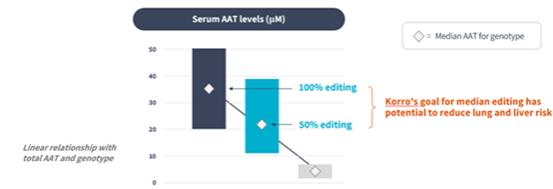
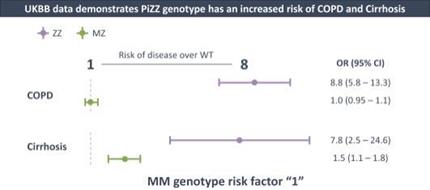
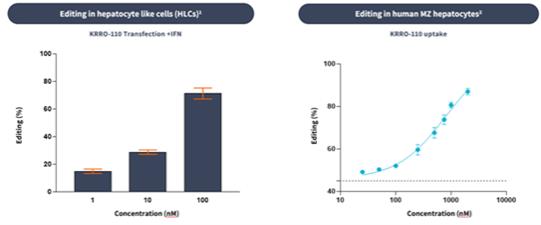
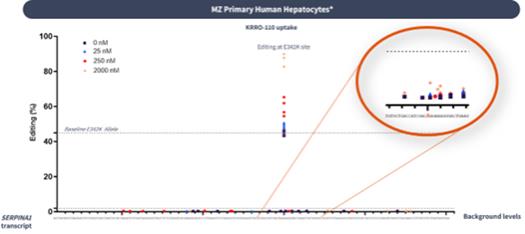
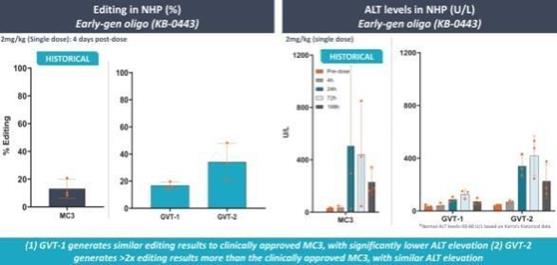
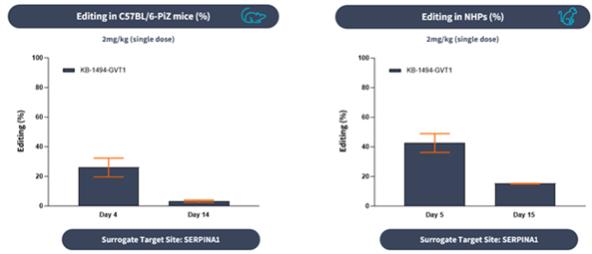
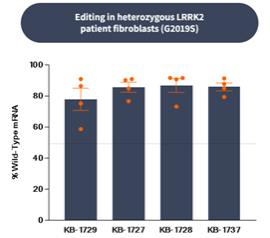
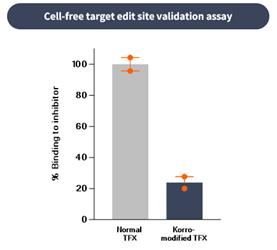
if our competitors develop and commercialize products that are safer, more effective, particularly if they represent cures, have fewer or less severe side effects, are more convenient, or are less expensive than any products that we may develop. Our competitors also may obtain FDA or other regulatory approval for their products more rapidly than we may obtain approval for ours, which could result in our competitors establishing a strong market position before we are able to enter the market. The key competitive factors affecting the success of all of our programs are likely to be their efficacy, safety, convenience, and availability of reimbursement.
Intellectual Property
Overview
We strive to protect the proprietary technology that we believe is important to our business, including seeking and maintaining patent protection in the United States and internationally for our current and future product candidates. We also rely on trademarks, copyrights, trade secrets, confidentiality procedures, employee disclosure, invention assignment agreements, know-how, continuing technological innovation and in-licensing opportunities to develop and maintain our proprietary position.
We seek to obtain domestic and international patent protection, and endeavors to promptly file patent applications for new commercially valuable inventions. We also rely on trade secrets to protect aspects of our business that are not amenable to, or that we do not consider appropriate for, patent protection.
