Exhibit 99.1

ANNUAL INFORMATION FORM
OF
IMMUNOPRECISE ANTIBODIES LTD.
July 28, 2022
TABLE OF CONTENTS
| 1 | ||||
| 3 | ||||
| 3 | ||||
| 10 | ||||
| 16 | ||||
| 30 | ||||
| 31 | ||||
| 31 | ||||
| 32 | ||||
| 32 | ||||
| 32 | ||||
| 38 | ||||
| 38 | ||||
| 40 | ||||
| 40 | ||||
| 40 | ||||
| 40 | ||||
| 41 | ||||
IMMUNOPRECISE ANTIBODIES LTD.
ANNUAL INFORMATION FORM
Date of Information
In this annual information form (“Annual Information Form”), ImmunoPrecise Antibodies Ltd., together with its subsidiaries, as the context requires, is referred to as “IPA” or the “Company”. All information contained in this Annual Information Form is as at April 30, 2022, unless otherwise stated, being the date of the most recently completed financial year of the Company, and the use of the present tense and of the words “is”, “are”, “current”, “currently”, “presently”, “now” and similar expressions in this Annual Information Form is to be construed as referring to information given as of that date.
Cautionary Statement Regarding Forward-Looking Statements and Information
This Annual Information Form contains forward-looking statements and information about the Company which reflect management’s expectations regarding the Company’s future growth, results of operations, operational and financial performance and business prospects and opportunities. In addition, the Company may make or approve certain statements or information in future filings with Canadian securities regulatory authorities, in news releases, or in oral or written presentations by representatives of the Company that are not statements of historical fact and may also constitute forward-looking statements or forward-looking information. All statements and information, other than statements or information of historical fact, made by the Company that address activities, events or developments that the Company expects or anticipates will or may occur in the future are forward-looking statements and information, including, but not limited to statements and information preceded by, followed by, or that include words such as “may”, “would”, “could”, “will”, “likely”, “expect”, “anticipate”, “believe”, “intends”, “plan”, “forecast”, “budget”, “schedule”, “project”, “estimate”, “outlook”, or the negative of those words or other similar or comparable words.
Forward-looking statements and information involve significant risks, assumptions, uncertainties and other factors that may cause actual future performance, achievements or other realities to differ materiality from those expressed or implied in any forward-looking statements or information and, accordingly, should not be read as guarantees of future performance, achievements or realities. Although the forward-looking statements and information contained in this Annual Information Form reflect management’s current beliefs based upon information currently available to management and based upon what management believes to be reasonable assumptions, the Company cannot be certain that actual results will be consistent with these forward-looking statements and information. A number of risks and factors could cause actual results, performance, or achievements to differ materially from the results expressed or implied in the forward-looking statements and information. Such risks and factors include, but are not limited to, the following:
| • | negative operating cash flow; |
| • | liquidity and future financing risk; |
| • | the financial position of the Company and its potential need for additional liquidity and capital in the future; |
| • | the success of any of the Company’s current or future strategic alliances; |
| • | the Company may become involved in regulatory or agency proceedings, investigations and audits; |
| • | the Company may be subject to litigation in the ordinary course of its business; |
| • | the ability of the Company to obtain, protect and enforce patents on its technology and products; |
| • | risks associated with applicable regulatory processes; |
| • | the ability of the Company to achieve publicly announced milestones; |
| • | the effectiveness of the Company’s business development and marketing strategies; |
| • | the competitive conditions of the industry in which the Company operates; |
| • | market perception of smaller companies; |
| • | the Company cannot assure the production of new and innovative processes, procedures or innovative approaches to antibody production or new antibodies; |
| • | the ability of the Company to manage growth; |
1
| • | the selection and integration of acquired businesses and technologies; |
| • | the Company may lose clients; |
| • | any reduction in demand; |
| • | any reduction or delay in government funding of research and development (“R&D”); |
| • | costs of being a public company in the United States; |
| • | the Company may fail to meet the delivery and performance requirements set forth in client contracts; |
| • | the Company may become subject to patent and other intellectual property litigation; |
| • | the Company’s dependence upon key personnel; |
| • | risks associated with the COVID-19 pandemic; |
| • | the Company may not achieve sufficient brand awareness; |
| • | the Company’s directors and officers may have interests which conflict with those of the Company; |
| • | the outsourcing trend in non-clinical discovery stages of drug discovery; |
| • | the Company’s products, services and expertise may become obsolete or uneconomical; |
| • | the effect of global economic conditions; |
| • | the Company has a limited number of suppliers; |
| • | the Company may become subject to liability for risks against which it cannot insure; |
| • | clients may restrict the Company’s use of scientific information; |
| • | the Company may experience failures of its laboratory facilities; |
| • | any contamination in animal populations; |
| • | any unauthorized access into information systems; |
| • | prospective investors’ ability to enforce civil liabilities; |
| • | the Company’s status as a foreign private issuer; |
| • | exposure to foreign exchange rates; |
| • | the effects of future sales or issuances of equity securities or debt securities; |
| • | the market price of the common shares may experience volatility; |
| • | the Company will maintain discretion in the use of proceeds of any offering of securities; |
| • | the Company has not declared or paid any dividends on the common shares and does not intend to do so in the foreseeable future; and |
| • | a liquid market for the common shares may not develop. |
For further details, see the “Risk Factors” section of this Annual Information Form.
Although the Company has attempted to identify important risks and factors that could cause actual actions, events or results to differ materially from those described in forward-looking statements or information, there may be other factors and risks that cause actions, events or results not to be as anticipated, estimated or intended. Further, any forward-looking statements and information contained herein are made as of the date of this Annual Information Form and, other than as required by applicable securities laws, the Company assumes no obligation to update or revise them to reflect new events or circumstances. New factors emerge from time to time, and it is not possible for management to predict all such factors and to assess in advance the impact of each such factor on the Company’s business or the extent to which any factor, or combination of factors, may cause actual realities to differ materially from those contained in any forward-looking statement or information. Accordingly, readers should not place undue reliance on forward-looking statements and information contained in this Annual Information Form and the documents incorporated by reference herein. All forward-looking statements and information disclosed in this Annual Information Form are qualified by this cautionary statement.
Share, Currency and Exchange Rate Information
The financial statements included herein are reported in Canadian dollars. References in this Annual Information Form to “$” are to the lawful currency of Canada, references to “€” are to the lawful currency of the European Union, and references to “US$” are to the lawful currency of the United States.
On July 27, 2022, the Bank of Canada average rate of exchange for one Canadian dollar in United States dollars was $1.00 = US$0.7769 and for one Canadian dollar in Euros was $1.00 = € 0.7661.
2
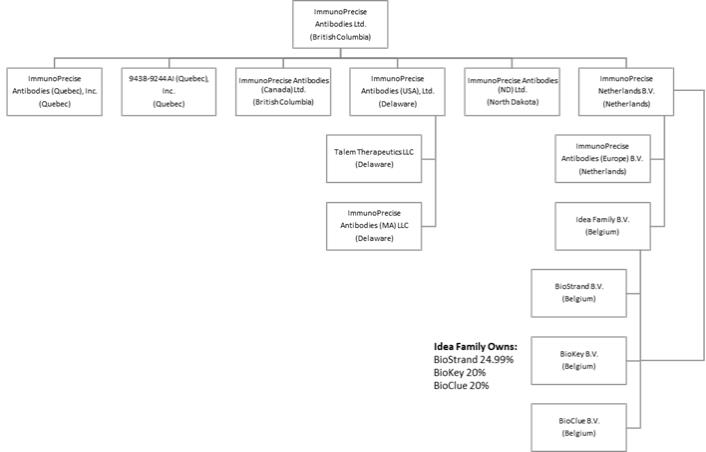
The Company was continued on September 2, 2016 under the Business Corporations Act (British Columbia) (“BCBCA”). On December 21, 2016, the Company changed its name to “ImmunoPrecise Antibodies Ltd.” The address of the Company’s head office is 3204 – 4464 Markham Street, Victoria, British Columbia V8Z 7X8. The registered and records office of the Company is located at 1800 – 510 West Georgia Street, Vancouver, British Columbia V6B 0M3.
The following chart sets out the Company’s intercorporate relationships with its subsidiaries, along with the jurisdiction in which such subsidiaries were formed. All of the Company’s subsidiaries are wholly owned by the Company.
GENERAL DEVELOPMENT OF THE BUSINESS
Overview
The Company is an innovation-driven, technology platform company that supports its pharmaceutical and biotechnology company partners in their quest to discover and develop novel, therapeutic antibodies against all classes of disease targets. See “Business – Overview” for further details.
3
The Company’s common shares are listed for trading on the TSX Venture Exchange (“TSXV”) under the trading symbol “IPA.” The Company’s common shares were approved for listing on the Nasdaq Global Market (“Nasdaq”) under the trading ticker symbol “IPA.”
Three Year History
Over the last three years, the Company has focused on growing its service and product offerings and revenues through organic growth as well as acquisitions, as set out below.
Fiscal Year Ended 2020
The Company achieved revenues of $14,057,927 during the year ended April 30, 2020, compared to revenues of $10,926,268 in the 2019 fiscal year. This represented a 29% increase in revenue for the year. The increasing revenue trend is due to increases in both volume and financial values of client contracts as a result of continued focus on expanding the breadth and depth of services offered, new client onboarding including top pharma companies, and growing the Company’s core existing client business.
Commitments
The Company entered into an operating lease for a piece of equipment for its Victoria, BC, Canada laboratory space on April 29, 2020. The lease commenced on May 15, 2020, with a 36-month term. The monthly lease payment is US$15,829. The Company has a right to purchase the equipment at fair market value at the end of the lease term.
Talem Therapeutics
On March 10, 2020, Talem entered into a research license agreement with Janssen Research & Development, LLC (“Janssen”), providing Janssen with exclusive access to a panel of novel, monoclonal antibodies against an undisclosed target. Pursuant to this license agreement, Janssen held an option to acquire all commercial rights to the antibodies. Under the terms of this license agreement, Janssen engaged the Company for an initial term of six months for aggregate consideration of less than US$500,000. In July 2020, Janssen requested a temporary extension of the license agreement on a no-fees basis. In December 2020, Janssen exercised its option to purchase the assets resulting from the work being conducted by Talem for aggregate consideration of less than US$1,000,000. The Company has committed few resources to this project and does not view this license agreement as being material to its operations.
New Laboratory Build, Manufacturing Capabilities and Service Offering
The Company has continued to invest significantly in ROI-generating capacity at its Utrecht location, committing to a new laboratory build and equipment purchases to support its growth. In January 2020, U-Protein Express B.V. (“UPE”) signed a long-term lease contract for a new multi-tenant building dedicated to the life sciences at the Utrecht Science Park alongside important stakeholders such as Genmab B.V. and Merus N.V. The Company expects UPE to take occupancy at this location in 2022.
Furthermore, along with Codex DNA, Inc. (formerly SGI-DNA, Inc.), the Company announced in January 2020 that UPE had integrated Codex DNA’s benchtop automated DNA printer, making the Company the first CRO in Europe to integrate the BioXp™ 3200 System in its workflow. As a result of this achievement, the Company aims to positively impact its manufacturing capacities by reducing the antibody design-synthesis-screening timeline, providing clear advantages to its partners, and to accelerate antibody discovery and manufacturing services at its European facilities.
The Company has continued to focus on advanced service offerings for therapeutic discovery and introduced the Abthena™ bispecific antibody platform. Bispecific antibodies can bind two different molecules with a single antibody, potentially increasing the therapeutic effectiveness of targeting infectious diseases, payload delivery and functional activity toward challenging targets. The Abthena platform enables the Company to test potential therapeutic lead candidates in a bispecific format early during the antibody discovery process.
4
Key Additions to the Board and Management Team
In October 2019, the Company appointed Dr. Stefan Lang as Chief Business Officer. Dr. Lang has more than 20 years of experience as a senior executive in the biotechnology industry. He has an impressive breadth of leadership within the biotech industry, including experience working at the organizational level and as a globally recognized and respected leader in antibody business development. In his most recent role, Dr. Lang worked in an executive role at Aldevron, LLC (“Aldevron”), as the Vice President of Business Development, with his main focus on corporate strategy, R&D innovation, sales and business development. Prior to Aldevron, he worked at GENOVAC, a pioneer in genetic immunization for antibody generation. In this newly created role, Dr. Lang is responsible for corporate and business development initiatives, as well as corporate and product strategic planning.
Also, in October 2019, the Company appointed Brian Lundstrom as a member of the Board of Directors. Mr. Lundstrom has over 30 years’ protein and antibody therapeutic business experience and was appointed to help strengthen the Company’s strategic and commercial growth in industry-leading therapeutic antibody discovery for both its clients and the Company’s emerging internal pipeline.
In April 2020, the Company appointed Dr. Yasmina Abdiche as Chief Scientific Officer (“CSO”). Dr. Abdiche was previously CSO at Carterra, where she helped to transition the LSA antibody screening technology to global commercialization. Prior to that, she had a twelve-year career at Rinat, where she led team of analytical scientists performing antibody characterization on label-free biosensors. Dr. Abdiche holds over twelve issued patents in the antibody space and is co-inventor of a PD-1 inhibitor (Sasanlimab, PF06801591, RN888) currently in clinical trials for various cancer types and of a market-approved anti-CGRP antibody for migraine, Ajovy. Dr. Abdiche was responsible for helping lead the Company’s research and development programs and services. Dr. Abdiche resigned from the Company effective June 30, 2021.
Fiscal Year Ended 2021
Private Placement Financing
On May 15, 2020, the Company closed a non-brokered private placement financing by issuing 10% convertible debentures (“New Debentures”) for total proceeds of $2,592,000. On May 27, 2020, the Company issued an additional $35,000 of the New Debentures. In total, the Company issued $2,627,000 of the New Debentures. The New Debentures are unsecured, bear interest at a rate of 10% per year and are payable at maturity. The maturity date is May 15, 2022, for $2,592,000 of the New Debentures and May 22, 2022, for $35,000 of the New Debentures. The principal amount of the New Debentures may be convertible, at the option of the holder, into common shares of the Company at a conversion price of $4.25 per share. The Company may force convert the principal amount of the New Debentures at $4.25 per Common Share if the average closing price is equal to or greater than $7.50 for 20 trading days. The Company paid finders cash commissions totaling $44,750. During the year $990,000 of the debentures were converted into 232,934 common shares.
Grant Funding
In April 2020, the Company was awarded US$75,000 in grant funds from the North Dakota Department of Agriculture through the state’s Bioscience Innovation Grant program to assist with expenses related to its PolyTope SARS-CoV-2 programs and was reimbursed in July 2020 after demonstrating a total spend in excess of $100,000 to satisfy the minimum 25% required match.
In June 2020, the Company was granted funding by TRANSVAC2, a European vaccine network, to cover the costs of a preclinical vaccine study of one of the Company’s vaccine candidates in a collaboration with LiteVax B.V.
5
In July 2020, the Company was awarded the Biosciences CARES grant from the Department of Agriculture of the State of North Dakota for the amount of US$1,500,000 to support the discovery, development and testing of SARS-CoV-2 therapeutic candidates. The total grant project cost is US$2,000,000 for which ImmunoPrecise Antibodies (USA) Ltd. (IPA USA) must contribute an amount not less than 25% of the grant project cost or US$500,000. The Company recorded other income for the years ended April 30, 2021, and 2020 of US$1,080,695 and US$158,000, respectively.
During the year ended April 30, 2021, the Company also received $583,347 from the Government of Canada through its Canadian Emergency Wage and Rent Subsidy Programs (CEWS and CERS). The subsidy was recorded in other income as subsidy income.
