UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 1-SA
x SEMIANNUAL REPORT PURSUANT TO REGULATION A
¨ SPECIAL FINANCIAL REPORT PURSUANT TO REGULATION A
For the fiscal semiannual period ended:
June 30, 2020
Virtuoso Surgical, Inc.
(Exact name of issuer as specified in its charter)
| Tennessee | 81-2994832 | |
| (State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) |
5701 Old Harding Pike, Suite 200
Nashville, Tennessee 37205
(Full mailing address of principal executive offices)
(615) 352-9519
(Issuer’s telephone number, including area code)
Table of Contents
| Item 1. Management’s Discussion and Analysis of Financial Condition and Results of Operation | 3 |
| Item 2. Other Information | 5 |
| Item 3. Financial Statements | 6 |
| Item 4. Exhibits | 7 |
| SIGNATURES | 8 |
INFORMATION TO BE INCLUDED IN REPORT
In this Semiannual Report on Form 1-SA (this “Report”), the terms “Virtuoso,” “Company,” “we,” “our,” or “us” refer to Virtuoso Surgical, Inc.
Item 1. Management’s Discussion and Analysis of Financial Condition and Results of Operation
Cautionary Note Regarding Forward-Looking Statements
The following discussion and analysis of our financial condition and results of operations for the six months ended June 30, 2020 should be read in conjunction with our financial statements and the related notes included elsewhere in this Report.
In addition to historical information, the following disclosure and analysis contains “forward-looking statements.” Forward-looking statements describe the Company’s current expectations or forecasts of future events as of the date of this Report and are not statements of historical fact. These forward-looking statements include information about possible or assumed future events, including, among other things, discussion, and analysis of the Company’s assets, business, capital expenditures, cash flows, cost management, condition (financial and otherwise), indebtedness, liquidity, profitability, prospects, results of operations, revenues, and strategic plans. Words such as “anticipates,” “believes,” “could,” “estimates,” “expects,” “intends,” “plans,” “potential,” “predicts,” “projects,” “seeks,” “should,” “will,” “would,” and variations of these words and similar expressions are intended to identify forward-looking statements.
Forward-looking statements are not guarantees of future performance and are subject to risks, uncertainties, and other factors, some of which are beyond the Company’s control, are difficult to predict, and/or could cause actual results and the timing of events to differ materially from those expressed or forecasted in the forward-looking statements. For a detailed discussion of the various factors that could cause our actual results to differ from expectations, please refer to the sections entitled “Cautionary Statement Regarding Forward-Looking Statements” and “Risk Factors” in our Offering Circular filed with the Securities and Exchange Commission on February 26, 2020. We assume no obligation to update any of these forward-looking statements except to the extent required by applicable law.
Discussion
The first half of 2020 was a good six months for Virtuoso, particularly given the circumstances of the COVID-19 pandemic.
In January, the Company continued high-level discussions with a number of major medical-device manufacturers, and those discussions continue and are ongoing. To aid the Company’s engineering efforts, to potentially bring additional capital to the Company, and to maintain open communications about a possible exit transaction, the Company will continue these discussions for the foreseeable future.
In January as well, the Company began work with Fletcher Spaght, Inc., a Boston-based marketing/consulting firm. The purpose of the engagement was to develop empirical data regarding physician acceptance of the Virtuoso System & Device for urologic and gynecologic surgery, and to model the possible financial possibilities of the Virtuoso system. Fletcher Spaght provided their initial report to the Company in March, and continued with a total of seven reports from March through August, including the results of physician surveys and financial modeling. The Company expects to continue to work with Fletcher Spaght regarding other future potential areas of operation, including orthopedic, pulmonary, and neurosurgery applications.
Liquidity and Capital Resources
In March 2020, the Company raised $2,500,000 through the sale of its common stock to a majority of its angel investors. From March 2020 through June 2020, the Company raised approximately $225,000 through the Company’s Regulation A, Tier 2, Offering (the “Reg A+ Offering”). Throughout the course of the summer, the pace of the Reg A+ Offering increased, and the Company has raised approximately $1,500,000 through September 28, 2020. The Company also filed for and ultimately received approximately $139,000 in Paycheck Protection Program funds through a loan guaranteed by the U.S. government.
3
For the period ended June 30, 2020, the Company had a net loss of approximately $1,000,000 and net cash used in operating activities of approximately $800,000. As of June 30, 2020, the Company had cash and cash equivalents of approximately $2,200,000, unbilled grants of approximately $750,000, and an accumulated deficit of approximately $2,000,000.
