
Exhibit 99.2 Clinical Stage Neurology Company Focused on Neuronal Excitability Disorders Corporate Presentation | October 5, 2022

Forward-Looking Statements This presentation contains forward-looking statements that involve substantial risks and uncertainties. All statements other than statements of historical facts contained in this presentation, including statements regarding our future financial condition, results of operations, business strategy and plans, and objectives of management for future operations, as well as statements regarding industry trends, are forward-looking statements. In some cases, you can identify forward-looking statements by terminology such as “anticipate,” “believe,” “continue,” “could,” “estimate,” “expect,” “intend,” “may,” “plan,” “potentially,” “predict,” “should,” “will” or the negative of these terms or other similar expressions. We have based these forward-looking statements largely on our current expectations and projections about future events and trends that we believe may affect our financial condition, results of operations, business strategy and financial needs. These forward-looking statements are subject to a number of risks, uncertainties and assumptions, including, among other things, risks related to: the success, cost and timing of our product development activities and clinical trials; our expectations about the timing of achieving regulatory approval and the cost of our development programs; our ability to obtain funding for our operations, including funding necessary to complete further development and commercialization of our product candidates; the impact of the COVID-19 pandemic on our operations; the commercialization of our product candidates, if approved; our plans to research, develop and commercialize our product candidates; our plans to develop additional product candidates; our ability to obtain, maintain, expand, protect and enforce our intellectual property rights; our ability to operate our business without infringing, misappropriating or otherwise violating the intellectual property rights of third parties; our ability to attract collaborators with development, regulatory and commercialization expertise; future agreements with third parties in connection with the commercialization of our product candidates; the size and growth potential of the markets for our product candidates and our ability to serve those markets; the rate and degree of market acceptance of our product candidates; regulatory developments in the United States and foreign countries; regulatory application, review and approval processes and our compliance with applicable legal and regulatory requirements; our ability to contract with third-party suppliers and manufacturers and their ability to perform adequately; the success of competing products that are or may become available; and our ability to attract and retain key scientific or management personnel. These risks are not exhaustive. New risk factors emerge from time to time and it is not possible for our management to predict all risk factors, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in, or implied by, any forward-looking statements. More information about the risks and uncertainties faced by Eliem is contained under the caption “Risk Factors” set forth in Eliem’s Quarterly Report on Form 10-Q for the quarterly period ended June 30, 2022, which is available on the SEC’s website at www.sec.gov, and in other subsequent reports and filings Eliem will make with the SEC from time to time. You should not rely upon forward-looking statements as predictions of future events. Although we believe that the expectations reflected in the forward-looking statements are reasonable, we cannot guarantee future results, levels of activity, performance or achievements. Except as required by law, we undertake no obligation to update publicly any forward-looking statements for any reason after the date of this presentation. Certain information contained in this presentation relates to, or is based on, studies, publications, surveys and other data obtained from third-party sources and the Company's own internal estimates and research. While we believe these third-party sources to be reliable as of the date of this presentation, we have not independently verified, and make no representation as to the adequacy, fairness, accuracy or completeness of, any information obtained from third-party sources. In addition, all of the market data included in this presentation involves a number of assumptions and limitations, and there can be no guarantee as to the accuracy or reliability of such assumptions. In addition, projections, assumptions and estimates of our future performance and the future performance of the markets in which we operate are necessarily subject to a high degree of uncertainty and risk. These and other factors could cause results to differ materially from those expressed in the estimates made by the independent parties and by us. Finally, while we believe our own internal research is reliable, such research has not been verified by any independent source. In addition, statements that “we believe” and similar statements reflect our beliefs and opinions on the relevant subject. These statements are based upon information available to us as of the date of this presentation, and while we believe such information forms a reasonable basis for such statements, such information may be limited or incomplete, and our statements should not be read to indicate that we have conducted an exhaustive inquiry into, or review of, all potentially available relevant information. These statements are inherently uncertain and investors are cautioned not to unduly rely upon these statements. 2

