AN OFFERING STATEMENT PURSUANT TO REGULATION A RELATING TO THESE SECURITIES HAS BEEN FILED WITH THE SECURITIES AND EXCHANGE COMMISSION. INFORMATION CONTAINED IN THIS PRELIMINARY OFFERING CIRCULAR IS SUBJECT TO COMPLETION OR AMENDMENT. THESE SECURITIES MAY NOT BE SOLD NOR MAY OFFERS TO BUY BE ACCEPTED BEFORE THE OFFERING STATEMENT FILED WITH THE COMMISSION IS QUALIFIED. THIS PRELIMINARY OFFERING CIRCULAR SHALL NOT CONSTITUTE AN OFFER TO SELL OR THE SOLICITATION OF AN OFFER TO BUY NOR MAY THERE BE ANY SALES OF THESE SECURITIES IN ANY STATE IN WHICH SUCH OFFER, SOLICITATION OR SALE WOULD BE UNLAWFUL BEFORE REGISTRATION OR QUALIFICATION UNDER THE LAWS OF SUCH STATE. THE COMPANY MAY ELECT TO SATISFY ITS OBLIGATION TO DELIVER A FINAL OFFERING CIRCULAR BY SENDING YOU A NOTICE WITHIN TWO BUSINESS DAYS AFTER THE COMPLETION OF THE COMPANY’S SALE TO YOU THAT CONTAINS THE URL WHERE THE FINAL OFFERING CIRCULAR OR THE OFFERING STATEMENT IN WHICH SUCH FINAL OFFERING CIRCULAR WAS FILED MAY BE OBTAINED.
PRELIMINARY OFFERING CIRCULAR DATED FEBRUARY 20, 2018
NowRx, Inc.

2224 Old Middlefield Way, Mountain View, CA 94043
(650) 386-5761
www.nowrx.com
UP TO 3,500,000 SHARES OF SERIES A PREFERRED STOCK
UP TO 3,500,000 SHARES OF COMMON STOCK INTO WHICH THE SERIES A PREFERRED STOCK MAY CONVERT*
SEE “SECURITIES BEING OFFERED” AT PAGE 52
MINIMUM INVESTMENT:
We are offering a minimum number of 225,000 shares of Series A Preferred Stock and a maximum number of 3,500,000 shares of Series A Preferred Stock on a "best efforts" basis.
| Series A Preferred Stock | Price Per Share to public | Underwriting discount and commissions** | Proceeds to issuer | |||||||||
| Total Minimum | $ | 2.00 | $ | 33,750 | $ | 416,250 | ||||||
| Total Maximum | $ | 2.00 | $ | 525,000 | $ | 6,475,000 | ||||||
| 1 |
*The Series A Preferred Stock is convertible into Common Stock either at the discretion of the investor or automatically upon the occurrence of certain events, like effectiveness of registration of the Common Stock in an initial public offering. The total number of shares of the Common Stock into which the Series A Preferred Stock may be converted will be determined by dividing the original issue price per share by the conversion price per share. See “Securities Being Offered” at page 52 for additional details.
** The company has engaged SI Securities, LLC to serve as its sole and exclusive placement agent to assist in the placement of its securities.The company will pay SI Securities, LLC in accordance with the terms of the Issuer Agreement between the company and SI Securities, LLC, a copy of which is filed as an exhibit to the Offering Statement of which this Offering Circular is a part. If the placement agent identifies all the investors and the maximum amount of shares is sold, the maximum amount the company would pay SI Securities, LLC is $525,000. Thisdoes not include transaction fees paid directly to SI Securities, LLC by investors. See “Plan of Distribution and Selling Securityholders” for details of compensation and transaction fees to be paid to the placement agent on page 59.
The company expects that the amount of expenses of the offering that it will pay will be approximately $75,000, not including commissions or state filing fees.
The company is selling shares of Series A Preferred Stock. Investors who invest less than $50,000 in this offering will be required to grant a proxy to vote their shares to SI Securities, LLC; see “Risk Factors” and “Securities Being Offered – Investor Proxy Agreement.” This means voting control of the company will be in the hands of persons other than those investors.
The company has engaged The Bryn Mawr Trust Company of Delaware as an escrow agent (the “Escrow Agent”) to hold funds tendered by investors, and assuming we sell a minimum of $450,000 in shares, may hold a series of closings at which we receive the funds from the escrow agent and issue the shares to investors. The offering will terminate at the earlier of: (1) the date at which the maximum offering amount has been sold, (2) one year from the date upon which the Securities and Exchange Commission qualifies the Offering Statement of which this Offering Circular forms a part, or (3) the date at which the offering is earlier terminated by the company in its sole discretion. In the event we have not sold the minimum amount of shares by _________, 2018, or sooner terminated by the company, any money tendered by potential investors will be promptly returned by the Escrow Agent. The company may undertake one or more closings on a rolling basis once the minimum offering amount is sold. After each closing, funds tendered by investors will be available to the company. The offering is being conducted on a best-efforts basis.
THE UNITED STATES SECURITIES AND EXCHANGE COMMISSION DOES NOT PASS UPON THE MERITS OR GIVE ITS APPROVAL OF ANY SECURITIES OFFERED OR THE TERMS OF THE OFFERING, NOR DOES IT PASS UPON THE ACCURACY OR COMPLETENESS OF ANY OFFERING CIRCULAR OR OTHER SOLICITATION MATERIALS. THESE SECURITIES ARE OFFERED PURSUANT TO AN EXEMPTION FROM REGISTRATION WITH THE COMMISSION; HOWEVER THE COMMISSION HAS NOT MADE AN INDEPENDENT DETERMINATION THAT THE SECURITIES OFFERED ARE EXEMPT FROM REGISTRATION
| 2 |
GENERALLY NO SALE MAY BE MADE TO YOU IN THIS OFFERING IF THE AGGREGATE PURCHASE PRICE YOU PAY IS MORE THAN 10% OF THE GREATER OF YOUR ANNUAL INCOME OR NET WORTH. DIFFERENT RULES APPLY TO ACCREDITED INVESTORS AND NON-NATURAL PERSONS. BEFORE MAKING ANY REPRESENTATION THAT YOUR INVESTMENT DOES NOT EXCEED APPLICABLE THRESHOLDS, WE ENCOURAGE YOU TO REVIEW RULE 251(d)(2)(i)(C) OF REGULATION A. FOR GENERAL INFORMATION ON INVESTING, WE ENCOURAGE YOU TO REFER TOwww.investor.gov.
This offering is inherently risky. See “Risk Factors” on page 12.
Sales of these securities will commence on approximately [date].
The company is following the “Offering Circular” format of disclosure under Regulation A.
| 3 |
TABLE OF CONTENTS
In this Offering Circular, the term “NowRx,” ”we,” “us” or “the company” refers to NowRx, Inc.
THIS OFFERING CIRCULAR MAY CONTAIN FORWARD-LOOKING STATEMENTS AND INFORMATION RELATING TO, AMONG OTHER THINGS, THE COMPANY, ITS BUSINESS PLAN AND STRATEGY, AND ITS INDUSTRY. THESE FORWARD-LOOKING STATEMENTS ARE BASED ON THE BELIEFS OF, ASSUMPTIONS MADE BY, AND INFORMATION CURRENTLY AVAILABLE TO THE COMPANY’S MANAGEMENT. WHEN USED IN THE OFFERING MATERIALS, THE WORDS “ESTIMATE,” “PROJECT,” “BELIEVE,” “ANTICIPATE,” “INTEND,” “EXPECT” AND SIMILAR EXPRESSIONS ARE INTENDED TO IDENTIFY FORWARD-LOOKING STATEMENTS, WHICH CONSTITUTE FORWARD LOOKING STATEMENTS. THESE STATEMENTS REFLECT MANAGEMENT’S CURRENT VIEWS WITH RESPECT TO FUTURE EVENTS AND ARE SUBJECT TO RISKS AND UNCERTAINTIES THAT COULD CAUSE THE COMPANY’S ACTUAL RESULTS TO DIFFER MATERIALLY FROM THOSE CONTAINED IN THE FORWARD-LOOKING STATEMENTS. INVESTORS ARE CAUTIONED NOT TO PLACE UNDUE RELIANCE ON THESE FORWARD-LOOKING STATEMENTS, WHICH SPEAK ONLY AS OF THE DATE ON WHICH THEY ARE MADE. THE COMPANY DOES NOT UNDERTAKE ANY OBLIGATION TO REVISE OR UPDATE THESE FORWARD-LOOKING STATEMENTS TO REFLECT EVENTS OR CIRCUMSTANCES AFTER SUCH DATE OR TO REFLECT THE OCCURRENCE OF UNANTICIPATED EVENTS.
| 4 |
Overview
NowRx is an on-demand pharmacy, leveraging the latest in software technology, artificial intelligence, robotics and logistics to provide the most convenient pharmacy experience available, with free same-day delivery of prescription medications, thereby avoiding the need to ever visit the pharmacy. Expedited one-hour delivery is also offered for a $5.00 charge. All pharmacy services are provided from a low cost, highly automated “virtual pharmacy” location, utilizing end-to-end robotic dispensing (“One-Click Fill”SM) and artificially intelligent chat bots, coupled with NowRx drivers and plug-in electric vehicles, to provide a more efficient and effective pharmacy experience for busy customers. Customers can easily and conveniently access NowRx services through the NowRx mobile app, text, telephone or through virtual voice-activated assistants such as Google Home.
NowRx’s business automatically generates a substantial database recording a wealth of medical prescription data affording the company the opportunity to develop, using machine-learning techniques, powerful tools to initiate pharmacy interventions for patients more likely to miss a dose or refill (NowRx Medication Adherence IndexSM), thereby providing better medication adherence and improved health outcomes.
NowRx is part of the rapidly growing on-demand economy, which is increasingly attracting consumers who are drawn by the user experience, added convenience and other benefits it provides. Recent studies show on-demand services have already moved beyond early adopters and are gaining traction among mainstream consumers who require that platforms be user-friendly and safe1.
Our Products and Services
NowRx is a fully licensed retail pharmacy that provides free same-day delivery of prescription drugs and some over-the-counter medications. For an additional $5.00, the company provides delivery within 1 hour of placing an order for in-stock pharmaceuticals. We offer our services through the NowRx app, by text, by telephone and through virtual assistants such as Google Home. Physicians are able to send prescriptions to NowRx through electronic prescribing, fax, the NowRx app or telephone. Current services provided include fulfilling new prescriptions or refills, transferring prescriptions from other pharmacies, pharmacist consultation via phone or secure video chat, and application of drug manufacturer coupons.
1 ”The On-Demand Economy is Growing, and Not Just for the Young and Wealthy”, Harvard Business Review, April 14 2016.
| 5 |
The Opportunity
NowRx is responding to the rapidly increasing consumer demand for services delivered same-day and managed by convenience of mobile apps, chat bots and voice-activated assistants. The on-demand economy has already attracted more than 22.4 million consumers annually and $57.6 billion in spending.2 Retail pharmacy is a $260 billion industry.3 We believe it’s only a matter of time before the bulk of the pharmacy industry will fulfill customer needs through same-day delivery and customers standing in line at a pharmacy counter will be a thing of the past. By offering a much more convenient, efficient, and zero cost service, NowRx eliminates the need for its customers to ever visit crowded pharmacies, enabling them to stay at home with their families or remain in their office or at their place of business. Furthermore, NowRx increases the likelihood of timely prescription fills, encourages medication adherence, reduces the number of missed refills, ultimately reducing costs to health plan insurers and providing better consumer health. For employers, NowRx can improve worker productivity by eliminating time employees spend off-campus having prescriptions filled and reduce overall health plan costs through better medication compliance.
Strategy
NowRx believes that its service can take advantage of this trend toward enhanced convenience with a four-pronged strategy embracing:
| 1. | Technology strategy. The focus of NowRx’s technology strategy, in addition to a unified communication platform, is to build a powerful back office technology infrastructure incorporating end-to-end robotic dispensing and delivery logistics. One priority for NowRx is to develop an in-house pharmacy management system incorporating an e-prescribing module and an insurance processing module, which would then be plugged directly into the company’s existing dispensing system. This would allow complete end-to-end automation from the moment a drug is ordered to the moment it is fulfilled by the robot dispensing system (other than the confirmation by the pharmacist). |
| 2. | Product strategy. NowRx will continue to evolve its product offering, focusing on two areas: rendering the customer experience as easy and convenient as possible, and secondly leveraging the company database to improve medication adherence. The company currently enables prescriptions to be ordered via the NowRx app, text, fax, email or telephone as well as through technology assistant tools such as Google Home. Each of these tools will continue to be upgraded to assure the most convenient, simplest, and fastest possible means of completing an order. For example, customers are increasingly interested in using standard text commands, which currently requires a NowRx employee to interpret. NowRx intends to develop a text based “chat bot” using artificial intelligence that will interpret customer’s requests, thereby fully automating the process. |
2 ”The On-Demand Economy is Growing, and Not Just for the Young and Wealthy”, Harvard Business Review, April 14 2016.
3 “The 2016 Economic Report on Retail, Mail, and Specialty Pharmacies”, Pembroke Consulting, January 2016.
| 6 |
| 3. | Geographical coverage strategy. NowRx expects in the next 18 months to cover the entire San Francisco Bay Area metropolitan area, in addition to opening locations in the Los Angeles basin. Additional locations are possible in this timeframe in neighboring cities such as San Diego or in another state such as Washington or Arizona. In the long-term the company seeks to cover the top twenty metropolitan areas in the United States. Geographical coverage is driven by a clustering approach, opening successive locations contiguous to an existing location (typically about 10 miles distant), thereby providing seamless service in a given region. We believe that clustering, by virtue of higher customer density, provides the most cost efficient vehicle both operationally (lowest possibleprescription fulfillment and delivery cost) and from a marketing perspective (lowest possible customer acquisition cost). |
| 4. | Marketing strategy. In the short-term, NowRx will continue to focus its marketing strategy on resource efficient channels such as physicians and healthcare facilities. Physician referrals carry considerable weight with patients who see little downside in trying the service. We believe the clustering approach will result in an increasingly lower customer acquisition cost as the company adds more contiguous locations. Word of mouth both from physicians and consumers themselves grows geometrically as the company expands its base. Consumer marketing via social media and other traditional media channels will be highly selective in the short term but grow as the company scales up in a given region in which it has achieved a critical mass of customers. |
Highlights and Metrics
| · | Revenues for the first six months of June 2017 were $994,588, an increase of 563% over the same period in 2016. |
| · | As of June 2017, NowRx had filled more than 16,000 prescriptions and served more than 2,700 customers. |
| · | Scalable growth model: Customer Acquisition Cost (CAC) is approximately $40 and Lifetime Value (LTV) is approximately $370.4 |
| · | NowRx had 945 referring physicians as of June 2017, an increase of 348% over June 2016. |
| · | The company engineered a HIPPA-compliant implementation and recently received approval as the first healthcare app in Google Home. Furthermore, Google Home’s discovery feature recognizes any users in our delivery area utilizing the key word “prescription” and prompts them to try NowRx. |
| · | Recently received an unsolicited request to serve as the recommended pharmacy for an assisted living facility (188 residents), an account NowRx successfully won in competition with Walgreens. |
| · | NowRx is advantaged over other startup competitors (NimbleRx, Alto, & Capsule) through: |
| · | 1-hour delivery capability; |
| · | “One-Click Fill” - end-to-end robotic dispensing; |
4 Customer Acquisition Cost (CAC) is calculated by dividing the total sales and marketing expense from inception through June 2017 by the number of new customers acquired over the same period. Lifetime Value (LTV) is calculated by projecting the cumulative gross margin contributed, based on the existing gross margin to date, projected over 5 years by the average customer.
| 7 |
| · | Unified communication platform with HIPAA-compliant texting solution; |
| · | Logistics algorithms for optimized refill routing. |
| · | Seasoned team with more than $1 billion in exits and a proven track record; |
| · | Proven ability to execute in a highly capital efficient manner, having built a complete initial technology platform and achieved monthly revenues over $200,000 with $2.1 million in capital raised to date. |
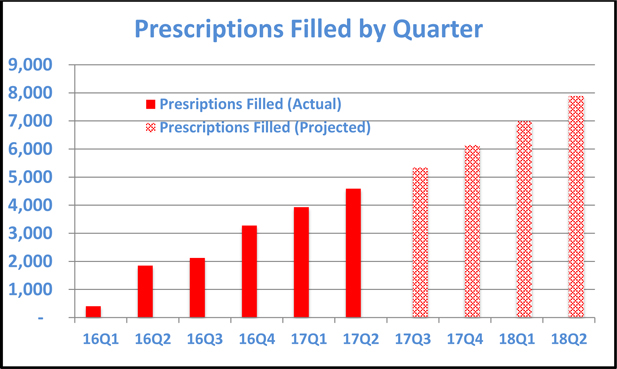
The following table illustrates the number of prescriptions filled in each quarter since the company commenced operations and projects near-term growth assuming that the historic trend continues.
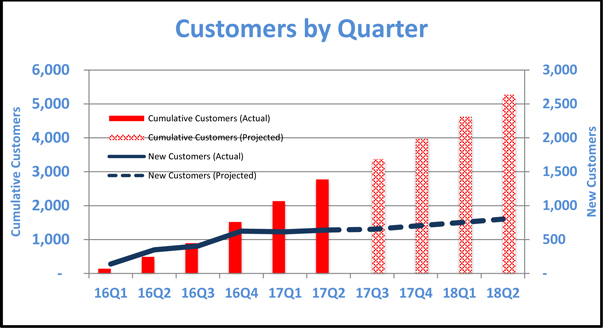
The following table illustrates (a) on the left axis the growth in total number of customers since the company commenced operations and projects near-term growth assuming that the rate of growth continues and (b) on the right axis the total number of new customers added each quarter since the company commenced operations and projects near-term growth assuming that the company acquires new customers at the same rate.
| 8 |
The Offering
| Securities offered: | Maximum of 3,500,000 shares of Series A Preferred Stock convertible into shares of Common Stock | |
| Series A Preferred Stock outstanding before the offering (1): | 4,282,738 shares | |
| Common Stock outstanding before the offering: (2) | 8,500,000 shares | |
| Use of proceeds: | The net proceeds of the offering will be used to continue building our software system, conduct research and development, support expansion and operations of our business and fund sales and marketing. |
(1) Assumes (a) conversion of all outstanding SAFE and KISS securities and Crowd Notes and all securities resulting from the exercise of warrants attached to certain outstanding SAFE securities into shares of Series A Preferred Stock and (b) payment of accrued interest on the KISS securities in cash immediately prior to the initial closing of this offering.
(2) Does not include shares issuable upon the exercise of options issued under the 2015 Stock Incentive Plan, shares allocated for issuance pursuant to the plan, outstanding warrants or shares into which the Series A Preferred Stock may convert.
| 9 |
Selected Risks Associated with Our Business
Our business is subject to a number of risks and uncertainties, including those highlighted in the section titled “Risk Factors” immediately following this summary. These risks include, but are not limited to, the following:
| · | We could be adversely affected by a decrease in the introduction of new brand name and generic prescription drugs as well as increases in the cost to procure prescription drugs. |
| · | We derive a significant portion of our sales from prescription drug sales reimbursed by a limited number of pharmacy benefit management companies. |
| · | Reductions in third party reimbursement levels, from private or government agency plans, and potential changes in industry pricing benchmarks for prescription drugs could materially and adversely affect our results of operations. |
| · | A shift in pharmacy mix toward lower margin plans and programs could adversely affect our results of operations. |
| · | We operate in a highly competitive industry that is dominated by several very large, well-capitalized market leaders and constantly evolving. New entrants to the market, existing competitor actions, or other changes in market dynamics could adversely impact us. |
| · | Consolidation in the healthcare industry could adversely affect us. |
| · | We purchase a substantial portion of our brand name and generic drugs from a single wholesaler. A disruption in this relationship may have a negative effect on us. |
| · | If we do not maintain the privacy and security of sensitive customer and business information, it could damage our reputation and we could suffer a loss of revenue, incur substantial additional costs and become subject to litigation and regulatory scrutiny. |
| · | Because we store, process and use data that contains personal information, we are subject to complex and evolving laws and regulations regarding privacy and data protection. Many of these laws and regulations are subject to change and uncertain interpretation, and could result in investigations, claims, changes to our business practices, increased cost of operations, and declines in customer retention, any of which could harm our business. |
| · | We are subject to payment-related risks that could increase our operating costs, expose us to fraud or theft, subject us to potential liability and potentially disrupt our business operations. |
| · | A significant change in, or noncompliance with, government regulations and other legal requirements could have a material adverse effect on our reputation and profitability. |
| · | We could be adversely affected by product liability, product recall, personal injury or other health and safety issues. |
| · | We only operate from one location. If we do not establish additional locations, we may not be able to successfully implement growth. |
| · | We could be adversely affected by a failure to correctly deliver prescriptions. |
| · | Our failure to attract and retain highly qualified personnel in the future could harm our business. |
| 10 |
| · | The company has a history of losses, and may not achieve or maintain profitability in the future. |
| · | We expect to raise additional capital through equity and/or debt offerings to support our working capital requirements and operating losses. |
| · | The auditor included a “going concern” note in its audit report. |
| · | The company is controlled by its officers and directors. |
| · | Other investors may have greater rights than you. |
| · | This investment is illiquid. |
| 11 |
The Commission requires the company to identify risks that are specific to its business and its financial condition. The company is still subject to all the same risks that all companies in its business, and all companies in the economy, are exposed to. These include risks relating to economic downturns, political and economic events and technological developments (such as hacking and the ability to prevent hacking). Additionally, early-stage companies are inherently more risky than more developed companies. You should consider general risks as well as specific risks when deciding whether to invest.
We could be adversely affected by a decrease in the introduction of new brand name and generic prescription drugs as well as increases in the cost to procure prescription drugs. The profitability of our business depends upon the utilization of prescription drugs. Utilization trends are affected by, among other factors, the introduction of new and successful prescription drugs as well as lower-priced generic alternatives to existing brand name drugs. Inflation in the price of drugs also can adversely affect utilization, particularly given the increased prevalence of high-deductible health insurance plans and related plan design changes. New brand name drugs can result in increased drug utilization and associated sales, while the introduction of lower priced generic alternatives typically results in relatively lower sales, but relatively higher gross profit margins. Accordingly, a decrease in the number or magnitude of significant new brand name drugs or generics successfully introduced, delays in their introduction, or a decrease in the utilization of previously introduced prescription drugs could materially and adversely affect our results of operations.
In addition, if we experience an increase in the amounts we pay to procure pharmaceutical drugs, including generic drugs, it could have a material adverse effect on our results of operations. Our gross profit margins would be adversely affected to the extent we are not able to offset such cost increases. Any failure to fully offset any such increased prices and costs or to modify our activities to mitigate the impact could have a material adverse effect on our results of operations. Additionally, any future changes in drug prices could significantly differ from our expectations.
We derive a significant portion of our sales from prescription drug sales reimbursed by a limited number of pharmacy benefit management companies and other third party payors. We derive a significant portion of our sales from prescription drug sales reimbursed through prescription drug plans administered by a limited number of pharmacy benefit management (“PBM”) companies. PBM companies typically administer multiple prescription drug plans that expire at various times and provide for varying reimbursement rates, and often limit coverage to specific drug products on an approved list, known as a formulary, which might not include all of the approved drugs for a particular indication. We cannot assure you that we will continue to participate in any particular PBM company’s pharmacy provider network in any particular future time period. If our participation in the pharmacy provider network for a prescription drug plan administered by one or more of the large PBM companies is restricted or terminated, we expect that our sales would be adversely affected, at least in the short-term. If we are unable to replace any such lost sales, either through an increase in other sales or through a resumption of participation in those plans, our operating results could be materially and adversely affected. If we exit a pharmacy provider network and later resume participation, we cannot assure you that we will achieve any particular level of business on any particular pace, or that all clients of the PBM company will choose to include us again in the pharmacy network for their plans, initially or at all. In addition, in such circumstances we may incur increased marketing and other costs in connection with initiatives to regain former patients and attract new patients covered by such plans.
| 12 |
Reductions in third party reimbursement levels, from private or government agency plans, and potential changes in industry pricing benchmarks for prescription drugs could materially and adversely affect our results of operations. The substantial majority of the prescriptions we fill are reimbursed by third-party payors, including private and government agency payors. The continued efforts of health maintenance organizations, managed care organizations, PBM companies, government agencies, and other third-party payors to reduce prescription drug costs and pharmacy reimbursement rates, as well as litigation and other legal proceedings relating to how drugs are priced, may adversely impact our results of operations. Typically, health plan changes with rate adjustments often occur in January and our reimbursement arrangements may provide for rate adjustments at prescribed intervals during their term. In addition, in an environment where some PBM clients utilize narrow or restricted pharmacy provider networks, some of these entities may offer pricing terms that we may not be willing to accept or otherwise restrict our participation in their networks of pharmacy providers.
