Exhibit 99.1
Overview
We were founded on the belief that engineered cells will be one of the most important transformations in medicine over the next several decades. The burden of diseases that can be addressed at their root cause through engineered cells is significant. We view engineered cells as having the potential to be as therapeutically disruptive as biologics to clinical practice. Our long-term aspirations are to be able to control or modify any gene in the body, to replace any cell that is damaged or missing, and to markedly improve access to cellular and gene- based medicines. We have brought together an experienced group of scientists, engineers, and company builders and combined them with the necessary technologies to move this vision forward. We are developing ex vivo and in vivo cell engineered cell therapies to revolutionize treatment across a broad array of therapeutic areas with unmet treatment needs, including in oncology, diabetes, autoimmune diseases, and central nervous system disorders, among others. Our platform progress, broad capabilities, and strong balance sheet enable us to execute on a broad vision.
Frequently in disease, cells are damaged or missing entirely, and an effective therapy needs to replace the entire cell, an approach referred to as cell therapy or ex vivo cell engineering. A successful therapeutic requires an ability to manufacture cells at scale that engraft, function, and have the necessary persistence in the body. Of these requirements, long-term persistence related to overcoming immunologic rejection of another person’s cells has been the most challenging, which has led many to focus on autologous, or a patient’s own, cells as the therapeutic source. However, autologous therapies require a complex process of harvesting cells from the patients, manipulating them outside the body, and returning them to the patient. Products using this approach have had to manage significant challenges such as scalability, product variability, product quality, cost, patient accessibility, and limits on number of cell types that are amenable to this approach. Given these limitations, rather than using autologous cells to overcome immune rejection, we have invested in creating hypoimmune- modified cells that can “hide” from the patient’s immune system. We are striving to make therapies that use pluripotent stem cells with our hypoimmune genetic modifications as the starting material, which we then differentiate into a specific cell type, such as a pancreatic islet cell, before treating the patient. Additionally, there are cell types for which effective differentiation protocols from a stem cell have not yet been developed, such as T cells. For these cell types, instead of starting from a pluripotent stem cell, we can use allogeneic, fully- differentiated cells sourced from a donor as the starting material to which we then apply our hypoimmune genetic modifications.
The process of repairing and controlling genes in the body, referred to as gene therapy or in vivo cell engineering, requires in vivo delivery of a therapeutic payload and modification of the genome. There are multiple methods available to modify the genome, but limited ability to deliver therapeutic payloads in vivo. Thus, delivery of a therapeutic payload is at the core of our strategic focus for our in vivo gene therapy program, with our ultimate goal being the delivery of any payload to any cell in a specific and repeatable way. Our initial effort is on cell-specific delivery and increasing the diversity and size of payloads. Using our fusogen technology, we have shown in preclinical studies that we can specifically target numerous cell surface receptors that, when combined with delivery vehicles to form fusosomes, allow cell-specific delivery across multiple different cell types.
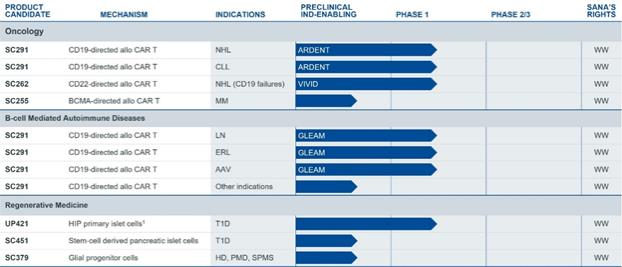
We believe the time is right to develop engineered cell therapies across a broad range of therapeutic areas. Substantial progress in the understanding of genetics, gene editing, protein engineering, stem cell biology, immunology, process analytics, and computational biology have converged to create an opportunity to markedly increase the breadth and depth of the potential impact of cellular medicines. We are focused on creating transformative engineered cell therapies across a range of therapeutic areas. We are developing a broad pipeline of product candidates, which are summarized below: