during the period. Diluted earnings per share is computed using the weighted average number of common shares outstanding during the period, plus additional shares to account for the dilutive effect of potential future issuances of common stock relating to stock options and other potentially dilutive securities using the treasury stock method. For the years ended December 31, 2019 and 2018, 2,080,800 and 2,060,000 options, respectively, were excluded from the computation of diluted earnings per share, as the effect would be anti-dilutive.
Comprehensive Loss
Comprehensive loss includes foreign currency translation adjustments related to the Company’s subsidiaries in Australia and Spain.
F-10
Recently Adopted Accounting Pronouncements
In May 2014, the Financial Accounting Standards Board (FASB) issued Accounting Standard Update (ASU) No. 2014-09, Revenue from Contracts with Customers (Topic 606) (ASU No. 2014-09), which amends the existing accounting standards for revenue recognition. ASU No. 2014-09 is based on principles that govern the recognition of revenue at an amount to which an entity expects to be entitled when products are transferred to customers. The Company adopted ASU No. 2014-09 as of January 1, 2019, using the full retrospective approach, which did not have an impact on the Company’s financial position or results of operations, as the Company is pre-commercial and has not generated any material revenue to date.
As of January 1, 2019, the Company adopted ASU No. 2018-07, Compensation—Stock Compensation (Topic 718), which expands the scope of Topic 718 to include share-based payment transactions for acquiring goods and services from non-employees that are currently covered under Accounting Standards Codification (ASC) 505-50, Equity-Based Payments to Non-employees. As a result of the adoption, the Company no longer records a mark-to-market adjustment at each reporting period for share-based payment transactions for acquiring goods and services from non-employees, which had an immaterial impact on its financial statements.
Recently Issued Accounting Pronouncements
In February 2016, the FASB issued ASU No. 2016-02, Leases (Topic 842) (ASU No. 2016-02), which changes the presentation of assets and liabilities relating to leases. The core principle of ASU No. 2016-02 is that a lessee should recognize the assets and liabilities that arise from leases. All leases create an asset and a liability for the lessee in accordance with FASB Concepts Statement No. 6, Elements of Financial Statements, and, therefore, recognition of those lease assets and lease liabilities represents an improvement over previous GAAP, which did not require lease assets and lease liabilities to be recognized for most leases.
In November 2019, the FASB issued ASU No. 2019-10, Financial Instruments - Credit Losses (Topic 326), Derivatives and Hedging (Topic 815), and Leases (Topic 842) (ASU No. 2019-10), which deferred the effective date of ASU No. 2016-02 for the Company from January 1, 2020 to January 1, 2021. The Company is currently evaluating the impact of this new standard on its financial statements.
In October 2018, the FASB issued ASU No. 2018-17, Consolidation (Topic 810) (ASU No. 2018-17), which adds an elective private-company scope exception to the variable interest entity (VIE) guidance for entities under common control. ASU No. 2018-17 will be effective for the Company beginning January 1, 2021, with early adoption permitted. The Company does not expect this new standard to have a material impact on its financial statements.
3. Related Party Transactions
Related Party Loan and Beneficial Conversion Feature
Since inception, the Company has received advances from EHS to fund its operations. In September 2017, the Company and EHS entered a revolving loan agreement (Related Party Loan), which was amended in January 2018. Borrowings under the loan, which EHP may draw down from time to time in one or more advances, will be evidenced by a demand grid promissory note (the Note). The Note will be revised to reflect the aggregate principal amount of the loan outstanding as of the date of each advance or repayment. In November 2019, the Related Party Loan was further amended to reduce the interest rate from 12% to 10%, compounded semiannually. The loan may be repaid by the Company or converted by EHS into shares of EHP at $2.00 per share or at a price to be equally agreed to between EHS and the Company (Conversion Feature). The Note is payable upon demand and has no expiration date. As of December 31, 2019 and 2018, $3,750,701 and $9,576,321, respectively, of principal, and $505,289 and $700,647, respectively, of accrued interest is due to EHS under the Related Party Loan.
In April 2019, the Company received written notice of demand (Notice) from EHS for payment of all accrued interest on the Related Party Loan as of March 31, 2019, which resulted in a cash payment of $1,044,901 to EHS. Also in April 2019, the Company received a second Notice from EHS that called for the following, upon qualification by the SEC of the Company’s Form 1-A Post-Qualification Offering Circular Amendment: (1) repayment of $2,000,000 of the unpaid principal balance under the loan, and (2) the conversion of an additional $2,500,000 of the unpaid principal balance under the loan at a conversion price of $2.00 per share. The Company’s Form 1-A Post-Qualification Offering Circular Amendment was qualified by the SEC on June 7, 2019 and the repayment of the $2,000,000 of unpaid principal was transacted as a cashless discharge and offset between the Related Party Loan and the Related Party Note Receivable (as defined below). Concurrently, 1,250,000 shares of EHP common stock were issued to EHS at a conversion price of $2.00 per share, further reducing the principal balance of the Related Party Loan by $2,500,000.
F-11
In November 2019, the Company received written notice of demand from EHS for payment of $3,000,000 of unpaid principal balance and $178,933 of accrued interest on the Related Party Loan, which was transacted as a cashless discharge and offset between the Related Party Loan and the remaining unpaid principal and accrued interest balances under the Related Party Note Receivable (as defined below) as of November 15, 2019.
The Conversion Feature of the loan agreement is not considered an embedded derivative under FASB Accounting Standards Codification (ASC) Topic 815, Derivatives and Hedging, since there are no provisions for net settlement nor is there a means for EHS to receive an asset that puts EHS in a position not substantially different from net settlement. The Company recorded a debt discount on the Related Party Loan of $1.4 million related to the beneficial conversion feature on advances under the loan during the year ended December 31, 2019. Subsequent to the recognition of the discount, due to the on-demand nature of the loan, the Company recognized $1.4 million in accretion of the discount which is included in related party interest expense.
Related Party Note Receivable
In May 2019, the Company’s Board of Directors authorized a funding arrangement with EHS (Related Party Note Receivable), which was amended in August 2019 and September 2019 to extend the repayment dates, whereby EHP may advance funds to EHS in the form of interest bearing (12%) short term notes, up to an aggregate principal amount of $6,000,000 under a Promissory Note between EHS and EHP (the Promissory Note). Advances under the Promissory Note were originally due for repayment with accrued and unpaid interest three months from the date of the advance. A total of $5,000,000 was advanced and $178,933 accrued as interest receivable under the Related Party Note Receivable, all of which was offset through cashless discharges against the unpaid principal and accrued interest payable balances, respectively, under the existing Related Party Loan with EHS. As of December 31, 2019, there are no outstanding principal or accrued interest receivable balances remaining under the Related Party Note Receivable, and all principal advances and related discharges have been recorded as equity transactions.
Shared Services with EHS and Related Entities
In June 2019, the Company entered into an Independent Contractor Agreement (the Independent Contractor Agreement) effective April 1, 2019, with EHS, pursuant to which EHS agreed to provide such services as are mutually agreed between the Company and EHS, including reimbursements for reasonable expenses incurred in the performance of the Independent Contractor Agreement. These services included, but were not limited to, corporate advisory services and technical expertise in the areas of business development, marketing, investor relations, information technology and product development. The Independent Contractor Agreement had an initial term of ten years. On November 15, 2019, the Board of Directors approved the termination of this agreement, effective as of December 31, 2019. During the year ended December 31, 2019, the Company recorded expenses totaling $339,627 for such services performed by EHS on behalf of the Company, $37,616 of which is recorded in related party liabilities as of December 31, 2019. See Note 8.
During the year ended December 31, 2018, the Company recorded $67,031, for certain general and administrative and research and development costs incurred by EHS on behalf of the Company.
The Company allocates certain operating expenses to entities which are subsidiaries of EHS for their share of facilities and office expenses. During the years ended December 31, 2019 and 2018, these allocations totaled $183,091 and $214,903, respectively, of which $37,260 is recorded in related party receivables as of December 31, 2019. See Note 8.
F-12
Board Observer Agreement
On November 15, 2019, Dr. Avtar Dhillon resigned as Chairman of the Board of Directors. The Company and EHS concurrently entered into a Board Observer Agreement, whereby the Company granted to EHS the right to designate an observer on the Board for so long as EHS maintains ownership of any securities of the Company. Dr. Avtar Dhillon was appointed as the initial Board Observer pursuant to the Board Observer Agreement.
Dr. Avtar Dhillon Independent Contractor Services Agreement
On December 5, 2019, following Dr. Avtar Dhillon’s resignation on November 15, 2019 from the Board of Directors, the Board approved an Independent Contractor Services Agreement, effective as of December 1, 2019, between the Company and Dr. Dhillon, pursuant to which Dr. Dhillon will provide ongoing corporate finance and strategic business advisory services to the Company. In exchange for his services, upon the Company completing a material financing, Dr. Dhillon will receive a monthly fee of $10,000, accruing from the effective date. The Board will review the monthly rate paid to Dr. Dhillon within 90 days of the end of each fiscal year. The Independent Contractor Services Agreement has an initial term of one year and will renew automatically thereafter unless terminated earlier by either party. The Independent Contractor Services Agreement may be terminated by either party for cause upon written notice to the other party if the other party defaults in the performance of the agreement in any material respect or materially breaches the terms of the agreement, or without cause upon 30 days’ prior written notice to the other party. As of December 31, 2019, accrued expenses include $10,000 due and payable to Dr. Dhillon under this agreement, upon completion of a material financing, as defined by the Board of Directors.
Subsidiary Agreements
In April 2018, the Company’s subsidiary, EHP Australia, executed three operating agreements – a Service Agreement, a Funding Agreement, and an IP License Agreement (Original IP License Agreement).
Under the Funding Agreement, which was amended and restated in November 2019, EHP Australia may borrow from the Company up to $AU5,000,000 in total, and $AU2,000,000 per year, in one or more advances, which will be evidenced by a drawdown notice. The loan bears interest at a rate relating to ‘inbound loans’ specified by the Australian Taxation Office from time to time, which was 3.76% per annum effective beginning January 1, 2019. The loan expires in April 2023. As of December 31, 2019, the outstanding unpaid principal balance of the loan was approximately $AU2.0 million, which amount is eliminated in consolidation.
In March 2020, the Service Agreement and Original IP License Agreement were terminated, retroactively effective to April 5, 2018 and concurrently, a new IP License Agreement (New IP License Agreement) was entered into between EHP and EHP Australia. Pursuant to the New IP License Agreement, the Company granted to EHP Australia a non-transferable license to use certain specified intellectual property of the Company. EHP Australia will pay the Company an arm’s length license fee, calculated in accordance with Australian Tax Office and internationally accepted transfer pricing principles, which will be eliminated upon consolidation. The IP License may be terminated at any time by either party.
4. Common Stock
On March 2, 2017, the Company issued 9,000,000 shares of common stock at $0.0001 per share to EHS for proceeds of $900. An additional 1,000,000 shares were issued to the founders of the Company for total proceeds of $100. The shares issued to founders vested 25% on the date of issuance and vest 25% annually thereafter until fully vested. Until the shares of common stock vest, the founders may not sell or transfer the unvested shares of common stock. In the event of the voluntary or involuntary termination of any of the founders, as an employee or director of the Company for any reason, the Company shall have the option to repurchase all or any portion of the shares of common stock for the same consideration which was originally paid by the founders. In October 2018, the Company exercised its option to repurchase 75,000 unvested shares from a founding member, which are currently held by the Company as treasury stock.
In January 2018, the Company filed a Certificate of Amendment of the Certificate of Incorporation which increased the number of authorized shares that the Company can issue from 20,000,000 to 100,000,000 shares of common stock with a par value of $0.0001 per share.
F-13
The Company’s initial Offering Statement on Form 1-A was qualified by the SEC in March 2018 and its Form 1-A Post-Qualification Offering Circular Amendment was qualified by the SEC on June 7, 2019. During the year ended December 31, 2019, the Company sold 3,176,385 shares of common stock under the Offering, for gross proceeds of $16.5 million, less issuance costs of $0.8 million.
In June 2019, the Company issued 1,250,000 additional shares of common stock to EHS in accordance with a written notice received from EHS in April 2019, for the conversion of $2,500,000 of the unpaid principal balance under the Related Party Loan at a conversion price of $2.00 per share. See Note 3.
In June 2019, the Company entered into a Broker-Dealer Agreement with Dalmore Group, LLC (Dalmore), a broker-dealer registered with the SEC and a member of FINRA, to perform administrative, compliance and placement agent related functions in connection with the Offering. The Company has agreed to pay Dalmore a 1.0% commission on the sale of common stock under the Offering, commencing with sales following regulatory approval by FINRA, which occurred on July 25, 2019. In addition, the Company paid Dalmore $28,000 in one-time set up fees, consisting of a $20,000 agreement fee and $8,000 for fees paid to FINRA. As of December 31, 2019, the Company has paid $16,568 and has accrued an additional $8,872 to be paid to Dalmore related to commission on the sale of common stock under the Offering.
In July 2019, the Company entered into an Escrow Services Agreement with Prime Trust, LLC. Under this agreement, the proceeds received from the Offering are deposited into an escrow account prior to distribution to the Company.
During the year ended December 31, 2019, the Company also sold 65,700 shares of common stock for proceeds of $328,500, less issuance costs of $51,776, in an exempt offshore offering under Regulation S under the Securities Act.
5. Equity Incentive Plan
In January 2018, the Company adopted the 2018 Equity Incentive Plan, which was amended on December 13, 2018 (the Plan). There are 2,200,000 shares of common stock reserved for issuance under the Plan. The Plan provides incentives to eligible employees, consultants, officers, and directors in the form of incentive stock options and non-qualified stock options, stock appreciation rights, restricted stock, restricted stock units and other rights or benefits. Recipients of stock options are eligible to purchase shares of the Company’s common stock at an exercise price equal to no less than the estimated fair market value of such stock on the date of grant. The maximum term of options granted under the Plan is ten years. Vesting schedules are determined by the Board of Directors. As of December 31, 2019, there were 119,200 shares available to grant under the Plan.
The following table summarizes stock-based compensation expense related to stock options granted to employees and non-employees included in the consolidated statements of operations as follows:
| Years Ended December 31, | ||||||||
| 2019 | 2018 | |||||||
| Research and development | $ | 227,273 | $ | 138,831 | ||||
| General and administrative | 1,868,497 | 435,691 | ||||||
| Total | $ | 2,095,770 | $ | 574,522 | ||||
Stock-based compensation for the years ended December 31, 2019 and 2018, includes expense of $1,651,345 and $208,863, respectively, related to option grants issued to non-employees.
F-14
The following table summarizes stock option activity:
| Options | Weighted Average Exercise Price | Weighted Average Remaining Contractual Term (in Years) | Aggregate Intrinsic Value (1) | |||||||||||||
| Outstanding at December 31, 2018 | 2,060,000 | $ | 2.85 | |||||||||||||
| Granted | 35,000 | $ | 6.00 | |||||||||||||
| Exercised | - | |||||||||||||||
| Forfeited/Expired | (14,200 | ) | $ | 3.68 | ||||||||||||
| Outstanding at December 31, 2019 | 2,080,800 | $ | 2.89 | 8.87 | $ | 6,465,550 | ||||||||||
| Options Vested and Exercisable as of December 31, 2019 | 1,387,800 | $ | 2.69 | 8.91 | $ | 4,591,550 | ||||||||||
| (1) | As of December 31, 2019, the fair value of the Company's common shares as determined by its board of directors was $6.00 per share. |
The following table summarizes certain information regarding stock options for the years ended December 31, 2019 and 2018:
| 2019 | 2018 | |||||||
| Weighted average grant date fair value per share of options granted during the period | $ | 4.40 | $ | 1.67 | ||||
| Fair value per share of options vested during the period | $ | 1.81 | $ | 1.85 | ||||
| Cash received from options exercised during the period | $ | - | $ | - | ||||
| Intrinsic value of options exercised during the period | $ | - | $ | - | ||||
As of December 31, 2019, unrecognized stock-based compensation expense for employee and non-employee stock options was approximately $1.2 million, which the Company expects to recognize over a weighted-average remaining period of 2.0 years, assuming all unvested options become fully vested.
The Company uses a Black-Scholes option-pricing model to value the Company’s option awards. Using this option-pricing model, the fair value of each employee and non-employee award is estimated on the grant date. The fair value is expensed on a straight-line basis over the vesting period. In general, the option awards vest partially upfront and then pro-rata annually thereafter. The expected volatility assumption is based on the volatility of the share price of comparable public companies. The expected life is determined using the “simplified method” permitted by Staff Accounting Bulletin Number 107 and 110 (the midpoint between the term of the agreement and the weighted average vesting term). The risk-free interest rate is based on the implied yield on a US Treasury security at a constant maturity with a remaining term equal to the expected term of the option granted. The dividend yield is zero, as the Company has never declared a cash dividend.
The fair value of the stock options granted was estimated on the date of grant using the Black-Scholes option pricing model with the following weighted-average assumptions for the periods indicated:
| Years Ended December 31, | ||||||||
| 2019 | 2018 | |||||||
| Expected term (in years) | 5.75 | 5.6 | ||||||
| Stock price volatility | 90 | % | 90 | % | ||||
| Risk-free interest rate | 1.77 | % | 2.68 | % | ||||
| Dividend yield | 0 | % | 0 | % | ||||
F-15
6. Commitments and Contingencies
On May 1, 2018, the Company entered into a two-year non-cancelable building lease for its corporate headquarters in San Diego, California. Effective August 15, 2019, the lease was amended to include additional space at the existing, premises and to extend the term of the original lease through August 31, 2022. Under the lease, the Company pays a base rent of $20,054 per month through April 30, 2020, after which time the base rent will increase by approximately 3% per year. The Company made security deposit payments of $24,805 and $16,698 in 2019 and 2018, respectively, which are recorded within other current assets. Rent expense for the years ended December 31, 2019 and 2018, was $81,968 and $53,782, respectively.
In June 2019, EHS entered into a three-year non-cancelable building lease agreement under which EHP has guaranteed the lease obligations. This lease was canceled by EHS in February 2020.
Future minimum payments under the non-cancelable operating lease as of December 31, 2019 were as follows:
| 2020 | $ | 247,663 | ||
| 2021 | 257,312 | |||
| 2022 | 174,818 | |||
| Total | $ | 679,793 |
In January 2020, EHP Australia entered into a loan agreement with Rocking Horse Nominees Pty Ltd (Rocking Horse), whereby Rocking Horse advanced $AU1.2 million (approximately $0.8 million) to EHP Australia. The loan is secured by the tax incentive refund anticipated to be received during 2020 for eligible spending incurred under the Australian research and development tax incentive program during 2019. The loan had an upfront establishment fee of 1.2% and bears interest at 1.25% per month compounded daily over a minimum term of at least 91 days. The loan and all outstanding interest are due upon the earlier of 1) the occurrence of an event of default, as defined in the loan agreement; 2) the date twenty-one business days after the notice of assessment is issued by the ATO; and 3) November 30, 2020.
