CYNK-001 ACUTE MYELOID LEUKEMIA UPDATE December 17, 2021 Exhibit 99.1


Address 3 Co-dependent variables affecting CYNK-001 potency and persistence in AML: CYNK-001 Cell dose Cyclophosphamide dose [IL-15 and Treg levels] IL-2 Use Determine optimal lymphodepletion regimen that: Enables safe outpatient administration Results in adequate IL-15 serum levels to promote CYNK-001 expansion, persistence and potency (Leclercq G. et al J Exp Med 184, 325-336 1996) Results in maximum suppression of T regulatory cells to enable co-administration of IL-2 to promote CYNK-001 expansion, persistence and potency (Miller J.S. et al Blood 105, 3051-3057 2005) Determine maximal tolerated CYNK-001 dosing regimen associated with greatest biologic effect size and durability during the “Window of Dosing Opportunity” when IL15 serum levels are elevated and T regulatory cells are low In patients with minimum residual disease (MRD) AML In patients with relapsed refractory AML CYNK-001 Acute myeloid leukemia (AML) Phase 1 Trial Goals
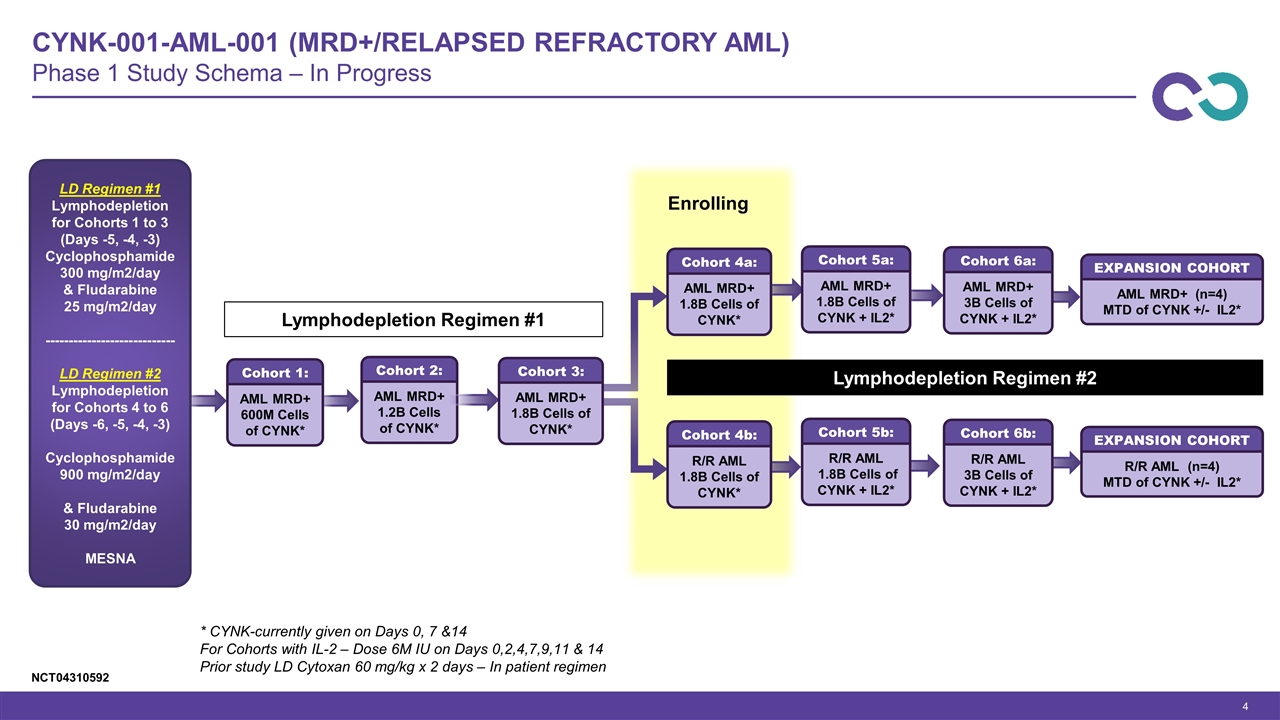
CYNK-001-AML-001 (MRD+/Relapsed Refractory AML) Phase 1 Study Schema – In Progress LD Regimen #1 Lymphodepletion for Cohorts 1 to 3 (Days -5, -4, -3) Cyclophosphamide 300 mg/m2/day & Fludarabine 25 mg/m2/day ---------------------------- LD