Free signup for more
- Track your favorite companies
- Receive email alerts for new filings
- Personalized dashboard of news and more
- Access all data and search results
Filing tables
Filing exhibits
BBIO similar filings
- 2 Nov 23 BridgeBio Pharma Reports Third Quarter 2023 Financial Results and Business Update
- 25 Sep 23 BridgeBio Pharma Announces $250 Million Private Placement Equity Financing
- 3 Aug 23 BridgeBio Pharma Reports Second Quarter 2023 Financial Results and Business Update
- 17 Jul 23 Regulation FD Disclosure
- 22 Jun 23 Submission of Matters to a Vote of Security Holders
- 4 May 23 Entry into a Material Definitive Agreement
- 4 May 23 BridgeBio Pharma Reports First Quarter 2023 Financial Results and Business Update
Filing view
External links
Exhibit 99.2

ATTRibute-CM Phase 3 Topline Results July 17, 2023 1

Forward-Looking Statements and Disclaimer 2 The presentation at the call may contain forward-looking statements. Statements made or presented at the call may include statements that are not historical facts and are considered forward-looking within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. Words such as “believe,” “anticipate,” “plan,” “expect,” “intend,” “will,” “may,” “goal,” “potential,” “should,” “could,” “aim,” “estimate,” “predict,” “continue” and similar expressions or the negative of these terms or other comparable terminology are intended to identify forward-looking statements, though not all forward-looking statements necessarily contain these identifying words. We intend these forward-looking statements to be covered by the safe harbor provisions for forward-looking statements contained in Section 27A of the Securities Act and Section 21E of the Exchange Act. These forward-looking statements, including statements relating to the clinical, therapeutic and market potential of our programs and product candidates, including our clinical development program for acoramidis for patients with transthyretin amyloid cardiomyopathy, the timing and success of our clinical development programs, the progress of our ongoing and planned clinical trials of acoramidis for patients with transthyretin amyloid cardiomyopathy, including our plans to file a new NDA with the FDA by end of year 2023, our planned interactions with regulatory authorities, the availability of data from our clinical trials of acoramidis, and the timing of these events, reflect our current views about our plans, intentions, expectations and strategies, which are based on the information currently available to us and on assumptions we have made. Such statements reflect the current views of the Company with respect to future events and are subject to known and unknown risks, including business, regulatory, economic and competitive risks, uncertainties, contingencies and assumptions about the Company, including, without limitation, risks inherent in developing therapeutic products, and those risks and uncertainties described under the heading “Risk Factors” in the Company’s most recent Annual Report on Form 10-K filed with the U.S. Securities and Exchange Commission (“SEC”) and in subsequent filings made by the Company with the SEC, which are available on the SEC’s website at www.sec.gov. In light of these risks and uncertainties, many of which are beyond the Company’s control, the events or circumstances referred to in the forward-looking statements, express or implied, may not occur. The actual results may vary from the anticipated results and the variations may be material. You are cautioned not to place undue reliance on these forward-looking statements, which speak to the Company’s current beliefs and expectations only as of the date of the call. Except as required by law, the Company disclaims any intention or responsibility for updating or revising any forward-looking statements made or presented at the call in the event of new information, future developments or otherwise. No representation is made as to the safety or effectiveness of the product candidates for the therapeutic use for which such product candidates are being studied. Certain information communicated at the call may relate to or is based on studies, publications, surveys and other data obtained from third-party sources and the Company’s own internal estimates and research. While the Company believes these third-party sources to be reliable as of the date of the call, it has not independently verified, and makes no representation as to the adequacy, fairness, accuracy or completeness of, any information obtained from third-party sources. In addition, certain information to be communicated at the call involves a number of assumptions and limitations, and there can be no guarantee as to the accuracy or reliability of such assumptions. Finally, such research has not been verified by any independent source. Such information is provided as of the date of the call and is subject to change without notice. The Company has not verified, and will not verify, any part of this presentation, and the Company makes no representation or warranty, express or implied, as to the accuracy or completeness of the information to be communicated at the call or as to the existence, substance or materiality of any information omitted from the presentation at the call. The Company disclaims any and all liability for any loss or damage (whether foreseeable or not) suffered or incurred by any person or entity as a result of anything contained or omitted from this document or the related presentation and such liability is expressly disclaimed.