7. Intellectual Property Transfer and Research Agreements
In June 2017, upon the execution of the Intellectual Property Transfer Agreement (IPTA), EHP paid EHBE approximately $112,000 for the purchase of three United States patents, two Japanese patents, one European patent and fourteen pending patent applications covering two series of molecules containing derivatives of CBD and CBG. Future payments of up to 2.7 million Euro (approximately $3.0 million, based upon the exchange rate at December 31, 2019) per product are due upon completion of certain development milestones. As further consideration, the Company will pay EHBE a 2.5% royalty on all net revenues of any drug developed from the transferred compounds. As of December 31, 2019, accrued expenses include approximately $0.4 million related to the first milestone payments due to EHBE for the Company’s completion of a Phase 1 clinical study for MS and SSc.
The IPTA is an asset acquisition under FASB ASC Topic 805, Business Combinations, as the intellectual property purchased from EHBE was determined by the Company to be a group of similar identifiable assets. Since the purchase consideration represents in-process research and development with no alternative future use the entire upfront payment was expensed to research and development expense in accordance with FASB ASC Topic 730, Research and Development.
Concurrent with the execution of the IPTA, the Company signed a Research Agreement with EHBE for an initial term of 5 years. Under the terms of the Research Agreement, EHBE is providing research services under the Company’s direction for consideration of cost plus a standard mark-up. Thereafter, the agreement will renew for successive one-year terms and may be terminated by either party on the expiration of the original term or any renewal term by delivering written notice at least 90 days prior to expiration. During the years ended December 31, 2019 and 2018, the Company recorded $152,133 and $359,005, respectively in research and development expense for services performed by EHBE under the Research Agreement. As of December 31, 2019 and 2018, $58,300 and $26,502, respectively, are included in accrued expenses and accounts payable, for amounts due to EHBE under the Research Agreement.
The Company performed a qualitative analysis to determine whether a variable interest in another entity represents a controlling financial interest in a variable interest entity. A controlling financial interest in a variable interest entity is characterized by having both the power to direct the most significant activities of the entity and the obligation to absorb losses or the right to receive benefits of the entity. Since EHP does not have voting control or other forms of control over the operations and decision making at EHBE, the Company determined that it does not have a variable interest in EHBE. This guidance requires on-going reassessments of variable interests based on changes in facts and circumstances. The Company continues to assess its variable interests and has determined that no significant changes have occurred as of April 15, 2020.
F-16
8. Balance Sheet Details
Other current assets consisted of the following:
| December 31, | ||||||||
| 2019 | 2018 | |||||||
| Prepaid contracts and expenses | $ | 468,232 | $ | 248,602 | ||||
| Related party receivables | 37,260 | - | ||||||
| Other | 42,788 | 43,609 | ||||||
| Total | $ | 548,280 | $ | 292,211 | ||||
Property and equipment consisted of the following:
| December 31, | ||||||||
| 2019 | 2018 | |||||||
| Furniture and fixtures | $ | 57,195 | $ | 57,195 | ||||
| Office equipment | 19,480 | 19,480 | ||||||
| Leasehold improvements | 20,638 | 20,638 | ||||||
| Property and equipment, gross | 97,313 | 97,313 | ||||||
| Accumulated depreciation | (44,855 | ) | (16,310 | ) | ||||
| Property and equipment, net | $ | 52,458 | $ | 81,003 | ||||
Depreciation expense for the years ended December 31, 2019 and 2018 was $28,545 and $16,310, respectively.
Accrued expenses are comprised of the following:
| December 31, | ||||||||
| 2019 | 2018 | |||||||
| Research and development liabilities | $ | 355,942 | $ | 269,329 | ||||
| Clinical trial related liabilities | 205,884 | 323,796 | ||||||
| Accrued payroll liabilities | 807,662 | 362,485 | ||||||
| Related party liabilities | 536,505 | - | ||||||
| Other liabilities | 71,634 | 34,663 | ||||||
| Total | $ | 1,977,627 | $ | 990,273 | ||||
9. Defined Contribution Plan
Effective January 1, 2018, the Company adopted a defined contribution savings plan pursuant to Section 401(k) of the Internal Revenue Code. The plan is for the benefit of all qualifying employees and permits voluntary contributions by employees up to 100% of eligible compensation, subject to the Internal Revenue Service imposed maximum limits. The terms of the plan allow for discretionary employer contributions.
On January 1, 2020, the Company commenced a safe harbor contribution of 3% of each eligible employee’s gross earnings, subject to Internal Revenue Service limitations. Employer matching contributions vest immediately.
F-17
10. Income Taxes
The Company has incurred net operating losses from inception through December 31, 2019. Therefore, no United States federal, state, or foreign income taxes are expected to be paid for 2019 or 2018 and no amounts payable have been recorded as of December 31, 2019 and 2018.
The Company’s loss before income taxes for the years ended December 31, 2019 and 2018, respectively, was generated in the following jurisdictions:
| Years Ended December 31, | ||||||||
| (amounts in thousands) | 2019 | 2018 | ||||||
| Domestic | $ | (14,157,884 | ) | $ | (7,618,692 | ) | ||
| Foreign | (2,211,026 | ) | (780,401 | ) | ||||
| Worldwide | $ | (16,368,910 | ) | $ | (8,399,093 | ) | ||
A reconciliation of income tax expense (benefit) to the amount computed by applying the statutory federal income tax rate to the loss from operations is summarized for the years ending December 31, 2019 and 2018, respectively, as follows:
| Years Ended December 31, | ||||||||
| 2019 | 2018 | |||||||
| Expected income tax benefit at US federal statutory rate | $ | (3,437,471 | ) | $ | (1,763,810 | ) | ||
| State income tax benefit, net of federal benefit | (750,286 | ) | (443,786 | ) | ||||
| Tax effect of: | ||||||||
| Change in valuation allowance | 2,874,738 | 1,645,567 | ||||||
| Uncertain tax positions | 731,462 | 422,675 | ||||||
| Australian tax incentive | 470,027 | 151,288 | ||||||
| Stock-based compensation | 110,817 | 29,042 | ||||||
| Other | 713 | (40,976 | ) | |||||
| Provision for income taxes | $ | - | $ | - | ||||
The Company’s net deferred tax assets are comprised of the following as of December 31, 2019 and 2018, respectively:
| As of December 31, | ||||||||
| 2019 | 2018 | |||||||
| Deferred tax assets: | ||||||||
| Tax loss carryforwards | $ | 4,129,060 | $ | 1,924,648 | ||||
| Stock-based compensation | 423,768 | 92,842 | ||||||
| Accrued expenses | 273,740 | 221,602 | ||||||
| Intangible assets | 22,991 | 24,914 | ||||||
| Other | 2,478 | 846 | ||||||
| Gross deferred tax assets | 4,852,037 | 2,264,852 | ||||||
| Less: Valuation allowance | (4,852,037 | ) | (2,264,852 | ) | ||||
| Total deferred tax assets | - | - | ||||||
| Deferred tax liabilities: | ||||||||
| - | - | |||||||
| Net deferred tax assets | $ | - | $ | - | ||||
F-18
At December 31, 2019, the Company had federal, state, and foreign net operating loss (NOL) carryforwards of approximately $19.2 million, $20.1 million and $0.3 million, respectively. The federal and certain state loss carryforwards generated in 2018 onwards of $16.5 million and $0.7 million, respectively, will carry forward indefinitely and can be used to offset up to 80% of future annual taxable income. Federal loss carryforwards generated prior to 2018 begin expiring in 2037, unless previously utilized. State loss carryforwards begin expiring in 2037, unless previously utilized, while the Company’s foreign loss carryforwards do not expire.
Realization of deferred tax assets is dependent upon future earnings, if any, the timing and amount of which are uncertain. Management assesses the available positive and negative evidence to estimate if sufficient future taxable income will be generated to use existing deferred tax assets. Based on the weight of evidence, including a history of operating losses, management has determined that it is more likely than not that the Company’s net deferred tax assets will not be realized. Accordingly, a valuation allowance of $4.9 million and $2.3 million has been established by the Company to fully offset these net deferred tax assets as of December 31, 2019 and 2018, respectively. The valuation allowance increased by $2.6 million during 2019.
Future utilization of the Company’s NOL carryforwards to offset taxable income may be subject to a substantial annual limitation as a result of ownership changes that may have occurred or that could occur in the future pursuant to Internal Revenue Code Sections 382 and 383. These ownership changes may limit the amount of NOL carryforwards that can be utilized to offset future taxable income and tax, respectively. In general, an ownership change, as defined by the tax code, results from a transaction or series of transactions over a three-year period resulting in an ownership change of more than 50 percent of the outstanding stock of a company by certain stockholders or public groups. The Company has not completed an analysis regarding the limitation of NOL carryforwards.
The Company applies the two-step approach to recognizing and measuring uncertain tax positions. The first step is to evaluate the tax position for recognition by determining if the weight of available evidence indicates it is more likely than not that the position will be sustained on audit, including resolution of related appeals or litigation processes, if any. The second step is to measure the tax benefit as the largest amount, which is more than 50% likely of being realized upon ultimate settlement. Income tax positions must meet a more likely than not recognition threshold at the effective date to be recognized upon the adoption of ASC 740 and in subsequent periods. This interpretation also provides guidance on measurement, derecognition, classification, interest and penalties, accounting in interim periods, disclosure and transition. The following table summarizes the activity related to the Company’s gross unrecognized tax benefits for the years ended December 31, 2019 and 2018:
| As of December 31, | ||||||||
| 2019 | 2018 | |||||||
| Balance at beginning of year | $ | 772,589 | $ | 237,554 | ||||
| Increases related to current year tax positions | 925,899 | 535,035 | ||||||
| Balance at end of year | $ | 1,698,488 | $ | 772,589 | ||||
At December 31, 2019 and 2018, the amount of unrecognized tax benefits that would affect the effective tax rate was $0. The Company’s policy is to recognize interest and penalties related to uncertain tax positions in income tax expense. The Company has not accrued any interest or penalties related to uncertain tax positions since inception. The Company does not anticipate that there will be a significant change in the amount of unrecognized tax benefits over the next twelve months.
The Company is subject to tax in the US federal jurisdiction as well as various state and foreign jurisdictions. The Company’s federal, state and foreign income tax returns beginning in 2017 are subject to examination by tax authorities; however, no such examinations have taken place.
F-19
11. Subsequent Events
As of April 28, 2020, the Company has sold 722,830 additional shares of common stock at $6.00 per share under its Offering pursuant to Regulation A, for gross proceeds of approximately $4.3 million. The Company is obligated to pay additional commissions of approximately $43,000 related to these sales of common stock under its Broker-Dealer Agreement.
In January 2020, EHP Australia entered into a loan agreement with Rocking Horse Nominees Pty Ltd (Rocking Horse), whereby Rocking Horse advanced $AU1.2 million (approximately $0.8 million) to EHP Australia. See Note 6.
In March 2020, the World Health Organization declared the outbreak of the novel coronavirus (COVID-19) as a pandemic. This contagious disease outbreak, which has continued to spread, has adversely affected workforces, economies, and financial markets globally. As a result, the Company is taking the steps to protect the health, safety and well-being of its employees, associates, and communities. The Company cannot reasonably estimate the length or severity of this pandemic, which may result in an adverse impact on its balance sheets, results of operations and cash flows in 2020. The evolving pandemic may affect patient enrollment for the Company’s Phase 2 clinical studies as patients may avoid or may not be able to travel to clinical study site locations. Similarly, this outbreak could result in the complete or partial closure of one or more of the Company’s clinical study site locations, the CRO, and/or impact the trial monitors and other critical vendors and consultants supporting the trials, and the impact of the COVID-19 pandemic on the global financial markets may reduce the Company’s ability to raise additional capital on attractive terms or at all.
On April 22, 2020, the Company received loan proceeds of $292,152 (PPP Loan) from Silicon Valley Bank pursuant to the Paycheck Protection Program (PPP) established as part of the Coronavirus Aid, Relief and Economic Security Act (CARES Act). The PPP Loan matures on April 21, 2022 and bears interest at a rate of 1% per annum, payable monthly commencing on November 21, 2020. The Note may be prepaid at any time prior to maturity with no prepayment penalties. The principal and interest accrued under the PPP Loan are forgivable after eight weeks as long as the loan proceeds are used for eligible purposes.
On April 24, 2020, the Company received written notice of demand from EHS for payment of $150,000 of accrued interest on the Related Party Loan.
F-20
Emerald Health Pharmaceuticals Inc.
Condensed Consolidated Balance Sheets
(Unaudited)
| June 30, 2020 | December 31, 2019 | |||||||
| Assets | ||||||||
| Current assets: | ||||||||
| Cash and cash equivalents | $ | 4,311,433 | $ | 983,261 | ||||
| Restricted cash | 1,203,617 | - | ||||||
| Incentive and other tax receivables | 271,410 | 1,420,107 | ||||||
| Other current assets | 881,656 | 548,280 | ||||||
| Total current assets | 6,668,116 | 2,951,648 | ||||||
| Property plant and equipment, net | 39,247 | 52,458 | ||||||
| Total assets | $ | 6,707,363 | $ | 3,004,106 | ||||
| Liabilities and stockholders’ deficit | ||||||||
| Current liabilities: | ||||||||
| Accounts payable | $ | 881,172 | $ | 1,945,549 | ||||
| Accrued expenses | 1,983,071 | 1,977,627 | ||||||
| Deposits held in escrow | 1,203,617 | - | ||||||
| Accrued interest payable | 367,286 | 505,289 | ||||||
| Related party loan | 3,750,701 | 3,750,701 | ||||||
| Total current liabilities | 8,185,847 | 8,179,166 | ||||||
| Loans payable | 292,152 | - | ||||||
| Total liabilities | 8,477,999 | 8,179,166 | ||||||
| Commitments and contingencies (Note 6) | ||||||||
| Stockholders’ deficit: | ||||||||
| Common stock, $0.0001 par value; 100,000,000 shares authorized, 16,000,711 shares issued and 15,925,711 shares outstanding at June 30, 2020; 100,000,000 shares authorized, 14,492,085 shares issued and 14,417,085 shares outstanding at December 31, 2019 | 1,600 | 1,449 | ||||||
| Additional paid-in-capital | 31,372,776 | 22,546,309 | ||||||
| Accumulated other comprehensive loss | (84,189 | ) | (38,724 | ) | ||||
| Accumulated deficit | (33,060,815 | ) | (27,684,086 | ) | ||||
| Treasury stock, at cost (common stock: 75,000 at June 30, 2020 and December 31, 2019) | (8 | ) | (8 | ) | ||||
| Total stockholders’ deficit | (1,770,636 | ) | (5,175,060 | ) | ||||
| Total liabilities and stockholders’ deficit | $ | 6,707,363 | $ | 3,004,106 | ||||
See accompanying Notes to Condensed Consolidated Financial Statements (Unaudited).
F-21
Emerald Health Pharmaceuticals Inc.
Condensed Consolidated Statements of Operations and Comprehensive Loss
(Unaudited)
| Six Months Ended June 30, | ||||||||
| 2020 | 2019 | |||||||
| Revenue | $ | - | $ | - | ||||
| Operating expenses: | ||||||||
| Research and development | 3,315,691 | 5,110,329 | ||||||
| General and administrative | 1,775,129 | 2,102,625 | ||||||
| Total operating expenses | 5,090,820 | 7,212,954 | ||||||
| Operating loss | (5,090,820 | ) | (7,212,954 | ) | ||||
| Other (income)/expenses: | ||||||||
| Related party interest income | - | (40,933 | ) | |||||
| Interest expense | 265,180 | 2,016,138 | ||||||
| Foreign exchange loss | 20,729 | 21,839 | ||||||
| Net loss | (5,376,729 | ) | (9,209,998 | ) | ||||
| Other comprehensive income (loss): | ||||||||
| Foreign currency translation adjustments | (45,465 | ) | (24,045 | ) | ||||
| Comprehensive loss | $ | (5,422,194 | ) | $ | (9,234,043 | ) | ||
| Net loss per share, basic and diluted | $ | (0.36 | ) | $ | (0.80 | ) | ||
| Weighted-average common shares outstanding, basic and diluted | 14,979,018 | 11,575,869 | ||||||
See accompanying Notes to Condensed Consolidated Financial Statements (Unaudited).
F-22
Emerald Health Pharmaceuticals Inc.
Condensed Consolidated Statements of Stockholders’ Deficit
For the Six Months Ended June 30, 2020
(Unaudited)
| Common Stock Outstanding | Additional Paid in | Accumulated Other Comprehensive | Accumulated | Treasury Stock | Total Stockholders’ | |||||||||||||||||||||||||||
| Shares | Amount | Capital | Loss | Deficit | Shares | Amount | Deficit | |||||||||||||||||||||||||
| Balance at December 31, 2019 | 14,417,085 | $ | 1,449 | $ | 22,546,309 | $ | (38,724 | ) | $ | (27,684,086 | ) | 75,000 | $ | (8 | ) | $ | (5,175,060 | ) | ||||||||||||||
| Issuance of common stock under Regulation A offering, net of issuance costs | 1,393,626 | 139 | 7,916,431 | 7,916,570 | ||||||||||||||||||||||||||||
| Issuance of common stock for services | 100,000 | 10 | 599,990 | 600,000 | ||||||||||||||||||||||||||||
| Issuance of restricted common stock under equity incentive plan | 15,000 | 2 | (2 | ) | - | |||||||||||||||||||||||||||
| Stock-based compensation expense | 310,048 | 310,048 | ||||||||||||||||||||||||||||||
| Net loss and comprehensive loss | (45,465 | ) | (5,376,729 | ) | (5,422,194 | ) | ||||||||||||||||||||||||||
| Balance at June 30, 2020 | 15,925,711 | $ | 1,600 | $ | 31,372,776 | $ | (84,189 | ) | $ | (33,060,815 | ) | 75,000 | $ | (8 | ) | $ | (1,770,636 | ) | ||||||||||||||
F-23
Condensed Consolidated Statements of Stockholders’ Deficit
For the Six Months Ended June 30, 2019
(Unaudited)
| Common Stock Outstanding | Additional Paid in | Note Receivable from | Accumulated Other Comprehensive | Accumulated | Treasury Stock | Total Stockholders’ | ||||||||||||||||||||||||||||||
| Shares | Amount | Capital | Stockholder | Loss | Deficit | Shares | Amount | Deficit | ||||||||||||||||||||||||||||
| Balance at December 31, 2018 | 9,925,000 | $ | 1,000 | $ | 574,522 | $ | - | $ | (1,808 | ) | $ | (11,315,176 | ) | 75,000 | $ | (8 | ) | $ | (10,741,470 | ) | ||||||||||||||||
| Issuance of common stock under Regulation A offering, net of issuance costs | 2,539,836 | 254 | 11,991,935 | 11,992,189 | ||||||||||||||||||||||||||||||||
| Issuance of common stock under Regulation S offering, net of issuance costs | 65,700 | 7 | 272,771 | 272,778 | ||||||||||||||||||||||||||||||||
| Issuance of common stock upon conversion of related party loan | 1,250,000 | 125 | 2,499,875 | 2,500,000 | ||||||||||||||||||||||||||||||||
| Recognition of beneficial conversion feature on related party loan | 1,360,840 | 1,360,840 | ||||||||||||||||||||||||||||||||||
| Funds advanced under note receivable from stockholder | (5,000,000 | ) | (5,000,000 | ) | ||||||||||||||||||||||||||||||||
| Discharge between related party loan and note receivable from stockholder | 2,000,000 | 2,000,000 | ||||||||||||||||||||||||||||||||||
| Stock-based compensation expense | 1,056,289 | 1,056,289 | ||||||||||||||||||||||||||||||||||
| Net loss and comprehensive loss | (24,045 | ) | (9,209,998 | ) | (9,234,043 | ) | ||||||||||||||||||||||||||||||
| Balance at June 30, 2019 | 13,780,536 | $ | 1,386 | $ | 17,756,232 | $ | (3,000,000 | ) | $ | (25,853 | ) | $ | (20,525,174 | ) | 75,000 | $ | (8 | ) | $ | (5,793,417 | ) | |||||||||||||||
F-24
Emerald Health Pharmaceuticals Inc.