Changes in political, economic and regulatory influences also may significantly affect healthcare financing and prescription drug reimbursement practices. For example, there have been multiple attempts through legislative action and legal challenges to repeal or amend the Patient Protection and Affordable Care Act (“ACA”). We cannot predict whether current or future efforts to repeal or amend these laws will be successful, nor can we predict the impact that such a repeal or amendment and any subsequent legislation would have on our business and reimbursement levels. There have also been a number of other proposals and enactments by the federal government and various states to reduce Medicare Part D and Medicaid reimbursement levels in response to budget deficits, and we expect additional proposals in the future. We cannot assure you that recent or future changes in prescription drug reimbursement policies and practices will not materially and adversely affect our results of operations. Efforts to control healthcare costs, including prescription drug costs, are continuous and reductions in third party reimbursement levels could materially and adversely affect our results of operations.
In addition, many payors are increasingly considering new metrics as the basis for reimbursement rates, such as average sales price, average manufacturer price, and actual acquisition cost. It is possible that the pharmaceutical industry or regulators may evaluate and/or develop an alternative pricing reference to replace average wholesale price, which is the pricing reference used for many of our contracts. Future changes to the pricing benchmarks used to establish pharmaceutical pricing, including changes in the basis for calculating reimbursement by third-party payors, could adversely affect us.
| 13 |
A shift in pharmacy mix toward lower margin plans and programs could adversely affect our results of operations. We seek to grow prescription volume while operating in a marketplace with continuous reimbursement pressure. A shift in the mix of pharmacy prescription volume towards 90-day, Medicare or other programs offering lower reimbursement rates could adversely affect our results of operations. We currently offer limited 90-day fulfillments. In addition, preferred Medicare Part D networks have increased in number in recent years; however, we do not participate in all such networks. In the future, we may accept lower reimbursement rates in order to secure preferred relationships with Medicare Part D plans serving senior patients with significant pharmacy needs. We intend to develop and expand our relationships with commercial third-party payors to enable new and/or improved market access via participation in the pharmacy provider networks they offer. If we are not able to generate additional prescription volume from patients participating in these programs that is sufficient to offset the impact of lower reimbursement, or if the degree or terms of our participation in such preferred networks declines from current levels in future years, our results of operations could be materially and adversely affected.
We operate in a highly competitive industry that is dominated by several very large, well-capitalized market leaders and constantly evolving. New entrants to the market, existing competitor actions, or other changes in market dynamics could adversely impact us. The level of competition in the retail pharmacy and pharmaceutical wholesale industries is high, with several very large, well-capitalized competitors holding a majority share of the market. Changes in market dynamics or actions of competitors or manufacturers, including industry consolidation and the emergence of new competitors and strategic alliances, could materially and adversely impact our business. Disruptive innovation by existing or new competitors could alter the competitive landscape in the future and require us to accurately identify and assess such changes and make timely and effective changes to our strategies and business model to compete effectively. We face intense competition from local, regional, national and global companies, including drugstore and pharmacy chains, independent drugstores and pharmacies, mail-order pharmacies and various other online retailers, some of which are aggressively expanding in California and markets we may seek to enter. Competition may also come from other sources in the future. As competition increases, a significant increase in general pricing pressures could occur, which could require us to reevaluate our pricing structures to remain competitive. For example, if we are not able to anticipate and successfully respond to changes in market conditions, it could result in a loss of customers or renewal of contracts or arrangements on less favorable terms.
| 14 |
Consolidation in the healthcare industry could adversely affect us. Many organizations in the healthcare industry have consolidated in recent years to create larger healthcare enterprises with greater bargaining power, which has resulted in greater pricing pressures on pharmaceuticals. If this consolidation trend continues, it could give the resulting enterprises even greater bargaining power, which may lead to further pressure on the prices for our products and services. If these pressures result in reductions in our prices, our business would become less profitable unless we are able to achieve corresponding reductions in costs or develop profitable new revenue streams.
We expect that market demand, government regulation, third-party reimbursement policies, government contracting requirements, and societal pressures will continue to cause the healthcare industry to evolve, potentially resulting in further business consolidations and alliances and increased vertical integration among the industry participants we engage with, which may adversely impact our business operations, financial condition and results of operations.
We purchase a substantial portion of our brand name and generic drugs from a single wholesaler. A disruption in this relationship may have a negative effect on us.We purchase approximately 70% of our brand name and generic drugs from a single wholesaler, McKesson. The remaining 30% is sourced from Independent Pharmacy Cooperative (“IPC”) and several small suppliers. Because McKesson acts as a wholesaler for drugs purchased from ultimate manufacturers worldwide, any disruption in the supply of a given drug, including supply shortages of key ingredients, or regulatory actions by domestic or foreign government agencies, or specific actions taken by drug manufacturers, could adversely impact McKesson's ability to fulfill our demands, which could adversely affect us. While we believe that alternative sources of supply for most generic and brand name pharmaceuticals are readily available, a significant disruption in our relationship with McKesson or IPC could make it difficult for us to continue to operate our business on a regular basis until we execute a replacement wholesaler agreement or develop and implement self-distribution processes. We believe we could obtain and qualify alternative sources, including through self-distribution, for substantially all of the prescription drugs we sell on an acceptable basis, and accordingly that the impact of any disruption would be temporary.
| 15 |
If we do not maintain the privacy and security of sensitive customer and business information, it could damage our reputation and we could suffer a loss of revenue, incur substantial additional costs and become subject to litigation and regulatory scrutiny.Our operations are dependent on our information systems and the information collected, processed, stored, and handled by these systems. We rely heavily on our computer systems to manage our ordering, pricing, fulfillment, inventory replenishment, claims processing and other processes. Throughout our operations, we receive, retain and transmit certain confidential information, including personally identifiable information that our customers provide to purchase products or services, interact with our personnel, or otherwise communicate with us. In addition, for these operations, we depend in part on the secure transmission of confidential information over public networks. Our information systems are subject to damage or interruption from power outages, facility damage, computer and telecommunications failures, computer viruses, security breaches, including credit card or personally identifiable information breaches, coordinated cyber attacks, vandalism, catastrophic events and human error. Although we deploy a layered approach to address information security threats and vulnerabilities, including ones from a cyber security standpoint, designed to protect confidential information against data security breaches, a compromise of our information security controls or of those businesses with whom we interact, which results in confidential information being accessed, obtained, damaged, or used by unauthorized or improper persons, could harm our reputation and expose us to regulatory actions and claims from customers, financial institutions, payment card associations and other persons, any of which could adversely affect our business, financial position, and results of operations. Because the techniques used to obtain unauthorized access, disable or degrade service, or sabotage systems change frequently and may not immediately produce signs of intrusion, we may not be able to anticipate these techniques or to implement adequate preventative measures. In addition, a security breach could require that we expend substantial additional resources related to the security of information systems and disrupt our businesses.
Because we store, process and use data that contains personal information, we are subject to complex and evolving laws and regulations regarding privacy and data protection. Many of these laws and regulations are subject to change and uncertain interpretation, and could result in investigations, claims, changes to our business practices, increased cost of operations, and declines in customer retention, any of which could harm our business. The regulatory environment surrounding information security and privacy is increasingly demanding, with the frequent imposition of new and changing requirements across businesses. We are required to comply with increasingly complex and changing data privacy regulations. Complying with these and other changing requirements could cause us to incur substantial costs and require us to change our business practices in certain jurisdictions, any of which could materially adversely affect our business operations and operating results. We may also face audits or investigations by one or more government agencies relating to our compliance with these regulations. Compliance with changes in privacy and information security laws and standards may result in significant expense due to increased investment in technology and the development of new operational processes. If we or those with whom we share information fail to comply with these laws and regulations or experience a data security breach, our reputation could be damaged and we could be subject to additional litigation and regulatory risks. Our security measures may be undermined due to the actions of outside parties, employee error, malfeasance, or otherwise, and, as a result, an unauthorized party may obtain access to our data systems and misappropriate business and personal information. Any such breach or unauthorized access could result in significant legal and financial exposure, damage to our reputation, and potentially have a material adverse effect on our business operations, financial condition and results of operations.
| 16 |
We are subject to payment-related risks that could increase our operating costs, expose us to fraud or theft, subject us to potential liability and potentially disrupt our business operations. We accept payments using a variety of methods, including cash, checks, credit and debit cards, gift cards and mobile payment technologies such as Apple Pay™, and we may offer new payment options over time. Acceptance of these payment options subjects us to rules, regulations, contractual obligations and compliance requirements, including payment network rules and operating guidelines, data security standards and certification requirements, and rules governing electronic funds transfers. These requirements and related interpretations may change over time, which could make compliance more difficult or costly. For certain payment methods, including credit and debit cards, we pay interchange and other fees, which could increase over time and raise our operating costs. We rely on third parties to provide payment-processing services, including the processing of credit cards, debit cards, and other forms of electronic payment. If these companies become unable to provide these services to us, or if their systems are compromised, it could disrupt our business. The payment methods that we offer also subject us to potential fraud and theft by persons who seek to obtain unauthorized access to or exploit any weaknesses that may exist in the payment systems. If we fail to comply with applicable rules or requirements, or if data is compromised due to a breach or misuse of data relating to our payment systems, we may be liable for costs incurred by payment card issuing banks and other third parties or subject to fines and higher transaction fees, or our ability to accept or facilitate certain types of payments could be impaired. In addition, our reputation could suffer and our customers could lose confidence in certain payment types, which could result in higher costs. As a result, our business and operating results could be adversely affected.
A significant change in, or noncompliance with, government regulations and other legal requirements could have a material adverse effect on our reputation and profitability. We operate in a complex, highly regulated environment and our operations could be adversely affected by changes to existing legal requirements including the related interpretations and enforcement practices, new legal requirements and/or any failure to comply with applicable regulations. Our business is subject to numerous federal, state and local regulations including licensing and other requirements for pharmacies and reimbursement arrangements. The regulations to which we are subject include, but are not limited to: federal and state registration and regulation of pharmacies and drug discount card programs; dispensing and sale of controlled substances and products containing pseudoephedrine; applicable governmental payor regulations including Medicare and Medicaid; data privacy and security laws and regulations including those under the Health Insurance Portability and Accountability Act of 1996 (“HIPAA”); the ACA or any successor to that act; laws and regulations relating to the protection of the environment and health and safety matters, including those governing exposure to, and the management and disposal of, hazardous substances; regulations regarding food and drug safety including those of the Food and Drug Administration (“FDA”) and Drug Enforcement Administration (“DEA”), trade regulations including those of the Federal Trade Commission, and consumer protection and safety regulations including those of the Consumer Product Safety Commission, as well as state regulatory authorities, governing the availability, sale, advertisement and promotion of products we sell; anti-kickback laws; false claims laws; laws against the corporate practice of medicine; and federal and state laws governing health care fraud and abuse and the practice of the profession of pharmacy. For example, the DEA, FDA and various other regulatory authorities regulate the distribution and dispensing of pharmaceuticals and controlled substances. We are required to hold valid DEA and state-level licenses, meet various security and operating standards and comply with the federal and various state controlled substance acts and related regulations governing the sale, dispensing, disposal, holding and distribution of controlled substances. The DEA, FDA and state regulatory authorities have broad enforcement powers, including the ability to seize or recall products and impose significant criminal, civil and administrative sanctions for violations of these laws and regulations.
| 17 |
Changes in laws, regulations and policies and the related interpretations and enforcement practices may alter the landscape in which we do business and may significantly affect our cost of doing business. The impact of new laws, regulations and policies and the related interpretations and enforcement practices generally cannot be predicted, and changes in applicable laws, regulations and policies and the related interpretations and enforcement practices may require extensive system and operational changes, be difficult to implement, increase our operating costs and require significant capital expenditures. Untimely compliance or noncompliance with applicable laws and regulations could result in the imposition of civil and criminal penalties that could adversely affect the continued operation of our business, including: suspension of payments from government programs; loss of required government certifications; loss of authorizations to participate in or exclusion from government programs, including the Medicare and Medicaid programs; loss of licenses; and significant fines or monetary penalties. Any failure to comply with applicable regulatory requirements could result in significant legal and financial exposure, damage our reputation, and have a material adverse effect on our business operations, financial condition and results of operations.
We could be adversely affected by product liability, product recall, personal injury or other health and safety issues. We could be adversely impacted by the supply of defective or expired products, including the infiltration of counterfeit products into the supply chain, errors in re-labeling of products, product tampering, product recall and contamination or product mishandling issues. We are also exposed to risks relating to the services we provide. Errors in the dispensing and packaging of pharmaceuticals, including related counseling, and in the provision of other healthcare services could lead to serious injury or death. Product liability or personal injury claims may be asserted against us with respect to any of the pharmaceuticals we sell or services we provide. Should a product or other liability issue arise, the coverage limits under our insurance programs and the indemnification amounts available to us may not be adequate to protect us against claims and judgments. We also may not be able to maintain this insurance on acceptable terms in the future. We could suffer significant reputational damage and financial liability if we experience any of the foregoing health and safety issues or incidents, which could have a material adverse effect on our business operations, financial condition and results of operations.
| 18 |
We only operate from one location. If we do not establish additional locations, we may not be able to successfully implement growth. We currently operate out of one warehouse in the Bay Area. In order to grow our business and extend our market position, we will need to establish more warehouse locations in the Bay Area and establish operations in other patient-dense geographies in the United States. Our ability to expand the market for our products and services depends on a number of factors, including, among others, the cost of establishing and operating a warehouse location, customer acceptance of app or web-enabled ordering, our ability to scale customer acquisition and our ability to attract more physicians who can refer their patients to our services. If we are unable to expand to other locations and/or scale customer acquisition at an acceptable cost, we may not be able to successfully grow our business.
We could be adversely affected by a failure to correctly deliver prescriptions.A customer signature is required for approximately 80% of all deliveries. This includes all deliveries of narcotics or other controlled drugs, designated as Schedule 2 through 5. We require a customer to present picture identification to the driver in addition to signing for any such controlled drug deliveries. There is a risk for the remaining 20% of deliveries, for which we do not require a signature, that the delivery will be made incorrectly, whether because a prescription is delivered to the wrong address, the wrong medication is left at the right address, the right medication is left at the wrong address, the customer claims the medication was never delivered, or the medication is stolen by a third party. The company maintains an electronic record of the exact route taken by each driver and drivers are required to take a picture of the medication by the doorstep, thereby assuring that the delivery is fully documented. In addition, there is a risk that drivers, in a small number of cases, may fail to collect payment directly from a customer, in cases where the customer elects to pay by cash, or may fail to obtain a written prescription from the customer, in cases where the physician did not electronically submit the prescription. In such instances of mistaken delivery, we could suffer reputational damage or regulatory or financial harm.
Our failure to attract and retain highly qualified personnel in the future could harm our business. As the company grows, it will be required to hire and attract additional qualified professionals such as pharmacists, pharmacist technicians, accounting, legal, finance, service and engineering experts. The company may not be able to locate or attract qualified individuals for such positions, which will affect the company’s ability to grow and expand its business.
The company has a history of losses, and may not achieve or maintain profitability in the future. The company has operated at a loss since inception and has raised additional capital and borrowed funds to meet its growth needs. We expect to make significant future investments in order to develop and expand our business, which we believe will result in additional sales and marketing and general and administrative expenses that will require increased sales to recover these additional costs. While net sales have grown in recent periods, this growth may not be sustainable or sufficient to cover the costs required to successfully compete.
| 19 |
We expect to raise additional capital through equity and/or debt offerings to support our working capital requirements and operating losses. In order to fund future growth and development, the company will likely need to raise additional funds in the future by offering shares of its common or preferred stock and/or other classes of equity or debt that convert into shares of common or preferred stock, any of which offerings would dilute the ownership percentage of investors in this offering. See “Dilution.” Furthermore, if the company raises debt, the holders of the debt would have priority over holders of common and preferred stock and the company may accept terms that restrict its ability to incur more debt. We cannot assure you that the necessary funds will be available on a timely basis, on favorable terms, or at all, or that such funds if raised, would be sufficient. The level and timing of future expenditure will depend on a number of factors, many of which are outside our control. If we are not able to obtain additional capital on acceptable terms, or at all, we may be forced to curtail or abandon our growth plans, which could adversely impact the company, its business, development, financial condition, operating results or prospects.
The auditor included a “going concern” note in its audit report.We may not have enough funds to sustain the business until it becomes profitable. Even if we raise funds through this offering, we may not accurately anticipate how quickly we may use the funds and whether these funds are sufficient to bring the business to profitability.
Projected financial data is included in this Offering Circular; projections are frequently inaccurate. We include projected financial data in "Management's Discussion and Analysis of Financial Condition and Results of Operations – Trend Information – Order Trends." Those projected results will only be achieved if the assumptions they are based on are correct. There are many reasons why the assumptions could be inaccurate, including customer acceptance of our products, competition, general economic conditions and our own inability to execute our plans. Potential investors should take the assumptions in consideration when reading those projections, and consider whether they think they are reasonable.
The company is controlled by its officers and directors. The company’s officers and directors currently hold all of the company’s voting stock, and at the conclusion of this offering will continue to hold a majority of the company’s voting stock. Investors in this offering will not have the ability to control a vote by the stockholders or the board of directors.
Other investors may have greater rights than you. Investors who invest less than $50,000 in the company will be required to sign the Investor Proxy Agreement, which will grant an irrevocable proxy to SI Securities, LLC to vote their shares of Series A Preferred Stock and any shares of Common Stock they receive if the shares of Series A Preferred Stock are converted at a later date. Furthermore, transferees of the investors party to the agreement are obligated to also become parties to the agreement. SI Securities, LLC owns securities that will convert into shares of Series A Preferred Stock immediately prior the initial closing of the offering and may have interests that conflict with those of investors who are party to the agreement. Investors who invest more than $50,000 will not be required to sign the Investor Proxy Agreement. For more information regarding the agreement, see “Securities Being Offered – Investor Proxy Agreement.”
| 20 |
This investment is illiquid. There is no currently established market for reselling these securities. If you decide that you want to resell these securities in the future, you may not be able to find a buyer.
| 21 |
Dilution means a reduction in value, control or earnings of the shares the investor owns.
Immediate dilution
An early-stage company typically sells its shares (or grants options over its shares) to its founders and early employees at a very low cash cost, because they are, in effect, putting their “sweat equity” into the company. When the company seeks cash investments from outside investors, like you, the new investors typically pay a much larger sum for their shares than the founders or earlier investors, which means that the cash value of your stake is diluted because all the shares are worth the same amount, and you paid more than earlier investors for your shares. Dilution may also be caused by pricing securities at a value higher than book value or expenses incurred in the offering
The following table demonstrates the price that new investors are paying for their shares with the effective cash price paid by existing stockholders, giving effect to full conversion of all outstanding stock options and other convertible instruments (SAFE agreements, KISS agreements, and Crowd Notes), and assuming that the shares are sold at $2.00 per share and that certain holders of SAFE agreements exercise their warrants to purchase additional shares of preferred stock at the time the SAFE agreements are converted. The table presents shares and pricing as issued and reflects all transactions since inception, which gives investors a better picture of what they will pay for their investment compared to the company’s insiders than just including such transactions for the last 12 months, which is what the SEC requires. The share numbers and amounts in this table assume (1) conversion of (a) all of the company’s outstanding SAFE and KISS securities and Crowd Notes (in the aggregate amount of $2,189,049) and (b) the outstanding Crowd Notes issued as compensation to SI Securities, LLC, which replaced the warrants that were outstanding at June 30, 2017 and subsequently rescinded, effective prior to the crowdfunding offering, into shares of Preferred Stock immediately prior to the initial closing of this offering, (2) conversion of all outstanding options into shares of Common Stock at weighted average exercise price and (3) all accrued interest on the KISS securities is paid in cash at the time of conversion.
| 22 |
| Dates Issued | Issued Shares | Potential Shares | Total Issued and Potential Shares | Effective Cash Price per Share at Issuance or Potential Conversion | ||||||||||||||||
| Common Shares | 2015 | 8,500,000 | 8,500,000 | $ | 0.0001 | (3) | ||||||||||||||
| Outstanding Stock Options | 2015-2016 | 447,000 | (4) | 447,000 | 0.0500 | (2) | ||||||||||||||
| Crowd Notes issued to broker | 2017 | 65,687 | (1) | 65,687 | 0.6000 | (1) | ||||||||||||||
| SAFE Agreements | 2015-2016 | 1,791,667 | (1) | 1,791,667 | 0.6000 | (1) | ||||||||||||||
| KISS Agreements Payable | 2016 | 1,506,667 | (1) | 1,506,667 | 0.6000 | (1) | ||||||||||||||
| Crowd Notes Payable | 2017 | 348,717 | (1) | 348,717 | 0.6000 | (1) | ||||||||||||||
| Total Common Share Equivalents | 8,500,000 | 4,159,738 | (5) | 12,659,738 | 0.1778 | |||||||||||||||
| Investors in this offering, assuming $7 million raised | 3,500,000 | 3,500,000 | 2.0000 | |||||||||||||||||
| Total after inclusion of this offering | 12,000,000 | 4,159,738 | (5) | 16,159,738 | 0.5725 | |||||||||||||||
| (1) | KISS notes payable, Crowd Notes, and SAFE agreements have conversion terms stipulating conversion pricing of a 20% discount or the price per share implied by a $6 million pre-money valuation. The terms of this offering would trigger conversion of all such instruments at the price implied by a $6 million pre-money valuation, or $0.60/share. The number of potential shares is calculated using the outstanding principal of each of these instruments applied at the $0.60/share conversion price. |
| (2) | Stock option pricing is the weighted average exercise price of outstanding options, including unvested shares. |
| (3) | Common shares issued for $0.0001/share in 2015 inception year. |
| (4) | Assumes conversion at exercise price of all outstanding options. |
| (5) | The dilution disclosures contained in this section are based upon the instruments issued and outstanding as of June 30, 2017, the date of our latest financial statements. Subsequent to that date, the company has issued additional KISS securities ($375,000) and SAFE securities ($195,000) to investors, which will have an additional dilutive effect to incoming investors in this offering. The terms of these KISS and SAFE securities are substantially similar to the existing issued and outstanding KISS and SAFE securities (as described below in “Securities Being Offered”), except that the conversion feature is instead based upon the lower of a $10 million valuation cap or 10% discount. Immediately prior to the initial closing of this offering, these KISS and SAFE agreements would convert into 570,000 shares of Series A Preferred Stock at a conversion price of $1.00 per share, compared to the $2.00 per share offering price. |
The following table demonstrates the dilution that new investors will experience upon investment in the company. This table uses the company’s net tangible book value as of June 30, 2017 of $(1,233,430), which is derived from the net equity of the company in the June 30, 2017 financial statements. This tangible net book value is then adjusted to contemplate conversion of all convertible instruments outstanding at June 30, 2017 that would provide proceeds to the company, which assumes exercise of all options (447,000 shares), SAFE agreements, Crowd Notes, and KISS notes outstanding through June 30, 2017. Such conversions would provide $22,350 of proceeds and result in the issuance of 4,159,738 shares of common stock, which are considered in the figures used in the calculations presented in the table.
| 23 |
The following table presents three scenarios for the convenience of the reader: the company raises (a) the minimum offering amount of $450,000, (b) $3,725,000, representing the midpoint between the minimum and maximum offering amounts, and (c) the maximum offering amount of $7,000,000.
| On Basis of Full Conversion of Issued Instruments | Minimum Offering $450,000 | Midpoint $3,725,000 | Maximum Offering $7,000,000 | |||||||||
| Price per share | $ | 2.00 | $ | 2.00 | $ | 2.00 | ||||||
| Shares issued | 225,000 | 1,862,500 | 3,500,000 | |||||||||
| Capital raised | $ | 450,000 | $ | 3,725,000 | $ | 7,000,000 | ||||||
| Less: Estimated offering costs | $ | (103,750 | ) | $ | (349,375 | ) | $ | (595,000 | ) | |||
| Net offering proceeds | $ | 346,250 | $ | 3,375,625 | $ | 6,405,000 | ||||||
| Net tangible book value before offering | $ | (1,211,080 | )(2) | $ | (1,211,080 | )(2) | $ | (1,211,080 | )(2) | |||
| Net tangible book value after offering | $ | (864,830 | ) | $ | 2,164,545 | $ | 5,193,920 | |||||
| Shares issued and outstanding before offering, assuming full conversion | 12,659,738 | (1) | 12,659,738 | (1) | 12,659,738 | (1) | ||||||
| Shares issued and outstanding after offering | 12,884,738 | 14,522,238 | 16,159,738 | |||||||||
| Net tangible book value per share before offering | $ | (0.096 | ) | $ | (0.096 | ) | $ | (0.096 | ) | |||
| Increase/(Decrease) per share attributable to new investors | $ | 0.029 | $ | 0.245 | $ | 0.417 | ||||||
| Net tangible book value per share after offering | $ | (0.067 | ) | $ | 0.149 | $ | 0.321 | |||||
| Dilution per share to new investors ($) | $ | 2.067 | $ | 1.851 | $ | 1.679 | ||||||
| Dilution per share to new investors (%) | 103.36 | % | 92.55 | % | 83.93 | % | ||||||
(1) Assumes conversion of all outstanding stock options (providing proceeds of $22,350 to net tangible book value), SAFE agreements, KISS agreements, and Crowd Notes.
