
C O P YR IG H T © A B C E LL E R A Q1 2024 BUSINESS UPDATE MAY 7, 2024

C O PY RI G H T © A B C EL LE RA DISCLAIMER 2 This presentation contains forward-looking statements, including statements made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. The forward-looking statements are based on management’s beliefs and assumptions and on information currently available to management. All statements contained in this presentation other than statements of historical fact are forward-looking statements, including statements regarding our ability to develop, commercialize and achieve market acceptance of our current and planned products and services, our research and development efforts, and other matters regarding our business strategies, use of capital, results of operations and financial position, and plans and objectives for future operations. In some cases, you can identify forward-looking statements by the words “may,” “will,” “could,” “would,” “should,” “expect,” “intend,” “plan,” “anticipate,” “believe,” “estimate,” “predict,” “project,” “potential,” “continue,” “ongoing” or the negative of these terms or other comparable terminology, although not all forward- looking statements contain these words. These statements involve risks, uncertainties and other factors that may cause actual results, levels of activity, performance, or achievements to be materially different from the information expressed or implied by these forward-looking statements. These risks, uncertainties and other factors are described under "Risk Factors," "Management's Discussion and Analysis of Financial Condition and Results of Operations" and elsewhere in the documents we file with the Securities and Exchange Commission from time to time. We caution you that forward-looking statements are based on a combination of facts and factors currently known by us and our projections of the future, about which we cannot be certain. As a result, the forward-looking statements may not prove to be accurate. The forward-looking statements in this presentation represent our views as of the date hereof. We undertake no obligation to update any forward-looking statements for any reason, except as required by law. Q 1 2 0 2 4 B U S IN ES S U PD AT E