Condensed Consolidated Statements of Cash Flows
(Unaudited)
| Six Months Ended June 30, | ||||||||
| 2020 | 2019 | |||||||
| Operating activities | ||||||||
| Net loss | $ | (5,376,729 | ) | $ | (9,209,998 | ) | ||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||
| Depreciation | 13,211 | 14,273 | ||||||
| Stock-based compensation | 910,048 | 1,056,289 | ||||||
| Accretion of beneficial conversion feature on related party loan | - | 1,360,840 | ||||||
| Changes in operating assets and liabilities: | ||||||||
| Incentive and other tax receivables | 1,148,697 | (811,264 | ) | |||||
| Other current assets | (333,376 | ) | (261,081 | ) | ||||
| Accounts payable | (1,110,677 | ) | 514,840 | |||||
| Accrued expenses | 8,505 | 491,642 | ||||||
| Accrued interest payable | (138,003 | ) | (389,603 | ) | ||||
| Net cash used in operating activities | (4,878,324 | ) | (7,234,062 | ) | ||||
| Investing activities | ||||||||
| Net cash used in investing activities | - | - | ||||||
| Financing activities | ||||||||
| Issuance of common stock | 8,361,756 | 13,027,680 | ||||||
| Funds received under loans payable | 1,087,373 | - | ||||||
| Funds repaid under loans payable | (795,221 | ) | - | |||||
| Deposits held in escrow | 1,203,617 | - | ||||||
| Funds received under related party loan | - | 1,674,380 | ||||||
| Funds advanced under note receivable from stockholder | - | (5,000,000 | ) | |||||
| Stock issuance costs | (401,947 | ) | (430,444 | ) | ||||
| Net cash provided by financing activities | 9,455,578 | 9,271,616 | ||||||
| Effect of exchange rate changes on cash | (45,465 | ) | (24,045 | ) | ||||
| Net increase in cash and cash equivalents, and restricted cash | 4,531,789 | 2,013,509 | ||||||
| Cash, cash equivalents, and restricted cash at beginning of period | 983,261 | 137,706 | ||||||
| Cash, cash equivalents, and restricted cash at end of period | $ | 5,515,050 | $ | 2,151,215 | ||||
| Supplemental disclosure of cash flow information: | ||||||||
| Interest paid to related party | $ | 350,000 | $ | 1,044,901 | ||||
| Interest paid on loans payable | $ | 42,597 | $ | - | ||||
| Non-cash investing and financing activities: | ||||||||
| Conversion of related party loan to common stock | $ | - | $ | 2,500,000 | ||||
| Discharge between related party loan and note receivable from stockholder | $ | - | $ | 2,000,000 | ||||
| Deferred stock issuance costs in accounts payable and accrued expenses | $ | 60,194 | $ | 58,977 | ||||
See accompanying Notes to Condensed Consolidated Financial Statements (Unaudited).
F-25
Emerald Health Pharmaceuticals Inc.
Notes to Condensed Consolidated Financial Statements
(Unaudited)
1. Description of Business and Going Concern
Emerald Health Pharmaceuticals Inc. (EHP, or the Company) was incorporated in the state of Delaware in March 2017. The Company is a biotechnology/pharmaceutical company, formed to acquire, discover, develop and commercialize drug product candidates containing novel, patented molecules chemically derived from two non-psychoactive cannabinoids (molecules found in cannabis), cannabidiol (CBD) and cannabigerol (CBG). EHP is focused on developing product candidates to treat diseases with unmet medical needs primarily in inflammatory, autoimmune, metabolic, neurodegenerative and fibrotic diseases. The Company is currently developing two initial product candidates that together target four initial diseases, multiple sclerosis (MS), systemic sclerosis (SSc), a severe form of scleroderma, Parkinson’s disease (PD) and Huntington’s disease (HD).
The Company acquired certain intellectual property from Emerald Health Biotechnology España, S.L.U. (EHBE), formerly known as VivaCell Biotechnology España S.L. (VivaCell). During the year ended December 31, 2018, EHBE became a wholly owned subsidiary of Emerald Health Research Inc. (EHR) which is a wholly owned subsidiary of Emerald Health Sciences Inc. (EHS). EHS is also the majority stockholder of EHP. EHP has no ownership or voting rights related to EHBE. See Note 7.
The Company is subject to risks common to other life science companies in the development stage including, but not limited to, uncertainty of product development and commercialization, lack of marketing and sales history, development by its competitors of new technological innovations, dependence on key personnel, market acceptance of products, product liability, protection of proprietary technology, ability to raise additional financing, and compliance with FDA and other government regulations. If the Company does not successfully commercialize any product candidates, it will be unable to generate recurring product revenue or achieve profitability.
The global spread of the novel coronavirus (COVID-19) has created significant volatility, uncertainty and economic disruption. The ultimate effects of the COVID-19 on the Company’s business, operations and financial condition are unknown at this time. In the near term, the potential exists for enrollment in its Phase 2a clinical trial to be delayed or slowed based on this, as patients may elect to postpone voluntary treatments and physicians’ offices are either closed or operating at a reduced capacity. However, the extent to which COVID-19 impacts the Company’s business will depend on future developments, which are highly uncertain and cannot be predicted, including new information which may emerge concerning the severity of COVID-19 and the actions to contain it or treat its impact, among others.
The Company’s financial statements have been prepared assuming the Company will continue as a going concern, which contemplates the realization of assets and the satisfaction of liabilities in the normal course of business. The Company has experienced losses and recurring cash outflows from operations since inception and has an accumulated deficit of $33,060,815 and negative working capital as of June 30, 2020. The Company has funded operations with capital raised from an ongoing Tier 2 offering (the Offering) pursuant to Regulation A (Regulation A) under the Securities Act of 1933, as amended (the Securities Act), as well as an exempt offshore offering under Regulation S under the Securities Act. In addition, the Company has received loan proceeds from three separate loan arrangements, including a revolving loan with its majority stockholder. Amounts advanced under the revolving loan and accrued interest are due upon demand. See Note 3 and Note 6.
The future viability of the Company is largely dependent upon its ability to raise additional capital to finance its operations. Management expects that future sources of funding may include sales of equity, obtaining loans, or other strategic transactions. Although management continues to pursue these plans, there is no assurance that the Company will be successful in obtaining sufficient financing on terms acceptable to the Company to fund continuing operations, if at all. These circumstances raise substantial doubt on the Company’s ability to continue as a going concern. The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
The Company is undertaking a “best efforts” offering of its common stock to raise additional capital. See Note 4.
F-26
2. Significant Accounting Policies
Basis of Presentation
The accompanying unaudited condensed consolidated financial statements have been prepared in conformity with accounting principles generally accepted in the United States (GAAP). and in accordance with the instructions to Form 1-SA and Regulation S-X. As permitted under these rules, certain footnotes or other financial information that are normally required by GAAP have been condensed or omitted. The Company has made estimates and judgments affecting the amounts reported in the condensed consolidated financial statements and the accompanying notes. The actual results experienced by the Company may differ materially from our estimates. The financial information is unaudited but reflects all normal adjustments that are, in the opinion of management, necessary to provide a fair statement of results for the interim period presented. The results for the six months ended June 30, 2020 are not necessarily indicative of the results expected for the year ended December 31, 2020 or any future periods. These condensed consolidated financial statements should be read in conjunction with the consolidated financial statements and notes thereto for the year ended December 31, 2019 included in the Company’s Annual Report on Form 1-K filed with the Securities and Exchange Commission (the SEC) on April 28, 2020.
