
4th Quarter & Full Year 2021 Financial Results & Corporate Update March 30, 2022 Exhibit 99.2

Titelmasterformat durch Klicken bearbeitenThis Slide Presentation Includes Forward-looking Statements 2 This presentation contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, as amended, including, but not limited to, statements concerning: BioNTech's expected revenues and net profit related to sales of BioNTech's COVID-19 vaccine, referred to as COMIRNATY® where approved for use under full or conditional marketing authorization, in territories controlled by BioNTech's collaboration partners, particularly for those figures that are derived from preliminary estimates provided by BioNTech's partners; BioNTech's pricing and coverage negotiations with governmental authorities, private health insurers and other third-party payors after BioNTech's initial sales to national governments; the extent to which initial or booster doses of a COVID-19 vaccine continue to be necessary in the future; competition from other COVID-19 vaccines or related to BioNTech's other product candidates, including those with different mechanisms of action and different manufacturing and distribution constraints, on the basis of, among other things, efficacy, cost, convenience of storage and distribution, breadth of approved use, side-effect profile and durability of immune response; the rate and degree of market acceptance of BioNTech's COVID-19 vaccine and, if approved, BioNTech's investigational medicines; the initiation, timing, progress, results, and cost of BioNTech's research and development programs and BioNTech's current and future preclinical studies and clinical trials, including statements regarding the timing of initiation and completion of studies or trials and related preparatory work, the period during which the results of the trials will become available and BioNTech's research and development programs; the timing of and BioNTech's ability to obtain and maintain regulatory approval for BioNTech's product candidates; the collaboration between BioNTech and Pfizer to develop a COVID-19 vaccine (including a potential booster dose of BNT162b2 and/or a potential booster dose of a variation of BNT162b2 having a modified mRNA sequence); the ability of BNT162b2 to prevent COVID-19 caused by emerging virus variants; BioNTech's ability to identify research opportunities and discover and develop investigational medicines; the ability and willingness of BioNTech's third-party collaborators to continue research and development activities relating to BioNTech's development candidates and investigational medicines; the impact of the COVID-19 pandemic on BioNTech's development programs, supply chain, collaborators and financial performance; unforeseen safety issues and claims for personal injury or death arising from the use of BioNTech's COVID-19 vaccine and other products and product candidates developed or manufactured by us; BioNTech's ability to progress BioNTech's Malaria, Tuberculosis and HIV programs, including timing for selecting clinical candidates for these programs and the commencement of a clinical trial, as well as any data readouts; the nature of the collaboration with the African Union and the Africa CDC; the nature and duration of support from WHO, the European Commission and other organizations with establishing infrastructure; the development of sustainable vaccine production and supply solutions on the African continent and the nature and feasibility of these solutions; BioNTech's estimates of research and development revenues, commercial revenues, cost of sales, research and development expenses, sales and marketing expenses, general and administrative expenses, capital expenditures, income taxes, shares outstanding; BioNTech's ability and that of BioNTech's collaborators to commercialize and market BioNTech's product candidates, if approved, including BioNTech's COVID-19 vaccine; BioNTech's ability to manage BioNTech's development and expansion; regulatory developments in the United States and foreign countries; BioNTech's ability to effectively scale BioNTech's production capabilities and manufacture BioNTech's products, including BioNTech's target COVID-19 vaccine production levels, and BioNTech's product candidates; and other factors not known to BioNTech at this time. In some cases, forward-looking statements can be identified by terminology such as “will,” “may,” “should,” “expects,” “intends,” “plans,” “aims,” “anticipates,” “believes,” “estimates,” “predicts,” “potential,” “continue,” or the negative of these terms or other comparable terminology, although not all forward-looking statements contain these words. The forward-looking statements in this presentation are neither promises nor guarantees, and you should not place undue reliance on these forward-looking statements because they involve known and unknown risks, uncertainties, and other factors, many of which are beyond BioNTech’s control and which could cause actual results to differ materially from those expressed or implied by these forward- looking statements. You should review the risks and uncertainties described under the heading “Risk Factors” in BioNTech's annual report on Form 20-F for the quarter and year ended December 31, 2021 and in subsequent filings made by BioNTech with the SEC, which are available on the SEC’s website at https://www.sec.gov/. Except as required by law, BioNTech disclaims any intention or responsibility for updating or revising any forward-looking statements contained in this presentation in the event of new information, future developments or otherwise. These forward-looking statements are based on BioNTech’s current expectations and speak only as of the date hereof.

Safety Information 3 COMIRNATY® ▼(the Pfizer-BioNTech COVID-19 vaccine) has been granted conditional marketing authorization (CMA) by the European Commission to prevent coronavirus disease 2019 (COVID-19) in people from 5 years of age. The vaccine is administered as a 2-dose series, 3 weeks apart. In addition, the CMA has been expanded to include a booster dose (third dose) at least 6 months after the second dose in individuals 18 years of age and older. For immunocompromised individuals, the third dose may be given at least 28 days after the second dose. The European Medicines Agency’s (EMA’s) human medicines committee (CHMP) has completed its rigorous evaluation of COMIRNATY®, concluding by consensus that sufficiently robust data on the quality, safety and efficacy of the vaccine are now available. IMPORTANT SAFETY INFORMATION: • Events of anaphylaxis have been reported. Appropriate medical treatment and supervision should always be readily available in case of an anaphylactic reaction following the administration of the vaccine. • Very rare cases of myocarditis and pericarditis have been observed following vaccination with Comirnaty. These cases have primarily occurred within 14 days following vaccination, more often after the second vaccination, and more often in younger men. Available data suggest that the course of myocarditis and pericarditis following vaccination is not different from myocarditis or pericarditis in general. • Anxiety-related reactions, including vasovagal reactions (syncope), hyperventilation or stress‐related reactions (e.g. dizziness, palpitations, increases in heart rate, alterations in blood pressure, tingling sensations and sweating) may occur in association with the vaccination process itself. Stress-related reactions are temporary and resolve on their own. Individuals should be advised to bring symptoms to the attention of the vaccination provider for evaluation. It is important that precautions are in place to avoid injury from fainting. • The efficacy, safety and immunogenicity of the vaccine has not been assessed in immunocompromised individuals, including those receiving immunosuppressant therapy. The efficacy of COMIRNATY® may be lower in immunosuppressed individuals. • As with any vaccine, vaccination with COMIRNATY® may not protect all vaccine recipients. Individuals may not be fully protected until 7 days after their second dose of vaccine. • In clinical studies, adverse reactions in participants 16 years of age and older were injection site pain (> 80%), fatigue (> 60%), headache (> 50%), myalgia and chills (> 30%), arthralgia (> 20%), pyrexia and injection site swelling (> 10%) and were usually mild or moderate in intensity and resolved within a few days after vaccination. A slightly lower frequency of reactogenicity events was associated with greater age. • The overall safety profile of COMIRNATY® in participants 5 to 15 years of age was similar to that seen in participants 16 years of age and older. • The most frequent adverse reactions in children 5 to 11 years of age were injection site pain (>80%), fatigue (>50%), headache (>30%), injection site redness and swelling (>20%), myalgia and chills (>10%). • The most frequent adverse reactions in clinical trial participants 12 to 15 years of age were injection site pain (> 90%), fatigue and headache (> 70%), myalgia and chills (> 40%), arthralgia and pyrexia (> 20%). • There is limited experience with use of COMIRNATY® in pregnant women. Administration of COMIRNATY® in pregnancy should only be considered when the potential benefits outweigh any potential risks for the mother and fetus. • It is unknown whether COMIRNATY® is excreted in human milk. • Interactions with other medicinal products or concomitant administration of COMIRNATY® with other vaccines has not been studied. • For complete information on the safety of COMIRNATY® always make reference to the approved Summary of Product Characteristics and Package Leaflet available in all the languages of the European Union on the EMA website. The black equilateral triangle ▼ denotes that additional monitoring is required to capture any adverse reactions. This will allow quick identification of new safety information. Individuals can help by reporting any side effects they may get. Side effects can be reported to EudraVigilance or directly to BioNTech using email medinfo@biontech.de, telephone +49 6131 9084 0, or via the website www.biontech.de