The Paycheck Protection Program was implemented in the United States to help businesses impacted by COVID-19 keep their workforce employed. Borrowers were eligible for full forgiveness if certain conditions were met. The Company was approved for a loan of US$209,000 and the loan was fully forgiven during the year ended April 30, 2021. The Company recognized $261,000 during the year ended April 30, 2021 in other income as subsidy income.
The Company had grants approved in the amount of approximately $55,000 in the form of reduced costs of services performed from the Canadian National Research Council’s (“NRC”) Innovation Research Assistance Program, to support collaborative research with the NRC.
Deferred Settlement Payments
The Company made the third and final deferred cash payment for the acquisition of ModiQuest Research B.V. pursuant to the Amendment, Termination and Settlement Agreement, by making a cash payment of €335,555 ($518,533) to Immusys.
On December 18 and December 31, 2020, the Company issued an aggregate of 203,178 common shares pursuant to a Share Exchange Agreement for the final deferred payment for the acquisition of UPE. The common shares were valued at $1,047,097 and issued to the sellers (Universiteit Utrecht Holding B.V., Bionomics B.V., Wieger Hemrika, and Roland Romijn) pursuant to the final deferred payment for the acquisition of UPE.
Issuance of Stock Options
On August 13, 2020, the Company granted 50,000 stock options, exercisable at $7.50 per option. The options are subject to vesting conditions and 25% of the options vest every three months. The options have an expiration date of August 13, 2023.
On September 1, 2020, the Company granted 270,000 stock options to officers and an employee of the Company, exercisable at $8.50 per option. The options are subject to vesting conditions as follows: one-third six months after the grant date, one-third 12 months after the grant date and one-third 18 months after the grant date. The options have an expiration date of September 1, 2025.
On January 6, 2021, the Company granted 25,000 stock options to directors of the Company, exercisable at $20.30 per option. The options are subject to vesting conditions as follows: one-quarter three months after the grant date, one-quarter six months after the grant date, one-quarter nine months after the grant date and one-quarter 12 months after the grant date. The options have an expiration date of January 6, 2026.
On January 6, 2021, the Company granted 238,000 stock options to employees of the Company, exercisable at $20.30 per option. The options are subject to vesting conditions as follows: one-third six months after the grant date, one-third 12 months after the grant date and one-third 18 months after the grant date. The options have an expiration date of January 6, 2026.
6
Collaboration Agreement
In October 2020, Talem entered into a collaboration agreement with Twist Bioscience Corporation (“Twist”) in order to expand its antibody pipeline on a wider range of oncology targets, combining their expertise in a highly collaborative manner to discover novel antibody therapeutics. The Company will contribute targets of interest with relevant background data, and the genetic sequences encoding for lead antibodies against the selected targets. Twist Biopharma, a division of Twist, will design synthetic antibody libraries based on the provided antibody repertoire sequences from immunized animals to discover optimized, humanized lead antibody candidates.
Talem Antibody Sale
In December 2020, a client purchased a set of antibodies against an undisclosed target from Talem. The Company recognized revenue of $1,156,967 in financial year 2021, and had previously recognized $229,849 in financial year 2020.
Nasdaq Listing
On September 10, 2020, the Company announced that it had commenced the application process to list the common shares on Nasdaq. The Company’s common shares were approved for listing on the Nasdaq under the trading ticker symbol “IPA.” Trading on Nasdaq commenced at market open on December 30, 2020.
Common Share Consolidation
On November 4, 2020, the Company announced that it intended to complete a consolidation of its issued and outstanding common shares on the basis of one new Common Share for every five issued and outstanding common shares (the “Consolidation”). The TSXV approved the Consolidation and on November 23, 2020, the Company consolidated its issued and outstanding common shares. All references to share and per share amounts in this Annual Information Form have been retroactively restated to reflect the Consolidation.
Base Shelf Prospectus Filing
On November 6, 2020, the Company filed a preliminary base shelf prospectus (the “Preliminary Shelf Prospectus”) with the securities commissions of each of the provinces of Canada except Quebec and on November 9, 2020, the Company filed a corresponding registration statement on Form F-10 (the “Registration Statement”) with the United States Securities and Exchange Commission (the “SEC”) under the multi-jurisdictional disclosure system adopted by the securities regulatory authorities in United States and Canada (“MJDS”).
The short form base shelf prospectus was receipted in each of the provinces of Canada except Quebec on December 14, 2020 and the corresponding Registration Statement was declared effective by the SEC on November 9, 2020, and allow the Company to undertake offerings of common shares, preferred shares, debt securities, warrants, units and subscription receipts, or any combination thereof, up to an aggregate total of $150,000,000 from time to time during the 25-month period that the final short form base shelf prospectus remains effective. The securities qualified thereunder may be offered in amounts, at prices and on terms to be determined at the time of sale and, subject to applicable regulations, may include “at-the-market” transactions, public offerings or strategic investments. The specific terms of any offering of the Company’s securities, including the use of proceeds from any offering, will be set forth in one or more shelf prospectus supplement(s) to be filed with applicable securities regulators.
Public Offering
On February 8, 2021, the Company closed a public offering of 1,616,293 common shares, at a price of US$13.45 per Common Share for gross proceeds of US$21,739,141 and net proceeds less underwriting discounts and commissions of US$19,575,364. On February 10, 2021, the Company issued an additional 242,443 common shares pursuant to the over-allotment option at a price of US$13.45 per Common Share for gross proceeds of US$3,260,858 and net proceeds less underwriting discounts and commissions of US$2,967,735.
7
Restructuring of the Netherland Entities
As of January 1, 2021, UPE and Immulease merged with IPA Europe to form one legal entity. UPE, now referred to as the Utrecht location, continues its operations in the biotechnology hub of Utrecht, the Netherlands.
Fiscal Year Ended 2022
Funding
Issuance of Stock Options
On May 9, 2021, the Company granted 10,000 stock options to strategic board members of the Company, exercisable at US$7.72 per option. The options are subject to vesting conditions as follows: one-third one year after the grant date, one-third two years after the grant date, and one-third three years after the grant date. The options have an expiration date of May 9, 2026.
On June 13, 2021, the Company granted 43,750 stock options to a consultant of the Company, exercisable at US$7.14 per option. The options vested on June 13, 2021. The options have an expiration date of June 1, 2024.On January 3, 2022, the Company granted 28,250 stock options to board members of the Company, exercisable for a period of five years from the date of grant at an exercise price of $6.89 per option. One quarter of the options will vest every three months from the date of grant.
On January 7, 2022, the Company granted 170,000 stock options to certain officers of IPA. Each option is exercisable for a period of five years from the date of grant at an exercise price of $7.94 per option. One third of the options will vest every six months from the date of grant.
On January 13, 2022, the Company granted 15,000 stock options to a certain officer of IPA. Each Option is exercisable for a period of six months from the date of grant at an exercise price of $8.30 per option. One third of the options vest every two months from the date of grant.
ATM Offering
On October 13, 2021, the Company and H.C. Wainwright & Co., LLC (the “Agent”) entered into an At-The-Market Offering Agreement (the “ATM Agreement”). Under the terms of the ATM Agreement, the Company is entitled, at its discretion and from time-to-time during the term of the ATM Agreement, to sell, through the Agent, acting as sole sales agent, common shares of the Company having an aggregate gross sales price of up to US$50 million. Sales of the common shares will be made in transactions that are deemed to be “at-the-market distributions” as defined in National Instrument 44-102 – Shelf Distributions, including, without limitation, sales made directly on Nasdaq or any other existing trading market for the common shares in the United States. No offers or sales of common shares will be made in Canada on the TSXV or other trading markets in Canada. The Company will determine, at its sole discretion, the date, minimum price and maximum number of common shares to be sold under the ATM Facility. The common shares will be distributed from time to time in negotiated transactions, at market prices prevailing at the time of sale, at prices relating to such prevailing market prices, and/or in any other manner permitted by applicable law. As such, the prices may vary between purchasers over time. The Company is not required to sell any common shares at any time during the term of the ATM facility. The ATM Agreement will remain in full force until (i) the date that it is terminated by the Company or the Agent, (ii) such date on which the maximum amount of common shares contemplated by the ATM Agreement has been sold, or (iii) or otherwise by mutual agreement of the parties.
8
Final Deferred Settlement Payment
The Company made the third and final deferred share payment for the acquisition of ModiQuest Research B.V. pursuant to the Amendment, Termination and Settlement Agreement on May 3, 2021, issuing 41,488 common shares of the Company with a fair value of $503,243 to the seller (Immusys) of ModiQuest Research B.V, now ImmunoPrecise Antibodies (Europe) B.V.
Key additions and changes to the board and management team
On June 2, 2021, the Company announced that Dr. Yasmina Abdiche resigned as CSO effective June 30, 2021, to pursue other opportunities. The Board appointed Dr. Ilse Roodink as CSO effective as of July 1, 2021.
The Company promoted Barry Duplantis, Ph.D., to Vice President of Client Relations and appointed Ms. Carla Dahl, as Vice President of Marketing. Paul Andreola resigned from the Company’s board of directors effective November 9, 2021, for personal reasons unrelated to the Company.
In April 2022, Chief Business Officer, Stefan Lang, departed the Company.
Strategic Advisory Board
On May 25, 2021, the Company announced the addition of Dr. Dion Neame to the Company’s Strategic Advisory Board (“SAB”). Dr. Neame is the second addition from the large pharma community to join IPA’s SAB. Each member of the SAB holds senior leadership roles and has been strategically onboarded to assist IPA with executing its growth and expansion plans.
SARS-CoV-2 Therapeutic Research
COVID-19 R&D
There is an ongoing need for therapeutics to protect against COVID-19 despite the availability of vaccines, as vaccines do not provide protection for all individuals. This is particularly true for immunocompromised individuals such as the elderly, cancer patients, individuals with HIV or those undergoing bone marrow and organ transplants, whose immune systems are too weak to mount an effective response upon vaccination. In addition, SARS-CoV-2 vaccines appear to be less effective against variants of the virus. Without 100% protection, important segments of higher exposure risk populations will likely be left unprotected – namely frontline workers and those living in group care.
Therapeutic antibodies are providing breakthrough medicines for cancer, inflammation, autoimmune and infectious diseases due in part to their high on-target affinity and exquisite specificity making them highly efficacious with good safety profiles.
Technological advances in antibody discovery methods such as B cell sorting now enable the rapid and systematic generation of high-quality fully human antibodies from healthy donors, diseased patients, and transgenic animals. Furthermore, when therapeutic antibodies are combined into cocktails, they can provide unique protection against infectious diseases by working synergistically to neutralize pathogens via multiple mechanism of action engaged in concert, boosting potency beyond the sum of their individual components. Single antibodies are vulnerable to mutagenic escape and can be rendered ineffective by a single point mutation in the pathogen. In contrast, antibody cocktails may protect against mutagenic escape because they cover a larger epitope footprint on the pathogen’s surface than possible with a single antibody, providing longer-lasting protection against emerging mutations.
The Company’s diverse panel of anti-SARS-CoV-2 antibodies with therapeutic potential can be curated into synergistic cocktails, providing opportunities for out-licensing and sponsorship deals which the Company believes would enable it to respond quickly to emerging viral variants as well as formulation into bi- or multi-specifics. The Company has successfully completed pre-clinical studies in Syrian hamsters and could demonstrate powerful in vivo efficacy in both therapeutic and prophylactic settings.
9
On January 27, 2022, Talem, filed for patent protection of its PolyTope® TATX-03 antibody cocktail via the PCT (Patent Cooperation Treaty) system (which has 154 member states) as well as national filings in the US, Taiwan, Argentina, and Paraguay, enabling the Company to pursue patent protection of the PolyTope TATX-03 cocktail in all sizeable potential markets. In the meantime, the PolyTope TATX-03 remaining non-clinical safety studies are expected to conclude in Q2 of CY2022. At the request of the FDA, the Company has prepared a comprehensive status update demonstrating the performance of PolyTope TATX-03 toward Omicron (B.1.1.529), and other variants of concern, for review. The Company expects that, upon completion of its ongoing studies, the data will enable the Company to file an Investigational New Drug (“IND”) application or a Clinical Trial Application, in Q3 of CY2022. The approval of the FDA or EMA with respect to the IND or EMA application will be required prior to commencing first-in-human clinical studies.
The Company is presently manufacturing clinical batches of the fully human, lead monoclonal antibodies of its TATX-03 cocktail and aims to use the resulting data to support conversations with sponsors, potential partners, and funding agencies. The Company anticipates similar cocktail formulations, including its bi-specific formulations, to also follow into pre-clinical testing in the near-term. As result, the Company anticipates that such developments will provide on-going opportunities for commercialization.
On April 14, 2022, the Company completed the acquisition of control over BioStrand BV, BioKey BV, and BioClue BV (hereinafter collectively referred to as “BioStrand”), a group of Belgian biotech entities and pioneers in the field of bioinformatics and biotechnology, through its wholly owned subsidiary ImmunoPrecise Netherlands BV. The Company paid a consideration of approximately € 20 million to the vendors, consisting of an aggregate of 4,077,774 common shares of IPA and a cash payment of approximately € 3,734,500. The consideration also includes a contingent earnout payment based on the profitability of BioStrand over a 7-year period, which shall not exceed in total €12 million.
Subsequent to fiscal year 2022
Grant Funding
In May 2022, Biostrand received a €460,000 round of grant funding from VLAIO (Flanders Innovation & Entrepreneurship), the research fund of the Flemish regional government in Belgium. Conditionally awarded in January 2022, BioStrand satisfied the remaining criteria for the award, which follows an original grant of €235,000 received from VLAIO in 2020.
Overview
The Company is an innovation-driven, technology platform company that supports its pharmaceutical and biotechnology company partners in their quest to discover and develop novel, therapeutic antibodies against all classes of disease targets. The Company aims to transform the conventional, multi-vendor, product development model by bringing innovative and high-throughput technologies to its partners, incorporating the advantages of diverse antibody repertoires with the Company’s therapeutic antibody discovery suite of technologies, to exploit antibodies of broad epitope coverage, multiple antibody formats, valency, and size, and to discover antibodies against multiple/rare epitopes.
The Company offers comprehensive support to its partners, starting with customized, computational project design, antigen preparation, an on-site vivarium, immunization services, high-throughput discovery platforms, functional antibody testing, lead candidate selection, antibody optimization, antibody engineering and manufacturing, all under one contract.
The Company believes that its experience, innovation, technologies, scientific rigor, and focus on producing quality products, provide a unique experience in one-stop service offerings, and assist the Company in its aim to reduce the time required for, and the inherent risk associated with, conventional multi-vendor product development.
10
The Company has achieved organic revenue growth through market penetration and service diversification in the biologics, CRO space, as well as accretive growth through strategic expansion of its operations in Europe, by acquiring and integrating innovative technologies, and through investments in R&D.
Products and Services
CRO Services
The Company’s CRO services include, but are not limited to, proprietary B cell sorting, screening and sequencing; custom, immune and naïve phage display production and screening; expertise with transgenic animals and multi-species antibody discovery; bi-specific, tri-specific, VHH, and VNAR (shark) antibody manufacturing; DNA cloning, protein and antibody downstream processing, purification in gram scale levels, characterization and validation; antibody characterization on label-free biosensors, antibody engineering; transient and stable cell line generation; antibody optimization and humanization; hybridoma production with multiplexed, high-throughput screening and clone-picking; cryopreservation; and custom antigen modeling, design and manufacturing.