Below is a graph of the Company’s spending patterns (except as qualified in the paragraph following the graph), by spending category and quarter, for 2019 and the first half of 2020:
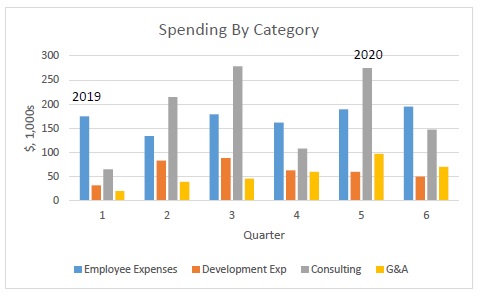
The Company’s spending is increasing steadily over time. The most variable expenditure is consulting, as outside resources often remain advisable only on a project-by-project basis. Non-employee development expenses are also variable, depending on the build-state of the Company’s prototypes at an given point in time. (Note: The graph above excludes the impact of a one-time stock-based compensation grant and related taxes for certain employees of the Company. Also, in Q1 2019, the Company altered the classification of its part-time officers who were accounted for as an employee expense, converting those expenditures to a consulting expense. The resulting change, were it reflected in Q1 2019, would have shown a steadier growth curve in employee expenses and somewhat less variability in consulting expenses.)
The Company believes that its cash and cash equivalents as of June 30, 2020, combined with remaining grant funds, periodic sales of the Company’s common stock, and Paycheck Protection Program funds, are sufficient to fund its operations for at least 24 months from this Report. The Company expects to continue to incur additional losses in the foreseeable future as a result of the Company’s research and development activities.
Impact of the COVID-19 Pandemic
In March 2020, COVID-19 became a national pandemic, and the Company altered operations to protect the safety of its workers. All workers were able to operate from home for a number of weeks, and the development of the Virtuoso System & Device continued apace. After several weeks, the Company decided that one engineer should be able to work on-site in order to maintain the development pace of the physical engineering efforts. Eventually, in June, the Company permitted other employees to return on-site as needed and where appropriate. The Company implemented rules for wearing masks and for socially distancing in the workplace. Employees are asked to continue working remotely as much as is feasible.
4
From the Company’s perspective, COVID-19 has not materially harmed its engineering operations, but COVID-19 may have affected the timing and success of the Reg A+ Offering. Specifically, as a result of COVID-19, there were appreciable delays in establishing sales efforts through broker/dealer networks in connection with the Reg A+ Offering. In September 2020, the Company, through its placement agent, C2M Securities, LLC (“Placement Agent”), began to work with Deer Isle Group, LLC to market the securities through Deer Isle’s platform. The Company, through its Placement Agent, is also building out the broker-dealer network for other channels of sales and distribution of the securities under the Reg A+ Offering. In the event that the ongoing efforts to fill the Reg A+ Offering are not complete by December 31, 2020, the Company may decide to extend the Reg A+ Offering into 2021.
Subsequent Events
In August, the Company began formally appointing physicians and surgeons to the Company’s Medical Advisory Board, including leaders in the fields of minimally invasive urology, gynecology, and pulmonology. The members of the Medical Advisory Board are expected to assist the Company as it accelerates the process of clinical testing and feedback.
From an engineering perspective, the Company is on the cusp of entering a new phase of the development process. The Company’s current prototype, called Bach, is fully operational. The Company’s engineering team is currently focused on reliability and repeatability testing, which requires multiple models of Bach to be fabricated and tested. That engineering work is expected to be the primary focus of the engineering team in the near-term. Once that phase of development is over, the development process will require formal testing, verification, and validation of the system, for regulatory approval by the U.S. Food and Drug Administration. That process is expected to require significant additional capital.
In June, the Company submitted its first application to the FDA, seeking Breakthrough Device Designation under the Breakthrough Device Program (“BDP”). Receiving BDP status has the potential to accelerate the Company’s communications with the FDA prior to the Company’s primary filing. The FDA initially rejected the Company’s BDP application. After the Company requested supervisory review, the decision denying BDP state was upheld, with a suggestion that the Company limit its proposed application for BDP designation. Based on that decision, the Company has decided to proceed with a Section 513(g) application for general device classification and to pursue the regular Q-Sub process of communication with the FDA. Depending upon the pace of the Company’s testing, verification, and validation work, the Company currently expects to file its application for De Novo Classification in the first half of 2022, which would ultimately permit the Company to begin commercial sales upon FDA’s expected eventual grant of the classification.
On September 3, 2020, and in light of the capital that has been raised to date, the Company’s Board approved a hiring plan that anticipates hiring at least four engineers. This is almost a 50% increase in staff FTEs. The Company’s Board also approved an acceleration of spending on outside consultants, particularly Precision Systems, Inc., who will assist the Company with FDA compliance of the system’s software components.
| Item 2. | Other Information |
None.
5
| Item 3. | Financial Statements |
INDEX TO FINANCIAL STATEMENTS
6
Virtuoso Surgical, Inc. June 30, 2010 (Unaudited) and December 31, 2019 |
| June 30, 2020 | December 31, 2019 | |||||||
| (Unaudited) | ||||||||
| Assets | ||||||||
| Cash and cash equivalents | $ | 2,212,285 | $ | 204,832 | ||||
| Deferred offering costs | - | 94,167 | ||||||
| Total current assets | 2,212,285 | 298,999 | ||||||
| Property and equipment, net | 299,364 | 257,332 | ||||||
| Total assets | $ | 2,511,649 | $ | 556,331 | ||||
| Liabilities | ||||||||
| Accounts payable | $ | 40,408 | $ | 49,533 | ||||
| Accrued liabilities | 10,673 | 294 | ||||||
| PPP loan | 139,672 | - | ||||||
| Total current liabilities | 190,753 | 49,827 | ||||||
| Stockholders’ Equity | ||||||||
| Convertible Class A preferred stock, $1 par value, 50,000,000 shares authorized, 1,535,000 and 1,305,000 shares issued and outstanding on June 30, 2020 and December 31, 2019, respectively, aggregate liquidation preference of $1,695,636 and $1,419,204 on June 30, 2020 and December 31, 2019, respectively | 1,535,000 | 1,305,000 | ||||||
| Common stock, no par value, 2,000,000 shares authorized, 1,040,000 and 985,000 shares issued and outstanding on June 30, 2020 and December 31, 2019, respectively, 5,000 shares reserved for stock-based awards | 2,750,000 | - | ||||||
| Accumulated deficit | (1,964,104 | ) | (798,496 | ) | ||||
| Total stockholders’ equity | 2,320,896 | 506,504 | ||||||
| Total liabilities and stockholders’ equity | $ | 2,511,649 | $ | 556,331 | ||||
See notes to financial statements.
F-1
Virtuoso Surgical, Inc. Statements of Operations (Unaudited) For the six months ended June 30, 2020 and 2019 |
| 2020 | 2019 | |||||||
| Operating Expenses | ||||||||
| Research and development | $ | 599,088 | $ | 598,485 | ||||
| General and administrative | 731,091 | 181,216 | ||||||
| Total operating expenses | 1,330,179 | 779,701 | ||||||
| Other income (expense) | ||||||||
| Government grants | 274,872 | 614,641 | ||||||
| Interest income | - | 930 | ||||||
| Total other income (expense) | 274,872 | 615,571 | ||||||
| Net loss | $ | (1,055,307 | ) | $ | (164,130 | ) | ||
See notes to financial statements.
F-2
Virtuoso Surgical, Inc. Statement of Stockholders’ Equity (Unaudited) For the six months ended June 30, 2020 and 2019 |
| Preferred stock | Common Stock | Accumulated | ||||||||||||||||||||||
| Class A shares | Amount | Shares | Amount | Deficit | Total | |||||||||||||||||||
| Balance on January 1, 2019 | 1,075,000 | $ | 1,075,000 | 985,000 | $ | - | $ | (383,756 | ) | $ | 691,244 | |||||||||||||
| Issuance of Class A shares | 30,000 | 30,000 | - | - | - | 30,000 | ||||||||||||||||||
| Net loss | - | - | - | - | (164,130 | ) | (164,130 | ) | ||||||||||||||||
| Balance on June 30, 2019 | 1,105,000 | $ | 1,105,000 | 985,000 | - | $ | (547,886 | ) | $ | 557,114 | ||||||||||||||
| Balance on January 1, 2020 | 1,305,000 | $ | 1,305,000 | 985,000 | - | $ | (798,496 | ) | $ | 506,504 | ||||||||||||||
| Stock issuance costs | - | - | - | - | (110,301 | ) | (110,301 | ) | ||||||||||||||||
| Issuance of Class A shares | 230,000 | 230,000 | - | - | - | 230,000 | ||||||||||||||||||
| Issuance of common shares | - | - | 50,000 | 2,500,000 | - | 2,500,000 | ||||||||||||||||||
| Stock-based compensation awards | - | - | 5,000 | 250,000 | - | 250,000 | ||||||||||||||||||
| Net loss | - | - | - | - | (1,055,307 | ) | (1,055,307 | ) | ||||||||||||||||
| Balance on June 30, 2020 | 1,535,000 | $ | 1,535,000 | 1,040,000 | $ | 2,750,000 | $ | (1,964,104 | ) | $ | 2,320,896 | |||||||||||||
See notes to financial statements.
F-3
Virtuoso Surgical, Inc. Statements of Cash Flows (Unaudited) For the six months ended June 30, 2020 and 2019 |
| 2020 | 2019 | |||||||
| Cash flows from operating activities | ||||||||
| Net loss | $ | (1,055,307 | ) | $ | (164,130 | ) | ||
| Adjustments to reconcile net loss to net cash used in operating activities: | ||||||||
| Depreciation | 16,702 | 14,547 | ||||||
| Stock-based compensation awards | 250,000 | - | ||||||
| Increase in grants receivable | - | (42,965 | ) | |||||
| Decrease in accounts payable | (9,126 | ) | (77,789 | ) | ||||
| Increase (decrease) in accrued liabilities | 10,380 | (11,015 | ) | |||||
| Net cash used in operating activities | (787,351 | ) | (281,352 | ) | ||||
| Cash flows from investing activities | ||||||||
| Purchases of property and equipment | (58,734 | ) | (68,565 | ) | ||||
| Net cash used in investing activities | (58,734 | ) | (68,565 | ) | ||||
| Cash flows from financing activities | ||||||||
| Proceeds from PPP loan | 139,672 | - | ||||||
| Proceeds from sale of common stock | 2,500,000 | - | ||||||
| Payments for stock issuance costs | (16,134 | ) | - | |||||
| Proceeds from sale of convertible preferred stock | 230,000 | 30,000 | ||||||
| Net cash provided by financing activities | 2,853,538 | 30,000 | ||||||
| Net increase (decrease) in cash and cash equivalents | 2,007,453 | (319,917 | ) | |||||
| Cash and cash equivalents at beginning of period | 204,832 | 602,748 | ||||||
| Cash and cash equivalents at end of period | $ | 2,212,285 | $ | 282,831 | ||||
See notes to financial statements.
F-4
Virtuoso Surgical, Inc.
Notes to Financial Statements (Unaudited)
For the six months ended June 30, 2020 and 2019
Note 1: Organization and Nature of Operations
Nature of business
Virtuoso Surgical, Inc. (the Company) was incorporated in April 2016, as a Tennessee corporation, with operations based in Nashville, Tennessee. The Company was formed to design, develop, and market medical devices to transform minimally invasive surgery by providing dexterous, accurate and cost-effective robotic tools. Since incorporation, the Company has devoted substantially all efforts to research and development, business planning, recruiting management and technical staff, acquiring operating assets, and raising capital. The Company has not commenced its planned principal operations.
Liquidity and Capital Resources
The Company is subject to a number of risks similar to other medical device companies, including, but not limited to, raising additional capital, development by its competitors of new technological innovations, safety and efficacy of the product in clinical trials, the regulatory approval process governing medical devices, market acceptance of the Company’s products, and protection of proprietary technology. The Company has funded its operations to date primarily through federal grants, a state of Tennessee SBIR matching grant and the sale of preferred and common stock.
The Company has sold 1,071,500 shares of preferred stock from July to August of 2020 yielding $1,071,500.
The Company believes that its cash and cash equivalents as of June 30, 2020, combined with remaining grant funds, periodic sales of the Company’s preferred and common stock are sufficient to fund its operations for at least 12 months from the issuance of these financial statements. The Company expects to continue to incur additional losses in the foreseeable future due to the Company’s research and development activities.
Note 2: Summary of Significant Accounting Policies
Basis of Presentation
The accompanying financial statements are presented in U.S. dollars and have been prepared in accordance with accounting principles generally accepted in the United States of America (US GAAP) and include all adjustments necessary for the fair presentation of the Company’s financial position, results of operations, and cash flows for the periods presented. The Company operates in one operating segment and, accordingly, no segment disclosures have been presented herein. The Company’s management performed an evaluation of its activities through the date of filing of these financial statements and concluded that there are no subsequent events requiring disclosure, other than as disclosed.
Interim Unaudited Financial Data
The Company’s financial statements reflect all adjustments, which are, in our opinion, of a normal recurring nature and necessary for a fair presentation of our financial position, results of operations, and cash flows for the interim periods. Interim results of operations are not necessarily indicative of the results to be expected for the full year. These financial statements, including notes, are unaudited, and exclude some of the disclosures required for annual financial statements, and should be read in conjunction with our audited financial statements for the year ended December 31, 2019, included in the Company’s annual report on Form 1-K filed with the SEC on May 12, 2020.
Use of estimates
The preparation of financial statements in conformity with accounting principles generally accepted in the United States of America requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and the disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of expenses during the reporting period. Actual results could differ from those estimates.
Cash and cash equivalents
Cash equivalents include all highly liquid investments with original maturities within 90 days from the date of purchase.
F-5
Virtuoso Surgical, Inc.
Notes to Financial Statements (Unaudited)
For the six months ended June 30, 2020 and 2019
Note 2: Summary of Significant Accounting Policies (Continued)
Concentrations of credit risk
Cash and cash equivalents are financial instruments that potentially subject the Company to concentrations of credit risk.
Property and equipment
Property and equipment are recorded at cost and depreciated over their estimated useful lives using the straight-line method. Repairs and maintenance costs are expensed as incurred, whereas major improvements are capitalized as additions to property and equipment.
Capitalized software costs represent costs incurred after technological feasibility was established. It is anticipated further costs will be capitalized until development is complete and a working model is ready for customer testing. Costs thereafter will be expensed as incurred.
Depreciation is provided using the straight-line method over the following estimated useful lives when the corresponding asset is placed in service:
| Asset Classification | Useful Life | |
| Laboratory equipment | Five years | |
| Computer equipment | Five years | |
| Office furniture and equipment | Five years | |
| Software (capital) | Three years | |
| Software (developed) | Three years | |
| Leasehold improvements | Lesser of useful life or remaining lease term |
Stock-based compensation
The Company records stock-based compensation at fair market value at the date of award.
PPP Loan
On January 30, 2020, the World Health Organization declared the COVID-19 outbreak a “Public Health Emergency of International Concern” and on March 11, 2020, declared it to be a pandemic. The Company received a loan in accordance with the Paycheck Protection Program (PPP) section of the Coronavirus Air, Relief, and Economic Security Act (the CARES Act). U.S. GAAP provides companies with several alternatives for reporting the loan and any future forgiveness: 1) proceeds can be treated as debt and future forgiveness recognized as income when the loan or any portion thereof is formally discharged; 2) proceeds can be treated as an income grant where they recognize a deferred income liability and derecognize the liability, and recognize income or reduce expenses, as they incur and recognize qualifying payroll and other operating costs that they estimate with reasonable assurance meet the conditions necessary for forgiveness; 3) proceeds can be treated as a conditional contribution where they recognize a refundable advance and derecognize the liability, and recognize income, as the conditions for forgiveness are substantially met or explicitly waived; or 4) proceeds can be recognized as a liability and derecognize the liability, and recognize income, as all conditions for forgiveness are met. The Company has elected to treat the PPP loan as a liability.
Government grants
The Company’s grants consist of United States Health and Human Services’ research and development and related matching awards. As each is a cost-reimbursement grant, the Company recognizes revenues up to the amount of incurred, allowable, and paid grant expenditures. Accordingly, grants receivable represents the related amount of requests for reimbursement not yet received.
Research and development costs
Costs incurred in the research and development of the Company's products are expensed as incurred.
F-6
Virtuoso Surgical, Inc.
Notes to Financial Statements (Unaudited)
For the six months ended June 30, 2020 and 2019
Note 2: Summary of Significant Accounting Policies (Continued)
Patent costs
The Company entered into license agreements with two research institutions for patented technology owned by these institutions. The Company expenses as incurred all costs, including legal expenses, associated with obtaining patents until the patented technology becomes feasible. All costs incurred after the patented technology is feasible will be capitalized as an intangible asset. As of June 30, 2020, no costs had been capitalized since inception of the Company.
The patents under these license agreements require certain initial fees, paid in cash and Common Stock. Royalties, as defined in the agreements, are payable to each institution upon sales of licensed products.
Income taxes
The Company records deferred tax assets and liabilities for the expected future tax consequences of temporary differences between the Company's financial statement carrying amounts and the tax basis of assets and liabilities using enacted tax rates expected to be in effect in the years in which the differences are expected to reverse. A valuation allowance is used to reduce the net deferred tax assets to the amount that will more likely than not be realized.
Any interest and penalties are classified in expense in the Company’s financial statements.
Retirement plan
The Company sponsors a Simplified Employee Pension (SEP) retirement plan. The Company contributes to the SEP an amount equal to four percent of each employee’s annual salary. The Company contributed $1,045 and $13,467 for the six months ended June 30, 2020 and 2019, respectively.
Reclassification
Certain amounts in the prior period financial statements have been reclassified to conform to the presentation of the current period financial statements. These reclassifications had no effect on the previously reported net loss.
Note 3: Property and Equipment
Property and equipment as of June 30, 2020 and 2019 consist of the following:
| 2020 | 2019 | |||||||
| Laboratory equipment | $ | 88,397 | $ | 88,397 | ||||
| Computer equipment | 40,321 | 40,321 | ||||||
| Furniture and equipment | 526 | - | ||||||
| Software – capital | 19,826 | 19,826 | ||||||
| Software – developed | 210,129 | 114,370 | ||||||
| 359,199 | 262,914 | |||||||
| Less accumulated depreciation and amortization | (59,835 | ) | (26,957 | ) | ||||
| $ | 299,364 | $ | 235,957 | |||||
The Company incurred depreciation and amortization expense of $16,702 and $14,547 for the six months ended June 30, 2020 and 2019, respectively.
Note 4: PPP Loan
On May 4, 2020, the Company received a loan in the amount of $139,672 in accordance with the PPP section of the CARES Act. Under this loan program, the Company may be eligible for forgiveness of some portion of the loan up to 100%, if and when qualifying conditions are met. Accounting for the loan and any future forgiveness could have an impact on future financial reporting. As of June 30, 2020, management is actively monitoring qualifying conditions to maximize future loan forgiveness. The unsecured note bears interest at the rate of 1.00% and matures on May 4, 2022. As long as the Company submits its loan forgiveness application within 10 months of December 31, 2020, the Company will not be required to make any payments on the loan until the forgiveness amount is remitted to the lender by the U.S. Small Business Administration (SBA). If the loan if fully forgiven, the Company will not be responsible for any payments.
F-7
Virtuoso Surgical, Inc.
Notes to Financial Statements (Unaudited)
For the six months ended June 30, 2020 and 2019
Note 5: Government Grants
The Company received certain awards from Federal and local sources to support its research and development activities as follows:
| National Institute of Health – | ||||
| Small Business Innovation Research Program (SBIR) including amendment | $ | 3,132,895 | ||
| Small Business Technology Transfer Program (SBTT) | 224,598 | |||
| 3,357,493 | ||||
| Authorized spending through June 30, 2020 | 2,611,385 | |||
| Remaining authorized funds | $ | 746,108 | ||
Through June 30, 2020 and June 30, 2019, the Company has billed $2,393,723 and $1,302,129, respectively, under the terms of these Federal awards with $64,004 recorded as grants receivable as of June 30, 2019.
The Company was awarded $150,000 of SBIR matching funding for its Federal grants from Launch Tennessee, of which $150,000 was spent through June 30, 2019.
Note 6: Stockholders’ Equity
General
The authorized capital stock of the Company consists of 52,000,000 shares, of which 50,000,000 shares are designated as Class A Preferred Stock and 2,000,000 shares are designated as Common Stock.
Class A Preferred Stock
In 2018, the Company offered 3,000,000 shares of Class A Preferred Stock (Preferred Stock) at $1.00 per share. The Company has sold 1,075,000 shares in 2018, 230,000 shares in 2019, and 230,000 shares in 2020. The sales of preferred shares yielded gross proceeds of $1,075,000 in 2018, $230,000 in 2019, and $230,000 for the first six months of 2020.
The Preferred Stock has the following characteristics:
Voting
The holders of the Preferred Stock shall have no voting or other management rights, or other beneficial rights other than those disclosed in the Company Bylaws.
Dividends
Issued Preferred Stock shares carry a Paid-In-Kind (PIK) dividend of 7% per year, uncompounded. Cumulative dividends for the period from issuance through June 30, 2020 are $160,636.
Other Provisions
Other provisions related to the Preferred Stock are set forth in the Company Bylaws and include certain rights upon a change of control, as defined, including Success Bonus terms, and rights and priorities with respect to consideration received or conversion rights in redemption.
Preferred shares are redeemable upon a change of control of the Company or at any time, at the discretion of the Board of Directors, provided that the redemption must include 100% of a shareholders’ preferred shares at par, plus any accrued PIK and the applicable Success Bonus amount. Until all Preferred Stock shares are redeemed certain restrictions exist as to compensation levels and dividend distributions.