Safety Information 4 AUTHORIZED USE IN THE U.S. COMIRNATY® (COVID-19 Vaccine, mRNA) is an FDA-approved COVID-19 vaccine for active immunization to prevent coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in individuals 16 years of age and older. It is also authorized under EUA to provide a 2-dose primary series to individuals 5 years of age and older, a third primary series dose to individuals 5 years of age and older who have been determined to have certain kinds of immunocompromise, a single booster dose to individuals 12 years of age and older who have completed a primary series with Pfizer-BioNTech COVID-19 Vaccine or COMIRNATY®, a single booster dose to individuals 18 years of age and older who have completed primary vaccination with a different authorized COVID-19 vaccine, a second booster dose to individuals 50 years of age and older who have received a first booster dose of any authorized COVID-19 vaccine; and a second booster dose to individuals 12 years of age and older who have been determined to have certain kinds of immunocompromise and who have received a first booster dose of any authorized COVID-19 vaccine. The booster schedule is based on the labeling information of the vaccine used for the primary series. IMPORTANT SAFETY INFORMATION Individuals should not get the vaccine if they: • had a severe allergic reaction after a previous dose of this vaccine • had a severe allergic reaction to any ingredient of this vaccine Individuals should tell the vaccination provider about all of their medical conditions, including if they: • have any allergies • have had myocarditis (inflammation of the heart muscle) or pericarditis (inflammation of the lining outside the heart) • have a fever • have a bleeding disorder or are on a blood thinner • are immunocompromised or are on a medicine that affects the immune system • are pregnant, plan to become pregnant, or are breastfeeding • have received another COVID-19 vaccine • have ever fainted in association with an injection The vaccine may not protect everyone. Side effects reported with the vaccine include: • There is a remote chance that the vaccine could cause a severe allergic reaction o A severe allergic reaction would usually occur within a few minutes to 1 hour after getting a dose of the vaccine. For this reason, vaccination providers may ask individuals to stay at the place where they received the vaccine for monitoring after vaccination o Signs of a severe allergic reaction can include difficulty breathing, swelling of the face and throat, a fast heartbeat, a bad rash all over the body, dizziness, and weakness o If an individual experiences a severe allergic reaction, they should call 9-1-1 or go to the nearest hospital • Myocarditis (inflammation of the heart muscle) and pericarditis (inflammation of the lining outside the heart) have occurred in some people who have received the vaccine, more commonly in males under 40 years of age than among females and older males. In most of these people, symptoms began within a few days following receipt of the second dose of the vaccine. The chance of having this occur is very low. Individuals should seek medical attention right away if they have any of the following symptoms after receiving the vaccine: o chest pain o shortness of breath o feelings of having a fast-beating, fluttering, or pounding heart • Additional side effects that have been reported with the vaccine include: o severe allergic reactions; non-severe allergic reactions such as rash, itching, hives, or swelling of the face; myocarditis (inflammation of the heart muscle); pericarditis (inflammation of the lining outside the heart); injection site pain; tiredness; headache; muscle pain; chills; joint pain; fever; injection site swelling; injection site redness; nausea; feeling unwell; swollen lymph nodes (lymphadenopathy); decreased appetite; diarrhea; vomiting; arm pain; fainting in association with injection of the vaccine • These may not be all the possible side effects of the vaccine. Serious and unexpected side effects may occur. The possible side effects of the vaccine are still being studied in clinical trials. Call the vaccination provider or healthcare provider about bothersome side effects or side effects that do not go away Data on administration of this vaccine at the same time as other vaccines have not yet been submitted to FDA. Individuals considering receiving this vaccine with other vaccines should discuss their options with their healthcare provider. Patients should always ask their healthcare providers for medical advice about adverse events. Individuals are encouraged to report negative side effects of vaccines to the US Food and Drug Administration (FDA) and the Centers for Disease Control and Prevention (CDC). Visit https://www.vaers.hhs.gov or call 1‐800‐ 822‐7967. In addition, side effects can be reported to Pfizer Inc. at www.pfizersafetyreporting.com or by calling 1-800-438-1985.

Corporate Outlook Ryan Richardson, Chief Strategy Officer Financial Results Jens Holstein, CFO Oncology Pipeline Update Özlem Türeci, CMO COVID-19 Vaccine Update Özlem Türeci, CMO Fourth Quarter and Full Year 2021 Highlights Ugur Sahin, CEO 5
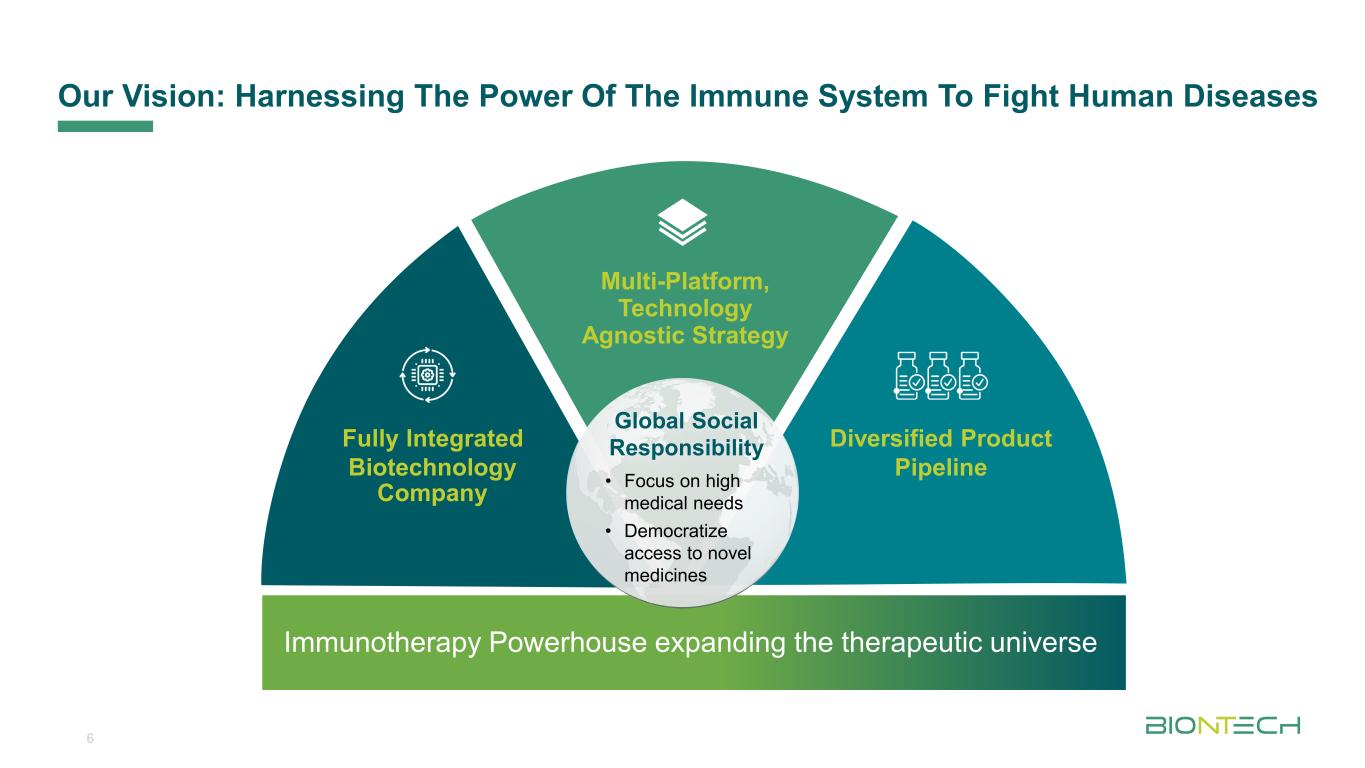
Titelmasterformat durch Klicken bearbeiten Our Vision: Harnessing The Power Of The Immune System To Fight Human Diseases 6 Multi-Platform, Technology Agnostic Strategy Diversified Product Pipeline Fully Integrated Biotechnology Company Immunotherapy Powerhouse expanding the therapeutic universe Global Social Responsibility • Focus on high medical needs • Democratize access to novel medicines

Titelmasterformat durch Klicken bearbeiten 2021 Key Highlights of Progress Towards Vision STRONG FINANCIAL PERFORMANCE €19.0 Bn Total 2021 Revenues2 €39.63 Diluted EPS2 EXPANDED GLOBAL ORGANIZATION 3,000+ team members >1 Bn to low- and middle- income countries1 Increased footprint with new offices in U.S., Europe and Asia COVID-19 VACCINE GLOBAL LEADERSHIP • 2 FixVac programs • 2 iNeST programs • 1 Bispecific Immunomodulator • RiboCytokines • RiboMabs • CAR-T cell therapy • NEOSTIM individualized neoantigen T cell therapy • Acquired Kite’s cell therapy facility in U.S. • Kite asset acquisition expanded personalized TCR platform • Expanded TCR pipeline with Medigene asset acquisition and research collaboration countries & territories1 >165~2.6 Bn doses delivered in 2021 1 As of end 2021 2 Estimated figures based on preliminary data shared between Pfizer and BioNTech as further described in our Annual Report on Form 20-F for the year ending December 31, 2021 to New platforms entered the clinic DROVE ADVANCEMENT IN ONCOLOGY Strategic M&A to complement existing technologies Multiple randomized Phase 2 trials 7
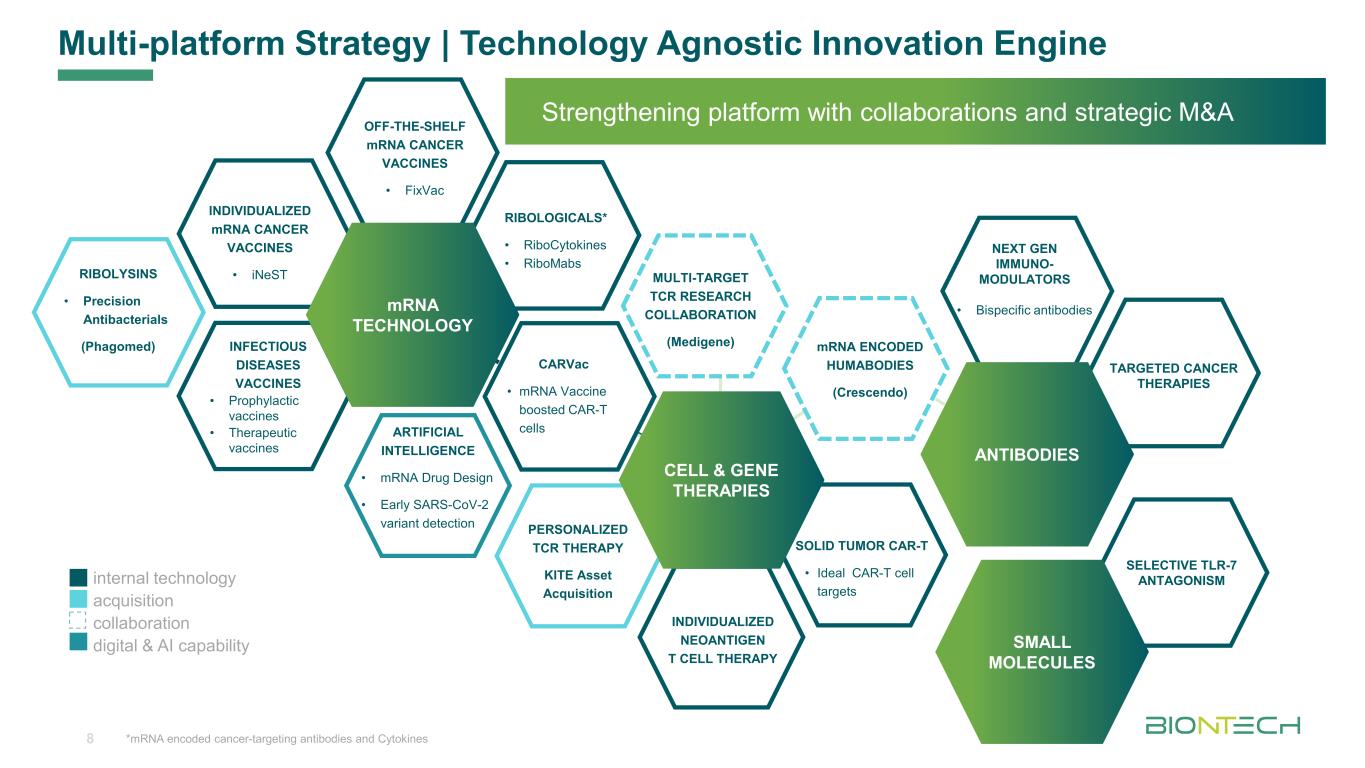
Multi-platform Strategy | Technology Agnostic Innovation Engine Strengthening platform with collaborations and strategic M&A SELECTIVE TLR-7 ANTAGONISM SMALL MOLECULES TARGETED CANCER THERAPIES ANTIBODIES NEXT GEN IMMUNO- MODULATORS • Bispecific antibodies *mRNA encoded cancer-targeting antibodies and Cytokines RIBOLYSINS • Precision Antibacterials (Phagomed) RIBOLOGICALS* • RiboCytokines • RiboMabs OFF-THE-SHELF mRNA CANCER VACCINES • FixVac INFECTIOUS DISEASES VACCINES • Prophylactic vaccines • Therapeutic vaccines INDIVIDUALIZED mRNA CANCER VACCINES • iNeST mRNA TECHNOLOGY PERSONALIZED TCR THERAPY KITE Asset Acquisition INDIVIDUALIZED NEOANTIGEN T CELL THERAPY SOLID TUMOR CAR-T • Ideal CAR-T cell targets CARVac • mRNA Vaccine boosted CAR-T cells CELL & GENE THERAPIES mRNA ENCODED HUMABODIES (Crescendo) MULTI-TARGET TCR RESEARCH COLLABORATION (Medigene) internal technology acquisition collaboration digital & AI capability ARTIFICIAL INTELLIGENCE • mRNA Drug Design • Early SARS-CoV-2 variant detection 8

