
C O P YR IG H T © A B C E LL E R A FULL YEAR 2023 BUSINESS UPDATE FEBRUARY 20, 2024

C O PY RI G H T © A B C EL LE RA DISCLAIMER 2 This presentation contains forward-looking statements, including statements made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. The forward-looking statements are based on management’s beliefs and assumptions and on information currently available to management. All statements contained in this presentation other than statements of historical fact are forward-looking statements, including statements regarding our ability to develop, commercialize and achieve market acceptance of our current and planned products and services, our research and development efforts, and other matters regarding our business strategies, use of capital, results of operations and financial position, and plans and objectives for future operations. In some cases, you can identify forward-looking statements by the words “may,” “will,” “could,” “would,” “should,” “expect,” “intend,” “plan,” “anticipate,” “believe,” “estimate,” “predict,” “project,” “potential,” “continue,” “ongoing” or the negative of these terms or other comparable terminology, although not all forward- looking statements contain these words. These statements involve risks, uncertainties and other factors that may cause actual results, levels of activity, performance, or achievements to be materially different from the information expressed or implied by these forward-looking statements. These risks, uncertainties and other factors are described under "Risk Factors," "Management's Discussion and Analysis of Financial Condition and Results of Operations" and elsewhere in the documents we file with the Securities and Exchange Commission