Consolidation
The consolidated financial statements include the accounts of the Company and its wholly-owned subsidiaries, Emerald Health Pharmaceuticals Australia Pty Ltd. (EHP Australia) and Emerald Health Pharmaceuticals, España Sociedad Limitada (EHP España). EHP Australia’s functional currency, the Australian dollar, is also its reporting currency, and its financial statements are translated to U.S. dollars prior to consolidation. EHP España’s functional currency, the Euro, is also its reporting currency, and its financial statements are translated to U.S. dollars prior to consolidation. All intercompany accounts and transactions have been eliminated in consolidation.
Use of Estimates
The preparation of financial statements in conformity with generally accepted accounting principles requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities at the date of the financial statements and the reported amounts of revenue and expenses during the reporting period. Actual results could differ from those estimates.
Cash, Cash Equivalents and Restricted Cash
The Company considers all highly liquid investments with an original maturity of three months or less from the date of purchase to be cash equivalents. As of June 30, 2020, the Company’s cash deposits are held in an FDIC-insured financial institution.
Restricted cash consists of cash held in an escrow account, received as deposits from potential investors towards purchases of common stock under the Offering which have not yet been fully consummated as of the balance sheet date, as described in Note 4.
The following table provides a reconciliation of cash, cash equivalents and restricted cash, reported within the condensed consolidated statements of cash flows:
| Six Months Ended June 30, | ||||||||
| 2020 | 2019 | |||||||
| Cash and cash equivalents | $ | 4,311,433 | $ | 2,151,215 | ||||
| Restricted cash | 1,203,617 | - | ||||||
| Total cash, cash equivalents and restricted cash presented in the condensed consolidated statements of cash flows | $ | 5,515,050 | $ | 2,151,215 | ||||
Incentive and Tax Receivables
The Company’s subsidiary, EHP Australia, is incorporated in Australia and is eligible to participate in an Australian research and development tax incentive program. As part of this program, EHP Australia is eligible to receive a cash refund from the Australian Taxation Office (ATO) for a percentage (currently 43.5%) of the research and development costs incurred by EHP Australia. The cash refund is available to eligible companies with an annual aggregate revenue of less than $AU20.0 million (Australian Dollars) during the reimbursable period. As of June 30, 2020 and December 31, 2019, the Company’s estimate of the amount of cash refunds expected to be received for eligible spending as part of this incentive program was $0.3 million and $1.3 million, respectively, which amounts are included in incentive and other tax receivables. In May 2020, the Company received $1.3 million as a cash refund from the ATO for eligible spending incurred during the year ended December 31, 2019.
In addition, EHP Australia incurs Goods and Services Tax (GST) on services provided by Australian vendors. As an Australian entity, EHP Australia is entitled to a refund of the GST paid. The Company’s estimate of the amount of cash refund expected to be received related to GST incurred as of June 30, 2020 and December 31, 2019, was $1,419 and $78,198, respectively, which amounts are included in incentive and other tax receivables.
F-27
Research and Development
Research and development costs are charged to expense as incurred and consist primarily of contract research fees, contract manufacturing costs, consultant fees, preclinical and clinical studies and study related costs, compensation and related benefits, and non-cash stock-based compensation. At the end of each reporting period, the Company compares the payments made to its vendors, clinical research organizations and consultants to the estimated progress towards completion of the related project. Factors that the Company considers in preparing these estimates include the number of patients enrolled in studies, milestones achieved, and other criteria related to the efforts of its vendors. These estimates will be subject to change as additional information becomes available. Depending on the timing of payments to vendors and estimated services provided, the Company will record net prepaid or accrued expenses related to these costs. Research and development expenses are recorded net of expected refunds of eligible research and development costs paid pursuant to the Australian research and development tax incentive program and GST incurred on services provided by Australian vendors.
Business Segments
The Company operates within the United States, Europe, and Australia, in one business segment, which is dedicated to research of drug candidates based on patented synthetic new chemical entities (NCEs) derived from non-psychoactive cannabinoid molecules.
Stock-Based Compensation
The Company accounts for stock option awards in accordance with Financial Accounting Standards Board Accounting Standards Codification (ASC) Topic No. 718, Compensation-Stock Compensation. Under FASB ASC Topic No. 718, compensation expense related to stock-based payments is recorded over the requisite service period based on the grant date fair value of the awards. Compensation previously recorded for unvested stock options that are forfeited is reversed upon forfeiture. The Company uses the Black-Scholes option pricing model for determining the estimated fair value for stock-based awards. The Black-Scholes model requires the use of assumptions which determine the fair value of stock-based awards, including the option’s expected term and the price volatility of the underlying stock. See Note 5.
Net Loss per Share
The Company computes basic net loss per common share by dividing the applicable net loss by the weighted average number of common shares outstanding during the period. Diluted earnings per share is computed using the weighted average number of common shares outstanding during the period, plus additional shares to account for the dilutive effect of potential future issuances of common stock relating to stock options and other potentially dilutive securities using the treasury stock method. For the six months ended June 30, 2020 and 2019, 2,075,000 and 2,060,000 options, respectively, were excluded from the computation of diluted earnings per share, as the effect would be anti-dilutive.
Comprehensive Loss
Comprehensive loss includes foreign currency translation adjustments related to the Company’s subsidiaries in Australia and Spain.
Significant Accounting Policies
There have been no changes to the significant accounting policies that were described in Note 2 to the 2019 Audited Financial Statements during the first six months of fiscal year 2020.
F-28
Recently Issued Accounting Pronouncements
In February 2016, the FASB issued ASU No. 2016-02, Leases (Topic 842) (ASU No. 2016-02), which changes the presentation of assets and liabilities relating to leases. The core principle of ASU No. 2016-02 is that a lessee should recognize the assets and liabilities that arise from leases. All leases create an asset and a liability for the lessee in accordance with FASB Concepts Statement No. 6, Elements of Financial Statements, and, therefore, recognition of those lease assets and lease liabilities represents an improvement over previous GAAP, which did not require lease assets and lease liabilities to be recognized for most leases.
In November 2019, the FASB issued ASU No. 2019-10, Financial Instruments - Credit Losses (Topic 326), Derivatives and Hedging (Topic 815), and Leases (Topic 842) (ASU No. 2019-10), which deferred the effective date of ASU No. 2016-02 for the Company from January 1, 2020 to January 1, 2021. The Company is currently evaluating the impact of this new standard on its financial statements.
In October 2018, the FASB issued ASU No. 2018-17, Consolidation (Topic 810) (ASU No. 2018-17), which adds an elective private-company scope exception to the variable interest entity (VIE) guidance for entities under common control. ASU No. 2018-17 will be effective for the Company beginning January 1, 2021, with early adoption permitted. The Company does not expect this new standard to have a material impact on its financial statements.
3. Related Party Transactions
Related Party Loan and Beneficial Conversion Feature
Since inception, the Company has received advances from EHS to fund its operations. In September 2017, the Company and EHS entered a revolving loan agreement (Related Party Loan), which was amended in January 2018. Borrowings under the loan, which EHP may draw down from time to time in one or more advances, are evidenced by a demand grid promissory note (the Note). The Note is revised to reflect the aggregate principal amount of the loan outstanding as of the date of each advance or repayment. In November 2019, the Related Party Loan was further amended to reduce the interest rate from 12% to 10%, compounded semiannually. The loan may be repaid by the Company or converted by EHS into shares of EHP at $2.00 per share or at a price to be equally agreed to between EHS and the Company (Conversion Feature). The Note is payable upon demand and has no expiration date. As of June 30, 2020 and December 31, 2019, $3,750,701 and $3,750,701, respectively, of principal, and $367,286 and $505,289, respectively, of accrued interest is due to EHS under the Related Party Loan.
F-29
In April 2019, the Company received written notice of demand (Notice) from EHS for payment of all accrued interest on the Related Party Loan as of March 31, 2019, which resulted in a cash payment of $1,044,901 to EHS. Also in April 2019, the Company received a second Notice from EHS that called for the following, upon qualification by the SEC of the Company’s Form 1-A Post-Qualification Offering Circular Amendment: (1) repayment of $2,000,000 of the unpaid principal balance under the loan, and (2) the conversion of an additional $2,500,000 of the unpaid principal balance under the loan at a conversion price of $2.00 per share. The Company’s Form 1-A Post-Qualification Offering Circular Amendment was qualified by the SEC on June 7, 2019 and the repayment of the $2,000,000 of unpaid principal was transacted as a cashless discharge and offset between the Related Party Loan and the Related Party Note Receivable (as defined below). Concurrently, 1,250,000 shares of EHP common stock were issued to EHS at a conversion price of $2.00 per share, further reducing the principal balance of the Related Party Loan by $2,500,000.
In November 2019, the Company received written notice of demand from EHS for payment of $3,000,000 of unpaid principal balance and $178,933 of accrued interest on the Related Party Loan, which was transacted as a cashless discharge and offset between the Related Party Loan and the remaining unpaid principal and accrued interest balances under the Related Party Note Receivable (as defined below) as of November 15, 2019.
The Conversion Feature of the loan agreement is not considered an embedded derivative under FASB Accounting Standards Codification (ASC) Topic 815, Derivatives and Hedging, since there are no provisions for net settlement nor is there a means for EHS to receive an asset that puts EHS in a position not substantially different from net settlement. The Company recorded a debt discount on the Related Party Loan of $1.4 million related to the beneficial conversion feature on advances under the loan during the year ended December 31, 2019. Subsequent to the recognition of the discount, due to the on-demand nature of the loan, the Company recognized $1.4 million in accretion of the discount which is included in related party interest expense. There were no such transactions recorded during the six months ended June 30, 2020, as there were no additional advances received under the loan during the period.