Titelmasterformat durch Klicken bearbeiten 2022 Strategic Priorities 9 POC, proof of concept; FIH, first–in-human; HSV 2, herpes simplex virus type 2 • Initiate 4 FIH vaccine trials: Expand in Infectious Disease • First randomized Phase 2 readout • Prepare for registrational trials • POC data for CAR-T cell therapy Execute in Oncology Continue COVID-19 Vaccine Leadership • Label & geographic expansion • Next-generation vaccines • Innovations for pandemic preparedness Invest in Foundation to Enable Accelerated Innovation and Expansion Digital & AI Capabilities | Technologies | Development Team | Manufacturing | Global Footprint • Autoimmune disease • Regenerative medicine • Cardiovascular disease Advance into New Therapeutic Areas • Shingles • HSV 2 • Tuberculosis • Malaria • 10+ additional mRNA vaccine programs • Precision antibacterials

Titelmasterformat durch Klicken bearbeiten Global Social Responsibility at Our Core 10 SBTi, Science Based Targets initiative (in line with Paris agreement) 1 base year: 2021 2 encompasses adhering to ten principles on human rights, the environment and anti-corruption COVID-19 vaccine pledge to COVAX and the world • 2+ bn doses to low- and middle-income countries by end of 2022 Address high medical need diseases such as: • Malaria • Tuberculosis Climate targets under SBTi • Scope 1 & 2: absolute emission reduction of 42% by 20301 Practice good corporate governance and social and societal responsibility • Signed UN Global Compact2 Strengthen recruiting and development • Diversified employee base from 60+ countries Democratize Access to Novel Medicine Pandemic response and access to medicines Launched BioNTainers as modular mRNA manufacturing facilities • Establishment of first mRNA manufacturing facility in African Union is expected to start in mid-2022 • Introduce drone vaccine delivery in Ghana Environmental & Climate Protection Responsible Governance Attractive Employer

05 Corporate Outlook Ryan Richardson, Chief Strategy Officer 04 Financial Results Jens Holstein, CFO 03 Oncology Pipeline Update Özlem Türeci, CMO COVID-19 Vaccine Update Özlem Türeci, CMO 01 Fourth Quarter and Full Year 2021 Highlights Ugur Sahin, CEO 11

COVID-19 Vaccine Accomplishments Position Company for Continued Success in 2022 12 Pediatrics • 5 to <12 yrs: • Obtained approvals in multiple markets1 for 2-dose regimen for BNT162b2 • Evaluating third dose • 6 mo to <5 yrs: • Evaluating 3-dose regimen; data expected in April 2022 • Data will be submitted to FDA and other regulators • Building state-of-the-art mRNA manufacturing sites in Africa and Asia • BioNTainers designed to enable rapid setup of new mRNA vaccine manufacturing nodes • Comprehensive variant-based vaccine development program • Development of Omicron-based vaccine on track • Scaled up manufacturing and started production • Generating clinical data to support potential regulatory submission; first data expected in April 2022 • Comprehensive research program to investigate evolution of SARS-CoV-2 directed immunity under vaccinations and infections to inform further vaccine development • InstaDeep collaboration to further develop early-warning system for high-risk variants Global Manufacturing Network Preemptive Approach to Variants Boosters • Obtained approvals in multiple markets1 for BNT162b2 booster (3rd dose) in 12 yrs+ • FDA approved 4th dose in 50 yrs+ and in 12 yrs+ with certain kinds of immunocompromise • Evaluating 3rd or 4th dose of variant-based versus wild-type vaccines 1 Includes U.S., EU, Canada and other countries Continued Label Expansion 2022 Order Book as of mid-March: 2.4Bn Doses

Need for Pandemic Vaccination Continues 13 1 shot 2 shots 3 or more shots Unvaccinated • ~36% of the world population remains completely unvaccinated • Only 19% of the world population has received a booster vaccination (3+ shots) • Infection rates remain high, driven by Omicron variant • Global weekly infection rate in March 2022 is >170% higher than global weekly infection rate in Q4 2021 • 54 countries reported an R0 >1 3 • 12 countries with cases doubling (or more) every week3 Unvaccinated 36% (2.86 bn) 1 shot 6% (0.46 bn) 3+ shots 19% (1.50 bn) 2 shots 39% (3.08 bn) Global Vaccination Status1 Global Confirmed COVID-19 Cases2 1 New York Times COVID-19 Vaccination Tracker, as of March 22, 2022 2 World Health Organization COVID-19 Dashboard, as of March 25, 2022 3 The Centre for the Mathematical Modelling of Infectious Diseases at the London School of Hygiene & Tropical Medicine, as of March 22, 2022 Week of February 28 Week of March 7 Week of March 14 12.6 bn 10.5 bn 11.6 bn