Highly experienced management team Clinical and preclinical pipeline based on clinically validated mechanisms of action Rethinking Two differentiated programs in depression and epilepsy with treatment for expansion opportunities in chronic pain nervous system disorders ~$130M* cash runway into 2025 allows for topline clinical data readouts and advancement of preclinical asset into clinic * Unaudited cash, cash equivalents and investments as of September 30, 2022, 3 including receipt of $6.2m in tax reimbursements within the quarter

Deep expertise in neuroscience Powered by successful and talented research, clinical development executives from pioneering organizations and commercialization • Lyrica, Neurontin, Trobalt, Vyepti, Vixotrigine, Nimotop, Aptiom, Lunesta, Geodon General Management, Commercial & Corporate Development Leadership experience in both large pharma and small biotech Robert Azelby, MBA Erin Lavelle James Bucher, J.D. • Large: Amgen, GSK, Novartis, Biogen, Chief Executive Officer Chief Operating Officer & EVP and General Counsel Bayer, Pfizer Chief Financial Officer • Small: Alder, Juno, Convergence, Cavion, Exelixis Research & Development Highly skilled in public/private capital raising and corporate development with successful exits Valerie Morisset, Ph.D. Joanne Palmer, Ph.D. Mark Versavel, M.D., Ph.D. • Exits: Alder, Convergence, Juno, EVP R&D and Chief Scientific Officer Chief Development Officer Interim Chief Medical Officer Immunomedics, Cascadian, Cavion 4 4

Addressing multiple interrelated diseases with two distinct, clinically validated mechanisms of action Approaching interrelated disease states with Depression distinct MOAs (GABA PAM and Kv7.2/3) A / Anxiety Innovating within clinically validated mechanisms of action ETX-155 Kv7 Chronic Pain Epilepsy Multiple “pipeline-in-a-product” opportunities 5
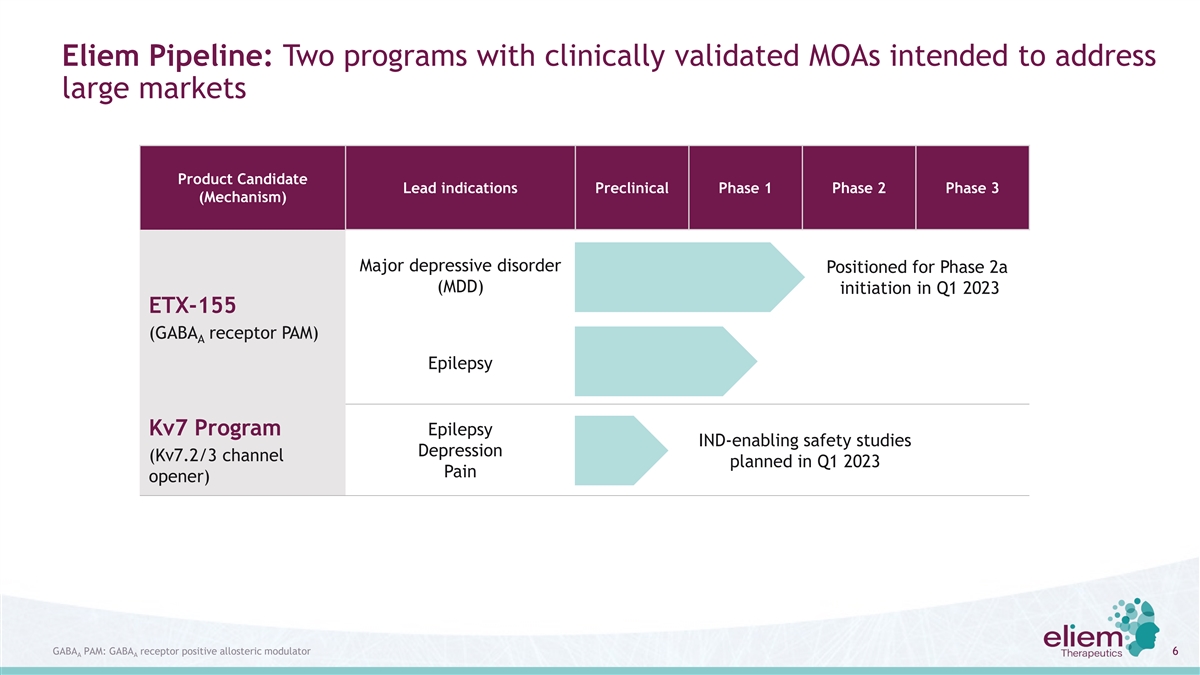
Eliem Pipeline: Two programs with clinically validated MOAs intended to address large markets Product Candidate Lead indications Preclinical Phase 1 Phase 2 Phase 3 (Mechanism) Major depressive disorder Positioned for Phase 2a (MDD) initiation in Q1 2023 ETX-155 (GABA receptor PAM) A Epilepsy Kv7 Program Epilepsy IND-enabling safety studies Depression (Kv7.2/3 channel planned in Q1 2023 Pain opener) GABA PAM: GABA receptor positive allosteric modulator 6 A A

Proof of concept Phase 2a trial in Major Depressive Disorder (MDD) ETX-155 positioned for Q1 2023 initiation 7

ETX-155: A differentiated neuroactive steroid GABA positive allosteric modulator A Clinical validation for MOA (GABA PAM) A Dual potent activity at synaptic and extrasynaptic GABA receptors, with high intrinsic efficacy A No clinically meaningful food effect Convenient once-daily dosing with ~40-hr half-life Well tolerated at exposure levels that have translated to clinical efficacy for other GABA PAMs A Strong IP position with patent protection to 2039 8

Clinical development focused on MDD with opportunity to expand into other large markets with considerable unmet need Proof of concept planned Potential future indication opportunities Major Depressive Perimenopausal Epilepsy / Focal Disorder (MDD) Depression (PMD) Onset Seizure (FOS) • Reduced GABA levels à • Reduced neurosteroid levels à • GABAergic deficits à 1 increased MDD severity PMD symptoms epileptic state MoA Rationale • Clinically validated • Clinically validated in neurosteroid- • Clinically validated in orphan (zuranolone) driven PPD (zuranolone) epilepsies (ganaxolone) • Faster onset of action • Same as MDD • Novel MoAsà better seizure control Unmet Needs • Improved tolerability/efficacy • Novel MoAs directly addressing reduced neurosteroid levels • Positive impact on mood as #1 4 • Novel MoAs comorbidity is depression Estimated annual ~32m ~8m ~2m 2 3 5 prevalence (US+EU) (~9m failed ≥1 prior therapy) (~2m with no history of MDD) (~0.8m with uncontrolled seizures) 1. Luscher et al, Mol Psychiatry, 2011;16(4):383-406 2. Decision Resources Group (DRG)– Unipolar Depression Disease Landscape and Forecast 3. Freeman et al, JAMA Psychiatry, 2014;72(1):36-43 4. Kanner AM, Biol Psychiatry, 2003;54(3):388-98 9 5. DRG – Epilepsy Disease Landscape and Forecast, May 2021

ETX-155 Differentiation: Similar dual GABA R potency to clinically validated A GABA PAMs, with differentiated pharmacokinetics A GABA R Clinical Validation A Pharmacokinetics (positive RCT) Potency Company Molecule Extra- Synaptic Food effect Half-life MDD PPD Epilepsy synaptic No ~40 hrs - - - ETX-155 zuranolone Yes 14-18 hrs - (SAGE-217) ZTALMY® Yes 2-3 hrs - - (ganaxolone) Sources: Zuranolone: Hoffmann et al, Clin Pharmacokinet, 2020;59(1):111-120; Hoffmann et al, ASCP 2018, poster #782; Botella et al, J Med * Evening QD dosing for ETX-155, zuranolone; Chem, 2017;60(18)7810-7819; Phase 3 WATERFALL topline data press release 10 Ganaxolone: ZTALMY Prescribing Information; Hulihan et al., American Epilepsy Society Annual Meeting 2020, poster TID dosing for ganaxolone
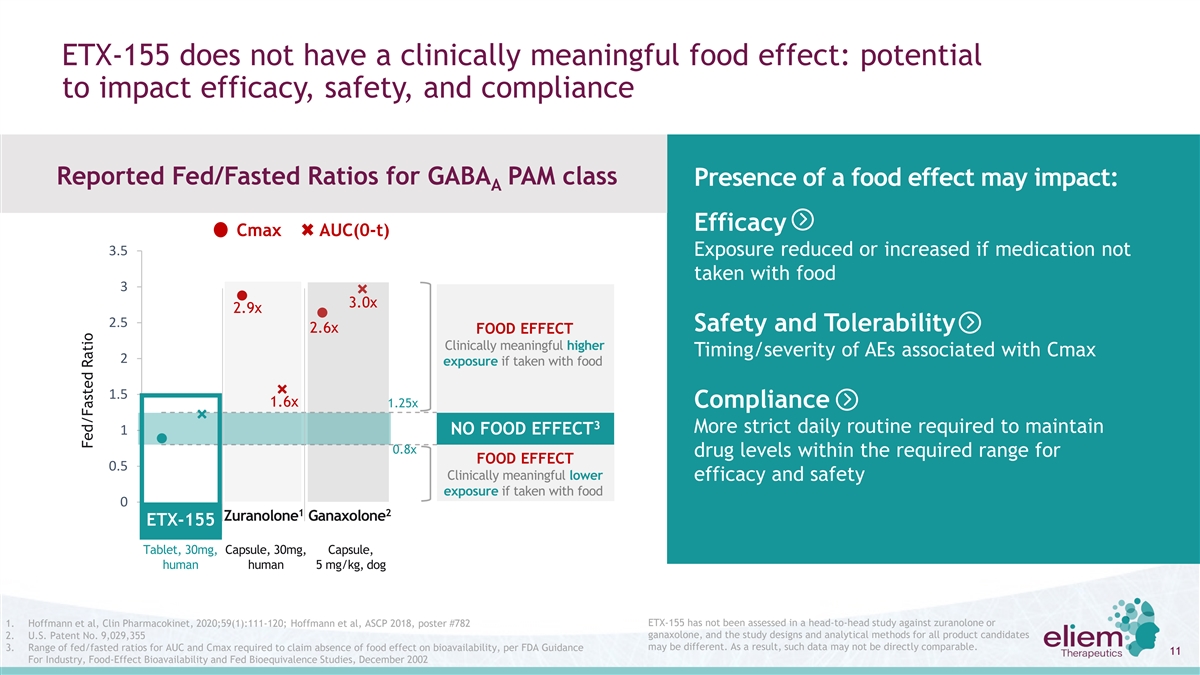
ETX-155 does not have a clinically meaningful food effect: potential to impact efficacy, safety, and compliance Reported Fed/Fasted Ratios for GABA PAM class Presence of a food effect may impact: A Efficacy Cmax AUC(0-t) 3.5 Exposure reduced or increased if medication not taken with food 3 3.0x 2.9x 2.5 Safety and Tolerability 2.6x FOOD EFFECT Clinically meaningful higher Timing/severity of AEs associated with Cmax 2 exposure if taken with food 1.5 1.6x 1.25x Compliance 3 More strict daily routine required to maintain 1 NO FOOD EFFECT 0.8x drug levels within the required range for FOOD EFFECT 0.5 Clinically meaningful lower efficacy and safety exposure if taken with food 0 1 2 Zuranolone Ganaxolone ETX-155 Tablet, 30mg, Capsule, 30mg, Capsule, human human 5 mg/kg, dog ETX-155 has not been assessed in a head-to-head study against zuranolone or 1. Hoffmann et al, Clin Pharmacokinet, 2020;59(1):111-120; Hoffmann et al, ASCP 2018, poster #782 2. U.S. Patent No. 9,029,355 ganaxolone, and the study designs and analytical methods for all product candidates may be different. As a result, such data may not be directly comparable. 3. Range of fed/fasted ratios for AUC and Cmax required to claim absence of food effect on bioavailability, per FDA Guidance 11 For Industry, Food-Effect Bioavailability and Fed Bioequivalence Studies, December 2002 Fed/Fasted Ratio