(2) Net tangible book value is adjusted for conversion proceeds for the outstanding stock options discussed at (1).
The next table is the same as the previous one, but adds in consideration of authorized but unissued stock options, presenting the fully diluted basis. This adds 1,053,000 pre-offering shares outstanding and is not adjusted for potential conversion proceeds on the hypothetical exercise of these options.
| 24 |
| On Basis of Full Conversion of Issued Instruments and Authorized but Unissued Stock Options | Minimum Offering $450,000 | Midpoint $3,725,000 | Maximum Offering $7,000,000 | |||||||||
| Price per share | $ | 2.00 | $ | 2.00 | $ | 2.00 | ||||||
| Shares issued | 225,000 | 1,862,500 | 3,500,000 | |||||||||
| Capital raised | $ | 450,000 | $ | 3,725,000 | $ | 7,000,000 | ||||||
| Less: Estimated offering costs | $ | (103,750 | ) | $ | (349,375 | ) | $ | (595,000 | ) | |||
| Net offering proceeds | $ | 346,250 | $ | 3,375,625 | $ | 6,405,000 | ||||||
| Net tangible book value before offering | $ | (1,211,080 | )(2) | $ | (1,211,080 | )(2) | $ | (1,211,080 | )(2) | |||
| Net tangible book value after offering | $ | (864,830 | ) | $ | 2,164,545 | $ | 5,193,920 | |||||
| Shares issued and outstanding before offering, assuming full conversion and authorized but unissued stock options | 13,712,738 | (1) | 13,712,738 | (1) | 13,712,738 | (1) | ||||||
| Shares issued and outstanding after offering | 13,937,738 | 15,575,238 | 17,212,738 | |||||||||
| Net tangible book value per share before offering | $ | (0.088 | ) | $ | (0.088 | ) | $ | (0.088 | ) | |||
| Increase/(Decrease) per share attributable to new investors | $ | 0.026 | $ | 0.227 | $ | 0.390 | ||||||
| Net tangible book value per share after offering | $ | (0.062 | ) | $ | 0.139 | $ | 0.302 | |||||
| Dilution per share to new investors ($) | $ | 2.062 | $ | 1.861 | $ | 1.698 | ||||||
| Dilution per share to new investors (%) | 103.10 | % | 93.05 | % | 84.91 | % | ||||||
| (1) | Assumes conversion of all outstanding stock options (providing proceeds of $22,350 to net tangible book value), SAFE agreements, KISS agreements, and Crowd Notes, and conversion of authorized but unissued stock options of 1,053,000 shares (no adjustment for proceeds contemplated in the calculations). |
| (2) | Net tangible book value is adjusted for conversion proceeds for the outstanding stock options discussed at (1). |
Future dilution
Another important way of looking at dilution is the dilution that happens due to future actions by the company. The investor’s stake in a company could be diluted due to the company issuing additional shares. In other words, when the company issues more shares, the percentage of the company that you own will go down, even though the value of the company may go up. You will own a smaller piece of a larger company. This increase in number of shares outstanding could result from a stock offering (such as an initial public offering, another crowdfunding round, a venture capital round or an angel investment), employees exercising stock options, or by conversion of certain instruments (such as convertible bonds, preferred shares or warrants) into stock.
| 25 |
If the company decides to issue more shares, an investor could experience value dilution, with each share being worth less than before, and control dilution, with the total percentage an investor owns being less than before. There may also be earnings dilution, with a reduction in the amount earned per share (though this typically occurs only if the company offers dividends, and most early stage companies are unlikely to offer dividends, preferring to invest any earnings into the company).
The type of dilution that hurts early-stage investors most occurs when the company sells more shares in a “down round,” meaning at a lower valuation than in earlier offerings. An example of how this might occur is as follows (numbers are for illustrative purposes only):
| · | In June 2017 Jane invests $20,000 for shares that represent 2% of a company valued at $1 million. |
| · | In December the company is doing very well and sells $5 million in shares to venture capitalists on a valuation (before the new investment) of $10 million. Jane now owns only 1.3% of the company but her stake is worth $200,000. |
| · | In June 2018 the company has run into serious problems and in order to stay afloat it raises $1 million at a valuation of only $2 million (the “down round”). Jane now owns only 0.89% of the company and her stake is worth only $26,660. |
This type of dilution might also happen upon conversion of convertible notes into shares. Typically, the terms of convertible notes issued by early-stage companies provide that in the event of another round of financing, the holders of the convertible notes get to convert their notes into equity at a “discount” to the price paid by the new investors, i.e., they get more shares than the new investors would for the same price. Additionally, convertible notes may have a “price cap” on the conversion price, which effectively acts as a share price ceiling. Either way, the holders of the convertible notes get more shares for their money than new investors. In the event that the financing is a “down round” the holders of the convertible notes will dilute existing equity holders, and even more than the new investors do, because they get more shares for their money. Investors should pay careful attention to the amount of convertible notes that the company has issued (and may issue in the future, and the terms of those notes.
If you are making an investment expecting to own a certain percentage of the company or expecting each share to hold a certain amount of value, it’s important to realize how the value of those shares can decrease by actions taken by the company. Dilution can make drastic changes to the value of each share, ownership percentage, voting control, and earnings per share.
| 26 |
The net proceeds of a fully subscribed offering after deducting offering expenses and commissions will be approximately $6.4 million. We plan to use the net proceeds to expand to 8 locations by year-end 2019, allocated as follows:
| · | Approximately $3.4 million for general and administrative expenses, which we estimate includes |
| o | $1.8 million for employee compensation. Of this amount, $800,000 would go towards executive officer compensation; |
| o | Delivery expenses of $600,000; |
| o | $350,000 for new location startup expenses (initial inventory and equipment); |
| o | Office and equipment leases would contribute $350,000; |
| o | The remaining $300,000 would go towards other operating expenses including legal, and office expenses; |
| · | Approximately $1.2 million for research and development of the company’s technology; and |
| · | Approximately $1.8million for sales and marketing. |
If the offering size is equal to or less than $5.25 million, representing 75% of the maximum offering amount, then we estimate that the net proceeds after deducting offering expenses and commissions would be approximately $4.78 million, which we would utilize to expand to 6 locations by year-end 2019, allocated as follows:
| · | Approximately $2.95 million for general and administrative expenses, which we estimate includes |
| o | $1.7 million for employee compensation. Of this amount, $800,000 would go towards executive officer compensation; |
| o | Delivery expenses of $400,000; |
| o | $250,000 for new location startup expenses (initial inventory and equipment); |
| o | Office and equipment leases would contribute $300,000; |
| o | The remaining $300,000 would go towards other operating expenses including legal and office expenses;, |
| · | Approximately $800,000 for research and development of the company’s technology; and |
| · | Approximately $1.8 million for sales and marketing expense through December 2019. |
If the offering size is equal to or less than $3.5 million, representing 50% of the maximum offering amount, then we estimate that the net proceeds after deducting offering expenses and commissions would be approximately $3.16 million, which we would utilize to expand to 3 locations by year-end 2019, allocated as follows:
| 27 |
| · | Approximately $2.16 million for general and administrative expenses, which we estimate includes |
| o | $1.4 million for employee compensation. Of this amount, $700,000 would go towards executive officer compensation; |
| o | Delivery expenses of $300,000; |
| o | $750,000 for new location startup expenses (initial inventory and equipment); |
| o | Office and equipment leases would contribute $200,000; |
| o | The remaining $185,000 would go towards other operating expenses including legal and office expenses; |
| · | Approximately $600,000 for research and development of the company’s technology; and |
| · | Approximately $400,000 million for sales and marketing expense through December 2019. |
If the offering size is equal to or less than $1.75 million, representing 25% of the maximum offering amount, then we estimate that the net proceeds after deducting offering expenses and commissions would be approximately $1.54 million, which we would utilize to expand to 2 locations by the end of June 2019, allocated as follows:
| · | Approximately $1.1 million for general and administrative expenses, which we estimate includes |
| o | $700,000 for employee compensation. Of this amount, $300,000 would go towards executive officer compensation; |
| o | Delivery expenses of $200,000; |
| o | $50,000 for new location startup expenses (initial inventory and equipment); |
| o | Office and equipment leases would contribute $50,000; |
| o | The remaining $100,000 would go towards other operating expenses including legal and office expenses; |
| · | Approximately $300,000 for research and development of the company’s technology; and |
| · | Approximately $150,000 million for sales and marketing expense through December 2019. |
If the offering size were to be equal to the minimum offering amount of $450,000, we estimate that the net proceeds would be approximately $340,000. In this case, we would significantly reduce our marketing and research and development efforts. In this scenario, emphasis would be placed on achieving cash-flow positive results at our existing location.
Because the offering is a “best efforts” offering, we may close the offering without sufficient funds for all the intended purposes set out above, or even to cover the costs of this offering.
| 28 |
The company reserves the right to change the above use of proceeds if management believes it is in the best interests of the company.
| 29 |
Overview
NowRx was founded with the goal of developing software, artificial intelligence, robotics and logistics to create the most convenient retail pharmacy service available, including free same-day delivery for prescription medications. For urgent medications, expedited one-hour delivery is available for an additional $5 charge. NowRx operates from within a low-cost, highly automated “virtual pharmacy” location that employs end-to-end robotic dispensing (“One-Click Fill”SM) and artificially intelligent chat bots to provide a more efficient and effective pharmacy experience for busy customers and their physicians. Medications are hand delivered by trained NowRx drivers and plug-in electric vehicles. By eliminating the dependency on retail space, NowRx is able to maintain fixed overhead costs that are substantially lower than traditional pharmacy providers with storefronts on busy streets. Further margin advantages are provided by robotics and automation. For added convenience, customers can manage their prescriptions through the NowRx mobile app, text, intelligent chat bots, phone, or through virtual voice-activated assistants such as Google Home.
 |
|
| |
| Robotic Dispensing Unit | NowRx App | Google Home |
NowRx responds to an obvious market need—solving the inconvenience of traditional brick and mortar pharmacies by providing all of the same pharmacy services, along with the additional benefit of safe, reliable delivery directly to the customer’s door free of charge. With NowRx, customers can conveniently manage their existing prescriptions, upload new prescriptions and even consult with a licensed pharmacist – all without ever having to leave their home or make a trip to a local pharmacy. No need to visit the pharmacy to obtain a prescription medication; no need for transportation to the pharmacy, or to stand in long lines, or to wait for the prescription to be filled. Recent studies have shown the median pharmacy wait time was 45 minutes5, and a second trip to the pharmacy was required 33% of the time.6 Customers can also use the app to easily set daily reminders to minimize the risk of missing a medication dose and to automatically request an upcoming refill order.
5 “Wait Time for Filling Rx”, Boehringer Ingelheim Pharmaceuticals, 2013.
6 “% Who Had to Return To or Got To a Different Pharmacy at Least Once in Past Year”, Boehringer Ingelheim Pharmaceuticals, 2013.
| 30 |
With free-same day delivery and one-hour expedited delivery, NowRx is part of a much broader global trend towards services delivered to directly to customers home rapidly and free, a trend recently described as the on-demand economy. The size and rapid growth of the on-demand economy clearly shows that consumers are attracted by the user experience, added convenience and other benefits it provides. Recent studies show on-demand services have already moved beyond early adopters and are gaining traction among mainstream consumers who require that platforms be user-friendly, safe and fast7.
NowRx also offers advantages over mail-order pharmacies, which are able to save customers trips to the pharmacy under certain circumstances, but can be confusing to use and are inherently unable to address many important pharmacy needs, including: first time prescriptions; urgent and refrigerated medications; or others not permitted to be sent through the mail such as narcotics and other DEA-controlled medications. We believe NowRx combines the best of current pharmacy offerings to create an exciting new pharmacy solution. Several of the NowRx services are designed to also make the prescription process more convenient for physicians who also can be frustrated by the bottenecks and inefficiencies in the pharmacy process, including our automated manufacturer coupon software that automatically identifies and applies available manufacturer coupons without the physicians having to manage physical coupons, and a streamlined insurance prior authorization process that reduces the amount of time physicians and their staff spend obtaining insurance approvals for drugs not on the health plan formularies.
7 “The On-Demand Economy is Growing, and Not Just for the Young and Wealthy”, Harvard Business Review, April 2016.
| 31 |
We plan to establish multiple locations in strategic patient-dense areas across the country. The company’s service area currently covers the Mountain View locality in the Bay Area, where we launched our first warehouse in January 2016. Over the next 18 months, we seek to expand within the Bay Area, to Redwood City and Campbell, as well as the Los Angeles/Orange County area, by establishing warehouses to support those localities. Our longer-term strategy is to expand to additional West Coast areas, including Seattle and San Diego, and ultimately to the rest of the country. Any expansion plans will be based on the competitive landscape, partnerships and other strategic considerations.
NowRx was incorporated in Delaware under the name YouRx, Inc. on February 19, 2015 and changed its name to NowRx, Inc. on May 19, 2015.
Industry Trends
On-Demand Economy
NowRx is responding to the rapidly increasing consumer demand for services delivered same-day and managed by convenience of mobile apps, chat bots and voice-activated assistants. The on-demand economy is attracting more than 22.4 million consumers annually and $57.6 billion in spending. Recent studies show on-demand services have already moved beyond early adopters and are gaining traction among mainstream consumers who require that platforms be user-friendly, safe and fast. We believe it’s just a matter of time before few people will be standing in traditional pharmacy lines.
The on-demand economy is also rapidly expanding across the global economy. According to Crowd Companies, a firm that tracks on- demand platform businesses, more than 280 companies provide on-demand goods and services across sixteen industries today. This is up from only 76 companies operating in just six industries two years ago. There are numerous recent examples of major corporations entering on- demand markets; such as GM’s partnership with Lyft, and hotel giant Accor’s recent acquisition of Airbnb competitor Onefinestay83.
8 “The On-Demand Economy is Growing, and Not Just for the Young and Wealthy”, Harvard Business Review, April 2016.
| 32 |
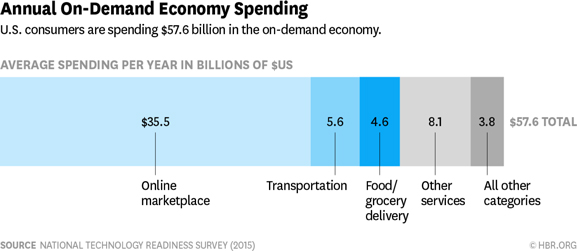
Traditional Pharmacy Industry
Traditional retail pharmacy is a $250 billion industry, growing by $20 billion annually9. The rate of sales growth in the United States has slowed in recent years, driven by a decline in new blockbuster drugs, a longer FDA approval process, drug safety concerns, higher copayments and an increase in the use of generic drugs, which are less expensive but generate higher gross margins. New drug development in the next few years is expected to be concentrated in specialty prescriptions, which are targeted toward a specific disease state. These drugs are often complex and expensive. We expect prescription usage to continue to grow in the coming years due to the aging U.S. population, increased life expectancy, "baby boomers" continuing to become eligible for the federally funded Medicare prescription program and new drug therapies.
Generic prescription drugs continue to help lower overall costs for customers and third-party payors. We believe the utilization of existing generic pharmaceuticals will continue to increase, although the pace of introduction of new generic drugs is expected to slow. In general, generic versions of drugs generate lower sales dollars per prescription, but higher gross profit dollars, as compared with patent-protected brand name drugs. The impact on retail pharmacy gross profit dollars can be significant in the first several months after a generic version of a drug is first allowed to compete with the branded version, which is generally referred to as a “generic conversion.” In any given year, the number of major brand name drugs that undergo a conversion from branded to generic status can vary and the timing of generic conversions can be difficult to predict, which can have a significant impact on retail pharmacy sales and gross profit dollars.
9“The 2016 Economic Report on Retail, Mail, and Specialty Pharmacies”, Pembroke Consulting, January 2016.
| 33 |
Products and Services
As an on-demand pharmacy, NowRx offers all of the same medication prescription services as a traditional retail pharmacy but does so remotely from a low cost, highly automated “virtual pharmacy” warehouse location, providing free same-day delivery of prescription drugs and some over-the-counter medications. For an additional $5.00, the company provides delivery within 1 hour of placing an order for in-stock pharmaceuticals; a choice that may be important if a drug is needed urgently.
We offer our services through the NowRx app, by text, by telephone and through voice-activated virtual assistants such as Google Home. Customers can download the NowRx app and complete their profile, including providing details of their health insurance plan and payment method, and delivery address information. Once a customer has completed their profile, they can instruct their doctors to send their prescriptions directly to NowRx when e-prescribing. The customer is then notified via the app that their prescription has been received and verified with their insurance company; the amount of the copay; and that the prescription will be delivered to the address of their choice the same day at no cost. With a single click the customer confirms the order. Alternatively, customers can scan a paper prescription securely through the NowRx app or transfer existing prescriptions from other pharmacies.
For a prescription covered by an insurance health plan (also known as a “third-party payor”), we collect from the customer the co-payment amount dictated by the third-party payor. If there is no third-party payor, we charge the customer a cash price that is set based on our own competitive pricing plan. We accept payments using a variety of methods, including cash, checks, credit and debit cards and health savings plans. Physicians are able to send prescriptions to NowRx through electronic prescribing, fax, the NowRx app or telephone.
Services provided include fulfilling new prescriptions or refills, transferring prescriptions from other pharmacies, and application of drug manufacturer coupons. The latter service is especially useful since many traditional pharmacies do not have the capability of automatically handling coupon driven prescription orders. NowRx also provides reminders through app and text to help customers remember to take medications and to process refills, to help promote better health and wellness. The NowRx app also has a secure video chat feature that allows customers to conveniently speak to a NowRx pharmacist from a private location of their own choosing.
The company uses employees and third-party delivery firms to deliver to a customer’s home or office. In instances in which a customer provides a scanned copy of their prescription, delivery personnel collect the original prescription prior to completing the delivery. Delivery personnel also collect cash payments when a customer does not make payment through the app or online. A customer signature and picture identification are required for approximately 80% of all deliveries. When customers are not present for a delivery that does not require a signature, drivers are required to take a picture of the medication by the doorstep, thereby assuring that the delivery is fully documented.
| 34 |
Suppliers
As a licensed pharmacy, NowRx purchases pharmaceutical products through wholesalers, then sells those products to patients, and then receives its revenue in the form of reimbursements from the patient’s insurance health plans and from the patients themselves in the form of co-pays or cash payments. We purchase approximately 70% of our pharmaceutical products from McKesson, with the remainder supplied by IPC. Orders are placed with McKesson at the end of each workday and those ordered items are delivered to NowRx the next business morning via truck delivery. IPC delivers products via UPS to NowRx. If our relationship with McKesson or IPC were disrupted, we could temporarily have difficulty filling prescriptions for branded and generic drugs until we execute a replacement wholesaler agreement or develop and implement self-distribution processes. There are several well-established wholesalers that are competitors to McKesson, including AmerisourceBergen Corp, Cardinal Health, Inc., and several smaller companies. We believe we could obtain and qualify alternative sources for substantially all of the prescription drugs we sell on an acceptable basis, and accordingly that the impact of any disruption would be temporary
Customers
Through June 30, 2017, we filled approximately 16,000 prescriptions and served more than 2,700 customers. A portion of our customers order one or more prescriptions each and every month, whereas some of our other customers order only one prescription every few months. During the month of June 2017, there were 764 customers who ordered a total of 1,833 prescriptions, for an average of 2.4 prescriptions per order and an average basket size of $285. The loss of any one customer would not have a material adverse impact on our results of operations.
The following table illustrates (a) on the left axis the growth in total number of customers since the company commenced operations and projects near-term growth assuming that the rate of growth continues and (b) on the right axis the total number of new customers added each quarter since the company commenced operations and projects near-term growth assuming that the company acquires new customers at the same rate.
| 35 |
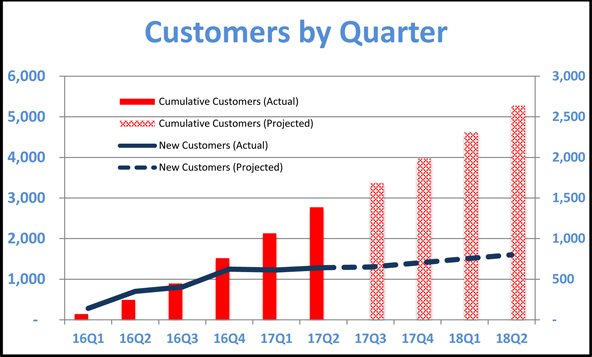
Third-party Payors
We enter into contracts with third-party payors (such as insurance companies, prescription benefit management companies, government agencies, private employers or other managed care providers) that agree to pay for all or a portion of a customer's eligible prescription purchases based on negotiated and contracted reimbursement rates for each prescription drug included in the third-party payor’s formulary. We collect from the customer any amount that remains due after reimbursement from third-party payors, if any. Reimbursement amounts from third-party payors are recorded as accounts receivable until payment is received, typically 20-45 days after the prescription is filled.
In 2016, 82 % of our net sales were to customers covered by third-party payors. During 2016, the top third-party payor (Access Health) accounted for approximately 48% of our net sales.
Technology
NowRx has developed and deployed a comprehensive suite of technologies, including the following:
Unified Communication Layer: Virtual Assistants, Mobile App and Chat Bots
NowRx has developed an extensive communication layer that unifies communication from customers from across a variety of modalities, including mobile app, chat bot that interacts with native mobile texting, and voice-activated voice assistants such as Alexa and Google Home. Recently, NowRx became the first HIPPA-compliant healthcare application approved on any voice assistant platform (Google Home) and is under review for approval on Amazon’s Alexa. Customers can interact with NowRx using any of these communication modalities to manage new prescriptions and refills, and receive helpful daily reminders.
| 36 |
“One Click FillSM”: Fully Integrated, End-to-end Robotic Dispensing
NowRx has developed a proprietary software method of interfacing with a commercially available robotic dispensing system allowing automated dispensing of refill requests with the press of one button on the mobile app by the customer (“One-Click FillSM”). Similarly, automated dispensing can be triggered by chat bot or voice-activated assistant command. Transfer requests from other pharmacies can also be made by the user via the app, chat bot or voice-activated assistant. Using Internet Protocol, these requests are automatically routed to the NowRx location where the robot performs automatic medication filling (sort, count, bottle, label and cap) without human intervention. A licensed pharmacist verifies every prescription before packaging for delivery, in accordance with regulations.