Discussion topics Q&A Session 5 Introduction & Overview 1 Neil Kumar, PhD Chief Executive Officer ATTRibute-CM Phase 3 Topline Results 2 Jonathan Fox, MD, PhD Chief Medical Officer, Cardiorenal Next Steps 3 Uma Sinha, PhD Chief Scientific Officer Commercial Launch Plans 4 Matt Outten, MBA Chief Commercial Officer 3

4 4 A sincere THANK YOU to patients and families, advocates, physicians, clinical research staff, and collaborating research partners (acoramidis for ATTR-CM)

ATTR-CM Idea Phase 3 Achondroplasia Idea Phase 3 LGMD2i Idea ADH1 Idea Phase 3 CAH Idea Program context 5

Acoramidis was designed to achieve maximal stabilization and preserve native TTR TTR = Transthyretin; ATTR-CM = TTR amyloid cardiomyopathy. 1Hammarstrom, P et al., PNAS. 2002;99:16427-16432. 2Damy, T., et al., Eur J Heart Fail. 2021;23(2):277-285. 3Coelho, T. et al., Neurology. 2012;79:785–792. 4Berk, JL et al , JAMA. 2013;310:2658-2667. 5Adams, DA. et al., N Engl J Med. 2018;379:11-21. 6Benson, M.D., et al., N Engl J Med. 2018;379:22-31. 7Richardson SJ, et al. Front Endocrinol. 2015;5:1-9. 6 Design Objectives Rationale Maximize TTR stabilization/minimize toxic monomer Preserve circulating native TTR Strong genotype/phenotype correlation between TTR instability and disease severity1 Dose-dependent improvements in both TTR stabilization and clinical outcomes demonstrated by tafamidis in ATTR-CM2 Extent of TTR stabilization or knockdown associated with degree of clinical benefit in ATTR-PN3-6 TTR has been highly conserved throughout evolution7 TTR is an abundant plasma protein with relatively rapid turnover requiring sustained metabolic energy expenditure 1 2 We plan to enter the ATTR-CM market with acoramidis, a next generation, more potent TTR stabilizer

Acoramidis is a next generation stabilizer that employs multiple strategies to maximize potency 7 Native TTR circulates in blood as a tetramer Dissociation into monomers initiates pathogenesis Monomers aggregate, causing disease Acoramidis was designed to mimic protective T119M mutation. Acoramidis sees more target (superior free fraction), binds more target (superior kd2), and glues the target together stronger (enthalpic binding mode).1,2 Disease mechanism Therapeutic hypothesis ~130 known destabilizing mutations ProtectiveT119M mutation 7 Acoramidis is an investigational molecule. The safety and efficacy have not been established by regulatory authorities. 1Data on File. 2Miller, M. et al. J Med Chem. 2018;61:7862-7876.

Data supporting more potent TTR stabilization 8 Near-Complete TTR Stabilization2 at target trough clinical concentrations Rapid, durable increases in serum TTR an in vivo marker of native tetramer stability Superior Binding to TTR in vitro1 facilitated by enthalpic interactions Tafamidis 20 mg Tafamidis 80 mg Acoramidis HCl 800 mg 22% 30% 39% ATTR-ACT Month 12 Data3 ATTRibute-CM Month 12 Data4 Kcal/mol -15 -12 -9 -6 -3 0 3 -15 -12 -9 -6 -3 0 3 Favorable Unfavorable Acoramidis Tafamidis △G △H -T△S DSMO TAF 16µM TAF 26µM Acoramidis 10µM 27% 57% 66% ~100% % Stabilization % Increase in Serum TTR 1Miller, M. et al. J Med Chem. 2018;61:7862-7876. 2Ji, A.X., et al. American Heart Association Scientific Sessions, 2019. 3Estimated from Damy, T., et al., Eur J Heart Fail. 2021;23(2):277-285. 4BridgeBio Part A press release, December 27, 2021. Note: Direct cross-study comparisons may suggest misleading similarities or differences. The values shown are directional and do not report robust comparative analysis.