Regimen #2 Lymphodepletion for Cohorts 4 to 6 (Days -6, -5, -4, -3) Cyclophosphamide 900 mg/m2/day & Fludarabine 30 mg/m2/day MESNA AML MRD+ 600M Cells of CYNK* Cohort 1: AML MRD+ 1.2B Cells of CYNK* Cohort 2: AML MRD+ 1.8B Cells of CYNK* Cohort 4a: R/R AML 1.8B Cells of CYNK* Cohort 4b: AML MRD+ 1.8B Cells of CYNK + IL2* Cohort 5a: R/R AML 1.8B Cells of CYNK + IL2* Cohort 5b: AML MRD+ 3B Cells of CYNK + IL2* Cohort 6a: R/R AML 3B Cells of CYNK + IL2* Cohort 6b: AML MRD+ (n=4) MTD of CYNK +/- IL2* EXPANSION COHORT R/R AML (n=4) MTD of CYNK +/- IL2* EXPANSION COHORT * CYNK-currently given on Days 0, 7 &14 For Cohorts with IL-2 – Dose 6M IU on Days 0,2,4,7,9,11 & 14 Prior study LD Cytoxan 60 mg/kg x 2 days – In patient regimen AML MRD+ 1.8B Cells of CYNK* Cohort 3: Enrolling Lymphodepletion Regimen #1 Lymphodepletion Regimen #2 NCT04310592
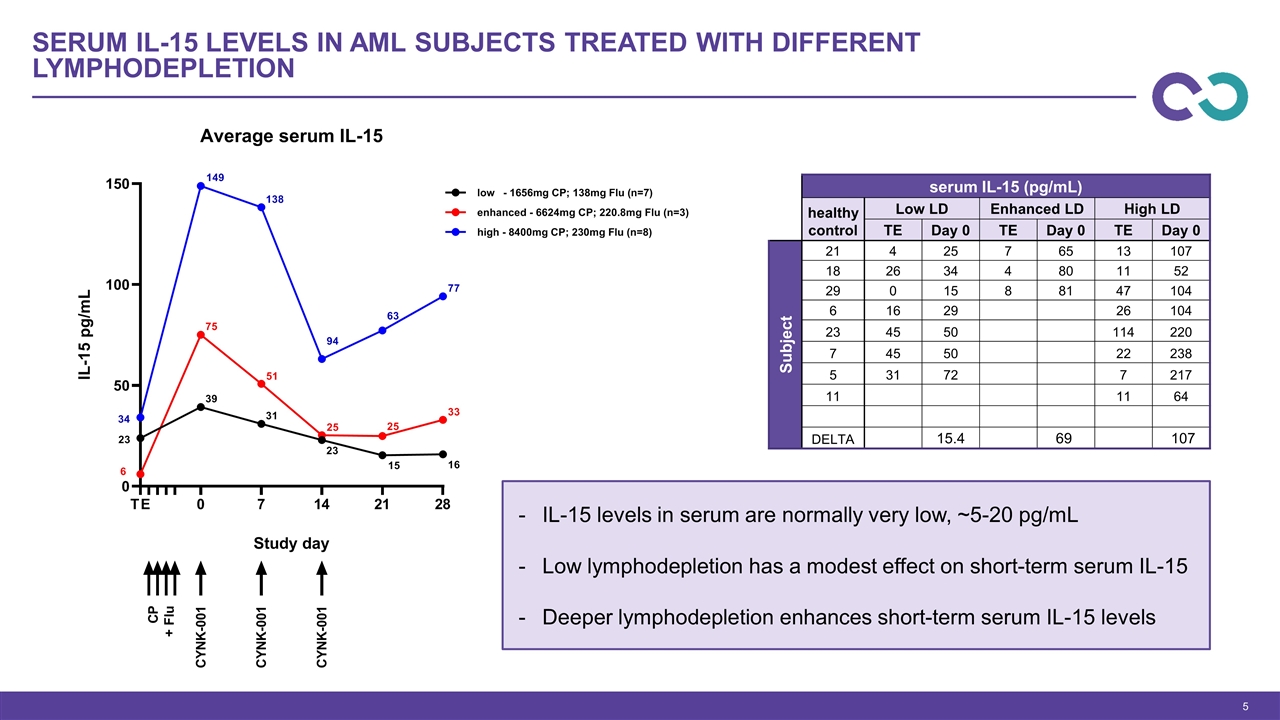
Serum IL-15 levels in AML subjects treated with different lymphodepletion IL-15 levels in serum are normally very low, ~5-20 pg/mL Low lymphodepletion has a modest effect on short-term serum IL-15 Deeper lymphodepletion enhances short-term serum IL-15 levels serum IL-15 (pg/mL) healthy control Low LD Enhanced LD High LD TE Day 0 TE Day 0 TE Day 0 Subject 21 4 25 7 65 13 107 18 26 34 4 80 11 52 29 0 15 8 81 47 104 6 16 29 26 104 23 45 50 114 220 7 45 50 22 238 5 31 72 7 217 11 11 64 DELTA 15.4 69 107
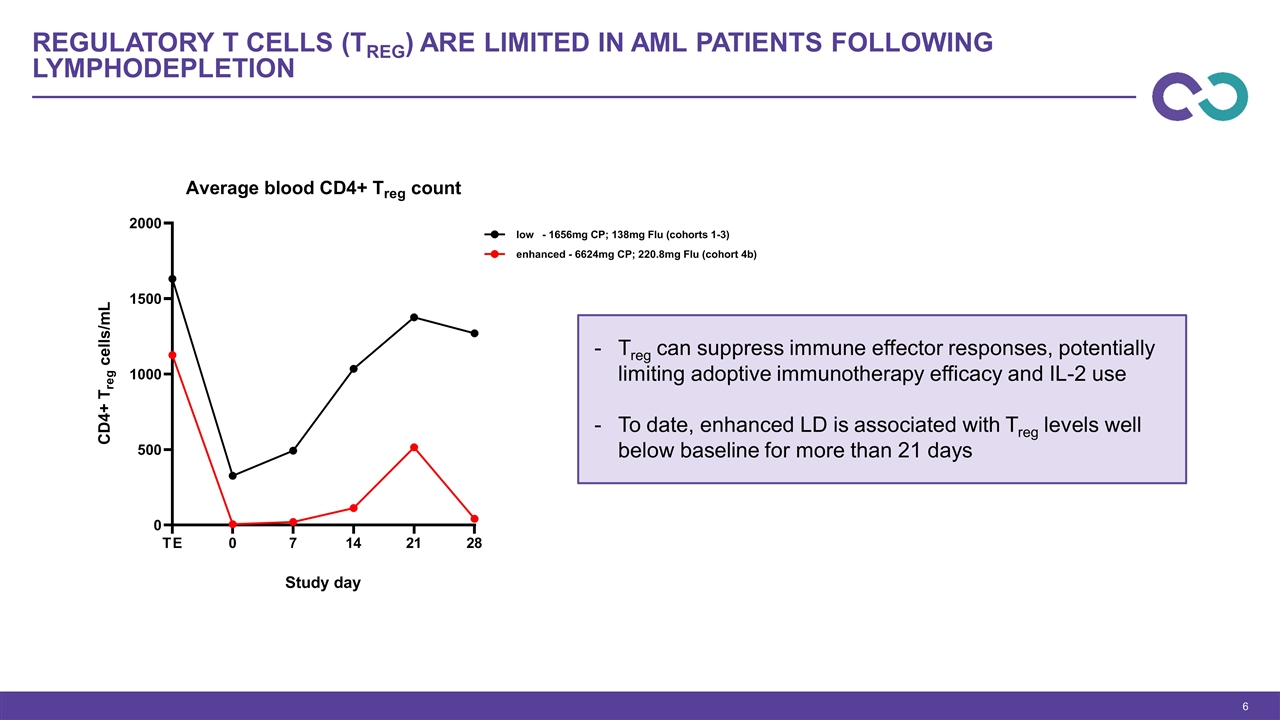
Regulatory T cells (Treg) are limited in AML patients following lymphodepletion Treg can suppress immune effector responses, potentially limiting adoptive immunotherapy efficacy and IL-2 use To date, enhanced LD is associated with Treg levels well below baseline for more than 21 days
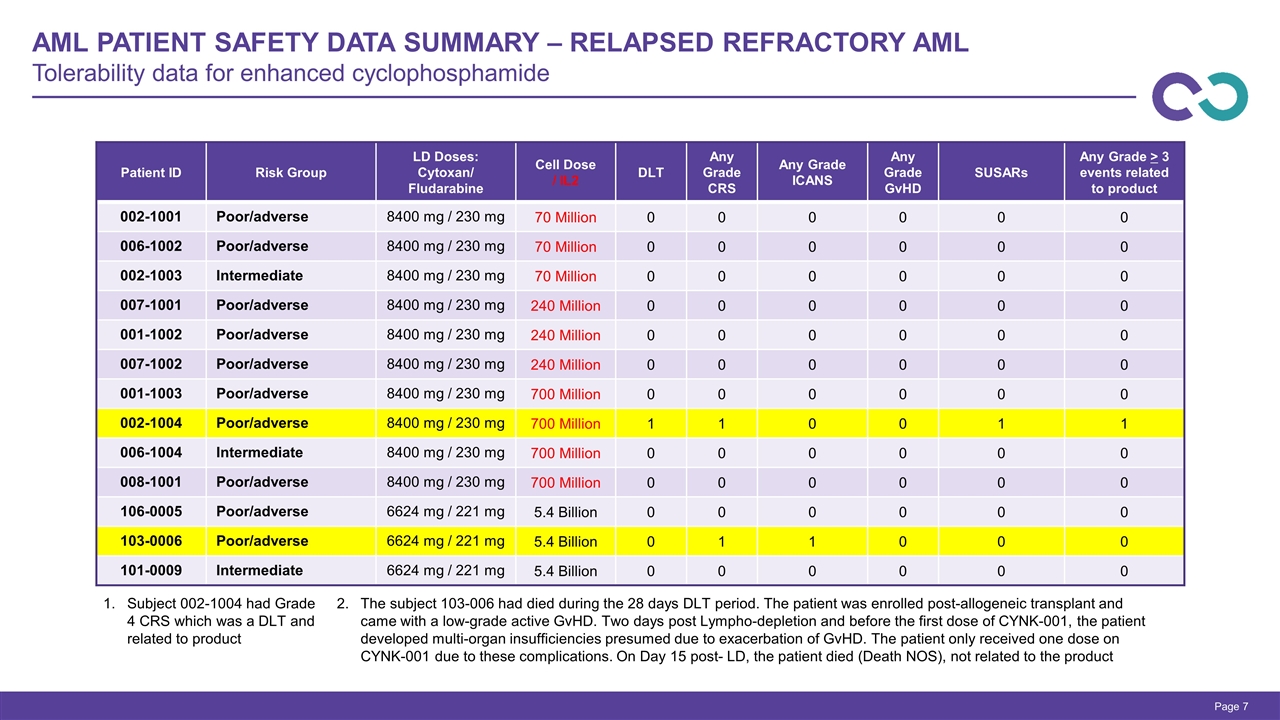
Page Subject 002-1004 had Grade 4 CRS which was a DLT and related to product 2.The subject 103-006 had died during the 28 days DLT period. The patient was enrolled post-allogeneic transplant and came with a low-grade active GvHD. Two days post Lympho-depletion and before the first dose of CYNK-001, the patient developed multi-organ insufficiencies presumed due to exacerbation of GvHD. The patient only received one dose on CYNK-001 due to these complications. On Day 15 post- LD, the patient died (Death NOS), not related to the product AML Patient Safety Data Summary – Relapsed Refractory AML Tolerability data for enhanced cyclophosphamide Patient ID Risk Group LD Doses: Cytoxan/ Fludarabine Cell Dose / IL2 DLT Any Grade CRS Any Grade ICANS Any Grade GvHD SUSARs Any Grade > 3 events related to product 002-1001 Poor/adverse 8400 