Delivery Logistics Layer and Opportunistic Refill Processing
NowRx has developed an extensive logistics platform and dashboard to optimize delivery routes based on traffic, geography and driver load. Furthermore, a large portion of medications are refilled and delivered to customers at regular intervals, usually once per month on or about the same day of the month (“ordinary refill queue”). However, refills may be opportunistically processed and delivered several days early, when permitted by the insurance health plan (typically 7 days in advance), to optimize delivery efficiency as measured by the number of refills delivered to the same neighborhood on the same day. As an illustration, if a new or refill prescription request is scheduled for delivery today to a certain neighborhood, the ordinary refill queue is scanned for any refills destined for the same neighborhood albeit several days in the future (“opportunistic refill”). These opportunistic refills are thus processed several days earlier than originally scheduled, thereby optimizing today’s deliveries to the target neighborhood, resulting in a lower cost delivery. As is always the case with deliveries, the customer is contacted first to confirm that an earlier delivery is acceptable.
| 37 |
Future Development
Patient Analytics: NowRx Medication Adherence IndexSM
NowRx is developing a method to improve the consistency with which patients adhere to their prescription plans (“medication adherence”). Patient frustration with the pharmacy process contributes to delays or postponement of medication regimes, non-adherence and in some cases prescription abandonment. Non-adherence is common. 110 million prescriptions are abandoned each year.10 For chronic disease patients, it is estimated that between 20% and 30% of prescriptions are never filled and up to 50% of medications are not taken as prescribed. Failure to follow prescriptions causes some 125,000 deaths per year and up to 10% of all hospitalizations. Medication non-adherence has been estimated to cause between $100 billion to $289 billion in waste to the U.S. healthcare system.11 One study found that 35% of patients who failed to fill their prescriptions cite the following reasons: 1) too busy to go to pharmacy, or too inconvenient, 2) forgot, or 3) lack of transportation.12 Patients that fail to adhere to their medication regimes, particularly those medications for chronic health conditions such as heart disease, diabetes and COPD, sometimes risk critical health episodes that lead to emergency healthcare services (hospitalizations, emergency rooms, or urgent care facilities). We believe it is possible to identify which patients are more likely to become non-adherent. NowRx intends to utilize data analytics and machine learning to calculate the NowRx Medication Adherence IndexSM, which will trigger additional communication and service protocols, such as additional text messages, phone calls from a pharmacist or coordination with a physician, to help improve medication adherence.
Mastering “The Last Mile of Healthcare”
Pharmacy represents a key entry point into the healthcare system for many customers. In fact, there are few aspects of healthcare that do not at some point intersect with pharmacy. Pharmacy data is some of the most valuable healthcare data available and we receive this data in its raw form, directly from physicians. In this way, we think about NowRx as a technology and Big Data company, and we plan to continue to develop advanced technologies across a wide array of pharmacy applications. All use of aggregate data is strictly subject to all applicable HIPAA and other privacy laws. In addition, pharmacists rank very high as authority figures in the healthcare planning for many customers. We believe there is an opportunity for technology-enabled, delivery-based pharmacies to become “health hubs” of the future, offering a platform to deploy additional healthcare services directly to customers, wherever they consume their healthcare services: at home, online or on their smartphone.
Wholesale Spot Purchasing Automation
Drug prices on the wholesale market have a high degree of variability. Each drug compound is available from multiple manufacturers, sold by a variety of wholesalers, each with their own individual price, which varies daily. Most pharmacies do not have sufficient staff resources to check all of the price variations each day in order to find the lowest price, and as a result frequently overpay for their inventory. NowRx is developing a software system to automatically analyze prices across wholesalers, manufacturers and drug formulation, and identify the lowest price each day, thereby optimizing wholesale purchases and increasing gross margins.
10 Understanding Prescription Abandonment”, CVS, 2008.
11 Interventions to Improve Adherence to Self-administered Medications for Chronic Diseases in the United States: A Systematic Review”, Annals of Internal Medicine.
12 “The Consent and Prescription Compliance (COPRECO) Study”, Academy of Emerging Medication, 2008.
| 38 |
Marketing
NowRx has pursued a very capital efficient marketing strategy, having expended very few resources on direct-to-consumer marketing, typically a very expensive proposition, and instead focused on a physician referral channel along with customer word of mouth. We market to physicians utilizing marketing representatives who visit directly with physicians and their staff to promote the benefits of NowRx not only to their patients but also to the physician’s office itself. This strategy has been very successful as evidenced by the graph below showing the steady growth of physician referrals as of June 2017 NowRx receives new customer referrals business from 945 physicians. Physicians obtain several benefits from the NowRx service, including reduced office resources spent managing patient prescriptions, streamlined insurance plan approvals, and more consistent application of drug coupons, all of which reduce the number of inbound calls to physicians, reduce time spent by physicians’ staff speaking with customers, drive higher patient satisfaction and potentially better patient health outcomes. NowRx also markets directly to healthcare facilities such as assisted living facilities that generate high volume prescription orders from multiple patients, an especially resource efficient marketing channel. We have continued to strengthen our presence on social media sites such as Facebook. We intend to use a portion of the net proceeds from this offering to expand our sales and marketing efforts, primarily by hiring additional marketing representatives, with a modest increase in social marketing for brand awareness purposes. As the company scales up and achieves broad-based consumer recognition in any given region, we will increasingly focus on consumer driven marketing to strengthen the pace of growth.
The follow table illustrates the growth on a quarterly basis in revenues generated from referrals from physicians already in the company’s network and from physicians added in that quarter.
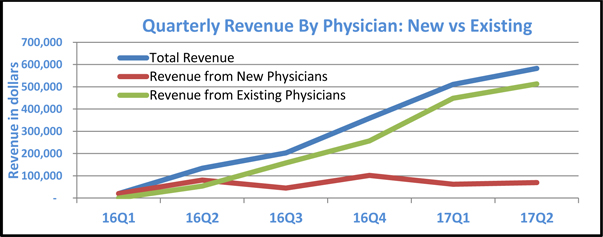
| 39 |
Market Opportunity
We believe it’s only a matter of time before the entire pharmacy industry will fulfill customer needs through same-day delivery and customers standing in line at a pharmacy counter will be a thing of the past. We believe recent trends in the on-demand economy indicate that the historic pharmacy model of requiring “in-store pickup” is becoming outdated, as consumers are increasingly demanding more services delivered the same-day or even same-hour. The large players in the industry are currently committed financially to a business model that is dependent on customers coming into stores to pick up their prescriptions and then buying additional items such as over-the-counter drugs and sundries. In 2015, there were approximately 64,000 traditional pharmacies that dispensed 5.2 billion 30-day prescriptions.13 The traditional pharmacy model with expansive retail space offering other products beyond prescription medications (such as over-the-counter medications and sundries) creates a financial incentive for large pharmacy chains to maintain their in-store pickup model for the upsell. We believe adopting a free same-day delivery would necessarily reduce the upsell opportunities and undermine the financial viability of the thousands of brick and mortar locations that are the backbone of the industry. Furthermore, we believe the large pharmacy chains currently lack the logistics and automation technology necessary to make the NowRx on-demand model work successfully. Notwithstanding, we believe that eventually the large chain store pharmacies will eventually be compelled to provide same-day delivery but are likely to do so through acquisitions or strategic partnerships with existing players such as NowRx. This can be a much easier path of entry since it facilitates quicker participation by the large chain store companies in the space without ever enduring the risks faced by startups to drive this industry. The acquirer gets immediate access to all of the needed technology, the backbone infrastructure and the delivery infrastructure. This could serve NowRx well since it affords the company the opportunity for a liquidity event other than an initial public offering. We cannot assure you, however, that any such acquisition will take place.
Competition
The retail pharmacy industry is highly competitive. NowRx competes with, among others, other startups in the on-demand prescription drug delivery space, retail drugstore chains, independently owned drugstores, wellness offerings and mail order pharmacies. We compete on the basis of convenience and customer service that is made possible through our technology. We believe continued consolidation of the drugstore industry and the aggressive discounting of generic drugs by mass merchandisers will further increase competitive pressures in the industry.
Startups
We are aware of several startups in the on-demand prescription drug delivery space, including Alto Pharmacy, NimbleRx and Capsule. Two of these startups, Alto Pharmacy and NimbleRx, are in the San Francisco Bay Area, with whom we compete directly on a day-to-day basis. Capsule currently serves the New York City area. Several of these competitors have raised significant venture capital rounds of funding. We believe we are the most technologically advanced pharmacy solution on the market, and offer the most customer-centric and reliable on-demand service available, and believe our revenue and customer growth supports this view.
13 “The 2016 Economic Report on Retail, Mail, and Specialty Pharmacies”, Pembroke Consulting, January 2016.
| 40 |
Large Chain Pharmacies
The strategy of national pharmacy chains is fundamentally based on in-store upsell opportunities (over-the-counter and sundries). Any move by the pharmacy chains into free delivery would necessarily decrease customer foot traffic, undermine their upsell opportunities, and jeopardize the financial results of their expansive retail infrastructure. Free delivery of prescription drugs will inevitably disrupt their existing business model. We believe that ultimately the large chain pharmacies will be compelled by virtue of consumer demand to enter the on-demand delivery space and likely will do so by acquiring existing players such as NowRx. As such, NowRx is building its business with the possibility of such a sale in mind, positioning itself to assure that its back-office infrastructure, its technology, and its delivery platform, are robust enough to support a business with thousands of locations. While there can be no assurance that a large pharmacy chain will seek to acquire the company, we believe this approach will make the company highly attractive to a large strategic buyer.
Mail Order Pharmacies
Mail-order pharmacy is a $75 billion market the number of prescriptions being filled by mail-order pharmacies has been in decline the last several years14. It is a convenient, but not an immediate service. It does not address “same-day fill” prescriptions, which is the $260 billion market that NowRx is addressing. Furthermore, acute, refrigerated (antibiotics, insulin, etc.) and controlled medications (schedule II narcotics, psychotropics, etc.), and first time prescriptions that require pharmacist consultation, are better suited for local, retail or on-demand pharmacy dispensing.
Amazon and Other New Entrants
There have been rumors and reports that Amazon may be considering entering the on-demand pharmacy space. Amazon traditionally competes on the basis of offering the lowest price to the consumer and the widest possible selection, characteristics that are not present in the pharmacy industry. In addition, the pharmacy industry is heavily regulated at the state and federal level. We believe NowRx has ample runway to build our business before and if Amazon enters the space. In fact, recent reports have shown that Amazon’s licensing to date has been in the wholesaler pharmacy space, not the retail space.15 Other new, unknown entrants may also enter this space.
14 The 2016 Economic Report on Retail, Mail, and Specialty Pharmacies”, Pembroke Consulting, January 2016.
15 Amazon gains wholesale pharmacy licenses in multiple states”, St. Louis Dispatch, October 2017.
| 41 |
Employees
Currently, we have 12 full-time employees and 4 part-time employees working out of our headquarters in Mountain View, California, including 3 licensed pharmacists.
Intellectual Property
The company does not currently hold any patents or trademarks. The company has filed a provisional patent covering several areas of our technology discussed above under “ — Technology.” This patent filing was registered with the US Patent Office on January 17, 2018 with serial no. 62618154.
Research and Development
In 2015 and 2016, we spent and $40,790 and $1,392, respectively, for research and development.
Regulation
Our business is subject to federal, state and local laws, regulations, and administrative practices concerning the provision of and payment for health care services, including, without limitation: federal, state and local licensure and registration requirements concerning the operation of pharmacies and the practice of pharmacy; Medicare, Medicaid and other publicly financed health benefit plan regulations prohibiting kickbacks, beneficiary inducement and the submission of false claims; the ACA; regulations of the FDA and the DEA, including regulations governing the purchase, sale, storing and dispensing of controlled substances and other products, as well as regulations promulgated by state and other federal agencies concerning automated outbound contacts such as phone calls, text messages and emails.
NowRx is licensed by the DEA and the state of California. Our pharmacy is regulated by the state of California. As we expand our operations within California and in other states, each warehouse will constitute a pharmacy, which will require a separate state license, and the company will become subject to additional states’ licensure and registration requirements.
Our business is also subject to patient privacy and other obligations, including responsibility imposed by HIPAA. As a covered entity, we are required to implement privacy standards, train our employees on the permitted uses and disclosures of protected health information, provide a notice of privacy practice to our customers and permit customers to access and amend their records and receive an accounting of disclosures of protected health information. We are also subject to federal and state privacy and data security laws with respect to our receipt, use and disclosure by us of personally identifiable information, which laws require us to provide appropriate privacy and security safeguards for such information.
| 42 |
In addition, in connection with the operation of our distribution center, we are subject to laws and regulations relating to the protection of the environment and health and safety matters, including those governing the management and disposal of hazardous substances and the cleanup of contaminated sites.
The Medicare Part D program has undergone significant legislative and regulatory changes since its inception, and continues to attract a high degree of legislative and regulatory scrutiny. The applicable government rules and regulations are expected to continue to evolve in the future.
NowRx currently leases its premises and owns one Parata Max robotic dispensing system. The company’s facility in Mountain View, California serves as its headquarters.
MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
The following discussion of our financial condition and results of operations should be read in conjunction with our financial statements and the related notes included in this Offering Circular. The following discussion contains forward-looking statements that reflect our plans, estimates, and beliefs. Our actual results could differ materially from those discussed in the forward-looking statements.
Overview
The company is an on-demand pharmacy, leveraging the latest in software technology, artificial intelligence, robotics and logistics to provide free same-day delivery of prescription medications, thereby avoiding the need to ever visit the pharmacy. NowRx was founded in February 2015 and commenced operations and revenue generation in January 2016. In 2015 the company was focused on obtaining its pharmacy licenses, developing the technology for the pharmacy platform and the mobile app, and establishing its first pharmacy location.
The company’s net sales consist of payments for prescription and some over-the-counter (“OTC”) items. For a prescription medication covered by a third-party payor, such as an insurance company, PBM or manufacturer coupon plan, the company receives a portion of its revenues from the patient, in the form of a co-payment paid or charged at the time the prescription is filled, and the remainder as a reimbursement from the third-party payor, at contracted prices. For prescription medication not covered by a third-party payor, the payment is collected entirely from the patient. The company records the amounts subject to reimbursement in accounts receivable until payment is received, typically 20-45 days after the prescription is filled. Cost of goods sold consists primarily of prescription and OTC medications that are acquired from wholesale suppliers.
| 43 |
Our net sales, gross profit margin and gross profit are impacted by, among other things, the percentage of prescriptions that we fill that are generic versus brand name, the rate at which new generic and brand name drugs are introduced to the market, the mix of business between prescription medications and OTC items, and variations in wholesale pricing. Because any number of factors outside of our control can affect timing for a generic conversion, we face substantial uncertainty in predicting when such conversions will occur and what effect they will have on particular future periods. Further consolidation among generic manufacturers coupled with changes in the number of major brand name drugs anticipated to undergo a conversion from branded to generic status may also result in gross margin pressures within the industry. We continuously face reimbursement pressure from PBM companies and other commercial third-party payors. In addition, plan changes with rate adjustments often occur in January and our reimbursement arrangements may provide for rate adjustments at prescribed intervals during their term. We experienced lower reimbursement rates as a percentage of revenue in fiscal 2017 as compared to the same period in the prior year. Wholesale pricing plans provide volume discounts that present an opportunity to increase gross margins as we grow our business. Increasing the percentage of revenue contributed by OTC items can also expand margins, as OTC items typically have higher margins than prescription medications. We expect downward pressure on reimbursements to be offset by improvements in wholesale volume discounting and increased OTC sales. However there is significant uncertainty in predicting the result of these offsetting factors on margins.
Results of operations
Year ended December 31, 2016 Compared to Year ended December 31, 2015
The company’s net sales for the year ended December 31, 2016 were $668,695. The company hired its first employees during the second half of 2015 and did not generate any revenues that year. Cost of goods sold was $573,791 in 2016, resulting in gross profit of $94,904, and a gross margin of 16.5%.
The company’s operating expenses consist of general and administrative, sales and marketing, depreciation, and research and development expenses. Operating expenses in 2016, the company’s first full year of operations, were $717,202, compared to $247,240 in 2015, an increase of 190% that resulted from the ramping up of the company’s operations in its first full year in business. General and administrative expenses represented the largest component of this increase, from $196,716 in 2015 to $655,755 in 2016, as the company company’s payroll increased from $116,709 to $413,325, legal and professional services increased from $15,482 to $36,064, travel expenses increased from $9,307 to $43,361, rent increased from $25,699 to $53,196 (the lease started August 2015) and the company began to incur delivery costs, which totaled $85,825. Sales and marketing expenses grew 563% from $8,293 in 2015 to $55,002 in 2016 as the company commenced marketing efforts with the launch of its services in January 2016. The company intends to use a portion of the net proceeds of this offering on marketing, including advertising and hiring sales representatives, to drive further sales, which will result in a significant increase in these costs. Research and development expenses decreased 97% from $40,790 in 2015 to $1,392 in 2016, reflecting the completion of the company’s initial phase of software development. The company also intends to use a portion of the net proceeds of this offering on the next phase of software development.
| 44 |
Other expenses consist of interest expense, which amounted to $237 in 2016, as the company entered into short-term promissory notes in December 2016. See “—Liquidity and Capital Resources – Indebtedness.”
As a result of the foregoing factors, the company’s net loss was $622,535 in 2016, a 152% increase from a net loss of $247,240 in 2015.
Six Months ended June 30, 2017 Compared to Six Months ended June 30, 2016
The company’s net sales for the six months ended June 30, 2017 were $994,588, compared to $149,882 for the six months ended June 30, 2016, an increase of 564%. The increase is attributable to an increase in number of prescriptions filled, from 2,253 during the first six months of 2016 to 8,525 during the first six months of 2017. Cost of goods sold was $823,073 through June 30, 2017, compared to $116,148 through June 30,2016, an increase of 609%, an increase reflecting similar growth in the number of prescriptions filled. Gross profit and gross margin for the first six months of 2017 were $171,515 and 17.2%, respectively, compared to gross profit and gross margin of $33,734 and 22.5%, respectively, during the same period in 2016. The higher gross margin during the first six months of 2016 reflects a higher mix of generic drugs as compared to the same period in 2017.
Operating expenses for the first six months of 2017 were $512,854, versus $345,061 for the first six months of 2016, an increase of 49%. General and administrative expenses increased from $312,296 in the 2016 period to $461,278 in the 2017 period, an increase of 47%. This increase was attributable to the hiring of 6 additional employees, including 2 additional drivers, the investment of $86,100 in the Parata robotic dispensing system, and expenses of $68,367 incurred in connection with the offering Crowd Notes described in “—Liquidity and Capital Resources.” Sales and marketing expenses for the first six months of 2017 were $47,468, compared to $28,846 in the same period in 2016, an increase of 68%, as the company increased its sales efforts.
Interest expense in the first six months of 2017 was $23,166, consisting of interest paid on short-term loans entered into in December 2016 and recognized on the Crowd Notes issued in the crowdfunding offering. As a result of the foregoing factors, the company’s net loss in the 2017 six-month period was $364,505, compared to $311,327 in the 2016 six-month period.
| 45 |
Liquidity and Capital Resources
As of June 30, 2017, the company’s cash on hand was $391,723. To date, the company has not made any profits and is still a “development stage company.” The company has recorded losses from the time of its inception in the total amount of $1,234,280.
In accordance with ASU No. 2014-15 Presentation of Financial Statements – Going Concern (subtopic 205-40), our management evaluates whether there are conditions or events, considered in the aggregate, that raise substantial doubt about our ability to continue as a going concern within one year after the date that the unaudited interim financial statements are issued. We have incurred substantial losses since our inception and we expect to continue to incur operating losses in the near-term. We expect that we will need to raise additional capital to meet anticipated cash requirements for the 24-month period following the filing date of this Offering Circular. In addition, we regularly consider fundraising opportunities and will determine the timing, nature and amount of financings based upon various factors, including market conditions and our operating plans. As we have done historically, we may again in the future elect to finance operations by selling equity or debt securities or borrowing money. If we raise funds by issuing equity securities, dilution to stockholders may result. Any equity securities issued may also provide for rights, preferences or privileges senior to those of holders of our common and preferred stock. If additional funding is required, we cannot assure you that additional funds will be available to us on acceptable terms on a timely basis, if at all, or that we will generate sufficient cash from operations to adequately fund our operating needs. If we are unable to raise additional capital or generate sufficient cash from operations to adequately fund our operations, we will need to curtail planned activities to reduce costs. Doing so will likely have an unfavorable effect on our ability to execute on our business plan, and have an adverse effect on our business, results of operations and future prospects.
Issuances of SAFE and KISS securities and Crowd Notes
Between July 2015 and November 2016, the company entered into simple agreements for future equity (“SAFE securities”) with investors, including Cary Breese and Sumeet Sheokand, for total proceeds of $1,067,500. Since June 30, 2017, the company has issued additional SAFE securities for total proceeds of $195,000. The SAFE securities do not bear interest and have no maturity date. As described below under “Securities Being Offered,” the SAFE securities contain a conversion provision that will be triggered by this offering.
Between December 19, 2016 and March 11, 2017, the Company entered into KISS agreements (Keep it Simple Security) with investors for total proceeds of $904,000. Since June 30, 2017, the company has issued additional KISS securities for total proceeds of $375,000. The instruments mature 24 months after issuance and bears 5% interest per annum. As described below under “Securities Being Offered,” the KISS agreements contain a conversion provision that will be triggered by this offering.
| 46 |
On March 11, 2017, the company issued $209,230 in Crowd Notes pursuant to Regulation Crowdfunding and Regulation D under the Securities Act (the “crowdfunding offering”). The Crowd Notes have no maturity date and bear 5% interest per annum. In connection with the crowdfunding offering, which was conducted on a portal affiliated with SI Securities, LLC, the company issued to SI Securities, LLC an additional $39,411.50 in Crowd Notes. This $39,411.50 in Crowd Notes represents a combination of the fees due to SI Securities, LLC as compensation for the crowdfunding offering and a previous offering conducted under Regulation D, for which SI Securities, LLC was issued warrants that were voided prior to the crowdfunding offering, with the understanding that the fees would be combined and paid in total as Crowd Notes. As described below under “Securities Being Offered,” the Crowd Notes contain a conversion provision that will be triggered by this offering.
Indebtedness
On December 21, 2016, the company entered into an inventory financing arrangement of $62,100 with Kabbage, an inventory financier. The loan was secured by all assets of the company, bore an interest rate of 24.04% per annum, and was payable over six months for a total repayment of $66,447. The loan was paid in full in June 2017.
On December 21, 2016, the company also entered into promissory notes totaling $70,000 with five individuals. The notes bore 12% interest and matured on April 30, 2017. The notes were collateralized by receivables, cash accounts, inventories, and all related proceeds. The notes, together with accrued interest, were paid at maturity.
The company currently has no material commitments for capital expenditures. The company maintains inventory used in the normal course of business, and had $156,892 of inventory on hand as of June 30, 2017.