C O PY RI G H T © A B C EL LE RA C O PY RI G H T © A B C EL LE RA 3 As we move programs toward the clinic, we are prioritizing: 2 0 2 4 P R I O R I T I E S Engine & Capabilities Strategic Partnerships Internal Programs & Pipeline 1 2 3 Q 1 2 0 2 4 B U S IN ES S U PD AT E

C O PY RI G H T © A B C EL LE RA C O PY RI G H T © A B C EL LE RA 4 New data demonstrates highly differentiated T-cell engager platform. O U R E N G I N E . P L A T F O R M D E V E L O P M E N T Q 1 2 0 2 4 B U S IN ES S U PD AT E Presented at AACR 2024
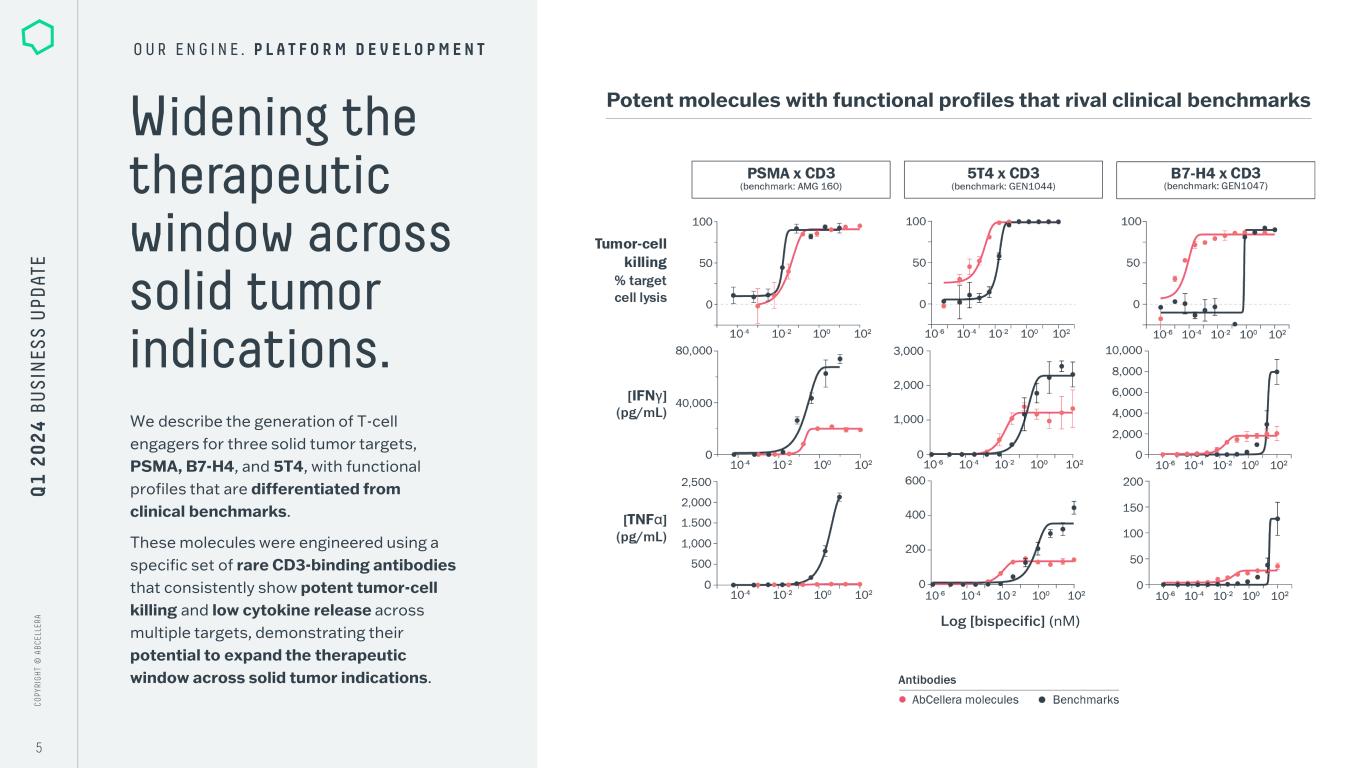
C O PY RI G H T © A B C EL LE RA C O PY RI G H T © A B C EL LE RA 5 Widening the therapeutic window across solid tumor indications. O U R E N G I N E . P L A T F O R M D E V E L O P M E N T We describe the generation of T-cell engagers for three solid tumor targets, PSMA, B7-H4, and 5T4, with functional profiles that are differentiated from clinical benchmarks. These molecules were engineered using a specific set of rare CD3-binding antibodies that consistently show potent tumor-cell killing and low cytokine release across multiple targets, demonstrating their potential to expand the therapeutic window across solid tumor indications. Potent molecules with functional profiles that rival clinical benchmarks Log [bispecific] (nM) Q 1 2 0 2 4 B U S IN ES S U PD AT E

C O PY RI G H T © A B C EL LE RA C O PY RI G H T © A B C EL LE RA 6 Q 1 2 0 2 4 B U S IN ES S U PD AT E We are using our T-cell engager platform as a source of internal programs and a basis for strategic partnerships. Our TCE platform is nearly complete. In addition to having highly differentiated proprietary CD3s, we have also developed panels of antibodies that can be used to enhance T-cell activation, survival via signal-2 costimulation, and have demonstrated the ability to target MHC peptides. O U R E N G I N E . P L A T F O R M D E V E L O P M E N T Our focus is to test in vivo and to advance a subset of programs.