Related Party Note Receivable
In May 2019, the Company’s Board of Directors authorized a funding arrangement with EHS (the Related Party Note Receivable), which was amended in August 2019 and September 2019 to extend the repayment dates, whereby EHP may advance funds to EHS in the form of interest bearing (12%) short term notes, up to an aggregate principal amount of $6,000,000 under a Promissory Note between EHS and EHP (the Promissory Note). Advances under the Promissory Note were originally due for repayment with accrued and unpaid interest three months from the date of the advance. During the year ended December 31, 2019, a total of $5,000,000 was advanced and $178,933 accrued as interest receivable under the Related Party Note Receivable, all of which was offset through cashless discharges against the unpaid principal and accrued interest payable balances, respectively, under the existing Related Party Loan with EHS. As of June 30, 2020 and December 31, 2019, there are no outstanding principal or accrued interest receivable balances remaining under the Related Party Note Receivable, and all principal advances and related discharges have been recorded as equity transactions.
Shared Services with EHS and Related Entities
In June 2019, the Company entered into an Independent Contractor Agreement (the Independent Contractor Agreement) effective April 1, 2019, with EHS, pursuant to which EHS agreed to provide such services as are mutually agreed between the Company and EHS, including reimbursements for reasonable expenses incurred in the performance of the Independent Contractor Agreement. These services included, but were not limited to, corporate advisory services and technical expertise in the areas of business development, marketing, investor relations, information technology and product development. The Independent Contractor Agreement had an initial term of ten years. On November 15, 2019, the Board of Directors approved the termination of this agreement, effective as of December 31, 2019. During the six months ended June 30, 2020 and 2019, the Company recorded expenses totaling $0 and $175,530, respectively, for such services performed by EHS on behalf of the Company.
The Company allocates certain operating expenses to entities which are subsidiaries of EHS for their share of facilities and office expenses. During the six months ended June 30, 2020 and 2019, these allocations totaled $71,895 and $67,806, respectively.
F-30
Dr. Avtar Dhillon
On November 15, 2019, Dr. Avtar Dhillon resigned as Chairman of the Board of Directors. The Company and EHS concurrently entered into a Board Observer Agreement, whereby the Company granted to EHS the right to designate an observer on the Board for so long as EHS maintains ownership of any securities of the Company. Dr. Avtar Dhillon was appointed as the initial Board Observer pursuant to the Board Observer Agreement.
On December 5, 2019, the Board of Directors approved an Independent Contractor Services Agreement, effective as of December 1, 2019, between the Company and Dr. Dhillon, pursuant to which Dr. Dhillon will provide ongoing corporate finance and strategic business advisory services to the Company. In exchange for his services, upon the Company completing a material financing, Dr. Dhillon will receive a monthly fee of $10,000, accruing from the effective date. The Board will review the monthly rate paid to Dr. Dhillon within 90 days of the end of each fiscal year. The Independent Contractor Services Agreement has an initial term of one year and will renew automatically thereafter unless terminated earlier by either party. The Independent Contractor Services Agreement may be terminated by either party for cause upon written notice to the other party if the other party defaults in the performance of the agreement in any material respect or materially breaches the terms of the agreement, or without cause upon 30 days’ prior written notice to the other party. As of June 30, 2020 and December 31, 2019, accrued expenses include $70,000 and $10,000, respectively due and payable to Dr. Dhillon under this agreement, upon completion of a material financing, as defined by the Board of Directors.
4. Common Stock
On March 2, 2017, the Company issued 9,000,000 shares of common stock at $0.0001 per share to EHS for proceeds of $900. An additional 1,000,000 shares were issued to the founders of the Company for total proceeds of $100. The shares issued to founders vested 25% on the date of issuance and vest 25% annually thereafter until fully vested. Until the shares of common stock vest, the founders may not sell or transfer the unvested shares of common stock. In the event of the voluntary or involuntary termination of any of the founders, as an employee or director of the Company for any reason, the Company shall have the option to repurchase all or any portion of the shares of common stock for the same consideration which was originally paid by the founders. In October 2018, the Company exercised its option to repurchase 75,000 unvested shares from a founding member, which are currently held by the Company as treasury stock.
In January 2018, the Company filed a Certificate of Amendment of the Certificate of Incorporation which increased the number of authorized shares that the Company can issue from 20,000,000 to 100,000,000 shares of common stock with a par value of $0.0001 per share.
In June 2019, the Company issued 1,250,000 additional shares of common stock to EHS in accordance with a written notice received from EHS in April 2019, for the conversion of $2,500,000 of the unpaid principal balance under the Related Party Loan at a conversion price of $2.00 per share. See Note 3.
The Company is currently selling common stock through the Offering. The Company’s initial Offering Statement on Form 1-A was qualified by the SEC in March 2018 and its Form 1-A Post-Qualification Offering Circular Amendments were subsequently qualified by the SEC on June 7, 2019 and July 14, 2020. During the six months ended June 30, 2020, the Company sold 1,393,626 shares of common stock under the Offering, for gross proceeds of $8.4 million, less issuance costs of $0.5 million; and during the six months ended June 30, 2019, the Company sold 2,539,836 shares of common stock under the Offering, for gross proceeds of $12.7 million, less issuance costs of $0.7 million.
In June 2019, the Company issued 1,250,000 additional shares of common stock to EHS in accordance with a written notice received from EHS in April 2019, for the conversion of $2,500,000 of the unpaid principal balance under the Related Party Loan at a conversion price of $2.00 per share. See Note 3.
During the six months ended June 30, 2019, the Company also sold 65,700 shares of common stock for gross proceeds of $328,500, less issuance costs of $51,776 in an exempt offshore offering under Regulation S under the Securities Act.
In June 2019, the Company entered into a Broker-Dealer Agreement with Dalmore Group, LLC (Dalmore), a broker-dealer registered with the SEC and a member of FINRA, to perform administrative, compliance and placement agent related functions in connection with the Offering. The Company has agreed to pay Dalmore a 1.0% commission on the sale of common stock under the Offering, commencing with sales following regulatory approval by FINRA, which occurred on July 25, 2019. In addition, the Company paid Dalmore $28,000 in one-time set up fees, consisting of a $20,000 agreement fee and $8,000 for fees paid to FINRA. As of June 30, 2020, the Company has paid $71,630 and has accrued an additional $37,102 to be paid to Dalmore related to commission on the sale of common stock under the Offering.
F-31
In July 2019, the Company entered into an Escrow Services Agreement with Prime Trust, LLC. Under this agreement, the proceeds received from the Offering are deposited into an escrow account prior to distribution to the Company. As of June 30, 2020, there was $1.2 million in this escrow account as deposits received from potential investors towards purchases of common stock under the Offering, which are still in process. The balance has been recorded as restricted cash, offset by deposits held in escrow liability.
In June 2020, the Company issued 100,000 shares of common stock to a consultant as payment for services. At the time of issuance, the Company recognized $600,000 of stock-based compensation expense, of which $300,000 was for research and development and $300,000 was for general and administrative services.
The Company also issued 15,000 shares of restricted common stock under the Plan (as defined below), to a consultant as payment for services.
5. Equity Incentive Plan
In January 2018, the Company adopted the 2018 Equity Incentive Plan, which was amended on December 13, 2018 and on August 12, 2020 (the Plan). As of June 30, 2020, there were 2,200,000 shares of common stock reserved for issuance under the Plan. On August 12, 2020 the Company adopted an amendment to the Plan, which increased the number of shares of Common Stock authorized to be issued under the 2018 Plan to equal 18% of the number of issued and outstanding shares of common stock of the Company as of the applicable date of issuance. As of August 12, 2020, 2,896,319 shares of Common Stock were reserved for issuance pursuant to awards under the 2018 Plan.
The Plan provides incentives to eligible employees, consultants, officers, and directors in the form of incentive stock options and non-qualified stock options, stock appreciation rights, restricted stock, restricted stock units and other rights or benefits. Recipients of stock options are eligible to purchase shares of the Company’s common stock at an exercise price equal to no less than the estimated fair market value of such stock on the date of grant. The maximum term of options granted under the Plan is ten years. Vesting schedules are determined by the Board of Directors. As of June 30, 2020, there were 125,000 shares available to grant under the Plan.
The following table summarizes stock-based compensation expense related to stock options granted to employees and nonemployees included in the condensed consolidated statements of operations as follows:
| Six Months Ended June 30, | ||||||||
| 2020 | 2019 | |||||||
| Research and development | $ | 92,430 | $ | 109,446 | ||||
| General and administrative | 217,618 | 946,843 | ||||||
| Total | $ | 310,048 | $ | 1,056,289 | ||||
Stock Options
There were no stock options granted during the six months ended June 30, 2020 and 2019. In August 2020, the Company granted 782,500 additional stock options.
As of June 30, 2020, unrecognized stock-based compensation expense for employee and non-employee stock options was approximately $0.8 million, which the Company expects to recognize over a weighted-average remaining period of 1.5 years, assuming all unvested options become fully vested.