Trend Information
Margin trends
The company has several initiatives underway to increase gross margins and improve operating margins, as follows:
| · | Pharmacy Management System – The company currently utilizes an “off-the-shelf” industry software system to manage pharmacy operations. Management has noted several areas of inefficiencies in the pharmacy management system that cause higher than necessary labor costs for filling prescriptions. With the net proceeds from this offering, the company plans to develop a proprietary pharmacy management system, which it expects to significantly reduce labor costs per prescription order. |
| · | Optimized Robotic Dispensing – The company has six months of experience with a robotic dispensing system, the usage of which is currently well below capacity. As order volume grows, we believe there is an opportunity to substantially increase the portion of medications filled robotically to reduce labor costs per order. |
| 47 |
| · | Pharmacy Refill Process Optimization – The company is developing proprietary algorithms to opportunistically process refills in advance of customary refill date, based on geographic location, in order to optimize delivery routing and reduce delivery costs per order. See “The Company’s Business – Technology — Delivery Logistics Layer and Opportunistic Refill Processing.” |
| · | Reduced Delivery Expense per Order – As the company increases revenue and customer volume per geography, the customer density increases and the run time per delivery is reduced, increasing the number of orders delivered per driver-hour and reducing the delivery cost per order. |
| · | Wholesale Volume Discounts – Wholesale pharmaceutical suppliers offer discounts for increasing volume of purchases and longer term contracts. As we grow our business, management anticipates being able to purchase products at a reduced cost. |
| · | Increased OTC sales – Many OTC products on average have a larger gross margin than the average prescription medication. Management intends to increase OTC as a percentage of sales over time, increasing average revenue per order (“basket size”) as well as increasing the overall gross margin for the company. |
| · | Medication Synchronization – Many customers have multiple prescription medications that refill monthly, but often at times such that the refill dates fall on different days of the month causing multiple trips to the same customer’s house per month. By synchronizing medications, with the customer’s approval, to refill on the same day of the month, delivery efficiency can be enhanced, reducing delivery costs per order. |
Order trends
The company seeks to continually grow the number of orders and its revenues by focusing on two initiatives:
| · | Increase sales and marketing through a combination of direct consumer advertising and sales representatives, which has generated steady growth from inception in the number of prescriptions, the number of referring physicians, and in revenues. A portion of the net proceeds of this offering will be allocated to increased sales and marketing efforts. |
| · | The company intends to add new pharmacy locations. The company anticipates that raising the maximum amount sought in this offering will permit it to establish as many as 8 new locations, which we plan to open by December 2019. We anticipate that each of these new locations will experience growth rates and other business performance metrics equal to or better than those achieved at the company’s existing location. Even if we are able to meet our projected timeline for establishing the new locations, these operations may not generate the anticipated growth in customers, orders and revenues at the pace that we project. The company recorded preliminary unaudited revenues of approximately $331,000 in the month of December 2017. Management believes that, given the growth pattern of revenues during 2016 and 2017 and, assuming we hire additional marketing resources with a portion of the net proceeds from this offering, it is likely that revenues will continue to increase. However, if NowRx were to experience zero growth and zero decline in revenues, the revenues in December 2017 translate to a run rate, based on 12 months, of approximately $4 million, the result of multiplying $331,000 by 12. Corresponding losses for the month of December 2017 and 12 month run rate, are $96,000 and $1.2 million, respectively. Note that these revenue and loss numbers are based on internal company financial statements that have not yet been audited or reviewed. When such audit or review takes place, we may need to make adjustments, which may be significant. We are not required to include audited financial information for 2017 in the Offering Circular until 90 days after the end of our fiscal year. Even if we are able to meet our projected timeline for establishing the new locations, these operations may not generate the anticipated growth in customers, orders, revenue and profit(loss) at the pace that we project. |
| 48 |
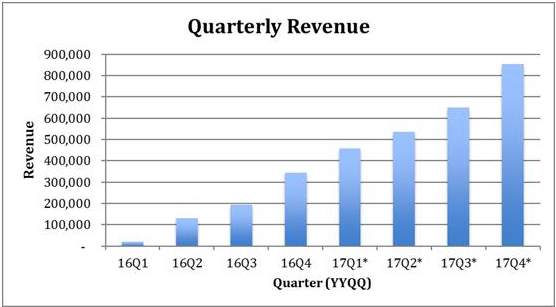
* Financial data for 2017 is unaudited
DIRECTORS, EXECUTIVE OFFICERS AND SIGNIFICANT EMPLOYEES
The company’s executive officers and directors are listed below. The executive officers are full-time employees.
| Name | Position | Age | Date Appointed to Current Position | |||
| Executive Officers | ||||||
| Cary Breese | Chief Executive Officer, Chief Financial Officer | 51 | June 24, 2015 | |||
| Sumeet Sheokand | Chief Technology Officer | 43 | June 24, 2015 | |||
| Directors | ||||||
| Cary Breese | Director | 51 | June 24, 2015 | |||
| Sumeet Sheokand | Director | 43 | June 24, 2015 | |||
| Barry Karlin | Executive Chairman | 63 | January 2, 2018 | |||
| Significant Employee | ||||||
| Melissa Bostock | Pharmacist-in-Charge | 37 | October 15, 2015 |
| 49 |
Cary Breese, CEO and Co-founder
Cary Breese has served as the company’s CEO and CFO since the formation of the company in 2015. Prior to founding NowRx, Cary was CEO of GenieDB, a venture-funded tech startup that provided [distributed MySQL database-as-a-service to enterprises], from 2012 until 2014. At GenieDB, Cary maintained overall responsibility for all operations, financing, marketing and sales. From 2011 to 2015, Cary was also a founding member and CFO of Frost Data Capital, a Big Data new company incubator and venture fund focused on new innovations in data management, data analytics and internet of things. Cary also was CEO of a successful financial tech company, Trafalgar Insurance Services, which he acquired in March 2003 and then led through financial turnaround and ultimate sale to a large regional financial services firm. Cary holds a Bachelor of Science degree in Electrical Engineering from Drexel University, and holds an ACAS designation from the Casualty Actuarial Society, a professional society whose members have demonstrated expertise in finance, economics, insurance and risk management.
Sumeet Sheokand, CTO and Co-founder
Sumeet Sheokand has served as the company’s CTO, a position in which Sumeet since the formation of the company in 2015. During 2014, Sumeet was involved with a startup incubator as an Entrepreneur-in-Residence. He also consulted with a top-3 US telecom provider to build their social media customer listener platform. Sumeet was CTO of GenieDB from 2012 until 2014, where he was responsible for all technology and software. Prior to joining GenieDB, Sumeet worked in technology management in a number of enterprises. Sumeet holds an undergraduate degree in Electrical and Computer Engineering from National Institute of Technology Kurukshetra, India, and a Masters of Business and Administration from UCLA Anderson School of Management.
Melissa Bostock, Pharmacist-in-Charge
Melissa Bostock has served as NowRx’s Pharmacist-in-Charge since the company began operations in November 2015. Prior to joining NowRx, Melissa was Pharmacy Executive Team Manager at Target, from July 2012 to November 2015. Melissa holds a BS degree in Microbiology and a Master of Public Health degree in Community Health, from UCLA, and holds a Doctor of Pharmacy from University of California, San Francisco.
| 50 |
Barry Karlin, Executive Chairman
Barry Karlin joined the company as Executive Chairman of the Board in January 2018. Since May 2014, he has been a private investor in real estate, high technology, and healthcare companies, and regularly consults to private equity firms as well as to public and privately held wealth management companies, primarily in the healthcare services sector. Barry was the founder, Chairman and CEO of Prospira PainCare, a physician-based rollup of pain management practices, from August 2012 to May 2014. He was also the founder, Chairman and CEO of CRC Health Group from 1995 to 2010, a specialized behavioral healthcare treatment provider in the United States, Canada and the United Kingdom. In 2000, Barry founded eGetgoing, which offered treatment through live, audio/video based online technology. He served as Chairman and CEO before eGetgoing was acquired by CRC in 2002. He previously cofounded and served as Chairman and CEO of Navigation Technologies, formerly served as a general partner at a venture firm specializing in the wireless communications industry, and served as a strategy management consultant; first with Strategic Decisions Group (1981 – 1984) and subsequently with Decision & Risk Analysis, Inc. (1991 – 1995). Barry has received numerous awards including Ernst & Young 2008 Northern California Regional Entrepreneur of the Year in Life Sciences, Silicon Valley Business Journal 2002 award for Fastest Growing Private Company, Certificate of Special Congressional Recognition in 2005 and the California Association of Drug Abuse Counselors award for exceptional contribution to the addiction community. He received his Ph.D. and M.S. from Stanford University in the Department of Engineering-Economic-Systems (specialty in Decision Sciences) and a B.S. in Electrical Engineering from the University of Witwatersrand in South Africa.
COMPENSATION OF DIRECTORS AND EXECUTIVE OFFICERS
For the fiscal year ended December 31, 2016, we compensated our three highest-paid directors and executive officers as follows:
| Name | Capacities in which compensation was received | Cash compensation | Other compensation | Total compensation | ||||||||
| Cary Breese | CEO | $ | 63,403 | 0 | $ | 63,403 | ||||||
| Sumeet Shoekand | CTO | $ | 56,461 | 0 | $ | 56,461 | ||||||
| Melissa Bostock | Pharmacist | $ | 131,456 | 2% equity, ISO options | $ | 131,456 | ||||||
Other than cash compensation, health benefits and stock options, no other compensation was provided to the executive officers, including in their capacity as directors of the company.
SECURITY OWNERSHIP OF MANAGEMENT AND CERTAIN SECURITYHOLDERS
The following table sets out, as of January 2, 2018, the voting securities of the company that are owned by executive officers and directors, and other persons holding more than 10% of any class of the company’s voting securities, or having the right to acquire those securities. The table assumes that all options and warrants have vested. The company’s voting securities include all shares of Common Stock and, upon the amendment of the amended certificate of incorporation immediately prior to the closing of this offering, all shares of Preferred Stock.
| 51 |
| Name and address of beneficial owner | Title of class | Amount and nature of beneficial ownership | Amount and nature of beneficial ownership acquirable | Percent of class | ||||||||||
| Cary Breese 6 Trapani Laguna Niguel, CA 92677 | Common Stock | 3,617,600 | 0 | 42.56 | % | |||||||||
| Sumeet Sheokand 995 W. Homestead Road Sunnyvale, CA 94087 | Common Stock | 3,182,400 | 0 | 37.44 | % | |||||||||
| Barry Karlin 120 Atherton Ave Atherton, CA 94027 | Common Stock | 1,700,000 | 0 | 20.00 | % | |||||||||
| All current officers and directors as a group (3 people) | Common Stock | 8,500,000 | 0 | 100.00 | % | |||||||||
| All current officers and directors as a group (3 people) | Series A Preferred Stock | 441,666 | (1) | 10.31 | % | |||||||||
(1) Represents shares of Series A Preferred Stock issuable upon conversion of KISS and SAFE securities immediately prior to the initial closing of the offering.
INTEREST OF MANAGEMENT AND OTHERS IN CERTAIN TRANSACTIONS
The company’s co-founders and an employee extended financing of $50,000 and $15,000, respectively, in 2015 to the company through SAFE securities. In 2016, that employee provided an additional $35,000 of SAFE financing.
General
The company is offering shares of Series A Preferred Stock in this offering. The Series A Preferred Stock may be converted into shares of the Common Stock of the company at the discretion of each investor, or automatically upon the occurrence of certain events, like an initial public offering. The company is therefore qualifying up to 3,500,000 shares of Series A Preferred Stock, convertible into an additional 3,500,000 shares of Common Stock, under the Offering Statement of which this Offering Circular is a part.
The following description summarizes the most important terms of the company’s capital stock. This summary does not purport to be complete and is qualified in its entirety by the provisions of NowRx’s amended certificate of incorporation and bylaws, copies of which have been filed as exhibits to the Offering Statement of which this Offering Circular is a part. For a complete description of NowRx’s capital stock, you should refer to the amended and restated certificate of incorporation and bylaws of the company and to the applicable provisions of Delaware law.
| 52 |
The authorized capital stock of the company consists solely of Common Stock, par value $0.00001 per share. Immediately prior to the initial closing of this offering, the company will amend its amended certificate of incorporation to authorize a second class of capital stock, Preferred Stock. The form of amended and restated certificate of incorporation setting forth the terms of the Series A Preferred Stock is filed as Exhibit 2.2 to the Offering Statement of which this Offering Circular is a part. Upon the filing of the amendment, the total number of authorized shares of Common Stock of NowRx will be 18,500,000 and the total number of authorized shares of Preferred Stock will be 8,500,000, all of which will be designated as Series A Preferred Stock. The total number of shares of Common Stock subject to awards under the 2015 Stock Incentive Plan is 1,500,000.
As of January 2, 2018, the outstanding shares and options of the company included 8,500,000 shares of Common Stock and 389,500 shares of Common Stock that are issuable upon exercise of employee stock options. Immediately prior to the initial closing of this offering, the SAFE securities will convert into 1,986,667 shares of Series A Preferred Stock, the KISS securities will convert into 1,881,667 shares of Series A Preferred Stock and the Crowd Notes will convert into 414,404 shares of Series A Preferred Stock. Accrued interest on the KISS securities will be paid in cash at the time of conversion.
Common Stock
Voting Rights
Each holder of the company’s Common Stock is entitled to one vote for each share on all matters submitted to a vote of the stockholders, including the election of directors.
Dividend Rights
Holders of Common Stock are entitled to receive dividends, as may be declared from time to time by the Board of Directors out of legally available funds and on a pari passu basis with holders of the Series A Preferred Stock, as detailed in the company’s amended and restated certificate of incorporation. The company has never declared or paid cash dividends on any of its capital stock and currently does not anticipate paying any cash dividends after this offering or in the foreseeable future.
Liquidation Rights
In the event of a voluntary or involuntary liquidation, dissolution, or winding up of the company, the holders of Common Stock are entitled to share ratably in the net assets legally available for distribution to stockholders after the payment of all debts and other liabilities of the company and the satisfaction of any liquidation preference granted to the holders of all shares of the outstanding Preferred Stock.
| 53 |
Rights and Preferences
Holders of the company's Common Stock have no preemptive, conversion, or other rights, and there are no redemptive or sinking fund provisions applicable to the company's Common Stock.
Series A Preferred Stock
Voting Rights
Each holder of Series A Preferred Stock will be entitled to one vote for each share of Common Stock into which such share of Preferred Stock could be converted. Fractional votes will not be permitted and if the conversion results in a fractional share, it will be disregarded. Holders of Preferred Stock will be entitled to vote on all matters submitted to a vote of the stockholders, including the election of directors, as a single class with the holders of Common Stock. Investors who invest less than $50,000 in this offering will be required to sign the Investor Proxy Agreement, described below in “—Investor Proxy Agreement.”
Dividend Rights
Holders of Series A Preferred Stock will be entitled to receive dividends as may be declared from time to time by the Board of Directors out of legally available funds and on a pari passu basis with holders of the Common Stock. The company has never declared or paid cash dividends on any of its capital stock and currently does not anticipate paying any cash dividends after this offering or in the foreseeable future.
Conversion Rights
Shares of Series A Preferred Stock will be convertible, at the option of the holder, at any time, into fully paid and nonassessable shares of the company’s Common Stock at the then-applicable conversion rate. Initially, the conversion rate for each series of Preferred Stock will be one share of Common Stock per share of Series A Preferred Stock. The conversion rate is subject to adjustment in the event of stock splits, reverse stock splits or the issuance of a dividend or other distribution payable in additional shares of Common Stock.
Additionally, each share of Series A Preferred Stock will automatically convert into common stock immediately prior to the closing of a firm commitment underwritten public offering, registered under the Securities Act or upon the affirmative election of the holders of a majority of the outstanding shares of Preferred Stock, voting as a single class and on an as-converted basis. The shares will convert in the same manner as a voluntary conversion.
| 54 |
Right to Receive Liquidation Distributions
In the event of a liquidation, dissolution or winding up of the company, whether voluntary or involuntary, or certain other events (each a “Deemed Liquidation Event”) such as the sale or merger of the company, as further set forth in the amended and restated certificate of incorporation, all holders of Series A Preferred Stock will be entitled to a liquidation preference that is senior to holders of the Common Stock. Holders of Series A Preferred Stock will receive a liquidation preference equal to the greater of (a) an amount for each share equal to the Original Issue Price for such share, adjusted for any stock dividends, combinations, splits, recapitalizations and the like (the “liquidation preference”) plus any declared but unpaid dividends with respect to such shares or (b) such amount per share as would have been payable had all shares of Series A Preferred Stock been converted into Common Stock immediately prior to such liquidation, dissolution or winding up or Deemed Liquidation Event.. Initially, the liquidation preferences for the shares of Series A Preferred Stock will be $2.00 per share (the “Original Issue Price”).
If, upon such liquidation, dissolution, or winding up or Deemed Liquidation Event, the assets (or the consideration received in a transaction) that are distributable to the holders of Preferred Stock are insufficient to permit the payment to such holders of the full amount of their respective liquidation preference, then all of such funds will be distributed ratably among the holders of the Preferred Stock in proportion to the full amounts to which they would otherwise be entitled to receive.
After the payment of the full liquidation preference of the Series A Preferred Stock, the remaining assets of the company legally available for distribution (or the consideration received in a transaction), if any, will be distributed ratably to the holders of the Common Stock in proportion to the number of shares of Common Stock held by each such holder.
Convertible Securities
The company has outstanding 3 series of convertible securities, all of which will be converted into shares of Series A Preferred Stock immediately prior to the initial closing of this offering. Upon the conversion, the company will retire the convertible securities.
SAFE Securities
The company issued 2 series of simple agreements for future equity (“SAFE securities”) for $1,067,500 in 2015 and 2016 (the “2015-2016 series”) and $195,000 in 2017 (the “2017 series”). $40,000 of the 2015-2016 series were rescinded during the first six months of 2017. Four individual holders of 2015-2016 series SAFE securities were issued warrants that granted them the right to increase their investment by 100% before conversion, with the identical terms, up to $47,500 in total.
| 55 |
Interest Rate and Maturity
The SAFE securities issued in the 2015-2016 series have a $6,000,000 valuation cap, an 80% discount rate, do not bear interest, and have no maturity date. The SAFE securities issued in the 2017 series have a $10,000,000 valuation cap, a 90% discount rate, do not bear interest, and have no maturity date.
Conversion terms
The SAFE securities issued in the 2015-2016 series convert upon a qualified equity financing into a number of shares of preferred stock equal to the amount purchased by a holder divided by either the price per share implied by a pre-money valuation of $6,000,000 on the company’s fully diluted capitalization or the price per share calculated at a 20% discount to the pricing in the triggering equity financing. The SAFE securities issued in the 2017 series convert upon a qualified equity financing into a number of shares of preferred stock equal to the amount purchased by a holder divided by either the price per share implied by a pre-money valuation of $10,000,000 on the company’s fully diluted capitalization or the price per share calculated at a 10% discount to the pricing in the triggering equity financing. The initial closing of this offering will trigger the conversion provision of all SAFE securities.
KISS Agreement
The company entered into KISS agreements for $50,000 in 2016 and for $854,000 during the first six months of 2017. Since June 30, 2017, the company has issued additional KISS securities for total proceeds of $375,000.
Interest Rate and Maturity
The KISS agreements bear an interest rate of 5% and have a maturity date 24 months from issuance.
Conversion terms
The KISS agreements issued in 2016 and early 2017 convert upon the company completing a qualified preferred financing that raises at least $1,000,000 in proceeds into preferred stock at a 20% discount to the pricing in the triggering financing or at a price per share implied by a pre-money valuation of $6,000,000 on the company’s fully diluted capitalization. The KISS agreements issued since June 30, 2017 convert upon the company completing a qualified preferred financing that raises at least $1,000,000 in proceeds into preferred stock at a 10% discount to the pricing in the triggering financing or at a price per share implied by a pre-money valuation of $10,000,000 on the company’s fully diluted capitalization. Should the proceeds of the initial closing of this offering be less than $1,000,000, the company will grant the holders most-favored-nation status with the holders of the SAFE securities and convert all the KISS agreements immediately prior to the initial closing.
| 56 |
Alternatively, the KISS agreements convert upon a corporate transaction at the share price implied by a $6,000,000 or $10,000,000 pre-money valuation, as the case may be, or are payable to the holder at two times the principal amount, at the holder’s election.
Crowd Notes
The company issued $209,230 in Crowd Notes during the first six months of 2017, plus an additional $39,412 of Crowd Notes issued to SI Securities, LLC.
Interest Rate and Maturity
The Crowd Notes of the company bear an interest rate of 5% and have no maturity date.
Conversion terms
The Crowd Notes convert upon a qualified preferred financing that raises at least $1,000,000 into preferred stock at a 20% discount to the pricing in the triggering financing or at a the price per share implied by the pre-money valuation of $6,000,000 on the company’s fully diluted capitalization. Should the proceeds of the initial closing of this offering be less than $1,000,000, the company will grant the holders most-favored-nation status with the holders of the SAFE securities and convert the Crowd Notes.
Investor Proxy Agreement
Investors who invest less than $50,000 in this offering will be required to sign the Investor Proxy Agreement with SI Securities, LLC. Investors who invest more than $50,000 will not be parties to the Investor Proxy Agreement. The following summary of the Investor Proxy Agreement is qualified in its entirety by the Investor Proxy Agreement itself, which is filed as Exhibit 4.2 to the Offering Statement of which this Offering Circular is a part:
| · | The investor irrevocably appoints SI Securities, LLC, to vote, exercise and/or waive any right, power and authority, and convert the investor’s shares of Series A Preferred Stock, as well as the investor’s shares of Common Stock of the company following the conversion of the Series A Preferred Stock; |
| · | Prior to undertaking any action or conversion, SI Securities, LLC may be required to request approval of the investors party to the agreement on a majority or super-majority basis, as determined by the Investor Proxy Agreement. Investors will have seven calendar days to approve or deny the action. If an investor does not approve or deny the action within that seven calendar day period, the investor relinquishes the right to approve or deny the action. Should majority or super-majority approval or denial be received, SI Securities, LLC will act for all of the shares as if unanimous approval or denial was received; |
| 57 |
| · | SI Securities, LLC is not restricted from acquiring a financial interest in the company, creating a potential for a conflict of interest. SI Securities, LLC holds Crowd Notes that it received as compensation in connection with earlier offerings by the company, which will convert into 65,687 shares of Series A Preferred Stock of the company immediately prior to the initial closing of the offering. If investors do not indicate their voting preference prior to the expiration of the voting period described above, SI Securities, LLC may vote those investors' shares in a way with which the investors do not agree; |
| · | Except as otherwise approved by a majority of investors party to the agreement, investors are not obligated to compensate SI Securities, LLC for its activities the Investor Proxy Agreement and SI Securities, LLC is required to bear the expense of its activities under the Investor Proxy Agreement; |
| · | SI Securities, LLC may only be removed from its activities and powers under the Investor Proxy Agreement by super-majority vote of investors party to the agreement; |
| · | The Investor Proxy Agreement is effective until one of the following events occurs: (1) upon the liquidation or sale of the company, (2) approval by a majority of investors party to the agreement to terminate the Investor Proxy Agreement, or (3) the resignation or removal of SI Securities, LLC as the appointee under the agreement without the appointment of a new appointee by investors; |
| · | Investors party to the agreement may only transfer their shares once their transferee has executed the Investor Proxy Agreement and agrees to assume all of the duties and obligations under the agreement and to be bound by and subject to all of the terms and conditions of the Investor Proxy Agreement; |
| · | Under the Investor Proxy Agreement, SI Securities, LLC is not liable to investors party to the agreement for any act or failure to act except in the event of fraud, gross negligence, willful malfeasance, intentional and material breach of the Investor Proxy Agreement, or conduct that is the subject of a criminal proceeding, provided that SI Securities, LLC knew the conduct was illegal; and |
| · | Investors party to the agreement are required to indemnify and reimburse expenses of SI Securities, LLC for any legal claims against SI Securities, LLC as a result of any act or failure to act in connection with its activities under the Investor Proxy Agreement |
| 58 |
PLAN OF DISTRIBUTION AND SELLING SECURITYHOLDERS
The company is offering a minimum of 225,000 and up to 3,500,000 shares of Series A Preferred Stock, as described in this Offering Circular. The company has engaged SI Securities, LLC as its sole and exclusive placement agent to assist in the placement of its securities. SI Securities, LLC is under no obligation to purchase any securities or arrange for the sale of any specific number or dollar amount of securities.