Unprecedented and consistent benefit on survival and morbidity Achieve statistical significance on primary endpoint:p-value <0.04 Primary endpoint met (p<0.0001) Unprecedented survival: Highest ever 30-month survival rate on drug (>80%) with clinically meaningful separation from placebo 81% 30-month survival on acoramidis 6.4% absolute & 25% relative risk reduction compared to placebo Best-in-class CVH data: Profound reduction in event rates 50% relative risk reduction for cumulative frequency of CVH (p<0.0001) Win Ratio better than 1.7: Significant impact on mortality and morbidity Win Ratio = 1.8 Best-in-class treatment effect on serum biomarkers: NT-proBNP, serum TTR, TTR stabilization Clinically and statistically significant (p<0.0001) benefit on NT-proBNP and serum TTR; sustained impact on TTR stabilization Best Case Target Clinical Profile Outcome Observed 9 CVH = Cardiovascular-related hospitalization; NT-proBNP = N-terminal prohormone of brain natriuretic peptide.

Discussion topics Q&A Session 5 Introduction & Overview 1 Neil Kumar, PhD Chief Executive Officer ATTRibute-CM Phase 3 Topline Results 2 Jonathan Fox, MD, PhD Chief Medical Officer, Cardiorenal Next Steps 3 Uma Sinha, PhD Chief Scientific Officer Commercial Launch Plans 4 Matt Outten, MBA Chief Commercial Officer 10
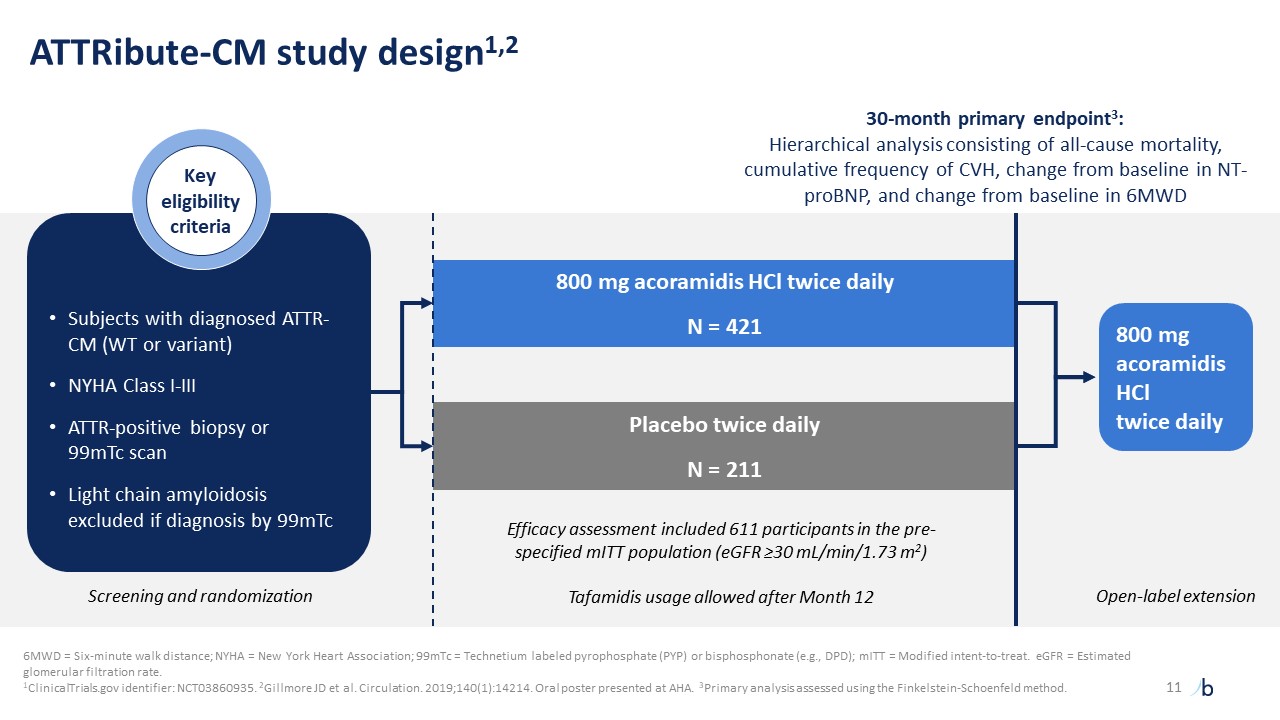
ATTRibute-CM study design1,2 Screening and randomization Open-label extension 30-month primary endpoint3: Hierarchical analysis consisting of all-cause mortality, cumulative frequency of CVH, change from baseline in NT-proBNP, and change from baseline in 6MWD Efficacy assessment included 611 participants in the pre-specified mITT population (eGFR ≥30 mL/min/1.73 m2) Tafamidis usage allowed after Month 12 800 mg acoramidis HCl twice daily N = 421 Placebo twice daily N = 211 11 800 mg acoramidis HCltwice daily Subjects with diagnosed