mg / 230 mg 70 Million 0 0 0 0 0 0 006-1002 Poor/adverse 8400 mg / 230 mg 70 Million 0 0 0 0 0 0 002-1003 Intermediate 8400 mg / 230 mg 70 Million 0 0 0 0 0 0 007-1001 Poor/adverse 8400 mg / 230 mg 240 Million 0 0 0 0 0 0 001-1002 Poor/adverse 8400 mg / 230 mg 240 Million 0 0 0 0 0 0 007-1002 Poor/adverse 8400 mg / 230 mg 240 Million 0 0 0 0 0 0 001-1003 Poor/adverse 8400 mg / 230 mg 700 Million 0 0 0 0 0 0 002-1004 Poor/adverse 8400 mg / 230 mg 700 Million 1 1 0 0 1 1 006-1004 Intermediate 8400 mg / 230 mg 700 Million 0 0 0 0 0 0 008-1001 Poor/adverse 8400 mg / 230 mg 700 Million 0 0 0 0 0 0 106-0005 Poor/adverse 6624 mg / 221 mg 5.4 Billion 0 0 0 0 0 0 103-0006 Poor/adverse 6624 mg / 221 mg 5.4 Billion 0 1 1 0 0 0 101-0009 Intermediate 6624 mg / 221 mg 5.4 Billion 0 0 0 0 0 0
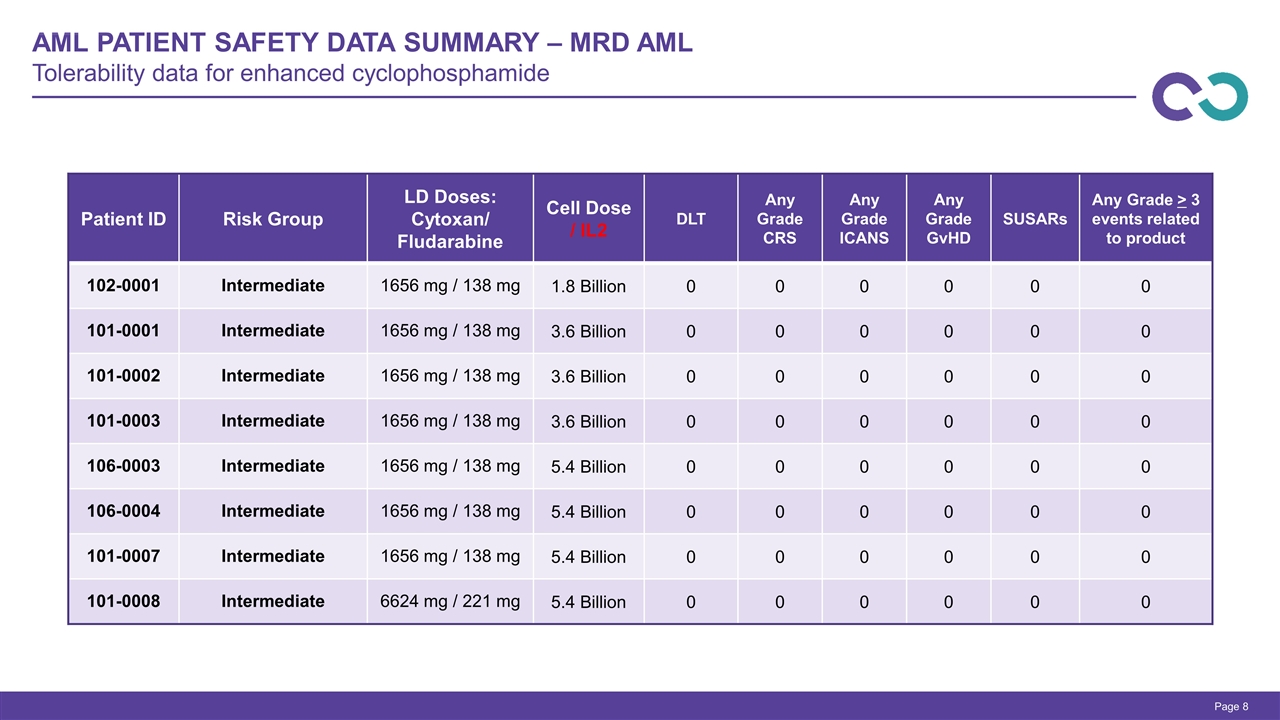
AML Patient safety Data Summary – MRD AML Tolerability data for enhanced cyclophosphamide Page Patient ID Risk Group LD Doses: Cytoxan/ Fludarabine Cell Dose / IL2 DLT Any Grade CRS Any Grade ICANS Any Grade GvHD SUSARs Any Grade > 3 events related to product 102-0001 Intermediate 1656 mg / 138 mg 1.8 Billion 0 0 0 0 0 0 101-0001 Intermediate 1656 mg / 138 mg 3.6 Billion 0 0 0 0 0 0 101-0002 Intermediate 1656 mg / 138 mg 3.6 Billion 0 0 0 0 0 0 101-0003 Intermediate 1656 mg / 138 mg 3.6 Billion 0 0 0 0 0 0 106-0003 Intermediate 1656 mg / 138 mg 5.4 Billion 0 0 0 0 0 0 106-0004 Intermediate 1656 mg / 138 mg 5.4 Billion 0 0 0 0 0 0 101-0007 Intermediate 1656 mg / 138 mg 5.4 Billion 0 0 0 0 0 0 101-0008 Intermediate 6624 mg / 221 mg 5.4 Billion 0 0 0 0 0 0
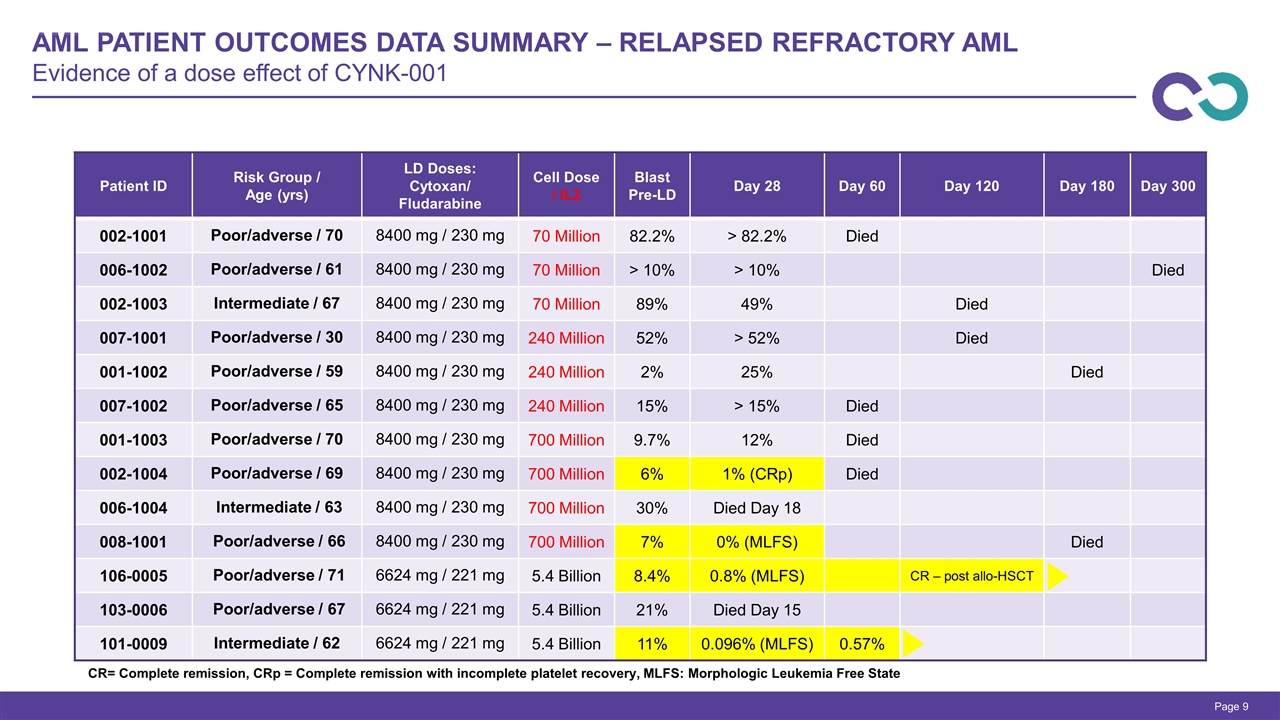
AML Patient Outcomes Data Summary – Relapsed Refractory AML Evidence of a dose effect of CYNK-001 Page CR= Complete remission, CRp = Complete remission with incomplete platelet recovery, MLFS: Morphologic Leukemia Free State Patient ID Risk Group / Age (yrs) LD Doses: Cytoxan/ Fludarabine Cell Dose / IL2 Blast Pre-LD Day 28 Day 60 Day 120 Day 180 Day 300 002-1001 Poor/adverse / 70 8400 mg / 230 mg 70 Million 82.2% > 82.2% Died 006-1002 Poor/adverse / 61 8400 mg / 230 mg 70 Million > 10% > 10% Died 002-1003 Intermediate / 67 8400 mg / 230 mg 70 Million 89% 49% Died 007-1001 Poor/adverse / 30 8400 mg / 230 mg 240 Million 52% > 52% Died 001-1002 Poor/adverse / 59 8400 mg / 230 mg 240 Million 2% 25% Died 007-1002 Poor/adverse / 65 8400 mg / 230 mg 240 Million 15% > 15% Died 001-1003 Poor/adverse / 70 8400 mg / 230 mg 700 Million 9.7% 12% Died 002-1004 Poor/adverse / 69 8400 mg / 230 mg 700 Million 6% 1% (CRp) Died 006-1004 Intermediate / 63 8400 mg / 230 