Phase 1 Study in Healthy Subjects: Encouraging safety & tolerability profile observed with no severe or serious adverse events Most common treatment-emergent AEs ETX-155 Phase 1 Repeat-Dose Results (In ≥10% of ETX-155 treated subjects across repeat dose studies) • Favorable pharmacokinetics 7-day Repeat 14-day Repeat • Steady state reached at day 8 Combined Dose Dose • ~40-hour half-life at steady state • 60 mg evening dosing was well tolerated ETX-155 ETX-155 ETX-155 Placebo Placebo Placebo 60 mg 60 mg 60 mg • No SAEs or discontinuations (n=6) (n=5) (n=11) (n=9) (n=15) (n=24) • All AEs were mild/moderate and transient n (%) n (%) n (%) n (%) n (%) n (%) • CNS AE details • The rate of CNS AEs were comparable in ETX-155 and ≥1 TEAE 5 (56) 3 (50) 9 (60) 4 (80) 14 (58) 7 (64) placebo groups • Most CNS AEs occurred at Tmax (3-4 hrs post-dose) Somnolence 1 (11) 2 (33) 6 (40) 2 (40) 7 (29) 4 (36) • 7 reports of somnolence out of 24 ETX-155-treated patients (no subject reported somnolence more than Fatigue 0 0 4 (27) 1 (20) 4 (17) 1 (9) once during dosing period) Headache 2 (22) 2 (33) 1 (7) 0 3 (13) 2 (18) • Leeds Sleep Evaluation Questionnaire indicates no difference in next-morning alertness or disruption in Dizziness 1 (11) 0 2 (13) 0 3 (13) 0 sleep quality compared to placebo 12

Analysis of ETX-155 pharmacokinetic profile confirmed comparable 60mg exposures obtained with different batches, enabling path forward in Phase 2a MDD trial Original Phase 1 studies PSE study findings (April ‘22) Extensive Investigation • SAD (5-200mg, n=6 active/cohort) • Proof-of-concept model in patients 1) CMC investigations on all batches of with photosensitive epilepsy (PSE) API and DP • MAD 7-day (60mg AM and PM, n=9 à No findings active/cohort) • Single-dose of 135 mg ETX-155 • MAD 14-day (60mg PM, n=15 • N=3 patients; evaluation of activity 2) Dog PK to compare preclinical active/cohort) in this model was inconclusive due exposure across all batches to ~50% lower-than-expected à Comparable across batches • Well tolerated, especially with PM exposures dosing 3) 5 single dose cohorts of healthy • Achievement of predicted • Encouraging PK profile subjects to confirm PK profile therapeutic exposure levels is Ø Moderate CV% of ~30% à Comparable exposure across batches Ø Half-life of ~40 hrs critical for success of planned Phase 4) Comparison of exposures from all Ø Steady state at day 8 2a MDD trial Ø Moderate acc. ratio of ~2 batches with population PK model • Prompted investigation to Ø No food effect based on original Phase 1 data understand potential root causes Ø Exposures within preclinical efficacy à Exposures at 60mg fit the model range prior to progressing to MDD trial 5) Confirmed 75mg well tolerated as a • Enabled selection of a Phase 2 single-dose clinical dose of 60mg à Plan to evaluate 75mg in repeat-dose Investigation concluded there was no meaningful difference between 60mg exposures obtained with different batches 13

At steady-state, modeled exposures of ETX-155 60mg and 75mg doses are well within the therapeutic range Modeled 60mg and 75mg ETX-155 AUC at steady state 0-24 Preclinical Robust ED range (3 mg/kg) Mean: 2810 Benchmark zuranolone modeled AUC for 0-24 30mg and 50mg capsules at steady state* Mean: 2248 Mean: 1540 Mean: 924 Preclinical MED range (1 mg/kg) Center lines represent the median, the top and bottom of the boxes represent the 25th and 75th percentiles (i.e. interquartile range) * Derived from 30mg pharmacokinetic data in Bullock et al, Marcé of North America Congress, Oct 21-24, 2021, extrapolated to 50 mg based on dose linearity; modeled CV% derived from Hoffmann et al, 2018 Annual Meeting of the American Society for Clinical Pharmacology and Therapeutics, March 21-24, 2018 The two graphs are not generated from head-to-head trials and therefore are not direct comparisons. ETX-155 preclinical AUC ranges are derived from rat PK study for 1 mg/kg and 3 14 mg/kg groups, which were efficacious dose levels in depression, anxiety, seizure, and EEG preclinical models; ED: Efficacious Dose; MED: Minimal Efficacious Dose

Positioned to progress ETX-155 into Phase 2a RCT in MDD in Q1 2023 with topline data expected in mid-2024 Screening Outpatient Treatment Follow-up Primary Endpoint Week 28 days 14 days Change from baseline in HAMD17 1 2 3 4 over days 4, 8, 15, 22, and 29 Secondary Endpoints ETX-155 qd x 28 days MDD Phase 2a Change from baseline in HAMD17 130 patients at days 4, 8, 15, 22, 29 and 42* 1:1 randomization Placebo 50% response rate in HAMD17 qd x 28 days at days 4, 8, 15, 22, 29 and 42* d1 d8 d15 d22 d29 d42 * Day 42 assessments are exploratory 15 HAMD17: Hamilton Depression Rating Scale

ETX-155 Market Opportunity 16
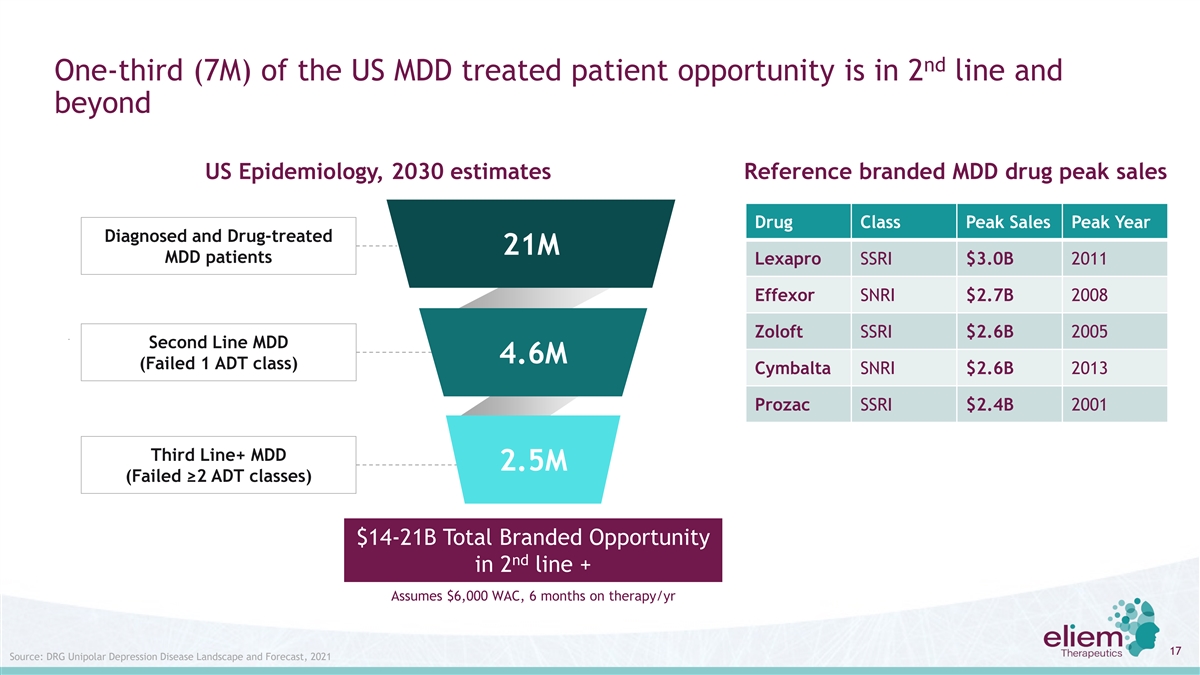
nd One-third (7M) of the US MDD treated patient opportunity is in 2 line and beyond US Epidemiology, 2030 estimates Reference branded MDD drug peak sales Drug Class Peak Sales Peak Year Diagnosed and Drug-treated 21M MDD patients Lexapro SSRI $3.0B 2011 Effexor SNRI $2.7B 2008 Zoloft SSRI $2.6B 2005 Second Line MDD 4.6M (Failed 1 ADT class) Cymbalta SNRI $2.6B 2013 Prozac SSRI $2.4B 2001 Third Line+ MDD 2.5M (Failed ≥2 ADT classes) $14-21B Total Branded Opportunity nd in 2 line + Assumes $6,000 WAC, 6 months on therapy/yr 17 Source: DRG Unipolar Depression Disease Landscape and Forecast, 2021

Zuranolone precedent GABA PAM efficacy in line with approved MDD drugs, A achieving statistical significance and ~50% reduction in HAMD17 despite smaller delta to placebo Estimated Mean HAMD17 at Mean HAMD17 change from Baseline end of treatment Year of Baseline Duration of Drug: Study* Reference Study HAMD17 Treatment Delta to Active Placebo Active Placebo (top dose, if >1 arm) placebo 1980s- -7.6 -10.4 38 studies of SSRIs/SNRIs Kirsch 2002 21.0 – 29.7 4-8 weeks -2.8 ~14 ~17 1990s (range: -5.9 to -14.2) (range: -3.0 to -10.5) Cymbalta: Study 1 Cymbalta label 2001 21 9 weeks -10.9 -6.1 -4.9 10.1 14.9 Cymbalta: Study 2 Cymbalta label 2001 20 9 weeks -10.5 -8.3 -2.2 9.5 11.7 Cymbalta: Study 3 Cymbalta label 2001 18 8 weeks -8.6 -5.0 -3.6 9.4 13 Cymbalta: Study 4 Cymbalta label 2001 20 8 weeks -12.1 -8.8 -3.3 7.9 11.2 Pristiq: Study 332 Liebowitz et al 2008 23 8 weeks -11.5 -9.5 -2.0 11.5 13.5 Pristiq: Study 333 Boyer et al 2008 24 8 weeks -13.7 -10.7 -3.0 10.3 13.3 Rexulti: Pyxis Study Thase et al (a) 2013 21 6 weeks -5.89 -3.59 -2.29 15.1 17.4 Rexulti: Polaris Study Thase et al (b) 2013 21 6 weeks -6.26 -4.57 -1.69 14.7 16.4 Rexulti: Sirius Study Hobart et al 2016 21 6 weeks -7.1 -5.9 -1.16 13.9 15.1 Average of all drugs -9.7 -7.0 -2.7 11.6 14.4 6-8 wks (-5.9 to -13.7) (-3.6 to -10.7) (-1.16 to -4.9) (7.9 to 15.1) (11.7 to 17.4) (range) Waterfall 2020-21 26.8 2 weeks -14.1 -12.3 -1.7 12.7 14.6 Sage zuranolone data Coral 2021 26.8 2 weeks -13.7 -12.9 -0.8 13.1 13.7 ~50% reduction in mean HAMD17 from severe depression (>24) to mild depression (8-16) 18 * Studies reporting HAMD as primary endpoint shown here; recent MDD registrational trials for Trintellix, Spravato, and Axsome used MADRS as endpoint, rather than HAMD17, so are not included