We plan to continue to expand our intellectual property estate by filing patent applications directed to platform technologies and improvements thereof, pharmaceutical compositions, methods of treatment, methods of manufacture or identified from our ongoing development of our product candidates. Our success will depend on our ability to obtain and maintain patent and other proprietary protection for commercially important technology, inventions and know-how related to our business, defend and enforce any patents that we may obtain, preserve the confidentiality of our trade secrets and operate without infringing the valid and enforceable patents and proprietary rights of third parties.
The patent positions of companies like us are generally uncertain and involve complex legal, scientific and factual questions. In addition, the coverage claimed in a patent may be challenged in courts after issuance. Moreover, many jurisdictions permit third parties to challenge issued patents in administrative proceedings, which may result in further narrowing or even cancellation of patent claims. We cannot guarantee that our pending patent applications, or any patent applications that we may in the future file or license from third parties, will result in the issuance of patents. We cannot predict whether the patent applications we are currently pursuing will issue as patents in any particular jurisdiction or at all, whether the claims of any patent applications, should they issue, will cover our product candidates, or whether the claims of any issued patents will provide sufficient protection from competitors or otherwise provide any competitive advantage. We cannot predict the scope of claims that may be allowed or enforced in our patents. In addition, the coverage claimed in a patent application can be significantly reduced before the patent is issued, and its scope can be reinterpreted after issuance. Consequently, we may not obtain or maintain adequate patent protection for any of our product candidates.
Because patent applications in the United States and certain other jurisdictions are maintained in secrecy for 18 months or potentially even longer, and because publication of discoveries in the scientific or patent literature often lags behind actual discoveries and patent application filings, we cannot be certain of the priority of inventions covered by pending patent applications. Accordingly, we may not have been the first to invent the subject matter disclosed in some of our patent applications or the first to file patent applications covering such subject matter, and we may have to participate in interference proceedings or derivation proceedings declared by the USPTO to determine priority of invention. For more information regarding the risks related to our intellectual property, see “Risk Factors—Risks Related to Our Business—Risks Related to Intellectual Property.”
Patent Portfolio
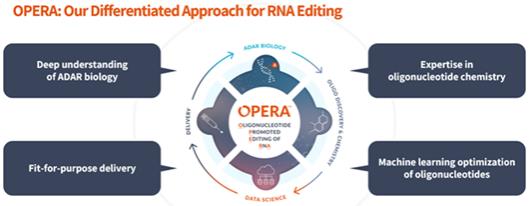
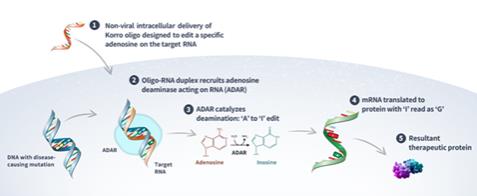
We strive to protect our proprietary RNA editing platform OPERA and related technologies and our product candidates, including seeking and maintaining patent protection intended to cover various target-specific editing strategies, the composition of matter of our product candidates, their methods of use, related delivery technologies, and other inventions. The intellectual property that is available to us is critical to our business and we strive to protect it, including by obtaining, maintaining, defending, and enforcing patent protection in the United States and internationally. As of December 31, 2023, our patent portfolio in total consisted of 32 patent families, with two U.S. patents and one patent in foreign jurisdictions (e.g., Canada), including five pending Patent Cooperation Treaty, or PCT, applications, various pending non-provisional applications world-wide (e.g., United States, Australia, Canada, China, Europe, South Korea, and Japan), and nine families with pending provisional patent applications.
We have a patent portfolio that relates to our RNA editing platform OPERA, as well as numerous disease programs listed below, and includes 13 patent families. These families are directed to various oligonucleotide formats, nucleotide compositions, oligonucleotide chemistries, modifications, specific linkage chemistries, oligonucleotides having a specific structures, methods of deaminating an adenosine using such oligonucleotides, methods of oligonucleotide delivery, and methods of treating disease by administering such oligonucleotides. The first patent family is pending in Australia, Canada, China, Europe, Japan, South Korea, Taiwan and the United States, and includes a U.S. patent. The first three patent families are pending in Australia, Canada, China,
96