Titelmasterformat durch Klicken bearbeiten BNT162b2 Boosters to Address Partial Immune Escape by Omicron 14 Potential to replace traditional treatment modalities Future pandemic preparedness: Monitoring of emerging variants Rapid data-guided vaccine adaptation 1 Collie SH, et al. N Engl J Med 2022; 386:494-496 DOI: 10.1056/NEJMc2119270; 2 UK Health Security Agency. COVID-19 Vaccine Surveillance Report - Week 8. 24 February 2022; 3 Tartof SY, et al. Available at SSRN: https://ssrn.com/abstract=4011905; 4 Hansen CH, et al. MedRXiv. doi: https://doi.org/10.1101/2021.12.20.21267966; 5 Thompson MG, et al. MMWR Morb Mortal Wkly Rep 2022;71:139–145. DOI: http://dx.doi.org/10.15585/mmwr.mm7104e3external icon; 6 Lauring AS, et al. BMJ 2022; 376 doi: https://doi.org/10.1136/bmj-2021-069761; 7 Andrews N, et al. NEJM 2022. DOI: 10.1056/NEJMoa2119451; 8 Ferdinands JM, et al. MMWR Morb Mortal Wkly Rep 2022;71:255–263. DOI: http://dx.doi.org/10.15585/mmwr.mm7107e2external icon; 9 Bar-On YM, et al MedRxiv [Preprint] https://doi.org/10.1101/2022.02.01.22270232. BNT162b2 3rd dose required to reinstall immunity and effectiveness against Omicron1 • Overall infections (~70-80%)1-4 • Symptomatic disease (~50-85%)1-5 • Hospitalizations (~75-90%)2-6 However: Vaccine effectiveness against Omicron starts waning after the first few months post booster7,8 Israel real-world data suggest a 4th dose increases immunogenicity and lowers rates of confirmed infections and severe illness in elderly population9 • In subjects >60 years of age, confirmed infection and severe disease after 4th dose1 was lower compared to individuals who did not receive 4th dose9 • At 12 days+ post 4th dose, reduced risk was demonstrated compared to only 3 doses9: • Infection by a factor of 2.0 (95% CI 2.0 to 2.1) • Severe disease by a factor of 4.3 (95% CI 2.2 to 7.5)
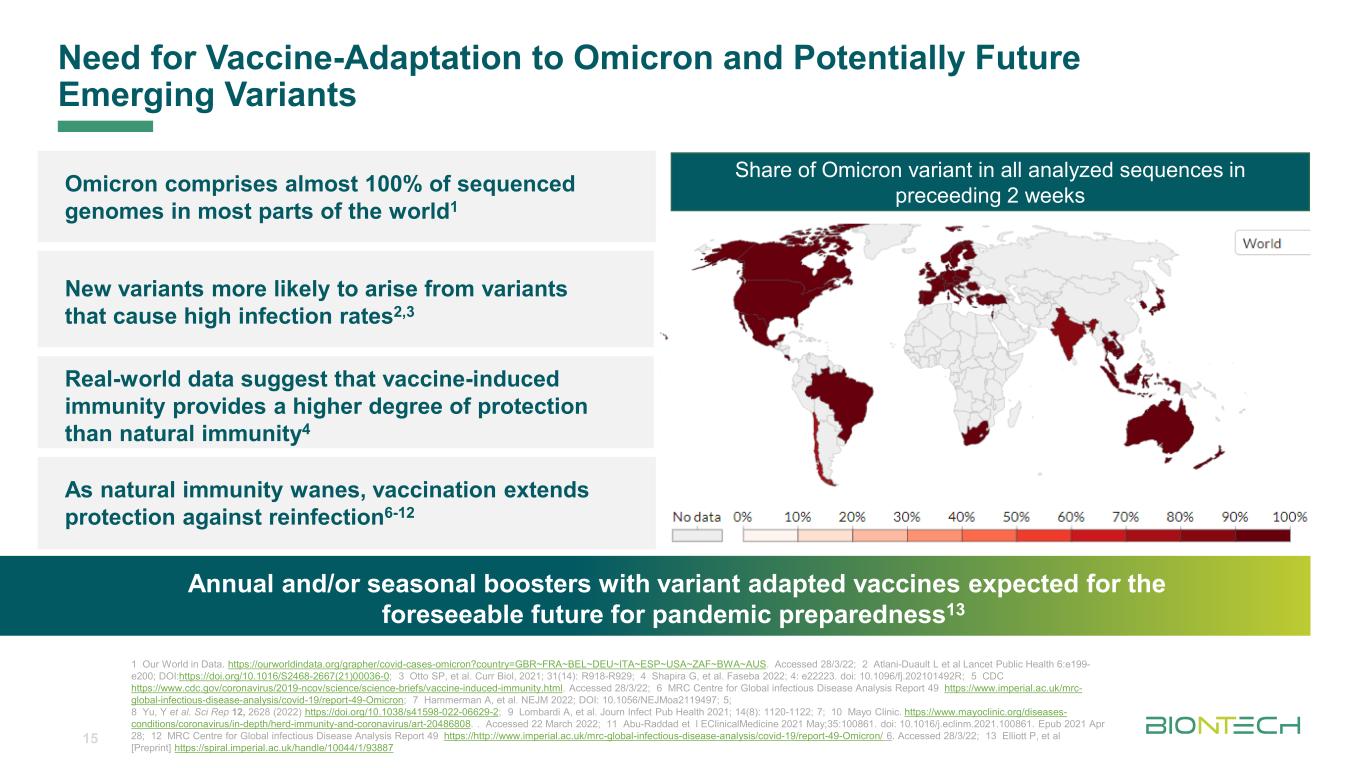
Titelmasterformat durch Klicken bearbeitenNeed for Vaccine-Adaptation to Omicron and Potentially Future Emerging Variants Potential to replace traditional treatment modalities Annual and/or seasonal boosters with variant adapted vaccines expected for the foreseeable future for pandemic preparedness13 New variants more likely to arise from variants that cause high infection rates2,3 Omicron comprises almost 100% of sequenced genomes in most parts of the world1 As natural immunity wanes, vaccination extends protection against reinfection6-12 Real-world data suggest that vaccine-induced immunity provides a higher degree of protection than natural immunity4 15 Share of Omicron variant in all analyzed sequences in preceeding 2 weeks 1 Our World in Data. https://ourworldindata.org/grapher/covid-cases-omicron?country=GBR~FRA~BEL~DEU~ITA~ESP~USA~ZAF~BWA~AUS. Accessed 28/3/22; 2 Atlani-Duault L et al Lancet Public Health 6:e199- e200; DOI:https://doi.org/10.1016/S2468-2667(21)00036-0; 3 Otto SP, et al. Curr Biol, 2021; 31(14): R918-R929; 4 Shapira G, et al. Faseba 2022; 4: e22223. doi: 10.1096/fj.202101492R; 5 CDC https://www.cdc.gov/coronavirus/2019-ncov/science/science-briefs/vaccine-induced-immunity.html. Accessed 28/3/22; 6 MRC Centre for Global infectious Disease Analysis Report 49 https://www.imperial.ac.uk/mrc- global-infectious-disease-analysis/covid-19/report-49-Omicron; 7 Hammerman A, et al. NEJM 2022; DOI: 10.1056/NEJMoa2119497; 5; 8 Yu, Y et al. Sci Rep 12, 2628 (2022) https://doi.org/10.1038/s41598-022-06629-2; 9 Lombardi A, et al. Journ Infect Pub Health 2021; 14(8): 1120-1122; 7; 10 Mayo Clinic. https://www.mayoclinic.org/diseases- conditions/coronavirus/in-depth/herd-immunity-and-coronavirus/art-20486808. . Accessed 22 March 2022; 11 Abu-Raddad et l EClinicalMedicine 2021 May;35:100861. doi: 10.1016/j.eclinm.2021.100861. Epub 2021 Apr 28; 12 MRC Centre for Global infectious Disease Analysis Report 49 https://http://www.imperial.ac.uk/mrc-global-infectious-disease-analysis/covid-19/report-49-Omicron/ 6. Accessed 28/3/22; 13 Elliott P, et al [Preprint] https://spiral.imperial.ac.uk/handle/10044/1/93887

Titelmasterformat durch Klicken bearbeiten Omicron-based vaccine or Clinical Program to Assess Safety, Tolerability and Immunogenicity of an Omicron-Adapted Vaccine 3rd dose or 3rd+4th dose 4th dose Omicron-based vaccine 3-dose primary regimen Omicron-based vaccine 1 BioNTech. Available at: https://investors.biontech.de/news-releases/news-release-details/pfizer-and-biontech-initiate-study-evaluate-omicron-based-covid. Accessed January 2022; 2 ClinicalTrials.gov. Available at: https://www.clinicaltrials.gov/ct2/show/NCT04955626. Accessed March 2022. Evaluating different Omicron monovalent vaccine regimens • N~1500, 18-55 years and >55 years • Vaccine experienced and naïve subjects • N~650, >55 years • Two dosages: 30 µg and 60 µg BNT162b2 BNT162b2 30 µg or 60 µg Vaccine naïve subjects 2 31 16 Evaluating bivalent and combination of Omicron-based vaccine and BNT162b2

05 Corporate Outlook Ryan Richardson, Chief Strategy Officer 04 Financial Results Jens Holstein, CFO Oncology Pipeline Update Özlem Türeci, CMO 02 COVID-19 Vaccine Update Özlem Türeci, CMO 01 Fourth Quarter and Full Year 2021 Highlights Ugur Sahin, CEO 17

Titelmasterformat durch Klicken bearbeiten Strong Clinical Execution in Oncology in 20211 R/R, refractory/relapsed; HNSCC, head and neck squamous cell carcinoma; NSCLC, non-small cell lung cancer; FPD, first patient dosed; 1 Includes updates through March 30, 2022; 2 BNT311 (Gen1046) and BNT312 (Gen1042) are partnered with Genmab; 3 Partnered with Regeneron; 4 Autogene Cevumeran is partnered with Genentech18 Data Updates Q4 2021 BNT211 – CARVac BNT221 – NEOSTIM BNT151 – RiboCytokines 5 First-in-human Phase 1 Trial Starts ✓ ✓ ✓ ✓ ✓ 4 Randomized Phase 2 Trial Starts BNT111 – R/R Melanoma3 BNT113 – HPV16+ HNSCC ✓ ✓ ✓ ✓ ✓ Milestone achieved in Q4 2021 and early 2022✓ Milestone achieved in full year 2021 BNT311 – R/R NSCLC – FPD Dec 2021 Autogene Cevumeran4 (BNT122) - Adjuvant colorectal cancer ✓ SITC 2021 ESMO-IO 2021 BNT211 Phase 1/2 BNT111 Phase 1 BNT112 Phase 1/2 BNT211 Phase 1/2 BNT3112 Phase 1/2 BNT3122 Phase 1/2 BNT411 Phase 1/2 ✓ ✓ ✓ ✓ ✓ ✓ BNT152+153 – RiboCytokines BNT141 – RiboMabs – FPD Jan 2022

Titelmasterformat durch Klicken bearbeiten Peripheral and Tumoral Immunologic Responses Supportive of Proposed Mechanism of Action in CPI-experienced NSCLC Patients • PD-L1 Inhibitor–Pre-treated patients • 25 NSCLC patients with evaluable baseline tumors SITC 2021 BNT311 Phase 1/2: First-in-Human Study of DuoBody-PD-L1x4-1BB Next Generation Immunomodulator designed to prime and activate anti-tumor T cell and NK cell function via • PD-L1 blockade and • Conditional 4-1BB stimulation Program partnered with Genmab; 50:50 profit/loss NSCLC, non-small cell lung cancer; PD-L1, programmed death-ligand 1; NK cell, natural killer cell; CPI, checkpoint inhibitor • Patients with tumor reduction mainly PD-L1+ tumors • Tumor reduction in 7 of 11 patients with PD-L1+ tumors Patient selection by PD-L1 status and combination with anti-PD-L1 may improve efficacy of BNT311 19

Titelmasterformat durch Klicken bearbeiten BNT311: Phase 2 Trial Targeting CPI-experienced PD-L1+ R/R NSCLC 20 Partnered with Genmab; 50:50 profit/loss collaboration R/R, refractory/relapsed; NSCLC, non-small cell lung cancer; PD-L1, programmed death-ligand 1; SOC, Standard of Care; CPI, check point inhibitor; TPS, tumor proportion score; ORR; objective response rate; PFS, progression free survival; DoR, duration of response; OS, Overall Survival *Following Safety run-in 1Bray et al., 2018; 2https://www.cancer.net/cancer-types/lung-cancer-non-small-cell/statistics; 3Cancer statistics, 2018.Siegel et al., CA Cancer J Clin. 2018 Jan; 68(1):7-30 2Qu et al., 2022; https://journals.sagepub.com/doi/10.1177/1758835921992968 BNT311 as monotherapy and in combination with Pembrolizumab after treatment with SOC immune checkpoint inhibitor • Stage IV metastatic R/R NSCLC (2L+) • PD-L1 TPS ≥1% Primary Endpoints • ORR per RECIST 1.1 Secondary Endpoints • PFS • DoR N~130 R A: BNT311 monotherapy B*: BNT311 + pembrolizumab • ~1.8 million lung cancer deaths worldwide annually1 • NSCLC is most common type (~85%)2 • 5-year survival only 4% for advanced or metastatic NSCLC3 • CPI therapy fails in majority of NSCLC patients due to evolution of resistance • Poor prognosis for CPI R/R NSCLC • Estimated PFS of < 6 months and OS of <1 year Significant unmet need in R/R NSCLC Standard of Care Benchmark • Docetaxel, ORR: 4-15%2 FPD in December 2021 C*: BNT311 + pembrolizumab (alternative treatment schedule) 1:1:1 Open-label, randomized Phase 2 trial New strategies needed to overcome resistance and maximize efficacy
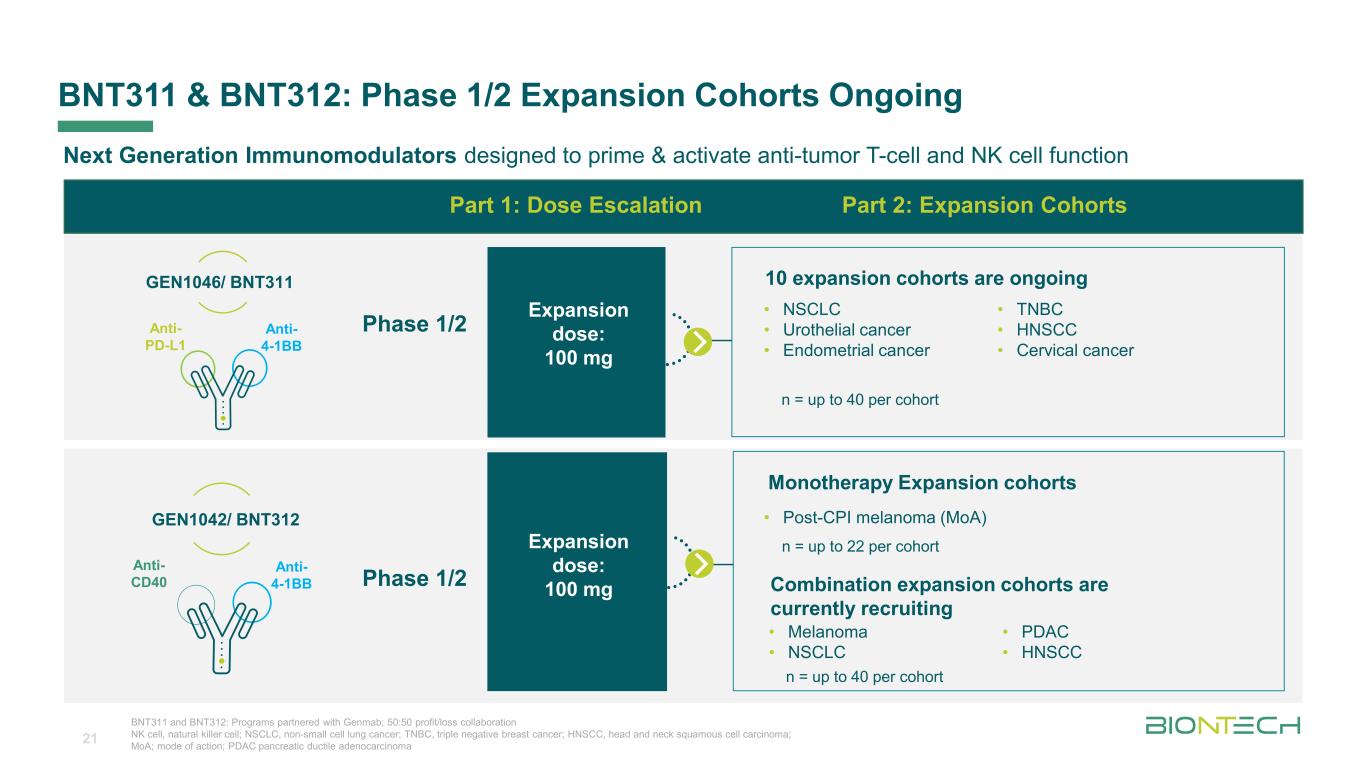
Titelmasterformat durch Klicken bearbeiten BNT311 & BNT312: Phase 1/2 Expansion Cohorts Ongoing 21 Next Generation Immunomodulators designed to prime & activate anti-tumor T-cell and NK cell function • Melanoma • NSCLC • PDAC • HNSCC Combination expansion cohorts are currently recruiting Part 1: Dose Escalation Part 2: Expansion Cohorts n = up to 40 per cohort Expansion dose: 100 mg GEN1042/ BNT312 Anti- 4-1BB Anti- CD40 Phase 1/2 BNT311 and BNT312: Programs partnered with Genmab; 50:50 profit/loss collaboration NK cell, natural killer cell; NSCLC, non-small cell lung cancer; TNBC, triple negative breast cancer; HNSCC, head and neck squamous cell carcinoma; MoA; mode of action; PDAC pancreatic ductile adenocarcinoma GEN1046/ BNT311 Anti- 4-1BB Anti- PD-L1 10 expansion cohorts are ongoing • NSCLC • Urothelial cancer • Endometrial cancer • TNBC • HNSCC • Cervical cancer n = up to 40 per cohort Expansion dose: 100 mg Phase 1/2 Monotherapy Expansion cohorts • Post-CPI melanoma (MoA) n = up to 22 per cohort
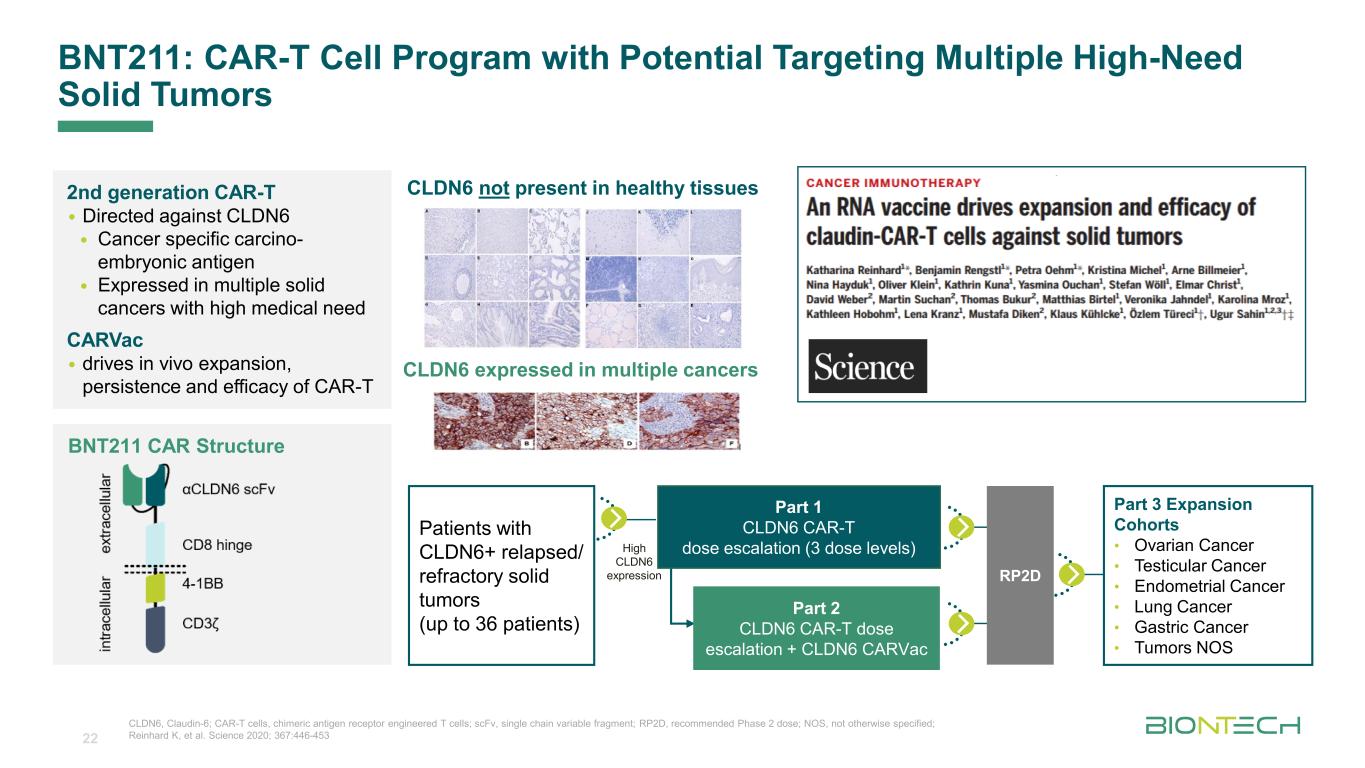
Titelmasterformat durch Klicken bearbeitenBNT211: CAR-T Cell Program with Potential Targeting Multiple High-Need Solid Tumors CLDN6, Claudin-6; CAR-T cells, chimeric antigen receptor engineered T cells; scFv, single chain variable fragment; RP2D, recommended Phase 2 dose; NOS, not otherwise specified; Reinhard K, et al. Science 2020; 367:446-453 BNT211 CAR Structure CLDN6 not present in healthy tissues CLDN6 expressed in multiple cancers 2nd generation CAR-T • Directed against CLDN6 • Cancer specific carcino- embryonic antigen • Expressed in multiple solid cancers with high medical need CARVac • drives in vivo expansion, persistence and efficacy of CAR-T Patients with CLDN6+ relapsed/ refractory solid tumors (up to 36 patients) Part 3 Expansion Cohorts • Ovarian Cancer • Testicular Cancer • Endometrial Cancer • Lung Cancer • Gastric Cancer • Tumors NOS High CLDN6 expression RP2D Part 1 CLDN6 CAR-T dose escalation (3 dose levels) Part 2 CLDN6 CAR-T dose escalation + CLDN6 CARVac 22