C O PY RI G H T © A B C EL LE RA C O PY RI G H T © A B C EL LE RA 7 We advanced strategic partnerships with established biopharmas and top-tier investment groups. O U R P A R T N E R S 1 Program NEUROLOGY Multiple Programs IMMUNOLOGY ESTABLISHED BIOPHARMA COMPANY CREATION with Investment Groups Q 1 2 0 2 4 B U S IN ES S U PD AT E

C O P Y R IG H T © A B C E LL E R A Q1 2024 FINANCIALS UPDATE

C O PY RI G H T © A B C EL LE RA C O PY RI G H T © A B C EL LE RA 9 Q 1 2 0 2 4 B U S IN ES S U PD AT E Continued progress on key business metrics and a strong cash position. in total cash, cash equivalents, & marketable securities cumulative partner- initiated program starts with downstreams internal programs advanced into IND-enabling studies $725M 90 2 molecules in the clinic13 In available liquidity to execute on our strategy~$1B
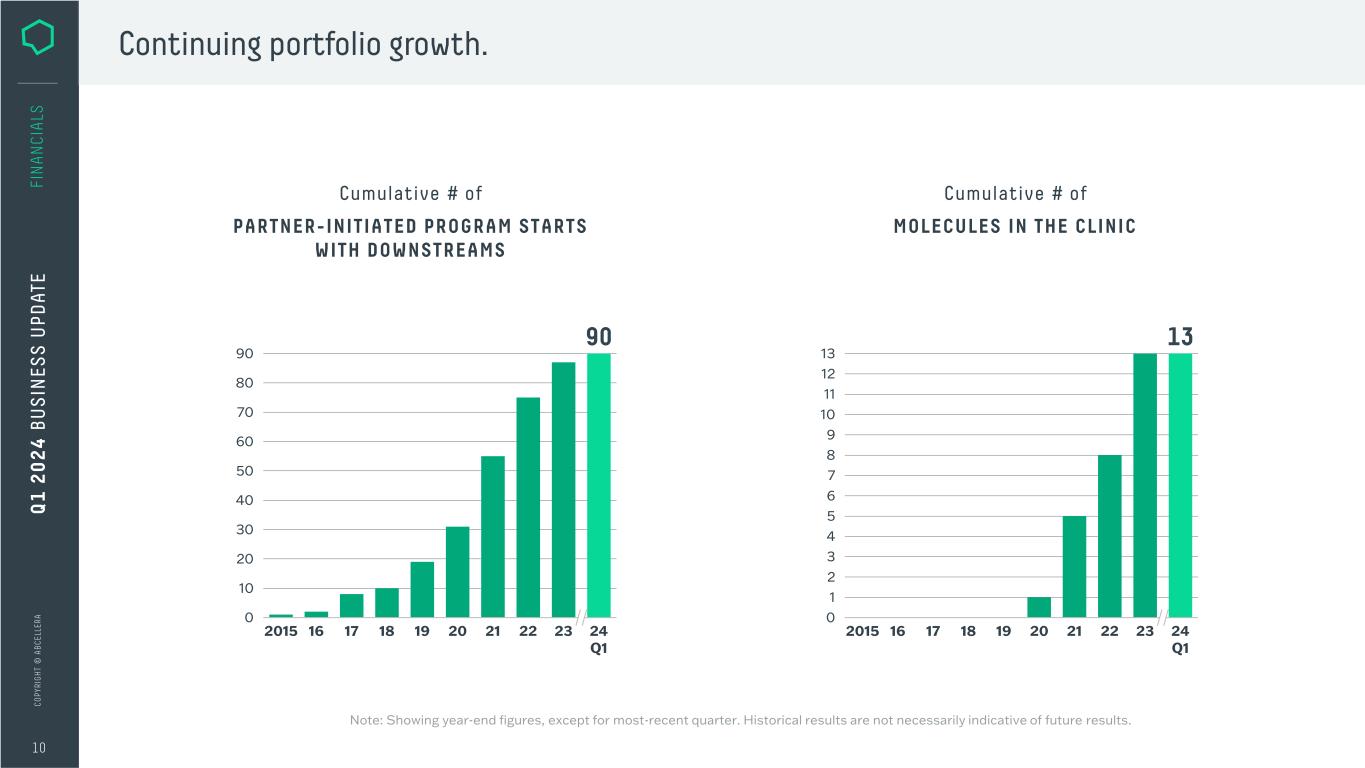
C O PY RI G H T © A B C EL LE RA PARTNER-INITIATED PROGRAM STARTS WITH DOWNSTREAMS Continuing portfolio growth. 10 FI N A N CI A LS Cumulative # of Q 1 2 0 2 4 B U S IN ES S U PD AT E 13 1 2 3 4 5 6 7 8 9 10 11 12 13 0 19 20 21 22 23 24 Q1 1817162015 MOLECULES IN THE CLINIC Cumulative # of 90 10 20 30 40 50 60 70 80 90 0 19 20 21 22 23 24 Q1 18162015 17
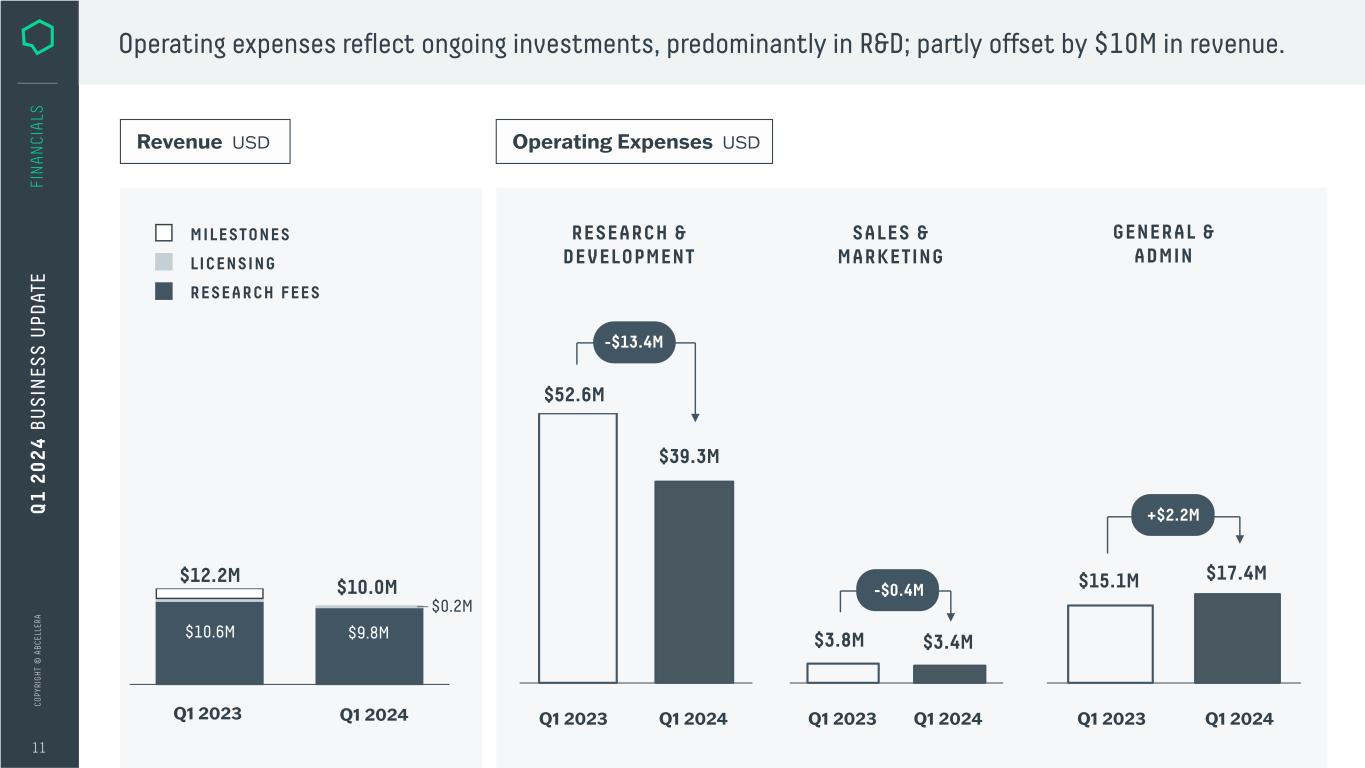