ATTR-CM (WT or variant) NYHA Class I-III ATTR-positive biopsy or 99mTc scan Light chain amyloidosis excluded if diagnosis by 99mTc Keyeligibilitycriteria 6MWD = Six-minute walk distance; NYHA = New York Heart Association; 99mTc = Technetium labeled pyrophosphate (PYP) or bisphosphonate (e.g., DPD); mITT = Modified intent-to-treat. eGFR = Estimated glomerular filtration rate. 1ClinicalTrials.gov identifier: NCT03860935. 2Gillmore JD et al. Circulation. 2019;140(1):14214. Oral poster presented at AHA. 3Primary analysis assessed using the Finkelstein-Schoenfeld method.

Hierarchical analysis consisting of: All-cause mortality2 Cumulative frequency of CVH Change from baseline in NT-proBNP Change from baseline in 6MWD p<0.0001 Win Ratio 1.8 Cumulative frequency of CVH3 p<0.0001 Change from baseline in 6MWD4 p<0.0001 Change from baseline in KCCQ-OS4 p<0.0001 Change from baseline in serum TTR4 p<0.0001 Change from baseline in NT-proBNP5 p<0.0001 All-cause mortality2,6 p=0.057 Primary endpoint1 p-value Select secondary endpoints p-value Highly statistically significant result achieved on primary and select secondary endpoints 12 KCCQ-OS = Kansas City cardiomyopathy questionnaire overall summary score. 1Primary analysis assessed using the Finkelstein-Schoenfeld method. 2Heart transplant and implantation of cardiac mechanical assistance device were treated as death for this analysis. 3Negative binomial regression with treatment group, stratification factors and the offset term is used to analyze the cumulative frequency of adjudicated CV-related hospitalization.4Least squares mean difference change from baseline at 30 months. 5Ratio of adjusted geometric mean fold change from baseline at 30 months. 6Assessed by Cochran-Mantel-Haenszel test; p=0.15 as assessed by Cox Proportional Hazard Model. 58% of ties broken by first two components of Win Ratio analysis