F-8
Virtuoso Surgical, Inc.
Notes to Financial Statements (Unaudited)
For the six months ended June 30, 2020 and 2019
Stock Issuance Costs
The Company recorded stock issuance costs for its Regulation D capital raise in the amount of $6,085 as contra-equity in 2018. The Company recorded stock issuance costs of $110,301 in relation to the Company’s 2020 Regulation A+ capital raise as a contra-equity in the statements of stockholders’ equity.
Common Stock
The Company issued 820,000 shares of common stock to certain founding parties who have been responsible for incubating and forming the Company and 90,000 shares to research institutions, in exchange for certain technology and contractual rights. The Company has issued 80,000 shares to employees. The Company sold 50,000 shares of common stock between January 1, 2020 and March 30, 2020 at $50 per share yielding proceeds of $2,500,000 and issued 5,000 shares of stock-based compensation awards.
Effective August 7, 2019, the Board of Directors approved an increase in the number of Common shares from 100 shares to 2,000,000 shares. The Board also approved a 10,000 for one Common share stock split that increased the number of Common shares issued and outstanding from 98.5 to 985,000. The financial statements have been adjusted retroactively for all periods presented to reflect the Common share stock split.
Note 7: Stock-Based Compensation
In 2020, the Company awarded 5,000 shares of common stock to certain employees and reserved 5,000 shares to these employees subject to a risk of forfeiture in 2021 if the employees were no longer employed. The Company recognized $250,000 as compensation expense in 2020 due to this award.
Note 8: Related Party Transactions
The Company had the following transactions with stockholders for the six months ended June 30, 2020 and 2019:
| 2020 | 2019 | |||||||
| Consulting fees | $ | 34,803 | $ | 62,457 | ||||
| Rent | - | 14,378 |
The Company paid consulting fees to certain board members and investors for research and development services.
On February 6, 2019, a Company officer and stockholder executed a five-year lease agreement for the Company’s office. On May 1, 2019, the Company entered into a one-year oral sublease agreement with the officer and stockholder whereby the Company pays the lease at cost, $6,000 per month.
The Company received a loan from an officer who is also a stockholder of $100,000 in February 2020 which was repaid in full in March 2020.
Note 9: Income Taxes
Since inception, the Company has experienced net operating losses (NOL) which is consistent with the company conducting extensive research and development (R&D) activities. Prior to 2018, the Company, as a Subchapter S Corporation, passed these losses and credits to the stockholders. Beginning January 1, 2018, the Company converted to C Corporation status, and as such, these NOLs and R&D tax credits create net deferred tax assets (DTA); however, because of the current operating status of the Company these DTAs have been reduced to zero for reporting purposes through a 100% valuation allowance at December 31, 2019 and 2018.
As of December 31, 2019, the Company had net deferred tax assets of $198,400. Due to uncertainties surrounding the Company’s ability to generate future taxable income to realize these assets, a full valuation allowance has been established to offset the net deferred tax asset.
F-9
Virtuoso Surgical, Inc.
Notes to Financial Statements (Unaudited)
For the six months ended June 30, 2020 and 2019
As of December 31, 2019, and 2018, the Company had available federal NOL carryforwards of approximately $781,000 and $442,000, respectively. The NOL generated in 2019 of $339,000 and 2018 of $442,000 will carry forward indefinitely and be available to offset up to 100% of future taxable income each year (per the CARES Act) and for two carryback years (per the CARES Act). In addition, the Company had federal research and development credits carryforwards of $58,300 and $37,700 as of December 31, 2019 and 2018, respectively, to reduce future income taxes, if any. These carryforwards begin to expire in 2038 and are subject to review and possible adjustment by the Internal Revenue Code (IRC). The Company also has available state NOL carryforwards of approximately $787,000 and $389,000 as of December 31, 2019 and 2018, respectively, which expire from 2033 to 2034.
The Company has evaluated the positive and negative evidence bearing upon its ability to realize the deferred tax assets. Management has considered the Company’s history of cumulative net losses incurred since inception and its lack of commercialization of any products and has concluded that it is more likely than not that the Company will not realize the benefits of the deferred tax assets. Accordingly, a full valuation allowance has been established against the deferred tax assets as of December 31, 2019 and 2018. Management reevaluates the positive and negative evidence at each reporting period. The Company’s valuation allowance increased by $103,200 for year ended December 31, 2019. For the year ended December 31, 2018, the valuation allowance increased by $83,300.
The Company recognizes liabilities for uncertain tax positions based on a two-step process. The first step is to evaluate the tax position for recognition by determining if the weight of available evidence indicates that it is more likely than not that the position will be sustained on audit, including resolution of related appeals or litigation processes, if any. The second step is to measure the tax benefit as the largest amount that is more than 50% likely of being realized upon settlement. While the Company believes that it has appropriate support for the positions taken on its tax returns, the Company regularly assesses the potential outcome of examinations by tax authorities in determining the adequacy of its provision for income taxes.
As of December 31, 2019, the Company’s U.S. federal and state tax returns remain subject to examination by tax authorities beginning with the tax year ended December 31, 2016. However, due to NOLs and credit carryforwards being generated and carried forward from prior tax years, substantially all tax years may also be subject to examination.
During the years ended December 31, 2019 and 2018, the Company did not recognize any interest and penalties relating to taxes, nor were any accrued as of December 31, 2019 and 2018.
Note 10: Fair Value Measurements
Fair value is defined as the price that would be received to sell an asset in the principal or most advantageous market for the asset in an orderly transaction between market participants on the measurement date. Fair value should be based on the assumptions market participants would use when pricing an asset. U.S. Generally Accepted Accounting Principles establishes a fair value hierarchy that prioritizes investments based on those assumptions. The fair value hierarchy gives the highest priority to quoted prices in active markets (observable inputs) and the lowest priority to an entity’s assumptions (unobservable inputs). The Company groups assets at fair value in three Fair Value Measurements levels, based on the markets in which the assets and liabilities are traded, and the reliability of the assumptions used to determine fair value. These levels are as follows:
Level 1 – Unadjusted quoted market prices for identical assets or liabilities in active markets as of the measurement date.
Level 2 – Other observable inputs, either directly or indirectly, including:
| · | Quoted prices for similar assets/liabilities in active markets; | |
| · | Quoted prices for identical or similar assets in non-active markets; | |
| · | Inputs other than quoted prices that are observable for the asset/liability; and, | |
| · | Inputs that are derived principally from or corroborated by other observable market data. |
Level 3 – Unobservable inputs that cannot be corroborated by observable market data.
F-10
Virtuoso Surgical, Inc.
Notes to Financial Statements (Unaudited)
For the six months ended June 30, 2020 and 2019
The Company’s significant financial instruments are cash and cash equivalents and other short-term assets and liabilities. For these financial instruments carrying values approximate fair value.
Note 11: Recent Accounting Pronouncements
In February 2016, the FASB issued ASU 2016-02, Leases (Topic 842), which requires lessees to recognize assets and liabilities for most leases. The recognition, measurement, and presentation of expenses and cash flows arising from a lease by a lessee is not expected to significantly change under such guidance. The standard will be effective for annual reporting periods beginning after December 15, 2021. Accordingly, this ASU will be effective for the Company for the year ending December 31, 2022. Adoption of this ASU will not have a significant impact on the Company’s financial position and results of operations.
Note 12: Subsequent Events
The Company has evaluated subsequent events through September 23, 2020, the date which the financial statements were available to be issued. There were no subsequent events that need disclosure that have not already been disclosed.
F-11
INDEX OF EXHIBITS
* Filed as an exhibit to Virtuoso Surgical, Inc.’s Regulation A Offering Statement on Form 1-A on December 27, 2019 (Commission File No. 024-11136) and incorporated herein by reference.
** Filed as an exhibit to Amendment No. 1 to Virtuoso Surgical, Inc.’s Regulation A Offering Statement on Form 1-A on January 23, 2020 (Commission File No. 024-11136) and incorporated herein by reference.
7
Pursuant to the requirements of Regulation A, the issuer has duly caused this Report to be signed on its behalf by the undersigned, thereunto duly authorized.
| VIRTUOSO SURGICAL, INC. | ||
| By: | /s/ Robert Webster, III | |
| Robert Webster, III | ||
| President, Chief Technology Officer, and Director (principal executive officer) | ||
| Date: | September 28, 2020 | |
Pursuant to the requirements of Regulation A, this report has been signed below by the following persons on behalf of the issuer and in the capacities and on the dates indicated.
| Signature | Title | Date | ||
| /s/ Robert Webster, III | ||||
| Robert Webster, III | President, Chief Technology Officer, and Director (principal executive officer) | September 28, 2020 | ||
| /s/ C. Mark Pickrell | ||||
| C. Mark Pickrell | Chief Administrative Officer, General Counsel, Secretary, and Director (principal financial officer and principal accounting officer) | September 28, 2020 |
8