Titelmasterformat durch Klicken bearbeitenBNT211: CAR-T Engraftment and Tolerable Safety Profile with CLDN6 CAR-T without and with CARVac Data cutoff = NOV 18, 2021; CRS, cytokine release syndrome; DL, dose level; DLT, dose-limiting toxicity; HDCT, high dose chemotherapy; ASCT, autologous stem cell transplant; Haanen J., et al. Oral presentation at the ESMO Immuno-Oncology Congress, December 08–14, 2021; Haanen J. et al. Anals fo Concology (2021) 32 (suppl._7): S1392-S1397 2 Abstract control #8172, Clinical Trials Plenary Session; will be presented by John Haanen, discussant is Vincent K. Lam, Johns Hopkins University, Baltimore; Session Title: Clinical Trials of Cellular Immunotherapies; Session Date and Time: Sunday Apr 10, 2022 1:31 PM - 1:46 PM; Presentation Number: CT002: BNT211: A Phase I trial to evaluate safety and efficacy of CLDN6 CAR-T cells and CARVac- mediated in vivo expansion in patients with CLDN6-positive advanced solid tumors Cohort/Patient Characteristics Part 1 DL1 (n=3) Part 2 DL1 (n=3) Part 1 DL2 (n=6) Part 2 DL2 w/ LD (n=2) Part 2 DL2 w/o LD (n=1) All patients (n=15) Median (range) age, years 33 (25-68) 41 (27-56) 56 (35-66) 53.5 (46-61) 56 54 (25-68) Cancer type, n Testicular Ovarian Endometrial Fallopian tube Sarcoma Gastric 1 1 0 0 1 0 3 0 0 0 0 0 2 1 1 1 0 1 0 2 0 0 0 0 1 0 0 0 0 0 7 4 1 1 1 1 Median (range) CLDN6 II/III+ cells, % 60 (60-80) 90 (90-95) 82.5 (50-90) 95 (90-100) 85 85 (50-100) Median (range) of prior treatment lines 4 (3-5) 4 (3-4) 5 (2-11) 6 (5-7) 4 4 (2-11) • CLDN6 CAR-T cells alone or combined with CARVac well tolerated with only 1 DLT observed • CRS grade 1-2 in 1 patient at DL1 + CARVac and 6 patients at DL2, and was manageable by administration of tocilizumab • Robust engraftment of CAR-T cells resulting in a total amount of around 109 achieved in most patients and seems predictive for clinical activity • 9 of 10 patients showed initial disease control including 4 PRs; 3 in testicular cancer patients with recent relapse after HDCT/ASCT Safety Efficacy Upcoming AACR Presentation, April 10th 2 23

05 Corporate Outlook Ryan Richardson, Chief Strategy Officer Financial Results Jens Holstein, CFO 03 Oncology Pipeline Update Özlem Türeci, CMO 02 COVID-19 Vaccine Update Özlem Türeci, CMO 01 Fourth Quarter and Full Year 2021 Highlights Ugur Sahin, CEO 24
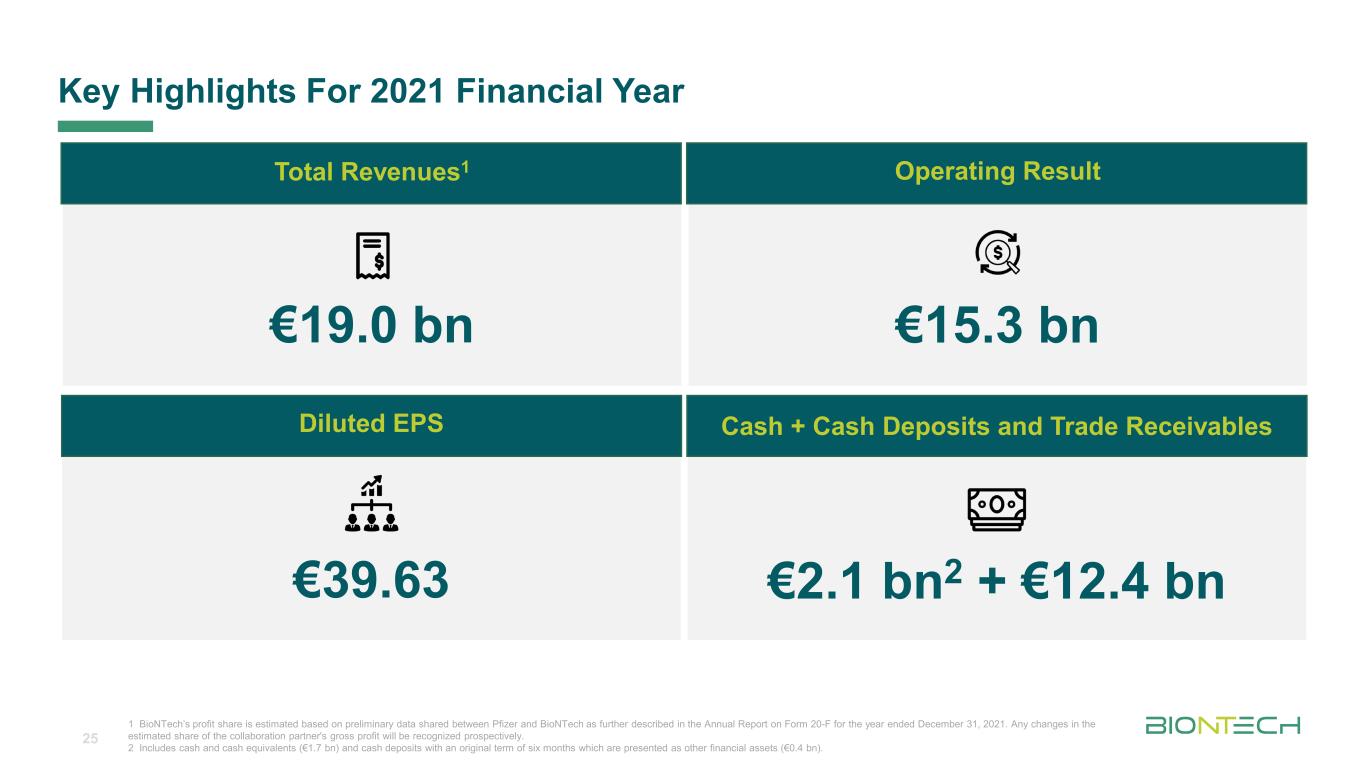
Titelmasterformat durch Klicken bearbeiten €19.0 bn €15.3 bn €39.63 €2.1 bn2 + €12.4 bn 1 BioNTech’s profit share is estimated based on preliminary data shared between Pfizer and BioNTech as further described in the Annual Report on Form 20-F for the year ended December 31, 2021. Any changes in the estimated share of the collaboration partner's gross profit will be recognized prospectively. 2 Includes cash and cash equivalents (€1.7 bn) and cash deposits with an original term of six months which are presented as other financial assets (€0.4 bn). Key Highlights For 2021 Financial Year 25 Total Revenues1 Operating Result Diluted EPS Cash + Cash Deposits and Trade Receivables

Titelmasterformat durch Klicken bearbeiten Direct COVID-19 vaccine sales to customers in BioNTech’s territory Share of gross profit and sales milestones from COVID-19 vaccine sales in the Pfizer and Fosun Pharma territory (100% gross margin) €1.0 bn €3.0 bn €14.8 bn FY 2021 1 BioNTech’s profit share is estimated based on preliminary data shared between Pfizer and BioNTech as further described in the Annual Report on Form 20-F for the year ended December 31, 2021. Any changes in the estimated share of the collaboration partner's gross profit will be recognized prospectively. Key Highlights For 2021 Financial Year 26 €18.8 bn COVID-19 Vaccine Commercial Revenues1: €18.8 bn Doses: ~2.6 bn delivered Low- and Middle-Income Countries 40% High-Income Countries 60%COVID-19 Sales to collaboration partners of products manufactured by BioNTech Revenues and margins exceeded expectations