The Company uses a Black-Scholes option-pricing model to value the Company’s option awards. Using this option-pricing model, the fair value of each employee and non-employee award is estimated on the grant date. The fair value is expensed on a straight-line basis over the vesting period. In general, the option awards vest partially upfront and then pro-rata annually thereafter. The expected volatility assumption is based on the volatility of the share price of comparable public companies. The expected life is determined using the “simplified method” permitted by Staff Accounting Bulletin Number 107 and 110 (the midpoint between the term of the agreement and the weighted average vesting term). The risk-free interest rate is based on the implied yield on a U.S. Treasury security at a constant maturity with a remaining term equal to the expected term of the option granted. The dividend yield is zero, as the Company has never declared a cash dividend.
The fair value of the stock options granted was estimated on the date of grant using the Black-Scholes option pricing model.
F-32
Restricted Stock
There were 15,000 shares of restricted common stock issued under the Plan, to a non-employee, during the six months ended June 30, 2020. The shares vest over a 6-month service period beginning July 2020. The Company recognized no stock-based compensation expense during the six months ended June 30, 2020 for restricted stock.
As of June 30, 2020, unrecognized stock-based compensation expense related to the unvested restricted common stock was $90,000, which the Company expects to recognize over the remaining 6 months of 2020.
6. Commitments and Contingencies
On May 1, 2018, the Company entered into a two-year non-cancelable building lease for its corporate headquarters in San Diego, California. Effective August 15, 2019, the lease was amended to include additional space at the existing premises and to extend the term of the original lease through August 31, 2022. Under the lease, the Company pays a base rent of $20,624 per month through August 31, 2020, after which time the base rent will increase by approximately 3% per year. The Company made security deposit payments of $24,805 and $16,698 in 2019 and 2018, respectively, which are recorded within other current assets.
Future minimum payments under the non-cancelable operating lease as of June 30, 2020 were as follows:
| 2020 | $ | 126,199 | ||
| 2021 | 257,312 | |||
| 2022 | 174,818 | |||
| Total | $ | 558,329 |
In June 2019, EHS entered into a three-year non-cancelable lease agreement for unrelated lab space under which the Company was a guarantor on the lease. This lease was cancelled by EHS in February 2020.
In July 2020, the company entered into an agreement to sublease a portion of its existing non-cancelable building lease to a tenant, effective August 1, 2020, and continuing through August 31, 2022. The Company paid commissions of $7,000 related to the execution of the sublease and expects to incur an additional loss of approximately $55,000 as a result of the transaction.
Loans payable
On January 23, 2020, EHP Australia entered into a loan agreement with Rocking Horse Nominees Pty Ltd (Rocking Horse), whereby Rocking Horse advanced $AU1.2 million (approximately $0.8 million) to EHP Australia. The loan was secured by the tax incentive refund anticipated to be received during 2020 for eligible spending incurred under the Australian research and development tax incentive program during 2019. The loan had an upfront establishment fee of 1.2% and bore interest at 1.25% per month compounded daily. Approximately $53,000 in interest expense was incurred during the six months ended June 30, 2020, and the loan and all outstanding interest were repaid to Rocking Horse during June 2020.
On April 22, 2020, The Company received loan proceeds of $292,152 (PPP Loan) from Silicon Valley Bank pursuant to the Paycheck Protection Program (PPP) established as part of the Coronavirus Aid, Relief and Economic Security Act (CARES Act). The PPP Loan, which is evidenced by a Note dated April 21, 2020, matures on April 21, 2022 and bears interest at a rate of 1% per annum, payable monthly commencing on November 21, 2020. The Note may be prepaid at any time prior to maturity with no prepayment penalties. The principal and interest accrued under the PPP Loan may be forgiven after eight weeks as long as the loan proceeds are used for eligible purposes, including payroll, benefits, rent and utilities. The amount of loan forgiveness will be reduced if the Company terminates employees or reduces salaries during the eight-week period. The Company intend to use the proceeds of the PPP Loan for purposes consistent with the PPP. The Company believes its use of the PPP Loan proceeds will meet the conditions for forgiveness; however, no assurances can be made that the Company will not take actions that could cause the Company to be ineligible for forgiveness of the PPP Loan, in whole or in part. The PPP loan is classified as a long-term liability within loans payable on the balance sheet.
7. Intellectual Property Transfer and Research Agreements
In June 2017, upon the execution of the Intellectual Property Transfer Agreement (IPTA), EHP paid EHBE approximately $112,000 for the purchase of three United States patents, two Japanese patents, one European patent and fourteen pending patent applications covering two series of molecules containing derivatives of CBD and CBG. Future payments of up to 2.7 million Euro (approximately $3.0 million, based upon the exchange rate at June 30, 2020) per product are due upon completion of certain development milestones. As further consideration, the Company will pay EHBE a 2.5% royalty on all net revenues of any drug developed from the transferred compounds. As of June 30, 2020, accrued expenses include approximately $0.1 million related to the first milestone payments due to EHBE for the Company’s completion of a Phase 1 clinical study for MS and SSc.
F-33
Concurrent with the execution of the IPTA, the Company signed a Research Agreement with EHBE for an initial term of 5 years. Under the terms of the Research Agreement, EHBE is providing research services under the Company’s direction for consideration of cost plus a standard mark-up. Thereafter, the agreement will renew for successive one-year terms and may be terminated by either party on the expiration of the original term or any renewal term by delivering written notice at least 90 days prior to expiration. During the six months ended June 30, 2020 and 2019, the Company recorded $63,386 and $93,833, respectively in research and development expense for services performed by EHBE under the Research Agreement. As of June 30, 2020 and December 31, 2019, $39,582 and $58,300, respectively, are included in accrued expenses and accounts payable for amounts due to EHBE under the Research Agreement.
The Company performed a qualitative analysis to determine whether a variable interest in another entity represents a controlling financial interest in a variable interest entity. A controlling financial interest in a variable interest entity is characterized by having both the power to direct the most significant activities of the entity and the obligation to absorb losses or the right to receive benefits of the entity. Since EHP does not have voting control or other forms of control over the operations and decision making at EHBE, the Company determined that it does not have a variable interest in EHBE. This guidance requires on-going reassessments of variable interests based on changes in facts and circumstances. The Company continues to assess its variable interests and has determined that no significant changes have occurred as of September 25, 2020.
8. Balance Sheet Details
Other current assets consisted of the following:
| June 30, 2020 | December 31, 2019 | |||||||
| Prepaid contracts and expenses | $ | 727,745 | $ | 468,232 | ||||
| Related party receivables | 108,621 | 37,260 | ||||||
| Other | 45,290 | 42,788 | ||||||
| Total | $ | 881,656 | $ | 548,280 | ||||
Property and equipment consisted of the following:
| June 30, 2020 | December 31, 2019 | |||||||
| Furniture and fixtures | $ | 57,195 | $ | 57,195 | ||||
| Office equipment | 19,480 | 19,480 | ||||||
| Leasehold improvements | 20,638 | 20,638 | ||||||
| Property and equipment, gross | 97,313 | 97,313 | ||||||
| Accumulated depreciation | (58,066 | ) | (44,855 | ) | ||||
| Property and equipment, net | $ | 39,247 | $ | 52,458 | ||||
Depreciation expense for the six months ended June 30, 2020 was $13,211.
Accrued expenses are comprised of the following:
| June 30, 2020 | December 31, 2019 | |||||||
| Research and development liabilities | $ | 301,392 | $ | 355,942 | ||||
| Clinical trial related liabilities | 308,964 | 205,884 | ||||||
| Accrued payroll liabilities | 1,125,133 | 807,662 | ||||||
| Related party liabilities | 149,729 | 536,505 | ||||||
| Other liabilities | 97,853 | 71,634 | ||||||
| Total | $ | 1,983,071 | $ | 1,977,627 | ||||
F-34
9. Defined Contribution Plan
Effective January 1, 2018, the Company adopted a defined contribution savings plan pursuant to Section 401(k) of the Internal Revenue Code. The plan is for the benefit of all qualifying employees and permits voluntary contributions by employees up to 100% of eligible compensation, subject to the Internal Revenue Service imposed maximum limits. The terms of the plan allow for discretionary employer contributions.
On January 1, 2020, the Company commenced a safe harbor contribution of 3% of each eligible employee’s gross earnings, subject to Internal Revenue Service limitations. Employer matching contributions vest immediately.
10. Subsequent Events
As of September 24, 2020, the Company has completed the sale of 214,574 additional shares of common stock at $6.00 per share under its Offering pursuant to Regulation A, for gross proceeds of approximately $1.3 million. In addition, the Company has received subscription agreements under the Offering for the purchase of up to 3,323,066 shares of common stock for an estimated $19.9 million, which are still in process as of September 24, 2020. In total, as of September 24, 2020, since the commencement of the Offering in March 2019, the Company has received commitments for the sale of 8,107,651 shares of common stock pursuant to the Offering for estimated total gross proceeds of $46.1 million (inclusive of both sales and pending sales in process).
In July 2020, the company entered into an agreement to sublease a portion of its existing non-cancelable building lease to a tenant, effective August 1, 2020, and continuing through August 31, 2022. The Company paid commissions of $7,000 related to the execution of the sublease and expects to incur an additional loss of approximately $55,000 as a result of the transaction.
In July 2020, the Company entered into a consulting agreement with a third party to provide business advisory services in connection with strategic development and private financing matters. Pursuant to this agreement, the Company will pay a consulting fee in the amount as shall be determined in good faith by the Board of Directors of the Company in evaluating the services performed by the consultant. The agreement terminates on December 31, 2020 but may be extended by mutual consent of both parties.
F-35