C O PY RI G H T © A B C EL LE RA 11 FI N A N CI A LS RESEARCH & DEVELOPMENT SALES & MARKETING GENERAL & ADMIN Operating Expenses USD Q1 2023 Q1 2024 $3.8M $3.4M Q1 2023 Q1 2024 $52.6M $39.3M -$13.4M Q 1 2 0 2 4 B U S IN ES S U PD AT E MILESTONES LICENSING RESEARCH FEES Revenue USD $0.2M $10.6M $9.8M Q1 2023 $12.2M $10.0M Q1 2024 Operating expenses reflect ongoing investments, predominantly in R&D; partly offset by $10M in revenue. -$0.4M Q1 2023 Q1 2024 +$2.2M $15.1M $17.4M
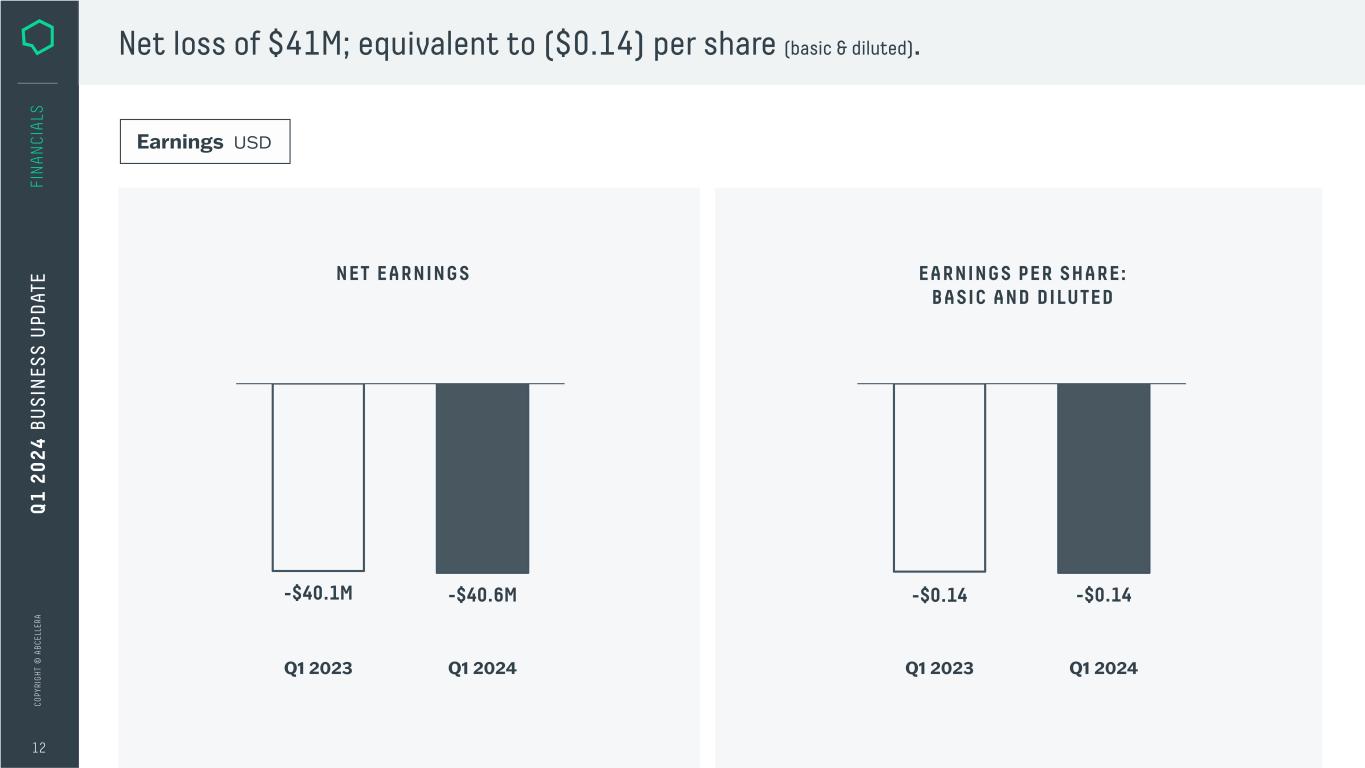
C O PY RI G H T © A B C EL LE RA 12 FI N A N CI A LS Net loss of $41M; equivalent to ($0.14) per share (basic & diluted). NET EARNINGS EARNINGS PER SHARE: BASIC AND DILUTED Q1 2023 