Titelmasterformat durch Klicken bearbeiten Key Highlights For 2021 Financial Year Funds to Finance our Growth2 1 Cash deposit with an original term of six months are presented as other financial assets. 2 Additional influencing factors (i.e. cash outlays) as well as certain collection risk with trade receivables exist. Cash and cash equivalents as of December 31, 2021 €1.7 bn Cash Cash deposits as of December 31, 2021 €0.4 bn Cash Deposits1 Trade receivables as of December 31, 2021 €12.4 bn Trade Receivables 27
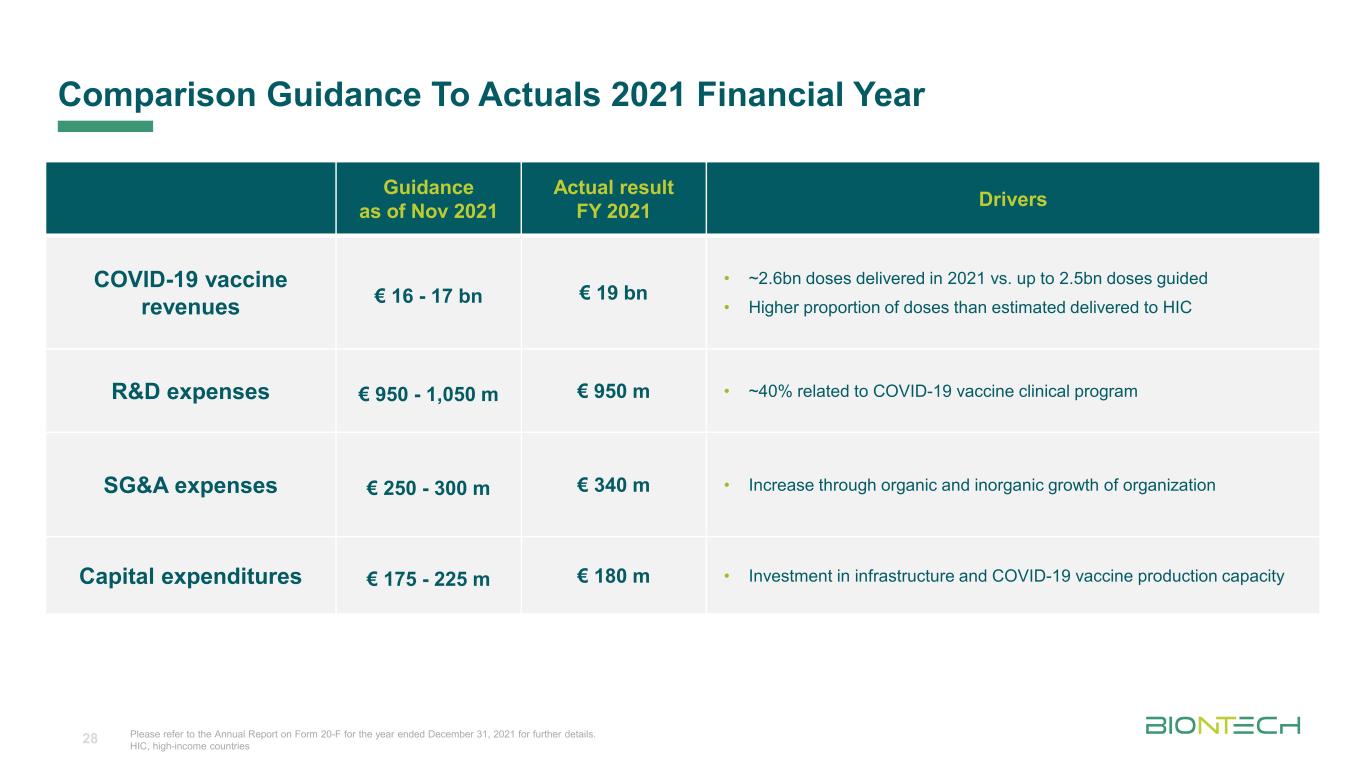
Titelmasterformat durch Klicken bearbeiten Comparison Guidance To Actuals 2021 Financial Year 28 Guidance as of Nov 2021 Actual result FY 2021 Drivers COVID-19 vaccine revenues € 16 - 17 bn € 19 bn • ~2.6bn doses delivered in 2021 vs. up to 2.5bn doses guided • Higher proportion of doses than estimated delivered to HIC R&D expenses € 950 - 1,050 m € 950 m • ~40% related to COVID-19 vaccine clinical program SG&A expenses € 250 - 300 m € 340 m • Increase through organic and inorganic growth of organization Capital expenditures € 175 - 225 m € 180 m • Investment in infrastructure and COVID-19 vaccine production capacity Please refer to the Annual Report on Form 20-F for the year ended December 31, 2021 for further details. HIC, high-income countries
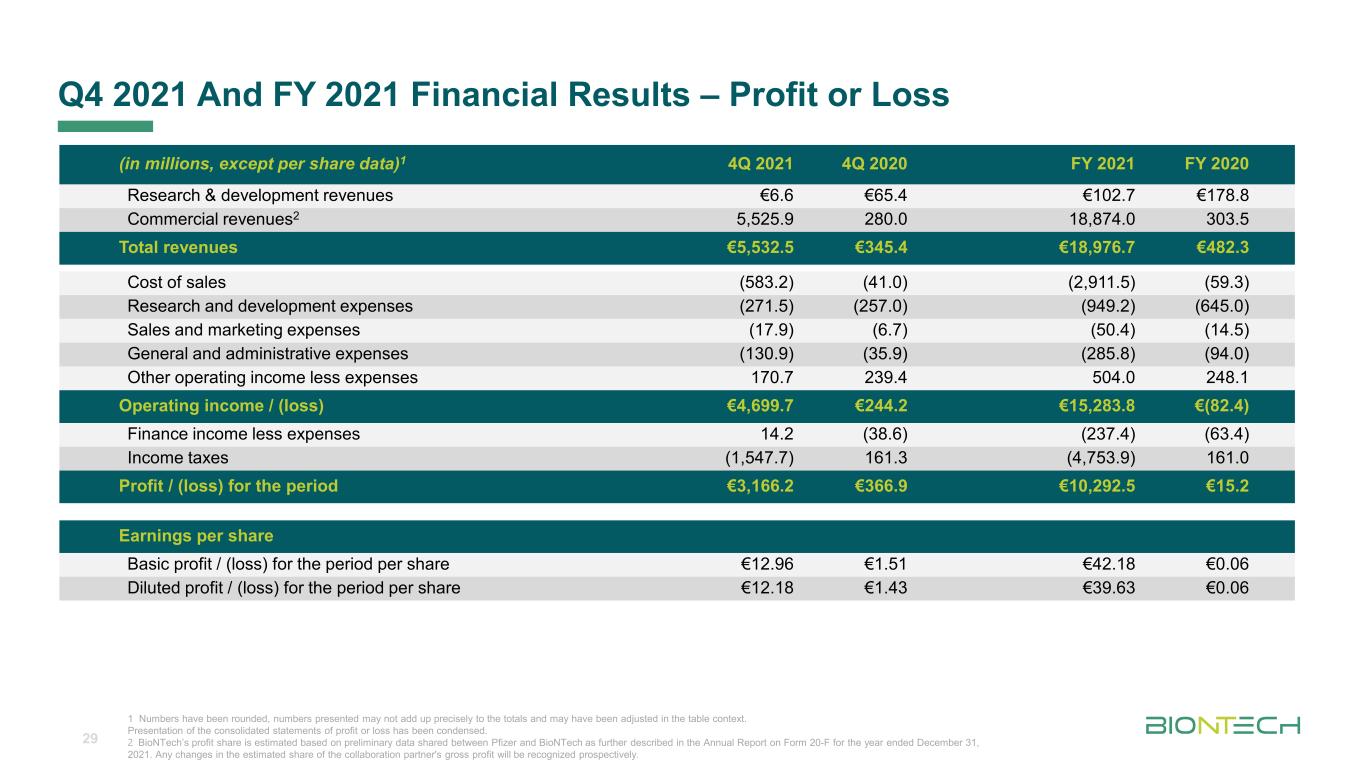
Titelmasterformat durch Klicken bearbeiten Q4 2021 And FY 2021 Financial Results – Profit or Loss 29 (in millions, except per share data)1 4Q 2021 4Q 2020 FY 2021 FY 2020 Research & development revenues €6.6 €65.4 €102.7 €178.8 Commercial revenues2 5,525.9 280.0 18,874.0 303.5 Total revenues €5,532.5 €345.4 €18,976.7 €482.3 Cost of sales (583.2) (41.0) (2,911.5) (59.3) Research and development expenses (271.5) (257.0) (949.2) (645.0) Sales and marketing expenses (17.9) (6.7) (50.4) (14.5) General and administrative expenses (130.9) (35.9) (285.8) (94.0) Other operating income less expenses 170.7 239.4 504.0 248.1 Operating income / (loss) €4,699.7 €244.2 €15,283.8 €(82.4) Finance income less expenses 14.2 (38.6) (237.4) (63.4) Income taxes (1,547.7) 161.3 (4,753.9) 161.0 Profit / (loss) for the period €3,166.2 €366.9 €10,292.5 €15.2 Earnings per share Basic profit / (loss) for the period per share €12.96 €1.51 €42.18 €0.06 Diluted profit / (loss) for the period per share €12.18 €1.43 €39.63 €0.06 1 Numbers have been rounded, numbers presented may not add up precisely to the totals and may have been adjusted in the table context. Presentation of the consolidated statements of profit or loss has been condensed. 2 BioNTech’s profit share is estimated based on preliminary data shared between Pfizer and BioNTech as further described in the Annual Report on Form 20-F for the year ended December 31, 2021. Any changes in the estimated share of the collaboration partner's gross profit will be recognized prospectively.
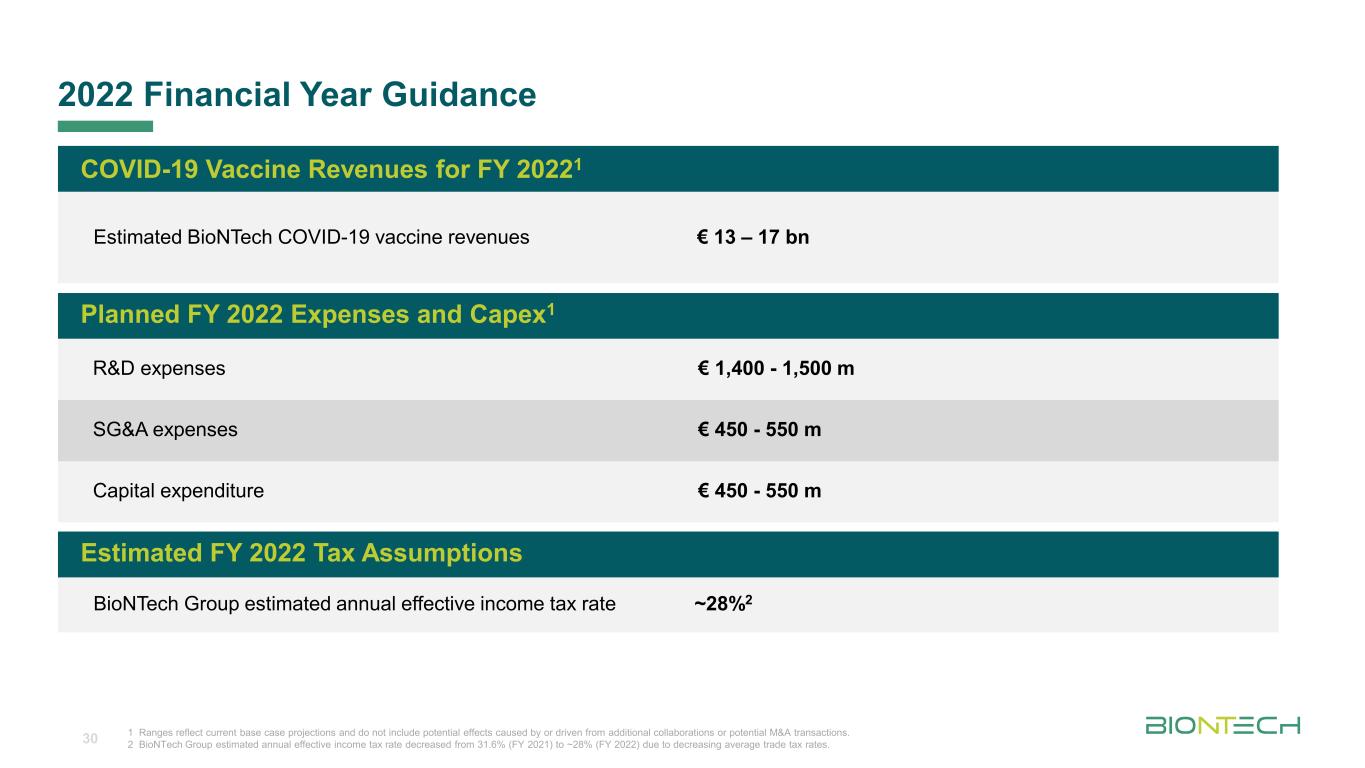
Titelmasterformat durch Klicken bearbeiten 2022 Financial Year Guidance 30 1 Ranges reflect current base case projections and do not include potential effects caused by or driven from additional collaborations or potential M&A transactions. 2 BioNTech Group estimated annual effective income tax rate decreased from 31.6% (FY 2021) to ~28% (FY 2022) due to decreasing average trade tax rates. COVID-19 Vaccine Revenues for FY 20221 Estimated BioNTech COVID-19 vaccine revenues € 13 – 17 bn Planned FY 2022 Expenses and Capex1 R&D expenses € 1,400 - 1,500 m SG&A expenses € 450 - 550 m Capital expenditure € 450 - 550 m Estimated FY 2022 Tax Assumptions BioNTech Group estimated annual effective income tax rate ~28%2
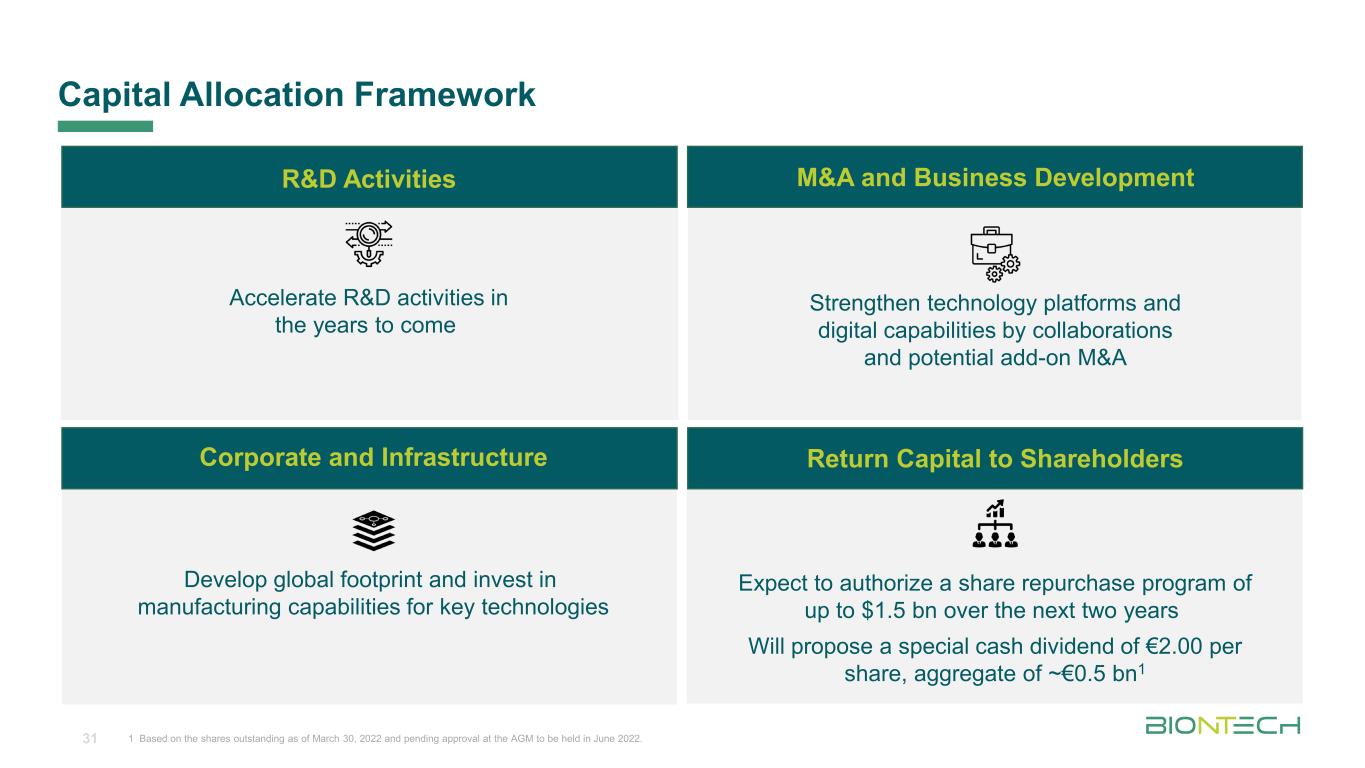
Titelmasterformat durch Klicken bearbeiten Accelerate R&D activities in the years to come Strengthen technology platforms and digital capabilities by collaborations and potential add-on M&A Develop global footprint and invest in manufacturing capabilities for key technologies Expect to authorize a share repurchase program of up to $1.5 bn over the next two years Will propose a special cash dividend of €2.00 per share, aggregate of ~€0.5 bn1 Capital Allocation Framework 31 1 Based on the shares outstanding as of March 30, 2022 and pending approval at the AGM to be held in June 2022. R&D Activities M&A and Business Development Corporate and Infrastructure Return Capital to Shareholders