from time to time. We caution you that forward-looking statements are based on a combination of facts and factors currently known by us and our projections of the future, about which we cannot be certain. As a result, the forward-looking statements may not prove to be accurate. The forward-looking statements in this presentation represent our views as of the date hereof. We undertake no obligation to update any forward-looking statements for any reason, except as required by law. FU LL Y EA R 2 0 2 3 B U S IN ES S U PD AT E

C O PY RI G H T © A B C EL LE RA C O PY RI G H T © A B C EL LE RA 3 FU LL Y EA R 2 0 2 3 B U S IN ES S U PD AT E We are evolving into a vertically integrated biotech. to include manufacturing, regulatory, and clinical capabilities including T-cell engagers, and GPCRs & ion channels by moving two programs into IND-enabling studies 2 0 2 3 H I G H L I G H T S Forward integration Unlock high-value drug programs Build partnered portfolio with high-quality programs & partnerships Advance internal pipeline Secured $220M in non-dilutive funding for our facilities, engine, and pipeline

C O PY RI G H T © A B C EL LE RA C O PY RI G H T © A B C EL LE RA 4 O U R E N G I N E . F O R W A R D I N T E G R A T I O N We are completing investments in clinical and manufacturing capabilities. After over a decade of building our platform, we are near completion. Beginning in 2024, we are shifting our capital allocation from building capabilities to using them to advance our pipeline and partnerships. 2024 Late 2025 Manufacturing OnlineMove to New Headquarters FU LL Y EA R 2 0 2 3 B U S IN ES S U PD AT E