Q1 2024 -$40.1M -$40.6M -$0.14 -$0.14 Q1 2023 Q1 2024 Q 1 2 0 2 4 B U S IN ES S U PD AT E Earnings USD
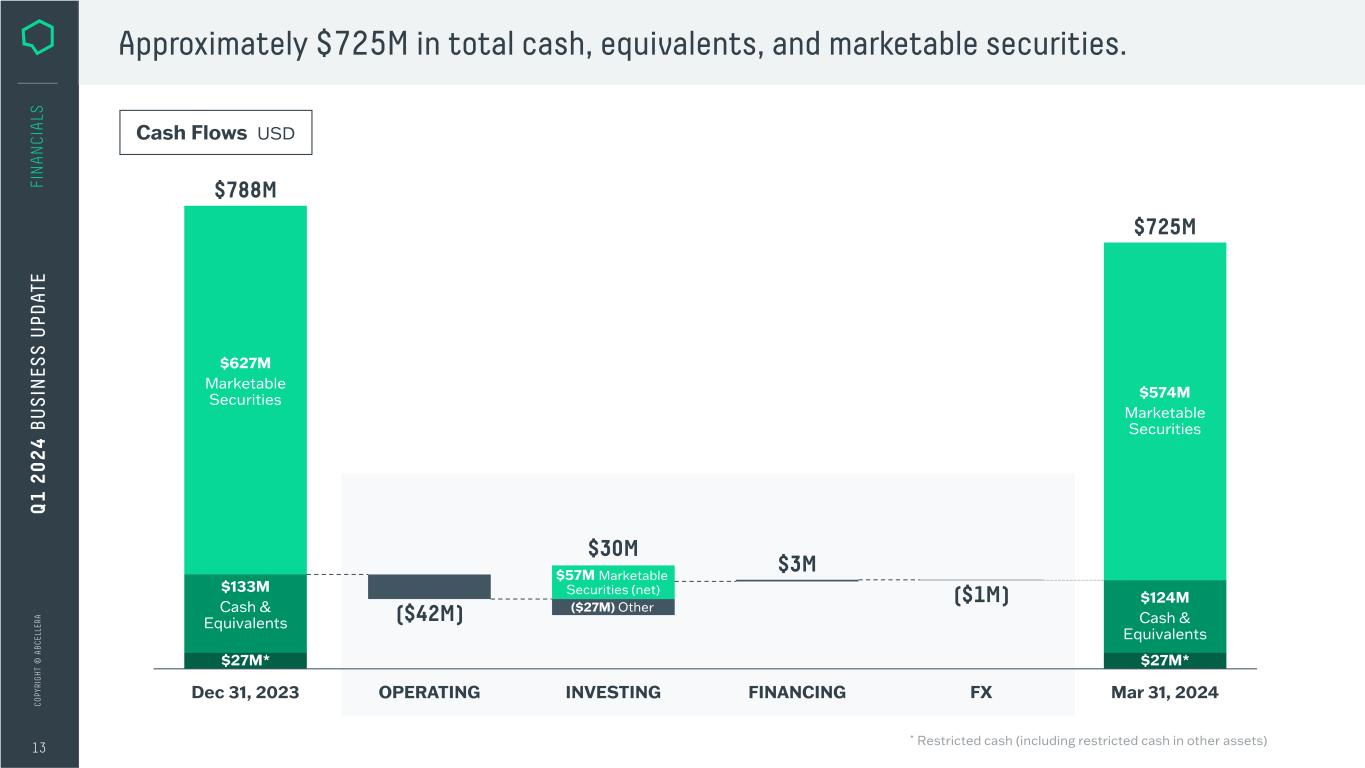
C O PY RI G H T © A B C EL LE RA 13 FI N A N CI A LS Approximately $725M in total cash, equivalents, and marketable securities. Cash Flows USD FINANCINGINVESTING FX $574M Marketable Securities $124M Cash & Equivalents $27M* $627M Marketable Securities ($27M) Other $57M Marketable Securities (net) OPERATINGDec 31, 2023 $27M* $788M ($42M) $30M $3M ($1M) $725M $133M Cash & Equivalents Mar 31, 2024 Q 1 2 0 2 4 B U S IN ES S U PD AT E

C O PY RI G H T © A B C EL LE RA 14 THANK YOU Q 1 2 0 2 4 B U S IN ES S U PD AT E