Moreover, in the past two years, the Company has gained increasing recognition as a rising leader in the biologics CRO space, with a focus on organic growth through market penetration and service diversification, as well as strategic expansion with platform and process integration. Furthermore, end-to-end services have been leveraged through acquisition, enabling a steady foundation for future growth.
In fiscal 2021, the Company’s CRO services accounted for 89% (2020: 96%) of the Company’s revenue.
The Company’s wholly owned subsidiaries, IPA (Canada) Ltd. (“IPA Canada”) and IPA Europe, have both been designated as approved CROs for the world’s leading, transgenic animal platform producing human antibodies, and exercised an advantage in optimizing services for various transgenic animal vendors. The Company made strategic investments in R&D activities to develop proprietary technologies enabling the application of their B cell Select™ and DeepDisplay™ platforms to address a range of transgenic animal species and strains and efficiently deliver fully-human, clinically relevant antibodies to its clients.
The Company’s key CRO services are set forth in detail below:
Service | Details | |
| B cell SelectTM | In 2018, the Company built on its decade of experience in single B cell interrogation to offer B cell services in both North America and Europe on species agnostic platforms, including the use of transgenic, humanized animals. These services are offered for a broad range of therapeutically relevant protein families, including GPCRs and other challenging, membrane-spanning proteins. The Company’s B cell Select™ platforms enable antibody screening directly from B cells, facilitating the analysis of a more diverse set of antibodies, and for faster, deeper screening compared to traditional technologies. By adding a high throughput, label-free Octet HTX biosensor (under the tradenames FortéBio, Sartorius) at IPA Canada, the Company uses a state-of-the-art high throughput platform that facilitates the rapid characterization and development of lead antibody candidates and addresses the need for increased speed and sample throughput when characterizing large panels of therapeutic antibody candidates, which are generated with its B cell or library-based platforms. |
11
Service | Details | |
| Phage Display | The Company’s phage display services are based on building custom immune libraries from multiple species, including transgenic animals, or, alternatively, the selection of antigen-specific, recombinant antibody fragments from its proprietary human or llama phage libraries. The proprietary libraries have been made from human auto-immune (diseased) patients and naïve (healthy donors) scFv (single chain fragment variable) repertoires, as well as from naïve llama (VHH) repertoires. Custom immune libraries are prepared from blood, spleen, lymph nodes, and bone marrow of immunized animals and aim to capture the entire immune repertoire for panning, rescue, and identification of unique antibodies with pre-specified characteristics. | |
| DeepDisplayTM | A powerful new technology utilizing a combination of transgenic animal platforms, like e.g. Ligand’s OmniAb®, and IPA’s custom phage display antibody selection. | |
| AbthenaTM Bispecifics | The Company’s bispecific Abthena™ technology complements its diverse discovery process, integrating seamlessly with the Artemis™ Intelligence Metadata (AIM)™ capabilities, to enable rapid turnaround on additional algorithmic outputs in therapeutic antibody optimization, stability, affinity, and manufacturability. | |
| LucinaTechTM Humanization | The Company provides a robust and efficient antibody humanization service, which consistently retains affinity and specificity levels. The approach is based on state-of-the-art in silico antibody modeling to identify essential framework and CDR residues for grafting onto a human antibody framework. | |
| Affinity Maturation | Antibody affinity is important in therapeutic and diagnostic applications. The Company’s affinity maturation service can improve antibody affinities. The Company applies different strategies to increase the affinity of the antibody, including gene shuffling and random mutagenesis. | |
| Immunization, hybridoma, sequencing | The Company offers antibody development services including a variety of immunization methods: Rapid Prime™ immunization, DNA immunization (NonaVac™), cell-based immunization (ModiVacc™), electro-fusion and hybridoma generation using semi-solid media and clone picking, as well as high throughput, multiplexed screening methods. With ImmunoProtect™, the DNA sequence of the antibody is determined and can be used to express the antibody recombinantly. | |
| rPExTM protein manufacturing | The Company provides large-scale production of recombinant mammalian proteins and antibodies for research and non-clinical applications. With a track record of successfully producing difficult-to-express proteins and antibodies (e.g. Fc-fusion proteins and bispecific antibodies), the Company offers gram scale production with low endotoxin levels. | |
| Cell line development | Using its proprietary vectors, the Company offers stable cell line development services (non GMP) of target proteins or antibodies adapted to specific growth conditions and media. | |
12
Therapeutic Discovery Program
While CRO services are the mainstay of the Company, IPA has worked continuously on building an IP estate and portfolio of proprietary methods and physical assets through collaborations, acquisitions and in-licensing. The Company has strategically invested in the development and licensing of antibody discovery platforms and related IP assets. The onboarding of existing assets with regard to equipment, technologies, IP and licenses within the Company’s European Union operations has been compounded by active research and development at all operational sites over the past two years, including the on-going development of new service offerings rolled out in the fiscal years 2020 and 2021, but more notably, internal discovery programs focused on novel, therapeutic antibodies, primarily in the field of immuno-oncology.
The Company formed Talem, based in Cambridge, Massachusetts, to support its internal and partnered therapeutic discovery programs. Talem offers strategic partnerships with pharma and biotech companies and is the only company to offer these services as a partnership in OmniAb® transgenic animals using their own license. The depth and speed of IPA’s offerings enables Talem to customize each program and leverages the Company’s expertise and technologies in the antibody discovery.
Locations of Operations
The Company’s operations are carried out globally in Victoria, British Columbia (IPA Canada) and Utrecht and Oss, the Netherlands (IPA Europe), and Fargo, North Dakota (IPA USA).
IPA Canada’s laboratory facilities in Victoria offer a variety of innovative services including B cell screening, sequencing and cloning, an on-site vivarium, a dedicated hybridoma suite, and modern screening services including semi-automated multiplexed screening and label-free biosensor analyses. The site continues to invest in developing new and supporting technologies to optimize the client experience and ensure state-of-the-art, high throughput capabilities.
IPA Europe at the location in Utrecht, the Netherlands, has been a staple in the recombinant protein community, operating for over 18 years, and specializing in the manufacture of complex proteins and antibodies in a variety of formats and from a range of mammalian cell types. Its streamlined and efficient operations have enabled it to successfully support over 6,000 different programs, with over a 90% success rate, for pharmaceutical and biotechnology industries as well as leading, academic institutions. In a seamless coordination, its operations also support the downstream expression and purification of the antibodies originating from the B cell Select programs, enabling validation of the platform’s outputs and comprehensive deliverables for clients. The site also has a global, exclusive license from Stanford University for the marketing and sales of the novel protein, Wnt surrogate Fc, which IPA Europe co-developed.
IPA Europe at the location in Oss, the Netherlands, contributed substantially in services and IP to the Company after its acquisition. The integration of the site significantly expanded the Company’s services portfolio including affinity maturation, humanization, functional assay design and development, naïve and disease human scFv libraries, naïve llama VHH libraries, and proprietary methods of immunization against conformational targets (e.g. ModiVacc™ mouse lymphoid tumor immunization and DNA immunization technologies). Adding to its proprietary services, IPA Europe developed and rolled-out the aforementioned DeepDisplay service for the discovery of fully human antibodies using transgenic animal immunization and custom phage display.
The Company established its executive headquarters in Fargo, North Dakota in 2018 in an effort to bring key members of management under a streamlined chain of command that is responsible for pipeline selection and oversight, policy establishment, finances and accounting, sales and marketing, communication, contracts, information technology governance and administration. The Fargo site is also the address of IPA (ND) Ltd. and IPA USA and offers the potential for future growth plans in the United States.
As of April 30, 2022 the Company had 85 employees.
13
Intellectual Property
The Company has initiated the protection of new innovation in its product pipeline and has trademarked its ImmunoProtect™, Rapid Prime™, DeepDisplay™, NonaVac™, Abthena™, Artemis™, ModiVacc™, LucinaTec™, B cell Select™ and rPExTM technologies. Currently, the Company has filed four patent applications (CA 2964907, CA 2947878, CA 2965017 and CA 2940065) relating to its proprietary technology and products, and has filed a provisional patent to protect its PolyTope SARS-CoV-2 intellectual property. Its IP strategy has been to protect its intellectual property primarily through a combination of trade secrets and copyright. See also “Risk Factors”.
The Company continues to develop new products such as novel biotherapeutics in a broad range of indications. New screening methodologies, screening services and data mining methodologies may also provide an expansion and new commercial opportunities for the Company.
Specialized Skill and Knowledge
The Company’s qualified staff of research and development scientists have experience in biotechnology and the pharmaceutical sector, academic research and government. The Company brings 30 years of experience in the production of antibodies and has a strong reputation for the delivery of a high standard of quality and professional antibody services and products.
Further, the Company has an in-house research staff, including a number of research scientists with MSc and a cadre of technical staff, innovating proprietary Rapid Prime immunization, single step cloning using semi- solid media for HAT selection of hybridomas, and B cell selection and screening.
Competitive Conditions
The Company competes primarily against other full-service CROs as well as services provided by in-house research and development, or R&D, departments of biopharmaceutical companies. The Company’s major CRO competitors include Abveris Inc., Genovac GmbH (formerly part of Aldevron LLC), Antibody Solutions, Genscript Biotech Corp, Lake Pharma Inc. (now part of Curia Inc.), and several specialty and regional CROs.
Competitive factors in the industry in which the Company operates include, but are not limited to, experience within specific therapeutic areas, quality of staff and services, reliability, range of provided services, ability to recruit principal investigators and patients into studies expeditiously, ability to organize and manage large-scale, global clinical trials, global presence with strategically located facilities, speed to completion, price and overall value. The Company believes it competes effectively with its competitors across these factors, particularly due to its full-service operating model, its therapeutic expertise, its global platform and its experienced and committed management team. However, some of the Company’s competitors have greater financial resources and a wider range of service offerings over a greater geographic area than the Company, which could put the Company at a competitive disadvantage with respect to these competitors. Many are also well known for niche specialities such as antibody development against glycosylated peptides or specific chemical modifications, specialties that the Company also houses, but is not yet well known for, which could put the Company at a competitive disadvantage with respect to these competitors.
Many competitors offer custom antibody production services in addition to large catalogues of antibodies available for sale through their websites. Over the years a number of competitors have been acquired and merged into larger companies, particularly larger laboratory facilities.
The R&D antibodies market is highly fragmented and served by numerous small suppliers of a similar size and scale to the Company, and no single company appears to dominate the market.
Regulatory Environment
The development, testing, manufacturing, labeling, storage and approval of antibody and therapeutic products are subject to regulation by various government authorities in Canada and in Europe. Companies
14
in the pharmaceutical and biotechnology industries, such as the Company’s clients, that carry out clinical trials are subject to stringent regulations. These regulations apply to the Company’s clients and are generally applicable to the Company when it provides services to its clients. Consequently, the Company must comply with relevant laws and regulations in the conduct of its business. The Company is in compliance with all Canadian and European regulations regarding the on-going operation of its laboratory facilities and delivery of all its products and services.
Seasonality
Sales of the Company’s products and services have not been subject to seasonality fluctuations.
Changes to Contracts
The Company uses a standard Master Services Agreement (“MSA”) with all customers for custom monoclonal and polyclonal antibodies and peptide protection and does not anticipate any changes in its MSA. The Company has a standard form of contract for its other services and anticipates development of a standard license agreement to take advantage of new licensing opportunities.
Foreign Operations
The Company currently conducts business activities in Canada and a significant portion of the Company’s business activities depend on foreign operations in the Netherlands and the United States. The Company distributes and offers its products and services globally. Significant portions of our revenues are from global sales. In fiscal 2021, 44% of our revenues came from sales to the United States, 42% from Europe and 8% to countries other than Canada.
Market for Products
Market Segment and Geographic Areas
The market for therapeutic antibodies was worth US$115 billion in 2018. According to a study published in the Journal of Biomedical Science in January 2020, it is estimated that the human therapeutic antibody market will grow to US$300 billion in 2025. Growth drivers in the antibody market are as follows:
| • | Increasing research and development expenditures in the life science sector and in the therapeutics industry |
| • | Emergence of innovative, facilitating platforms |
| • | Growing demand for revolutionary therapies for major diseases as populations age and life expectancies increase |
| • | Growing emphasis on antibody development at CROs |
| • | Increasing applications in the environmental sectors |
| • | Biopharmaceuticals is the fastest growing pharma sector. This market is mainly dominated by large pharmaceutical companies, like Abbvie, Novartis, Roche and Johnson & Johnson. Companies are currently sponsoring clinical studies for more than 570 monoclonal antibodies (mAbs). Of these, approximately 90% are early-stage studies designed to assess safety (Phase I) or safety and preliminary efficacy (Phase I/II or Phase II) in patient populations. |
The global immunoassay market is estimated to accumulate US$37,987.8 million by the year 2027. According to MarketStudyReport.com, the global immunoassay market was worth US$21,800 million in 2018 and is anticipated to grow with a compound annual growth rate (“CAGR”) of 6.5% through the year 2027.
15
In recent years, the number of monoclonal antibody drugs approved for commercialization has proliferated, with the 100th monoclonal antibody approved by the United States Food and Drug Administration (“FDA”) as of May 2021 (Nature Reviews Drug Discovery) and further 17 investigational antibody therapeutics in regulatory review in either the United States or Europe as of June 2021 according to AntibodySociety.org. According to a 2017 report from FiercePharma.com, it is expected that 9 of the 15 best-selling drugs worldwide in 2022 will be monoclonal antibody drugs, the fastest growing segment in the bio-pharmaceutical market.
The protein- and antibody-related service and product market is expected to grow with a CAGR of 6.2% by 2027 to US$5.6 billion, according to GrandViewResearch.com.
Prior to the acquisitions of UPE and IPA Europe, the Company focused on serving primarily the diagnostic antibody market in North America. Since such acquisitions, the Company has redirected most of its focus to the therapeutic antibody market and delivering an expanded portfolio of products and services to customers in Europe, a broader segment of North America and the rest of the world.
Marketing Plan and Strategies
Market Acceptance
The Company has a long-standing acceptance of its customized antibodies and protein production services in the market. The Company believes that the market acceptance of its products will continue as it organically grows its business, optimizes its laboratory, new sales and marketing capacity and production process to support long-term growth. Further, the Company is one of the few approved CROs for multiple transgenic animal providers on the market, enabling the faster development of therapeutic antibodies. Among 28 human antibodies approved by the FDA between 2002 and 2019, 19 were animal derived and nine were generated by phage display.
Bankruptcy and Similar Procedures
The Company does not have any bankruptcy, receivership or similar proceedings or any voluntary bankruptcy, receivership or similar proceedings within the three most recently completed financial years or completed during or proposed for the current financial year.
There are numerous and varied risks, known and unknown, that may prevent the Company from achieving its goals. The risks described below are not the only ones the Company will face. If any of these risks actually occurs, the Company’s business, financial condition or results of operations may be materially and adversely affected. In that case, the trading price of the Company’s securities could decline and investors in such securities could lose all or part of their investment.
Negative Operating Cash Flow
The Company has negative cash flow from operating activities and has historically incurred net losses. There is no assurance that the Company will generate sufficient revenues in the near future. To the extent that the Company has negative operating cash flows in future periods, it may need to deploy a portion of its existing working capital to fund such negative cash flows. The Company expects to need to raise additional funds through issuances of securities or through loan financing. There is no assurance that additional capital or other types of financing will be available if needed or that these financings will be on terms at least as favourable to the Company as those previously obtained, or at all. If the Company is unable to obtain additional financing from outside sources and eventually generate enough revenues, the Company may be forced to sell a portion or all of the Company’s assets, or curtail or discontinue the Company’s operations. If any of these events happen, investors may lose all or part of their investment.