C O PY RI G H T © A B C EL LE RA C O PY RI G H T © A B C EL LE RA 5 O U R E N G I N E . P L A T F O R M D E V E L O P M E N T • Hundreds of diverse CD3-binding antibodies to combine to create optimized precision cancer therapies • Clinically-validated OrthoMab bispecifics platform, to pair CD3-targeting and tumor-targeting antibodies • Internal validation of panel through work on three well-recognized and high-value targets T-Cell Engager Platform We are unlocking high-value drug targets. GPCR & Ion Channel Platform • Clinically validated, membrane-protein targets with large commercial potential that have proven largely intractable using traditional methods for antibody discovery • Internal platform integrated across the best of modern data science and biotechnology to generate first-in-class and best-in- class candidates FU LL Y EA R 2 0 2 3 B U S IN ES S U PD AT E

C O PY RI G H T © A B C EL LE RA C O PY RI G H T © A B C EL LE RA 6 Molecule Target Indication Therapeutic Area Potential Differentiation ABCL635 undisclosed GPCR or ion channel undisclosed metabolic & endocrine conditions first in class ABCL575 OX40L atopic dermatitis immunology & inflammation best in class We are advancing an internal pipeline of potential first-in- class and best-in-class assets as a top priority to drive growth. • First two IND submissions anticipated in 2025 • We intend to take additional first-in-class development candidates into IND-enabling studies in 2024 and 2025 • $220M in non-dilutive funding from the governments of Canada and British Columbia O U R P I P E L I N E FU LL Y EA R 2 0 2 3 B U S IN ES S U PD AT E

C O PY RI G H T © A B C EL LE RA C O PY RI G H T © A B C EL LE RA 7 Molecule ABCL635 Status IND-enabling studies (Preclinical) Target Undisclosed - GPCR or ion channel MOA Antagonist Indication Undisclosed Therapeutic Area Metabolic & endocrine conditions Market Opportunity >$2B Potential Differentiation First-in-class Summary Program details and indication will only be disclosed once the molecule reaches the clinic. A potential first-in-class antibody therapy for metabolic and endocrine conditions. ABCL635 is the first AbCellera-led asset derived from our GPCR and ion channel platform. FU LL Y EA R 2 0 2 3 B U S IN ES S U PD AT E O U R P I P E L I N E . A B C L 6 3 5

C O PY RI G H T © A B C EL LE RA C O PY RI G H T © A B C EL LE RA 8 O U R P I P E L I N E . A B C L 5 7 5 Molecule ABCL575 Status IND-enabling studies (Preclinical) Target OX40 ligand (OX40L) MOA Blocking and non-depleting Indication • Atopic dermatitis (AD) • Other indications in autoimmunity and inflammation Therapeutic Area Immunology & inflammation Market Opportunity $17B by 2032* Potential Differentiation ABCL575 has been designed with potency, pharmacokinetics, and developability to enable less frequent dosing, which provides a potential for differentiation. Summary Targeting OX40L has the potential to address a broad range of inflammatory conditions and autoimmune diseases, some of which include colitis/inflammatory bowel disease (IBD), and asthma/atopy1. OX40L blocking is currently under investigation for atopic dermatitis2, asthma2, alopecia areata3, hidradenitis suppurativa (HS)4. *Source: IQVIA ABCL575 is a potential best-in-class and second-in-class antibody therapy for the treatment of atopic dermatitis. 1. Croft M, et al. (2009) Immunol Rev. 229(1):173-91. 2. https://clinicaltrials.gov/search?intr=Amlitelimab 3. https://clinicaltrials.gov/search?intr=IMG-007 4. https://clinicaltrials.gov/search?intr=%20SAR-442970 FU LL Y EA R 2 0 2 3 B U S IN ES S U PD AT E

C O PY RI G H T © A B C EL LE RA C O PY RI G H T © A B C EL LE RA 9 We are increasingly focused on strategic partnerships. We look for partnerships that bring new insights into novel biology or access to unique capabilities to create opportunities in therapeutic areas of shared interest. Expanding relationships with large pharma and large biotech T-cell engager platform deals in oncology and autoimmune conditions Co-development to access new targets and technology AbbVie Company creation with top-tier venture capital groups FU LL Y EA R 2 0 2 3 B U S IN ES S U PD AT E O U R P A R T N E R S

C O PY RI G H T © A B C EL LE RA C O PY RI G H T © A B C EL LE RA 10 We use our capabilities to advance programs and partnerships. Expand high value strategic partnerships, leveraging T-cell engager platform. 2 0 2 4 P R I O R I T I E S Invest in the final stage of our engine, including our manufacturing facility. Engine & capabilities Partnered programs Elect additional first-in-class development candidates from GPCR / ion channel platform and advance ABCL635 and ABCL575 through IND-enabling studies. Internal programs & pipeline Capital Allocation: ~40% ~20% ~40% FU LL Y EA R 2 0 2 3 B U S IN ES S U PD AT E

C O P Y R IG H T © A B C E LL E R A FULL YEAR 2023 FINANCIALS UPDATE
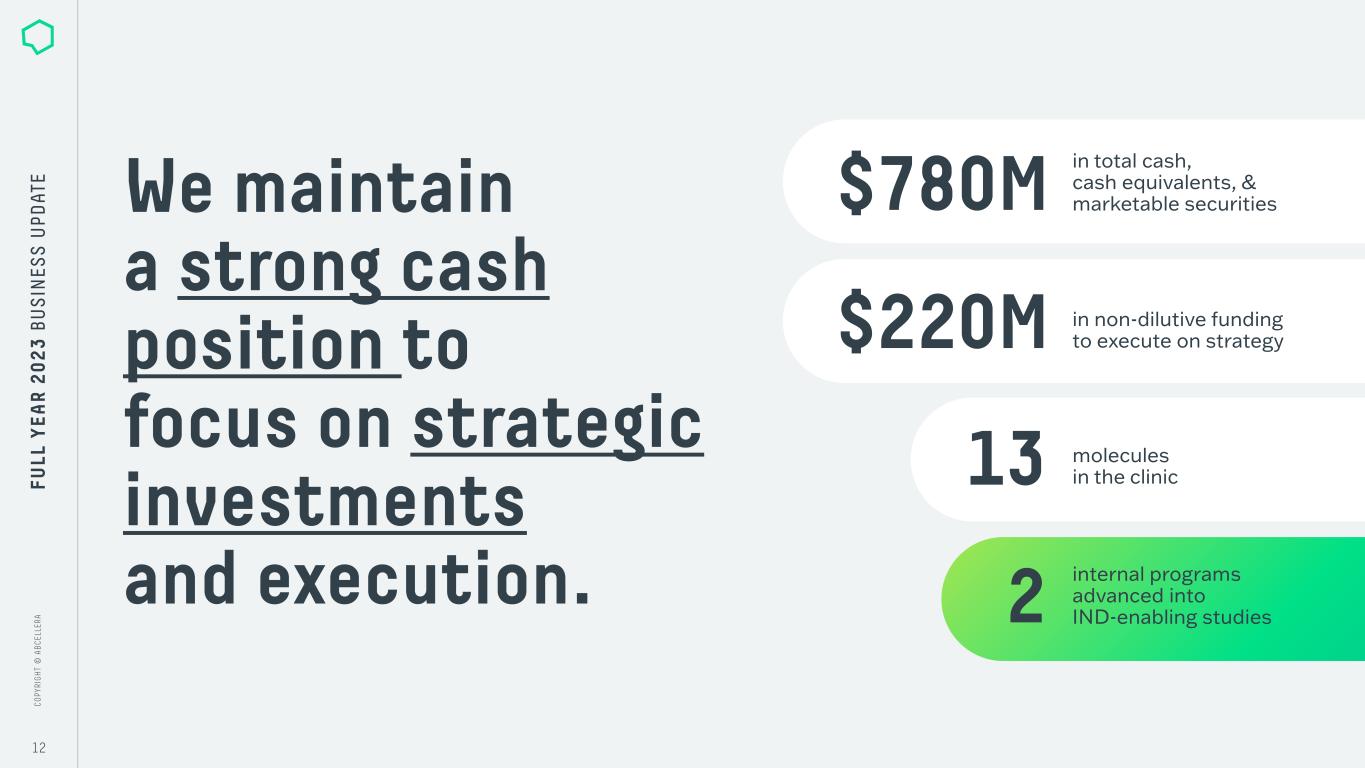
C O PY RI G H T © A B C EL LE RA C O PY RI G H T © A B C EL LE RA 12 FU LL Y EA R 2 0 2 3 B U S IN ES S U PD AT E We maintain a strong cash position to focus on strategic investments and execution. in non-dilutive funding to execute on strategy in total cash, cash equivalents, & marketable securities molecules in the clinic internal programs advanced into IND-enabling studies $780M 13 $220M 2