Commissions and Discounts
The following table shows the total discounts and commissions payable to the placement agents in connection with this offering:
| Per Share | ||||
| Public offering price | $ | 2.00 | ||
| Placement Agent commissions | $ | 0.15 | ||
| Proceeds, before expenses, to us | $ | 1.85 | ||
Placement Agent Equity
The company has agreed to issue to SI Securities, LLC, for nominal consideration, 5% of the total number of shares of Series A Preferred Stock successfully placed by SI Securities, LLC (“Equity Compensation”). The shares of Series A Preferred Stock issuable will have identical rights, preferences, and privileges to those being offered by this Offering Circular.
The Equity Compensation has been deemed compensation by FINRA and is therefore subject to a 180-day lock-up pursuant to FINRA Rule 5110(g)(1). In accordance with FINRA Rule 5110(g)(1), the Equity Compensation may not be sold, transferred, assigned, pledged or hypothecated, or be the subject of any hedging, short sale, derivative, put, or call transaction that would result in the effective economic disposition of such securities by any person for a period of 180 days immediately following the qualification date or commencement of sales of this offering, except to any placement agent and selected dealer participating in the offering and their bona fide officers or partners and except as otherwise provided for in FINRA Rule 5110(g)(2).
Other Terms
Except as set forth above, the company is not under any contractual obligation to engage SI Securities, LLC to provide any services to the company after this offering, and has no present intent to do so. However, SI Securities, LLC may, among other things, introduce the company to potential target businesses or assist the company in raising additional capital, as needs may arise in the future. If SI Securities, LLC provides services to the company after this offering, the company may pay SI Securities, LLC fair and reasonable fees that would be determined at that time in an arm’s length negotiation.
| 59 |
SI Securities, LLC intends to use an online platform provided by SeedInvest Technology, LLC, an affiliate of SI Securities, LLC, at the domain name www.seedinvest.com (the “Online Platform”) to provide technology tools to allow for the sales of securities in this offering. SeedInvest will charge you a non-refundable transaction fee equal to 2% of the amount you invest (up to $300) at the time you subscribe for our shares. This fee will be refunded in the event the company does not reach its minimum fundraising goal. In addition, SI Securities, LLC may engage selling agents in connection with the offering to assist with the placement of securities.
Selling Security holders
No securities are being sold for the account of security holders; all net proceeds of this offering will go to the company.
Investors’ Tender of Funds
After the Offering Statement has been qualified by the Commission, the company will accept tenders of funds to purchase the Series A Preferred Stock. The company may close on investments on a “rolling” basis (so not all investors will receive their shares on the same date), provided that the minimum offering amount has been met. Investors may subscribe by tendering funds via wire or ACH only, checks will not be accepted, to the escrow account to be setup by the Escrow Agent. Tendered funds will remain in escrow until both the minimum offering amount has been reached and a closing has occurred. However, in the event we have not sold the minimum amount of shares by _____, 2018, or sooner terminated by the company, any money tendered by potential investors will be promptly returned by the Escrow Agent. Upon closing, funds tendered by investors will be made available to the company for its use.
In order to invest you will be required to subscribe to the offering via the Online Platform and agree to the terms of the offering, Series A Preferred Stock Purchase Agreement, and any other relevant exhibit attached thereto. In addition, investors who invest less than $50,000 will be required to sign the Investor Proxy Agreement, a copy of which has been filed as an exhibit to the Offering Statement of which this Offering Circular is a part
In the event that it takes some time for the company to raise funds in this offering, the company will rely on income from sales, funds raised in any offerings from accredited investors.
At stepped investment levels, the company plans to offer investment packages that provide various incentives. The company plans to offer the following benefits at various levels of investment:
Tier 1 ($10,000+)
| · | Free T-Shirt and Mug to commemorate the offering. |
Tier 2 ($50,000+)
All of the above plus:
| · | option to name a dispensing robot in a location, including receiving a signed photo of the "christened" robot. |
Tier 3 ($150,000+)
All of the above plus:
| · | regular scheduled quarterly call with senior management. |
Tier 4 ($250,000+)
All of the above plus:
| · | Paid airfare to a NowRx location for a personal tour and dinner with management team. |
| · | Regular annual dinner with the NowRx senior management team. |
| 60 |
TAX CONSEQUENCES FOR RECIPIENT (INCLUDING FEDERAL, STATE, LOCAL AND FOREIGN INCOME TAX CONSEQUENCES) WITH RESPECT TO THE INVESTMENT BENEFIT PACKAGES ARE THE SOLE RESPONSIBILITY OF THE INVESTOR. INVESTORS MUST CONSULT WITH THEIR OWN PERSONAL ACCOUNTANT(S) AND/OR TAX ADVISOR(S) REGARDING THESE MATTERS.
| 61 |
A Delaware Corporation
Financial Statements and Independent Auditor’s Report
December 31, 2016 and 2015
| NowRx, Inc. |
| TABLE OF CONTENTS |
| Page | |
| INDEPENDENT AUDITOR’S REPORT | F-1 |
| FINANCIAL STATEMENTS AS OF DECEMBER 31, 2016 AND 2015 AND FOR THE PERIODS THEN ENDED: | |
| Balance Sheets | F-2 |
| Statements of Operations | F-3 |
| Statements of Changes in Stockholders’ Equity (Deficit) | F-4 |
| Statements of Cash Flows | F-5 |
| Notes to Financial Statements | F-6 |
To the Board of Directors and Stockholders
of NowRx, Inc.
We have audited the accompanying financial statements of NowRx, Inc., which comprise the balance sheets as of December 31, 2016 and 2015, and the related statements of operations, changes in stockholders’ equity (deficit), and cash flows for the years then ended, and the related notes to the financial statements.
Management’s Responsibility for the Financial Statements
Management is responsible for the preparation and fair presentation of these financial statements in accordance with accounting principles generally accepted in the United States of America; this includes the design, implementation, and maintenance of internal control relevant to the preparation and fair presentation of financial statements that are free from material misstatement, whether due to fraud or error.
Auditor’s Responsibility
Our responsibility is to express an opinion on these financial statements based on our audit. We conducted our audit in accordance with auditing standards generally accepted in the United States of America. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free from material misstatement.
An audit involves performing procedures to obtain audit evidence about the amounts and disclosures in the financial statements. The procedures selected depend on the auditor’s judgment, including the assessment of the risks of material misstatement of the financial statements, whether due to fraud or error. In making those risk assessments, the auditor considers internal control relevant to the entity’s preparation and fair presentation of the financial statements in order to design audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the entity’s internal control. Accordingly, we express no such opinion. An audit also includes evaluating the appropriateness of accounting policies used and the reasonableness of significant accounting estimates made by management, as well as evaluating the overall presentation of the financial statements.
We believe that the audit evidence we have obtained is sufficient and appropriate to provide a basis for our audit opinion.
Opinion
In our opinion, the financial statements referred to above present fairly, in all material respects, the financial position of NowRx, Inc. as of December 31, 2016 and 2015, and the results of its operations and its cash flows for the years then ended in accordance with accounting principles generally accepted in the United States of America.
Going Concern
The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. As disclosed in Note 3 of the financial statements, NowRx, Inc. has not yet generated profits and has an accumulated deficit that raise substantial doubt about its ability to continue as a going concern. Management’s plans in regard to these matters are also described in Note 3. The financial statements do not include any adjustments that might result from the outcome of this uncertainty.

Spokane, Washington
December 27, 2017
| F-1 |
| NOWRX, INC. |
| BALANCE SHEETS |
| As of December 31, 2016 and 2015 |
| 2016 | 2015 | |||||||
| ASSETS | ||||||||
| Current Assets: | ||||||||
| Cash and equivalents | $ | 175,519 | $ | 160,765 | ||||
| Accounts receivable, net | 128,552 | 12,826 | ||||||
| Inventory | 97,994 | 86,993 | ||||||
| Deposits | 13,898 | 13,898 | ||||||
| Total Current Assets | 415,963 | 274,482 | ||||||
| Property and equipment, net | 43,522 | 33,679 | ||||||
| TOTAL ASSETS | $ | 459,485 | $ | 308,161 | ||||
| LIABILITIES AND STOCKHOLDERS' EQUITY (DEFICIT) | ||||||||
| Current Liabilities: | ||||||||
| Accounts payable | $ | 67,207 | $ | - | ||||
| Accrued liabilities | 11,366 | 14,715 | ||||||
| Accrued interest | 237 | - | ||||||
| Short-term inventory financing | 62,100 | 94,836 | ||||||
| Short-term notes payable | 70,000 | - | ||||||
| Total Current Liabilities | 210,910 | 109,551 | ||||||
| Non-Current Liabilities: | ||||||||
| SAFE liability | 1,067,500 | 445,000 | ||||||
| KISS liability | 50,000 | - | ||||||
| Total Non-Current Liabilities | 1,117,500 | 445,000 | ||||||
| Total Liabilities | 1,328,410 | 554,551 | ||||||
| Stockholders' Equity (Deficit): | ||||||||
| Common stock, $0.00001 par value, 10,000,000 shares authorized, 8,500,000 and 8,500,000 shares issued and outstanding, 3,364,583 and 1,239,583 vested as of December 31, 2016 and 2015, respectively. | 85 | 85 | ||||||
| Additional paid-in capital | 765 | 765 | ||||||
| Accumulated deficit | (869,775 | ) | (247,240 | ) | ||||
| Total Stockholders' Equity (Deficit) | (868,925 | ) | (246,390 | ) | ||||
| TOTAL LIABILITIES AND STOCKHOLDERS' EQUITY (DEFICIT) | $ | 459,485 | $ | 308,161 | ||||
See Independent Auditor’s Report and accompanying notes, which are an integral part of these financial statements.
| F-2 |
| NOWRX, INC. |
| STATEMENTS OF OPERATIONS |
| For the year ended December 31, 2016 and for the period from February 19, 2015 (inception) to December 31, 2015 |
| 2016 | 2015 | |||||||
| Sales, net | $ | 668,695 | $ | - | ||||
| Cost of goods sold | (573,791 | ) | - | |||||
| Gross profit | 94,904 | - | ||||||
| Operating Expenses: | ||||||||
| General and administrative | 655,755 | 196,716 | ||||||
| Sales and marketing | 55,002 | 8,293 | ||||||
| Depreciation expense | 5,053 | 1,441 | ||||||
| Research and development | 1,392 | 40,790 | ||||||
| Total Operating Expenses | 717,202 | 247,240 | ||||||
| Loss from operations | (622,298 | ) | (247,240 | ) | ||||
| Other Expenses: | ||||||||
| Interest expense | (237 | ) | - | |||||
| Total Other Expenses | (237 | ) | - | |||||
| Net Loss | $ | (622,535 | ) | $ | (247,240 | ) | ||
| Weighted-average vested common shares outstanding: | ||||||||
| - Basic and Diluted | 2,302,083 | 450,758 | ||||||
| Net loss per common share | ||||||||
| - Basic and Diluted | $ | (0.27 | ) | $ | (0.55 | ) | ||
See Independent Auditor’s Report and accompanying notes, which are an integral part of these financial statements.
| F-3 |
| NOWRX, INC. |
| STATEMENTS OF CHANGES IN STOCKHOLDERS’ EQUITY (DEFICIT) |
| For the year ended December 31, 2016 and for the period from February 19, 2015 (inception) to December 31, 2015 |
| Common Stock | Additional | Total Stockholders' | ||||||||||||||||||
| Number of Shares | Amount | Paid-In Capital | Accumulated Deficit | Equity (Deficit) | ||||||||||||||||
| Balance at February 19, 2015 (Inception) | - | $ | - | $ | - | $ | - | $ | - | |||||||||||
| Issuance of common stock | 8,500,000 | 85 | 765 | - | 850 | |||||||||||||||
| Net loss | - | - | - | (247,240 | ) | (247,240 | ) | |||||||||||||
| Balance at December 31, 2015 | 8,500,000 | 85 | 765 | (247,240 | ) | (246,390 | ) | |||||||||||||
| Net loss | - | - | - | (622,535 | ) | (622,535 | ) | |||||||||||||
| Balance at December 31, 2016 | 8,500,000 | $ | 85 | $ | 765 | $ | (869,775 | ) | $ | (868,925 | ) | |||||||||
See Independent Auditor’s Report and accompanying notes, which are an integral part of these financial statements.
| F-4 |
| NOWRX, INC. |
| STATEMENTS OF CASH FLOWS |
| For the year ended December 31, 2016 and for the period from February 19, 2015 (inception) to December 31, 2015 |
| 2016 | 2015 | |||||||
| Cash Flows from Operating Activities | ||||||||
| Net Loss | $ | (622,535 | ) | $ | (247,240 | ) | ||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||
| Depreciation and amortization | 5,053 | 1,441 | ||||||
| Changes in operating assets and liabilities: | ||||||||
| Change in receivables | (115,727 | ) | (12,826 | ) | ||||
| Change in inventory | (11,001 | ) | (86,993 | ) | ||||
| Change in deposits | - | (13,898 | ) | |||||
| Change in accounts payable and accrued liabilities | 63,859 | 14,715 | ||||||
| Change in accrued interest | 237 | - | ||||||
| Net Cash Used in Operating Activities | (680,114 | ) | (344,801 | ) | ||||
| Cash Flows from Investing Activities | ||||||||
| Purchases of property and equipment | (14,896 | ) | (35,120 | ) | ||||
| Net Cash Used in Investing Activities | (14,896 | ) | (35,120 | ) | ||||
| Cash Flows from Financing Activities | ||||||||
| Proceeds/(repayments) short-term inventory financing | (32,736 | ) | 94,836 | |||||
| Proceeds from KISS agreements | 50,000 | - | ||||||
| Proceeds from SAFE agreements | 622,500 | 445,000 | ||||||
| Proceeds from short-term notes payable | 70,000 | - | ||||||
| Issuance of common stock to founders | - | 850 | ||||||
| Net Cash Provided by Financing Activities | 709,764 | 540,686 | ||||||
| Net Change In Cash | 14,754 | 160,765 | ||||||
| Cash at Beginning of Period | 160,765 | - | ||||||
| Cash at End of Period | $ | 175,519 | $ | 160,765 | ||||
| Supplemental Disclosure of Cash Flow Information: | ||||||||
| Cash paid for interest | $ | - | $ | - | ||||
| Cash paid for income taxes | $ | - | $ | - | ||||
See Independent Auditor’s Report and accompanying notes, which are an integral part of these financial statements.
| F-5 |
| NOWRX, INC. |
| NOTES TO FINANCIAL STATEMENTS |
| For the year ended December 31, 2016 and for the period from February 19, 2015 (inception) to December 31, 2015 |
NOTE 1: NATURE OF OPERATIONS
NowRx, Inc. (the “Company”), is a corporation organized February 19, 2015 under the laws of Delaware. The Company is an on-demand pharmacy that fulfills and delivers customer prescriptions using a web and application based platform.
NOTE 2: SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Basis of Presentation
The accounting and reporting policies of the Company conform to accounting principles generally accepted in the United States of America (GAAP).
The Company adopted the calendar year as its basis of reporting.
Use of Estimates
The preparation of financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosures of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. Actual results could differ from those estimates.
Risks and Uncertainties
The Company is dependent upon additional capital resources for its planned full scale operations and is subject to significant risks and uncertainties; including failing to secure funding to continue to operationalize the Company’s plans or failing to profitably operate the business.
Cash Equivalents and Concentration of Cash Balance
The Company considers all highly liquid securities with an original maturity of less than three months to be cash equivalents. The Company’s cash and cash equivalents in bank deposit accounts, at times, may exceed federally insured limits. As of December 31, 2016 and 2015, the Company’s cash balances did not exceed federally insured limits.
Accounts Receivable
The Company assesses its receivables based on historical loss patterns, aging of the receivables, and assessments of specific identifiable customer accounts considered at risk or uncollectible. The Company also considers any changes to the financial condition of its customers and any other external market factors that could impact the collectability of the receivables in the determination of the allowance for doubtful accounts. The Company has recorded allowances against its accounts receivable balances of $6,000 and $0 as of December 31, 2016 or 2015, respectively.
See accompanying Independent Auditor’s Report
| F-6 |
| NOWRX, INC. |
| NOTES TO FINANCIAL STATEMENTS |
| For the year ended December 31, 2016 and for the period from February 19, 2015 (inception) to December 31, 2015 |
Inventory
Inventory is stated at the lower of cost or market and accounted for using the weighted average cost method. The inventory balances of $97,994 and $86,993 as of December 31, 2016 and 2015, respectively, consisted of pharmaceuticals and related products. The Company records impairment and obsolescence reserves against its inventory balances as deemed necessary.
Property and Equipment
Property and equipment are recorded at cost when purchased. Depreciation is recorded for property and equipment using the straight-line method over the estimated useful lives of assets, which range from 5-15 years. The Company reviews the recoverability of all long-lived assets, including the related useful lives, whenever events or changes in circumstances indicate that the carrying amount of a long-lived asset might not be recoverable. Depreciation charges on property and equipment totaled $5,053 and $1,441 for the periods ended December 31, 2016 and 2015, respectively. The Company’s property and equipment consisted of the following as of December 31, 2016 and 2015:
| 2016 | 2015 | |||||||
| Automobiles | $ | 13,975 | $ | 12,646 | ||||
| Furniture and equipment | 13,929 | 5,020 | ||||||
| Leasehold improvements | 22,113 | 17,454 | ||||||
| Property and equipment, at cost | $ | 50,017 | $ | 35,120 | ||||
| Less: accumulated depreciation | (6,495 | ) | (1,441 | ) | ||||
| Property and equipment, net | $ | 43,522 | $ | 33,679 | ||||
Fair Value of Financial Instruments
Financial Accounting Standards Board (“FASB”) guidance specifies a hierarchy of valuation techniques based on whether the inputs to those valuation techniques are observable or unobservable. Observable inputs reflect market data obtained from independent sources, while unobservable inputs reflect market assumptions. The hierarchy gives the highest priority to unadjusted quoted prices in active markets for identical assets or liabilities (Level 1 measurement) and the lowest priority to unobservable inputs (Level 3 measurement). The three levels of the fair value hierarchy are as follows:
Level 1 - Unadjusted quoted prices in active markets for identical assets or liabilities that the reporting entity has the ability to access at the measurement date. Level 1 primarily consists of financial instruments whose value is based on quoted market prices such as exchange-traded instruments and listed equities.
Level 2 - Inputs other than quoted prices included within Level 1 that are observable for the asset or liability, either directly or indirectly (e.g., quoted prices of similar assets or liabilities in active markets, or quoted prices for identical or similar assets or liabilities in markets that are not active).
Level 3 - Unobservable inputs for the asset or liability. Financial instruments are considered Level 3 when their fair values are determined using pricing models, discounted cash flows or similar techniques and at least one significant model assumption or input is unobservable.
See accompanying Independent Auditor’s Report
| F-7 |
| NOWRX, INC. |
| NOTES TO FINANCIAL STATEMENTS |
| For the year ended December 31, 2016 and for the period from February 19, 2015 (inception) to December 31, 2015 |
The carrying amounts reported in the balance sheets approximate their fair value.
Revenue Recognition
The Company recognizes revenue when: (1) persuasive evidence exists of an arrangement with the customer reflecting the terms and conditions under which products or services will be provided; (2) delivery has occurred or services have been provided; (3) the fee is fixed or determinable; and (4) collection is reasonably assured. Cost of goods sold includes product costs, while delivery related costs of $85,825 for the year ended December 31, 2016 are included in general and administrative costs in the statement of operations.
Stock-Based Compensation
The Company accounts for stock-based compensation in accordance with ASC 718, Compensation - Stock Compensation. Under the fair value recognition provisions of ASC 718, stock-based compensation cost is measured at the grant date based on the fair value of the award and is recognized as expense ratably over the requisite service period, which is generally the option vesting period. The Company uses the Black-Scholes option pricing model to determine the fair value of stock options.
Research and Development
Research and development costs are expensed as incurred. Total expense related to research and development was $1,392 and $40,790 for the periods ended December 31, 2016 and 2015, respectively.
Income Taxes
The Company uses the liability method of accounting for income taxes as set forth in ASC 740, Income Taxes. Under the liability method, deferred taxes are determined based on the temporary differences between the financial statement and tax basis of assets and liabilities using tax rates expected to be in effect during the years in which the basis differences reverse. A valuation allowance is recorded when it is unlikely that the deferred tax assets will be realized.
The Company assesses its income tax positions and records tax benefits for all years subject to examination based upon its evaluation of the facts, circumstances and information available at the reporting date. In accordance with ASC 740-10, for those tax positions where there is a greater than 50% likelihood that a tax benefit will be sustained, our policy is to record the largest amount of tax benefit that is more likely than not to be realized upon ultimate settlement with a taxing authority that has full knowledge of all relevant information. For those income tax positions where there is less than 50% likelihood that a tax benefit will be sustained, no tax benefit will be recognized in the financial statements. The Company has determined that there are no material uncertain tax positions.
See accompanying Independent Auditor’s Report
| F-8 |
| NOWRX, INC. |
| NOTES TO FINANCIAL STATEMENTS |
| For the year ended December 31, 2016 and for the period from February 19, 2015 (inception) to December 31, 2015 |
The Company accounts for income taxes with the recognition of estimated income taxes payable or refundable on income tax returns for the current period and for the estimated future tax effect attributable to temporary differences and carryforwards. Measurement of deferred income items is based on enacted tax laws including tax rates, with the measurement of deferred income tax assets being reduced by available tax benefits not expected to be realized in the immediate future. The Company had net operating loss carryforwards of $814,252 and $231,986 as of December 31, 2016 and 2015, respectively. The Company pays Federal and California income taxes at rates of approximately 34% and 8.8%, respectively, and has used an effective blended rate of 39.8% to derive net tax assets of $329,553 and $96,384 as of December 31, 2016 and 2015, respectively, resulting from its net operating loss carryforwards and other temporary book to tax differences. Due to uncertainty as to the Company’s ability to generate sufficient taxable income in the future to utilize the net operating loss carryforwards before they begin to expire in 2035, the Company has recorded a full valuation allowance to reduce the net deferred tax asset to zero.
The Company files U.S. federal and state income tax returns. All previous tax returns have been filed. All tax periods since inception remain open to examination by the taxing jurisdictions to which the Company is subject.
Net Earnings or Loss per Share
Net earnings or loss per share is computed by dividing net income or loss by the weighted-average number of common shares outstanding during the period, excluding shares subject to redemption or forfeiture. The Company presents basic and diluted net earnings or loss per share. Diluted net earnings or loss per share reflect the actual weighted average of common shares issued and outstanding during the period, adjusted for potentially dilutive securities outstanding. Potentially dilutive securities are excluded from the computation of the diluted net earnings or loss per share if their inclusion would be anti-dilutive. As all potentially dilutive securities are anti-dilutive as of December 31, 2016 and 2015, diluted net loss per share is the same as basic net loss per share for each year.
NOTE 3: GOING CONCERN
The accompanying financial statements have been prepared on a going concern basis, which contemplates the realization of assets and the satisfaction of liabilities in the normal course of business. The Company is a business that has not yet generated profits, has sustained net losses of $622,535 and $247,240 during the periods ended December 31, 2016 and 2015, respectively, and has an accumulated deficit of $869,775 and $247,240 as of December 31, 2016 and 2015, respectively.
The Company’s ability to continue as a going concern in the next twelve months following the date the financial statements were available to be issued is dependent upon its ability to produce revenues and/or obtain financing sufficient to meet current and future obligations and deploy such to produce profitable operating results. Management has evaluated these conditions and plans to generate revenues and raise capital as needed to satisfy its capital needs. No assurance can be given that the Company will be successful in these efforts.
See accompanying Independent Auditor’s Report
| F-9 |
| NOWRX, INC. |
| NOTES TO FINANCIAL STATEMENTS |
| For the year ended December 31, 2016 and for the period from February 19, 2015 (inception) to December 31, 2015 |
These factors, among others, raise substantial doubt about the ability of the Company to continue as a going concern for a reasonable period of time. The financial statements do not include any adjustments relating to the recoverability and classification of recorded asset amounts or the amounts and classification of liabilities.
NOTE 4: FINANCING ARRANGEMENTS
Short-Term Inventory Financing
On December 4, 2015, the Company entered into an inventory financing agreement for $94,836 with its largest vendor. Terms of the loan called for a security interest in all personal property, 0% interest, and six monthly payments of $15,806 beginning in January 2016. During 2016, the Company repaid the loan in full. As of December 31, 2016 and 2015, $0 and $94,836 was outstanding under this arrangement, respectively.
On December 21, 2016, the Company entered into an inventory financing arrangement of $62,100 with Kabbage, an inventory financier. The loan is secured by all assets of the Company, bears an interest rate of 24.04% per annum, and is payable over six months, with expected average monthly payments of approximately $11,075, for a total repayment of $66,447. During the remainder of 2016, the Company deemed accrued interest to be trivial and therefore did not recognize interest expense during the period or an accrual as of December 31, 2016. As of December 31, 2016, the outstanding loan balance was $62,100.
Short-Term Notes Payable
On December 21, 2016, the Company entered into promissory notes totaling $70,000 with five individuals. All notes bear 12% interest, mature on April 30, 2017, and are due in full with accrued interest at maturity. These notes are collateralized by receivables, cash accounts, inventories, and all related proceeds. The notes are immediately callable in the event of (a) an equity financing of at least $1 million, or (b) insolvency or bankruptcy proceedings. As of December 31, 2016, principal of $70,000 and accrued interest of $237 were outstanding. During 2016, the Company recognized $237 of interest expense related to these notes.
KISS Agreements
On December 19, 2016, the Company entered into a KISS agreement (Keep it Simple Security) with a third party for $50,000. The instrument matures in 24 months and bears 5% interest per annum.
If and upon a qualified financing where the Company sells preferred stock of $1,000,000 or greater, the instrument’s face value along with accrued interest will automatically convert to preferred stock at a discount of 20% to the pricing in the triggering round or the price per share implied by a pre-money valuation of $6,000,000 on the Company’s fully diluted capitalization. Such a conversion does include a “shadow series” provision, which limits the holder’s liquidation preference to the discounted purchase price.
See accompanying Independent Auditor’s Report
| F-10 |
| NOWRX, INC. |
| NOTES TO FINANCIAL STATEMENTS |
| For the year ended December 31, 2016 and for the period from February 19, 2015 (inception) to December 31, 2015 |
If and upon a corporate transaction, the KISS becomes convertible into the number of shares determined by dividing the then outstanding principal and interest by the price per share implied by a pre-money valuation of $6,000,000 on the Company’s fully diluted capitalization, or is payable to the noteholder in the amount of two times the principal amount, at the noteholder’s election.
If at maturity neither of the aforementioned conversions have occurred, the noteholder may elect to convert the KISS and any unpaid accrued interest into the number of shares determined by dividing the then outstanding principal and interest by the price per share implied by a pre-money valuation of $6,000,000 on the Company’s fully diluted capitalization.
As of December 31, 2016, the KISS had not been converted and remained outstanding in the full principal amount. The Company analyzed the note for beneficial conversion features, and concluded the conversion terms did not constitute beneficial conversion features.
SAFE Agreements
In 2016 and 2015, the Company issued simple agreements for future equity (SAFE Agreement) in exchange for cash investments of $622,500 and $445,000, respectively. The SAFE Agreements entitle the holder to convert the SAFE agreements into the Company’s preferred stock (this classification of stock has not yet been authorized or established). The terms provide for automatic conversion of the SAFE agreements’ purchase amounts of $1,067,500 as of December 31, 2016 (the “Purchase Amount”) into the Company’s preferred stock if and upon a qualified equity financing event, which is generally defined as a transaction or series of transactions involving the issuance of the Company’s preferred stock at a fixed pre-money valuation. The number of shares of preferred stock the SAFE agreement converts into is the Purchase Amount divided by the price per share determined by either: a) a $6,000,000 pre-money valuation on the Company’s then outstanding capitalization (as further defined in the agreements), or b) a 20% discount to the share pricing in the triggering equity financing. $87,500 of the 2016 issuances contain an additional provision providing a future purchase right to additional purchases of preferred stock at the same terms and pricing as the SAFE conversions, which results in a potential increase to the aggregate purchase amount of $87,500.
In the case of a liquidation event (as defined in the SAFE agreement), the SAFE agreement is convertible into either: A) cash of the Purchase Amount; B) the number of shares determined by dividing the Purchase Amount by the Company’s then outstanding capitalization (as defined in the agreement).
The SAFE agreements provide holders with various additional protections, including preferences over unitholders in a dissolution event for payment of the Purchase Amount and anti-dilution protections. If the SAFE agreement converts into the Company’s preferred stock, it will have all the same rights and privileges of the preferred stock from the triggering financing, except that the liquidation preference will be equal to the Purchase Amount.
See accompanying Independent Auditor’s Report
| F-11 |
| NOWRX, INC. |
| NOTES TO FINANCIAL STATEMENTS |
| For the year ended December 31, 2016 and for the period from February 19, 2015 (inception) to December 31, 2015 |
NOTE 5: STOCKHOLDERS’ EQUITY (DEFICIT)
Common Stock
The Company has authorized 10,000,000 shares of $0.00001 par value common stock. As of each December 31, 2016 and 2015, 8,500,000 shares of common stock were issued and outstanding, respectively.
In June of 2015, the Company issued its two founders a total of 8,500,000 shares of common stock at $0.0001 per share, in exchange for $850 of cash and intellectual property. These stock issuances were conducted under terms of restricted stock purchase agreements and are subject to vesting terms contingent upon continuous service with the Company, which provide the Company the right to repurchase unvested shares at the original purchase price. These shares vest at 1/48 per month, commencing May 2015. As of December 31, 2016 and 2015, 3,364,583 and 1,239,583 of the shares had vested, respectively.
2015 Equity Incentive Plan
The Company adopted the2015 Equity Incentive Plan (the “Plan”), as amended and restated. The Plan permits the grant of stock options, stock appreciation rights and restricted stock to attract and retain employees and consultants. Under the plan, the Company issues stock appreciation rights and options having a term of up to ten years and a strike price of no less than fair market value of common stock. Restricted stock is subject to vesting restrictions determined on a case-by-case basis. While shares may be restricted, the restricted stockholder retains voting rights for each share, regardless of restriction. Upon termination of employment or services, the Company may exercise its repurchase option over unvested equity interests.
See accompanying Independent Auditor’s Report
| F-12 |
| NOWRX, INC. |
| NOTES TO FINANCIAL STATEMENTS |
| For the year ended December 31, 2016 and for the period from February 19, 2015 (inception) to December 31, 2015 |
The Company has reserved 1,500,000 shares of common stock under the Plan. As of December 31, 2016 and 2015, 1,053,000 and 1,105,500 shares of common stock were available for grant under the Plan, respectively. A summary of options activities during 2015 and 2016 is as follows:
| December 31, 2016 | December 31, 2015 | |||||||||||||||
| Options | Weighted Average Exercise Price | Options | Weighted Average Exercise Price | |||||||||||||
| Outstanding - beginning of year | 394,500 | $ | 0.05 | - | $ | 0.05 | ||||||||||
| Granted | 52,500 | $ | 0.05 | 394,500 | $ | 0.05 | ||||||||||
| Exercised | - | $ | 0.05 | - | $ | 0.05 | ||||||||||
| Forfeited | - | $ | 0.05 | - | $ | 0.05 | ||||||||||
| Outstanding - end of year | 447,000 | $ | 0.05 | 394,500 | $ | 0.05 | ||||||||||
| Exercisable at end of year | 168,486 | $ | 0.05 | 9,486 | $ | 0.05 | ||||||||||
| Weighted average grant date fair value of options granted during year | $ | 0.17 | $ | - | ||||||||||||
| Weighted average duration (years) to expiration of outstanding options at year-end | 9.0 | 9.8 | ||||||||||||||
The Company measures employee stock-based awards at grant-date fair value and recognizes employee compensation expense on a straight-line basis over the vesting period of the award. Determining the appropriate fair value of stock-based awards requires the input of subjective assumptions, including the fair value of the Company’s common stock, and for stock options, the expected life of the option, and expected stock price volatility. The Company used the Black-Scholes option pricing model to value its stock option awards. The assumptions used in calculating the fair value of stock-based awards represent management’s best estimates and involve inherent uncertainties and the application of management’s judgment. As a result, if factors change and management uses different assumptions, stock-based compensation expense could be materially different for future awards.
See accompanying Independent Auditor’s Report
| F-13 |
| NOWRX, INC. |
| NOTES TO FINANCIAL STATEMENTS |
| For the year ended December 31, 2016 and for the period from February 19, 2015 (inception) to December 31, 2015 |
The expected life of stock options was estimated using the “simplified method,” which is the midpoint between the vesting start date and the end of the contractual term, as the Company has limited historical information to develop reasonable expectations about future exercise patterns and employment duration for its stock options grants. The simplified method is based on the average of the vesting tranches and the contractual life of each grant. For stock price volatility, the Company uses comparable public companies as a basis for its expected volatility to calculate the fair value of options grants. The risk-free interest rate is based on U.S. Treasury notes with a term approximating the expected life of the option. The estimation of the number of stock awards that will ultimately vest requires judgment, and to the extent actual results or updated estimates differ from the Company’s current estimates, such amounts are recognized as an adjustment in the period in which estimates are revised.
The Company calculated its estimate of the value of the stock compensation granted during the years ended December 31, 2016 and 2015 under FASB ASC 718, and concluded the value of these options was trivial and therefore did not record compensation costs related to the stock option grants.
NOTE 6: RELATED PARTY TRANSACTIONS
The Company’s co-founders and an employee extended financing of $65,000 and $25,000 in 2015 to the Company through the SAFE agreements discussed in Note 4. In 2016, that employee issued another $10,000 of SAFE financing.
NOTE 7: LEASE ARRANGEMENTS
In June 2015, the Company entered into a 3-year operating lease agreement for office space. The agreement called for a security deposit of $8,898 and monthly payments of $4,194 for the first year, with subsequent annual rent increases of 3% over the next two years. Additionally, the Company is responsible for 18% of the homeowners’ association dues, which currently totals $200-$300 per months. Future minimum payments under this lease are as follows:
| 2017 | $ | 52,227 | ||
| 2018 | 40,041 | |||
| Total future minimum lease payments | $ | 92,268 |
In addition to this lease, the Company reimburses the CEO for an apartment on a month-to-month basis. Rents are $1,525 per month and are available for reimbursement monthly.
In 2016 and 2015, the Company recognized $53,196 and $25,699 of lease expense related to these leases, respectively.
See accompanying Independent Auditor’s Report
| F-14 |
| NOWRX, INC. |
| NOTES TO FINANCIAL STATEMENTS |
| For the year ended December 31, 2016 and for the period from February 19, 2015 (inception) to December 31, 2015 |
NOTE 8: COMMITMENTS AND CONTINGENCIES
The Company may be subject to pending legal proceedings and regulatory actions in the ordinary course of business. The results of such proceedings cannot be predicted with certainty, but the Company does not anticipate that the final outcome, if any, arising out of any such matter will have a material adverse effect on its business, financial condition or results of operations.
NOTE 9: CONCENTRATIONS
The Company has significant concentrations in its accounts receivables, where 81% and 100% of its accounts receivable balance as of December 31, 2016 and 2015, respectively, were held with one insurance payor. Should these insurance payors no longer be able to satisfy these statutory obligations to the Company, it would have a significant adverse effect to the Company.
Approximately 70% of the Company’s products are procured through a single supplier. Should this supplier no longer be willing or able to satisfy the Company’s product needs it could adversely affect the Company’s business.
NOTE 10: RECENT ACCOUNTING PRONOUNCEMENTS
In July 2014, the FASB issued the ASU No. 2015-11 on “Inventory (Topic 330): Simplifying the Measurement of Inventory”, which proposed that inventory should be measured at the lower of cost and the estimated selling prices in the ordinary course of business, less reasonably predictable costs of completion, disposal, and transportation. These amendments are based on existing guidance that requires measuring inventory at the lower of cost or market to consider the replacement cost of inventory less an approximately normal profit margin along with net value in determining the market value. It is effective for reporting periods beginning after December 15, 2016. Management is assessing the impact of this pronouncement on our financial statements.
Management does not believe that any recently issued, but not yet effective, accounting standards could have a material effect on the accompanying financial statements. As new accounting pronouncements are issued, the Company will adopt those that are applicable under the circumstances.
NOTE 11: SUBSEQUENT EVENTS
2017 Convertible Note Issuances
During 2017, the Company issued $876,902 of convertible notes payable, with terms similar to those discussed in Note 4.
Short-Term Notes Payable
The short-term note payable discussed in Note 4 was repaid in full during 2017.
See accompanying Independent Auditor’s Report
| F-15 |
| NOWRX, INC. |
| NOTES TO FINANCIAL STATEMENTS |
| For the year ended December 31, 2016 and for the period from February 19, 2015 (inception) to December 31, 2015 |
Management’s Evaluation
Management has evaluated subsequent events through December 27, 2017, the date the financial statements were available to be issued. Based on this evaluation, no additional material events were identified which require adjustment or disclosure in these financial statements.
See accompanying Independent Auditor’s Report
| F-16 |
NowRx, Inc.
A Delaware Corporation
Financial Statements (Unaudited)
June 30, 2017 and 2016
| F-17 |
NowRx, Inc.
TABLE OF CONTENTS
| Page | |
| FINANCIAL STATEMENTS AS OF JUNE 30, 2017 (UNAUDITED) AND DECEMBER 31, 2016 (AUDITED) AND FOR THE SIX-MONTH PERIODS ENDED JUNE 30, 2017 AND 2016 | |
| (UNAUDITED). | |
| Balance Sheets | F-19 |
| Statements of Operations | F-20 |
| Statements of Changes in Stockholders’ Equity (Deficit) | F-21 |
| Statements of Cash Flows | F-22 |
| Notes to Financial Statements | F-23 |
| F-18 |
NOWRX, INC.
As of June 30, 2017 (unaudited) and December 31, 2016 (audited)
| 6/30/2017 | 12/31/2016 | ||||||||
| ASSETS | |||||||||
| Current Assets: | |||||||||
| Cash and equivalents | $ | 391,723 | $ | 175,519 | |||||
| Accounts receivable, net | 172,543 | 128,552 | |||||||
| Other receivable | 78,329 | - | |||||||
| Inventory | 156,892 | 97,994 | |||||||
| Deposits | 18,898 | 13,898 | |||||||
| Total Current Assets | 818,385 | 415,963 | |||||||
| Property and equipment, net | 136,446 | 43,522 | |||||||
| TOTAL ASSETS | $ | 954,831 | $ | 459,485 | |||||
| LIABILITIES AND STOCKHOLDERS' EQUITY (DEFICIT) | |||||||||
| Current Liabilities: | |||||||||
| Accounts payable | $ | 105,609 | $ | 67,207 | |||||
| Accrued liabilities | - | 11,366 | |||||||
| Accrued interest | 9,921 | 237 | |||||||
| Short-term inventory financing | - | 62,100 | |||||||
| Short-term notes payable | - | 70,000 | |||||||
| Total Current Liabilities | 115,530 | 210,912 | |||||||
| Non-Current Liabilities: | |||||||||
| SAFE liability | 1,027,500 | 1,067,500 | |||||||
| Crowd notes payable | 184,288 | - | |||||||
| KISS liability | 860,943 | 50,000 | |||||||
| Total Non-Current Liabilities | 2,072,731 | 1,117,500 | |||||||
| Total Liabilities | 2,188,261 | 1,328,410 | |||||||
| Stockholders' Equity (Deficit): | |||||||||
| Common stock, $0.00001 par value, 10,000,000 shares authorized, 8,500,000 and 8,500,000 shares issued and outstanding, 4,427,083 and 3,364,583 vested as of June 30, 2017 and December 31, 2016 respectively. | 85 | 85 | |||||||
| Additional paid-in capital | 765 | 765 | |||||||
| Accumulated deficit | (1,234,280 | ) | (869,775 | ) | |||||
| Total Stockholders' Equity (Deficit) | (1,233,430 | ) | (868,925 | ) | |||||
| TOTAL LIABILITIES AND STOCKHOLDERS' EQUITY (DEFICIT) | $ | 954,831 | $ | 459,485 | |||||
See accompanying notes, which are an integral part of these financial statements.
| F-19 |
NOWRX, INC.
STATEMENTS OF OPERATIONS (UNAUDITED)
For the six-month periods ended June 30, 2017 and 2016
| 6/30/2017 | 6/30/2016 | |||||||
| Sales, net | $ | 994,588 | $ | 149,882 | ||||
| Cost of goods sold | (823,073 | ) | (116,148 | ) | ||||
| Gross profit | 171,515 | 33,734 | ||||||
| Operating Expenses: | ||||||||
| General and administrative | 461,278 | 312,296 | ||||||
| Sales and marketing | 47,768 | 28,846 | ||||||
| Depreciation expense | 2,677 | 2,527 | ||||||
| Research and development | 1,131 | 1,392 | ||||||
| Total Operating Expenses | 512,854 | 345,061 | ||||||
| Loss from operations | (341,339 | ) | (311,327 | ) | ||||
| Other Expenses: | ||||||||
| Interest expense | (23,166 | ) | - | |||||
| Total Other Expenses | (23,166 | ) | - | |||||
| Net Loss | $ | (364,505 | ) | $ | (311,327 | ) | ||
| Weighted-average vested common shares outstanding: | ||||||||
| - Basic and Diluted | 3,895,833 | 1,770,833 | ||||||
| Net loss per common share | ||||||||
| - Basic and Diluted | $ | (0.09 | ) | $ | (0.18 | ) | ||
See accompanying notes, which are an integral part of these financial statements. In the opinion of management all adjustments necessary in order to make the interim financial statements not misleading have been included
| F-20 |
NOWRX, INC.
STATEMENTS OF CHANGES IN STOCKHOLDERS’ EQUITY (DEFICIT) (UNAUDITED)
For the six-month periods ended June 30, 2017 and 2016
| Common Stock | ||||||||||||||||||||
| Number of Shares | Amount | Additional Paid-In Capital | Accumulated Deficit | Total Stockholders' Equity (Deficit) | ||||||||||||||||
| Balance at December 31, 2015 | 8,500,000 | $ | 85 | $ | 765 | $ | (247,240 | ) | $ | (246,390 | ) | |||||||||
| Net loss | - | - | - | (311,327 | ) | (311,327 | ) | |||||||||||||
| Balance at June 30, 2016 | 8,500,000 | $ | 85 | $ | 765 | $ | (558,567 | ) | $ | (557,717 | ) | |||||||||
| Net loss | - | - | - | (311,208 | ) | (311,208 | ) | |||||||||||||
| Balance at December 31, 2016 | 8,500,000 | $ | 85 | $ | 765 | $ | (869,775 | ) | $ | (868,925 | ) | |||||||||
| Net loss | - | - | - | (364,505 | ) | (364,505 | ) | |||||||||||||
| Balance at June 30, 2017 | 8,500,000 | $ | 85 | $ | 765 | $ | (1,234,280 | ) | $ | (1,233,430 | ) | |||||||||
See accompanying notes, which are an integral part of these financial statements.
| F-21 |
NOWRX, INC.
STATEMENTS OF CASH FLOWS (UNAUDITED)
For the six-month periods ended June 30, 2017 and 2016
| 6/30/2017 | 6/30/2016 | |||||||
| Cash Flows from Operating Activities | ||||||||
| Net Loss | $ | (364,505 | ) | $ | (311,327 | ) | ||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||
| Depreciation | 2,677 | 2,527 | ||||||
| Loan fee amortization | 6,268 | - | ||||||
| Changes in operating assets and liabilities: | ||||||||
| Change in accounts receivables | (43,991 | ) | (51,145 | ) | ||||
| Change in other receivables | (78,330 | ) | - | |||||
| Change in inventory | (58,898 | ) | 34,595 | |||||
| Change in deposits | (5,000 | ) | - | |||||
| Change in accounts payable and accrued liabilities | 27,036 | (14,713 | ) | |||||
| Change in accrued interest | 9,684 | - | ||||||
| Net Cash Used in Operating Activities | (505,059 | ) | (340,063 | ) | ||||
| Cash Flows from Investing Activities | ||||||||
| Purchases of property and equipment | (95,600 | ) | (4,623 | ) | ||||
| Net Cash Used in Investing Activities | (95,600 | ) | (4,623 | ) | ||||
| Cash Flows from Financing Activities | ||||||||
| Proceeds/(repayments) short-term inventory financing | (62,100 | ) | - | |||||
| Proceeds from Crowd Note issuances, net of placement fees | 184,288 | - | ||||||
| Proceeds from KISS note issuances, net of placement fees | 764,675 | - | ||||||
| Proceeds from SAFE agreements | - | 370,000 | ||||||
| Repayments of short-term notes payable | (70,000 | ) | (94,836 | ) | ||||
| Net Cash Provided by Financing Activities | 816,863 | 275,164 | ||||||
| Net Change In Cash | 216,204 | (69,522 | ) | |||||
| Cash at Beginning of Period | 175,519 | 160,765 | ||||||
| Cash at End of Period | $ | 391,723 | $ | 91,243 | ||||
| Supplemental Disclosure of Cash Flow Information: | ||||||||
| Cash paid for interest | $ | 13,482 | $ | - | ||||
| Cash paid for taxes | $ | - | $ | - | ||||
| Supplemental Disclosure of Non-Cash Financing Activities: | ||||||||
| Conversion of SAFE agreements to KISS notes payable | $ | 40,000 | $ | - | ||||
See accompanying notes, which are an integral part of these financial statements.
| F-22 |
NOWRX, INC.
As of June 30, 2017 (unaudited) and December 31, 2016 (audited) and for the six-month periods ended June 30, 2017 and 2016 (unaudited)
NOTE 1: NATURE OF OPERATIONS
NowRx, Inc. (the “Company”), is a corporation organized February 19, 2015 under the laws of Delaware. The Company is an on-demand pharmacy that fulfills and delivers customer prescriptions using a web and application based platform.
NOTE 2: SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Basis of Presentation
The accounting and reporting policies of the Company conform to accounting principles generally accepted in the United States of America (GAAP).
The Company adopted the calendar year as its basis of reporting.
Use of Estimates
The preparation of financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosures of contingent assets and liabilities at the date of the financial statements and the reported amounts of revenues and expenses during the reporting period. Actual results could differ from those estimates.
Risks and Uncertainties
The Company is dependent upon additional capital resources for its planned full scale operations and is subject to significant risks and uncertainties; including failing to secure funding to continue to operationalize the Company’s plans or failing to profitably operate the business.
Cash Equivalents and Concentration of Cash Balance
The Company considers all highly liquid securities with an original maturity of less than three months to be cash equivalents. The Company’s cash and cash equivalents in bank deposit accounts, at times, may exceed federally insured limits. As of June 30, 2017 and December 31, 2016, the Company’s cash balances exceeded federally insured limits by $125,723 and $0, respectively.
Accounts Receivable
The Company assesses its receivables based on historical loss patterns, aging of the receivables, and assessments of specific identifiable customer accounts considered at risk or uncollectible. The Company also considers any changes to the financial condition of its customers and any other external market factors that could impact the collectability of the receivables in the determination of the allowance for doubtful accounts. The Company has recorded an allowance against its accounts receivable balances of $6,000 and $6,000 as of June 30, 2017 and December 31, 2016, respectively.
Inventory
Inventory is stated at the lower of cost or market and accounted for using the weighted average cost method. The inventory balances of $156,892 and $97,994 as of June 30, 2017 and December 31, 2016, respectively, consisted of pharmaceuticals and related products. The Company records impairment and obsolescence reserves against its inventory balances as deemed necessary.
| F-23 |
NOWRX, INC.