16
Liquidity and Future Financing Risk
Although the Company is a going concern and, according to management’s estimates, has sufficient cash reserves to sustain existing operations for at least two years, the Company does not have cash reserves to fund all its strategic future growth and expansion plans. The Company’s ability to secure any required financing to sustain its operations will depend in part upon prevailing capital market conditions, as well as the Company’s business success. There can be no assurance that the Company will be successful in its efforts to secure any additional financing or additional financing on terms satisfactory to the Company’s management. If additional financing is raised by issuing shares of the Company, control of the Company may change, and shareholders may suffer additional dilution. If adequate funds are not available, or are not available on acceptable terms, the Company may be required to scale back its business plan.
Financial Position and Additional Needs for Liquidity and Capital
The Company is a biopharmaceutical company focused on the development of novel, therapeutic antibodies. Investment in biopharmaceutical product development is highly speculative because it entails substantial upfront capital expenditures and significant risk that a product candidate will fail to prove effective, gain regulatory approval or become commercially viable. The Company does not have any products approved by regulatory authorities and has not generated substantial revenues from collaboration and licensing agreements or clinical product sales to date, and has incurred significant research, development and other expenses related to ongoing operations and expects to continue to incur such expenses. As a result, the Company has not been profitable and has incurred operating losses in every reporting period since its inception and has a significant accumulated deficit. Operating costs are expected to increase in the near term as the Company continues product development efforts and expects to continue until such time as any future product sales, royalty payments, licensing fees, and/or milestone payments are sufficient to generate revenues to fund continuing operations. The Company is unable to predict the extent of any future losses or when this business section will become profitable, if ever. Even if the Company achieves profitability, it may not be able to sustain or increase profitability on an ongoing basis.
Strategic Alliances
The Company currently has, and may in the future enter into, strategic alliances with third parties that the Company believes will complement or augment its existing business. The Company’s ability to enter into strategic alliances is dependent upon, and may be limited by, the availability of suitable candidates and capital. In addition, strategic alliances could present unforeseen integration obstacles or costs, may not enhance the Company’s business, and may involve risks that could adversely affect the Company, including significant amounts of management time that may be diverted from operations in order to pursue and complete such transactions or maintain such strategic alliances. Future strategic alliances could result in the incurrence of additional debt, costs and contingent liabilities, and there can be no assurance that future strategic alliances will achieve, or that the Company’s existing strategic alliances will continue to achieve, the expected benefits to the Company’s business or that the Company will be able to consummate future strategic alliances on satisfactory terms, or at all. Any of the foregoing could have a material adverse effect on the Company’s business, financial condition and results of operation.
The Company may not be able to enter into collaboration agreements on terms favorable to the Company or at all. Furthermore, some of those agreements may give substantial responsibility over the Company’s drug candidates to the collaborator.
If the Company enters into collaboration agreements for one or more of its drug candidates, the success of such drug candidates will depend in great part upon the Company’s and its collaborators’ success in promoting them as superior to other treatment alternatives. The Company believes that its drug candidates may be proven to offer disease treatment with notable advantages over other drugs. However, there can be no assurance that the Company will be able to prove these advantages or that the advantages will be sufficient to support the successful commercialization of its drug candidates.
17
Regulatory or Agency Proceedings, Investigations and Audits
The Company’s business requires compliance with many laws and regulations. Failure to comply with these laws and regulations could subject the Company to regulatory or agency proceedings or investigations and could also lead to damage awards, fines and penalties. The Company may become involved in a number of government or agency proceedings, investigations and audits. The outcome of any regulatory or agency proceedings, investigations, audits, and other contingencies could harm the Company’s reputation, require the Company to take, or refrain from taking, actions that could harm its operations or require the Company to pay substantial amounts of money, harming its financial condition. There can be no assurance that any pending or future regulatory or agency proceedings, investigations and audits will not result in substantial costs or a diversion of management’s attention and resources or have a material adverse impact on the Company’s business, financial condition and results of operations.
Litigation Risk
The Company may become party to litigation from time to time in the ordinary course of business including, but not limited to, in connection with its operations or pursuant to the terms of any of its commercial agreements, which could adversely affect its business. Should any litigation in which the Company becomes involved be decided against the Company, such a decision could adversely affect the Company’s ability to continue operating and the value of the Company’s securities and could use significant resources. Even if the Company is involved in litigation and wins, litigation can redirect significant Company resources, including the time and attention of management and available working capital. Litigation may also create a negative perception of the Company’s brand.
Intellectual Property Protection
The Company’s success will depend on its ability to obtain, protect and enforce patents on its technology and products. Any patents that the Company may own or license in the future may not afford meaningful protection for its technology and products. The Company’s efforts to enforce and maintain its intellectual property rights may not be successful and may result in substantial costs and diversion of management time. In addition, others may challenge patents the Company may obtain in the future and, as a result, these patents could be narrowed, invalidated or rendered unenforceable or it may be forced to stop using the technology covered by these patents or to license the technology from third parties. In addition, current and future patent applications on which the Company depends may not result in the issuance of patents. Even if the Company’s rights are valid, enforceable and broad in scope, competitors may develop products based on similar technology that is not covered by the Company’s patents. Further, since there is a substantial backlog of patent applications at the various patent offices, the approval or rejection of the Company and its competitors’ patent applications may take several years.
In addition to patent protection, the Company also relies on copyright and trademark protection, trade secrets, know-how, continuing technological innovation and licensing opportunities. In an effort to maintain the confidentiality and ownership of the Company’s trade secrets and proprietary information, the Company requires its employees, consultants and advisors to execute confidentiality and proprietary information agreements. However, these agreements may not provide the Company with adequate protection against improper use or disclosure of confidential information and there may not be adequate remedies in the event of unauthorized use or disclosure. Furthermore, like many companies in the Company’s industry, the Company may from time to time hire scientific personnel formerly employed by other companies involved in one or more areas similar to the activities the Company conducts. In some situations, the Company’s confidentiality and proprietary information agreements may conflict with, or be subject to, the rights of third parties with whom its employees, consultants or advisors have prior employment or consulting relationships. Although the Company require its employees and consultants to maintain the confidentiality of all confidential information of previous employers, the Company or these individuals may be subject to allegations of trade secret misappropriation or other similar claims as a result of their prior affiliations. Finally, others may independently develop substantially equivalent proprietary information and techniques, or otherwise gain access to its trade secrets. The Company’s failure to protect its proprietary information and techniques may inhibit or limit its ability to exclude certain competitors from the market and execute its business strategies.
18
Regulatory Approval Processes
The Company’s businesses are subject to certain laws, regulations, and guidelines. Although the Company intends to comply with all such laws, regulations, and guidelines there is no guarantee that the governing laws and regulations will not change, which will be outside of the Company’s control. Numerous statutes and regulations govern the preclinical and clinical development, manufacture and sale, and post-marketing responsibilities for non-therapeutic and human therapeutic products in the United States, the European Union, Canada, Australia and other countries that are the intended markets for current and future product candidates. Such legislation and regulation governs the approval of manufacturing facilities, the testing procedures, and controlled research that must be carried out, and the preclinical and clinical data that must be collected prior to marketing approval. The Company’s R&D efforts, as well as any future clinical trials, and the manufacturing and marketing of any products the Company may develop, will be subject to and restricted by such extensive regulation.
The process of obtaining necessary regulatory approvals is lengthy, expensive, and uncertain. The Company may fail to obtain the necessary approvals to commence or continue clinical testing or to manufacture or market potential products in reasonable time frames, if at all. In addition, governmental authorities may enact regulatory reforms or restrictions on the development of new therapies that could adversely affect the regulatory environment in which the Company operates or the development of any products the Company may develop.
Completing clinical testing and obtaining required approvals is expected to take several years and to require the expenditure of substantial resources of the Company. There can be no assurance that clinical trials will be completed successfully within any specified period of time, if at all. Furthermore, clinical trials may be delayed or suspended at any time by the Company or by the various regulatory authorities if it is determined at any time that the subjects or patients are being exposed to unacceptable risks.
Any failure or delay in obtaining regulatory approvals would adversely affect the Company’s ability to utilize its technology and would therefore adversely affect its operations. Furthermore, no assurance can be given that the Company’s current or future product candidates will prove to be safe and effective in clinical trials or that such product candidates will receive the requisite regulatory approval. Moreover, any regulatory approval of a drug which is eventually obtained may be granted with specific limitations on the indicated uses for which that drug may be marketed. Furthermore, product approvals may be withdrawn if problems occur following initial marketing or if compliance with regulatory standards is not maintained.
Publicly Announced Milestones
From time to time, the Company may announce the timing of certain events which are expected to occur, such as the anticipated timing of results from clinical trials. These statements are forward-looking and are based on the best estimates of management at the time. However, the actual timing of such events may differ significantly from what has been publicly disclosed. The timing of events such as the initiation or completion of a clinical trial, filing of an application to obtain regulatory approval, or an announcement of additional clinical trials for a product candidate may ultimately vary from what is publicly disclosed. These variations in timing may occur as a result of different events, including the nature of the results obtained during a clinical trial or during a research phase, problems with a contract manufacturing organization or CRO or any other event having the effect of delaying the publicly announced timeline. The Company undertakes no obligation to update or revise any forward-looking information, whether as a result of new information, future events or otherwise, except as otherwise required by law. Any variation in the timing of previously announced milestones could have a material adverse effect on the Company’s business plan, financial condition or operating results, and the trading price of the common shares.
Business Development and Marketing Strategies
The Company’s future growth and profitability will depend on the effectiveness and efficiency of its national and international business development and marketing and sales strategy, including the Company’s ability to (i) grow brand recognition for its services internationally; (ii) determine appropriate business development, marketing and sales strategies and (iii) maintain acceptable operating margins on such costs.
19
There can be no assurance that business development, marketing and sales costs will result in revenues for the Company’s business in the future, or will generate awareness of the Company’s products and services. In addition, no assurance can be given that the Company will be able to manage its business development, marketing and sales costs on a cost-effective basis.
Competition
Although the Company believes that there are only a limited number of full-service, biologics, CRO firms, the Company may face intense competition in selling its products and services. Some competitors may have marketing, financial, development and personnel resources which exceed those of the Company. As a result of this competition, the Company may be unable to maintain its operations or develop them as currently proposed on terms it considers acceptable or at all. Increased competition by larger, better-financed competitors with geographic advantages could materially and adversely affect the Company’s business, financial condition and results of operations. To remain competitive, the Company believes that it must effectively and economically provide: (i) products and services that satisfy client demands, (ii) superior client service, (iii) high levels of quality and reliability, and (iv) dependable and efficient distribution networks. Increased competition may require the Company to reduce prices or increase spending on sales and marketing and client support, which may have a material adverse effect on its financial condition and results of operations. Any decrease in the quality of the Company’s products or level of service to clients or any occurrence of a price war among the Company’s competitors may adversely affect the business and results of operations. Client reach, service and on-time delivery will continue to be a hallmark of the Company’s ability to compete with other market players. Further, the acquisitions translate to spreading the Company’s footprint on two continents. In addition, the Company has deployed a sales team tasked with continually sourcing and providing market intelligence as part of its activities.
Market Perception of Smaller Companies
Market perception of smaller companies may change, potentially affecting the value of investors’ holdings and the ability of the Company to raise further funds through the issue of further common shares or otherwise. The share price of publicly traded smaller companies can be highly volatile. The value of the common shares may go down as well as up and, in particular, the share price may be subject to sudden and large falls in value given the restricted marketability of the common shares, results of operations, changes in earnings estimates or changes in general market, economic and political conditions.
Research and Development and Product Development
The Company is a life science company that makes customized antibodies and is engaged in the research and product development of new antibodies, processes, procedures and innovative approaches to antibody production. The Company has been engaged in such research and development activities for over 30 years and has had significant success. Continued investment in retaining key scientific staff, as well as an ongoing commitment in research and development activities, will continue to be a cornerstone in the Company’s development of new services, processes, and competitive advantages such as Rapid Prime, B cell Select, DeepDisplay and its methods for the production of human antibodies. The Company realizes that such research and product development activities endeavour, but cannot assure, the production of new and innovative processes, procedures or innovative approaches to antibody production or new antibodies. Furthermore, if the Company does not achieve sufficient market acceptance of its expansion of its commercialization of its products and services, it will be difficult for the Company to achieve consistent profitability. The Company’s marketing and sales approach and external sales personnel continue to introduce a steady stream of new clients.
Management of Growth
The Company may be subject to growth-related risks including pressure on its internal systems and controls. The Company’s ability to manage its growth effectively will require it to continue to implement and improve its operational and financial systems and to expand, train and manage its employee base. The inability of the Company to deal with this growth could have a material adverse impact on its business, operations and prospects. The Company may experience growth in the number of its employees and the
20
scope of its operating and financial systems, resulting in increased responsibilities for the Company’s personnel, the hiring of additional personnel and, in general, higher levels of operating expenses. In order to manage its current operations and any future growth effectively, the Company will also need to continue to implement and improve its operational, financial and management information systems and to hire, train, motivate, manage and retain its employees. There can be no assurance that the Company will be able to manage such growth effectively, that its management, personnel or systems will be adequate to support the Company’s operations or that the Company will be able to achieve the increased levels of revenue commensurate with the increased levels of operating expenses associated with this growth.
Selection and Integration of Acquired Businesses and Technologies
The Company has expanded its business through acquisitions. The Company may plan to continue to acquire businesses and technologies and form strategic alliances. However, businesses and technologies may not be available on terms and conditions the Company finds acceptable. The Company risks spending time and money investigating and negotiating with potential acquisition or alliance partners, but not completing transactions.
Acquisitions and alliances involve numerous risks which may include:
| • | difficulties in achieving business and financial success; |
| • | difficulties and expenses incurred in assimilating and integrating operations, services, products, technologies or pre-existing relationships with the Company’s clients, distributors and suppliers; |
| • | challenges with developing and operating new businesses, including those that are materially different from the Company’s existing businesses and that may require the development or acquisition of new internal capabilities and expertise; |
| • | potential losses resulting from undiscovered liabilities of acquired companies that are not covered by the indemnification the Company may obtain from the seller or the insurance acquired in connection with the transaction; |
| • | loss of key employees; |
| • | the presence or absence of adequate internal controls and/or significant fraud in the financial systems of acquired companies; |
| • | diversion of management’s attention from other business concerns; |
| • | a more expansive regulatory environment; |
| • | acquisitions could be dilutive to earnings, or in the event of acquisitions made through the issuance of the Company’s common stock to the shareholders of the acquired company, dilutive to the percentage of ownership of the Company’s existing shareholders; |
| • | differences in foreign business practices, customs and importation regulations, language and other cultural barriers in connection with the acquisition of foreign companies; |
| • | new technologies and products may be developed that cause businesses or assets the Company acquires to become less valuable; and |
| • | disagreements or disputes with prior owners of an acquired business, technology, service or product that may result in litigation expenses and diversion of the Company’s management’s attention. |
If an acquired business, technology or an alliance does not meet the Company’s expectations, its results of operations may be adversely affected.
Some of the same risks exist when the Company decides to sell a business, site or product line. In addition, divestitures could involve additional risks, including the following:
| • | difficulties in the separation of operations, services, products, and personnel; |
21
| • | diversion of management’s attention from other business concerns; and |
| • | the need to agree to retain or assume certain current or future liabilities in order to complete the divestiture. |
The Company continually evaluates the performance and strategic fit of its businesses (including specific product lines and service offerings) to determine whether any divestitures are appropriate. Any divestitures may result in significant write-offs, including those related to goodwill and other intangible assets and which could have an adverse effect on the Company’s results of operations and financial condition. In addition, the Company may encounter difficulty in finding buyers or alternative exit strategies at acceptable prices and terms, and in a timely manner. The Company may not be successful in managing these or any other significant risks that it encounters in divesting a business, site or product line or service offering and, as a result, may not achieve some or all of the expected benefits of the divestiture.