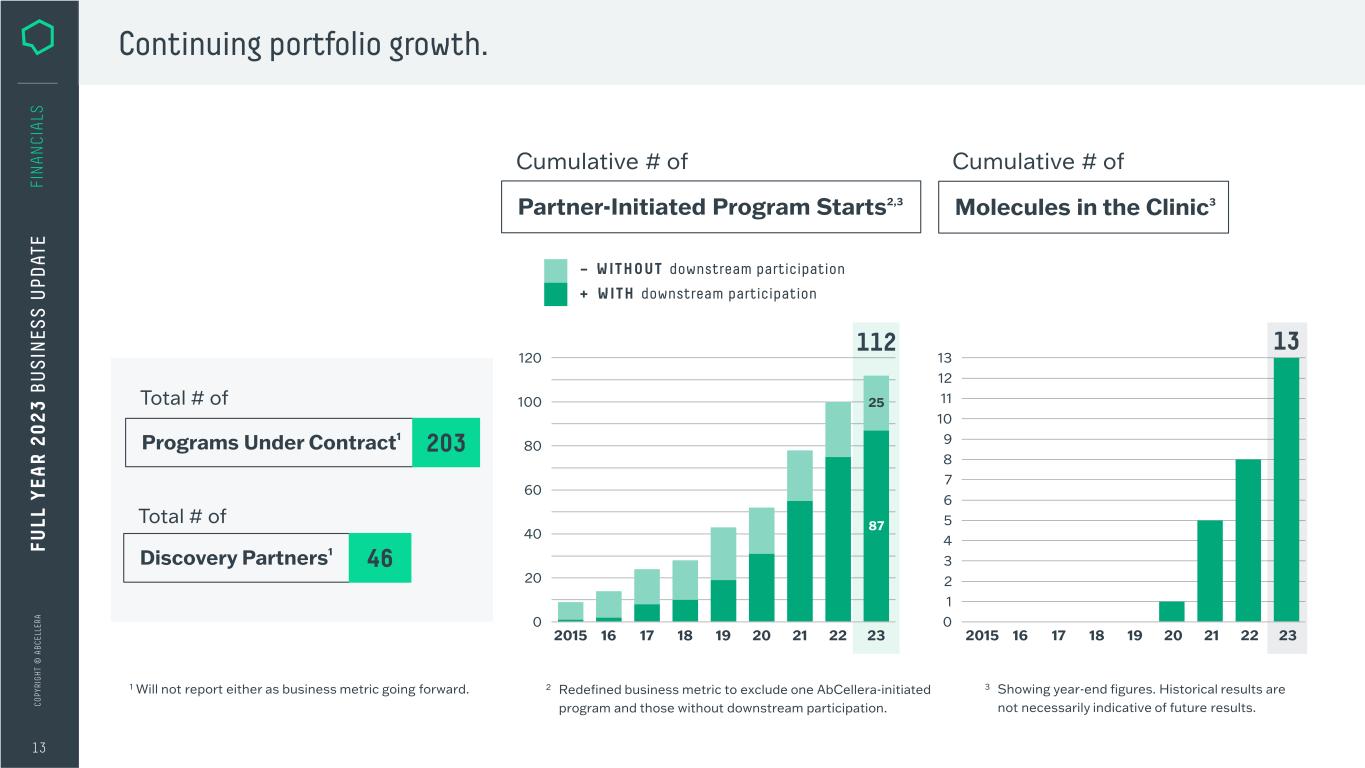
C O PY RI G H T © A B C EL LE RA 87 25 20 40 60 80 100 0 120 16 17 18 19 20 21 22 23 112 2015 Continuing portfolio growth. 13 FI N A N CI A LS 3 Showing year-end figures. Historical results are not necessarily indicative of future results. Partner-Initiated Program Starts2,3 Cumulative # of + WITH downstream participation – WITHOUT downstream participation Molecules in the Clinic3 Cumulative # of Discovery Partners1 Programs Under Contract1 203 Total # of Total # of 46 13 1 2 3 4 5 6 7 8 9 10 13 0 12 11 19 20 21 22 2318172015 16 FU LL Y EA R 2 0 2 3 B U S IN ES S U PD AT E 1 Will not report either as business metric going forward. 2 Redefined business metric to exclude one AbCellera-initiated program and those without downstream participation.
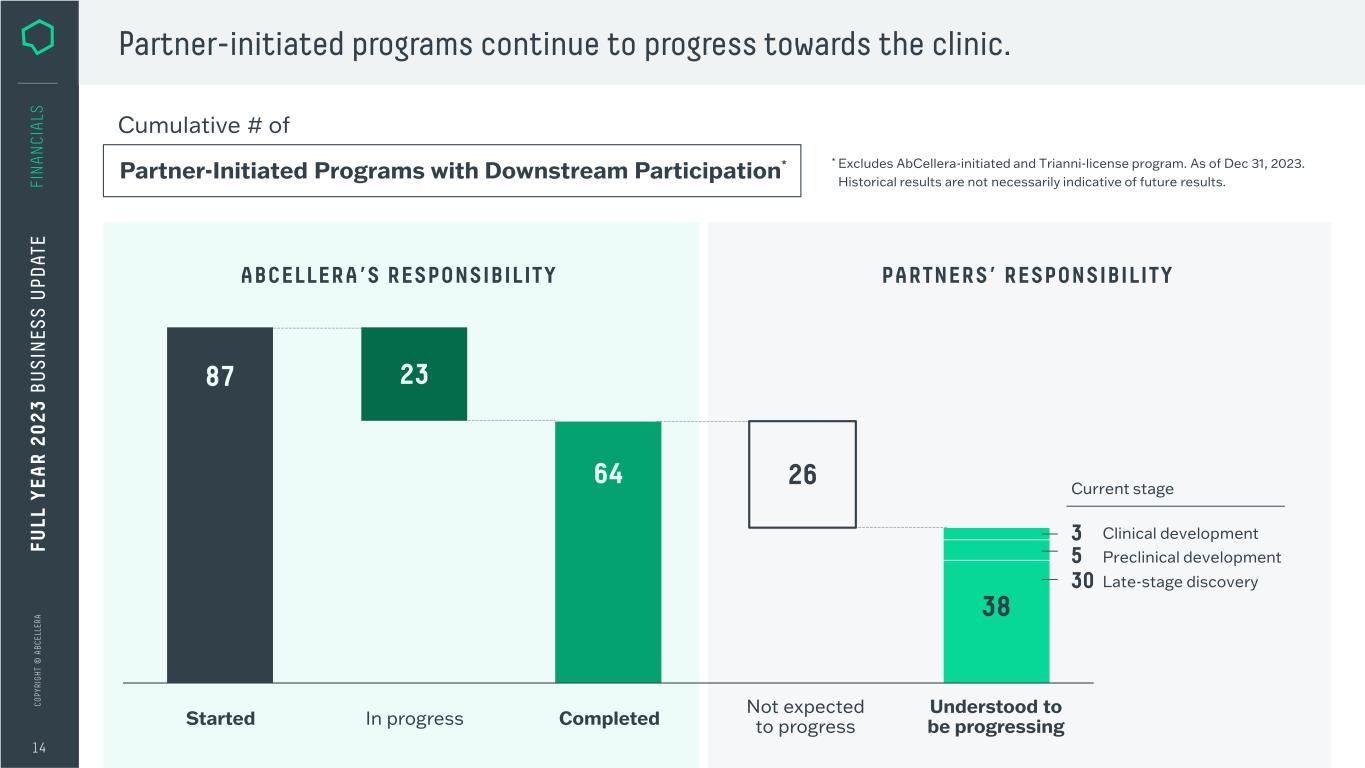
C O PY RI G H T © A B C EL LE RA Partner-initiated programs continue to progress towards the clinic. 14 FI N A N CI A LS FU LL Y EA R 2 0 2 3 B U S IN ES S U PD AT E Partner-Initiated Programs with Downstream Participation* Cumulative # of 87 64 23 26 Understood to be progressingStarted Not expected to progressCompletedIn progress 38 Late-stage discovery Clinical development Preclinical development Current stage * Excludes AbCellera-initiated and Trianni-license program. As of Dec 31, 2023. Historical results are not necessarily indicative of future results. 3 5 30 ABCELLERA’S RESPONSIBILITY PARTNERS’ RESPONSIBILITY
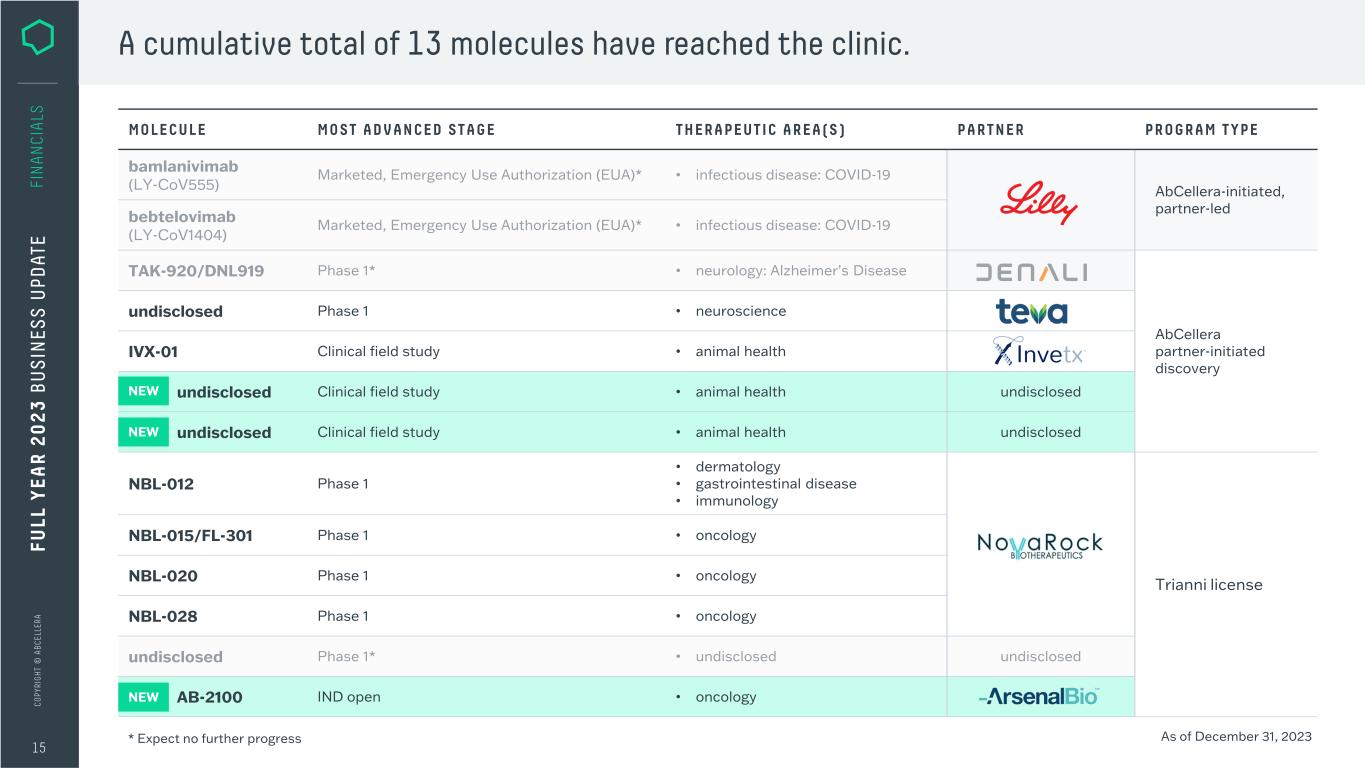