No safety signals of potential clinical concern identified 13 Any treatment-emergent adverse events (TEAEs) 413 (98.1%) 206 (97.6%) TEAE with fatal outcome 60 (14.3%) 36 (17.1%) TEAE leading to hospitalization 212 (50.4%) 128 (60.7%) TEAE leading to study drug discontinuation 39 (9.3%) 18 (8.5%) Any treatment-emergent serious adverse events (SAEs) 230 (54.6%) 137 (64.9%) Treatment-emergent SAEs leading to study drug discontinuation 21 (5.0%) 15 (7.1%) Severe TEAEs1 157 (37.3%) 96 (45.5%) Subjects with one or more event(s) AcoramidisN=421 N (%) PlaceboN=211 N (%) All Adverse Events (AEs) occurring during the treatment period are considered treatment-emergent adverse events (TEAEs). Serious Adverse Event (SAE) meets seriousness criteria. 1Severity as assessed by the investigator.

Exploratory post hoc analysis: serum TTR levels Serum TTR Level Mean Change from Baseline1 (mg/dL) +42% Placebo + Tafamidis2 35 Placebo 100 Acoramidis 234 N = 14 1Mean change from baseline in serum TTR at Month 30 in mITT population. 2Mean exposure on tafamidis = 11 months in mITT population. Acoramidis + Tafamidis2 49

Exploratory post hoc analysis: median NT-proBNP NT-proBNP Level Median Change from Baseline1 (pg/mL) Placebo + Tafamidis2 35 Placebo 98 Acoramidis 232 N = 15 1Median change from baseline in NT-proBNP at Month 30 in mITT population. 2Mean exposure on tafamidis = 11 months in mITT population. Acoramidis + Tafamidis2 48 9% experienced improvement from baseline 45% experienced improvement from baseline Worse Better

Summary of topline results 16 Unprecedented 30-month survival of >80% for a targeted intervention in ATTR-CM Achieved primary endpoint with highly statistically significant result with Win Ratio of 1.8 6.4% ARR & 25% RRR in all-cause mortality 50% RRR for cumulative frequency of CVH Well-tolerated with no safety signals of potential clinical concern Acoramidis was observed to consistently outperform placebo on survivaland established measures of ATTR-CM morbidity ARR = Absolute risk reduction. RRR = Relative risk reduction.
Discussion topics Q&A Session 5 Introduction & Overview 1 Neil Kumar, PhD Chief Executive Officer ATTRibute-CM Phase 3 Topline Results 2 Jonathan Fox, MD, PhD Chief Medical Officer, Cardiorenal Next Steps 3 Uma Sinha, PhD Chief Scientific Officer Commercial Launch Plans 4 Matt Outten, MBA Chief Commercial Officer 17

First regulatory submission planned for year-end 2023 Present ATTRibute-CM Primary Results European Society of Cardiology 2023 August 27th, 2023 File New Drug Application (NDA) with FDA End of 2023 Submit additional regulatory filings (EMA & others) 2024 Execute lifecycle management Initiate primary prevention study (ACT-EARLY) 2024 18

Discussion topics Q&A Session 5 Introduction & Overview 1 Neil Kumar, PhD Chief Executive Officer ATTRibute-CM Phase 3 Topline Results 2 Jonathan Fox, MD, PhD Chief Medical Officer, Cardiorenal Next Steps 3 Uma Sinha, PhD Chief Scientific Officer Commercial Launch Plans 4 Matt Outten, MBA Chief Commercial Officer 19

Commercial launch plans 20 We have a world-class commercial team and we are prepared to go to market 20+ FTEs (Pharmacyclics, Vertex, and Schering Plough alumni) and a distinctive commercial advisory board, inclusive of Fred Hassan, Jennifer Cook and Jim Robinson Have initiated discussions with key partners (payers and distributors) to bring this drug to patients Our goal is to continue working closely with current and future partners to bring this next generation stabilizer to as broad a patient and provider community as possible Access Global reach More details on commercial execution to come

ATTRibute-CM Phase 3 Topline Results Q&A Session 21