Zuranolone precedent suggests potential GABA PAM advantages relative to existing A ADTs: would be attractive in a “direct to patient” commercial marketplace Background Potential Differentiation Points • SSRI’s treatment duration undefined – many • “Treat depressive episode”: zuranolone uses a patients on SSRIs for multiple years/life two-week regimen, 80% of patients needed only 4 weeks of therapy in a year • SSRI’s can take 6 to 8 weeks to work, if they work; not accounting for titration period • Rapid onset: zuranolone achieved activity by day 3, with no titration, which should enable patient • Side effects including weight gain, sexual to know within two weeks if product is working dysfunction, withdrawal symptoms • Transient side effects: somnolence/fatigue but • Unsatisfied market with new MDD patient on no weight gain, sexual dysfunction, or withdrawal therapy for ~ 5.5 months, adherence rates observed in zuranolone trials 1 of 51% at week 16, 21% at week 33 • Enhanced adherence: two-week course of therapy should dramatically improve adherence Short treatment duration combined with rapid effect enables dosing aligned with the depressive episode 19 1. Sawada et al, BMC Psychiatry, 2009;9(38).

ETX-155: Being Developed as a Potentially Clinically Differentiated Oral Neuroactive Steroid ETX-155 Opportunities Improve Efficacy: Increasing AUC • Absence of clinically meaningful food effect • Significantly longer half-life • Higher exposures observed at well tolerated doses Improve Tolerability • CNS AE rates similar to placebo in healthy subjects • No difference from placebo in sleep quality and next day alertness • No reports of somnolence on more than one day in repeat dosing studies Improve Durability • Evaluate longer dosing regimens (i.e., 28 days) 20 Statements are not based on data resulting from head-to-head trials and are not direct comparisons of safety or efficacy

Pre-candidates identified IND-enabling safety studies planned for Kv7 Opener Q1 2023 21

Kv7.2/3 Program: Developing a differentiated Kv7.2/3 opener for multiple neuronal excitability indications Kv7 Opportunity Eliem Kv7 Program Goal Maintain efficacy with improved tolerability Human genetic validation and safety Strong clinical validation in pain and Program Status epilepsy (retigabine, flupirtine, XEN1101) Foundational IP filed Metabolic/safety liabilities with existing Multiple lead and backup candidates in molecules novel chemical space Improved metabolic stability Clear translational path to clinical efficacy Potent at Kv7.2/3, selective vs Kv7.1/4, and active in MES rat model Two pre-candidates being scaled up for IND-enabling safety studies anticipated to initiate in Q1 2023 22 MES: maximal electroshock seizure

Highly experienced management team Clinical and preclinical pipeline based on clinically validated mechanisms of action Rethinking Two differentiated programs in depression and epilepsy with treatment for expansion opportunities in chronic pain nervous system disorders ~$130M* cash runway into 2025 allows for topline clinical data readouts and advancement of preclinical asset into clinic * Unaudited cash, cash equivalents and investments as of September 30, 2022, 23 including receipt of $6.2m in tax reimbursements within the quarter

For more information: www.eliemtx.com InvestorRelations@eliemtx.com