mg 700 Million 30% Died Day 18 008-1001 Poor/adverse / 66 8400 mg / 230 mg 700 Million 7% 0% (MLFS) Died 106-0005 Poor/adverse / 71 6624 mg / 221 mg 5.4 Billion 8.4% 0.8% (MLFS) CR – post allo-HSCT 103-0006 Poor/adverse / 67 6624 mg / 221 mg 5.4 Billion 21% Died Day 15 101-0009 Intermediate / 62 6624 mg / 221 mg 5.4 Billion 11% 0.096% (MLFS) 0.57%
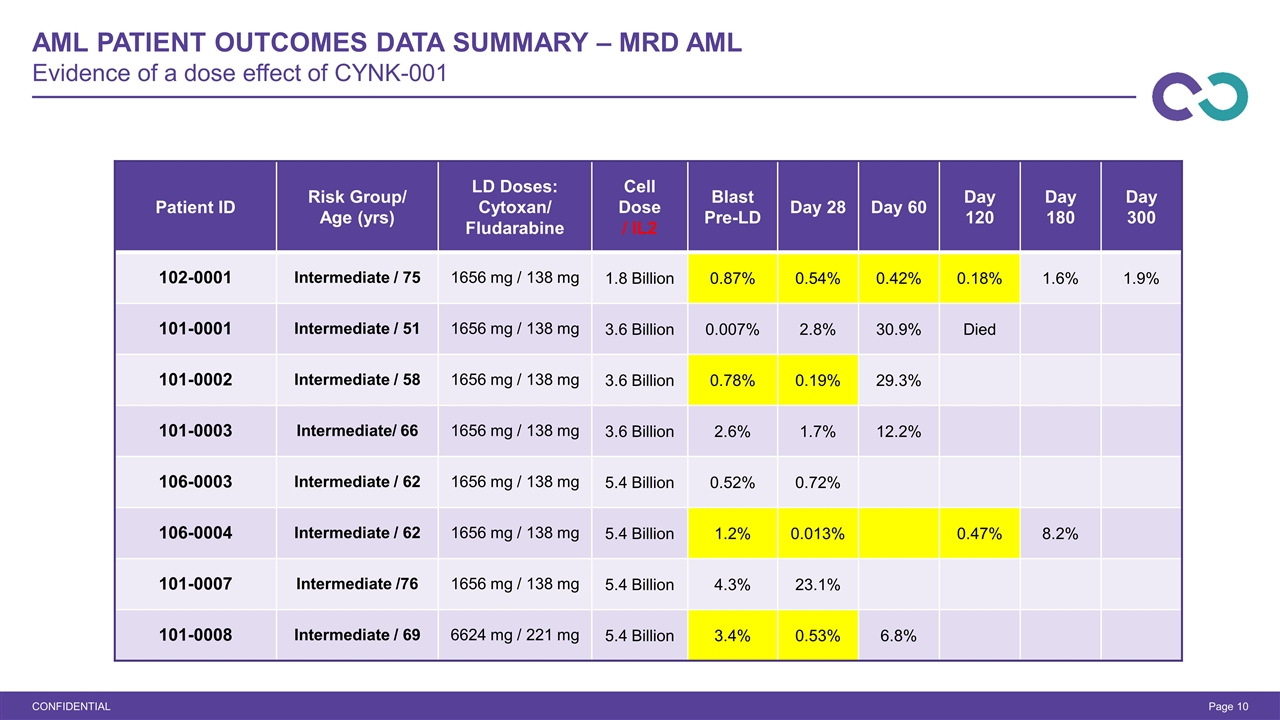
AML Patient Outcomes Data Summary – MRD AML Evidence of a dose effect of CYNK-001 CONFIDENTIAL Page Patient ID Risk Group/ Age (yrs) LD Doses: Cytoxan/ Fludarabine Cell Dose / IL2 Blast Pre-LD Day 28 Day 60 Day 120 Day 180 Day 300 102-0001 Intermediate / 75 1656 mg / 138 mg 1.8 Billion 0.87% 0.54% 0.42% 0.18% 1.6% 1.9% 101-0001 Intermediate / 51 1656 mg / 138 mg 3.6 Billion 0.007% 2.8% 30.9% Died 101-0002 Intermediate / 58 1656 mg / 138 mg 3.6 Billion 0.78% 0.19% 29.3% 101-0003 Intermediate/ 66 1656 mg / 138 mg 3.6 Billion 2.6% 1.7% 12.2% 106-0003 Intermediate / 62 1656 mg / 138 mg 5.4 Billion 0.52% 0.72% 106-0004 Intermediate / 62 1656 mg / 138 mg 5.4 Billion 1.2% 0.013% 0.47% 8.2% 101-0007 Intermediate /76 1656 mg / 138 mg 5.4 Billion 4.3% 23.1% 101-0008 Intermediate / 69 6624 mg / 221 mg 5.4 Billion 3.4% 0.53% 6.8%
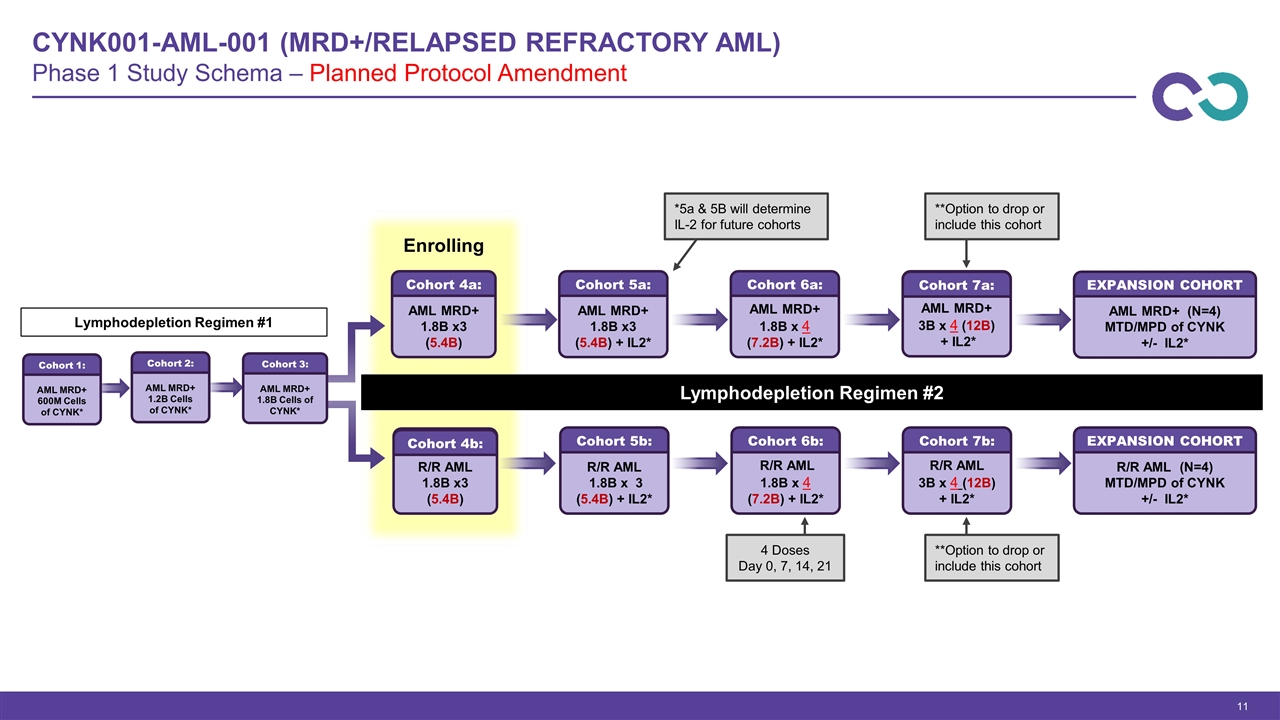