Loss of Clients
The Company’s clients may terminate their contracts with it upon 30 to 90 days’ notice for a number of reasons or, in some cases, for no reason. Although the Company’s clients are currently comprised of a number of small and larger pharma entities, the Company is making a strategic shift to increase the number of larger pharma and biotech clients, including the size of each service contract. If any one of the Company’s major clients cancels its contract with the Company, its revenue may decrease.
Reduction in Demand
The Company’s business could be adversely affected by any significant decrease in drug R&D expenditures by pharmaceutical and biotechnology companies, as well as by academic institutions, government laboratories or private foundations. Similarly, economic factors and industry trends that affect the Company’s clients in these industries also affect their R&D budgets and, consequentially, the Company’s business as well.
The Company’s clients include researchers at pharmaceutical and biotechnology companies. The Company’s ability to continue to grow and win new business is dependent in large part upon the ability and willingness of the pharmaceutical and biotechnology industries to continue to spend on molecules in the non-clinical phases of R&D and to outsource the products and services the Company provides. Furthermore, the Company’s clients (particularly larger biopharmaceutical companies) continue to search for ways to maximize the return on their investments with a focus on lowering R&D costs per drug candidate. Fluctuations in the expenditure amounts in each phase of the R&D budgets of these researchers and their organizations could have a significant effect on the demand for the Company’s products and services. R&D budgets fluctuate due to changes in available resources, mergers of pharmaceutical and biotechnology companies, spending priorities, general economic conditions, institutional budgetary policies and the impact of government regulations, including potential drug pricing legislation. Available funding for biotechnology clients in particular may be affected by the capital markets, investment objectives of venture capital investors and priorities of biopharmaceutical industry sponsors.
Reduction or Delay in Government Funding of R&D
A small portion of revenue is derived from clients at academic institutions and research laboratories whose funding is partially dependent on both the level and timing of funding from government sources in Canada, such as NRC, and the United States, such as the United States’ National Institutes of Health, and international agencies, which can be difficult to forecast. Government funding of R&D is subject to the political process, which is inherently fluid and unpredictable. The Company’s revenue may be adversely affected if its clients delay purchases as a result of uncertainties surrounding the approval of government budget proposals, included reduced allocations to government agencies that fund R&D activities. Government proposals to reduce or eliminate budgetary deficits have sometimes included reduced allocations to government agencies that fund R&D activities, or such funding may not be directed towards projects and studies that require the use of the Company’s products and services, both of which could adversely affect the Company’s business and financial results.
22
Public Company in the United States
As a public company in the United States, the Company incurs additional legal, accounting, reporting and other expenses that it did not incur as a public company solely listed in Canada. The additional demands associated with being a United States public company may disrupt regular operations of business by diverting the attention of some of the Company’s senior management team away from revenue-producing activities to additional management and administrative oversight, adversely affecting the Company’s ability to attract and complete business opportunities and increasing the difficulty in both retaining professionals and managing and growing its business. Any of these effects could harm the Company’s business, results of operations and financial condition. In general, the United States tends to be more litigious than Canada and being a public company in the United States may make it more likely that the Company is subjected, from time to time, to the types of lawsuits that affect public companies in the United States.
Delivery and Performance Requirements in Client Contracts
In order to maintain its current client relationships and to meet the performance and delivery requirements in its client contracts, the Company must be able to provide products and services at appropriate levels and with acceptable quality and at an acceptable cost. The Company’s ability to deliver the products and provide the services it offers to its clients is limited by many factors, including the difficulty of the processes associated with its products and services, the lack of predictability in the scientific process and the shortage of qualified scientific personnel. In particular, a large portion of the Company’s revenue depends on producing biologics and the current rate at which the Company is producing them. Some of the Company’s clients can influence when it will deliver products and perform services under their contracts. If the Company is unable to meet its contractual commitments, it may delay or lose revenue, lose clients or fail to expand its existing relationships.
Patent and Other Intellectual Property Litigation
The drug research and development industry has a history of patent and other intellectual property litigation and these lawsuits will likely continue. Because the Company produces and provides many different products and services in this industry, it faces potential patent infringement suits by companies that control patents for similar products and services. In order to protect or enforce the Company’s intellectual property rights, it may have to initiate legal proceedings against third parties. In addition, others may sue the Company for infringing their intellectual property rights or the Company may initiate a lawsuit seeking a declaration from a court that it does not infringe the proprietary rights of others. The patent positions of pharmaceutical, biotechnology and drug discovery companies are generally uncertain and involve complex legal and factual questions. No consistent policy has emerged from the United States Patent and Trademark Office or the courts regarding the breadth of claims allowed or the degree of protection afforded under patents like those for which the Company has applied. Legal proceedings relating to intellectual property would be expensive, take significant time and divert management’s attention from other business concerns, whether the Company wins or loses. The cost of such litigation could affect the Company’s profitability.
Further, if the Company does not prevail in an infringement lawsuit brought against it, the Company might have to pay substantial damages, including treble damages, and it could be required to stop the infringing activity or obtain a license to use the patented technology. Any required license may not be available to the Company on acceptable terms, or at all. In addition, some licenses may be nonexclusive, and therefore, the Company’s competitors may have access to the same technology licensed to the Company. If the Company fails to obtain a required license or is unable to design around a patent, it may be unable to sell some of its products or services.
Key Personnel Risk
The Company’s success will depend on its directors’ and officers’ ability to develop the Company’s business and manage its operations, and on the Company’s ability to attract and retain the Chief Executive Officer, management team and other key technical, sales, public relations and marketing staff or consultants to operate and grow the business. The loss of any key person or the inability to find and retain new key
23
persons could have a material adverse effect on the Company’s business. Competition for experienced scientists is intense. The Company competes with pharmaceutical and biotechnology companies, including its clients and collaborators, medicinal chemistry outsourcing companies, contract research companies, and academic and research institutions to recruit scientists. The Company’s inability to hire additional qualified personnel may also require an increase in the workload for both existing and new personnel. The Company may not be successful in attracting new scientists or management or in retaining or motivating its existing personnel. The shortage of experienced scientists, and other factors, may lead to increased recruiting, relocation and compensation costs for such scientists, which may exceed the Company’s expectations. These increased costs may reduce the Company’s profit margins or make hiring new scientists impracticable.
Pandemic Risk
The Company is currently unable to determine whether the ongoing COVID-19 pandemic will have a negative effect on the Company’s results for the duration of the outbreak. There has been minimal impact on the Company’s operations and results to date, and the Company has not experienced negative impact on client sales or the supply chain. The Company’s sales, operations and financial performance could suffer given a potential rapidly spreading virus. Internally, the virus may infect its employees resulting in operating at lower productivity levels or even a complete laboratory shutdown. The Company’s business is dependent on its laboratories to produce its products and services which if not operating will impact the financial performance of the company and its ability to meet its obligations. The Company has diversified geographic locations with the ability to perform similar services at other sites. In addition, certain roles have the ability to work remotely and the Company has business interruption insurance which may aid in the recovery of lost profits. External factors may also contribute to this risk, such as the impact of a pandemic on the Company’s clients and suppliers.
Brand Awareness
The Company’s expansion of its products and services depends on increasing brand awareness with respect to its products and services. There is no assurance that the Company will be able to achieve sufficient brand awareness. In addition, the Company must successfully develop a larger market for its services in order to increase the sales of its services. If the Company is not able to successfully develop a market for its services, then such failure will have a material adverse effect on the business, financial condition and operating results of the Company.
Conflicts of Interest Risk
Certain of the Company’s directors and officers are also involved as advisors for other companies. Situations may arise in connection with potential acquisitions or opportunities where the other interests of these directors and officers conflict with or diverge from the Company’s interests. In accordance with the BCBCA, directors who have a material interest in any person who is a party to a material contract or a proposed material contract are required, subject to certain exceptions, to disclose that interest and generally abstain from voting on any resolution to approve the contract.
In addition, the directors and the officers are required to act honestly and in good faith with a view to the Company’s best interests. However, in conflict of interest situations, the Company’s directors and officers may owe the same duty to another company and will need to balance their competing interests with their duties to the Company. Circumstances (including with respect to future corporate opportunities) may arise that may be resolved in a manner that is unfavourable to the Company.
Outsourcing Trend in Non-Clinical Discovery Stages of Drug Discovery
Over the past decade, pharmaceutical and biotechnology companies have generally increased their outsourcing of non-clinical research support activities, such as antibody discovery. While many industry analysts expect the outsourcing trend to continue to increase for the next several years (although with different growth rates for different phases of drug discovery and development), decreases in such outsourcing may result in a diminished growth rate in the sales of any one or more of the Company’s service lines and may adversely affect the Company’s financial condition and results of operations.
24
Competition and Obsolescence
The pharmaceutical and biotechnology industries are characterized by rapid and continuous technological innovation. The Company competes with companies around the world that are engaged in the development and production of products and services, including pharmaceutical companies, biotechnology companies, and contract research companies. Academic institutions, governmental agencies and other research organizations also are conducting research and developing technologies in areas in which the Company provides services, either on its own or through collaborative efforts. The Company’s pharmaceutical and biotechnology company clients have internal departments that provide products and services that directly compete with the Company’s products and services. Many of the Company’s competitors offer a broader range of products and services and have greater access to financial, technical, scientific, business development, recruiting and other resources than the Company does, and some of its competitors may also operate with a lower cost structure. The Company anticipates that it will face increased competition in the future as it expands its operations and its products and services and as new companies enter the market and advanced technologies become available. The Company’s products, services and expertise may become obsolete or uneconomical due to technological advances or entirely different approaches developed by the Company, its clients or one or more of its competitors. For example, advances in databases and molecular modeling tools that predict how effectively compounds will treat a targeted disease may render some of its technologies obsolete. While the Company plans to develop technologies that will give it a competitive advantage, it may not be able to develop the technologies necessary for it to successfully compete in the future. Additionally, the existing approaches of the Company’s competitors or new approaches or technologies developed by its competitors may be more effective than those it develops. The Company may not be able to compete successfully with existing or future competitors.
Other competitive factors could force the Company to lower prices or could result in reduced sales. In addition, new products developed by others could emerge as competitors to the Company’s drug candidates. If the Company is not able to compete effectively against current and future competitors, its business will not grow and its financial condition and operations will suffer.
Global Economic Conditions
Current global economic conditions could have a negative effect on the Company’s business and results of operations. Market disruptions have included extreme volatility in securities prices, as well as severely diminished liquidity and credit availability. The economic crisis may adversely affect the Company in a variety of ways. Access to lines of credit or the capital markets may be severely restricted, which may preclude the Company from raising funds required for operations and to fund continued development. It may be more difficult for the Company to complete strategic transactions with third parties. The financial and credit market turmoil could also negatively impact suppliers, clients and banks with whom the Company does business. Such developments could decrease the Company’s ability to source, produce and distribute its products or obtain financing and could expose it to risk that one of its suppliers, clients or banks will be unable to meet their obligations under agreements with the Company.
Limited Number of Suppliers
The Company currently purchases animals and certain key components of biological and chemical materials that it uses in its products and services from a limited number of outside sources. The Company’s reliance on its suppliers exposes it to risks, including: (i) the possibility that one or more of its suppliers could terminate their services at any time without penalty; (ii) the potential inability of its suppliers to obtain required materials; (iii) the potential delays and expenses of seeking alternative sources of supply; and (iv) reduced control over pricing, quality and timely delivery due to the difficulties in switching to alternative suppliers.
25
Consequently, if materials from the Company’s suppliers are delayed or interrupted for any reason, the Company may not be able to deliver its products and perform its services on a timely basis or in a cost-efficient manner.
Uninsured or Uninsurable Risk
The Company may become subject to liability for risks against which it cannot insure or against which the Company may elect not to insure due to the high cost of insurance premiums or other factors. The payment of any such liabilities would reduce the funds available for the Company’s usual business activities. Payment of liabilities for which the Company does not carry insurance may have a material adverse effect on the Company’s financial position and operations.
Restricted Use of Scientific Information
The Company’s ability to improve the efficiency of the CRO services it provides by, among other things, developing an effective database designed to predict how chemical compounds interact with a targeted disease-related protein, depends in part on the Company’s generation and use of information that is not proprietary to its clients and that it derives from performing these services. However, the Company’s clients may not allow it to use this information with other clients, such as the general interaction between types of chemistries and types of drug targets that the Company generates when performing drug discovery services for its clients. Without the ability to use this information, the Company may not be able to develop a database, which may limit its ability to improve the efficiency of the drug discovery services it provides.
Failure of Laboratory Facilities
The Company’s operations could suffer as a result of a failure of its laboratory facilities. The Company’s business will be dependent upon a laboratory infrastructure to produce products and services. These systems and operations are vulnerable to damage and interruption from fires, earthquakes, telecommunications failures, and other events. Any such errors or inadequacies in the software that may be encountered could adversely affect operations, and such errors may be expensive or difficult to correct in a timely manner.
Further, many of the Company’s operations are comprised of complex mechanical systems that are subject to periodic failure, including aging fatigue. Such failures are unpredictable, and while the Company has made significant capital expenditures designed to create redundancy within these mechanical systems, strengthened biosecurity, improved operating procedures to protect against contaminations, and replaced impaired systems and equipment in advance of such events, failures and/or contaminations may still occur.
The production of monoclonal and polyclonal antibodies requires state of the art laboratory facilities and the success of these laboratory services depends on the recruitment and retention of highly qualified technical staff to maintain the level and quality of standard of the Company’s products and services expected from clients. There is no assurance that the Company will be able to expand and operate such state of the art laboratory services and recruit and retain qualified staff.
The Company produces and supplies antibodies and there is no guarantee that such production will be successful and produce the desired results. As a result, the Company continues to be exposed to potential liability that may exceed any insurance coverage that the Company may obtain in the future. As a result, the Company may incur significant liability exposure, which may exceed any insurance coverage that the Company may obtain in the future. Even if the Company elects to purchase such insurance in the future, the Company may not be able to maintain adequate levels of insurance at reasonable cost and/or on reasonable terms. Excessive insurance costs or uninsured claims may increase the Company’s operating loss and affect its financial condition.
26
Contaminations in Animal Populations
Animals that the Company uses must be free of certain infectious agents, such as certain viruses and bacteria, because the presence of these contaminants can distort or compromise the quality of research results and could adversely impact animal health. The presence of these infectious agents in the Company’s animal facility and certain service operations could disrupt the Company’s animal service businesses, harm the Company’s reputation and result in decreased sales.
Contaminations are unanticipated and difficult to predict and could adversely impact the Company’s financial results. If they occur, contaminations typically require cleaning up, renovating, disinfecting, retesting and restarting production or services. Such clean-ups result in inventory loss, clean-up and start-up costs, and reduced sales as a result of lost client orders and potentially credits for prior shipments. Contaminations also expose the Company to risks that clients will request compensation for damages in excess of the Company’s contractual indemnification requirements.
Unauthorized Access into Information Systems
The Company operates large and complex information systems that contain significant amounts of client data. As a routine element of the Company’s business, the Company collects, analyzes and retains substantial amounts of data pertaining to the non-clinical research it conducts for its clients. Unauthorized third parties could attempt to gain entry to such information systems to steal data or disrupt the systems. The Company has taken measures to protect them from intrusion.