C O PY RI G H T © A B C EL LE RA MOLECULE MOST ADVANCED STAGE THERAPEUTIC AREA(S) PARTNER PROGRAM TYPE AbCellera-initiated, partner-led AbCellera partner-initiated discovery undisclosed Phase 1 • neuroscience IVX-01 Clinical field study • animal health undisclosed Clinical field study • animal health undisclosed undisclosed Clinical field study • animal health undisclosed NBL-012 Phase 1 • dermatology • gastrointestinal disease • immunology Trianni license NBL-015/FL-301 Phase 1 • oncology NBL-020 Phase 1 • oncology NBL-028 Phase 1 • oncology AB-2100 IND open • oncology 15 FU LL Y EA R 2 0 2 3 B U S IN ES S U PD AT E FI N A N CI A LS A cumulative total of 13 molecules have reached the clinic. As of December 31, 2023* Expect no further progress NEW NEW NEW
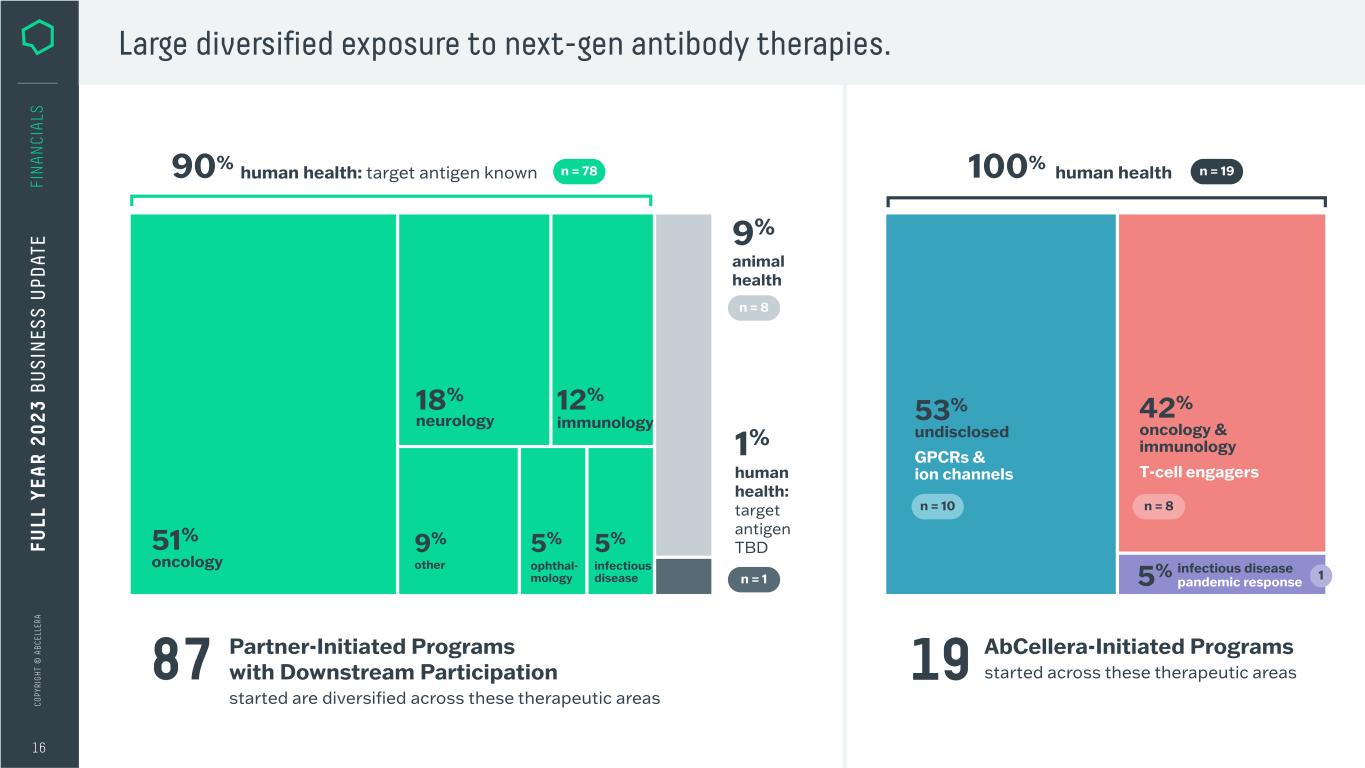
C O PY RI G H T © A B C EL LE RA Partner-Initiated Programs with Downstream Participation started are diversified across these therapeutic areas 16 Large diversified exposure to next-gen antibody therapies. ophthal- mology 5% other 9%51% oncology 18% neurology 87 AbCellera-Initiated Programs started across these therapeutic areas19 infectious disease 5% 12% immunology 42% oncology & immunology T-cell engagers 53% undisclosed GPCRs & ion channels 5% infectious disease pandemic response FU LL Y EA R 2 0 2 3 B U S IN ES S U PD AT E FI N A N CI A LS 1 100% human health n = 1990% human health: target antigen known n = 78 9% animal health n = 8 1% human health: target antigen TBD n = 1 n = 10 n = 8
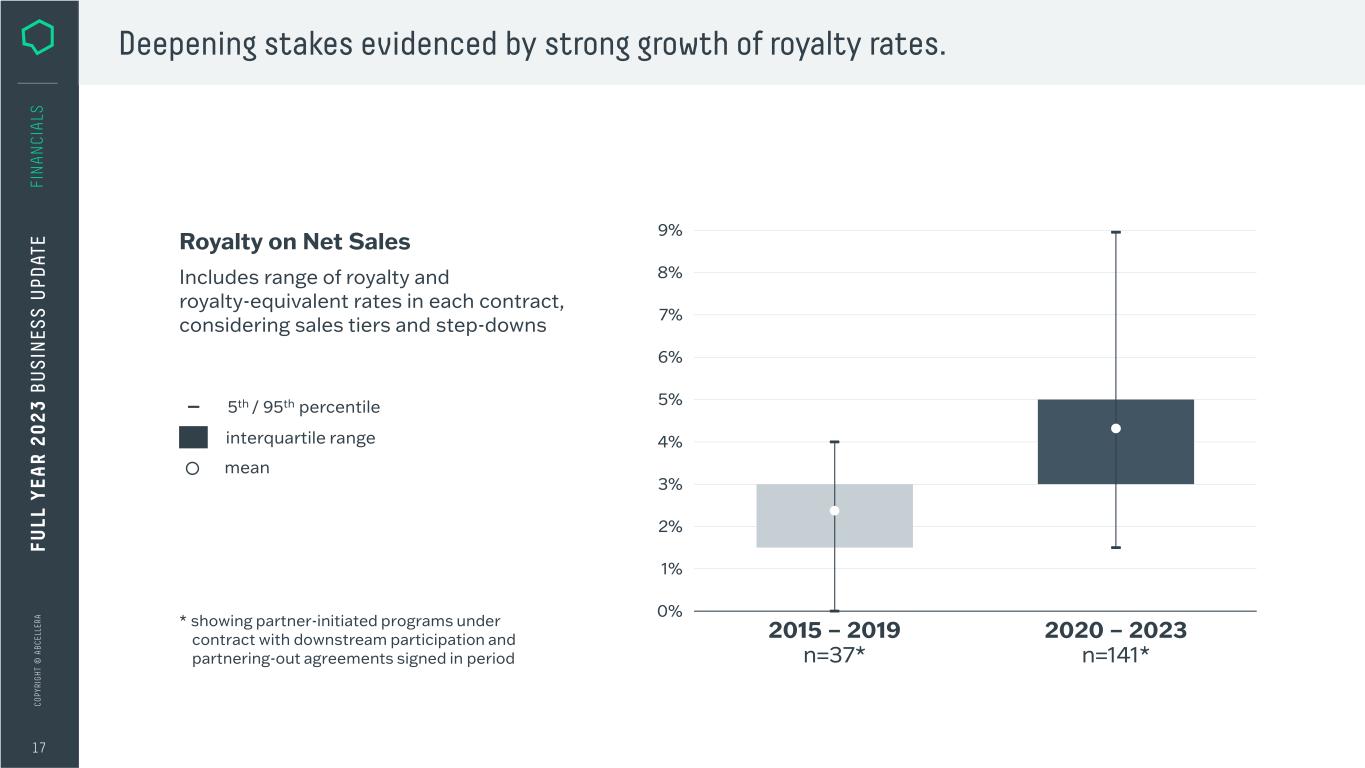
C O PY RI G H T © A B C EL LE RA 17 FI N A N CI A LS Deepening stakes evidenced by strong growth of royalty rates. * showing partner-initiated programs under contract with downstream participation and partnering-out agreements signed in period Includes range of royalty and royalty-equivalent rates in each contract, considering sales tiers and step-downs Royalty on Net Sales mean interquartile range 5th / 95th percentile FU LL Y EA R 2 0 2 3 B U S IN ES S U PD AT E 5% 0% 4% 7% 3% 8% 2% 9% 1% 6% 2020 – 2023 n=141* 2015 – 2019 n=37*