NOTES TO FINANCIAL STATEMENTS
As of June 30, 2017 (unaudited) and December 31, 2016 (audited) and for the six-month periods ended June 30, 2017 and 2016 (unaudited)
Property and Equipment
Property and equipment are recorded at cost when purchased. Depreciation is recorded for property and equipment using the straight-line method over the estimated useful lives of assets, which range from 5-15 years. The Company reviews the recoverability of all long-lived assets, including the related useful lives, whenever events or changes in circumstances indicate that the carrying amount of a long-lived asset might not be recoverable. Depreciation charges on property and equipment totaled $2,677 and $2,527 for the periods ended June 30, 2017 and 2016, respectively. The Company’s property and equipment consisted of the following as of June 30, 2017 and December 31, 2016:
| 6/30/2017 | 12/31/2016 | |||||||
| Automobiles | $ | 13,975 | $ | 13,975 | ||||
| Furniture and equipment | 13,929 | 13,929 | ||||||
| Leasehold improvements | 117,713 | 22,113 | ||||||
| Property and equipment, at cost | $ | 145,617 | $ | 50,017 | ||||
| Less: accumulated depreciation | (9,171 | ) | (6,495 | ) | ||||
| Property and equipment, net | $ | 136,446 | $ | 43,522 | ||||
Fair Value of Financial Instruments
Financial Accounting Standards Board (“FASB”) guidance specifies a hierarchy of valuation techniques based on whether the inputs to those valuation techniques are observable or unobservable. Observable inputs reflect market data obtained from independent sources, while unobservable inputs reflect market assumptions. The hierarchy gives the highest priority to unadjusted quoted prices in active markets for identical assets or liabilities (Level 1 measurement) and the lowest priority to unobservable inputs (Level 3 measurement). The three levels of the fair value hierarchy are as follows:
Level 1 - Unadjusted quoted prices in active markets for identical assets or liabilities that the reporting entity has the ability to access at the measurement date. Level 1 primarily consists of financial instruments whose value is based on quoted market prices such as exchange-traded instruments and listed equities.
Level 2 - Inputs other than quoted prices included within Level 1 that are observable for the asset or liability, either directly or indirectly (e.g., quoted prices of similar assets or liabilities in active markets, or quoted prices for identical or similar assets or liabilities in markets that are not active).
Level 3 - Unobservable inputs for the asset or liability. Financial instruments are considered Level 3 when their fair values are determined using pricing models, discounted cash flows or similar techniques and at least one significant model assumption or input is unobservable.
The carrying amounts reported in the balance sheets approximate their fair value.
Revenue Recognition
The Company recognizes revenue when: (1) persuasive evidence exists of an arrangement with the customer reflecting the terms and conditions under which products or services will be provided;
| F-24 |
NOWRX, INC.
NOTES TO FINANCIAL STATEMENTS
As of June 30, 2017 (unaudited) and December 31, 2016 (audited) and for the six-month periods ended June 30, 2017 and 2016 (unaudited)
(2) delivery has occurred or services have been provided; (3) the fee is fixed or determinable; and (4) collection is reasonably assured. Cost of goods sold includes product costs, while delivery related costs are included in general and administrative costs in the statement of operations.
Stock-Based Compensation
The Company accounts for stock-based compensation in accordance with ASC 718, Compensation - Stock Compensation. Under the fair value recognition provisions of ASC 718, stock-based compensation cost is measured at the grant date based on the fair value of the award and is recognized as expense ratably over the requisite service period, which is generally the option vesting period. The Company uses the Black-Scholes option pricing model to determine the fair value of stock options.
Research and Development
Research and development costs are expensed as incurred. Total expense related to research and development was $1,131 and $1,392 for the periods ended June 30, 2017 and 2016, respectively.
Income Taxes
The Company uses the liability method of accounting for income taxes as set forth in ASC 740, Income Taxes. Under the liability method, deferred taxes are determined based on the temporary differences between the financial statement and tax basis of assets and liabilities using tax rates expected to be in effect during the years in which the basis differences reverse. A valuation allowance is recorded when it is unlikely that the deferred tax assets will be realized.
The Company assesses its income tax positions and records tax benefits for all years subject to examination based upon its evaluation of the facts, circumstances and information available at the reporting date. In accordance with ASC 740-10, for those tax positions where there is a greater than 50% likelihood that a tax benefit will be sustained, our policy is to record the largest amount of tax benefit that is more likely than not to be realized upon ultimate settlement with a taxing authority that has full knowledge of all relevant information. For those income tax positions where there is less than 50% likelihood that a tax benefit will be sustained, no tax benefit will be recognized in the financial statements. The Company has determined that there are no material uncertain tax positions.
The Company accounts for income taxes with the recognition of estimated income taxes payable or refundable on income tax returns for the current period and for the estimated future tax effect attributable to temporary differences and carryforwards. Measurement of deferred income items is based on enacted tax laws including tax rates, with the measurement of deferred income tax assets being reduced by available tax benefits not expected to be realized in the immediate future. The Company had net operating loss carryforwards of $1,217,134 and $814,252 as of June 30, 2017 and December 31, 2016, respectively. The Company pays Federal and California income taxes at rates of approximately 34% and 8.8%, respectively, and has used an effective blended rate of 39.8% to derive net tax assets of $484,419 and $329,553 as of June 30, 2017 and December 31, 2016, respectively, resulting from its net operating loss carryforwards and other temporary book to tax differences. Due to uncertainty as to the Company’s ability to generate sufficient taxable income in the future to utilize the net operating loss carryforwards before they begin to expire in 2035, the Company has recorded a full valuation allowance to reduce the net deferred tax asset to zero.
| F-25 |
NOWRX, INC.
NOTES TO FINANCIAL STATEMENTS
As of June 30, 2017 (unaudited) and December 31, 2016 (audited) and for the six-month periods ended June 30, 2017 and 2016 (unaudited)
The Company files U.S. federal and state income tax returns. All previous tax returns have been filed. All tax periods since inception remain open to examination by the taxing jurisdictions to which the Company is subject.
NOTE 3: GOING CONCERN
The accompanying financial statements have been prepared on a going concern basis, which contemplates the realization of assets and the satisfaction of liabilities in the normal course of business. The Company is a business that has not yet generated profits, has sustained net losses of $364,505 and $311,327 during the periods ended June 30, 2017 and 2016, respectively, and has an accumulated deficit of $1,234,280 and $869,775 as of June 30, 2017 and December 31, 2016, respectively.
The Company’s ability to continue as a going concern in the next twelve months following the date the financial statements were available to be issued is dependent upon its ability to produce revenues and/or obtain financing sufficient to meet current and future obligations and deploy such to produce profitable operating results. Management has evaluated these conditions and plans to generate revenues and raise capital as needed to satisfy its capital needs. No assurance can be given that the Company will be successful in these efforts.
These factors, among others, raise substantial doubt about the ability of the Company to continue as a going concern for a reasonable period of time. The financial statements do not include any adjustments relating to the recoverability and classification of recorded asset amounts or the amounts and classification of liabilities.
NOTE 4: FINANCING ARRANGEMENTS
Short-Term Inventory Financing
On December 4, 2015, the Company entered into an inventory financing agreement for $94,836 with its largest vendor. Terms of the loan called for a security interest in all personal property, 0% interest, and six monthly payments of $15,806 beginning in January 2016. During 2016, the Company repaid the loan in full. As of both June 30, 2017 and December 31, 2016, $0 was outstanding under this arrangement, respectively.
On December 21, 2016, the Company entered into an inventory financing arrangement of $62,100 with Kabbage, an inventory financier. The loan is secured by all assets of the Company, bears an interest rate of 24.04% per annum, and is payable over six months, with expected average monthly payments of approximately $11,075, for a total repayment of $66,447. This loan was repaid in full during the period ended June 30, 2017. As of June 30, 2017 and December 31, 2016, the outstanding loan balance was $0 and $62,100, respectively.
Short-Term Notes Payable
On December 21, 2016, the Company entered into promissory notes totaling $70,000 with five individuals. All notes bear 12% interest, mature on April 30, 2017, and are due in full with accrued interest at maturity. These notes are collateralized by receivables, cash accounts, inventories, and all related proceeds. The notes are immediately callable in the event of (a) an equity financing of at least $1 million, or (b) insolvency or bankruptcy proceedings. The loans were repaid in full during the period ended June 30, 2017. As of June 30, 2017 and December 31, 2016, principal of $0 and $70,000 and accrued interest of $0 and $237 were outstanding, all respectively.
| F-26 |
NOWRX, INC.
NOTES TO FINANCIAL STATEMENTS
As of June 30, 2017 (unaudited) and December 31, 2016 (audited) and for the six-month periods ended June 30, 2017 and 2016 (unaudited)
KISS Agreements
On December 19, 2016, the Company entered into a KISS agreement (Keep it Simple Security) with a third party for $50,000. The instrument matures in 24 months and bears 5% interest per annum. During the six-month period ended June 30, 2017, the Company issued additional KISS notes for total principal of $854,819, maturing after terms of 24 months, and bearing interest at 5% per annum. Of that amount, $40,000 were converted to KISS agreements from previously outstanding SAFE agreements, while the remaining $814,819 were cash investments.
If and upon a qualified financing where the Company sells preferred stock of $1,000,000 or greater, the instruments’ face value along with accrued interest will automatically convert to preferred stock at a discount of 20% to the pricing in the triggering round or the price per share implied by a pre-money valuation of $6,000,000 on the Company’s fully diluted capitalization. Such a conversion does include a “shadow series” provision, which limits the holder’s liquidation preference to the discounted purchase price.
If and upon a corporate transaction, the KISS becomes convertible into the number of shares determined by dividing the then outstanding principal and interest by the price per share implied by a pre-money valuation of $6,000,000 on the Company’s fully diluted capitalization, or is payable to the noteholder in the amount of two times the principal amount, at the noteholder’s election.
If at maturity neither of the aforementioned conversions have occurred, the noteholder may elect to convert the KISS and any unpaid accrued interest into the number of shares determined by dividing the then outstanding principal and interest by the price per share implied by a pre-money valuation of $6,000,000 on the Company’s fully diluted capitalization.
As of June 30, 2017 and December 31, 2016, the KISS agreements had not been converted and remained outstanding in the full principal amounts. The Company analyzed the note for beneficial conversion features, and concluded the conversion terms did not constitute beneficial conversion features.
Loan placement fees of $50,144 were incurred in connection with the issuance of these notes and were recorded as discounts to the notse. The Company is amortizing such fees to interest expense over a 24-month period, which is the Company’s estimate of when these notes will convert
As of June 30, 2017 and December 31, 2016, $904,819 and $50,000, of KISS notes were issued and outstanding, which are presented net of unamortized loan expenses of $43,876 and $0, for a carrying balance of $860,943 and $50,000, all respectively. Interest expense of $8,420 was recognized during the period ended June 30, 2017 on such notes, of which none was paid so the full amount remains on the balance sheet as accrued interest payable.
| F-27 |
NOWRX, INC.
NOTES TO FINANCIAL STATEMENTS
As of June 30, 2017 (unaudited) and December 31, 2016 (audited) and for the six-month periods ended June 30, 2017 and 2016 (unaudited)
SAFE Agreements
In 2016 and 2015, the Company issued simple agreements for future equity (SAFE Agreement) in exchange for cash investments of $622,500 and $445,000, respectively. During the six-month period ended June 30, 2017, $40,000 of such agreements were converted to KISS notes. The SAFE Agreements entitle the holder to convert the SAFE agreements into the Company’s preferred stock (this classification of stock has not yet been authorized or established). The terms provide for automatic conversion of the SAFE agreements’ purchase amounts of $1,067,500 as of both June 30, 2017 and December 31, 2016 (the “Purchase Amount”) into the Company’s preferred stock if and upon a qualified equity financing event, which is generally defined as a transaction or series of transactions involving the issuance of the Company’s preferred stock at a fixed pre-money valuation. The number of shares of preferred stock the SAFE agreement converts into is the Purchase Amount divided by the price per share determined by either: a) a $6,000,000 pre-money valuation on the Company’s then outstanding capitalization (as further defined in the agreements), or b) a 20% discount to the share pricing in the triggering equity financing. $87,500 of the 2016 issuances contain an additional provision providing a future purchase right to additional purchases of preferred stock at the same terms and pricing as the SAFE conversions, which results in a potential increase to the aggregate purchase amount of $47,500.
In the case of a liquidation event (as defined in the SAFE agreement), the SAFE agreement is convertible into either: A) cash of the Purchase Amount; B) the number of shares determined by dividing the Purchase Amount by the Company’s then outstanding capitalization (as defined in the agreement).
The SAFE agreements provide holders with various additional protections, including preferences over unitholders in a dissolution event for payment of the Purchase Amount and anti-dilution protections. If the SAFE agreement converts into the Company’s preferred stock, it will have all the same rights and privileges of the preferred stock from the triggering financing, except that the liquidation preference will be equal to the Purchase Amount.
Crowd Notes Payable
The Company conducted an offering of convertible securities (“Crowd Notes”) under Regulation CF during 2017, resulting in the issuance of $209,230 of Crowd Notes during the period ended June 30, 2017. The Crowd Notes bear interest at 5%, compounded quarterly, and do not have a stated maturity date. The Crowd Notes are convertible into the Company’s stock upon a future qualified equity financing of $1,000,000 or greater, or upon a corporate transaction (as defined in the note agreements). The conversion price on the notes is the lesser of a 20% discount or the price per share implied by a $6,000,000 pre-money valuation on the Company’s fully diluted capitalization.
Loan placement fees of $26,942 were incurred in connection with this offering and were recorded as discounts to the notes. The Company is amortizing such fees to interest expense over a 24-month period, which is the Company’s estimate of when these notes will convert.
As of June 30, 2017, $209,230 of Crowd Notes were issued and outstanding, which are presented net of unamortized loan expenses of $26,942, for a carrying balance of $184,288 as of June 30, 2017. Interest expense of $261 was recognized during the period ended June 30, 2017 on such notes, of which none was paid so the full amount remains on the balance sheet as accrued interest payable.
| F-28 |
NOWRX, INC.
NOTES TO FINANCIAL STATEMENTS
As of June 30, 2017 (unaudited) and December 31, 2016 (audited) and for the six-month periods ended June 30, 2017 and 2016 (unaudited)
NOTE 5: STOCKHOLDERS’ EQUITY (DEFICIT)
Common Stock
The Company has authorized 10,000,000 shares of $0.00001 par value common stock. As of each June 30, 2017 and December 31, 2016, 8,500,000 shares of common stock were issued and outstanding, respectively.
In June of 2015, the Company issued its two founders a total of 8,500,000 shares of common stock at $0.0001 per share, in exchange for $850 of cash and intellectual property. These stock issuances were conducted under terms of restricted stock purchase agreements and are subject to vesting terms contingent upon continuous service with the Company, which provide the Company the right to repurchase unvested shares at the original purchase price. These shares vest at 1/48 per month, commencing May 2015. As of June 30, 2017 and December 31, 2016, 4,427,083 and 3,364,583 of the shares had vested, respectively.
2015 Equity Incentive Plan
The Company adopted the2015 Equity Incentive Plan (the “Plan”), as amended and restated. The Plan permits the grant of stock options, stock appreciation rights and restricted stock to attract and retain employees and consultants. Under the Plan, the Company issues stock appreciation rights and options having a term of up to ten years and a strike price of no less than fair market value of common stock. Restricted stock is subject to vesting restrictions determined on a case-by-case basis. While shares may be restricted, the restricted stockholder retains voting rights for each share, regardless of restriction. Upon termination of employment or services, the Company may exercise its repurchase option over unvested equity interests.
The Company has reserved 1,500,000 shares of common stock under the Plan. As of June 30, 2017 and December 31, 2016, 1,053,000 and 1,053,000 shares of common stock were available for grant under the Plan, respectively. A summary of options activities for the periods ended June 30, 2017 and 2016 is as follows:
| June 30, 2017 | December 31, 2016 | |||||||||||||||
| Options | Weighted Average Exercise Price | Options | Weighted Average Exercise Price | |||||||||||||
| Outstanding - beginning of year | 447,000 | $ | 0.05 | 394,500 | $ | 0.05 | ||||||||||
| Granted | - | 52,500 | $ | 0.05 | ||||||||||||
| Exercised | - | - | ||||||||||||||
| Forfeited | - | - | ||||||||||||||
| Outstanding - end of year | 447,000 | $ | 0.05 | 447,000 | $ | 0.05 | ||||||||||
| Exercisable at end of year | 240,846 | $ | 0.05 | 168,486 | $ | 0.05 | ||||||||||
| Weighted average grant date fair value of options granted during year | N/A | $ | 0.17 | |||||||||||||
| Weighted average duration (years) to expiration of outstanding options at year-end | 8.5 | 9.0 | ||||||||||||||
| F-29 |
NOWRX, INC.
NOTES TO FINANCIAL STATEMENTS
As of June 30, 2017 (unaudited) and December 31, 2016 (audited) and for the six-month periods ended June 30, 2017 and 2016 (unaudited)
The Company measures employee stock-based awards at grant-date fair value and recognizes employee compensation expense on a straight-line basis over the vesting period of the award. Determining the appropriate fair value of stock-based awards requires the input of subjective assumptions, including the fair value of the Company’s common stock, and for stock options, the expected life of the option, and expected stock price volatility. The Company used the Black-Scholes option pricing model to value its stock option awards. The assumptions used in calculating the fair value of stock-based awards represent management’s best estimates and involve inherent uncertainties and the application of management’s judgment. As a result, if factors change and management uses different assumptions, stock-based compensation expense could be materially different for future awards.
The expected life of stock options was estimated using the “simplified method,” which is the midpoint between the vesting start date and the end of the contractual term, as the Company has limited historical information to develop reasonable expectations about future exercise patterns and employment duration for its stock options grants. The simplified method is based on the average of the vesting tranches and the contractual life of each grant. For stock price volatility, the Company uses comparable public companies as a basis for its expected volatility to calculate the fair value of options grants. The risk-free interest rate is based on U.S. Treasury notes with a term approximating the expected life of the option. The estimation of the number of stock awards that will ultimately vest requires judgment, and to the extent actual results or updated estimates differ from the Company’s current estimates, such amounts are recognized as an adjustment in the period in which estimates are revised.
The Company calculated its estimate of the value of the stock compensation granted for the periods ended June 30, 2017 and 2016 under FASB ASC 718, and concluded the value of these options was trivial and therefore did not record compensation costs related to the stock option grants.
Warrants
In conjunction with its offering and issuance of Crowd Notes payable (discussed in Note 4), in March of 2017, the Company entered into an agreement with its funding portal providing the funding portal with warrants granting the funding portal the right to purchase the Company’s stock at the same terms as the investors in the offering. The number of shares the funding portal may obtain under this agreement is 5% of the number of shares converted by the investors in the offering upon a conversion event under the terms of the agreement. This agreement expires after a seven-year term in March 2024. As there has not yet been a conversion event, the number of shares to which the funding portal is entitled under this agreement is not yet determinable.
NOTE 6: RELATED PARTY TRANSACTIONS
The Company’s co-founders and an employee extended financing of $65,000 and $25,000 in 2015 to the Company through the SAFE agreements discussed in Note 4. In 2016, that employee issued another $10,000 of SAFE financing.
NOTE 7: LEASE ARRANGEMENTS
In June 2015, the Company entered into a 3-year operating lease agreement for office space. The agreement called for a security deposit of $8,898 and monthly payments of $4,194 for the first year, with subsequent annual rent increases of 3% over the next two years. Additionally, the Company is responsible for 18% of the homeowners’ association dues, which currently totals $200-$300 per month.
| F-30 |
NOWRX, INC.
NOTES TO FINANCIAL STATEMENTS
As of June 30, 2017 (unaudited) and December 31, 2016 (audited) and for the six-month periods ended June 30, 2017 and 2016 (unaudited)
Future minimum payments under this lease are as follows:
| 2017 | $ | 26,114 | ||
| 2018 | 40,041 | |||
| Total future minimum lease payments | $ | 66,155 |
In addition to this lease, the Company reimburses the CEO for an apartment on a month-to-month basis. Rent is $1,525 per month and is available for reimbursement monthly.
For the periods ended June 30, 2017 and 2016, the Company recognized $27,707 and $26,301 of lease expense related to these leases, respectively.
NOTE 8: COMMITMENTS AND CONTINGENCIES
The Company may be subject to pending legal proceedings and regulatory actions in the ordinary course of business. The results of such proceedings cannot be predicted with certainty, but the Company does not anticipate that the final outcome, if any, arising out of any such matter will have a material adverse effect on its business, financial condition or results of operations.
NOTE 9: CONCENTRATIONS
The Company has significant concentrations in its accounts receivables, where approximately 80% and 81% of its accounts receivable balance as of June 30, 2017 and December 31, 2016, respectively, were held with one insurance payor. Should this insurance payor no longer be able to satisfy these statutory obligations to the Company, it would have a significant adverse effect to the Company.
Approximately 70% of the Company’s products are procured through a single supplier. Should this supplier no longer be willing or able to satisfy the Company’s product needs it could adversely affect the Company’s business.
NOTE 10: RECENT ACCOUNTING PRONOUNCEMENTS
In July 2014, the FASB issued the ASU No. 2015-11 on “Inventory (Topic 330): Simplifying the Measurement of Inventory”, which proposed that inventory should be measured at the lower of cost and the estimated selling prices in the ordinary course of business, less reasonably predictable costs of completion, disposal, and transportation. These amendments are based on existing guidance that requires measuring inventory at the lower of cost or market to consider the replacement cost of inventory less an approximately normal profit margin along with net value in determining the market value. It is effective for reporting periods beginning after December 15, 2016. Management has implemented this guidance to these financial statements.
Management does not believe that any recently issued, but not yet effective, accounting standards could have a material effect on the accompanying financial statements. As new accounting pronouncements are issued, the Company will adopt those that are applicable under the circumstances.
| F-31 |
NOWRX, INC.
NOTES TO FINANCIAL STATEMENTS
As of June 30, 2017 (unaudited) and December 31, 2016 (audited) and for the six-month periods ended June 30, 2017 and 2016 (unaudited)
NOTE 11: SUBSEQUENT EVENTS
Conversion of Warrants to Crowd Notes
The warrants outstanding as of June 30, 2017 discussed in Note 5 were subsequently rescinded in their entirety and exchanged for Crowd Notes with total principal of $39,412 under the same terms as the Crowd Notes discussed at Note 4.
Management’s Evaluation
Management has evaluated subsequent events through December 27, 2017, the date the financial statements were available to be issued. Based on this evaluation, no additional material events were identified which require adjustment or disclosure in these financial statements.
| F-32 |
PART III
INDEX TO EXHIBITS
| 1 | Issuer Agreement with SI Securities, LLC* |
| 2.1 | Amended Certificate of Incorporation* |
| 2.2 | Form of Amended and Restated Certificate of Incorporation** |
| 2.3 | Bylaws* |
| 4.1 | Form of subscription agreement** |
| 4.2 | Investor Proxy Agreement* |
| 8 | Escrow Agreement |
| 11 | Auditor’s Consent |
| 12 | Opinion of CrowdCheck Law LLP** |
| 13 | Testing the waters materials** |
* Previously filed
** To be filed by amendment
| 62 |
SIGNATURES
Pursuant to the requirements of Regulation A, the issuer certifies that it has reasonable grounds to believe that it meets all of the requirements for filing on Form 1-A and has duly caused this Offering Statement to be signed on its behalf by the undersigned, thereunto duly authorized, in the City of Mountain View, State of California, on February 20, 2018.
| NOWRX, INC. | ||
| By | /s/ Cary Breese | |
| Cary Breese, Chief Executive Officer | ||
| NowRx, Inc. | ||
The following persons in the capacities and on the dates indicated have signed this Offering Statement.
| /s/ Cary Breese | ||
| Cary Breese, Chief Executive Officer, Principal Financial Officer, Principal Accounting Officer, Director | ||
| Date: | February 20, 2018 | |
| /s/ Sumeet Sheokand | ||
| Sumeet Sheokand, Director | ||
| Date: | February 20, 2018 | |
| /s/ Barry Karlin | ||
| Barry Karlin, Director | ||
| Date: | February 20, 2018 | |
| 63 |