The Company’s contracts with its clients typically contain provisions that require the Company to keep confidential the information generated from the research conducted. In the event the confidentiality of such information is compromised, whether by unauthorized access or other breaches, the Company could be exposed to significant harm, including termination of customer contracts, damage to its customer relationships, damage to its reputation and potential legal claims from customers, employees and other parties. In addition, the Company may face investigations by government regulators and agencies as a result of a breach.
Further, the Company is required to comply with data privacy and security laws in many jurisdictions. For example, the Company is required to comply with the European Union General Data Protection Regulation (“GDPR”), which became effective on May 25, 2018 and imposes heightened obligations and enhanced penalties for noncompliance (including up to four percent (4%) of global revenue). The cost of compliance, and the potential for fines and penalties for non-compliance, with GDPR may have a significant adverse effect on the Company’s business and operations. Also, the California legislature passed the California Consumer Privacy Act (“CCPA”), which became effective January 1, 2020. The CCPA creates new transparency requirements and grants California residents several new rights with regard to their personal information. Failure to comply with the CCPA may result in, among other things, significant civil penalties and injunctive relief, or potential statutory or actual damages. The Company has made changes to, and investments in, its business practices and will continue to monitor developments and make appropriate changes to help attain compliance with these evolving and complex regulations.
Enforcement of Civil Liabilities
The Company is organized under the laws of the Province of British Columbia, with its registered place of business in Canada, some of its directors and officers reside outside the United States and the majority of the Company’s assets and all or a substantial portion of the assets of these persons may be located outside the United States. Consequently, it may be difficult for investors who reside in the United States to effect service of process in the United States upon the Company or upon such persons who are not residents of the United States, or to realize upon judgments of courts of the United States predicated upon the civil liability provisions of the United States federal securities laws.
27
Foreign Private Issuer
The Company is a “foreign private issuer” as such term is defined in Rule 405 under the United States Securities Act of 1933, as amended (“U.S. Securities Act”) and is permitted, under a the MJDS, to prepare its disclosure documents filed under the United States Securities Exchange Act of 1934 (“U.S. Exchange Act”) in accordance with Canadian disclosure requirements. Under the U.S. Exchange Act, the Company is subject to reporting obligations that, in certain respects, are less detailed and less frequent than those of United States domestic reporting companies. As a result, the Company does not file the same reports that a United States domestic issuer files with the SEC, although it is required to file or furnish to the SEC the continuous disclosure documents that it is required to file in Canada under Canadian securities laws. In addition, the officers, directors, and principal shareholders of the Company are exempt from the reporting and “short swing” profit recovery provisions of Section 16 of the U.S. Exchange Act. Therefore, the Company’s shareholders may not know on as timely a basis when the officers, directors and principal shareholders of the Company purchase or sell shares, as the reporting thresholds and deadlines under the corresponding Canadian insider reporting requirements differ.
As a foreign private issuer, the Company is exempt from the rules and regulations under the U.S. Exchange Act related to the furnishing and content of proxy statements. It is also exempt from Regulation FD, which prohibits issuers from making selective disclosures of material non-public information. While the Company expects to comply with the corresponding requirements relating to proxy statements and disclosure of material non-public information under Canadian securities laws, these requirements differ from those under the U.S. Exchange Act and Regulation FD and shareholders should not expect to receive in every case the same information at the same time as such information is provided by United States domestic companies. In addition, as a foreign private issuer, the Company has the option to follow certain Canadian corporate governance practices, provided that the Company discloses the requirements that are not being followed and describes the Canadian practices being followed instead. The Company plans to rely on this exemption. As a result, the Company’s shareholders may not have the same protections afforded to shareholders of Unites States domestic companies that are subject to all Unites States corporate governance requirements.
Loss of Foreign Private Issuer Status
The Company may lose its status as a foreign private issuer if, as of the last business day of the Company’s second fiscal quarter for any year, more than 50% of the Company’s outstanding voting securities (as determined under Rule 405 of the U.S. Securities Act) are directly or indirectly held of record by residents of the United States. The regulatory and compliance costs under U.S. federal securities laws as a U.S. domestic issuer may be significantly more than the costs incurred as a Canadian foreign private issuer eligible to use the MJDS. If the Company is not a foreign private issuer, it would not be eligible to use the MJDS or other foreign issuer forms and would be required to file periodic and current reports and registration statements on U.S. domestic issuer forms with the SEC, which are more detailed and extensive than the forms available to a foreign private issuer. These increased costs may have a material adverse effect on the business, financial condition or results of operations of the Company.
“Emerging Growth Company” Status
The Company is an “emerging growth company” as defined in section 3(a) of the U.S. Exchange Act (as amended by the JOBS Act, enacted on April 5, 2012), and the Company will continue to qualify as an emerging growth company until the earliest to occur of: (a) the last day of the fiscal year during which the Company has total annual gross revenues of US$1,070,000,000 (as such amount is indexed for inflation every five years by the SEC) or more; (b) the last day of the fiscal year of the Company following the fifth anniversary of the date of the first sale of common equity securities of the Company pursuant to an effective registration statement under the U.S. Securities Act; (c) the date on which the Company has, during the previous three year period, issued more than US$1,000,000,000 in non-convertible debt; and (d) the date on which the Company is deemed to be a “large accelerated filer”, as defined in Rule 12b-2 under the U.S. Exchange Act. The Company will qualify as a large accelerated filer (and would cease to be an emerging growth company) at such time when on the last business day of its second fiscal quarter of such year the aggregate worldwide market value of its common equity held by non-affiliates will be US$700,000,000 or more.
28
For so long as the Company remains an emerging growth company, it is permitted to and intends to rely upon exemptions from certain disclosure requirements that are applicable to other public companies that are not emerging growth companies. These exemptions include not being required to comply with the auditor attestation requirements of Section 404 of the JOBS Act. The Company takes advantage of some, but not all, of the available exemptions available to emerging growth companies. The Company cannot predict whether investors will find the Common Shares less attractive because the Company relies upon certain of these exemptions. If some investors find the Common Shares less attractive as a result, there may be a less active trading market for the Common Shares and the Common Share price may be more volatile. On the other hand, if the Company no longer qualifies as an emerging growth company, the Company would be required to divert additional management time and attention from the Company’s development and other business activities and incur increased legal and financial costs to comply with the additional associated reporting requirements, which could negatively impact the Company’s business, financial condition and results of operations.
Foreign Exchange Rates
The Company may conduct business with clients, distributors, suppliers, other service providers and affiliates in currencies other than Canadian Dollars. Therefore, the Company’s business could be adversely affected by fluctuations in domestic or foreign currencies.
Effects of Future Sales or Issuances of Equity Securities or Debt Securities
The Company may sell additional equity securities in subsequent offerings (including through the sale of securities convertible into equity securities) and may issue additional equity securities to finance operations, acquisitions or other projects. The Company cannot predict the size of future issuances of equity securities or the size and terms of future issuances of debt instruments or other securities convertible into equity securities or the effect, if any, that future issuances and sales of securities will have on the market price of the common shares. Any transaction involving the issuance of previously authorized but unissued common shares, or securities convertible into common shares, would result in dilution, possibly substantial, to securityholders. Exercises of presently outstanding share options may also result in dilution to securityholders.
The Company’s board of directors has the authority to authorize certain offers and sales of additional securities without the vote of, or prior notice to, shareholders. Based on the need for additional capital to fund expected expenditures and growth, the Company expects that it will issue additional securities to provide such capital. Such additional issuances may involve the issuance of a significant number of common shares at prices less than the current market price for the common shares.
Sales of substantial amounts of securities, or the availability of such securities for sale, could adversely affect the prevailing market prices for securities and dilute investors’ earnings per share. A decline in the market prices of the securities could impair the Company’s ability to raise additional capital through the sale of securities should the Company desire to do so. Sales of common shares by shareholders might also make it more difficult for the Company to sell equity securities at a time and price that it may deem to be appropriate.
Common Share Price Volatility
An investment in the Company’s securities is highly speculative. The market prices for the securities of pharmaceutical companies, including the Company’s, have historically been highly volatile. The market has from time to time experienced significant price and volume fluctuations that are unrelated to the financial performance or prospects of any particular company. In addition, because of the nature of the Company’s business, certain factors such as announcements, competition from new therapeutic products or technological innovations, governmental regulations, fluctuations in operating results, results of clinical trials, public concern regarding the safety of drugs generally, general market conditions, developments in patent and proprietary rights, the Company’s financial condition or results of operations as reflected in its
29
quarterly and annual financial statements, operating performance and the performance of competitors and other similar companies, changes in earnings estimates or recommendations by research analysts who track the Company’s securities or securities of other companies in the life sciences sector, general market conditions, announcements relating to litigation, the arrival or departure of key personnel and the factors listed under the heading “Risk Factors” can have an adverse impact on the market price of the common shares.
Any negative change in the public’s perception of the Company’s prospects could cause the price of the Company’s securities, including the price of the common shares, to decrease dramatically. Furthermore, any negative change in the public’s perception of the prospects of life sciences companies in general could depress the price of the Company’s securities, including the price of the common shares, regardless of the Company’s financial and operating results. In the past, following declines in the market price of a company’s securities, securities class-action litigation often has been instituted against said company. Litigation of this type, if instituted, could result in substantial costs and a diversion of the Company’s management’s attention and resources.
Discretion in Use of Proceeds
The Company will have broad discretion over the use of proceeds from an offering by the Company of its securities. Because of the number and variability of factors that will determine the Company’s use of such proceeds, the Company’s ultimate use might vary substantially from any planned use disclosed by the Company. Investors and securityholders may not agree with how the Company allocates or spends the proceeds from an offering of securities. The Company may pursue acquisitions, collaborations or other opportunities that do not result in an increase in the market value of its securities, including the market value of the common shares, and that may increase its losses.
Dividend Policy
No dividends on the common shares have been paid by the Company to date. The Company does not intend to declare or pay any cash dividends in the foreseeable future. Payment of any future dividends will be at the discretion of the Board, after taking into account a multitude of factors appropriate in the circumstances, including the Company’s operating results, financial condition and current and anticipated cash needs.
Liquid Market for Common Shares
Shareholders of the Company may be unable to sell significant quantities of common shares into the public trading markets without a significant reduction in the price of their common shares, or at all. There can be no assurance that there will be sufficient liquidity of the Company’s common shares on the trading market, and that the Company will continue to meet the listing requirements of the TSXV or Nasdaq.
The Company’s common shares are newly listed on Nasdaq. If an active trading market does not develop in the United States, investors may have difficulty selling any of the common shares that they buy over a United States exchange. The Company cannot predict the extent to which investor interest in the Company will lead to the development of an active trading market on the Nasdaq or otherwise, or how liquid that market might become. Listing of the common shares on the Nasdaq in addition to the TSXV may increase price volatility on the TSXV and also result in volatility of the trading price on the Nasdaq because trading will be in two markets, which may result in less liquidity on both exchanges. In addition, different liquidity levels, volumes of trading, currencies and market conditions on the two exchanges may result in different prevailing trading prices.
The Company has not paid any dividends. The Company intends to retain its earnings, if any, to finance the future growth and development of its business and does not expect to pay dividends or to make any other distributions in the foreseeable future. Payment of dividends in the future is dependent upon the earnings and financial condition of the Company and other factors which the Board may deem appropriate at the time.
30
There are no restrictions in the constating documents of the Company, and it is not currently expected that there will exist such restriction elsewhere, which could prevent the Company from paying dividends.
DESCRIPTION OF CAPITAL STRUCTURE
Common Shares
The Company’s authorized share capital consists of an unlimited number of common shares. As at the date of this Annual Information Form, 24,836,723 common shares are issued and outstanding.
Registered holders of common shares are entitled to receive notice of and attend all meetings of shareholders of the Company, and are entitled to one vote for each Common Share held at a meeting of shareholders other than meetings at which only the holders of any other class or series of shares of the Company may be issued or outstanding from time to time or are entitled to vote as a separate class or series. In addition, holders of common shares are entitled to receive on a pro rata basis dividends if, as and when declared by the board of directors and, upon liquidation, dissolution or winding-up of the Company, are entitled to receive on a pro rata basis the net assets of the Company after payment of debts and other liabilities, in each case subject to the rights, privileges, restrictions and conditions attaching to any other series or class of shares, including preferred shares, ranking in priority to, or equal with, the holders of the common shares.
Trading Price and Volume
The common shares of the Company are listed for trading on the TSXV and Nasdaq under the symbol “IPA”. The following tables set out the market price range and trading volumes of the common shares on the TSXV and Nasdaq for the periods indicated.
TSXV Price Range ($)
Month and Year | High ($) | Low ($) | Volume (number of shares) | |||||||||
May 2021 | 11.360 | 8.180 | 741,913 | |||||||||
June 2021 | 9.200 | 7.400 | 609,101 | |||||||||
July 2021 | 16.560 | 6.580 | 4,605,758 | |||||||||
August 2021 | 10.500 | 8.180 | 1,231,898 | |||||||||
September 2021 | 9.230 | 7.880 | 684,250 | |||||||||
October 2021 | 8.190 | 7.290 | 517,160 | |||||||||
November 2021 | 8.470 | 6.740 | 608,666 | |||||||||
December 2021 | 7.580 | 6.510 | 433,035 | |||||||||
January 2022 | 8.300 | 6.750 | 474,862 | |||||||||
February 2022 | 7.740 | 5.950 | 494,703 | |||||||||
March 2022 | 6.400 | 4.670 | 1,044,284 | |||||||||
April 2022 | 7.590 | 6.510 | 392,932 | |||||||||
NASDAQ Price Range (US$)
Month and Year | High ($) | Low ($) | Volume (number of shares) | |||||||||
May 2021 | 9.230 | 6.880 | 1,138,165 | |||||||||
June 2021 | 7.670 | 6.020 | 1,142,160 | |||||||||
July 2021 | 13.220 | 5.140 | 139,793,087 | |||||||||
August 2021 | 8.340 | 6.430 | 7,665,205 | |||||||||
31
September 2021 | 7.310 | 6.190 | 1,850,405 | |||||||||
October 2021 | 6.690 | 5.850 | 1,659,722 | |||||||||
November 2021 | 6.420 | 5.350 | 1,297,498 | |||||||||
December 2021 | 5.860 | 5.060 | 1,021,055 | |||||||||
January 2022 | 6.490 | 5.350 | 754,449 | |||||||||
February 2022 | 6.120 | 4.670 | 640,935 | |||||||||
March 2022 | 5.140 | 3.640 | 870,858 | |||||||||
April 2022 | 6.080 | 5.190 | 669,481 |
The following table summarizes the issuance of unlisted securities of the Company during the 12-month period preceding April 30, 2022.
Date Issued | Type of Security Issued | Number of Securities Issued | Issuance / Exercise Price Per Security | |||||||
May 9, 2021 | Options | 10,000 | $ | 7.72 | (1) | |||||
June 13, 2021 | Options | 43,750 | $ | 7.14 | (1)(2) | |||||
August 7, 2021 | Options | 45,000 | $ | 9.19 | ||||||
January 2, 2022 | Options | 28,250 | $ | 6.89 | ||||||
January 7, 2022 | Options | 338,000 | $ | 7.94 | ||||||
January 13, 2022 | Options | 39,000 | $ | 8.30 | ||||||
March 11, 2022 | Options | 15,000 | $ | 6.35 | ||||||
Notes:
| (1) | Priced in USD. |
| (2) | Options were expired as of April 30, 2022 |
As at April 30, 2022, a total of 4,077,774 common shares of the Company were held in escrow, representing 16.7% of the common shares issued and outstanding. The common shares are held in escrow by Computershare Trust Company of Canada, and will be released over a three year period as per the terms of the BioStrand Share Purchase Agreement.