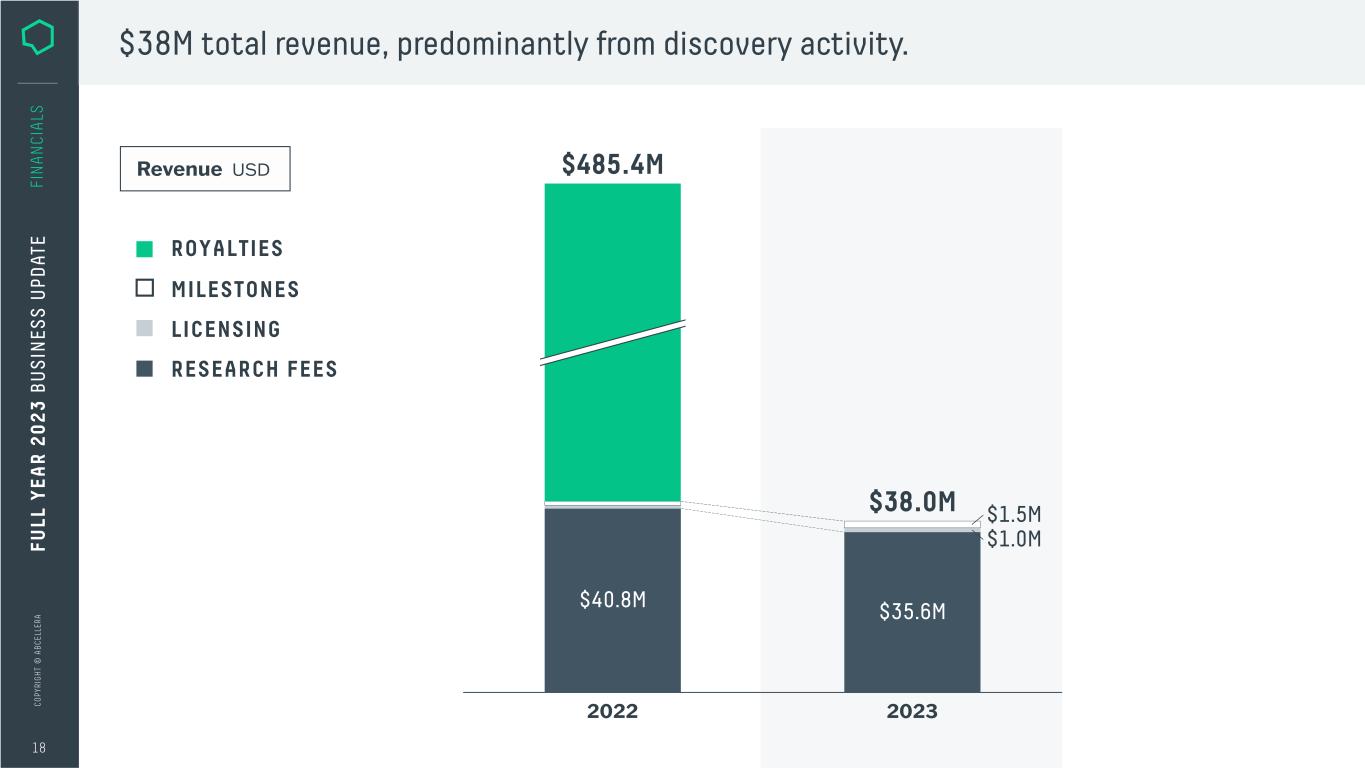
C O PY RI G H T © A B C EL LE RA 18 FI N A N CI A LS $38M total revenue, predominantly from discovery activity. MILESTONES ROYALTIES LICENSING RESEARCH FEES Revenue USD $35.6M 2022 $485.4M $38.0M 2023 $1.5M $1.0M $40.8M FU LL Y EA R 2 0 2 3 B U S IN ES S U PD AT E
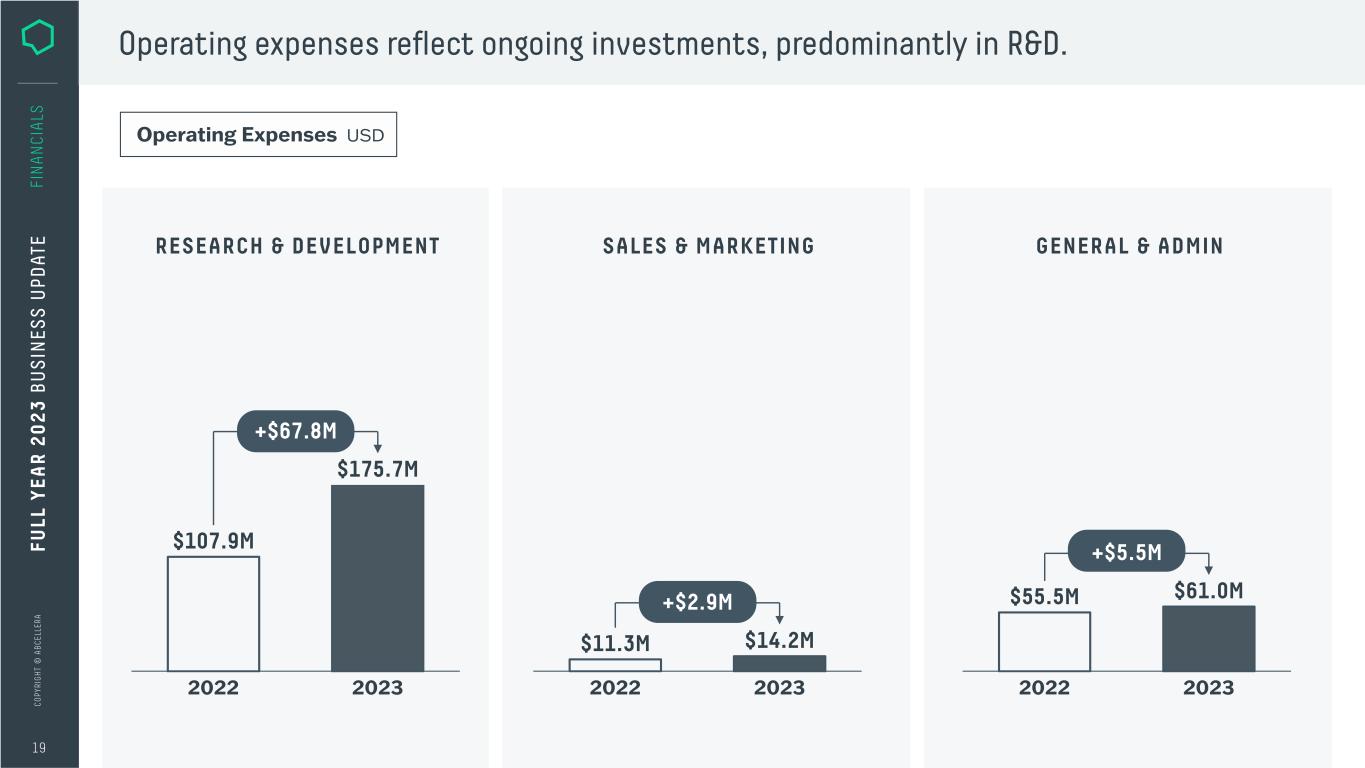
C O PY RI G H T © A B C EL LE RA 19 FI N A N CI A LS Operating expenses reflect ongoing investments, predominantly in R&D. RESEARCH & DEVELOPMENT SALES & MARKETING GENERAL & ADMIN Operating Expenses USD 20232022 $11.3M $14.2M +$2.9M 20232022 $55.5M $61.0M +$5.5M 20232022 $107.9M $175.7M +$67.8M FU LL Y EA R 2 0 2 3 B U S IN ES S U PD AT E
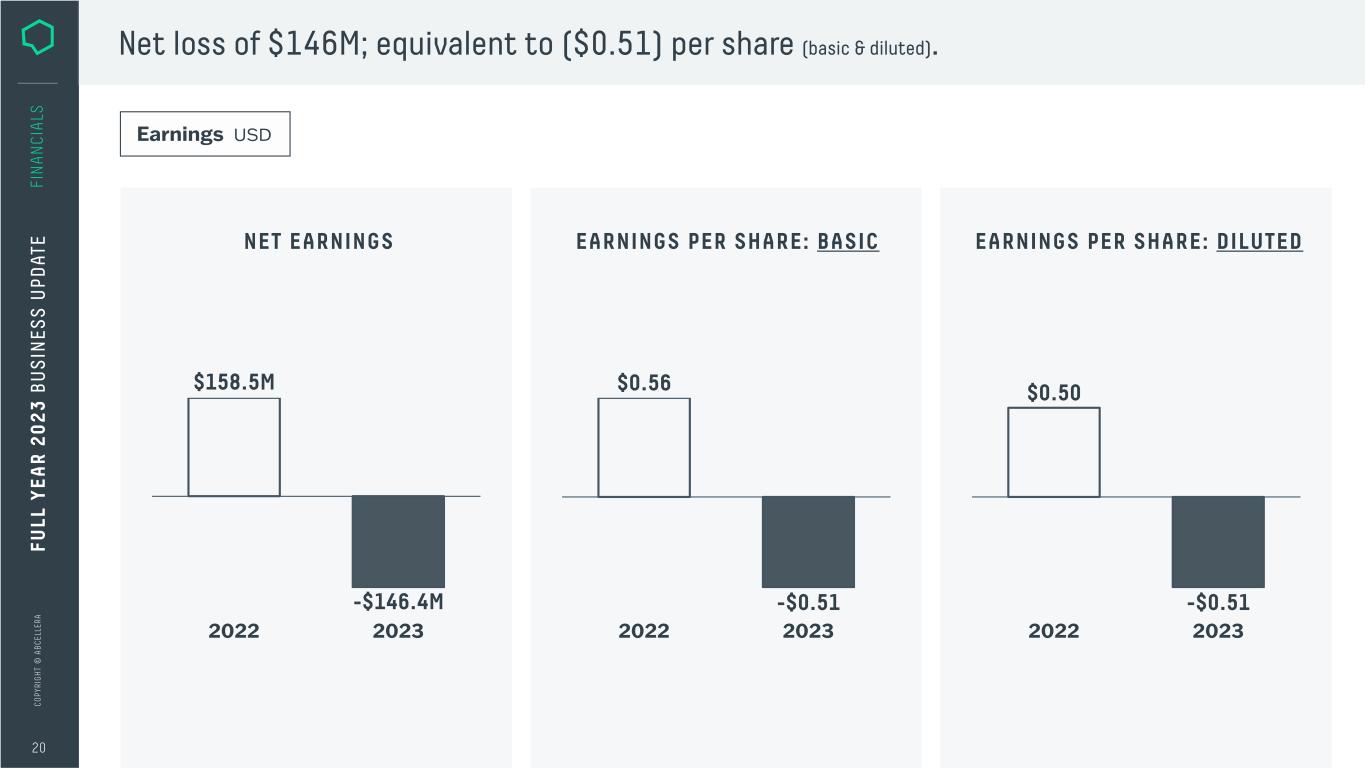
C O PY RI G H T © A B C EL LE RA 20 FI N A N CI A LS Net loss of $146M; equivalent to ($0.51) per share (basic & diluted). Earnings USD NET EARNINGS EARNINGS PER SHARE: BASIC 2022 2023 $158.5M -$146.4M $0.56 -$0.51 2022 2023 EARNINGS PER SHARE: DILUTED $0.50 -$0.51 2022 2023 FU LL Y EA R 2 0 2 3 B U S IN ES S U PD AT E
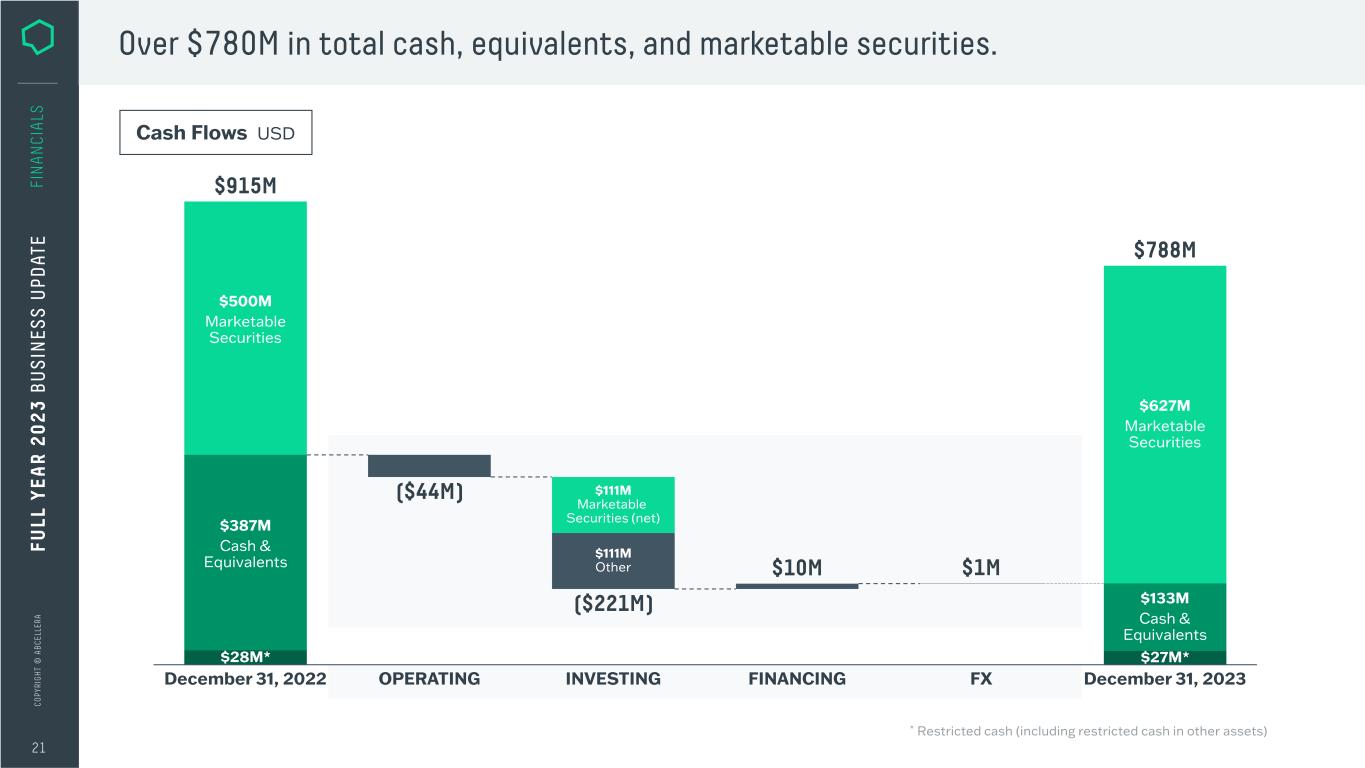
C O PY RI G H T © A B C EL LE RA 21 FI N A N CI A LS Over $780M in total cash, equivalents, and marketable securities. Cash Flows USD INVESTING FINANCING $111M Other FX $627M Marketable Securities $133M Cash & Equivalents $500M Marketable Securities December 31, 2023 $111M Marketable Securities (net) OPERATINGDecember 31, 2022 $28M* $387M Cash & Equivalents $915M ($44M) ($221M) $10M $1M $788M $27M* FU LL Y EA R 2 0 2 3 B U S IN ES S U PD AT E

C O PY RI G H T © A B C EL LE RA 22 THANK YOU FU LL Y EA R 2 0 2 3 B U S IN ES S U PD AT E