Corporate Outlook Ryan Richardson, Chief Strategy Officer 04 Financial Results Jens Holstein, CFO 03 Oncology Pipeline Update Özlem Türeci, CMO 02 COVID-19 Vaccine Update Özlem Türeci, CMO 01 Fourth Quarter and Full Year 2021 Highlights Ugur Sahin, CEO 32

Titelmasterformat durch Klicken bearbeiten Outlook for 2022 33 Global footprint & organization Corporate Development Expect data updates for up to 3 additional pipeline programs Global Development Organization transformation underway to support pipeline expansion and potential initiation of registration trials Expanding footprint in Europe, U.S., Asia, and Africa Investing to expand global mRNA manufacturing capacity with new production nodes – including deployment of our first BioNTainer Further pipeline expansion & acceleration2 3 4 1 Continued COVID-19 vaccine franchise leadership Multiple data read-outs expected throughout the year On track to submit regulatory data package for Omicron-adapted vaccine pending decision from regulators New partnerships & M&A to accelerate and enable long-term strategy Extend mRNA platform into new “white spaces” and further expand toolkit in synthetic biology following collaborations with Medigene and Crescendo

Titelmasterformat durch Klicken bearbeiten Expected Pipeline Milestones in 2022 (1 of 2) 34 BNT162b2 Timing • Data for 4th dose in adults, aged 16 to 65 years ongoing • Data for 3rd dose in children, aged 5 to <12 years ongoing • Data for 3-dose regimen in children, aged 6 months to <5 years April Next-Generation COVID-19 vaccines • Data for Omicron-based vaccine (monovalent) April • Multiple updates: Variant combinations and bivalent vaccines 2H Other pipeline programs • BNT161 – Influenza mRNA vaccine1 1H • BNT1222 Phase 2 – iNeST in combination w/Pembrolizumab, 1L Melanoma 2H • BNT211 Phase 1/2 – CAR-T/CLDN6+, multiple solid tumors 2H CLDN, Claudin; NSCLC, non-small cell lung cancer 1 Partnered with Pfizer 2 Partnered with Genentech 8+ Data Updates

Titelmasterformat durch Klicken bearbeiten Expected Pipeline Milestones in 2022 (2 of 2) 35 Infectious Diseases Timing • Shingles vaccine1 2H • Tuberculosis vaccine2 2H • HSV 2 vaccine 2H • Malaria vaccine 2H Oncology • BNT141 – RiboMab, solid tumors FPD in January • BNT142 – RiboMab, solid tumors 1H • BNT116 – Fixvac in combination w/Libtayo, NSCLC 2H ✓ HSV 2, herpes simplex virus type 2; FPD, first patient dosed; NSCLC, non-small cell lung cancer 1 Partnered with Pfizer 2 Partnered with Bill and Melinda Gates Foundation Up to 7 First-in-Human Trial Starts

36 SAVE THE DATE Capital Markets Day Annual General Meeting June 29, 2022 June 1, 2022 36