DIRECTORS AND EXECUTIVE OFFICERS
Directors
The directors of the Company are set forth below. Each director’s current term of office will expire at the next annual general meeting of the Company’s shareholders or when their successors are duly elected or appointed in accordance with the Company’s articles of incorporation or upon such director’s earlier death, resignation or removal.
32
Name and | Principal Occupation During Past Five Years | Director Since | Number of Voting | Percent of Voting | ||||
Jennifer Bath Minnesota, USA | CEO, President of the Company since February 2018; Global Director of Aldevron, LLC, a company specializing in the manufacturing of biologics, from July 2015 to February 2018. | May 2018 | 85,858 (Direct) | * | ||||
James Kuo(2) California, USA | Chairman of the Company since December 2016; Managing Director at Athena Bioventures, a venture capital firm investing in breakthrough medicine from 2007 to present. | December 2016 | 25,000 (Direct) | * | ||||
Greg Smith(2)(3) British Columbia, Canada | President & Owner of Broadway Refrigeration & Air Conditioning Co. Ltd., a company specialized in HVAC-R mechanical and maintenance services; Director of Atlas Engineered Products Inc. (TSXV: AEP), a company specialized in the design and manufacturing of trusses and engineered wood products through seven plants across Canada. Formerly, Chairman and Interim CEO of Lite Access Technologies (TSXV: LTE), a company specialized in micro/narrow trenching technologies and specialist fibre-optic products; | September 2016 | 35,000(6) (Direct and Indirect) | * | ||||
Robert Burke(2)(4) British Columbia, Canada | Professor at the University of Victoria. | December 2017 | 38,600(7) (Direct and Indirect) | * | ||||
Anna Pettersson(3)(4) New York, USA | Director, Client Services with Genioo, LLC and Owner / Founder of Barnegat Advisors, LLC since 2021; Senior director at Pfizer, Inc. from January 2012 to March 2021. | November 2021 | Nil | Nil | ||||
Notes:
| * | Denotes less than 1% of the issued and outstanding common shares. |
33
| (1) | The information as to the nature of common shares beneficially owned, or controlled or directed, directly or indirectly, by the directors, not being within the knowledge of the Company, has been furnished by such directors. |
| (2) | Member of the Audit Committee. |
| (3) | Member of the Compensation Committee. |
| (4) | Member of the Governance & Nominating Committee. |
| (5) | Of the 35,000 common shares, 34,000 common shares are held by Mr. Smith and 1,000 common shares are held by the spouse of Mr. Smith. |
| (6) | Of the 38,600 common shares, 30,000 common shares are held by Mr. Burke and 8,600 common shares are held by the spouse of Mr. Burke. |
| (7) |
Executive Officers
The executive officers of the Company are set forth below:
Name and Municipality of Residence | Principal Occupation During Past Five Years | Officer Since | Number of | Percent of | ||||
Jennifer Bath Minnesota, USA | As Above. | February 2018 | As Above | As Above | ||||
Lisa Helbling North Dakota, USA | Chief Financial Officer of the Company since January 2019; CFO of Anchor Ingredients, a specialty ingredients supplier to food and pet food industries worldwide, from January to August 2018; and CFO and Treasurer of TMI Hospitality, an operator and developer of hotels, from December 2011 to December 2017. | January 2019 | 2,000 (Direct) | * | ||||
Stefan Lang Freiburg, Germany | Chief Business Officer of the Company since October 2019 to April 28, 2022; Vice President of Business Development of Aldevron LLC, a company specialized in the manufacturing of biologics. | October 2019 | 2,000 (Direct) | * | ||||
Ilse Roodink Loenen, the Netherlands | Chief Scientific Officer of the Company since July 1, 2021; Chair of Talem’s Scientific Advisory Committee since 2019; since 2013 with ModiQuest Research and later at IPA Europe with her last role as Scientific Director | July 2021 | 942 (Direct) | Nil | ||||
Note:
| * | Denotes less than 1% of the issued and outstanding common shares. |
| (1) | The information as to the nature of common shares beneficially owned, or controlled or directed, directly or indirectly, by the executive officers, not being within the knowledge of the Company, has been furnished by such officers. |
34
Shareholdings of Directors and Executive Officers
As at the date of this Annual Information Form, the directors and executive officers of the Company, as a group, beneficially owned, or controlled or directed, directly or indirectly, 1,316,234 common shares, representing approximately 6.83% of the issued and outstanding common shares of the Company.
Biographical Information
The following is a brief description of each of the executive officers and directors of the Company (including details with regard to their principal occupations for the last five years).
Jennifer Bath, Chief Executive Officer, President and Director
Dr. Jennifer Bath has twenty years’ experience in biopharma industry, previously serving on the executive team at Aldevron, LLC. Prior, she was the executive director of the Global Vaccine Institute and has served as an international advisor with an experienced and proven leader in strategic planning and corporate growth as well as converting pharma companies’ scientific challenges into operational solutions. Dr. Bath specializes in strategic growth, business operations alignment, and value creation. She holds a Ph.D. in Cellular and Molecular biology from North Dakota State University.
James Kuo, MD, MBA, Chairman and Director
Dr. James (Jim) Kuo currently serves as Managing Director of Athena Bioventures in La Jolla, CA. He is an experienced biotech industry executive and investor, who brings financial and management experience to the Company. During his career, he has held executive positions in private as well as listed bioscience companies in the US, Canada, and in Europe. He previously served as CEO of Tryp Therapeutics, Synthetic Biologics, BioMicro Systems, and Discovery Laboratories. Prior to that, Dr. Kuo was Associate Director in Corporate Licensing and Development at Pfizer and Managing Director of HealthCare Ventures, a $378 million venture fund. He received his MD from the University of Pennsylvania School of Medicine and his MBA from the Wharton School of Business. Dr. Kuo’s undergraduate education is in molecular biology and music history from Haverford College.
Greg Smith, Director
Mr. Greg Smith is a seasoned capital markets veteran who held senior positions in investment banking and institutional fixed income portfolio management before transitioning to private equity with the acquisition of one of the largest HVAC companies in Western Canada. Mr. Smith held the position of Portfolio Manager for Phillips, Hagar & North & Executive Director, Canadian Securitization Group, CIBC World Markets in Toronto for close to ten years. Mr. Smith currently serves as President & Director of Broadway Refrigeration & Air Conditioning Co. Ltd. Mr. Smith earned an MBA from Dalhousie University, is a Chartered Financial Analyst and serves in advisory and board positions to multiple private and public ventures.
35
Robert Burke, Director
Dr. Robert D. Burke is an Emeritus Professor at the University of Victoria, where he was a faculty member for over 35 years. He has a longstanding research interest in the molecular basis of cellular signaling in early embryonic development. His research involves production and characterization of antibodies and he employs them extensively with high-resolution optical imaging methods. Dr. Burke has published over 100 peer-reviewed publications and has supervised numerous trainees. He was Chair of the Department of Biochemistry and Microbiology for 8 years, was on the University of Victoria Senate for 12 years, and served on numerous advisory and management committees nationally and internationally. Dr. Burke completed a BSc (Honours) and a PhD at the University of Alberta.
Anna Pettersson
Dr. Anna Pettersson is a Director, Client Service with Genioo, LLC and Owner / Founder of Barnegat Advisors, LLC. She has deep knowledge of, and experience from both the Life Sciences industry and strategy consulting. A previous Senior Director at Pfizer, Inc. and Principal at Booz & Company, she has over two decades of experience serving as a strategic advisor to top life science companies in a both internal and external capacity, with a particular emphasis on innovative therapeutics R&D. She has a longstanding interest in biopharmaceutical drug discovery and development strategy, and has led public policy efforts on behalf of Pfizer, Inc., in areas including, but not limited to Precision Medicine, Gene Therapy and Rare Diseases. Dr. Pettersson holds a BS in Biology from the University of Uppsala, Sweden, and a PhD in Genetics and Diabetology from the Karolinska Institute in Stockholm, Sweden.
Lisa Helbling, Chief Financial Officer
Ms. Lisa Helbling has 37 years experience in finance and accounting gained from diverse industries and roles. For the past 11 years, she served in the role of CFO, most recently for the Company. Prior to being a CFO, she was the VP of Internal Audit and Business Risk Management for Otter Tail Corporation (NASDAQ: OTTR) a diversified electric utility and Controller for Clarica Life Insurance Company-U.S., a subsidiary of a Clarica Life Insurance Co., then listed on the Toronto Stock Exchange. She began her career in public accounting. As the Company’s CFO Ms. Helbling provides strategic vision and leadership to create and execute finance strategy to support the Company’s global sites, management of debt and equity, financial planning, budgeting and cash management. Ms. Helbling also develops and oversees the accounting, financial reporting, financial internal controls, risk management, information technology and compliance activities. Ms. Helbling currently serves on the Board of Directors for Healthy Dakota Mutual Holdings and is Chair of the Audit and Compliance Committee and serves on the Board of Directors for Border States Industries, Inc. and is Chair of the Audit Committee. Ms. Helbling is a Certified Public Accountant and has a Bachelor of Science in Accounting.
Stefan Lang, Chief Business Officer (until April 28th, 2022)
Dr. Stefan Lang previously served as the Vice President of Business Development at Aldevron LLC. Dr. Lang brings extensive background knowledge in the therapeutic antibody sector including corporate strategy, R&D innovation, sales and business development. He has an impressive breadth of leadership within the biotech industry, including experience working at the organizational level and as a globally recognized and respected leader in antibody business development. Dr. Lang holds a Dr. rer. nat. in biology from the Technical University of Karlsruhe, Germany a diploma in biology from the University of Kassel, Germany. He started his career as a technical consultant and moved into the biotech industry in 2000.
Ilse Roodink, Chief Scientific Officer
Dr. Ilse Roodink serves as Chief Scientific Officer (CSO) of the Company from July 1, 2021 on, to support the Company’s global research and development teams. Prior to her appointment as CSO, she held different scientific positions at the Company’s Dutch facility in Oss from 2013 until 2021. In her last role as Scientific Director of IPA Europe, Dr. Roodink was overseeing contract research project execution and management and actively involved in the integration of innovative technologies supporting antibody characterization and engineering. Following its establishment in 2019, Dr. Roodink has served as
36
Chairwoman of Talem Therapeutics’ Scientific Advisory Committee, leading the development of Talem’s pipeline assets. Dr. Roodink graduated from Radboud University of Nijmegen, the Netherlands with a Master’s degree in Biomedical Health Sciences and a Ph.D. in Medical Sciences. Her work, resulting in several peer-reviewed publications, focused on platform development to facilitate the discovery of antibodies specifically recognizing native tumor targets.
Cease Trade Orders or Bankruptcies
To the knowledge of the Company:
| (a) | no director or executive officer of the Company is, as at the date of this Annual Information Form, or was within 10 years before the date of this Annual Information Form, a director, chief executive officer or chief financial officer of any company (including IPA), that: |
| (i) | was subject to an order that was issued while the director or executive officer was acting in the capacity as director, chief executive officer or chief financial officer; or |
| (ii) | was subject to an order that was issued after the director or executive officer ceased to be a director, chief executive officer or chief financial officer and which resulted from an event that occurred while that person was acting in the capacity as director, chief executive officer or chief financial officer. |
For the purposes of this subsection (a), “order” means a cease trade order, an order similar to a cease trade order or an order that denied the relevant company access to any exemption under securities legislation, and in each case that was in effect for a period of more than 30 consecutive days.
| (b) | no director or executive officer of the Company, or a shareholder holding a sufficient number of securities of the Company to affect materially the control of the Company: |
| (i) | is, as at the date of this Annual Information Form, or has been within the 10 years before the date of this Annual Information Form, a director or executive officer of any company (including IPA) that, while that person was acting in that capacity, or within a year of that person ceasing to act in that capacity, become bankrupt, made a proposal under any legislation relating to bankruptcy or insolvency or was subject to or instituted any proceedings, arrangement or compromise with creditors or had a receiver, receiver manager or trustee appointed to hold its assets; or |
| (ii) | has, within the 10 years before the date of this Annual Information Form, become bankrupt, made a proposal under any legislation relating to bankruptcy or insolvency, or was subject to or instituted any proceedings, arrangement or compromise with creditors, or had a receiver, receiver manager or trustee appointed to hold the assets of the director, executive officer or shareholder. |
The foregoing information, not being within the knowledge of the Company, has been furnished by the respective directors, officers and shareholders holding a sufficient number of securities of the Company to affect materially control of the Company.
Penalties or Sanctions
No director or executive officer of the Company, or a shareholder holding a sufficient number of securities of the Company to affect materially the control of the Company, has been subject to:
| (c) | any penalties or sanctions imposed by a court relating to securities legislation or by a securities regulatory authority or has entered into a settlement agreement with a securities regulatory authority; or |
37
| (d) | any other penalties or sanctions imposed by a court or regulatory body that would likely be considered important to a reasonable investor in making an investment decision regarding the Company. |
The foregoing information, not being within the knowledge of the Company, has been furnished by the respective directors, officers and shareholders holding a sufficient number of securities of the Company to affect materially control of the Company.
Conflicts of Interest
The Company’s directors and officers may serve as directors or officers of other companies or have significant shareholdings in other companies and, to the extent that such other companies may participate in ventures in which the Company may participate, the directors of the Company may have a conflict of interest in negotiating and concluding terms respecting the extent of such participation. In the event that such conflict of interest arises at a meeting of the Company’s board of directors, a director who has such a conflict will abstain from voting for or against the approval of such participation or such terms. In accordance with the BCBCA, the directors of the Company are required to act honestly, in good faith and with a view to the best interests of the Company. In determining whether or not the Company will participate in a particular program and the interest therein to be acquired by it, the directors will primarily consider the degree of risk to which the Company may be exposed and its financial position at that time.
The directors and officers of the Company are aware of the existence of laws governing the accountability of directors and officers for corporate opportunity and requiring disclosures by the directors of conflicts of interest and the Company will rely upon such laws in respect of any directors’ and officers’ conflicts of interest or in respect of any breaches of duty by any of its directors and officers. All such conflicts will be disclosed by such directors or officers in accordance with the BCBCA and they will govern themselves in respect thereof to the best of their ability in accordance with the obligations imposed upon them by law. See “Risk Factors”. The directors and officers of the Company are not aware of any such conflicts of interest.
LEGAL PROCEEDINGS AND REGULATORY ACTIONS
The Company is not aware of: (a) any legal proceedings to which it is a party, or by which any of its property is subject, which would be material to it and are not aware of any such proceedings being contemplated; (b) any penalties or sanctions imposed against the Company by a court relating to securities legislation or by a securities regulatory authority, or any other penalties or sanctions imposed by a court or regulatory body against it that would likely be considered important to a reasonable investor making an investment decision; and (c) any settlement agreements that it has entered into before a court relating to securities legislation or with a securities regulatory authority.
The Company is required to have an audit committee (the “Audit Committee”) comprised of not less than three directors, a majority of whom are not officers, control persons or employees of the Company or an affiliate of the Company.
Audit Committee Charter
The text of the Audit Committee’s charter is attached as Schedule “A” to this Annual Information Form.
Composition of Audit Committee, Independence, and Financial Literacy
The Company’s current Audit Committee consists of Greg Smith, Dr. James Kuo and Dr. Robert Burke. National Instrument 52-110 – Audit Committees (“NI 52-110”) provides that a member of an audit committee is “independent” if the member has no direct or indirect material relationship with the Company, which could, in the view of the Company’s Board, be reasonably expected to interfere with the exercise of the member’s independent judgment. All members of the current Audit Committee are considered independent as defined in NI 52-110.
38
NI 52-110 provides that an individual is “financially literate” if he or she has the ability to read and understand a set of financial statements that present a breadth and level of complexity of accounting issues that are generally comparable to the breadth and complexity of the issues that can reasonably be expected to be raised by the Company’s financial statements. All members of the Audit Committee are “financially literate” as defined in NI 52-110.
Relevant Education and Experience
Refer to “Biographical Information” for a description of the Audit Committee members’ education and experience.
Audit Committee Oversight
Since the commencement of the Company’s most recently completed financial year, the Audit Committee has not made any recommendations to nominate or compensate an external auditor which were not adopted by the Board.
Reliance on Certain Exemptions
At no time since the commencement of the Company’s most recently completed financial year has the Company relied on any exemptions identified in Section 4, 5 or 6 of NI 52-110F1.
Pre-Approval Policies and Procedures
From time to time, management recommends to and requests approval from the Audit Committee for audit and non-audit services to be provided by the Company’s independent registered public accounting firm. The Audit Committee satisfies the pre-approval requirement if: (a) the aggregate amount of all the non-audit services that were not pre-approved is reasonably expected to constitute no more than five per cent of the total amount of fees paid by the issuer and its subsidiary entities to the issuer’s external auditor during the financial year in which the services are provided; (b) the Company or the subsidiary of the Company, as the case may be, did not recognize the services as non-audit services at the time of the engagement; and (c) the services are promptly brought to the attention of the Audit Committee and approved by the Audit Committee or by one or more of its members to whom authority to grant such approvals has been delegated by the Audit Committee, prior to the completion of the audit.
The Audit Committee may delegate to one or more independent Members the authority to pre-approve non-audit services in satisfaction of the requirement. The pre-approval of non-audit services by any Member to whom authority has been delegated must be presented to the Audit Committee at its first scheduled meeting following such pre approval.
Audit Fees
Financial Year Ended April 30 | Audit Fees ($)(1) | Audit Related Fees ($)(2) | Tax Fees ($)(3) | All Other Fees ($)(4) | ||||||||||||
2021 | 187,313 | 93,118 | Nil | Nil | ||||||||||||
2022 | 335,252 | Nil | 46,679 | Nil | ||||||||||||
Notes:
| (1) | “Audit fees” include aggregate fees billed by the Company’s external auditor in each of the last two fiscal years for audit fees. |
| (2) | “Audited related fees” include the aggregate fees billed in each of the last two fiscal years for assurance and related services by the Company’s external auditor that are reasonably related to the performance of the audit or review of the Company’s financial statements and are not reported under “Audit fees” above. |
39
| (3) | “Tax fees” include the aggregate fees billed in each of the last two fiscal years for professional services rendered by the Company’s external auditor for tax compliance, tax advice and tax planning. |
| (4) | “All other fees” include the aggregate fees billed in each of the last two fiscal years for products and services provided by the Company’s external auditor, other than “Audit fees”, “Audit related fees” and “Tax fees” above. |
INTEREST OF MANAGEMENT AND OTHERS IN MATERIAL TRANSACTIONS
No director, executive officer or shareholder that beneficially owns, or controls or directs, directly or indirectly, more than 10% of the issued shares of the Company, or any of their respective associates or affiliates, has any material interest, direct or indirect, in any transaction in which the Company has participated prior to the date of this Annual Information Form, which has materially affected or is reasonably expected to materially affect the Company.
The transfer agent and registrar for the common shares is Computershare Investor Services Inc. at 3rd Floor, 510 Burrard Street, Vancouver, British Columbia.
Except for contracts entered into in the ordinary course of business, the only material contracts that the Company has entered in the financial year ended April 30, 2022, or before the last financial year but still in effect, are as follows:
| 2 | ATM Agreement dated October 13, 2021 between the Company and H.C. Wainwright & Co., LLC, the particulars of which can be found under the heading “General Development of the Business” of this Annual Information Form; |
| 3 | Share Purchase Agreement entered into on March 29, 2022 among Charmquark EEN, Charmquark TWEE, K&E BV (collectively, the “Vendors”) and ImmunoPrecise Netherlands B.V. the Company, (the “BioStrand Share Purchase Agreement”) in connection with the BioStrand acquisition, the particulars of which can be found under the heading “General Development of the Business” of this Annual Information Form; and |
| 4 | Escrow Agreement dated April 13, 2022 between the Company, the Vendors and Computershare Trust Company of Canada (the “Escrow Agreement”), whereby the Company and the Vendors appointed Computershare Trust Company of Canada to act as escrow agent with respect to the 4,077,774 common shares of the Company issued to the Vendors as per the terms of the BioStrand Share Purchase Agreement. These common shares will be released over a three-year period, in accordance with and subject to the terms of the Escrow Agreement. |
Copies of the above material contracts are available under the Company’s profile on the SEDAR website at www.sedar.com.
Grant Thornton LLP, Independent Registered Public Accounting Firm, provided an auditor’s report in respect to the Company’s financial statements for the year ended April 30, 2022. Grant Thornton LLP is independent with respect to the Company in accordance with the standards of the U.S. Public Company Accounting Oversight Board.
Crowe MacKay LLP, Chartered Professional Accountants, provided an auditor’s report in respect to the Company’s financial statements for the year ended April 30, 2021. Crowe MacKay LLP is independent with respect to the Company in accordance with the Chartered Professional Accountants of British Columbia Code of Professional Conduct.
40
Additional financial information is provided in the Company’s comparative financial statements and management’s discussion and analysis for the year ended April 30, 2022, which will be available under the Company’s profile on the SEDAR website at www.sedar.com. Additional information, including directors’ and officers’ remuneration and indebtedness and securities authorized for issuance under equity compensation plans, is contained in the Company’s information circular for its most recent annual meeting of securityholders.
Copies of all materials incorporated by reference herein and additional information relating to the Company are available under the Company’s profile on the SEDAR website at www.sedar.com and the United States Securities and Exchange Commission website at www.sec.gov/edgar/search/.
Dated July 28, 2022.
BY ORDER OF THE BOARD OF DIRECTORS
“Jennifer Bath”
Jennifer Bath
President and Chief Executive Officer
41
SCHEDULE “A”
IMMUNOPRECISE ANTIBODIES LTD.
(THE “COMPANY”)
AUDIT COMMITTEE CHARTER
(Implemented pursuant to National Instrument 52-110 – Audit Committees)
National Instrument 52-110 – Audit Committees (the “Instrument”) relating to the composition and function of audit committees was implemented for reporting issuers and, accordingly, applies to every TSX Venture Exchange listed company, including the Company. The Instrument requires all affected issuers to have a written audit committee charter which must be disclosed, as stipulated by Form 52-110F2, in the management information circular of the Company wherein management solicits proxies from the security holders of the Company for the purpose of electing directors to the board of directors. The Company, as a TSX Venture Exchange-listed company is, however, exempt from certain requirements of the Instrument.
This Charter has been adopted by the board of directors in order to comply with the Instrument and to more properly define the role of the Committee in the oversight of the financial reporting process of the Company. Nothing in this Charter is intended to restrict the ability of the board of directors or Committee to alter or vary procedures in order to comply more fully with the Instrument, as amended from time to time.
PART 1
Purpose:
The purpose of the Committee is to:
| (a) | improve the quality of the Company’s financial reporting; |
| (b) | assist the board of directors to properly and fully discharge its responsibilities; |
| (c) | provide an avenue of enhanced communication between the directors and external auditors; |
| (d) | enhance the external auditor’s independence; |
| (e) | increase the credibility and objectivity of financial reports; and |
| (f) | strengthen the role of the directors by facilitating in depth discussions between directors, management and external auditors. |
| 1.1 | Definitions |
“accounting principles” has the meaning ascribed to it in National Instrument 52-107 – Acceptable Accounting Principles, Auditing Standards and Reporting Currency;
“Affiliate” means a Company that is a subsidiary of another Company or companies that are controlled by the same entity;
1
“audit services” means the professional services rendered by the Company’s external auditor for the audit and review of the Company’s financial statements or services that are normally provided by the external auditor in connection with statutory and regulatory filings or engagements;
“Charter” means this audit committee charter;
“Committee” means the committee established by and among certain members of the board of directors for the purpose of overseeing the accounting and financial reporting processes of the Company and audits of the financial statements of the Company;
“Control Person” means any individual or company that holds or is one of a combination of individuals or companies that holds a sufficient number of any of the securities of the Company so as to affect materially the control of the Company, or that holds more than 20% of the outstanding voting shares of the Company except where there is evidence showing that the holder of those securities does not materially affect the control of the Company;
“financially literate” has the meaning set forth in Section 1.2;
“immediate family member” means a person’s spouse, parent, child, sibling, mother or father-in-law, son or daughter-in-law, brother or sister-in-law, and anyone (other than an employee of either the person or the person’s immediate family member) who shares the individual’s home;
“Instrument” means National Instrument 52-110 – Audit Committees;
“MD&A” has the meaning ascribed to it in National Instrument 51-102;
“Member” means a member of the Committee;
“National Instrument 51-102” means National Instrument 51-102 – Continuous Disclosure Obligations; and
“non-audit services” means services other than audit services.
| 1.2 | Meaning of Financially Literate |
For the purposes of this Charter, an individual is financially literate if he or she has the ability to read and understand a set of financial statements that present a breadth and level of complexity of accounting issues that are generally comparable to the breadth and complexity of the issues that can reasonably be expected to be raised by the Company’s financial statements.
PART 2
| 2.1 | Audit Committee |
The board of directors has hereby established the Committee for, among other purposes, compliance with the Instrument.
| 2.2 | Relationship with External Auditors |
The Company will require its external auditor to report directly to the Committee and the Members shall ensure that such is the case.
2
| 2.3 | Committee Responsibilities |
| 1. | The Committee shall be responsible for making the following recommendations to the board of directors: |
| (a) | the external auditor to be nominated for the purpose of preparing or issuing an auditor’s report or performing other audit, review or attest services for the Company; and |
| (b) | the compensation of the external auditor. |
| 2. | The Committee shall be directly responsible for overseeing the work of the external auditor engaged for the purpose of preparing or issuing an auditor’s report or performing other audit, review or attest services for the Company, including the resolution of disagreements between management and the external auditor regarding financial reporting. This responsibility shall include: |
| (a) | reviewing the audit plan with management and the external auditor; |
| (b) | reviewing with management and the external auditor any proposed changes in major accounting policies, the presentation and impact of significant risks and uncertainties, and key estimates and judgements of management that may be material to financial reporting; |
| (c) | questioning management and the external auditor regarding significant financial reporting issues discussed during the fiscal period and the method of resolution; |
| (d) | reviewing any problems experienced by the external auditor in performing the audit, including any restrictions imposed by management or significant accounting issues on which there was a disagreement with management; |
| (e) | reviewing audited annual financial statements, in conjunction with the report of the external auditor, and obtaining an explanation from management of all significant variances between comparative reporting periods; |
| (f) | reviewing the post-audit or management letter, containing the recommendations of the external auditor, and management’s response and subsequent follow up to any identified weakness; |
| (g) | reviewing interim unaudited financial statements before release to the public; |
| (h) | reviewing all public disclosure documents containing audited or unaudited financial information before release, including any prospectus, the annual report and management’s discussion and analysis; |
| (i) | reviewing the evaluation of internal controls by the external auditor, together with management’s response; |
| (j) | reviewing the terms of reference of the internal auditor, if any; |
| (k) | reviewing the reports issued by the internal auditor, if any, and management’s response and subsequent follow up to any identified weaknesses; and |
| (l) | reviewing the appointments of the chief financial officer and any key financial executives involved in the financial reporting process, as applicable. |
| 3. | The Committee shall pre-approve all non-audit services to be provided to the Company or its subsidiary entities by the issuer’s external auditor. |
3
| 4. | The Committee shall review the Company’s financial statements, MD&A, and annual and interim earnings press releases before the Company publicly discloses this information. |
| 5. | The Committee shall ensure that adequate procedures are in place for the review of the Company’s public disclosure of financial information extracted or derived from the Company’s financial statements, and shall periodically assess the adequacy of those procedures. |
| 6. | When there is to be a change of auditor, the Committee shall review all issues related to the change, including the information to be included in the notice of change of auditor called for under National Instrument 51-102, and the planned steps for an orderly transition. |
| 7. | The Committee shall review all reportable events, including disagreements, unresolved issues and consultations, as defined in National Instrument 51-102, on a routine basis, whether or not there is to be a change of auditor. |
| 8. | The Committee shall, as applicable, establish procedures for: |
| (a) | the receipt, retention and treatment of complaints received by the issuer regarding accounting, internal accounting controls, or auditing matters; and |
| (b) | the confidential, anonymous submission by employees of the issuer of concerns regarding questionable accounting or auditing matters. |
| 9. | As applicable, the Committee shall establish, periodically review and approve the Company’s hiring policies regarding partners, employees and former partners and employees of the present and former external auditor of the issuer, as applicable. |
| 10. | The responsibilities outlined in this Charter are not intended to be exhaustive. Members should consider any additional areas which may require oversight when discharging their responsibilities. |
| 2.4 | Non-Audit Services |
The Committee shall satisfy the pre-approval requirement in subsection 2.3(3) if:
| (a) | the aggregate amount of all the non-audit services that were not pre-approved is reasonably expected to constitute no more than five per cent of the total amount of fees paid by the issuer and its subsidiary entities to the issuer’s external auditor during the financial year in which the services are provided; |
| (b) | the Company or the subsidiary of the Company, as the case may be, did not recognize the services as non-audit services at the time of the engagement; and |
| (c) | the services are promptly brought to the attention of the Committee and approved by the Committee or by one or more of its members to whom authority to grant such approvals has been delegated by the Committee, prior to the completion of the audit. |
4
| 2.5 | Delegation of Pre-Approval Function |
| 1. | The Committee may delegate to one or more independent Members the authority to pre-approve non-audit services in satisfaction of the requirement in subsection 2.3(3). |
| 2. | The pre-approval of non-audit services by any Member to whom authority has been delegated pursuant to subsection 2.5(1) must be presented to the Committee at its first scheduled meeting following such pre-approval. |
PART 3
| 3.1 | Composition |
| 1. | The Committee shall be composed of a minimum of three Members. |
| 2. | Every Member shall be a director of the issuer. |
| 3. | The majority of Members shall not be employees, Control Persons or officers of the Company. |
| 4. | If practicable, given the composition of the directors of the Company, each audit committee member shall be financially literate. |
PART 4
| 4.1 | Authority |
Until the replacement of this Charter, the Committee shall have the authority to:
| (a) | engage independent counsel and other advisors as it determines necessary to carry out its duties; |
| (b) | set and pay the compensation for any advisors employed by the Committee; |
| (c) | communicate directly with the internal and external auditors; and |
| (d) | recommend the amendment or approval of audited and interim financial statements to the board of directors. |
PART 5
| 5.1 | Disclosure in Information Circular |
If management of the Company solicits proxies from the security holders of the Company for the purpose of electing directors to the board of directors, the Company shall include in its management information circular the disclosure required by Form 52-110F2 (Disclosure by Venture Issuers).
PART 6
| 6.1 | Meetings |
| 5. | Meetings of the Committee shall be scheduled to take place at regular intervals and, in any event, not less frequently than quarterly. |
| 6. | Opportunities shall be afforded periodically to the external auditor, the internal auditor and to members of senior management to meet separately with the Members. |
| 7. | Minutes shall be kept of all meetings of the Committee. |
5
PART 7
| 7.1 | Currency of this Charter |
| 1. | This charter was last revised and approved by the Board on March 16, 2017. |
6