CYNK001-AML-001 (MRD+/Relapsed Refractory AML) Phase 1 Study Schema – Planned Protocol Amendment AML MRD+ 1.8B x3 (5.4B) Cohort 4a: R/R AML 1.8B x3 (5.4B) Cohort 4b: AML MRD+ 1.8B x3 (5.4B) + IL2* Cohort 5a: R/R AML 1.8B x 3 (5.4B) + IL2* Cohort 5b: AML MRD+ 1.8B x 4 (7.2B) + IL2* Cohort 6a: R/R AML 3B x 4 (12B) + IL2* Cohort 7b: Lymphodepletion Regimen #2 AML MRD+ 3B x 4 (12B) + IL2* Cohort 7a: R/R AML 1.8B x 4 (7.2B) + IL2* Cohort 6b: *5a & 5B will determine IL-2 for future cohorts Enrolling **Option to drop or include this cohort **Option to drop or include this cohort R/R AML (N=4) MTD/MPD of CYNK +/- IL2* EXPANSION COHORT AML MRD+ (N=4) MTD/MPD of CYNK +/- IL2* EXPANSION COHORT AML MRD+ 600M Cells of CYNK* Cohort 1: AML MRD+ 1.2B Cells of CYNK* Cohort 2: AML MRD+ 1.8B Cells of CYNK* Cohort 3: Lymphodepletion Regimen #1 4 Doses Day 0, 7, 14, 21

Relapsed refractory cohort 4b expanded to 6 patients due to a DLT that was unrelated to CYNK-001 MRD cohorts screen rate is 1 in every 4 patients Window of dosing opportunity is out past 21 days, plan to add a fourth dose on Day 21 Currently adding new sites to accelerate and complete dose cohorts to proceed to expansion CYNK-001 Acute myeloid leukemia program
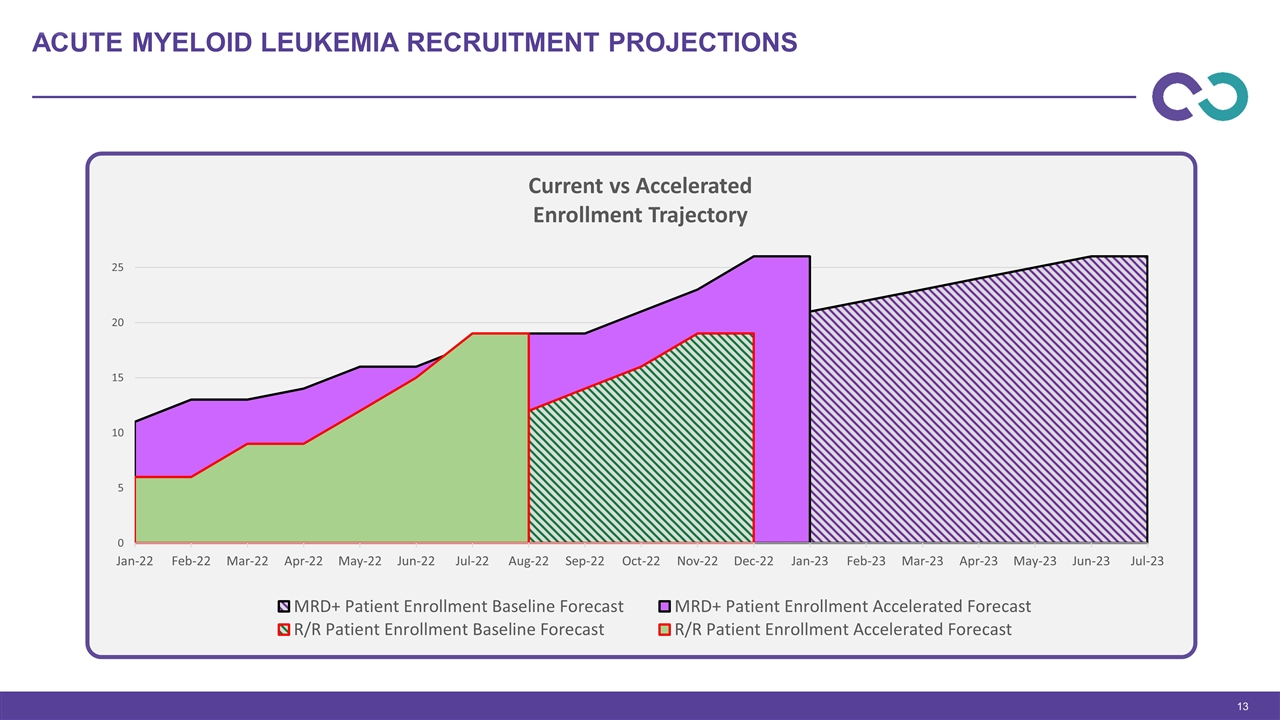
Acute Myeloid Leukemia Recruitment Projections

RECRUITED Investigative sites Page Currently Active Sites

AML annual incidence 20,240 Estimated patients eligible for CYNK-001 annually 11,000 Current Cynk-001 production potential Placenta CD34/Placenta Current 35 Day Process (Minaris Spinner Flask (SF) Process) Expected 35 Day Process (CELU Xuri Process) 1 2 Million 25 Billion (process not amenable to expansion) 50 Billion – 200 Billion (expandable process) 200/week 400 Million 5,000 Billion 10,000 – 40,000 Billion Production Metrics Total available market = 11,000 AML patients/52 weeks/year= 211 Patients/week CD34 7.2 billion CYNK-001 cell dose/patient X 211 Patients/week = 1,519 Billion CYNK-001 cells needed/week 1,519 Billion CYNK-001 cells needed each week / (25 Billion CYNK-001 cells current manufactured/placenta) = 60 Placentas/week with current SF process or 8 - 30 Placentas/week with expandable Xuri process

Enhanced LD with Regimen 2 is associated with increased IL-15 and suppressed Treg cell levels for over 21 days CYNK-001 has demonstrated evidence of a dose effect and evidence of biologic activity In our next cohorts we plan to add IL-2, add a 4th dose on day 21 and potentially increase the dose to 3 Billion CYNK-001 cells/infusion given the window of dosing opportunity observed We are adding more sites to accelerate accrual fINDINGs To Date